Abstract
Background
Endovascular aortic repair with fenestrated or branched endografts is technically challenging, and proper intraoperative assessment of all stent graft components with only angiography and fluoroscopy can be difficult. Intraoperative computed tomography (CT) imaging can be a valuable aid for the operators in the evaluation of stent grafting results prior to completion of surgery.
Purpose
To examine the feasibility of performing intraoperative CT imaging during fenestrated and branched endovascular aortic repair (f-bEVAR) under sterile conditions and with patients under general anesthesia.
Material and Methods
Intraoperative CT imaging was performed in 10 patients undergoing elective aortic repair with fenestrated or branched endografts. Adverse events, time consumption for CT set-up and image acquisition, and additional radiation dose to the patient were recorded. CT image quality was graded. Immediate corrective maneuvers performed based on the CT findings was registered.
Results
There were no adverse events related to intraoperative CT imaging. The median additional operating time by including intraoperative CT was 16 min (interquartile range [IQR] = 12–19), comprising 7% of the median total operating time. The median estimated additional radiation dose to the patient was 4.8 mSv (IQR = 3.8–4.9). All intraoperative CT examinations were considered to be of sufficient quality for stent graft evaluation. No immediate corrective procedures were performed on the basis of CT findings in this study cohort.
Conclusion
CT imaging intraoperatively during f-bEVAR is feasible with an acceptable increase in operating time and radiation dose.
Introduction
Endovascular treatment of thoracoabdominal and juxtarenal aortic aneurysms with fenestrated and branched endografts is well-established, although still considered technically demanding (1). Computed tomography (CT) is the gold standard in both preoperative planning and postoperative surveillance in endovascular treatment of aortic disease (2). The accurate placement of all endograft components is crucial for a durable result. In fenestrated and branched endovascular aortic repair (f-bEVAR), satisfactory assessment of all endograft components can be difficult with two-dimensional angiography and fluoroscopy alone (3–9). Unwanted findings such as stent compression or kinking, insufficient bridging or flaring of branch stents are easily detected on conventional CT or cone beam computed tomography (CBCT) imaging (4,7–11). Intraoperative CBCT for the evaluation of structural problems may detect findings requiring immediate corrective maneuvers, likely avoiding secondary interventions (5,7,10–12). The limitations of CBCT include a limited field of view, increased radiation dose to the patient, and reduced image quality due to metal artifacts (5,7). As a further development of hybrid operating suites, different manufacturers now offer the combination of an operating table with angiography and a CT scanner. In our institution, a system comprising a floor mounted c-arm (Siemens ARTIS pheno; Siemens Healthcare GmbH, Erlangen, Germany), a sliding CT scanner (Siemens SOMATOM Edge; Siemens Healthcare GmbH, Erlangen, Germany), and a radiolucent carbon fiber operating table (Maquet Magnus; Getinge AB, Gothenburg, Sweden) was installed in 2018. The use of a sliding CT scanner during stent graft procedures is however not without a set of logistical and practical challenges. The aim of the present study was to examine the feasibility of performing CT imaging intraoperatively during f-bEVAR under sterile conditions and with patients under general anesthesia. The secondary endpoints were additional time consumption, radiation dose to the patient, CT image quality compared to gold standard CT imaging, and number of corrective maneuvers performed on the basis of intraoperative CT findings.
Material and Methods
After approval from the regional ethics committee, 10 patients undergoing elective fEVAR or bEVAR in 2020–2021 were enrolled in this study. Written informed consent was obtained from all patients. Patient characteristics, pathology, and stent grafts used are presented in Table 1. All patients were treated in accordance with the standard protocol in our institution. In short, endovascular aortic repair was performed in the hybrid operating suite with the patient under general anesthesia. Image fusion with preoperative CT, with superimposed markings for the branch vessel ostia, was used (11). Unilateral surgical cut-down with exposure of the femoral vessels was performed, whereas the contralateral femoral artery was accessed percutaneously. After the main graft body deployment, target vessels were catheterized from a femoral access using a steerable sheet introducer (Heartspan®; Merit Medical, South Jordan, UT, USA or TourGuide™; Medtronic, Minneapolis, MN, USA) in both fEVAR and bEVAR procedures. After completed deployment of all endograft components, a uniplanar digital subtraction angiogram (DSA) was obtained using a power injector. Structural inadequacies of the stent graft components and early endoleaks depicted on the angiogram were corrected before proceeding with the CT scan, if possible.
Pathology and stent grafts used.

*Cook Medical, IN, USA.
Cook Zenith t-Branch® (Cook Medical, IN, USA).
CMD, custom-manufactured device; TAA, thoracoabdominal aortic aneurysm.
Before the CT scan, metal guidewires were exchanged with a diagnostic catheter in order to reduce metal artifacts. The patient was covered in transparent sterile drapes, and the arms were kept in position alongside the body to reduce the risk of compromising sterility. The CT gantry was slide-in from the patient’s head side. The C-arm, lead shields, operation lamps, monitors, infusion pumps, and other anesthesiology equipment were moved to avoid collision with the CT scanner. The ventilator tube was disconnected, pulled through the CT gantry, and reconnected. The set-up is displayed in Fig. 1.
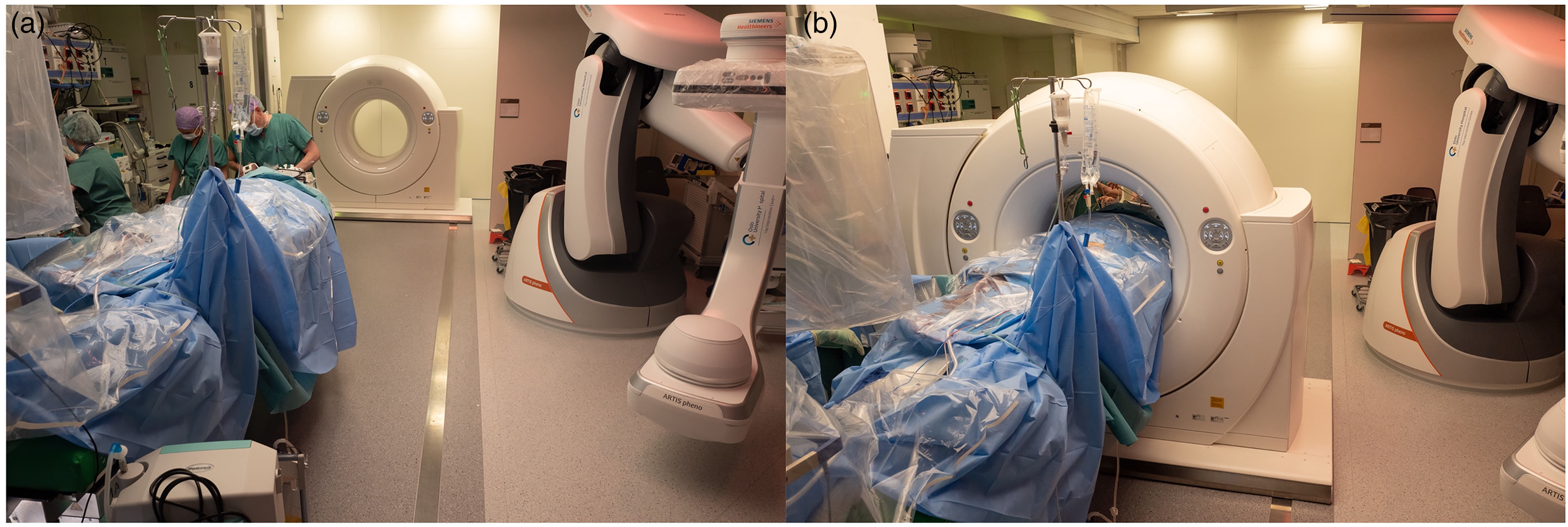
(a, b) The C-arm is parked away from the patient and the sliding CT scanner is positioned from the head of the operating table.
The scan range, including the entire stent graft, was defined on a scout view. Non-enhanced intraoperative CT scanning was performed with simultaneous breath-hold and automatic dose modulation. Scanning and reconstruction parameters are presented in Table 2. The reconstructed images were evaluated by the operator on the workstation. Provided that no structural problems were detected, the surgery proceeded towards completion. CT angiography was obtained in the radiology department on a dual-source scanner (Siemens SOMATOM Force; Siemens Healthcare GmbH, Erlangen, Germany) on the first postoperative day. The postoperative CT protocol included a non-contrast scan, an arterial phase scan, and a delayed contrast phase scan.
Scanning and reconstruction parameters.

Qref, quality reference.
Unanticipated problems with anesthesiologic equipment or patient condition hindering intraoperative CT as well as the inability to preserve sterility in the operating field were considered adverse events. Time elapsed from the assessment of completed DSA until reconstructed intraoperative CT images were available for evaluation was defined as intraoperative CT time consumption. Operating time was defined from time of skin incision to time of incision closure. Adverse events, time consumption for set-up and performing intraoperative CT, total operating time, radiation dose parameters from intraoperative and postoperative CT as well as intraoperative CT findings and number of corrective maneuvers were recorded. Technical success was defined as ability to obtain intraoperative CT imaging. Procedural technical success was defined as satisfactory deployment of aortic stent graft, iliac limbs, and all intended branch stents. Stent graft compression, infolding, kinking as well as inadequate branch stent length or adaption to the fenestration or target vessel wall were considered findings warranting immediate corrective intervention. An estimated effective radiation dose from intraoperative CT was calculated from the dose length product (DLP) multiplied with a conversion factor of 0.017; the normalized value of effective dose per DLP for abdominal scans (13). In a similar manner, an estimated effective radiation dose was calculated for the comparable non-enhanced scan from postoperative CT.
Two experienced vascular radiologists reviewed the intraoperative and postoperative CT scans independently and in a random order. The readers evaluated the image quality by utilizing a visual grading scale ranging from 1 (excellent) to 5 (poor) (14,15), and overall examination quality in terms of diagnostic acceptance. The statistical analysis with the Wilcoxon sign rank test was performed in SPSS version 26 (IBM Corp., Armonk, N.Y., USA). A P value ≤0.05 was considered statistically significant. Image examples from intraoperative and postoperative scans are displayed in Figs. 2–4.
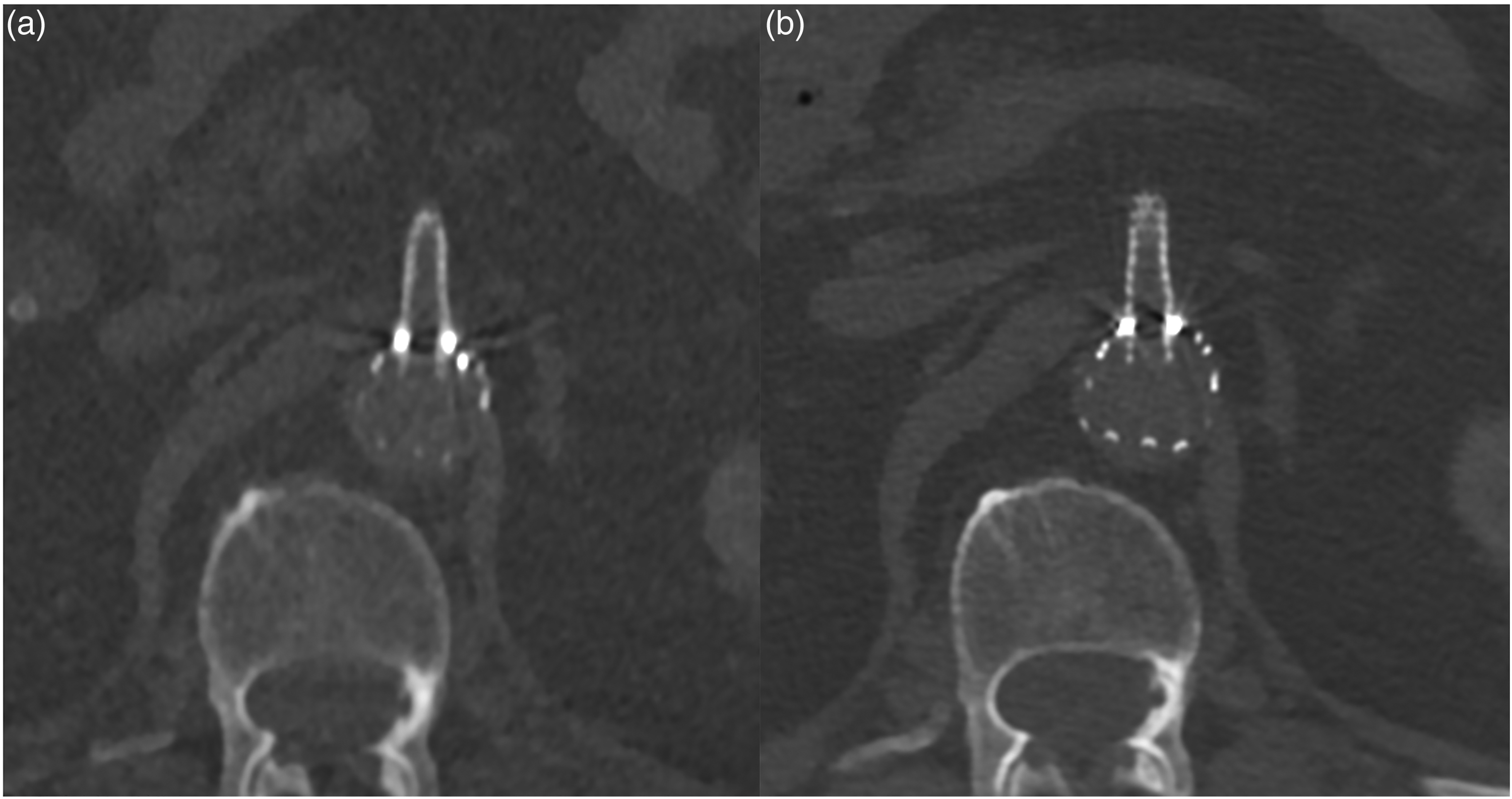
Axial image of proximal superior mesenteric branch stent (fEVAR) in (a) intraoperative and (b) postoperative CT imaging.
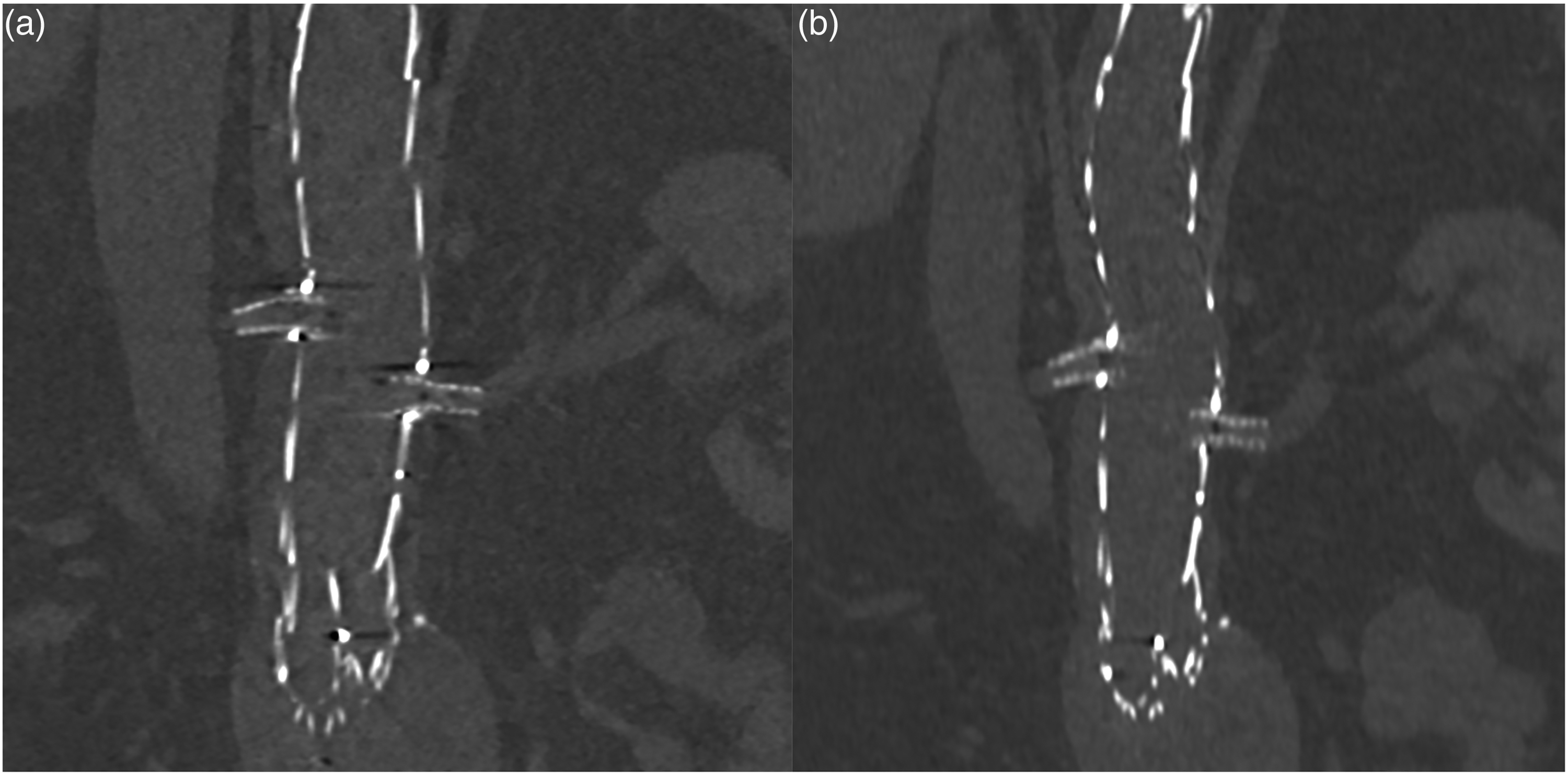
Reconstructed coronal image of proximal renal artery branch stents (fEVAR) in (a) intraoperative and (b) postoperative CT imaging.
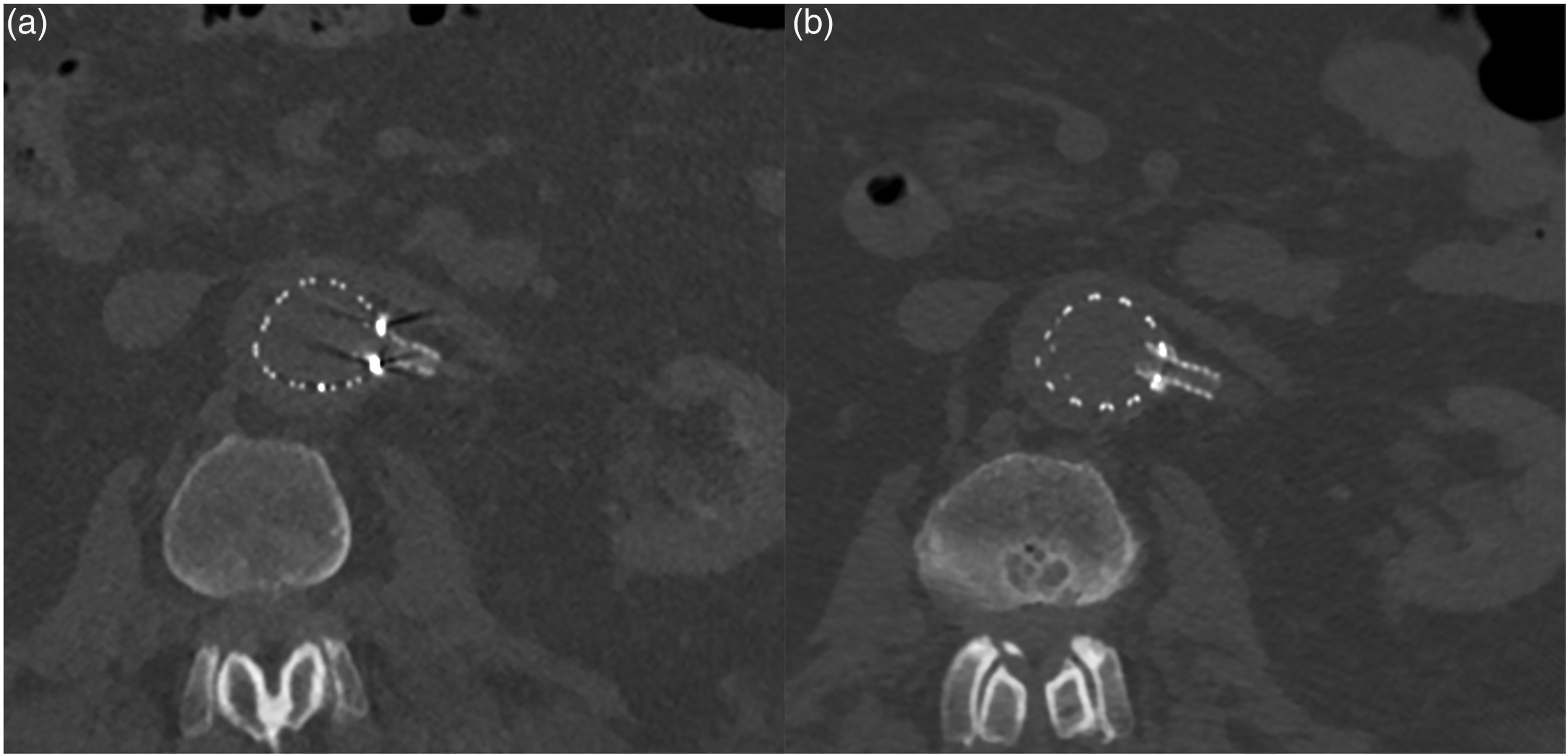
Axial image of proximal left renal artery branch stent (fEVAR) in (a) intraoperative and (b) postoperative CT imaging.
Results
Technical success with completed intraoperative CT imaging was achieved in all 10 patients. No adverse events related to intraoperative CT imaging were recorded. There were no findings on intraoperative CT requiring immediate corrective maneuvers. Procedural technical success was achieved in 9 out of 10 patients. In one patient treated with a branched endograft, the operators were not able to position a left renal artery branch stent due to pronounced vessel tortuosity. This branch stent was later successfully placed in a secondary procedure. In one patient, the distal sealing zone in the left iliac artery was deemed insufficient on DSA, and the iliac limb were extended before intraoperative CT.
Table 3 displays the median time consumption for set-up and completion of intraoperative CT, total operating time, and patient radiation dose. Intraoperative CT comprised a median 7% of the total operating time. The median estimated effective dose for non-enhanced scans on postoperative CT was significantly lower than intraoperative scans (P = 0.005). Relative visual grading showed that overall image quality in intraoperative CT was inferior to non-enhanced postoperative CT (P = 0.007). All examinations were evaluated to be of diagnostic quality. Overall image quality grading scores are presented in Fig. 5.
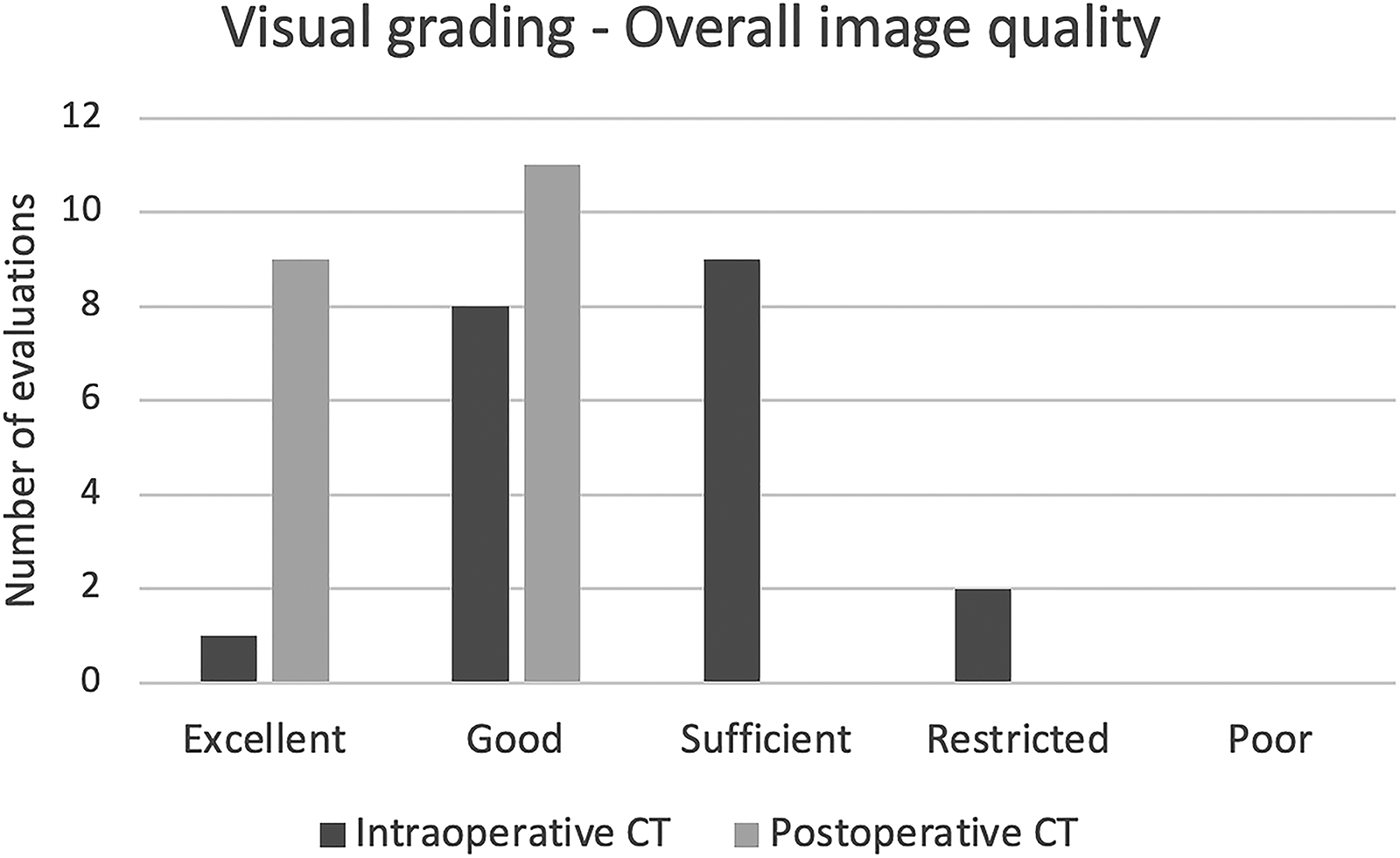
Grading distribution of overall image quality in intraoperative and postoperative CT examinations.
Investigational data.
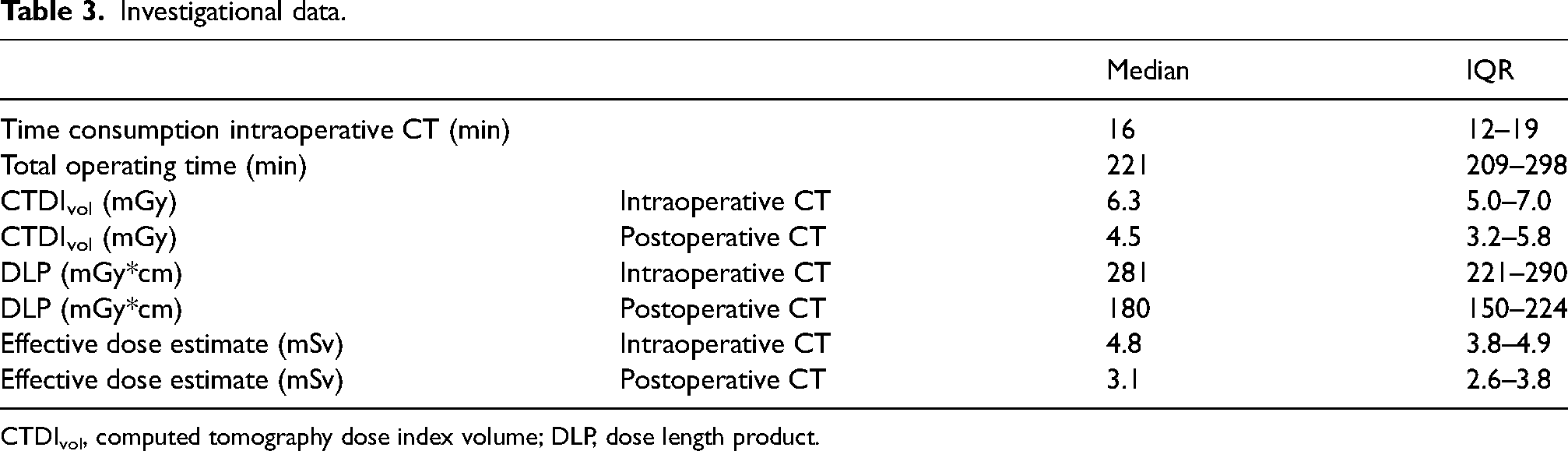
CTDIvol, computed tomography dose index volume; DLP, dose length product.
Discussion
The present study demonstrates the feasibility of performing CT imaging intraoperatively during f-bEVAR procedures. The intraoperative CT scans had sufficient quality for the evaluation of potential structural problems of the stent graft components. Several previous studies have with the same rationale demonstrated the feasibility and utility of performing CBCT both with and without contrast enhancement during aortic stent graft procedures (4–7,9–12). Although similar stent graft assessment can be made by CBCT, CT imaging has superior contrast resolution compared to CBCT, in addition to advantages with respect to field of view and scan length as well as to the possibility of applying artefact reduction algorithms.
Opposed to studies utilizing CBCT (5,7,12), we did not demonstrate CT findings requiring prompt corrective interventions. In one study, CBCT demonstrated stent graft kinks or compressions in 22% of f-bEVAR patients, and approximately 10% underwent immediate revision (7). The fact that we did not demonstrate any positive CT findings may be due to the small number of patients in our study. We did not perform additional CBCT for comparison to intraoperative CT as this would have resulted in an increased cumulated radiation dose without any expected clinical benefit for the patient.
A disadvantage of CT imaging compared to CBCT is the additional time necessary for readying the patient and positioning the CT scanner. However, with a median time of 16 min, comprising 7% of the median total operating time, we found the time consumption to be acceptable. In the earliest procedures of the study, moving and reconnecting the anesthesia equipment proved to be time consuming. Throughout the study, this improved to be executed very efficiently. Time spent on the acquisition and reconstruction of CT images was negligible. One extra CT radiographer was present during the CT examination for positioning the CT scanner and image acquisition. Advanced technological set-ups require user proficiency among operators and radiographers, and a certain amount of training seems mandatory to master the equipment efficiently during surgery.
The median estimated effective radiation dose to the patient on intraoperative CT was higher compared to postoperative CT. One cause is the positioning of the patient’s arms alongside the body on intraoperative CT. This may be addressed by not applying automatic dose modulation on intraoperative CT scans. More experience might allow for optimalization of the intraoperative CT protocol with concomitant reduction in radiation dose. The median estimated effective dose in both intraoperative and postoperative CT are well below the reference values for abdominal scans (16). A recent systematic review by Hansrani et al. finds the use of intraoperative contrast-enhanced CBCT appropriate and suggests reducing the total radiation dose to the patient by omitting the control CT exam at discharge (17). By demonstrating the feasibility of intraoperative CT imaging, it follows that contrast-enhanced intraoperative CT imaging might analogously be used to assess for potential endoleaks and complications, such as dissection and embolism. Thus, it possibly omits the need for early postoperative CT control.
The addition of a CT scanner in a hybrid operating suite is a substantial economic investment. In our institution, the sliding CT is located in a research unit and made available for multiple surgical specialties in two adjacent operating suites. The added value of such an investment to standard hospital routine has still to be determined.
The present study has some limitations. These include the low number of patients in the study cohort. With the implementation of new equipment we expect a learning curve. Gaining experience may allow for a reduction in radiation dose and time consumption.
In conclusion, performing CT imaging intraoperatively during f-bEVAR procedures is feasible. Additional time consumption and radiation dose is acceptable, and the CT examinations have sufficient quality for assessing stent graft architecture.
Footnotes
Data availability
The datasets analyzed in this study are available from the corresponding authors on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
