Abstract
Background
Safety data on routine clinical use of gadoxetate disodium for liver magnetic resonance imaging (MRI) is not reported yet.
Purpose
To assess the safety profile of gadoxetate disodium for liver MRI in the routine clinical setting.
Material and Methods
Six multicenter studies were performed in Europe, USA, Australia, and Asia to evaluate the safety and efficacy of gadoxetate disodium (Primovist®/Eovist®) enhanced liver MRI. Patients received a single intravenous bolus injection of the standard approved dose of 0.025 mmol/kg body weight (0.1 mL/kg). The number of patients, the characteristics of adverse events, related adverse events, and serious adverse events were analyzed.
Results
A total of 8194 patients were included in the database. A total of 141 patients (1.7%) reported 230 AEs of which 129 were considered being related to the use of gadoxetate disodium by the investigators. None of the AEs in the pediatric population (n = 52) were related. The most frequent AEs independent of relationship to the drug included dyspnea (25/0.31%), nausea (22/0.27%), liver disorders (13/0.16%), and renal disorders (9/0.11%). Nine related SAEs were recorded. No patient died during the studies.
Conclusion
Gadoxetate disodium for liver MRI is safe and well tolerated in the routine clinical setting.
Introduction
Gadoxetate disodium (Primovist®/Eovist®) is a magnetic resonance (MR) contrast agent specifically developed for detection, localization, and characterization of liver lesions. Previous work demonstrated that gadoxetate disodium-enhanced liver MR imaging (MRI) provides better diagnostic performance than computed tomography (CT) or conventional extracellular contrast-enhanced MRI (1,2). In particular, the highest rate of correctly detected lesions is for lesions with a diameter <1 cm (3). Therefore, gadoxetate disodium is widely used as an MR contrast agent for the evaluation of liver lesions.
Gadoxetate disodium belongs to the class of linear ionic gadolinium-based contrast agents (GBCAs) and features a high T1 relaxivity of 6.9 L mmol−1 s−1 at 1.5 T (in plasma) (4,5). After intravenous application, gadoxetate disodium is distributed in the extracellular space and quickly and selectively taken up by the hepatocytes, thus enabling both dynamic and hepatocyte-specific imaging. In healthy subjects about 50% is excreted via the kidneys and 50% via the biliary system (6,7). Contrast enhancement of the liver parenchyma and liver to lesion contrast is highest at about 20 min after administration with a plateau lasting for at least 45 min post injection (8,9). The strong enhancement of hepatic parenchyma in hepatobiliary phase images provides better lesion conspicuity, which is one of the advantages of this contrast agent.
A favorable safety profile of gadoxetate disodium (3,10,11) has been established in 12 clinical phase II–III studies and confirmed in post-marketing surveillance (12). So far, since approval in March 2004 through March 2014, more than 2.2 million patients have been exposed to gadoxetate disodium worldwide. The purpose of this analysis was to systematically evaluate the safety profile of gadoxetate disodium in the routine clinical setting.
Material and Methods
Studies
Essentials of studies included in the analysis.
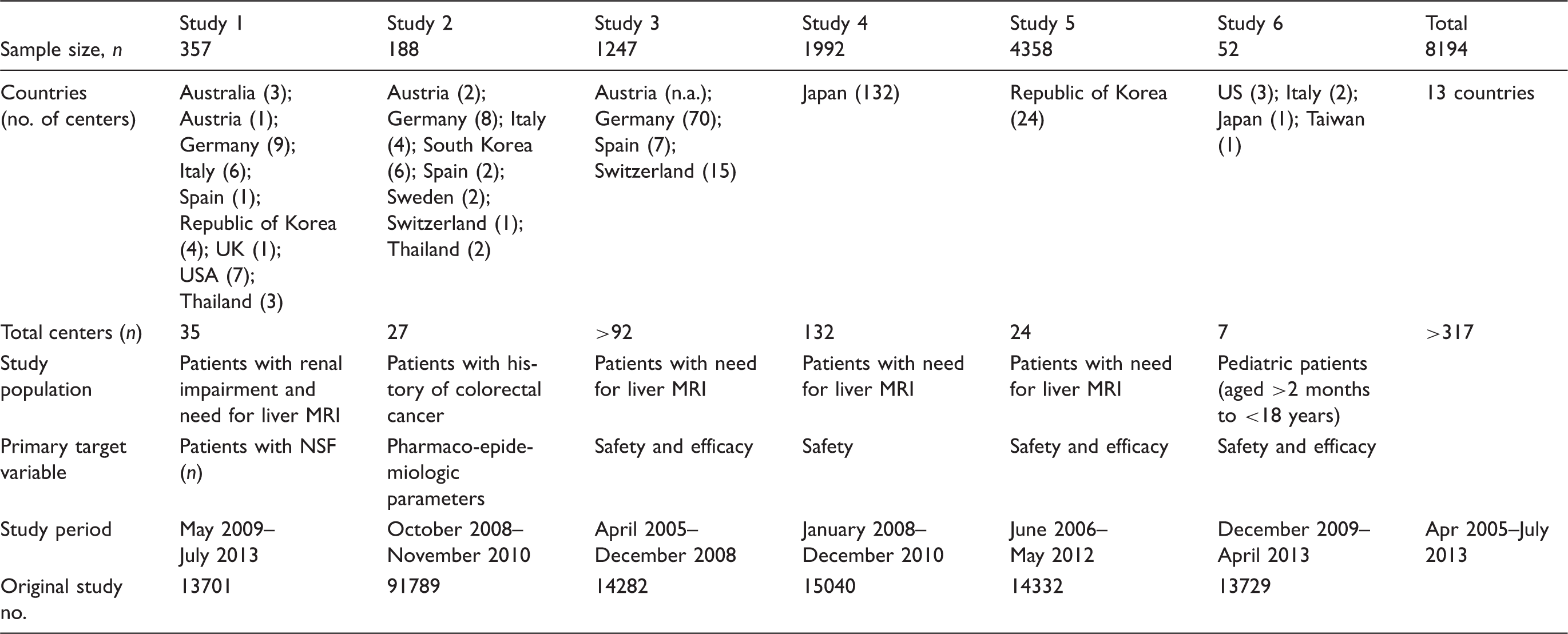
Three studies were designed to investigate the safety and efficacy of gadoxetate disodium (Studies 3, 5, and 6) and one focused on safety aspects only (Study 4). Study 1 specifically assessed the risk of nephrogenic systemic fibrosis (NSF), while Study 2 focused on pharmaco-epidemiologic parameters after liver imaging of patients with colorectal cancer (Table 1). All studies were conducted in accordance with all guidelines set forth by the approving institutional review board.
Study population
The study population consisted of patients scheduled for gadoxetate disodium-enhanced liver MRI within the approved indications and dose. One study specifically included patients with renal impairment (Study 1), and another study (Study 2) included patients with colorectal cancer. Study 6 focused on pediatric patients aged (>2 months and <18 years) (Table 1). Informed consent (IC) was obtained prior to study start, except for Japan where IC is not necessary for non-interventional studies.
Treatment
All patients were to receive a single intravenous bolus injection of 0.025 mmol/kg (0.1 mL/kg) body weight gadoxetate disodium (Primovist®/Eovist®, Bayer HealthCare AG, Leverkusen, Germany) at the recommended flow rate of about 2 mL/s followed by a saline chaser. Gadoxetate disodium is approved and marketed in all participating countries for adults. It was purchased locally by the centers at hospital pharmacies.
Target variables
The key target variables of this analysis were the number of patients with and the characteristics of adverse events (AEs), related adverse events, and serious adverse events (SAEs).
An AE was defined as any illness, sign or symptom, or unfavorable change in the clinical status that had appeared or worsened after study start, whether or not it was considered to be related to gadoxetate disodium administration. All AEs were evaluated for seriousness and potential relationship to gadoxetate disodium administration by experienced healthcare professionals in each institution. Related AEs comprised the categories “possibly”, “probably”, and “definitely” related to gadoxetate disodium administration.
An SAE was defined as any adverse event that: (i) resulted in death; (ii) was life-threatening; (iii) required subject hospitalization or prolongation of existing hospitalization; (iv) resulted in a persistent or significant disability/incapacity; (v) resulted in a congenital anomaly/birth defect; or (vi) was considered an otherwise medically significant event.
Study procedures
The observational study approach did not interfere with the routine clinical practice in the participating centers of all six studies. Demographic data, medical history – in particular history of renal diseases and allergies – and contrast media applications were recorded. Once gadoxetate disodium enhanced liver MRI was performed, patients were asked about their well-being in order to gather information about AEs. The follow-up period lasted from just the examination day (Study 5) up to 24 months (Study 1). These were rated by the treating physician as “definitively”, “probably”, “possibly”, or “not related” to the study.
Statistical and sample size
All patients who received gadoxetate disodium were included in the safety analysis. In Study 5 more than 3000 cases had to be collected based on Article 6, Paragraph 3, Subparagraph 1 of “Standards for new drug surveillance (Korea Food and Drug Administration Notification No. 2008-38, 2008.06.27)”. In the other studies the sample size was chosen according to feasibility criteria.
Descriptive statistics were applied. Qualitative variables were reported as frequency count and percentage. AE frequency percentages were calculated by dividing the number of patients reporting one specific AE or AE grouping by the number of patients exposed to gadoxetate disodium, times 100. In addition, AEs were analyzed by system organ classes (SOCs).
All analyses were performed for each study separately as predefined in the study protocols and overall evaluations were done post hoc. Statistical analyses were performed using the software package SAS release 9.2 (SAS Institute, Cary, NC, USA).
Results
A total of 8194 patients were included in the database. Three studies included more than 1000 patients: Study 5 from Korea recruited 4358, Study 4 from Japan 1992 patients, and Study 3 from Europe 1247 patients (Table 1).
Subject demographics at baseline (n = 8194).
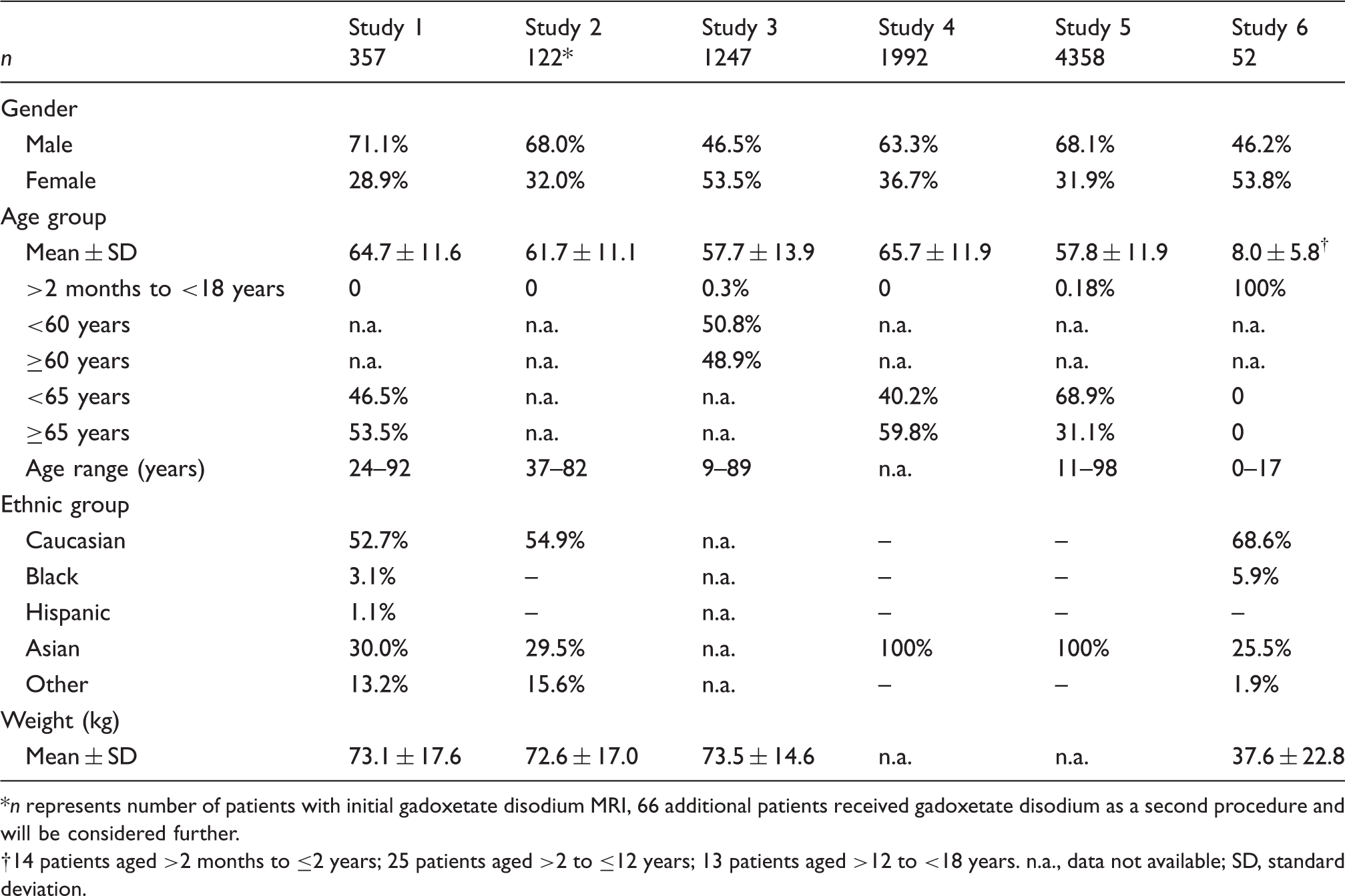
n represents number of patients with initial gadoxetate disodium MRI, 66 additional patients received gadoxetate disodium as a second procedure and will be considered further.
14 patients aged >2 months to ≤2 years; 25 patients aged >2 to ≤12 years; 13 patients aged >12 to <18 years. n.a., data not available; SD, standard deviation.
Number of patients with overall and related adverse events (AEs).
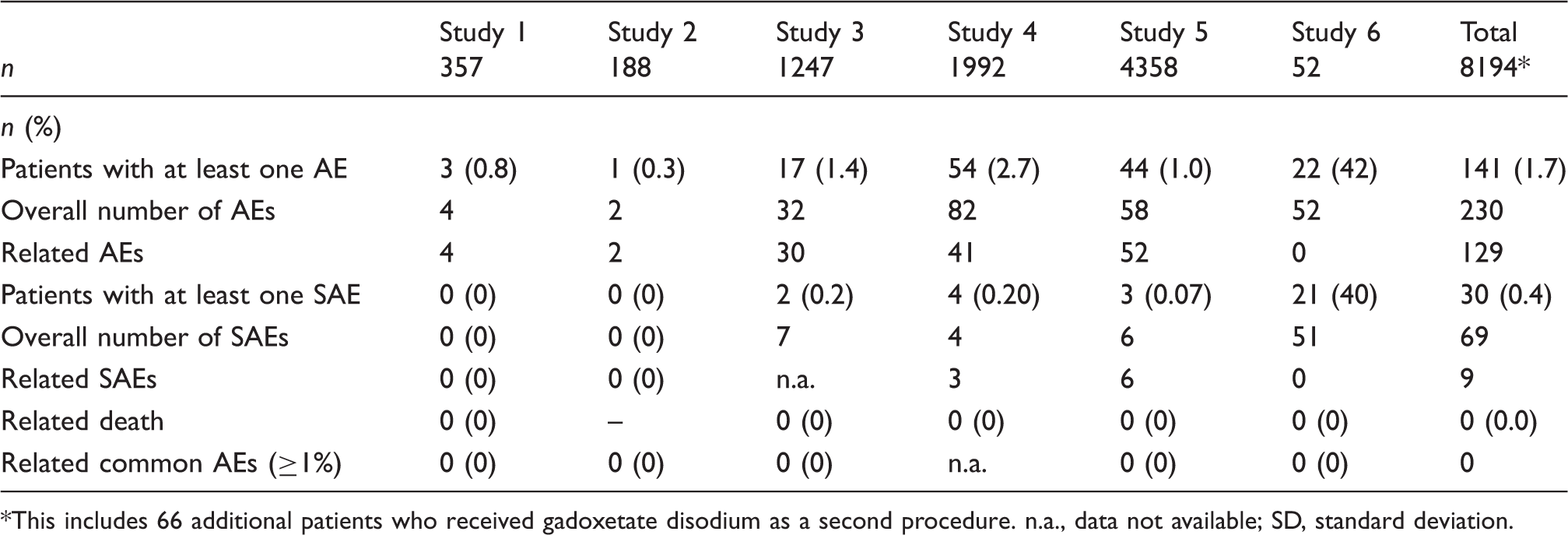
This includes 66 additional patients who received gadoxetate disodium as a second procedure. n.a., data not available; SD, standard deviation.
Details on related AEs and related SAEs.
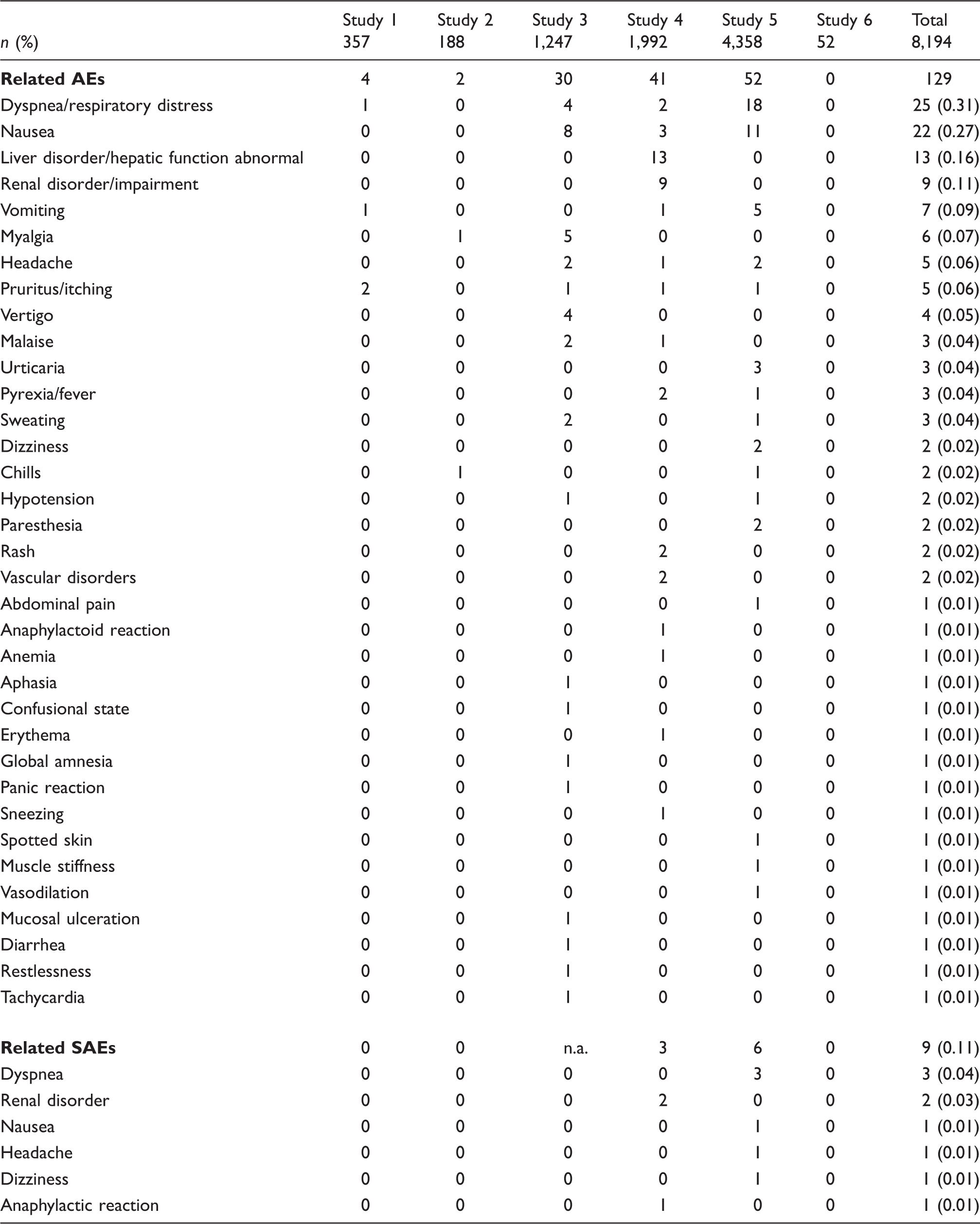
Number of patients with related and/or unrelated AEs by system organ class (SOC).
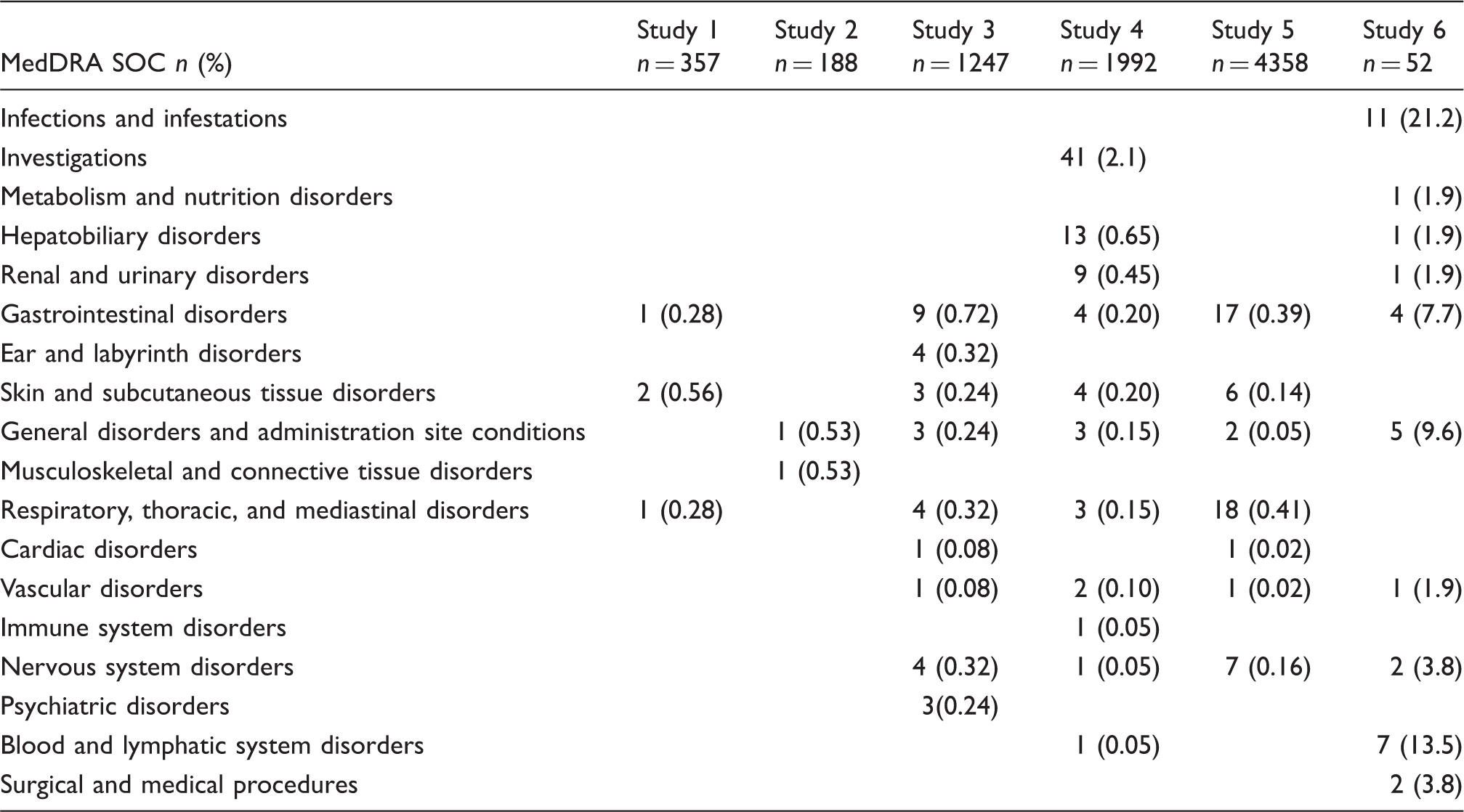
Discussion
This publication is a systematic analysis of safety data on gadoxetate disodium reported in six multicenter, prospective studies performed in Europe, USA, Australia, and Asia. It complements the concise analysis of phases II and III clinical development studies and post-marketing data published recently (12). The rate and quality of AEs and related AEs were consistent with those of other GBCAs (13–16). The findings did not give rise to any specific safety concerns regarding gadoxetate disodium.
The fact that patients were recruited on four continents (Asia, America, Australia and Europe) in 13 countries and at more than 317 centers is one particular feature of this analysis that differentiates it from others. The results of all six studies were similar, thus confirming the good safety profile of gadoxetate disodium in different ethnic groups. Furthermore, the safety profile was consistent within the broad age range (2 months to 98 years). The majority of patients irrespective of age received a dose of 0.025 mmol/kg body weight gadoxetate disodium. So far, no further data on newborns/infants have been published.
When comparing AE rates of liver-specific gadoxetic acid with other, non-liver-specific GBCAs, it is important to keep in mind that patients for liver imaging form a specific subgroup of patients because other GBCAs (e.g. Gd-DTPA) are used not only for liver imaging but for a wide spectrum of body regions. In addition, a reasonable comparison should be done preferably with other observational studies, as results from the tightly controlled phase II–III studies or from pharmacovigilance databases may yield higher or lower AE rates, respectively, due to the completely different study designs and ways of data capturing.
Forsting et al. analyzed the safety profile of gadobutrol in six observational studies including 14,299 patients. Seventy-eight patients (0.55%) reported at least one related AE (17). Similarly, Herborn et al. assessed the safety profile of gadobenate dimeglumine in 38,568 patients in 662 centers in Germany. They reported an AE rate of 1.2% (18). Bleicher et al. also looked at 23,553 patients after gadobenate dimeglumine and recorded related AEs in 0.76% (13). Also for gadoterate meglumine an observational study is available. In a total of 24,308 patients in 61 German radiologic institutions a total AE rate of 0.4% was recorded (14). All these results are well in line with the ones reported here for gadoxetic acid.
In contrast to the adult population, 22 AEs in 52 patients were recorded in the pediatric population (aged >2 months to 17 years). However, none of the AEs was categorized as drug-related. Comparative data for other GBCAs in children is rare. In a phase I–III study on gadobutrol in 2–17-year-old patients a rate for drug-related AEs of 5.8% was reported (19) while in an observational study in infants aged under 2 years, no patients experienced AEs related to gadobutrol (20).
Dyspnea, nausea, liver, and renal disorders were the most frequent related AEs. The terms “dyspnea”, “respiratory distress”, and “respiration abnormal” are MedDRA preferred terms (PTs) encompassing a broad range of respiratory symptoms from simple breath-holding difficulties to the feeling of suffocation. In this evaluation, only 3/24 cases of such respiratory events were classified as SAEs, indicating that just a fraction of events was considered clinically relevant by the medical staff. However, Davenport et al. described a phenomenon called “acute transient self-limiting dyspnea” in patients receiving gadoxetate disodium or gadobenate dimeglumine and the subsequent effects on image quality (21,22). Interestingly, also in their publication it remains unclear if “dyspnea” during breath-hold represents a sensation of breathlessness or the inability to hold one’s breath, which is common in severely ill patients with liver issues (23). Some patients had ascites that also causes problems with breath-holding. In 12 controlled phase II–III clinical trials gadoxetate disodium has been administered to 1989 patients and a dyspnea frequency was reported as low as 0.2% (12). Also in the post-marketing surveillance database for gadoxetate disodium more than 2.2 million administrations were recorded and the reporting rate for dyspnea was 0.004% (12). Gadobenate dimeglumine, also used for liver MRI, showed an AE rate for dyspnea of 0.05% in 38,568 patients (18). An overall comparison to other GBCAs is limited, as the population scheduled for liver imaging is a specific subset of patients scheduled for contrast-enhanced MR exams. Nausea was reported by 22 patients (0.27%). Nausea is also reported for all other GBCAs, e.g. with gadobutrol (0.25%) (17), with gadobenate dimeglumine (0.6%) (18), and gadoterate meglumine (0.17%) (14).
The major limitation of this evaluation is that it is a compilation of phase IV studies. Such studies have the advantage of reporting data on routine clinical use, but they lack the meticulousness of data capturing applied in phase I–III. Thus, reports may sometimes be missing critical data elements necessary for comprehensive evaluation, such as complete medical history, co-morbidities, or co-medications. On the other hand, phase IV studies reflect the real-life situation in day-to-day medical practice.
In conclusion, gadoxetate disodium for liver MRI is safe and well tolerated in the routine clinical setting.
Footnotes
Acknowledgements
We thank all investigators and patients who participated in the study program.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: With the exception of SYK, authors are employees of Bayer Healthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All studies were funded by Bayer.
