Abstract
Background
Iosimenol 340 injection is a new, dimeric, iso-osmolar, iodinated contrast medium for X-ray angiography.
Purpose
To compare the safety and efficacy of iosimenol injection to iodixanol injection in two randomized, controlled phase 2 trials.
Material and Methods
One hundred and forty-four adult patients were enrolled in the two trials, one for evaluation during arteriography and the other for evaluation during computed tomography. Safety was compared by assessing adverse events, vital signs, ECGs, and laboratory parameters. Efficacy was assessed as X-ray attenuation in the computed tomography (CT) trial and as the quality of contrast enhancement in the arteriography trial.
Results
There were no statistically significant differences in terms of safety or efficacy between the two contrast media. Both were well tolerated upon intravenous as well as intra-arterial injection. The most common adverse event was a feeling of warmth (observed in 35.1% of the patients with Iosimenol injection and 44.3% with iodixanol injection).
Conclusion
Iosimenol upon intravenous as well as upon intra-arterial injection exhibits a safety profile and shows an efficacy similar to that of iodixanol.
Introduction
More than 80 million doses of iodinated contrast media (CM) are administered annually (1). All approved radiographic CM are derivatives of tri-iodo-benzoic acid with X-ray attenuating properties proportional to the iodine concentration of the final, formulated product. Incremental improvements in safety and tolerance after vascular administration have been achieved by reducing the number of moles of solute per kg water in the final solution, thereby reducing the osmolality of the contrast medium. The number of adverse drug reactions was thus reduced by approximately 75% when outcomes after injection of ionic, monomeric CM are compared to that after injection of non-ionic, monomeric CM (2). However, only dimerization allows synthesis of CM which can be formulated as iso-osmolar to plasma (approximately 290 mOsm/kg) while providing a sufficient concentration of iodine for all clinical applications (3). The only commercially available non-ionic, dimeric, iso-osmolar CM (iodixanol, Visipaque™), has been shown to reduce injection associated discomfort (pain, heat) compared to non-ionic, monomeric CM (4). It has been claimed that the frequency of CM-induced nephropathy (CIN) is lower (5), however this remains a controversial topic, since other authors could not confirm that (6). Some data even suggest improved in-hospital outcomes and a reduction in major angiographic complications after coronary angiography (7). A superior safety profile was also reported in comparison to another dimeric (but ionic) compound (ioxaglate, Hexabrix™) (8). However, the low osmolality of iodixanol came at the expense of increased viscosity, rendering injection through very thin intra-arterial catheters harder (9). In addition, the high viscosity per se may pose a renal insult, as suggested by non-clinical data (10,11).
Iosimenol is a novel dimeric, non-ionic, iso-osmolar CM with lower molecular weight and viscosity than iodixanol at equal iodine concentration. Data from the preclinical evaluation of iosimenol have been promising (12,13). The electro-physiologic cardiac response in humans was excellent (i.e. minimal) (14) and pharmacokinetic and initial safety data collected during Phase 1 was similar to that of other contrast media (15). In order to further evaluate the safety and efficacy, two comparative clinical trials, one after intravenous injection (computed tomography [CT]) and one after intra-arterial administration (digital subtraction angiography [DSA]), were conducted.
Material and Methods
The safety, tolerance and efficacy of iosimenol injection (provided by Koehler Chemie, Bensheim, Germany; manufactured by Interpharma Praha a.s., Prague, Czech Republic]) were compared to that of iodixanol injection (Visipaque™, GE Healthcare, Chalfont St Giles, Buckinghamshire, UK) in two randomized and double-blind, bi-centric phase 2 trials conducted in Germany, between May 2003 and July 2005. The trials were approved by the local Ethics Committees and conducted in accordance with Good Clinical Practice, the German Drug Law and the 1964 Declaration of Helsinki with subsequent updates. Written informed consent was obtained from all patients. Only adult patients (≥18 years of age) were enrolled. Patients with renal dysfunction (defined in the intravenous trial as serum creatinine (SCr) >1.5 mg/100 mL and in the intra-arterial trial as >2.0 mg/100 mL) were excluded as were patients with severe hepatic dysfunction or a history of any allergic reactions to CM in both trials.
For the CT trial, 80 adult patients scheduled for CT were enrolled and randomized to receive either iosimenol 340 mg iodine/mL (42 patients) or iodixanol 320 mg iodine/mL (38 patients). A total of 100 mL of CM was applied by means of an automatic injector at a mean rate of 2.3 mL/s (±0.31).
For the angiography trial, 64 adult patients scheduled for aortography and lower limb arteriography were enrolled and randomly assigned to one of four treatment groups (iosimenol 270 mg iodine/mL; iosimenol 340 mg iodine/mL; iodixanol 270 mg iodine/mL; or iodixanol 320 mg iodine/mL). Patients were given as many intra-arterial injections as considered necessary for diagnostic purposes. Ranges of 15–60 mL of CM were administered for aortography and 30–35 mL for lower limb angiography, up to a total of 300 mL.
Safety assessments
Safety was assessed by physical examination, electrocardiogram (ECG) recordings, vital signs (blood pressure and pulse rate), and clinical laboratory parameters for hematology and clinical chemistry. Safety evaluations were performed at screening, baseline, and at regular intervals until day 4 (72 h). A follow-up visit was performed between days 6 and 8. Change in serum creatinine (SCr) after injection of CM was used as an indicator of CIN, defined as either a relative increase in SCr of >25% or an absolute increase of ≥44.2 µmol/L (6). Blood samples were collected at 24 and 72 h. The original ECGs were re-evaluated independently after the end of the trial.
Adverse events (AEs) were assessed at each of the time points for blood sampling and also by phone after 6–8 days. Patients reporting possible AEs during the phone call were further evaluated during a subsequent follow-up visit. AEs occurring 2 h or later after CM administration were classified as having a late onset. The severity of AEs was evaluated by the investigators as mild (did not interfere with routine activities), moderate (interfered with routine activities), or severe (prohibited routine activities). The causality of AEs was classified as not related, unlikely, possibly, probably, or highly probably related. A simplified binary outcome of “not related” or “related” to the CM was used for the sake of this publication, with not/unlikely related AEs included in the first category and the remaining AEs accounted for in the second. AEs were classified as serious when causing death, being life-threatening, causing hospitalization, significant, persistent, or permanent disability or requiring medical interventions to prevent permanent impairment.
Efficacy assessments
In the CT trial X-ray attenuation (Hounsfield units) in the aorta was measured 30–60 s after the start of the injections. Measurements (TomoScan AV, Philips Medical Systems Nederland B.V., Best, The Netherlands and GE Hi Speed, GE Healthcare, Chalfont St Giles, UK) were performed within regions of interest (1.0–1.5 cm in diameter) in the aortic arch, the descending thoracic, and/or suprarenal abdominal aorta.
In the angiography trial three readers, blinded for clinical and demographic data as well as trial drug, independently assessed the image sets by region (abdomen, pelvis, thighs, lower legs, and ankles/feet as available). The quality of contrast enhancement (QCE) for each region was assessed by a four-point rating scale. Ratings of “excellent” and “good” were considered to provide diagnostic quality contrast enhancement, while ratings of “poor” or “none” were classified as providing non-diagnostic quality contrast enhancement. The main efficacy endpoint was the overall QCE score for each patient.
Each reader determined first whether the images were technically adequate (evaluable) and only if they were, continued the evaluation. Technically inadequate was defined as improper positioning, artifacts and patient movement which was the case in 2% of the image sets.
Statistical analysis
Continuous safety variables were summarized with descriptive statistics (N, mean, standard deviation, median, minimum, and maximum), categorical variables by frequencies and percentages. 95% two-sided exact confidence intervals were constructed according to Clopper-Pearson. P values were calculated using Fisher’s exact test. A P value of <0.05 was considered statistically significant. All data processing and analyses were performed with SAS® Version 9.2 or higher (SAS Institute, Heidelberg, Germany).
Results
Subjects
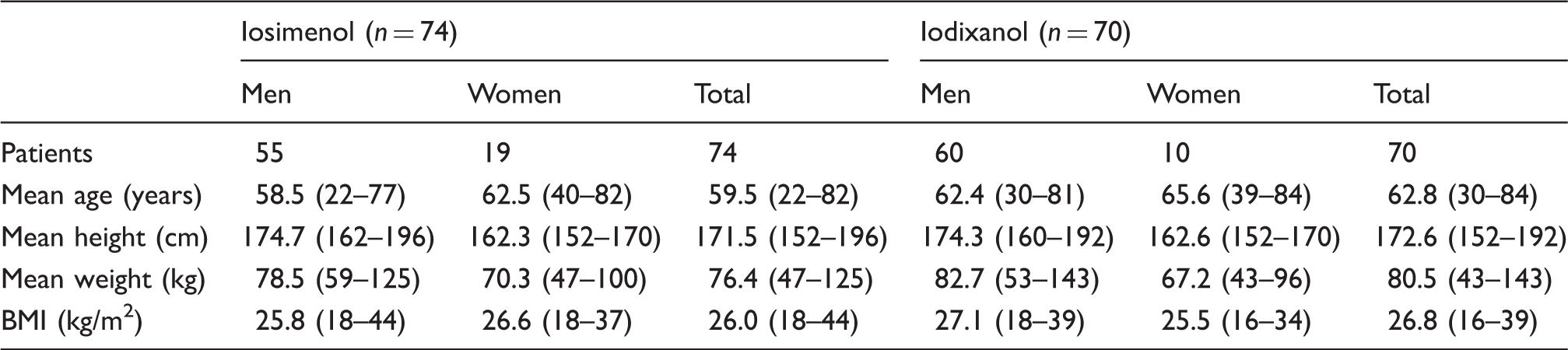
Patient demographics (mean values and ranges).
Safety
Summary of adverse events (AEs).
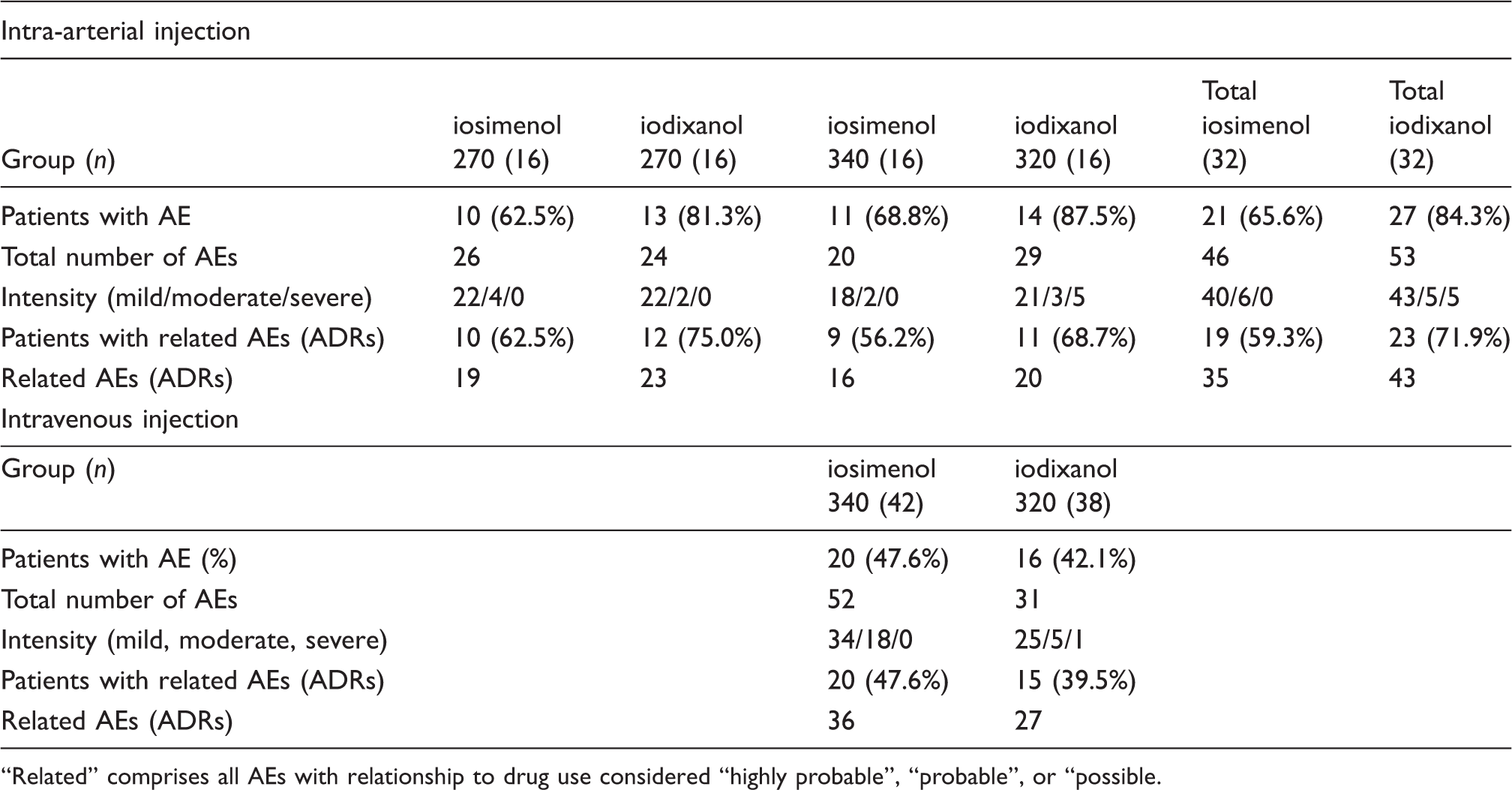
Related” comprises all AEs with relationship to drug use considered “highly probable”, “probable”, or “possible.
Individual changes in vital signs were small and within expected physiologic variability. None was considered as clinically significant.
No patient had an increase in heart rate exceeding 8 bpm and all recovered within a few minutes. Seven patients had a change in QTcF in the range of 30–60 ms after injection of both CM. One patient had a change >60 ms in the Iosimenol 270 group. There were few ECG changes after contrast administration and all occurred after intra-arterial injection, two in each group. Overall there were no differences with regard to effects on vital signs and ECG between the two CM.
Incidence of contrast-induced nephropathy.
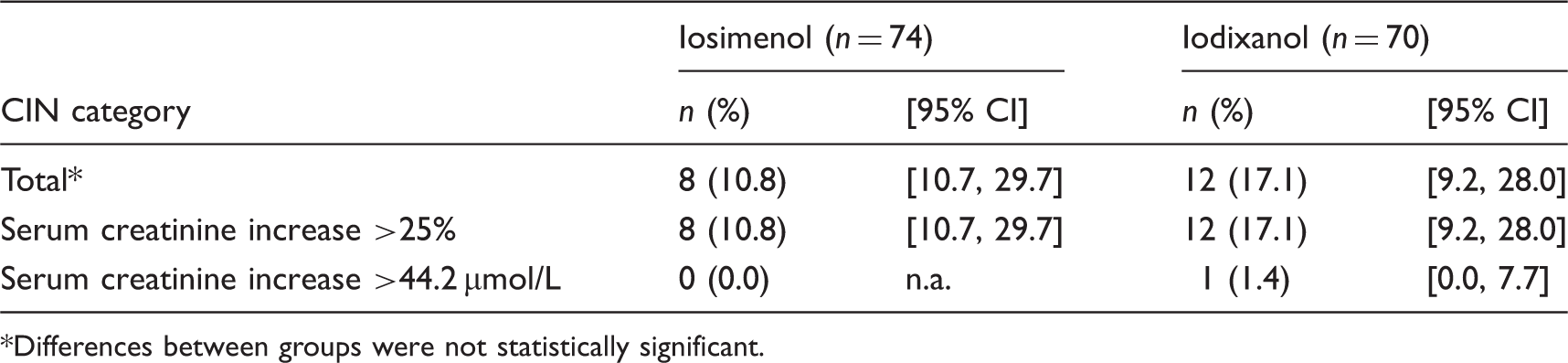
Differences between groups were not statistically significant.
Efficacy
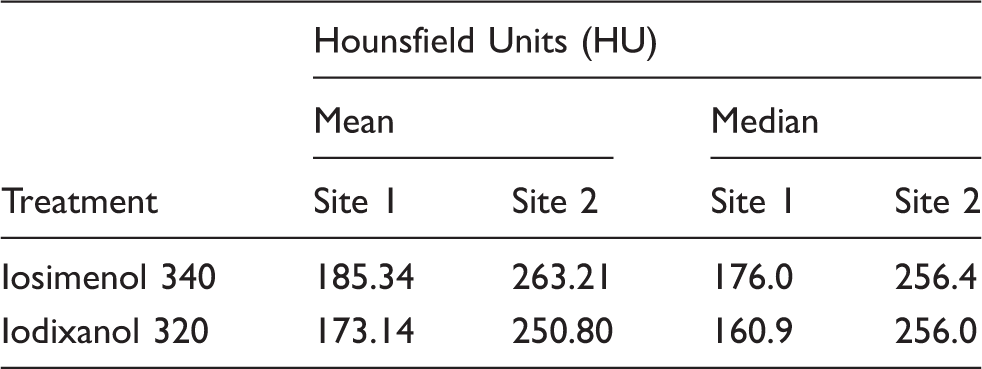
Contrast enhancement in Hounsfield Units/CT trial.
Overall image evaluation by reader/Arteriography trial.
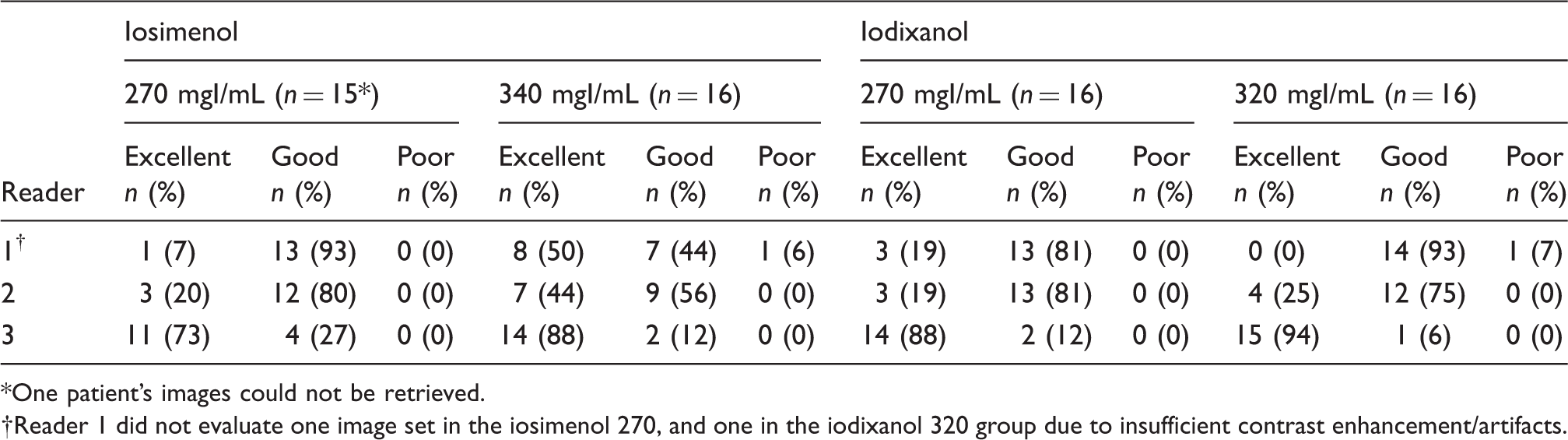
One patient’s images could not be retrieved.
Reader 1 did not evaluate one image set in the iosimenol 270, and one in the iodixanol 320 group due to insufficient contrast enhancement/artifacts.
Discussion
Most AEs after the injection of iodinated CM are known to be transient, self-limiting, and mild in intensity (16). Typical manifestations include a sensation of heat, nausea, occasional vomiting, skin rash, and hemodynamic changes of minor clinical importance (2,4). The frequency, type and severity of AEs in the present trials were not different. Moreover, no significant difference between the iodixanol and iosimenol groups was found. A feeling of warmth was by far the most frequent AE occurring in the present study, encountered in 43.7% and 59.4% of the patients after intra-arterial administration and 26.2% and 31.6% after intravenous administration of iosimenol and iodixanol, respectively. In a recent meta-analysis of 15 trials (n = 5899) (4) more than 90% of the patients described a sensation of heat after intra-arterial injection, irrespective of which CM was used. In this meta-analysis the effects of iodixanol, the only iso-osmolar CM in the analysis, compared favorably towards pooled comparators with higher osmolality with regard to incidence and severity of the sensation of heat.
Late onset of AEs is defined as an onset between 1 h and 1 week after CM administration (17); most of these AEs occur between 3 h and 2 days. (18). Therefore the definition used in the present study (2 h to 1 week) is considered adequate. Typically, the type of events does not vary much with the time of occurrence. Late onset AEs include nausea, vomiting, headache, itching, skin rash, musculoskeletal pain, and fever (17,19,20). While some authors report a higher incidence after injection of iso-osmolar dimeric CM compared to injection of monomeric CM with higher osmolality (21–23), others claim that no such difference exists (24,25). These seemingly divergent conclusions largely originate from design issues of the trials the conclusions are based on. The late AEs in our studies were in most cases of mild intensity; about half of them were skin reactions with a few more occurring in the iosimenol group.
CIN, defined as a condition of decreased renal function within 3 days of intravascular administration of a CM, in the absence of an alternative etiology, remains a concern (6). While the course of CIN is usually benign and transient, permanent renal failure requiring dialysis is known to occur (26,27). Even more concerning is that the frequency of patients in need of dialysis within a 2-year period after exposure to an iodinated CM has been reported as four times higher in patients with initial laboratory findings of CIN compared to those without, irrespective of whether the initial SCr increase returned to normal in the immediate period after the event (28). Bearing in mind that normal renal function as measured by SCr can be maintained in spite of significant loss of functional nephrons, permanent damage can occur also in patients who seemingly recover after an episode of CIN. Such patients would be more vulnerable to further renal insults from age, disease or repeated CM injections, which would explain the increased long-term renal morbidity.
In the two trials described here, CIN occurred in eight out of 74 patients (10.8%) in the iosimenol groups and in 12 out of 70 (17.1%) in the iodixanol group with slightly more cases in the arteriography trial which seems in line with historically reported data. For instance, Morcos et al. reported an incidence of 3.1–10.9% after intra-arterial administration and 2.6–8.5% after intravenous administration (27).
A potential drawback of higher viscosity in clinical practice concerns injectability (9). Considering the intravenous (CT) and intra-arterial (DSA) injections through low-diameter lines with high flow rates, intraluminal pressure may become a safety issue. Catheters with a larger diameter, on the other hand, may result in more frequent vascular complications requiring postprocedure transfusions and even causing major adverse cardiac events (29).
In both trials the efficacy of iosimenol was comparable to that of iodixanol. The higher iodine concentration of iosimenol potentially contributed to a somewhat higher rate of “excellent “images in the angiography trial compared to iodixanol. This higher iodine content probably also is the reason for the small difference in contrast enhancement in the CT trial. The additionally observed difference in contrast enhancement between the two trial centers is most likely due to different scanner types.
There are in particular three potential weaknesses of the trials that should be mentioned. The low number of patients enrolled does not allow for conclusions as to subtle differences in the safety profile of the CM that may yet be of clinical importance. Of particular interest with regard to further exploration in larger trials, as well as in high-risk populations, is the frequency of CIN observed after injection of iosimenol when compared to iodixanol. Second, the trials did not include any comparison with the older, monomeric CM still frequently used in clinical practice. Finally, the design of the trials does not allow for any in depth comparison of possibly clinically meaningful advantages attributable to the diagnostic potential of the higher iodine content of iosimenol.
In conclusion, iosimenol upon intravenous as well as upon intra-arterial injection, exhibits a safety profile and shows an efficacy similar to that of iodixanol.
Footnotes
Acknowledgements
The authors would like to thank Thomas Brzoska for his help to write this article.
Declaration of interests
The owner of the development rights for Iosimenol 340 is presently Otsuka Pharmaceutical Co., Ltd (Osaka, Japan). Karoline Meurer and Barry Hogstrom are both employees of subsidiaries of Otsuka Pharmaceutical Co., Ltd.
Funding
The study was funded by Interpharma Praha (Prague, Czech Republic).
