Abstract
X-ray contrast media have been reported to have inhibitory effects on bacterial growth. Despite its potentially beneficial effect on patients, these features of contrast media have received relatively little attention in the medical literature in the past decades. The aim of this review is to evaluate the literature concerning the bactericidal and bacteriostatic effects of X-ray contrast media, specifically if there is a known difference concerning these effects between ionic and non-ionic contrast media. Systematic literature review was performed for the years of publication between 1911 and 2019. Since the publication of Grossich in 1911, the effect of iodine on the treatment of superficial infections in surgical procedures has been established clinical knowledge. Bacteriostatic and bactericidal effects of ionic X-ray contrast media are well established. However, non-ionic contrast agents have been the subject of little research in this respect. In past decades, the hypothesis emerged in the literature that mainly the concentration of free iodine might be responsible for any bacteriostatic or bactericidal effect of ionic X-ray contrast media. Nowadays, however, only non-ionic contrast media are used. The question regarding the mechanism and magnitude of bacteriostatic or bactericidal effects of these, non-ionic contrast media, could not be answered conclusively from this review. Non-ionic contrast media could be used intentionally when a local antibacterial effect is intended (e.g. in percutaneous abscess drainage), as well as to reduce the overall dose of antibiotics administered to a patient. Thus, this question remains relevant and might constitute the area of future research.
Keywords
Introduction
The first description of iodine came from the French Salpeter chemist Bernhard Courtois in 1811 as brown-violet vapors emanating a distinct odor when boiling the ashes of seaweed with concentrated sulfuric acid. The naming of the substance followed in 1813 by Joseph Louis Gay-Lussac and Nicolas Clement-Desormes as “iodine” (Greek: iώδης, iodes = violet colored). In 1814, iodine was included by Jöns Jacob Berzelius with the symbol “J” in his atomic weight table.
It then took almost a century, until Grossich (1849–1926) used iodine as a disinfectant in wound care and disinfection in the surgical field in 1911 (1,2). The action principle of disinfection through external application of iodine has long been known and clinically successfully used. In particular, the operative disciplines employed various iodine solutions, tinctures, and ointments (e.g. Braunol®, 2-Pyrrolidinone, 1-ethenyl-, homopolymer, compound with iodine; B. Braun Medical Inc., Melsungen, Germany). Iodine tincture, iodoform, gauze strips soaked with iodine solution, and iodine-soaked deposits in body cavities should all be mentioned here.
This clinical knowledge was taken further in the 1950s by Svoboda and Ježková, who tested iodinated X-ray contrast media in vitro in order to elucidate any bactericidal or bacteriostatic effect on urologic germs (3). The iodine-containing X-ray contrast medium Diodon® (bis(2-hydroxyethyl)azanium;2-(3,5-diiodo-4-oxopyridin-1-yl)acetate; Spofa, Prague, Czech Republic) was tested for its possible germicidal effectiveness (Fig. 1). The effect of Diodon® was investigated in vitro in 70%, 50%, and 35% dilutions with respect to any bactericidal or bacteriostatic effect on various germ populations. Control experiments were carried out with penicillin. Growth stimulation of the investigated bacterial populations as evidence of the lack of bactericidal or bacteriostatic activity could not be detected in a total of 270 samples at constant body temperature (37° C). No further changes in the investigation parameters such as temperature, pH, or osmolality were undertaken. These germs were tested: Staphylococcus albus; S. aureus; S. pyogenes; S. haemolyticus; Sarcina; Streptococcus alpha; Pneumococcus; Corynebacterium pseudodiphthericum; Bacterium anthracoides; Neisseria; Pseudomonas; and Escherichia coli.
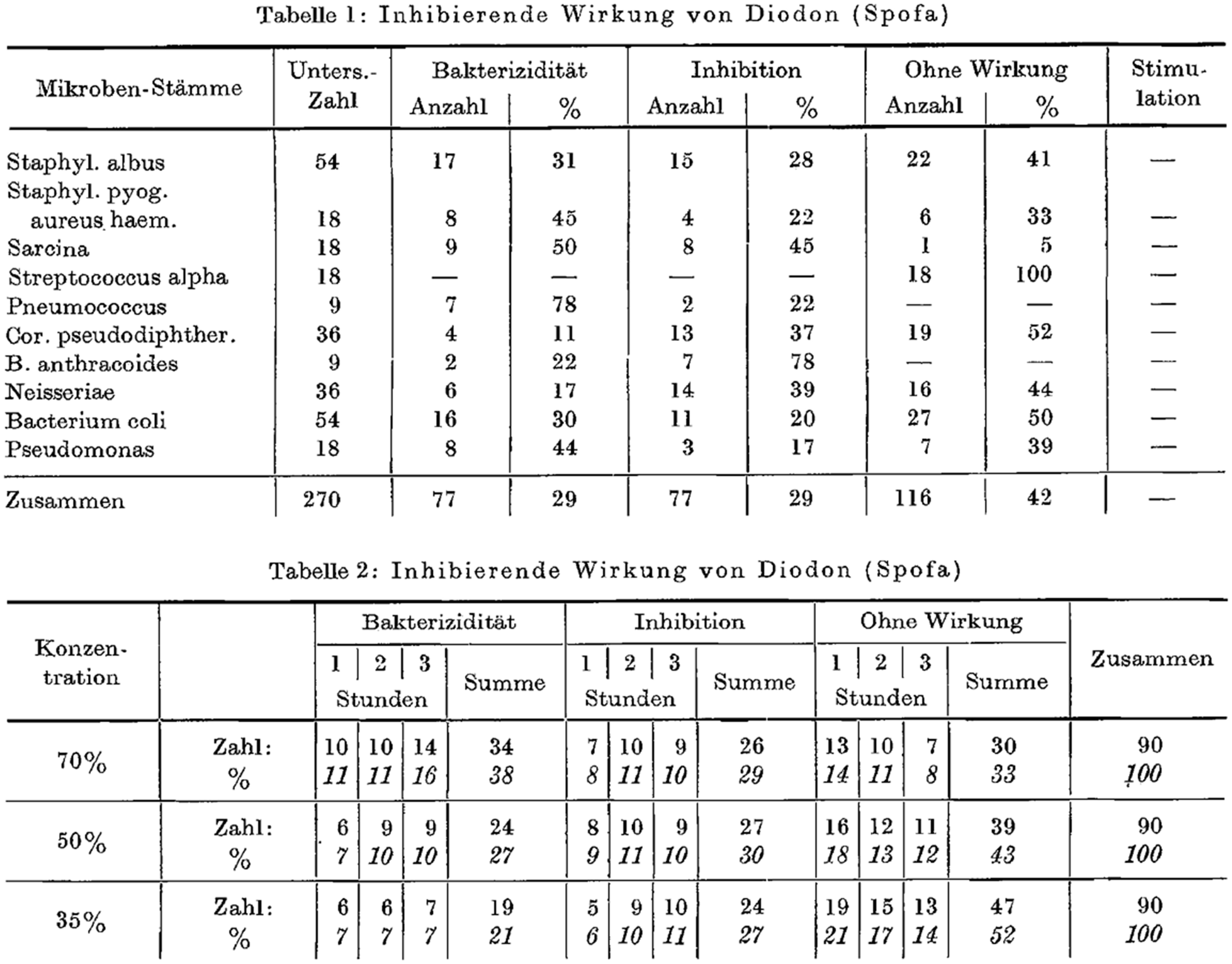
Original tables from Svoboda et al. (3) showing the bactericidal and bacteriostatic effects of Diodon®.
The experiments proved bactericidal activity of Diodon® in the range of 11%–50% (for Sarcina) and bacteriostatic activity in the range of 17%–78% (78% for Bacterium anthracoides).
Bactericidal and bacteriostatic activity was concentration-dependent and, tested over a period up to 3 h, showed the best results at the highest iodine concentration (70% Diodon® in solution).
It was the urologists in the 1960s who recognized the inhibitory effect of iodine in the X-ray contrast media on the growth of bacteria in cystitis (4). However, the iodine is firmly incorporated in the structure of the respective contrast agent molecule. This does not readily explain the inhibitory effect of X-ray contrast media on some bacteria. Since Grossich published his findings in 1911, the disinfecting effect of iodine was current clinical knowledge (1). At least in urology, the locally disinfectant effect of ionic X-ray contrast media was in clinical usage. However, it was commonly implied that only the free, unbound iodine, which is present in a much lesser concentration, can have this disinfecting effect.
Almén is credited with the invention of non-ionic X-ray contrast media, a description of which he published in 1969 (5). Non-ionic contrast media are, due to their chemical composition, susceptible to microbacteria and fungi as investigated in 1983 by Becker et al. (6). The question remained later on whether non-ionic X-ray contrast media have any measurable antimicrobial effect.
Results
In 1969, the urologist H Sachse investigated the influence of X-ray contrast media on urinary tract bacteria in terms of their bactericidal and bacteriostatic effects, with different results (4).
Several X-ray contrast media were investigated, including Urovision® (3,5-diacetamido-2,4,6-triiodobenzoic acid; Schering AG, Berlin, Germany), with an iodine content of 325 mg/mL, Conray® 70 (3-acetamido-2,4,6-triiodo-5-(methylcarbamoyl)benzoic acid; Byk Gulden, Constance, Germany), with an iodine content of 410 mg/mL, Triopac® 200 (sodium;3-acetamido-2,4,6-triiodobenzoate; Cilag AG, Schaffhausen, Switzerland), with an iodine content of 200 mg/mL and Joduron® (an aqueous solution of 2-(3,5-diiodo-4-oxopyridin-1-yl)acetate; Cilag AG, Schaffhausen, Switzerland).
When 0.2 mL of contrast media (either Conray® or Urovision®) were applied on an agar plate and incubated for 24 h at 37° C with S. aureus, Proteus, or E. coli, the bacteria showed no inhibition halo. However, when 0.5 mL of the same contrast media was applied, a very small inhibition halo was found on the agar plate (for the same bacterial strains).
Additionally, the contrast agents Joduron® and Triopac® were also investigated under the same conditions, including the same bacterial strains. However, these contrast agents did not display any signs of bacterial growth inhibition, i.e. there was no distinguishable inhibition halo on the agar plate. This lack of effect on bacterial growth made these contrast media the clinical choice when it was important to still be able to grow cultures from midstream urine after the i.v. administration of iodinated, intravenous X-ray contrast media.
In 1971, Narins et al. (7) investigated possible bacteriostatic and bactericidal effects of another X-ray contrast agent, Hypaque® (sodium;3,5-diacetamido-2,4,6-triiodobenzoate; Winthrop Laboratories, New York, NY, USA). This is a triple-iodinated organic iodide which has a very rapid glomerular filtration rate and has been widely used in the radiological assessment of renal anatomy and function. The studies with Hypaque® were conducted both in vitro and in vivo, and yielded different results. In vitro, Hypaque® showed a bactericidal effect on Proteus, E. coli, and Klebsiella as well as a mild inhibitory effect on Pseudomonas and Enterococcus. In vitro, neither a change in osmolality nor a change in pH showed a bactericidal or bacteriostatic effect on germ growth.
In 1972, Kuhns et al. (8) published in vitro bacteriological efficacy studies with Cystokon® (sodium;3-acetamido-2,4,6-triiodobenzoate 24%; Mallinckrodt Pharmaceutical, St. Louis, MO, USA) and Hypaque® (Figs. 2 and 3). They found that the latter was bactericidal to E. coli, Pseudomonas aeruginosa, Proteus mirabilis, and Proteus vulgaris, whereas Cystokon® showed no comparable bactericidal effect in the same cultures after 5 h of incubation on agar plates.
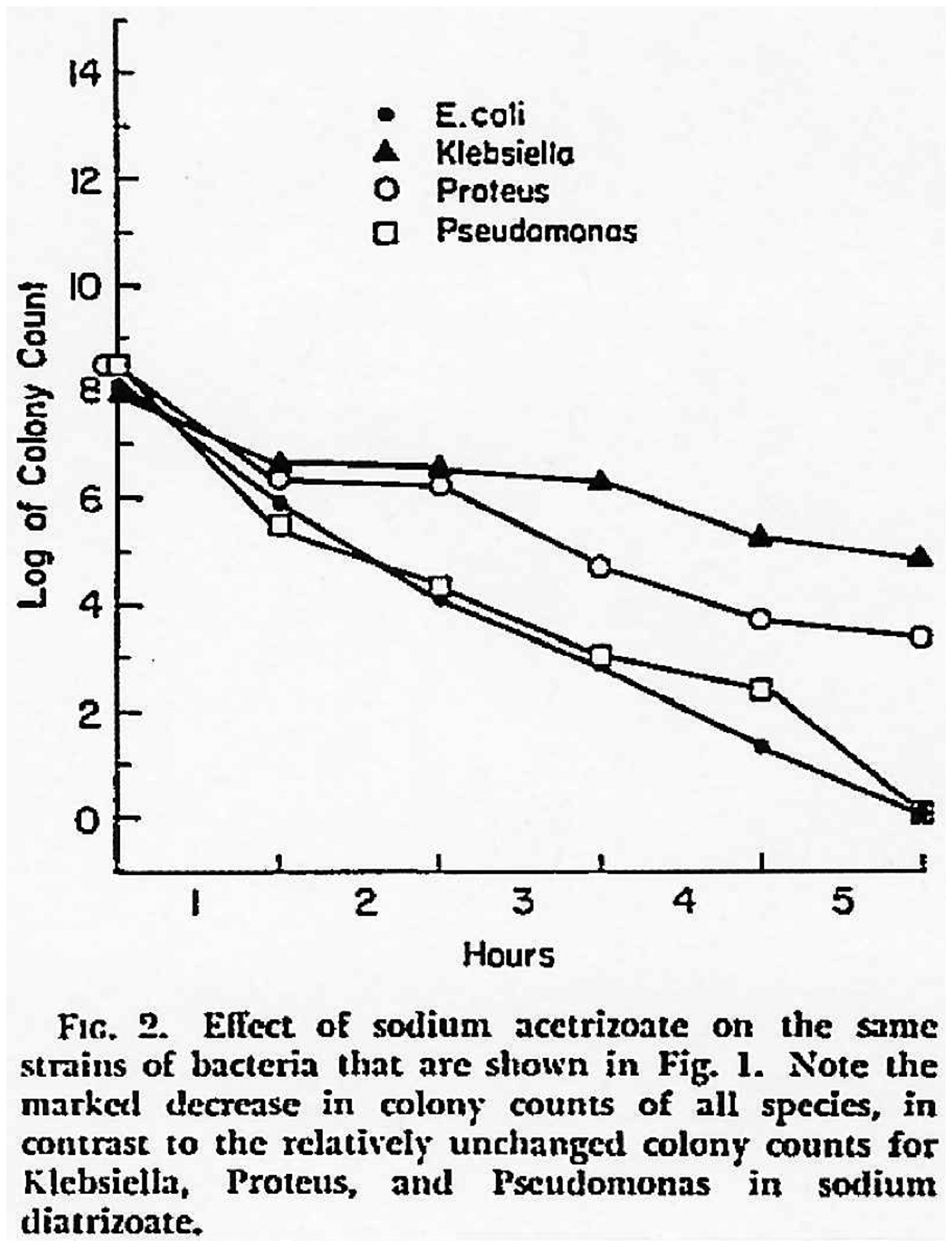
Effect of sodium acetrizoate over time, from the original publication by Kuhns et al. (8).
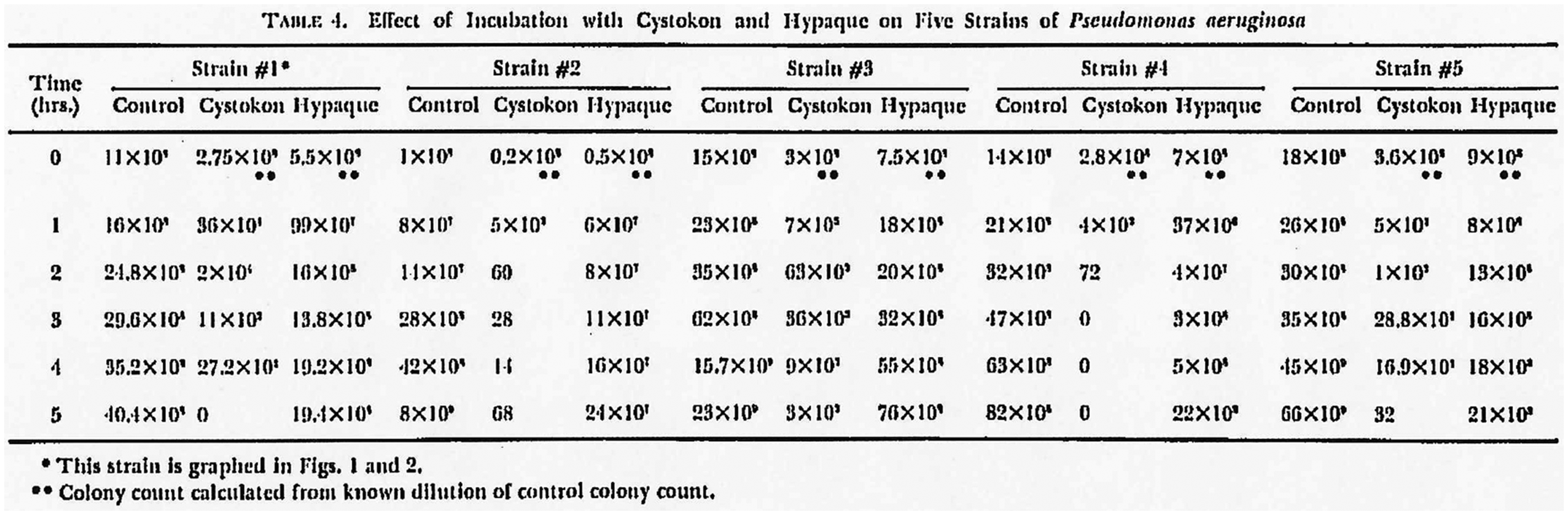
Effect of incubation with Cystokon®, from Kuhns et al. (8).
In 1956, Eyler et al. (9) concluded on a theoretical basis that Hypaque® was the contrast agent of choice in patients undergoing diagnostic cystouretherography for investigation of neurogenic bladder. In the diagnostic workup of bladder rupture, Cystokon® was recommended as a contrast media of choice. Due to its bactericidal nature, it showed fewer side effects when passing into the tissue surrounding the (ruptured) urinary bladder.
After the first basic experimental in vitro studies in the urological field, radiological studies with contrast agents used in arthrography were subsequently performed by Melson et al. (10).
The authors investigated in vitro the bactericidal and bacteriostatic activity of Conray® 400, Renografin® 76 (3,5-diacetamido-2,4,6-triiodobenzoic acid; ER Squibb & Sons, LLC, New York, NY, USA) and Hypaque® 50, which have found use for arthrographic imaging. The germs studied were Streptococcus pyogenes, group A Streptococcus, Haemophylus influenzae, S. aureus, Streptomyces albus, Pseudomonas aeruginosa, Neisseria gonorrhoeae, and Diplococcus pneumonia, over a period of 2–72 h. The study showed no significant bactericidal or bacteriostatic effects.
In 1974, Odiston® 75 (3,5-diacetamido-2,4,6-triiodobenzoic acid; Bayer Pharmaceuticals, Leverkusen, Germany) was investigated both in vitro and in vivo for its effect on urinary tract bacteria by Bignion et al. (11). Of note, Odiston® showed different bactericidal and bacteriostatic activities in vivo compared to in vitro. At iodine concentrations of 0.6 and 0.3 g%, solutions of Odiston® 75 showed in urine cultures in vitro a bactericidal effect on E. coli, Klebsiella, and Proteus. However, at an iodine concentration of 0.035 g%, only a bacteriostatic effect was recorded. For clinical in vivo applications, it was concluded that the administration of 1 mL Odiston® 75 per kilogram bodyweight is required to achieve at least a bacteriostatic effect. The clinical results showed bacteriostasis in 57% of Klebsiella, E. coli, and Proteus strains in the infected urine cultures. The persistent effect could still be detected in the examined urine cultures in 75% of the cases up to 24 h after application. Bactericidal activity, however, was not investigated in the urine cultures obtained in vivo and was subsequently not described.
In 1977, chemically novel X-ray contrast media were investigated by Johansen et al. (12). Isopaque® (3-acetamido-5-[acetyl(methyl)amino]-2,4,6-triiodobenzoic acid; GE Healthcare, Chicago, IL, USA) and Amipaque® (metrizamide, Sanofi, Paris, France) were tested in vitro at the concentrations of 100 mg and 260 mg iodine per mL, respectively. The organisms tested on were P. aeruginosa, E. coli, Proteus vulgaris, S. faecalis, and Streptococcus faecalis. Isopaque® and Amipaque® are water-soluble and were tested in dilution series of 1/10, 1/100, 1/1000, and 1/10,000 g with sterile water. Isopaque® and Amipaque® showed no bactericidal and only a discrete bacteriostatic effect on Proteus vulgaris at the concentrations of 100 mg iodide per mL and 260 mg iodide per mL, respectively. Experiments were performed at 22° C, at pH 7.0 and pH 7.3, over a period of up to 48 h after inoculation. The authors hypothesized that the iodine concentration might be the main factor influencing bacterial growth, although the iodine is bound the contrast media molecule. Even only traces of inorganic iodine (e.g. present by contamination during the manufacturing process) could influence the growth of germs, the authors argued. In the time span of the analyzed literature, this is the first time (1977) that the hypothesis is presented according to which primarily only free iodine might be responsible for the germicidal or at least bacteriostatic effect.
In the 1950s to 1980s, the relevant experiments concerning X-ray contrast media were performed predominantly in vitro. The scientific literature from this period tends to attribute bactericidal or bacteriostatic properties more to the ionic and less to the non-ionic contrast media. There were notable exceptions to this general attribution: in his publications in 1990 and 1991, Speck (13.14) pointed at the low bacteriostatic and bactericidal properties of the non-ionic X-ray contrast agents. In experiments with the non-ionic contrast media Ultravist® (1-N,3-N-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-5-[(2-methoxyacetyl)amino]-3-N-methylbenzene-1,3-dicarboxamide; Bayer Pharmaceuticals, Leverkusen, Germany), Speck showed that the growth of S. aureus was inhibited. However, three out of six examined microorganisms grew in Ultravist® solutions: P. aeruginosa; Candida albicans; and Aspergillus niger. To our knowledge, Speck was the first to prolong the incubation time of his growth tests to 14 days (Fig. 4). Until the studies of Speck, the maximum incubation time in the papers reviewed did not exceed three days. It should also be noted that there was no effect of the Ultravist® solution on the number of gram-positive, spore-forming bacteria Bacillus subtilis (aerobic) and Clostridium sporogenes (anerobic) (13).
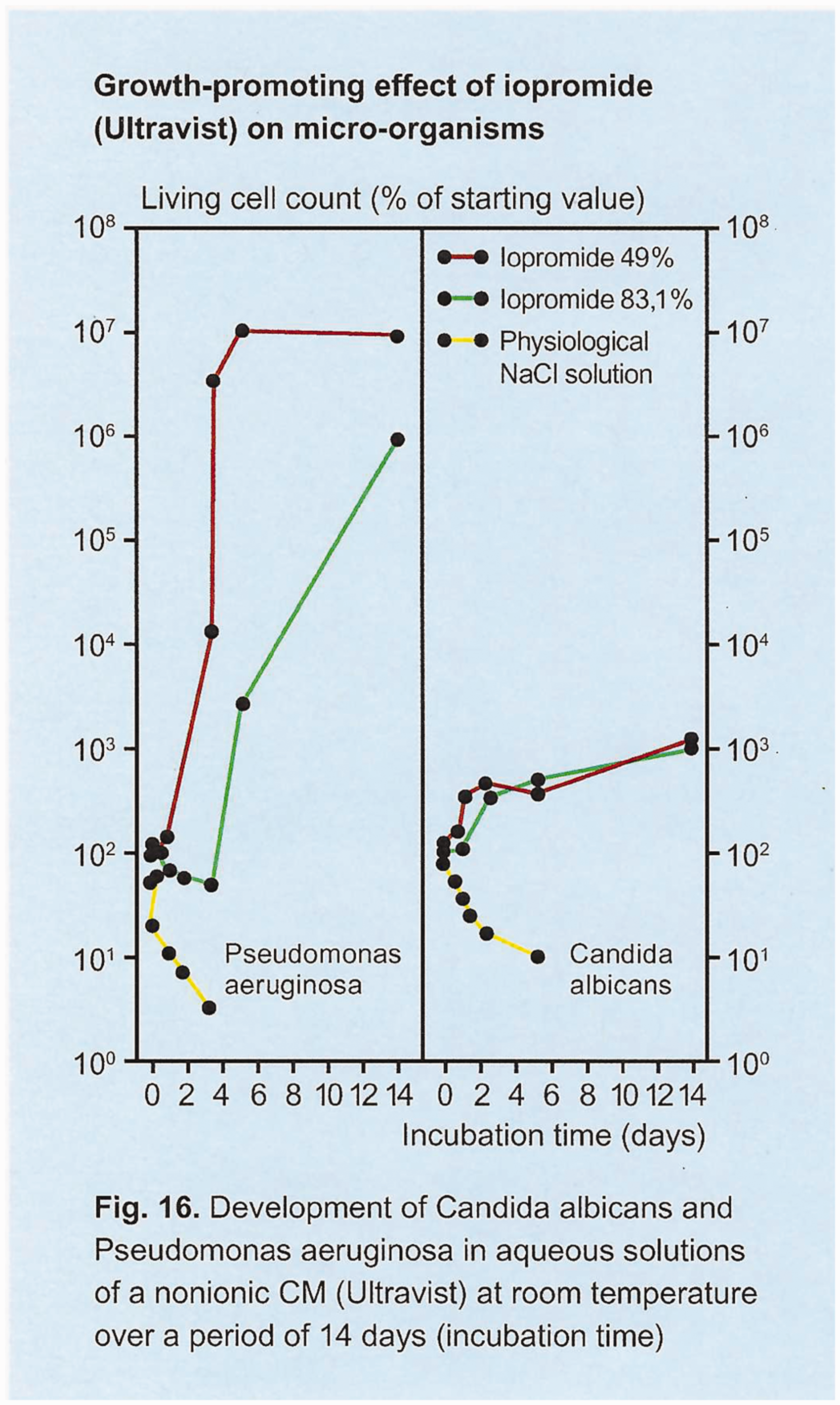
Non-ionic contrast media (Ultravist®) and its growth promoting effect during 14 days of incubation with Candida albicans and Pseudomonas aeruginosa, from Speck et al. (14).
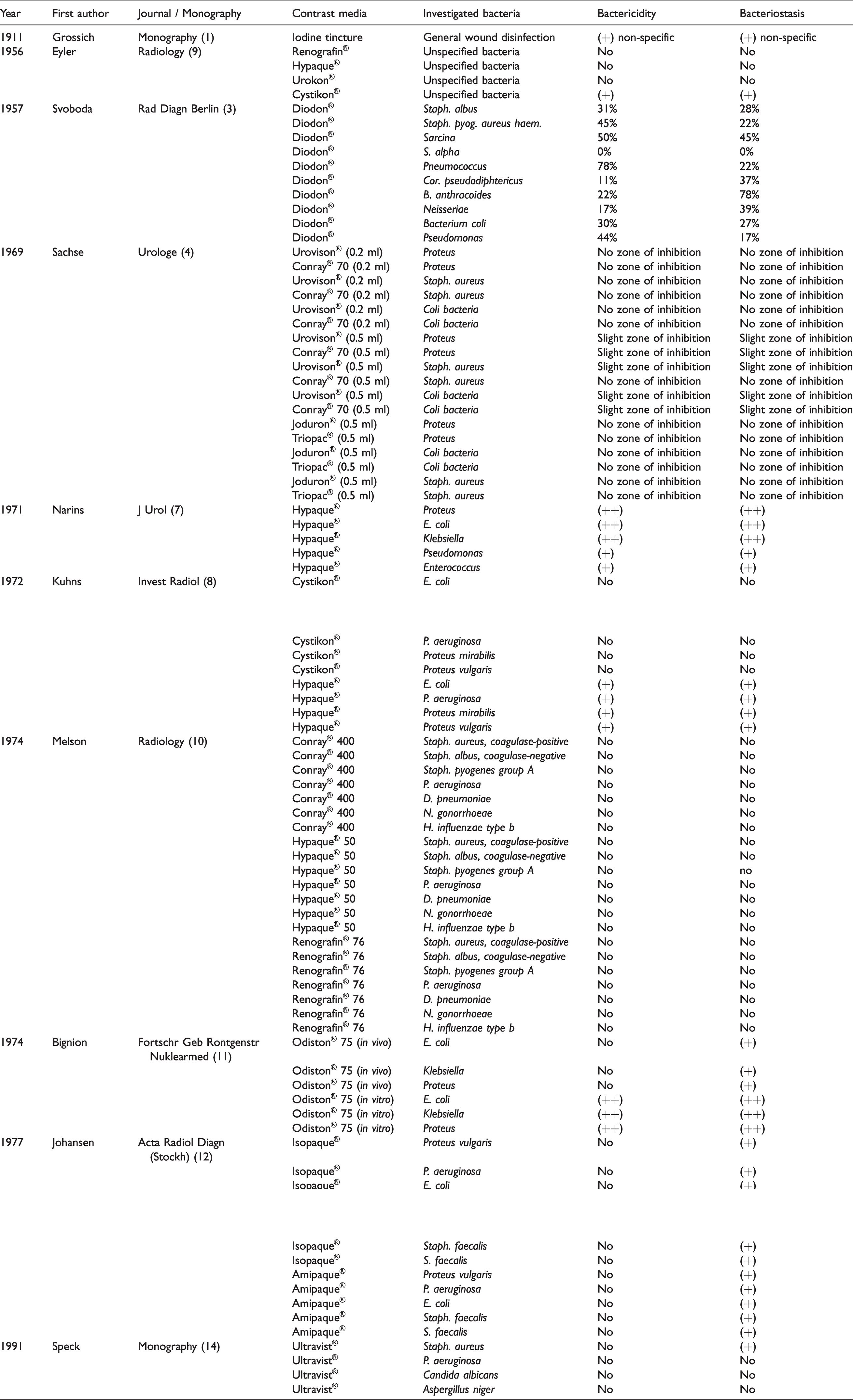
The bactericidal and bacteriostatic effects of the contrast media in the literature analyzed are listed in Table 1.
Chronological list of publications concerning bactericidity and bacteriostasis of X-ray contrast agents.
Based on the assumption that the bactericidal and/or bacteriostatic effects of the non-ionic contrast agents may depend essentially on the proportion of free iodine as a major agent on the hindrance of the growth of bacteria, there are useful data published by Rendl et al. (15) in 2001 (Figs. 5 and 6) concerning the content of free inorganic iodine in various ionic and non-ionic contrast media. These data could serve as a sound and useful basis for further microbiological studies, both qualitative and quantitative, on bactericidal and bacteriostatic effects of free iodine in non-ionic contrast media.
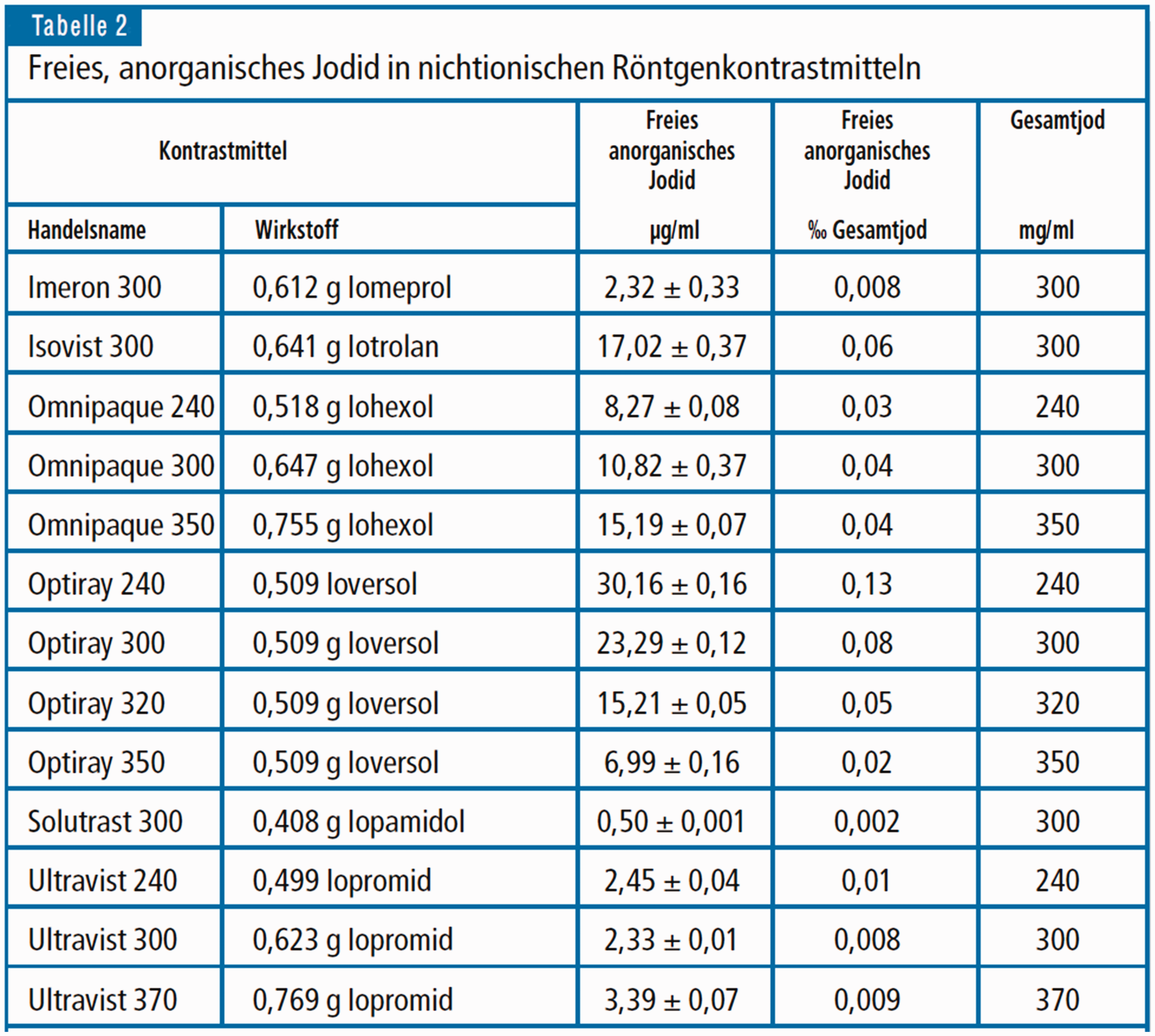
Concentration of unbound, anorganic iodine in non-ionic X-ray contrast media, from Rendl et al. (15).
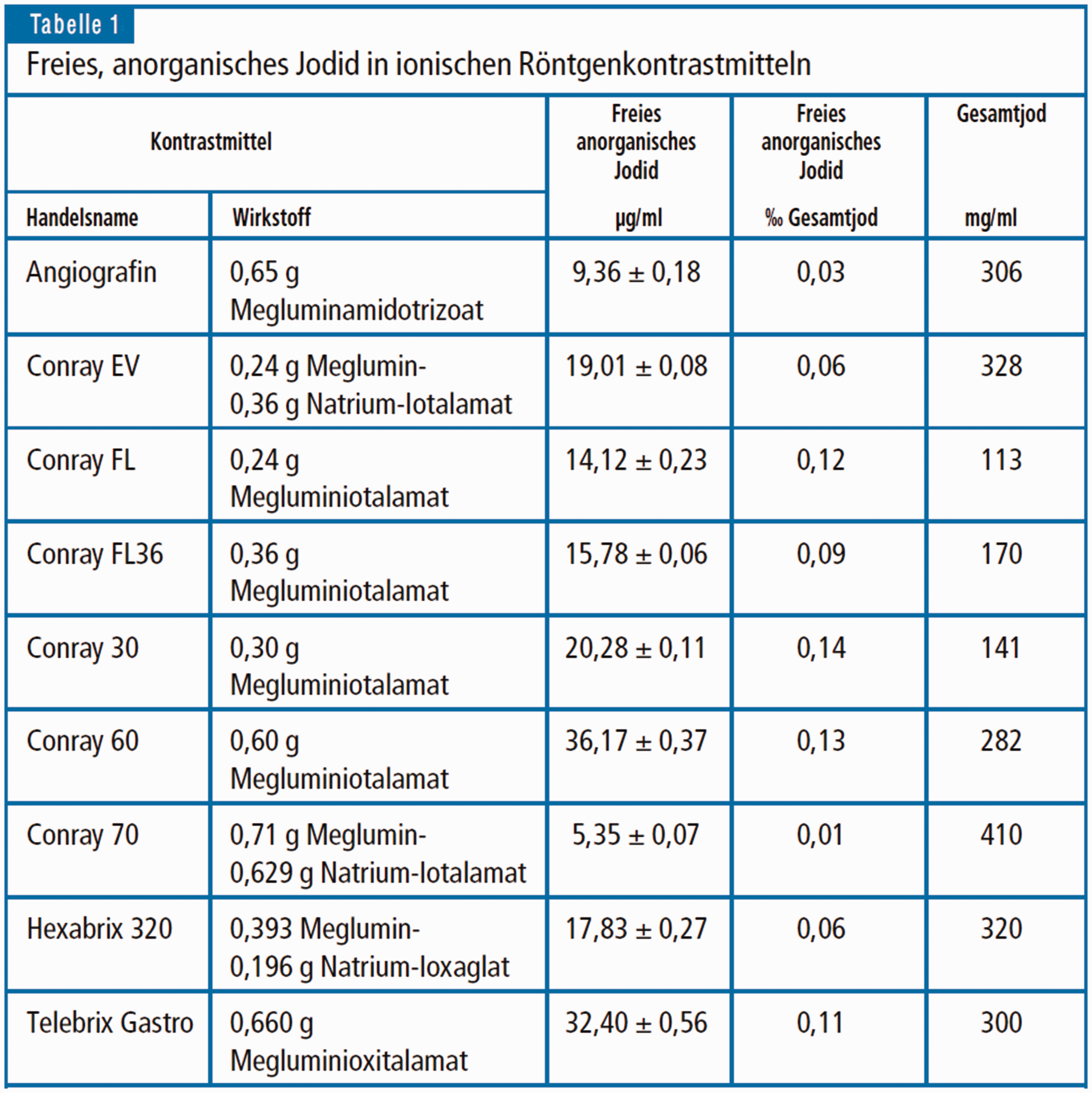
Concentration of unbound, anorganic iodine in ionic X-ray contrast media, from Rendl et al. (15).
Discussion
The clinical application of iodine-containing X-ray contrast media for its antiseptic properties, and not for imaging reasons, has been reported for several decades in the last century, especially in urology. One case of clinical use that stands out is in the presence of bacterially induced cystitis, where the goal was to treat the inflamed bladder mucosa through direct contact via the inserted bladder catheter by irrigation with iodine-containing X-ray contrast media, and not by systemic antibiotics. This local procedure also circumvents the risk of resistance build-up accompanied by systemic antibiotics.
As a parallel to the urologic, locally therapeutic administration of X-ray contrast media, a new in vivo study might be adequate to elucidate if abscess cavity irrigation with iodine-containing, non-ionic contrast medium is beneficial in treating inflammatory processes locally. In this case, a two-step procedure would seem stringent: the postinterventional control of the result of abscess drainage by means of radiological imaging with non-ionic contrast media would be followed by therapeutic irrigation, e.g. flushing of the abscess cavity via the percutaneous catheter with the iodine-containing contrast media.
An additional area of research still remains to be investigated: if and to what extent local proteases induced by microorganisms are able to disintegrate the chelates binding the iodine over a longer period of time. Thus, the concentration of free iodine might be inconsistent over time and consequently the bactericidal and bacteriostatic effects would also be expected to change over time.
Conclusion
While the published knowledge on bactericidal and bacteriostatic properties of non-ionic X-ray contrast media is far outweighed by the literature on these properties of ionic contrast media, it would seem very interesting to do more focused research on the non-ionic group, both in vitro and in vivo.
Footnotes
Authors’ note
We would like to dedicate this work to our friend and colleague, Professor Dr. Iris Baumgartner, Director of the Division of Angiology, Swiss Cardiovascular Centre, Inselspital, Bern University Hospital, Switzerland.
Acknowledgments
The authors thank Professor Dr. med. Iris Baumgartner Director, Division of Angiology, Swiss Cardiovascular Centre, University of Bern, Bern University Hospital, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
