Abstract
Objective:
To investigate the role of vascular endothelial growth factor (VEGF) in haemorrhagic fever with renal syndrome (HFRS).
Methods:
VEGF, soluble VEGF receptor (sVEGFR)-2, angiopoietin (Ang)-1, tumour necrosis factor (TNF)-α and interferon (IFN)-γ levels were measured in serum samples from 68 patients with HFRS. Cultured human umbilical vein endothelial cells (HUEVCs) were infected by Hantaan virus (HTNV) and/or stimulated with recombinant VEGF; dextran permeability of the cells was determined. Claudin-1 and vascular endothelial (VE)-cadherin levels were determined by real-time reverse transcription-polymerase chain reaction and Western blot analyses.
Results:
Serum VEGF, TNF-α and IFN-γ levels were significantly elevated, whereas sVEGFR2 and Ang-1 levels were reduced, during the acute phase of HFRS. In vitro cell permeability was unaffected by HTNV infection or VEGF stimulation alone, but the combination of HTNV infection and VEGF treatment significantly increased the permeability of endothelial cell monolayers in a time-dependent manner. Claudin-1 and VE-cadherin were downregulated at both the mRNA and protein level by combined HTNV infection and VEGF stimulation.
Conclusions:
Elevated VEGF induced by HTNV infection may play an important role in the vascular hyperpermeability that is characteristic of HFRS.
Keywords
Introduction
Haemorrhagic fever with renal syndrome (HFRS) is an acute infectious disease caused by a group of serologically related viruses including Hantaan virus (HTNV), which belongs to the Hantavirus genus of the Bunyaviridae family.1,2 Worldwide, approximately 60 000 – 100000 cases of HFRS are reported annually, with a mortality rate of 2 – 10%. 3 The majority of HFRS cases are reported in China, where symptom severity and mortality rates are the highest in the world.3,4
The principal pathophysiological change in HFRS is increased vascular permeability, which commonly affects small systemic blood vessels and capillaries, resulting in severe hypotension, oedema and multiple organ failure. 5 Mechanisms of hantavirus-mediated hyperpermeability are unclear; postmortem analyses show that endothelial cells from patients with HFRS commonly express viral antigens, suggesting that direct viral induction may be a possible cause.6,7 There is increasing evidence, however, that hantavirus is not directly cytopathic1,8 and that clinical outcomes of infection may be the result of complicated interactions between the virus and the host immune response. 8 Although elevated levels of cytokines with a permeability enhancing effect (such as tumour necrosis factor [TNF]-α, interferon [IFN]-γ, interleukin [IL]-4, IL-6 and IL-8) have been reported in HFRS, their precise role in plasma leakage remains unknown.9,10
Vascular endothelial growth factor (VEGF) is a key regulator of vascular permeability, and VEGF-mediated permeability commonly involves several signalling pathways that differ in their time courses. 11 Pathogenic hantaviruses (such as HTNV, Andes virus and New York-1 virus) markedly increase the permeability of endothelial cells in response to VEGF, whereas nonpathogenic hantaviruses (such as Leaky virus, Prospect Hill-like virus and Isla Vistra virus) have no such effect.12,13 The downstream involvement of VEGF pathways in hantavirus infection are, however, not fully understood.
The present study examined serum levels of VEGF, soluble VEGF receptor (sVEGF)-2 and related cytokines in Chinese patients who were in different phases of HFRS. In addition, the influence of HTNV and VEGF stimulation on the permeability of cultured cells was evaluated: levels of the tight junctional proteins claudin-1 and vascular endothelial (VE)-cadherin were investigated in vitro, under conditions of HTNV infection and VEGF stimulation.
Patients and methods
Study Population
Consecutive patients hospitalized with HFRS at Tangdu Hospital, Fourth Military Medical University, Xi'an, China, between January 2009 and January 2010, were enrolled in this study. All patients had symptoms typical of HFRS (fever, lumbalgia and proteinuria), and diagnosis was confirmed by detection of hantavirus-specific immunoglobulin (Ig) M and IgG using enzyme-linked immunosorbent assay (ELISA) kits (Xi'an Bosheng Biological Technology, Shaanxi, China). Diagnosis of HFRS was made according to the criteria of the Monitor Scheme of HFRS, issued by the Chinese Ministry of Health in 2005 (available at http://www.moh.gov.cn/publicfiles/business/htmlfiles/wsb/pzcjd/200804/21000.htm [in Chinese]). Exclusion criteria were: any other kidney disease; diabetes mellitus; cardiovascular disease; autoimmune disease; haematological disease; viral hepatitis (types A, B, C, D, E); any other liver disease. A group of age- and sex-matched healthy volunteers, recruited from the local population of Xi'an, Shaanxi Province, China, was included as the control group.
The study protocol was approved by the Ethics Committee of Tangdu Hospital and all participants or their next of kin provided written informed consent.
Elisa Analyses
Nonfasting blood samples (5 ml) were collected from all participants and centrifuged for 10 min at 1730
Cell Culture
Primary human umbilical vein endothelial cells (HUVECs; ScienCell™ Research Laboratories, Carlsbad, CA, USA) were grown in endothelial cell medium (ScienCell™ Research Laboratories; this includes penicillin and streptomycin and is also supplemented with essential and nonessential amino acids, vitamins, organic and inorganic compounds, hormones, growth factors and trace minerals) in 5% heat-inactivated fetal bovine serum (FBS; Gibco, Grand Island, NY, USA) at 37 °C in 5% CO2. All cells underwent fewer than eight passages before use.
Permeability Assay and Teer
The HUVECs were seeded onto polyester membrane transwell inserts (6.5-mm diameter, 0.33-cm2 insert membrane growth area, 3-μm pore size; Corning Inc., Corning, NY, USA) at a density of 2 × 105 cells and grown for 4 days, as described above, to reach confluence. Confluent HUVECs were infected with HTNV (strain 76-118; grown in the Centre for Infectious Diseases, Tangdu Hospital, Fourth Military Medical University) at a multiplicity of infection of 0.5 plaque-forming units/cell for an adsorption period of 2 h, or mock infected (i.e. without HTNV or VEGF) to serve as a control. Cells were then incubated in endothelial cell medium containing 0.5% FBS. Fluorescein-isothiocyanate (FITC)-labelled dextran (0.5 mg/ml; Sigma-Aldrich, St. Louis, MO, USA) was added to inserts 0, 1, 2 or 3 days postinfection in the presence or absence of 100 ng/ml recombinant VEGF (PeproTech, Rocky Hill, NJ, USA), TNF-α (100 ng/ml, PeproTech) or IFN-γ (100 IU/ml, PeproTech). Transendothelial electrical resistance (TEER) across HUVECs was measured using an electrical cell sensor system (Millicell®-ERS; World Precision Instruments, Sarasota, FL, USA). The TEER value was calculated using the following formula: (average resistance of experimental wells - average resistance of blank wells) × 0.33 (insert membrane growth area). Supernatant (100 μl) was harvested from the basolateral chamber at 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 h after treatment with VEGF and fluorescence intensity was quantified using a FluorImager® 595 (Molecular Devices, Sunnyvale, CA, USA). Each experiment was performed in triplicate.
RT–PCR Analyses
Total RNA was extracted from cultured HUVECs (1 × 106) using TRIzol® (Invitrogen, Carlsbad, CA, USA). Reverse transcription (RT) was undertaken with random hexamers using PrimeScript™ RT Master Mix (Takara Biotechnology, Dalian, China). Real-time RT-polymerase chain reaction (PCR) was performed using 100 ng of CDNA and SYBR® Premix Ex Taq (Takara Biotechnology) and primers for claudin-1, VE-cadherin and glyceraldehyde 3-phosphate dehydrogenase (GAPDH), as shown in Table 1. Real-time PCR analyses were carried out using the Applied Biosystems® 7500 Real-Time PCR system (Applied Biosystems, Foster, CA, USA), using a standard protocol provided with the thermal cycler. Relative expression levels were quantified by the 2–ΔΔCT method, 16 using the ABI Prism® 7500 Sequence Detection System software (Applied Biosystems, Foster, CA, USA), normalized to GAPDH.
Primer sequences used in real-time reverse transcription–polymerase chain reaction analyses, performed in total RNA extracted from cultured human umbilical vein endothelial cells, to investigate levels of tight junctional proteins under conditions of Hantaan virus infection and vascular endothelial growth factor stimulation

VE, vascular endothelial; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Western Blotting Analysis
Cells (5 × 106 per sample) were lysed on ice in lysis buffer (50 mM Tris-HCl, pH 8.0, 150 mM NaCl, 0.1% sodium dodecyl sulphate [SDS], 1% Nonidet™ P-40, 0.5% deoxycholic acid, 0.5% sodium azide and 100 μg/ml phenylmethanesulphonylfluoride) for 15 min. Lysates were transferred to 0.5-ml tubes and the supernatants were collected by centrifugation at 12 000
Statistical Analyses
All statistical analyses were performed using SPSS® statistical software, version 12.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Results were expressed as the mean ± SD. Data were analysed with Student's t-test (two-tailed). A P-value of < 0.05 was considered to be statistically significant.
Results
A total of 68 patients with HFRS (48 males, 20 females), median age 43 years (range, 12 – 65 years), were enrolled in the study. Patients were in the febrile (n = 15), hypotensive (n = 15), oliguric (n = 15), diuretic (n = 15) and convalescent (n = 8) disease phases. There were 20 gender-matched control subjects (11 males, 9 females), median age 36 years (range, 21 – 49 years).
Compared with control subjects, serum levels of VEGF were significantly increased in patients in the febrile, oliguric (P < 0.05) and hypotensive phases (P < 0.01) of HFRS (Table 2). No significant differences were observed between control subjects and patients in the diuretic phase or in convalescence. Conversely, serum levels of sVEGFR2 and Ang-1 were significantly reduced in HFRS patients in the hypotensive phase (P < 0.01) compared with control subjects, but gradually increased in the oliguric and diuretic phases, returning to control levels in the convalescence phase. Serum TNF-α and IFN-γ levels were also significantly elevated in HFRS patients in the hypotensive and oliguric phases (P < 0.05), demonstrating similar trends to VEGF serum levels across the disease phases (Table 2).
Serum levels of vascular endothelial growth factor (VEGF), soluble VEGF receptor (sVEGF)-2, angiopoietin (Ang)-1 and related cytokines, determined by enzyme-linked immunosorbent assay, in patients with haemorrhagic fever with renal syndrome

Data presented as mean ± SD.
Normal serum levels of VEGF, sVEGFR2 and Ang-1 are reported to be 335 ± 118 pg/ml, 1.0 ± 0.3 ng/ml and 3226 pg/ml (range: 1974.5 - 5307.0 pg/ml), respectively.14,15
P < 0.05 and
P < 0.01 compared with healthy controls; Student's t-test (two-tailed).
TNF, tumour necrosis factor; IFN, interferon.
There was no change in endothelial permeability in HUVECs infected with HTNV or in mock-infected cells treated with VEGF. At 3 days postinfection, permeability was increased more than 20-fold in cells infected with HTNV and treated with VEGF (P < 0.01; Fig. 1A). On day 3 postinfection, an increase in permeability over time was observed in HTNV-infected cells following VEGF treatment; modest increases were measured 1.5 h after stimulation, peaking at 3 h (P < 0.01; Fig. 1B). As shown in Fig. 1C, TEER was significantly decreased in HTNV-infected cells from 1.5 h (P < 0.05) poststimulation with VEGF and remained depressed for up to 3 h (P < 0.01). TNF-α and IFN-γ; also increased cell permeability in combination with HTNV infection by approximately 4-fold compared with controls (data not shown).
Human umbilical vein endothelial cells (HUVECs) infected with Hantaan virus (HTNV) strain 76-118 or mock infected at day 0 and incubated for 3 days in the presence/absence of 100 ng/ml recombinant vascular endothelial growth factor (VEGF). (A), (B) Endothelial permeability over time in HUVECs following treatment with 100 ng/ml vascular endothelial growth factor (VEGF). (C) Transendothelial electrical resistance (TEER) over time in HUEVCs following treatment with 100 ng/ml VEGF. Data presented as mean ± SD. Experiments performed in triplicate. Mock, mock-infected cells; HTNV, infected cells with no VEGF stimulation; VEGF, mock-infected cells stimulated with VEGF; HTNV + VEGF, infected cells stimulated with VEGF. *P < 0.05, **P < 0.01 versus mock-infected cells; Student's t-test (two-tailed)
Claudin-1 and VE-cadherin mRNA levels were significantly decreased by day 1 postinfection following VEGF stimulation and remained depressed on days 2 and 3 postinfection (P < 0.05; Fig. 2A). Similar findings were observed following Western blot analysis, with reductions in claudin-1 and VE-cadherin protein levels seen in HTNV-infected HUVECs over time, following VEGF treatment (Fig. 2B).
Human umbilical vein endothelial cells (HUVECs) infected with Hantaan virus (HTNV) strain 76-118 or mock infected at day 0 and incubated for 3 days in the presence/absence of 100 ng/ml recombinant vascular endothelial growth factor (VEGF). (A) Real-time reverse transcription–polymerase chain reaction analyses of claudin-1 and vascular-endothelial (VE)-cadherin mRNA levels over time in HUVECs infected with HTNV and stimulated with 100 ng/ml VEGF. Data presented as mean ± SD. *P < 0.05, **P < 0.01 versus mock-infected cells; Student's t-test (two-tailed). (B) Western blot analyses of claudin-1 and VE-cadherin protein levels over time in HTNV-infected HUEVCs following VEGF stimulation. GAPDH, glyceraldehyde 3-phosphate dehydrogenase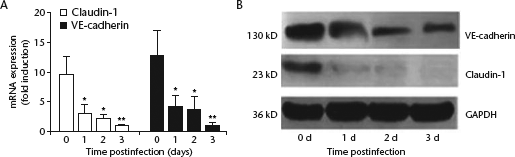
Discussion
Pathogenic hantaviruses, which cause HFRS, render human endothelial cells more sensitive to VEGF-mediated increases in permeability. 12 The present study is the first to demonstrate that dynamic changes in serum VEGF occur in the different diseases phases of HFRS: VEGF levels increased in the febrile phase, peaked during the hypotensive phase and slowly returned to normal during the oliguric, diuretic and convalescence phases. Furthermore, elevated serum levels of VEGF were associated with a decrease in serum levels of sVEGFR2 and Ang-1 (which are known to counter the permeability effects VEGF and, therefore, stabilize the vasculature).17–19
Plasma leakage is a central event in the pathogenesis of both HFRS and dengue haemorrhagic fever (DHF). Findings from the present study are consistent with other reports, where plasma VEGF levels in patients with DHF were found to be significantly higher than levels in patients with dengue fever alone, 20 and reductions in sVEGF2 were demonstrated in patients with DHF. 19 Furthermore, suppression of Ang-1 at both the mRNA and protein levels has been found in HTNV-infected HUVECs. 15 The dynamic changes in serum VEGF, sVEGFR2 and Ang-1 observed in the present study may therefore contribute to the plasma leakage that is characteristic of HFRS. Other research has also shown that high levels of cytokines including IL-10, TNF-α and IFN-γ are present in serum from patients with HFRS 21 and has demonstrated that TNF-α promotes VEGF production in vitro. 22 Consistent with these data, the present study also found that levels of TNF-α and IFN-γ were elevated in HFRS.
The role of VEGF in HFRS has not been studied extensively, although research has shown that while VEGF-mediated permeability differs in the time course of treatment, 23 it commonly involves many different cytokines,22,24 together with specific signalling pathways and ultrastructural changes. 11 Neither HTNV infection nor VEGF stimulation alone altered in vitro cell permeability in the present study, but the combination of both notably elevated the permeability of endothelial monolayers in a time-dependent manner, as determined by measurement of FITC-labelled dextran permeability and TEER. Similar to VEGF, TNF-α and IFN-γ were also found to increase cell permeability in combination with HTNV infection, but to a much lesser extent.
Viruses are known to influence the function and structure of intercellular junctional proteins, thereby breaching cellular barriers and allowing further spread of the virus. 25 IFN-γ has been shown to inhibit hepatitis C virus (HCV) infection by altering the distribution of HCV receptors and downregulating claudin-1, which results in a disruption of barrier function. 26 Studies have also shown that the pulmonary syndrome-associated hantavirus, Andes virus, disrupts the endothelial cell barrier by induction of VEGF and downregulation of VE-cadherin. 27 Thus, active Andes virus replication results in increased permeability and loss of endothelial cell barrier integrity. Furthermore, ultrastructural studies have demonstrated that there is a loss of junctional integrity with rapid VEGF activation, and a reduction in endothelial junctional proteins (such as zonula occluden-1, occludin and VE-cadherin), coincident with the onset of increased vascular permeability.28–30 The present study investigated two important components of adherens junctions, claudin-1 and VE-cadherin, and found that both were gradually downregulated at the mRNA and protein level in HTNV-infected cells following VEGF stimulation. These findings suggest that HTNV-induced VEGF secretion could cause ultrastructural changes by suppression of junctional proteins, in order to alter cellular permeability.
In conclusion, the present study demonstrated that dynamic changes in VEGF levels occurred in different disease phases of HFRS, with peak levels being observed during the hypotensive phase. Increases in VEGF were associated with downregulation of sVEGFR2 and Ang-1, and may have led to the secretion of inflammatory cytokines, suppression of tight junctional proteins and further ultrastructural modification of endothelial cell barriers. These morphological changes in the endothelium may result in an increase in vascular permeability, resulting in the phenomenon of plasma leakage that is characteristic of HFRS.
Footnotes
Acknowledgements
The authors wish to thank all the study participants. This work was supported by grants from the National Basic Research Programme of China (Programme 973; No. 2012CB518905) and the National Natural Science Foundation of China (No. 30872215).
The authors had no conflicts of interest to declare in relation to this article.
