Abstract
Background
The safety of gadolinium-based contrast agents is a hot topic in radiology.
Purpose
To evaluate the safety profile of gadobutrol during routine use.
Material and Methods
Prospective, non-interventional study in 80 centers in Germany. The primary outcome was the number of adverse drug reactions (ADR) following gadobutrol administration.
Results
A total of 3710 patients were included in the analysis, including 404 children (10.9%). A total of 6026 magnetic resonance imaging examinations of organs/organ systems and 872 magnetic resonance angiography examinations were performed. A total of 22 (0.59%) patients experienced at least one ADR. Nausea and vomiting were the most frequent ADRs, experienced by nine (0.24%) and three (0.08%) patients, respectively. One serious ADR was recorded (anaphylactoid reaction). No deaths were reported.
Conclusion
This non-interventional study in 3710 patients confirmed gadobutrol as a safe and reliable contrast agent in adults and children.
Keywords
Introduction
Gadobutrol (Gadovist® 1.0 mmol/mL, Bayer AG, Leverkusen, Germany) is a gadolinium-based contrast agent (GBCA) approved in Europe for magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) of all body regions in adults and children including term newborns (1). Gadobutrol is a macrocyclic, second-generation, extracellular GBCA (2,3) with particular physicochemical properties provided in a unique 1 molar formulation (2,4). This is twice the gadolinium concentration of other marketed GBCAs so that the injection volume is 50% of that of 0.5 molar GBCAs (2). Gadobutrol delivers the highest relaxivity of all macrocyclic GBCAs (4–6).
The macrocyclic structure of gadobutrol offers higher chelate stability and therefore a lower propensity of gadolinium release compared with linear GBCAs (7,8). This feature is clinically relevant in the context of nephrogenic systemic fibrosis (NSF) in patients with severe renal impairment (9,10). As a result, the American College of Radiology (ACR) and the European Medicines Agency (EMA) have categorized gadobutrol in the lowest risk group for development of NSF (11–13).
The recommended standard dose of gadobutrol is 0.1 mmol/kg body weight (bw). Doses up to 0.3 mmol/kg bw are approved in Europe and other regions for specific indications in adults, in particular MRA. So far, gadobutrol’s safety and efficacy have been studied in a number of clinical trials in different indications in patients of a broad age range, including term newborns and elderly (14–19).
The objective of this prospective, non-interventional safety study, PATRON (PATient’s profile in use of Gadobutrol for actual RadiOlogical DiagNostics), was to collect data on adverse drug reactions (ADRs) in the routine setting of gadobutrol-enhanced MRI in German patients.
Material and Methods
Study design
This prospective, non-interventional, multicenter study was performed in patients scheduled for routine contrast-enhanced MRI. Eighty centers in Germany participated, including 65 private practices and 15 radiological departments of hospitals, of which four departments were specialized in pediatric radiology. All centers routinely used gadobutrol which was administered in accordance with the German prescribing information. Ethical approval was obtained for all centers. The study was performed in accordance with the ICH-GCP-Guideline (E6).
Study population
Inpatients and outpatients scheduled for contrast-enhanced MRI with gadobutrol were enrolled between January 2009 and September 2011. Written informed consent (for children by their parents) was obtained after thorough counseling about the imaging procedure and after exclusion of contraindications for contrast-enhanced MRI.
Study procedures
Study start was defined as the time point of gadobutrol administration. After thoroughly informing the patient about the nature of the study, the patient’s demographic data, medical history, concomitant medication, indication for the MRI, dosage, injection parameters, etc. were documented on a standardized case report form (CRF). The observation period encompassed the time until the patient left the institution; however, patients were instructed to report any adverse event (AE) in the following hours and days. All AEs were thoroughly documented in the CRF by the investigators and categorized as either non-serious or serious. The investigator was also asked to assess whether the event was drug-related, the latter, hence classified as an ADR. Any medical intervention and the outcome were also to be recorded.
The sponsor summarized all AEs and ADRs using the MedDRA coding system version 14.0 by system organ classes (SOCs) and preferred terms (PTs). Coding was conducted according to the relevant guidelines or standard operating procedure (SOP), where applicable.
Target variable
The primary outcome was the number of ADRs following gadobutrol administration.
Statistics
The statistical analysis was carried out by the Institute Dr. Schauerte, Oberhaching, Germany. Patient data from the standardized CRF were pooled into an integrated database. Statistical analyses were of an explorative and descriptive nature. Categorical variables (e.g. demographic data, ADRs) were summarized by frequency and proportion.
Results
A total of 3710 patients were included in the analysis, including 404 children (10.9%) and 3306 adults (89.1%). Four patients (0.01%) were aged <2 years, 400 (10.7%) were aged 2–17 years, 2206 (59.5%) were aged 18–64 years, and 1098 (29.6%) were aged ≥65 years. The demographic data are shown in Table 1.
Demographic data of study population (n = 3710).

The majority of imaging procedures were performed to diagnose tumors or inflammatory diseases, 1716 (46.3%) and 852 (23.0%), respectively (Table 2).
Indications for contrast-enhanced MRI.

In total, 6026 MRI examinations of organs or organ systems and 872 MRA examinations were performed in the study cohort. Some patients received MR examinations of more than one body region. The vast majority of MRIs were performed for CNS and cranial imaging, 2452 (40.7%) and 1952 (32.4%), respectively. The vast majority of MRAs were performed for angiography of the head (229 MRAs; 30.8%) and extremities (212 MRAs; 24.3%) (Table 3).
MRI and MRA by number of body regions (n = 3710 patients; MRI n = 3302, MRA n = 708). Some patients received MRI in different body regions.
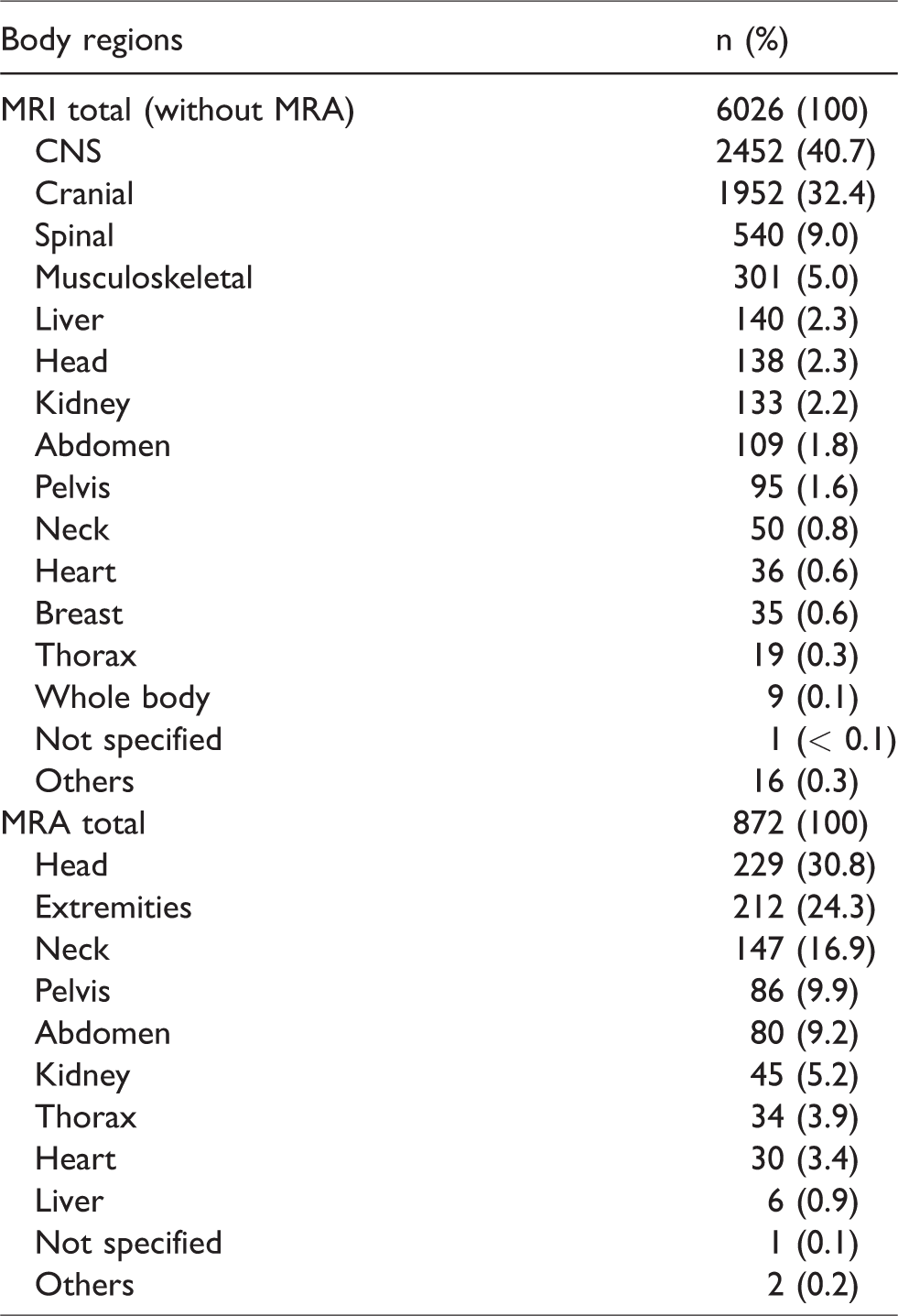
A total of 26 (0.7%) patients experienced at least one AE, of which 22 (0.59%) patients had an ADR. One serious ADR was recorded (0.03%). This was considered a delayed anaphylactoid reaction with erythema and dyspnea in a multimorbid patient, occurring about 10 h after a cranial MRI with 8 mL gadobutrol for tumor staging and exclusion of meningitis or a cerebral abscess. The patient was hospitalized and treated symptomatically and was discharged the next day. Although the patient received several other medications, a causal relationship to gadobutrol administration could not be excluded. There were no deaths (Table 4).
Patients with adverse events (AEs)/adverse drug reactions (ADRs) – overview.
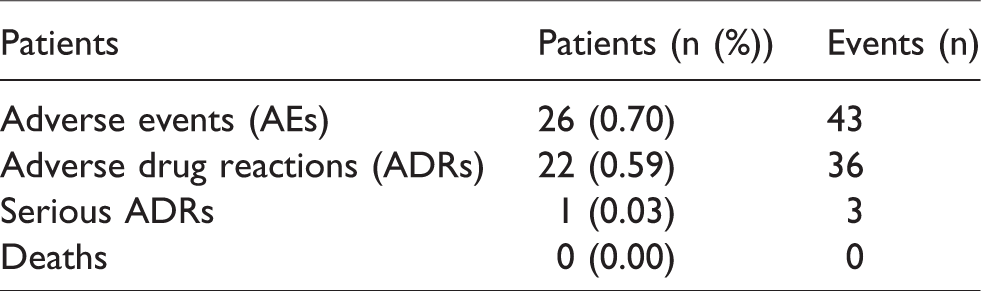
Twenty-two patients experienced a total of 36 ADRs. Thirty-two of 36 ADRs (88.9%) (experienced by 20 patients) occurred within 1 h after gadobutrol administration (Table 5).
Time to ADR onset. 22 patients experienced a total of 36 ADRs.
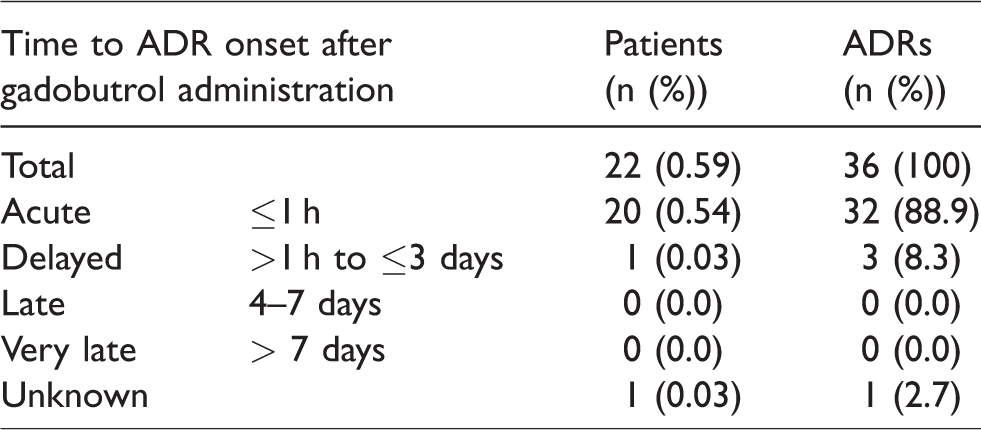
Reactions of the gastrointestinal system, i.e. nausea and vomiting, were the most frequent ADRs, experienced by nine (0.24%) and three (0.08%) patients, respectively. ADRs of the nervous system, i.e. dizziness, dysgeusia, headache, and hypoesthesia, were second with a total of five patients (0.14%). While no ADRs were seen in children aged <7 years, there was one in a 14-year-old patient who underwent a cranial MRI for a suspected brain tumor. The patient suffered from a burning sensation along the forearm immediately after injection and a headache for 5 min. Complete recovery without therapy was reported. All ADRs were transitory; in five patients, symptomatic therapy was necessary (Table 6).
ADRs by organ system (n = 22/3710 (0.59%) patients suffered at least one ADR).
*Multiple responses possible.
The contrast quality of gadobutrol was classified as very good or good in 3683 patients (99.3%).
Discussion
This prospective, non-interventional study in 3710 patients in Germany evaluated the safety profile and contrast quality of gadobutrol in routine clinical use.
A review on general safety of gadobutrol has been published recently. This publication analyzed 6809 patients from 42 clinical Phase II–IV studies and data from pharmacovigilance including 29 million applications (20). In the clinical Phase II–IV studies 2.6–4.9% of patients experienced ADRs with gadobutrol and similar figures were recorded for the comparator GBCAs, i.e. 1.7–4.9% of ADRs. The ADR reporting rate in pharmacovigilance was remarkably lower at 0.05% (20). The ADR rate of the study presented here was 0.59%. The discrepancies between clinical development studies, Phase IV studies and analyses of pharmacovigilance data can be explained by the different study designs and the degree of scrutiny of data collection and reporting behavior (21).
A study very similar in design published by Forsting et al. summarized six prospective observational studies in 14,299 patients in 300 institutions in Europe and Canada. A total of 78 patients (0.55%) reported at least one ADR, with nausea and vomiting as the most frequent events (16). Moreover, Prince et al. analyzed 23,708 patients and found 0.7% of patients with ADRs (19), a result that is also confirmed by our data. A Japanese study in 3337 patients will be published soon, showing also ADR rates in the same range (accepted by Japanese Journal of Radiology Sept 19, 2018).
There were no ADRs in children aged < 7 years and one ADR in one child aged 8–18 years indicating a similar safety profile in all age groups. This is in line with other studies looking at the safety of gadobutrol in children (14,17,22).
Comparing the safety profile of different GBCAs is a challenge as head-to-head prospective safety studies have not been published. With all caveats, one could put clinical phase IV studies with comparable study designs side by side. Doing so, the overall ADR rates reported for gadopentetate (23), gadoterate (24), and gadobenate (25) were 2.4%, 0.4%, and 0.76%, respectively. Once again, nausea and vomiting were always the most frequently reported ADRs. In summary, the safety profile and tolerability of the investigated GBCAs can be regarded as similar.
One serious ADR was reported in our study: an anaphylactoid reaction with erythema and dyspnea in a multi-morbid patient. Fortunately, the patient recovered within one day after symptomatic therapy. In general, anaphylactoid—or hypersensitivity reactions—encompass a broad range of symptoms, e.g. angioedema, shock, hypotension, erythema, rash, pruritus, edema, sneezing, and urticaria (26). Anayphylactoid reactions are mentioned in the prescribing information of all GBCAs, however with a low incidence rate. A reporting rate of 0.019% has been calculated from the gadobutrol post-marketing surveillance database (20).
In 2014, Kanda et al. reported increased signal intensity (SI) in the dentate nucleus and the globus pallidus of the brain on unenhanced T1-weighted MR images after multiple administrations of linear GBCAs (27–31). This phenomenon is most probably caused by gadolinium released from its chelate and binding to certain macromolecules in the brain and is robustly associated with linear GBCAs but not with gadobutrol or other macrocyclic GBCAs (32–39). However, minute amounts of gadolinium have been detected in brain and other body tissues post mortem after both linear and macrocyclic GBCA administration (40,41). As of today, no signs or symptoms of adverse health effects and no histopathological changes associated with hyperintensity and the presence of Gd in the brain have been observed.
Two limitations need to be addressed. First, this non-interventional study did not include a comparator group (neither active comparator nor a group with un-enhanced MRI). Second, the study was conducted in only one country. However, both limitations might be compensated by the plethora of Phase II–III data and pharmacovigilance results from 29 million applications worldwide (20).
Since the first market introduction of a GBCA (gadopentetate dimeglumine) in 1988, > 450 million GBCA doses have been administered globally. Compared to iodine-based X-ray contrast agents GBCAs cause fewer systemic effects, fewer allergic reactions and fewer renal problems (42). Gadobutrol was first introduced in 1998 and had been administered >50.6 million times by April 2018.
In conclusion, this study confirmed gadobutrol to be a safe and reliable contrast agent for adults and children.
Footnotes
Acknowledgements
The authors thank all the investigators and patients who participated in the study program.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PK and JE are employees of Bayer Vital GmbH and Bayer AG, respectively.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The investigators were reimbursed for running the study but did not get any financial support for authorship.
