Abstract
Background
Vascular malformations are rare diseases that should be treated in dedicated vascular anomaly centers (VAC). There is only a small amount of data on the diagnostic and therapeutic handling of these patients, before they are referred to a VAC.
Purpose
To demonstrate the disease-specific patient characteristics in a German VAC, which are required to determine diagnostic and therapeutic steps.
Material and Methods
In a retrospective study, all patients who were treated in the VAC from April 2014 until August 2021 were identified. In total, 593 patients were included in this study.
Results
Almost all patients had previously consulted a physician (591/593, 99.7%). A mean of two different physicians had been consulted (range 0–10). Patients with more complex, syndromal vascular malformations had significantly more previous appointments (P = 0.0018). In only 44% (261/593) of patients, the referral diagnosis was made correctly. Most patients had been previously treated for their vascular anomaly: pharmacotherapy (n = 130; 21.9%), compression garments (n = 141; 23.8%), surgical resection (n = 80; 17.3%) and sclerotherapy (n = 68; 11.5%). Fifty-two patients who had been falsely diagnosed had also received therapy prior to their referral to the VAC (8.8%). Most patients received an ultrasound examination in the VAC (n = 464; 78.2%). Most frequently, compression therapy was prescribed (n = 256; 43.2%), followed by sclerotherapy (n = 175, 29.5%) and pharmacotherapy (n = 55; 9.3%).
Conclusion
Patients suffering from vascular anomalies often go through a complicated scheduling with numerous outpatient appointments and have a high risk of misdiagnosis and mistreatment prolonging the medical condition. Therefore, patients with vascular anomalies should be treated in a dedicated vascular anomaly center.
Introduction
According to the established and commonly accepted International Society for the Study of Vascular Anomalies classification, vascular anomalies are stratified into vascular tumours and vascular malformations (1). Vascular malformations occur in 0.8 to 1.0% of the population and they are thus considered a group of rare diseases (2). Incidence and prevalence differ between different types of malformations, with venous malformations being the most common manifestation and arteriovenous malformations being the rarest form. They often present very heterogenous phenotypes and commonly occur combined with a wide range of comorbidities. It is recommended that complex vascular anomalies are treated in specialized vascular anomalies centers (VAC) (3,4).
Due to the lack of expertise, false diagnosis, resulting in delayed or inappropriate treatment, is still very common (5,6).
Vascular tumours are more frequent than malformations and occur in 5–10% of neonates and are even more frequent in preterm neonates with a prevalence of up to 30% (7). Vascular tumours include infantile and congenital hemangiomas, as well as hemangioendotheliomas (6). Females have a higher incidence than males (8,9). By contrast, the frequency of vascular malformations is not gender related (6). Histopathologically, vascular tumours are defined by densely packed layers of thin vascular channels and plump endothelial cells, infantile hemangiomas are GLUT1-positive, while congenital hemangiomas are GLUT1-negative (10).
There is only a small amount of data on the patient characteristics of a dedicated vascular anomalies center in Germany. Thus, the present study seeks to delineate the composition of the patient population at a VAC in a major population center in Germany. The findings will possibly influence dedicated patient care.
Material and Methods
In a retrospective review of medical records, all patients referred to the vascular anomaly center since its establishment in April 2014 until August 2021 were identified. In the present study, the main focus was on the identification of following parameters: verification of the diagnosis of the underlying vascular anomaly, patient history, previous diagnostics and treatment, and the treatment in our institution.
All reported data in the study are based on patient related medical information that has been derived from clinical consultation and treatment of patients. No further patient interviews were conducted. No third party had access to any patient related data during the conduction of this study and the entire stored dataset was initially anonymized. The study was granted approval by the local ethics committee (ID-Nr. 2016-522N-MA).
In the review of the patient records and the electronic inpatient hospital system we captured the following factors: patient demographics, such as gender, age, length of journey to the VAC in kilometers, number of pre-VAC doctor appointments regarding the vascular anomaly, referral diagnosis, localization and distribution of the vascular anomaly, past diagnostic measures and previous therapies, such as screening for coagulopathy, biopsy, ultrasound, computed tomography (CT) or magnetic resonance imaging (MRI) scans, intervention and surgical resection. Furthermore, all diagnostics, interventional procedures, complications during the treatment in the VAC, as well as the final diagnosis, were noted.
When analyzing the correctness of the referral diagnosis, patients with unspecific referral diagnoses (such as “vascular malformation”) were excluded from the analysis.
Statistical analysis
Normally distributed data was analyzed with Student’s t-test. For non-normally distributed data, the Mann–Whitney U-test was used. A chi-squared test was performed for categorical variables. Categorical variables are presented with frequencies (percentages) and continuous variables with the mean ± SD or median and range. P < 0.05 was considered statistically significant. All data analysis was conducted using Prism, version 9.2.0 (GraphPad Software Inc., San Diego, CA, USA).
Results
Patient demographics
In summary, 593 patients were treated in the Mannheim University VAC from 2017 to 2021. Some 41.5% of patients were male, 58,7% were females and the mean age was 26.4 years. Children and adolescents made up 38.7% of the patient population. Table 1 depicts the patient characteristics.
Patient characteristics.
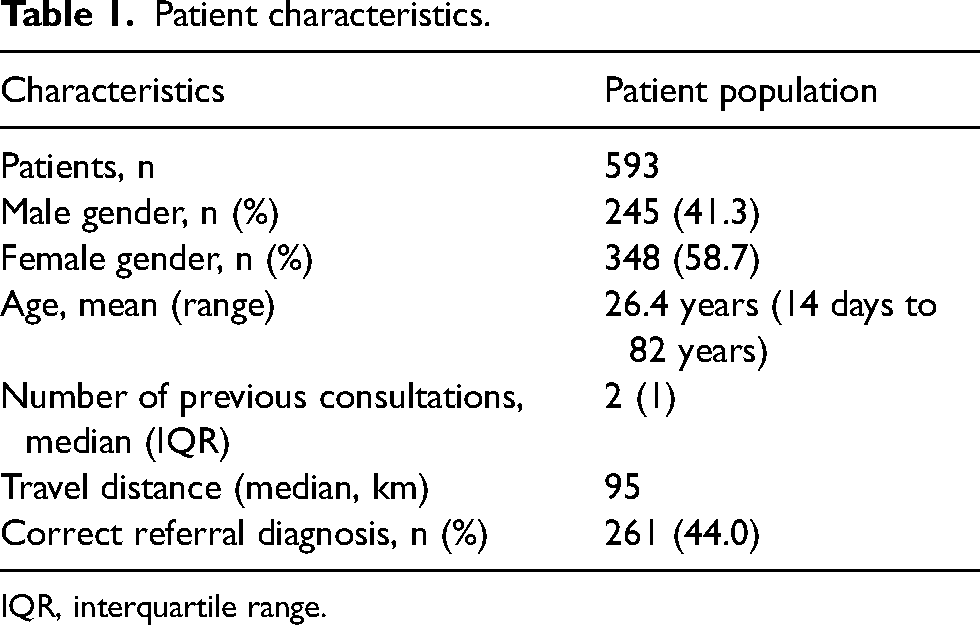
IQR, interquartile range.
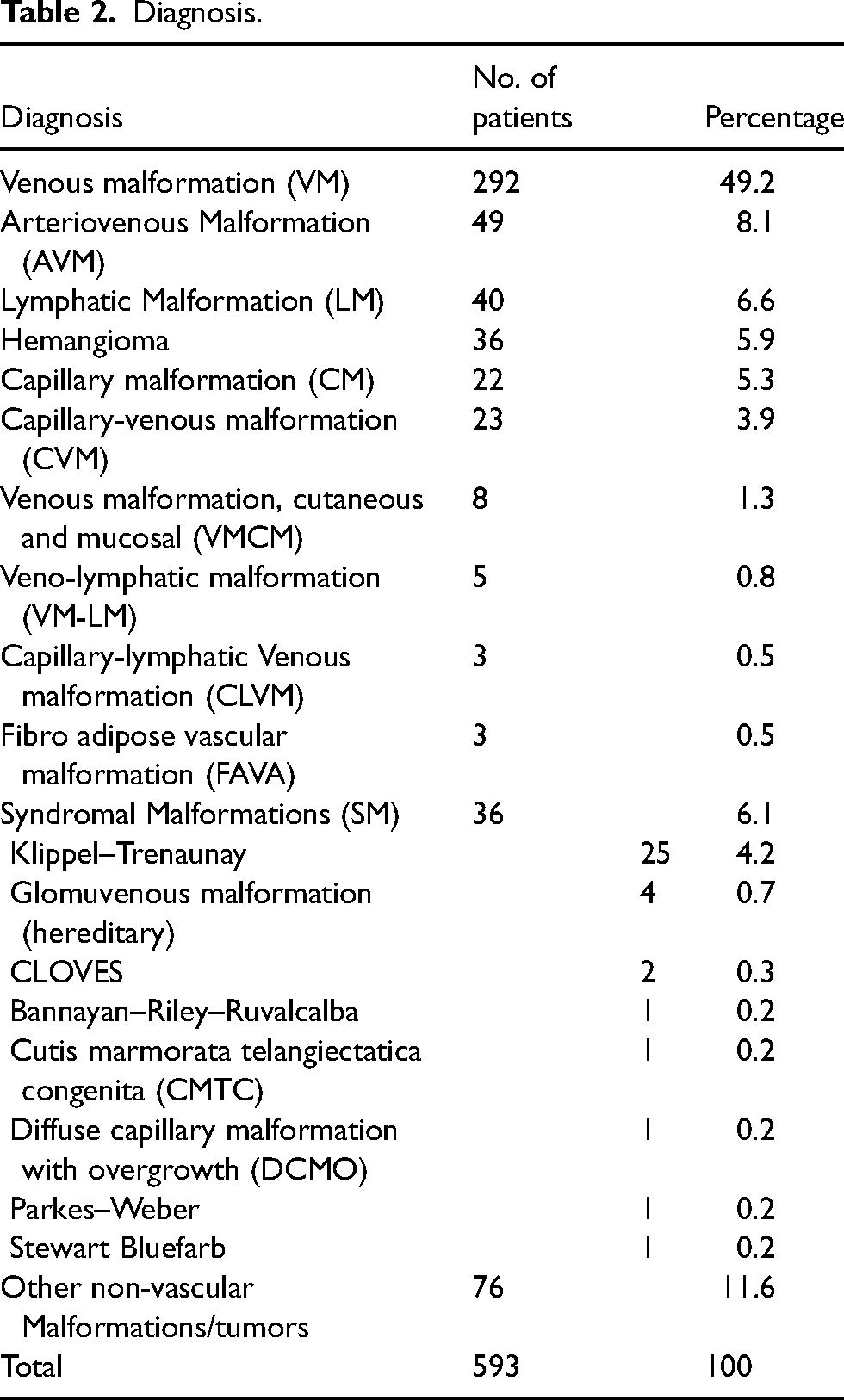
Tables 2 to 4 show the distribution of the different diagnoses in our patient population. In summary, venous malformations represented the largest group of vascular anomalies in 49.2% of patients, followed by non-vascular malformations or tumours ranging up to 11.6%. Arteriovenous malformations made up 8.1% of the patient population. Lymphatic malformations had a percentage of 6.6, while syndromal vascular malformations (syndromes known formerly as Klippel–Trenaunay syndrome; Parkes–Weber syndrome; Bannayan–Riley––Ruvalcaba syndrome; Cutis marmorata teleangiectatica congenita; hereditary glomuvenous malformation; diffuse capillary malformation with overgrowth) occurred in 6.1% of the patients.
Diagnosis.
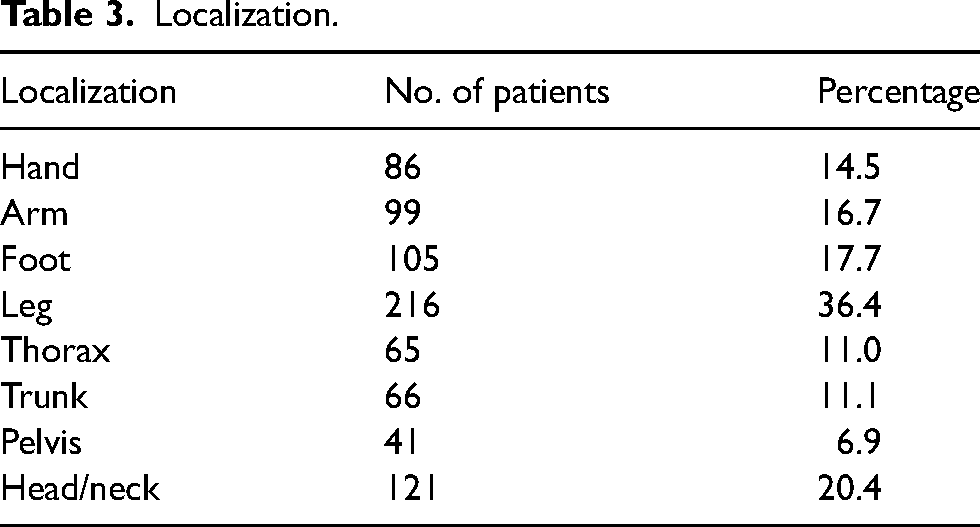
Localization.
Distribution.

Patient history prior to therapy in the VAC
Out of 593 patients, only two had not previously seen a physician for the vascular malformation. On average, patients who came to the VAC had previously seen 2.7 other doctors.
Patients with hemangiomas, venous malformations, capillary malformations, lymphatic malformations, and cutaneous and mucous venous malformations had previously seen a median of two different physicians. Patients suffering from arteriovenous malformations, capillary–venous malformations and syndromal malformations had a median of three previous appointments. Patients with a fibrous adipose vascular malformation or capillary lymphatic venous malformation had the highest median with four previous doctor's visits. Fig. 1 shows an overview over range and median of previous doctor's appointments for each entity.
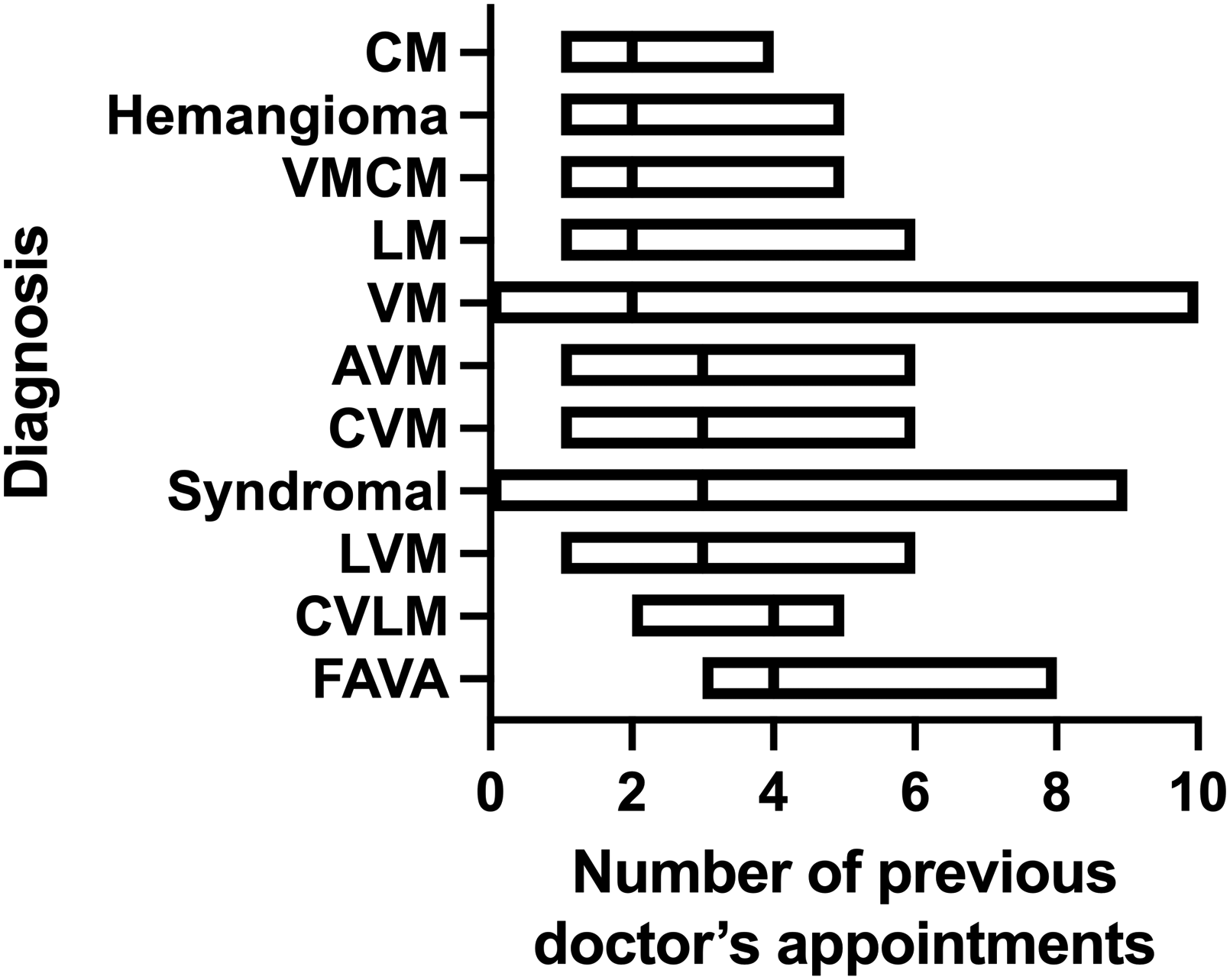
Number of previous doctor's appointments according to entity, bars show the range and median. CM, capillary malformation; VMCM, venous malformation, cutaneous and mucous; LM, lymphatic malformation; VM, venous malformation; CVM, capillary–venous malformation, syndromal: combined vascular malformations with overgrowth (formerly known as Klippel-Trenaunay syndromes) or hereditary malformations (glomuvenous malformation); LVM, lymphaticovenous malformation; CVLM, capillary venolymphatic malformation; AVM, arteriovenous malformation; FAVA, fibro-adipose vascular anomaly.
Patients with complex, syndromal vascular malformations had had significantly more previous appointments than patients with “regular” venous malformations (P = 0.0018).
When comparing the number of previous appointments among arteriovenous malformations, lymphatic malformations and venous malformations, no statistically significant differences were found. However, patients with vascular malformations (all entities) had had significantly more previous medical consultations than patients suffering from vascular tumors, like hemangiomas.
Only 261 out of 593 patients (44.0%) were referred to our VAC with the correct diagnosis. Fig. 2 depicts the rate between correct versus false referral diagnoses. Even though venous malformations are the most frequent vascular anomaly to be encountered, they were significantly less often diagnosed correctly than arteriovenous (P < 0.0001) or lymphatic malformations (P < 0.0001). Hemangiomas were misdiagnosed in 39.1% of cases, when compared to vascular malformations (all entities), there was no significant difference in the frequency of false diagnosis. Interestingly, complex syndromal vascular malformations, such as CLOVES, Klippel–Trenaunay syndrome, Bannayan–Riley–Ruvalcaba syndrome and Parkes–Weber syndrome were significantly more often diagnosed correctly than simple vascular malformations (arteriovenous, venous and lymphatic malformations) (P < 0.0001).
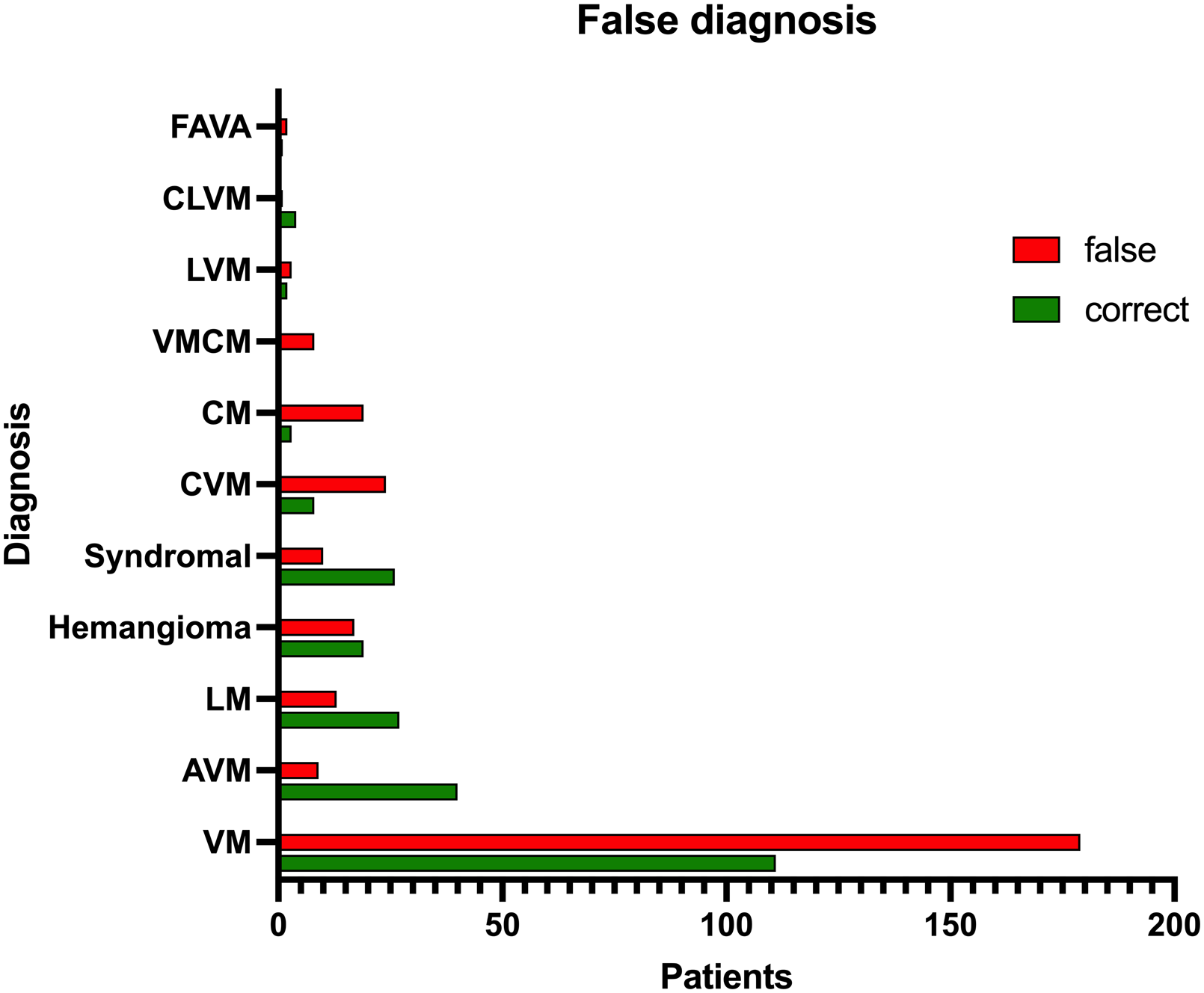
Number of incorrectly (red bar) and correctly (green bar) diagnosed patients listed according to their vascular anomaly. FAVA, fibro-adipose vascular anomaly; CVLM, capillary veno-lymphatic malformation; LVM, veno-lymphatic malformation; VMCM, venous malformation, cutaneous and mucous; CM, capillary malformation; CVM, capillary–venous malformation, syndromal, combined vascular malformations with overgrowth (formerly known as Klippel–Trenaunay) or hereditary malformations (glomuvenous malformation); LM, lymphatic malformation; AVM, arteriovenous malformation; VM, venous malformation.
Antecedent diagnostics and therapy
MRI was the most common diagnostic method applied prior to referral to the VAC (n = 442; 74.5%), followed by ultrasound (n = 68; 11.5%) and then diagnostic transcatheter angiography (n = 42; 7.1%). Twenty-nine patients underwent CT (4.9%). In 22 cases, conventional radiographs were available (3.7%). Conventional phlebography was used the least (n = 19; 3.2%).
Most patients had previously undergone treatment for their vascular anomaly. A large proportion (n = 130; 21.9%) had previously received pharmacotherapy, including anticoagulation (n = 47; 7.9%), propranolol (n = 6; 1.0%) and analgesics (n = 120; 20.2%). In total, 141 patients had been treated with compression garments (23.8%). The number of cases undergoing surgical resection was high, with 80 patients (17.3%). In 68 patients, sclerotherapy had been conducted (11.5%). Embolization was the least frequent antecedent therapy modality with only 24 cases (4.0%). Figs 3 and 4 give an overview of the performed diagnostics and therapies prior to and during therapy in the VAC.
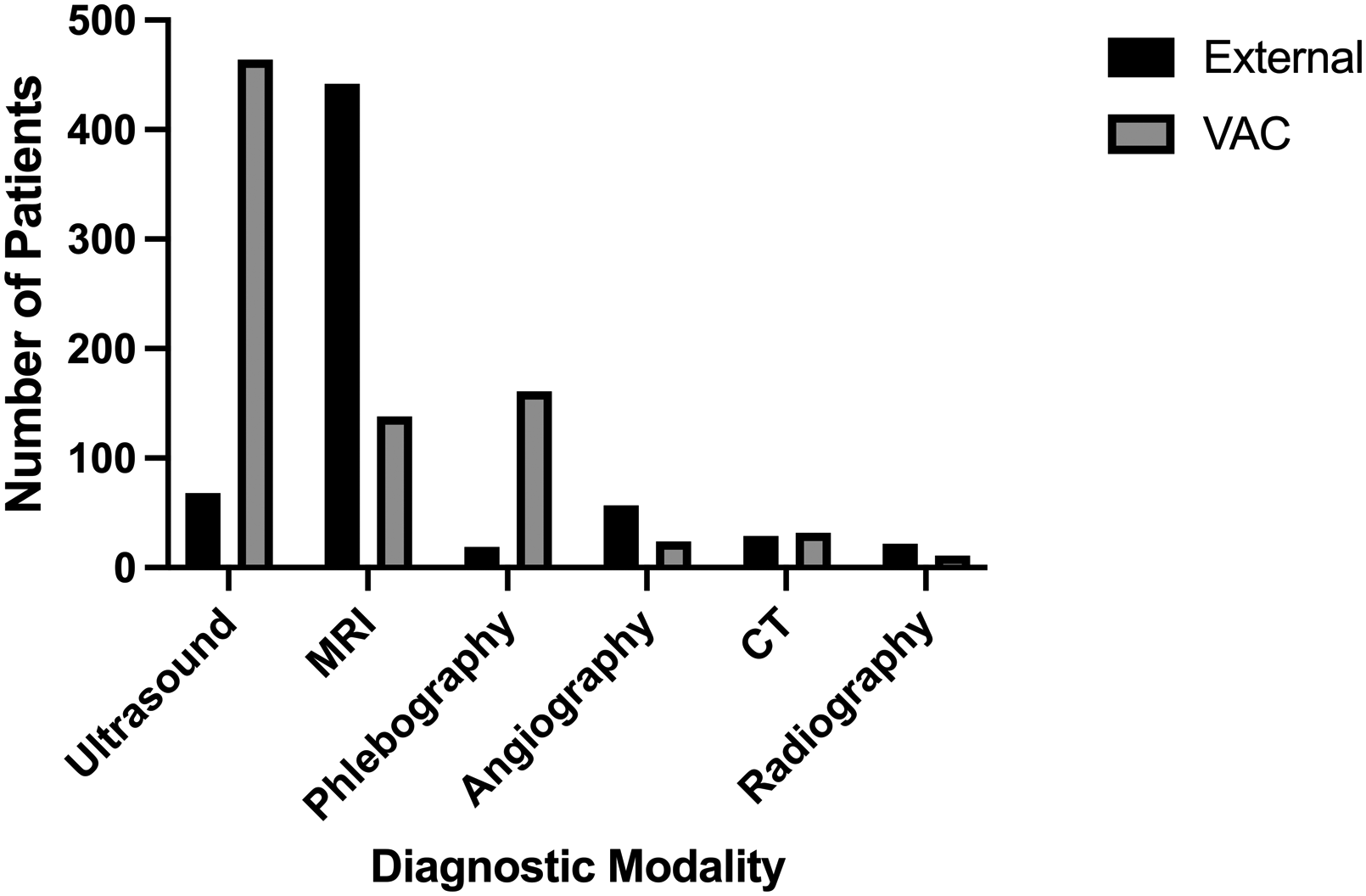
Diagnostic modalities used prior to referrals and during the therapy in the vascular anomaly center (VAC).
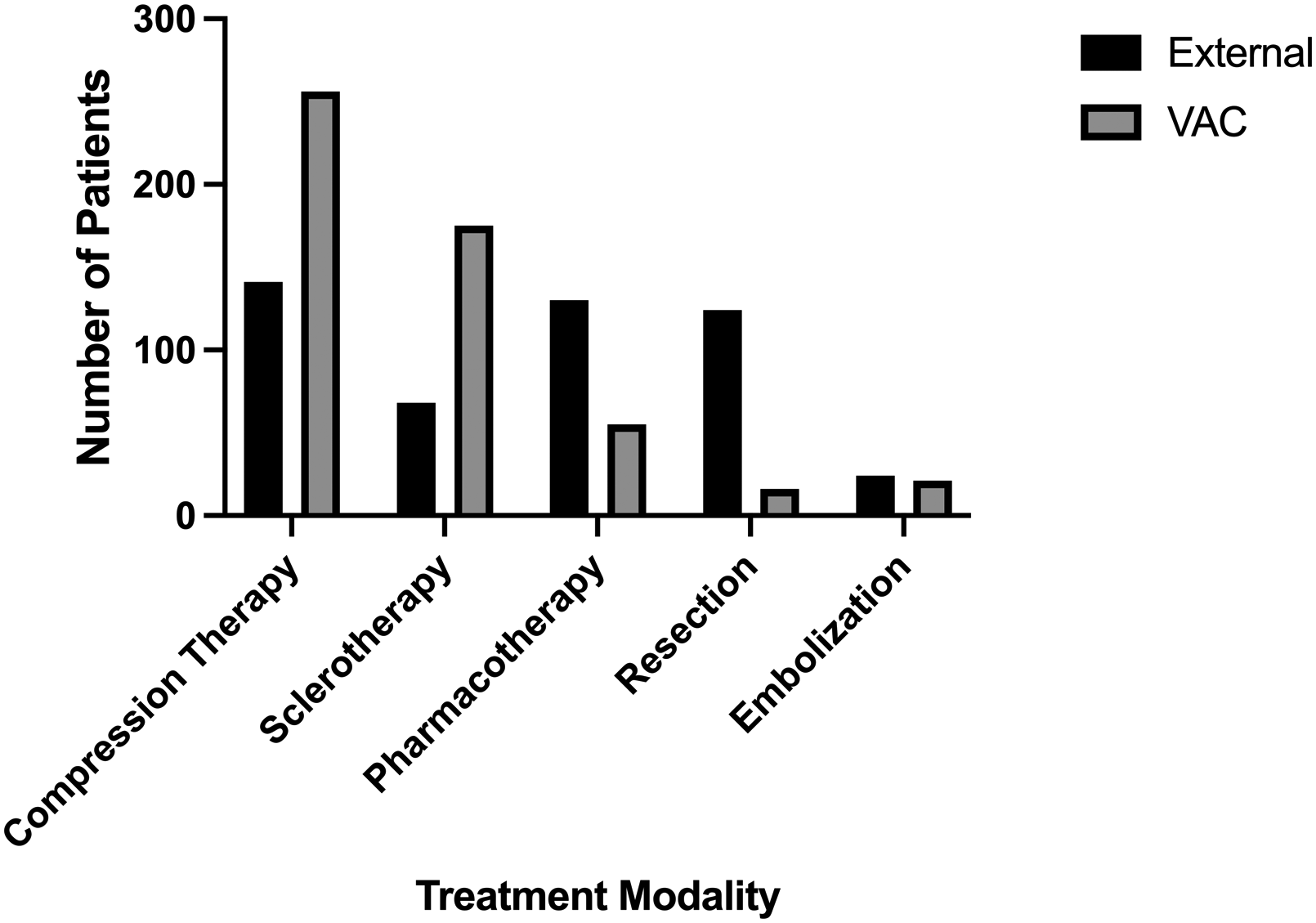
Treatment performed prior the referral to the vascular anomaly center (VAC) and initiated by the VAC.
Many of the patients with incorrect diagnosis before consulting the VAC had already undergone treatment. Table 5 gives an overview of the various interventions that had been carried out in those patients, stratified by the correct diagnosis as determined by the VAC. Of these falsely diagnosed patients, eight received transarterial catheter embolization despite the fact that the underlying diagnosis in these eight patients was a venous malformation. Nineteen patients had been treated with sclerotherapy, one of who suffered from an arteriovenous malformation. Most of the falsely diagnosed patients, referred from an external institution, had been treated surgically with resection of the vascular anomaly (n = 52). Patients suffering from venous malformations underwent resection in 31 cases. Pharmacological treatment with anticoagulation was initiated in six patients, out of whom one was diagnosed in our facility with a parenchymatous tumor and not a vascular malformation. Compression garments were prescribed in 43 misdiagnosed patients and four of these patients were suffering parenchymatous tumors. In total, 137 treatments were performed in patients that had not been correctly diagnosed. Interestingly, there were 35 cases in which a biopsy had been obtained and the final diagnosis was non-concordant with the pathology report.
Therapy of falsely diagnosed patients.

AVM, arteriovenous malformation; CM, capillary malformation; CVM, capillary–venous malformation; LM, lymphatic malformation; VM, venous malformation; VMCM, venous malformation, cutaneous and mucous; FAVA, fibro-adipose vascular anomaly.
Diagnostic and therapy in the VAC
In several patients diagnostic imaging had to be repeated in the. In 138 patients (23.3% of all 593 patients) new MRIs had to be obtained, as the region of interest was not fully documented on the previous scans and therefore relevant information, such as the patency of the deep venous system, could not be obtained. CT scans were performed in 32 cases (5.4%), mainly for planning of radiological interventional therapies. Conventional transcatheter digital subtraction angiographies (n = 24; 4.0%) and percutaneous phlebographies (n = 161; 27.2%) were frequently performed in the VAC in the context of a therapeutic approach to the vascular anomaly during the treatment sessions.
Native radiographs were acquired in eleven patients (1.9%). Ultrasound, indifferent of the underlying vascular anomaly, was by far the most applied diagnostic tool in 78.2% of the patients visiting the VAC (n = 464).
In the VAC, the most frequently prescribed conservative treatment approach was compression therapy (n = 256; 43.2%). A large number of patients were treated with sclerotherapy which was related to the high percentage of venous malformations in the patient population (n = 175; 29.5%). Pharmacotherapy was used in 9.3% of the patients (n = 55) and included analgesics (n = 32; 5.4%), anticoagulation (n = 48; 8.1%) and propranolol (n = 1; 0.2%). Surgical interventions were performed in 3.0% of patients (comprising of surgical resection and reconstruction of functional structures, tendon transfers and local or free flaps if needed). Figs 3 and 4 provide an overview of the diagnostic and therapeutic measures prior to and during the therapy in the VAC.
Discussion
Vascular anomalies and vascular tumors represent rare diseases. Their diagnosis and treatment remain a challenge, no matter which clinical specialty is confronted with them, either in the elective outpatient management or in the acute emergency setting. This has catalyzed the formation of dedicated vascular anomaly centers, where experts in the field can offer specialized consultation and care to affected patients. In a previous study, we analyzed the composition of referrals to a VAC and observed that, in our patient collective, venous malformations were the most frequently encountered vascular anomaly compared to lymphatic malformations and arteriovenous malformations (11). In the present study, we aimed to further analyze the patient characteristics and disease spectrum of a German vascular anomaly center.
All but two of the 593 patients had previously sought medical advice for their vascular malformations before their first visit to the VAC. It was common for patients to have multiple consultations with physicians from diverse specialties before being referred to, or independently arranging, an appointment at our VAC. Complex cases with involvement of multiple anatomical sites and/or comorbidities due to syndromal vascular malformations had significantly more previous doctor's appointments than patients with simpler vascular malformations. In certain cases, patients had seen as many as eight or 10 different doctors before being referred to a VAC. This underscores the diagnostic and therapeutic challenges confronting physicians without comprehensive knowledge in the field of vascular malformations, when approached by affected patients. Considering the overall incidence of approximately 1% of vascular anomalies in Germany, around 850,000 affected patients can be considered to be suffering from this rare disease. It is therefore reasonable to assume that many patients with vascular malformations are not under treatment in specialized centers. The problem of frequent misdiagnosis has been pointed out many times before and was indeed also confirmed in the present study (3,5,6,11,12). A retrospective analysis by Hassanein et al. (13) revealed that patients with misdiagnosed hemangiomas had a significantly higher risk of receiving incorrect treatment.
In the present study, several misdiagnosed patients received treatments that are usually not indicated for true vascular malformations. In one case, unfortunately, sclerotherapy was performed in an arteriovenous malformation with fast-flow arteriovenous shunting, although transcatheter embolization would have been the gold standard (11,14,15). Thirty-one patients suffering from venous malformations were treated by surgical resection. The consensus is that the therapy of choice for most venous malformations is sclerotherapy and surgical treatment is usually reserved for limited cases, where complete excision without significant morbidity is feasible or for staged approaches (16). Notably, one patient who did not have a vascular anomaly had nonetheless been treated for it. This highlights the importance of correct diagnosis of vascular anomalies. We therefore wish to emphasize that, if there is any doubt about the nature of a suspected vascular malformation or lesion, the patient should be referred to a specialized VAC.
Interestingly, we have shown a low frequency of ultrasound imaging, with only 11.5% of patients having had an ultrasound examination upon referral. These numbers require cautious interpretation because potential insufficient documentation of the ultrasound examination in the doctor's records may have misrepresented the real frequency. Yet, grey scale sonography with and without color coded Doppler is considered as a widely available diagnostic tool in pediatric and adult medicine and is commonly applicable for many physicians as a valuable screening tool.
Because our data have been gathered in a retrospective review and not via interviews, cases in which an ultrasound had been performed but not documented would not have been identified. In almost 80% of the patients referred to the VAC, a new ultrasound examination was performed. This fact once more underlines the importance of ultrasound in the management of vascular anomalies. The most used imaging modality in the outpatient setting was MRI, while native radiographs and CT scans were scarcely utilized. MRI, being the modality of choice for high resolution imaging of soft tissue, obviously shown to be the diagnostic gold standard. Imaging also includes MR-based dynamic perfusion angiography or venography and patient examinations, especially in the rather young population consulting the VAC, aiming to avoid unnecessary radiation exposure (16,17). The value of 4D CT scanning in our institution lies in the diagnostic assessment of fast flow arteriovenous malformations prior to catheter embolization.
We consider ultrasound as the optimal tool for the primary evaluation in case of a suspected vascular malformation. Chang et al. (18) have shown its usefulness for diagnosing early high-flow malformations in a clinical trial. Despite surprisingly little evidence supporting the use of ultrasonography for vascular malformations, most clinicians agree that ultrasound is crucial in the diagnostic work-up, intra-procedural management during treatment and post-therapeutic follow-up of vascular anomalies (19–23).
The spectrum of treatment in patients prior to their referral to the VAC was heterogenous. While 17.3% (n = 80) of cases had undergone surgical treatment, only 15.5% of patients had received interventional therapy, either in the form of sclerotherapy (11.5%; n = 68) or transcatheter embolization (4.0%; n = 24). Once admitted to the VAC, a large proportion of patients received sclerotherapy (29.5; n = 175) and transcatheter embolization (3.5%; n = 21). Surgery was rather underrepresented (3.0%; n = 16).
This distribution of therapies in the VAC is related to the vast number of venous malformations in our cohort and the fact that we do not routinely perform incisional biopsies in our institution. Pharmacotherapy was more common prior to referral to the VAC and mostly consisted of analgetic medication and anticoagulation. Additionally, a large percentage of patients received compression therapy using customized compression garments (43.2%; n = 256) in the VAC compared to the antecedent therapy (23.8%; n = 141).
However, there is a distinctive lack of prospective studies evaluating the use of compression therapy for vascular anomalies (24). Nonetheless, it seems to provide a clear clinical benefit to patients with vascular anomalies, leading to improved functionality and pain relief. Patient-reported outcome measures have gained more attention in the scientific community and could be well implemented in the further analysis of the care for vascular malformations. For example, Weitz-Tuoretmaa et al. (25) had successfully implemented such questionnaires in a study examining endovascular sclerotherapy for venous malformations.
Our study has some limitations. First, the data were derived from a retrospective chart analysis and therefore might have some bias. The incidence of ultrasound imaging prior to referral and the number of previous appointments might be too low, since these factors were not always documented well. Furthermore, the study is single-center design and the results may differ in other centers or in other countries. Yet one must consider that the formation of dedicated VACs with extensive experience in diagnosis and treatment are rare in Germany and data comparison with other institutions may prove difficult. Unfortunately, we do not have data on the medical care of patients with vascular malformations before the establishment of the VAC. To our knowledge, there has been no previous data published on this topic in Germany, which would have allowed us to draw conclusions about the frequency and treatment modalities of vascular anomalies in our country. Prior to the establishment of the VAC, patients consulted various clinical institutions without any standardized diagnostic or therapeutic approach. This situation applies to all centers currently dealing with vascular anomalies in Germany. Undoubtedly, more high-quality prospective trials and establishment of VACs are needed to further improve the understanding of vascular anomalies.
In conclusion, patients suffering from vascular anomalies often go through numerous appointments and have a high risk of misdiagnosis and mistreatment prolonging the medical condition. The present study reemphasizes once more that patients with vascular anomalies should be treated in a dedicated vascular anomaly center, where specialized diagnostic and therapeutic dedicated patient care can contribute to more precise and rapid treatment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
The study was granted approval by the local ethics committee (ID-Nr. 2016-522N-MA).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
