Abstract
Fermented beverages made from cactus fruits hold deep historical, cultural, and nutritional significance in arid and semi-arid regions of Mexico and the United States. Despite their longstanding role in local diets and food systems, these beverages remain understudied in ethnobiological research. One example is colonche, a traditional beverage made by open fermentation of prickly pear fruits, particularly Opuntia streptacantha, in the Central Mexican Plateau. Similarly, the Comcaac (Seri) people of the Sonoran Desert prepare imám hamaax (pitahaya wine) from Pachycereus pringlei fruit. This study combines ethnographic and microbiological approaches to investigate the production of these two beverages and their associated microbial communities. We documented traditional knowledge about harvesting, preparation, and fermentation practices in both regions. Forty-three yeast isolates were obtained from these fermented beverages, representing 12 species from 10 genera. These included both Saccharomyces and non-Saccharomyces yeast species, such as Kluyveromyces marxianus, Zygosaccharomyces bailii, Torulaspora delbrueckii, Meyerozyma guilliermondii, and Rhodotorula mucilaginosa. We observed regional differences in yeast composition, reflecting local ecological conditions and fermentation practices. This study contributes to the ethnobiology of traditional fermentations by documenting the microbial and cultural diversity of cactus-based beverages and highlighting the depth of the traditional ecological knowledge that sustains them. Our findings suggest that these fermentation systems provide valuable insights into microbial management, signs of domestication, biocultural resilience, and food heritage in arid environments.
Introduction
Arid and semi-arid regions, which cover more than half of Mexico (Secretaría del Medio Ambiente y Recursos Naturales [SEMARNAT], 2018), harbor exceptional biotic and cultural diversity. The Sonoran and Chihuahuan Deserts support a wide variety of flora adapted to extreme climatic conditions. Among the most iconic of these are cacti (Cactaceae), which are prized for their morphological diversity and remarkable drought resistance. Their succulent stems store water, enabling them to survive during prolonged dry periods (Ramírez-Rodríguez et al. 2020). Additionally, many cactus species produce edible fruits that have long sustained various populations in arid regions.
Despite their nutritional and cultural importance, cactus fruits are scarcely studied in arid regions of Mexico and the United States. A wide range of species, including those of the genera Echinocactus, Ferocactus, Hylocereus, Mammillaria, Escontria chiotilla, Myrtillocactus geometrizans, and various Opuntia and Stenocereus species, are still traditionally gathered or cultivated for their fruits (Pimienta-Barrios et al. 1997; Casas, Caballero and Valiente-Banuet 1999; Casas and G. Barbera 2002). While Opuntia ficus-indica dominates commercial production worldwide, other cacti continue to play an important role in regional diets (Ramírez-Rodríguez et al. 2020).
Fermented beverages made from cactus fruits are an integral part of these regional food traditions. Across cultures, these beverages have provided nutrition and medicinal benefits, as well as safe hydration in water-scarce environments. They have also played key roles in ritual practices and social cohesion (Koechlin Bokulich et al. 2014; Tamang et al. 2020; Lappe-Oliveras et al. 2025). These fermentation practices are deeply embedded in place-based knowledge systems, representing intergenerational ecological expertise (Flachs and Orkin 2019; Mutlu Sirakova 2023). Traditional fermented beverages are thus biocultural expressions of human–environment relationships (Ojeda-Linares et al. 2021, 2024, 2025). However, the knowledge required to produce these beverages is rapidly diminishing or being passed on poorly. As a result, many traditional beverages remain marginalized, and there is little documentation of the plant substrates used, and even less understanding of the microbial communities essential for their fermentation (Ojeda-Linares et al. 2021).
This study documents two cactus-based fermented beverages: colonche, made from Opuntia spp. fruits in the Central Mexican Plateau (also known as the Altiplano Central de México), and imám hamaax, a fermented beverage made by the Comcaac (also known as Seri) people from the cardón fruits (Pachycereus pringlei) in the Sonoran Desert. The current work uses ethnographic and microbiological data, aiming to explore Aridoamerica, a region spanning northern Mexico and the southwestern United States, which has a long history of cactus management and domestication (Casas and Barbera 2002; Casas et al. 2007; Lamia, Zouhir and Youcef 2018; Nabhan, Colunga-GarcíaMarín and Zizumbo-Villarreal 2022; Alvarado-Sizzo and Casas 2023; Bravo-Avilez and Rendón-Aguilar 2025). The long-standing relationship between cultural groups and cacti has led to a diverse array of practices, involving food, medicine, and fermentation (Bruman 2000; Mapes and Basurto 2016). However, fermentation is one of the least explored areas of ethnobiology (Ojeda-Linares et al. 2020; 2024).
Ecologically, Aridoamerica is a heterogeneous region that can be divided into two principal areas. The Sonoran Desert is characterized by bimodal rainfall patterns and the presence of large columnar cacti (Felger 1974). It spans parts of Sonora, Baja California, and the southwestern United States. In contrast, the Chihuahuan Desert, centered in northern Mexico and extending into the southwestern United States, is distinguished by its summer-dominant rainfall and rich xerophytic plant diversity (Schmidt 1989; Zavala-Hurtado and Jiménez 2020). The Chihuahuan Desert alone harbors more than 3,000 plant species, including several Opuntia species with edible fruits (Lamia, Zouhir and Youcef 2018). The political border between Mexico and the United States separates these two desert regions, each of which is shaped by distinct environmental dynamics, legal frameworks, social norms, and cultural conceptions of the landscape.
Opuntia fruits, commonly known as tunas in Spanish or prickly pear fruits, come in a variety of colors. They are widely consumed as fruit and processed into products such as queso de tuna candy or melcocha jam (Nabhan et al. 2020a, 2020b; Ojeda-Linares et al. 2020; Nabhan, Colunga-GarcíaMarín and Zizumbo-Villarreal 2021; Paz-Navarro et al. 2023). The Mayo people collect fruits from O. gosseliniana, O. leptocaulis, and O. wilcoxii, while the O’odham and Comcaac people collect fruits from O. acanthocarpa and O. fulgida, respectively (Ramírez-Rodríguez et al. 2020). In the Chihuahuan Desert, groups such as the Tarahumara used to collect fruits of Opuntia, columnar cacti, and Ferocactus to obtain a juice that was later fermented either on its own or added to another traditional fermented beverage made with malted maize, known as tesgüino (Pennington 1963). These fruits are often eaten fresh but are also traditionally fermented into beverages such as colonche, also known as nochoctli (from Nahuatl: nochtli = cactus fruit, octli = wine), which refers to a closely related form of cactus fruit fermentation rooted in different historical and linguistic traditions (Ojeda-Linares et al. 2020).
Colonche is a pre-Hispanic fermented beverage made from prickly pear fruits that was once widespread throughout the Central Mexican Plateau. Colonial texts often grouped their indigenous producers under the term “chichimecas,” a colonial exonym derived from the Nahuatl language that denotes perceived cultural otherness rather than a distinct group (Powell 1983; Soustelle 1993). Contemporary colonche production continues among mestizo and indigenous communities in both the Sonoran and Chihuahuan Deserts (Ojeda-Linares et al. 2020; Nabhan, Colunga-GarcíaMarín and Zizumbo-Villarreal 2021). Nevertheless, historical records suggest that colonche played an important role in the diets of various culturally settled and nomadic cultural groups in the Tunal Grande region, which encompasses the current states of Guanajuato, Zacatecas, and San Luis Potosí in Mexico, and is known for its dense prickly pear cactus diversity (Powell 1983).
Several columnar cactus fruits, including those of Pachycereus pringlei, P. weberi, P. pecten-aboriginum, Stenocereus thurberi, S. gummosus, Carnegiea gigantea, and Pachycereus schottii, are used to make fermented beverages by indigenous and non-Indigenous communities in the Sonoran Desert. These communities include the Guarijíos and Pimas of Sonora and Chihuahua, the Pápagos of Sonora, and the Comcaac of coastal Sonora (Felger and Moser 1974). These plants are not only key ecological components of arid ecosystems but also hold deep cultural and historical significance in the borderlands, where biocultural practices transcend contemporary political boundaries. Among these, imám hamaax is a fermented beverage that plays a significant role in the daily lives, diets, and ceremonies of the Comcaac (Hernández-Santana and Narchi 2018; Narchi, Marlett and Hernández-Santana 2020).
Fermentation is a dynamic process driven by an ecological succession of microorganisms, mainly yeasts and bacteria. These microorganisms gradually convert sugars into ethanol, acids, carbon dioxide, and a variety of aromatic and flavor molecules that are reflected in the beverage's chemical composition and sensory characteristics (Minervini et al. 2014; Schwan, Pereira and Fleet 2014; Wu et al. 2021). Saccharomyces yeasts play a pivotal role in producing ethanol and carbon dioxide, especially in the latter stages of fermentation. However, other groups of yeast, known as non-Saccharomyces, are equally important in the early stages. They contribute a wide range of aromatic compounds that enhance flavor complexity and produce other metabolites that influence the beverage's final profile (Wang, Mas and Esteve-Zarzoso 2016). In Mexican fermented beverages, Saccharomyces cerevisiae is the most frequently documented species (Ojeda-Linares et al. 2021). Several non-Saccharomyces genera, including Candida, Kluyveromyces, Pichia, Torulaspora, and Debaryomyces, are commonly associated with agave-based fermentations (Ojeda-Linares et al. 2021; Jara-Servín et al. 2024). In contrast, yeast communities in cactus-based beverages are not well studied. Only three species have been reported in colonche: Torulopsis taboadae, Saccharomyces cerevisiae, and Candida valida (Herrera and Ulloa 1981). To date, no microbiological studies have characterized the cultivable yeast diversity of imám hamaax.
This study provides the first update in nearly 30 years on the isolated yeast community of colonche and the first characterization of yeast diversity in imám hamaax. Using an integrative approach, we document both the microbial diversity and the traditional knowledge systems that sustain desert fermentation practices. This approach provides insights into the interconnected relationships among people, plants, microbes, and desert landscapes. These relationships are crucial for the conservation of this biocultural heritage.
Methods
Ethnographic Study and Study Sites
A series of ethnographic studies was conducted to document and analyze the production of colonche and imám hamaax. These studies included semi-structured interviews and participant observation with local producers (see Supplementary Material 1). Eighteen interviews were conducted in communities recognized for their traditional cactus fruit fermentation practices. The producers described the production process, including the fruit species traditionally and currently used, the fermentation techniques used, and the historical changes in methods. They also identified perceived threats to the continuity of these cultural practices, such as reduced fruit availability, market pressures, and generational gaps.
Participants were selected based on their active engagement in fermented beverage production or their recognized expertise within their communities. These individuals included household-level producers. This sampling strategy allowed us to document not only the technical aspects of fermentation but also local interpretations of fermentation activities, the symbolic and social significance of these beverages, and their role in shaping cultural identity and sense of place.
Informed consent was obtained orally from all participants, following ethical guidelines for research with Indigenous and rural communities, in which written consent may be culturally inappropriate. As has been discussed in the literature on participatory and decolonial methodologies (Defries and Chilisa 2014), oral consent is a legitimate and respectful form of agreement, especially when based on trust and continuous community involvement. Participants were explicitly informed of the study's purpose, their rights, and the intended uses of the information shared.
The study was conducted in several locations across Aridoamerica, specifically within the floristic provinces of the Sonoran and Chihuahuan Deserts. Five producers of colonche were interviewed in Laguna de Guadalupe, two in Mineral de Pozos, and one in San Luis de la Paz, all in the state of Guanajuato. Additional interviews were conducted with two producers in Pinos, Zacatecas, and four in the state of San Luis Potosí (two in Mexquitic de Carmona and two in Palmar Segundo). These localities are in the Central Mexican Plateau (see Figure 1) and are characterized by xerophytic vegetation dominated by Agave and Opuntia species.
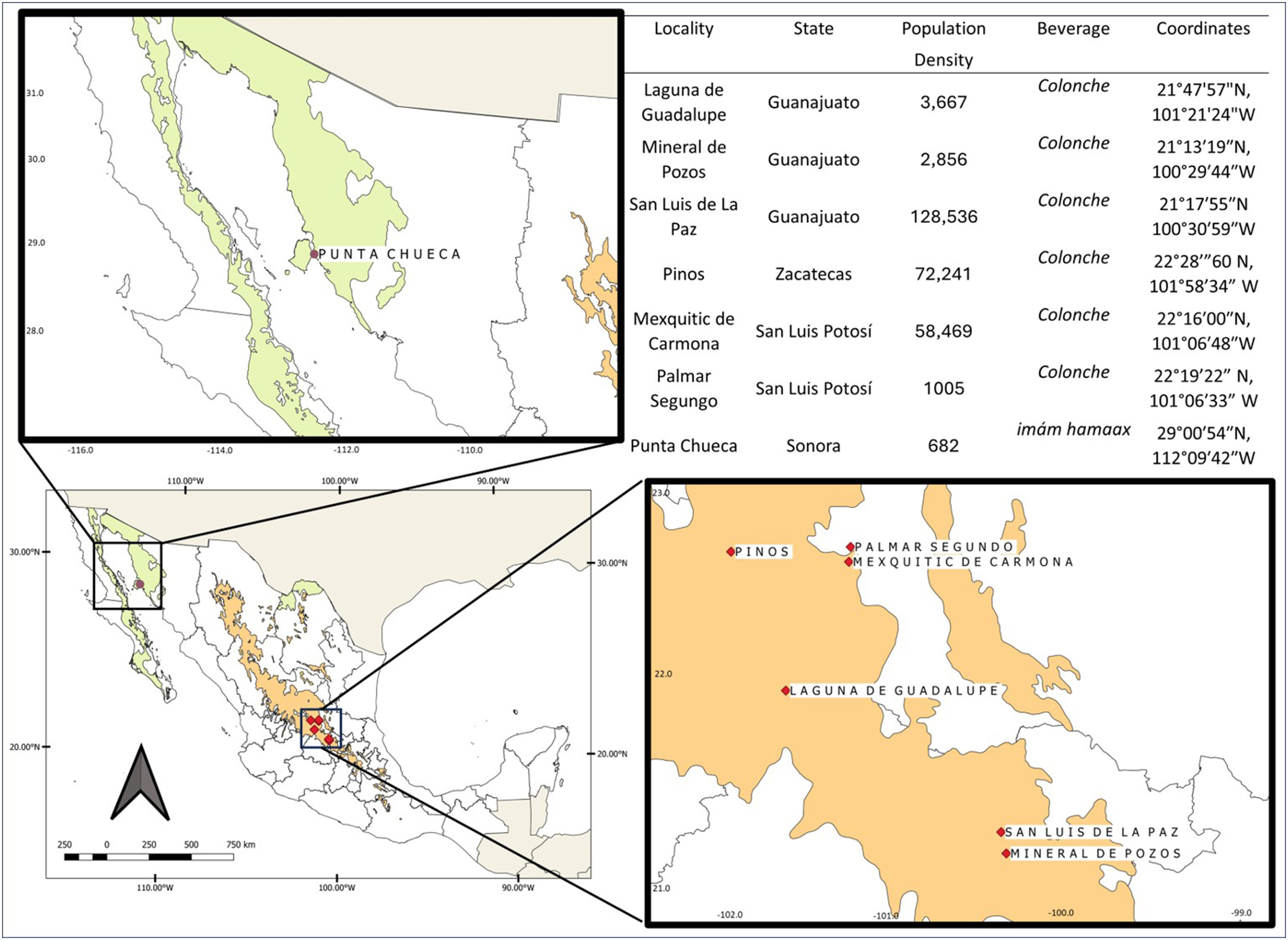
Study sites for traditional cactus fruit fermentations in arid and semi-arid Mexico. Map showing the locations of the study sites for colonche and imám hamaax production across the Aridoamerican region. The purple dot marks Punta Chueca in Sonora, home to the Comcaac community and imám hamaax production. Red diamonds indicate communities where colonche samples were collected, including sites in Guanajuato, Zacatecas, and San Luis Potosí. The green-shaded areas correspond to hot desert climates (BWh) according to the Köppen classification. These climates are characterized by high summer temperatures, low annual precipitation, and sparse desert-adapted vegetation (Peel, Finlayson and McMahon 2007). The orange-shaded areas represent semi-arid temperate climates (BS1Kw), as defined by the modified Köppen system for Mexico (García 2004). These climates are characterized by xerophytic shrublands, warm summers, and less than 5% annual rainfall in winter. These ecological zones frame the cultural and environmental contexts in which these traditional fermented beverages persist.
The mestizo communities involved in colonche production have long histories of small-scale agriculture and cactus fruit use, which are often tied to family-based knowledge transmission. These communities typically have a few hundred to a few thousand inhabitants, with older populations resulting from youth migration to urban centers. Their economies primarily rely on subsistence agriculture, seasonal factory work outside the locality, gathering in other areas, and remittances. Some also engage in artisanal food production, such as making the sweet quiote cocido (cooked agave stem) and borrachitos (cactus buds with agave sap). In Laguna de Guadalupe, we revisited producers who had previously engaged in colonche production. However, several had discontinued this activity due to generational shifts, migration, and evolving economic priorities (Ojeda-Linares et al. 2020). In other communities, we contacted the few remaining active producers, who were often elders or individuals recognized locally as skilled fermenters. Thus, the scope of the current work expanded to regions not previously addressed. Figure 1 shows population density.
Five semi-structured interviews were conducted with Comcaac producers in Punta Chueca, Sonora, for imám hamaax production (see Figure 1). The Comcaac are an indigenous group native to the central coast of the Sonoran Desert in northwestern Mexico. Traditionally semi-nomadic, they have relied on hunting, gathering, and marine harvesting, developing deep ecological knowledge of desert and coastal ecosystems. Punta Chueca (Socaaix in the Comcaac language) is one of the two main Comcaac settlements. It is located along the Gulf of California and is home to fewer than 800 people. Currently, the community's economic activities include artisanal fishing, handicrafts, government assistance programs, and occasional ecotourism.
The Comcaac's relationship with local cactus species is part of a broader ecological knowledge system shaped by long-term habitation and cultural adaptation to arid environments. The regional flora combines elements of the Sonoran plains and coastal vegetation (Felger 1974). As illustrated in Figure 1, population density in this area is low. By focusing on key knowledge holders in central Mexico, we acknowledge its geographic limitations. Notably, the northern regions of the Chihuahuan and Sonoran Deserts, particularly those spanning or adjacent to the United States–Mexico border, are underrepresented. These transboundary regions have complex histories of cultural exchange, migration, and restricted access to traditional gathering territories. Future research should explore how border dynamics affect the availability of cactus resources, the continuity and transformation of fermentation knowledge, and the development of hybrid practices among indigenous and mestizo communities that share a biocultural heritage across national boundaries.
Sampling and Physicochemical Characterization of Fermented Beverages
Eight colonche samples, ready for consumption, were collected from local producers in the aforementioned regions. Samples from Laguna de Guadalupe were prepared through open fermentation. The remaining colonche samples were produced using pulque (a fermented beverage made from agave sap) as an inoculum (Supplementary Table 1). The three imám hamaax samples were freshly prepared in collaboration with Comcaac producers through the collection of fruits and active participation in pulp extraction. Samples were taken 12 h after production. All samples were collected using sterile 50 mL Falcon tubes and handled with sterile disposable gloves. Yeast isolates were obtained from the samples immediately after collection and before freezing. The remaining aliquots were then placed in insulated coolers with ice packs (∼4 °C), transported to the laboratory, and stored in sterilized glycerol at −20 °C until further analysis.
Physicochemical parameters were measured both in the field and in the laboratory. Initial pH values were determined in the field using pH 0–6 indicator strips (Merck, Darmstadt, Germany) and confirmed later in the laboratory using a calibrated pH meter (HI 8010, Hanna Instruments, Smithfield, RI, United States). Total soluble solids were measured in the field using a handheld refractometer (Westover RHB-32ATC, Mill Creek, WA, United States), and the results were expressed in degrees Brix (°Brix). Laboratory verification of these values was performed using the same instrument (see Supplementary Table 1).
Yeast Isolation of Fermented Cactus Beverages
Yeast species from colonche and imám hamaax samples were isolated according to standard microbiological protocols. Serial decimal dilutions were prepared in triplicate in 15 mL sterile Falcon tubes containing 9 mL of sterile saline peptone solution (1 g peptone, 8.5 g NaCl, 1000 mL distilled water, pH 7.2). Starting with the fourth dilution, 100 µL of the aliquots were plated onto WL Nutrient Agar (Alpha Biosciences, Baltimore, MD, United States) supplemented with 0.03% chloramphenicol (Sigma-Aldrich, St. Louis, MO, United States) to inhibit bacterial growth. WL Nutrient Agar is commonly used to differentiate yeast species and strains in fermentation studies (Lodolo et al. 2008). The plates were then incubated at 27 °C for 2–6 days.
Colonies exhibiting distinct morphological characteristics (e.g., color, texture, edge, elevation, and surface) were selected and purified through repeated streaking on WL Nutrient Agar plates to obtain axenic cultures. The cultures were then transferred to YPD agar plates (20 g/L peptone, 10 g/L yeast extract, 20 g/L glucose, and 20 g/L agar in 1000 mL distilled water) and incubated at 27 °C for 4–6 days (Kurtzman, Fell and Boekhout 2011, 87–88).
Axenic cultures were stored in sterile distilled water at 4 °C for short-term storage or in YPD broth containing 25% (v/v) glycerol at −80 °C for long-term storage (Kurtzman, Fell and Boekhout 2011, 92). Of the 60 isolated yeast colonies, 47 (approximately 78%) were selected for sequencing of the 5.8S-ITS region, prioritizing diversity and frequency of colony morphology across replicates and locations. Before DNA extraction, each selected isolate was cultured in YPD broth.
DNA Extraction and PCR Amplification of Yeast Isolates
Genomic DNA was extracted and purified from 50 mg of wet yeast biomass obtained from overnight cultures in YPD medium. The silica-based protocol described by Rojas-Herrera et al. (2008) was used, with the addition of a step involving mechanical disruption of the cells with a pestle after three freezing/defrosting cycles. The ITS-5.8S region of the rDNA was amplified using the universal fungal primers ITS1 (5′-TCCGTAGGTGAACCTGCGG-3′) and ITS4 (5′-TCCTCCGCTTATTGATATGC-3′) (White et al. 1990). PCR reactions (30 μL) contained 30 ng of DNA template, 15 μL of Thermo Scientific (Waltham, MA, United States) DreamTaq PCR Master Mix (2×), and 0.5 μM of primers. The PCR conditions included an initial denaturation at 95 °C for three minutes, followed by 34 cycles of denaturation (94 °C, for one minute), annealing (51 °C, for one minute), and extension (72 °C, for one minute), with a final extension at 72 °C for eight minutes.
Amplicons were visualized on 1.2% agarose gels stained with GelRed. The PCR products were purified using the QIAquick PCR Purification Kit (Qiagen, Hilden, Germany) and sequenced using the BigDye Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, United States) at the Laboratorio de Biología Molecular de la Biodiversidad y la Salud, Instituto de Biología, UNAM. Sequences were aligned using Geneious v9.1.8 (Kearse et al. 2012), and taxonomic identification was performed using BLASTN (Altschul et al. 1997), with a 95% similarity threshold and an e-value of 10−6 against the NCBI nucleotide database (Sayers et al. 2021). The sequences were deposited in GenBank (see Supplementary Table 3).
Diversity Analysis
To evaluate the diversity of yeast communities in colonche and imám hamaax, we integrated data on chemical composition, producer characteristics, and operational taxonomic unit (OTU) abundance (see Supplementary Table 1). We used these data to calculate α-, β-, and γ-diversity, as well as the Shannon index. These metrics assess species richness and evenness and were computed per producer using standard ecological formulas implemented in the Phyloseq package in R (McMurdie and Holmes 2013). We only performed diversity analyses for colonche samples because they exhibit variation in fermentation practices and regional contexts. In contrast, imám hamaax samples were collected from a single site with standardized practices.
To explore variation in yeast community composition among colonche producers, we conducted a principal coordinate analysis (PCoA) using the
Phylogenetic Analysis
Molecular phylogeny was constructed using ITS-5.8S region sequences, with Entorrhiza citriformis (NCBI accession: NR_119597.1) as the outgroup. Sequences were aligned using MAFFT v7.313 (Katoh and Standley 2013), and a maximum likelihood tree was inferred using IQ-TREE v2.1.13 (Minh et al. 2020) with the GTR model. The analysis included 100 maximum likelihood (ML) tree searches and 1000 ultrafast bootstrap replicates. The resulting phylogeny was visualized in iTOL (Letunic and Bork 2007) and showed associations between yeast taxa and beverage sources.
Results
Traditional Knowledge and Practices of Colonche and Imám Hamaax Production
Although colonche production methods remain rooted in pre-Hispanic traditions, regional adaptations have emerged in response to species availability, ecological conditions, and socioeconomic factors (Ojeda-Linares et al. 2020). Producers throughout the Central Mexican Plateau consistently prefer tuna cardona (Opuntia streptacantha) because of its bright red pulp, easy peeling, and desirable masuda (doughy) texture. The fruit is harvested early in the morning to avoid saraza texture and flavor, which producers describe as overripening and loss of sugar.
In Laguna de Guadalupe (San Felipe, Guanajuato), colonche is made exclusively through open fermentation in large, unglazed clay vessels. This local pottery tradition is currently in decline, mostly due to a lack of artisans who craft these containers. The fruits are peeled in situ and crushed by hand directly into the fermentation vats, where they undergo open fermentation for 12–24 h. This process relies on naturally occurring microorganisms, such as yeasts, bacteria, and molds, that are present in the raw materials, the environment, and even on the hands of those involved in production. In Mineral de Pozos and San Luis de la Paz, producers cook mashed prickly pear cactus fruits to concentrate the juice. Sometimes, they add water or sugar to increase the yield. In addition, young entrepreneurs have begun adding pulque to start fermentation and increase volume for weekend sales. This practice arises from limited access to the quantity of prickly pear fruits required to produce colonche.
In Mexquitic de Carmona and Palmar Segundo, a semi-traditional approach involves cooking the mashed fruit in stainless steel vats and then fermenting it with starter cultures from previous batches. These cultures are known locally as zinaiste. Pulque can also be used as a substitute for zinaiste; however, all samples analyzed in this study were prepared with zinaiste. Occasionally, aromatic additions, such as cinnamon or anise, are included to enhance flavor. In Pinos, fermentation occurs in glazed clay vessels. While Opuntia streptacantha remains the main ingredient, other species are sometimes added to improve the color of colonche. Pulque, often sourced from Mexquitic de Carmona, serves as the starter culture to ensure microbial consistency and product stability. Fermentation in these localities typically lasts four to six hours (Figure 2).

Colonche production. (A) The Central Mexican Plateau is home to a variety of prickly pear cacti, including two species with red fruit: Opuntia streptacantha (commonly known as tuna cardona, shown above) and Opuntia ficus-indica (known as burrona, shown below). There is also a variety of green fruit, commonly called tuna blanca in Spanish. These varieties are particularly abundant in the town of Pinos, in the state of Zacatecas. (B) Opuntia streptacantha is the most commonly used species of prickly pear to produce colonche. Its fruits are widely distributed across different localities in Guanajuato. (C) The preparation process begins with collecting and crushing the fruits of O. streptacantha to extract their juice. (D) Local producers, such as those in Guanajuato and Zacatecas, harvest tuna cardona during the summer fruiting season. (E) A local producer stands next to the clay pots used to ferment colonche.
Imám Hamaax, Fermentation, and Ritual Among the Comcaac
The Comcaac are an indigenous nomadic people from the central Gulf region of the Sonoran Desert who have strong cultural ties to local columnar cacti (Figure 3). These cacti serve many purposes, including providing food and medicine, as well as materials for tattooing and winemaking. The traditional fermented beverage known as imám hamaax is made from the fruits of species such as cardón (Pachycereus pringlei), Stenocereus thurberi, Machaerocereus gummosus, and Carnegiea gigantea. Nonetheless, cardón fruits are primarily harvested today between April and June using a pitayero, a forked tool designed for collecting cactus fruits. Producers typically select fruits that have just opened, as this indicates optimal ripeness and high sugar content. While white fruits are considered sweeter, both red and white varieties are used in fermentation. According to local producers, pollinators such as bees and bats visit the open fruits to feed on their sugars. These animals act as vectors for wild yeasts, facilitating fermentation through the introduction of microbial communities rather than by consuming sugar. The Comcaac community believes that these interactions contribute to the onset of fermentation, reflecting local ecological knowledge of fermentation cues embedded in the dynamic desert landscape.

Imám hamaax production. (A) Open cardón fruits selected from the columnar cactus Pachycereus pringlei. (B) A pitayero, a traditional tool used to collect cardón fruits; it has a long piece of metal to pierce the fruit. (C) Producer collecting cardón fruit with the pitayero. (D) Unripe cardón fruit, unsuitable to produce imám hamaax. (E) Crushed cardón pulp prepared for fermentation. (F) Pulp is placed in a plastic bottle for fermentation, which takes about 12 hours.
Once harvested, the fruits are typically mashed and fermented in plastic bottles. In the past, however, fermentation was performed in 4–7-liter clay pots, which are no longer in use. The knowledge of how to make these pots is decaying because it is not being passed on. After fermentation, the seeds are removed from the beverage and dried for separate consumption. These seeds were used to make a yogurt-like product, but the production and consumption of this product are also declining. In fact, when we ask the producers about it, they do not remember its name. During the Comcaac New Year (Hant cmaa quiih Comcaac qui yaat), fermented imám hamaax is placed in a hollowed-out barrel cactus (Ferocactus spp.) for ceremonial use (Figure 3). Participants share the beverage while singing wine-specific songs and performing dances to invoke rain and ecological abundance.
Fermentation usually lasts 24 h, but it can extend to 48 h, depending on the temperature, which ranges from 22°C to 38°C, and the preferred alcohol content of the producers, who characterize it as strong or sweet. Nowadays, to preserve its red color, fermentation takes place in shaded areas and inside the house. For long-term storage, imám hamaax is sealed in glass bottles and sometimes buried underground. However, if the bottles are not properly sealed, the beverage can turn into vinegar, reflecting both traditional preservation techniques and the fragility of microbial processes.
Challenges to the Continuity of Traditional Cactus Fermented Beverages
The production of colonche and imám hamaax demonstrates remarkable continuity and cultural significance. However, these practices are increasingly vulnerable to converging sociocultural, economic, environmental, and climatic pressures. The persistence of these production methods since pre-Hispanic times reflects the resilience of traditional knowledge; however, ongoing changes threaten the conditions that have historically sustained these beverages. This section describes how shifts in land use, climate variability, intergenerational knowledge transfer, and material culture are eroding the foundations of traditional fermentation in Mexico's arid and semi-arid regions.
Local efforts, such as the annual Feria del Colonche (Colonche Festival) in Laguna de Guadalupe and the Comcaac New Year's celebrations, which include imám hamaax consumption, aim to preserve and promote these beverages. However, while these celebrations are meaningful, they cannot fully offset the structural threats to continuity. A key concern is the erosion of intergenerational knowledge transfer. Many current producers are older adults, and few young people are actively learning or continuing the practice. In areas such as Mineral de Pozos and San Luis de la Paz, colonche is produced only on a small scale, primarily for tourists who visit on weekends. In Punta Chueca, younger Comcaac people are interested in consuming imám hamaax but not producing it. This reflects a broader disconnection between cultural identity and practical engagement with fermentation processes. Economic marginalization, out-migration, and a lack of institutional support further exacerbate the risk of knowledge loss. This is a major trend observed in the states of Guanajuato, Zacatecas, and San Luis Potosí.
Ecological and infrastructural changes also put pressure on traditional production systems. In the Sonoran Desert, imám hamaax producers have expressed concern about the Saguaro Liquefied Natural Gas (LNG) project, which includes the 800-kilometer Sierra Madre Gas Pipeline (Energy Analytics Institute, 2024). The pipeline cuts through ecologically and culturally significant landscapes in the Sonoran and Chihuahua Deserts. The project threatens keystone cactus species such as Pachycereus pringlei. This specie is ecologically and symbolically important to the region's indigenous communities and to the production of fermented beverages such as imám hamaax.
Environmental stressors, particularly those related to climate variability, are also reducing the availability and quality of fermentation resources. Shorter rainy seasons, hailstorms, and rising temperatures have reduced cactus fruit yields and increased susceptibility to pests. Producers of colonche and imám hamaax report traveling greater distances to collect sufficient fruit, which increases production costs and reduces accessibility. In places such as Laguna de Guadalupe, the fragmentation of their historic majadas (fruit-gathering commons) due to land privatization and conversion to monocultures or intensive cattle ranching directly affects native Opuntia streptacantha populations (Figure 4). Wider land use changes, such as conversion to Opuntia ficus-indica plantations or intensified Agave cultivation driven by the growing market for distilled beverages, are replacing traditional cactus landscapes across the Central Mexican Plateau in the states of Zacatecas and San Luis Potosí.

Changes in the production of cactus-fermented beverages. (A) Local landscape where tunas are collected. Fires and intensive cattle grazing have contributed to the erosion of the nopaleras (Opuntia landscape). The significant removal of O. streptacantha (1) has led to the rapid expansion of O. robusta (2), a species not used by colonche producers. (B) Changes in the containers used by local producers of imám hamaax, a Ferocactus spp. used for the Comcaac New Year celebration (Hant cmaa quiih Comcaac qui yaat).
Meanwhile, ecological shifts in species composition further complicate harvesting. Accelerated grazing has impeded the regeneration of slow-growing species such as O. streptacantha. This has allowed faster-growing yet less desirable species, such as O. robusta and Cylindropuntia spp., to dominate historic landscapes that were once predominantly covered by O. streptacantha (Figure 4). Local producers consistently report a marked scarcity of tuna cardona, due to these factors.
Material transformations in fermentation practices reflect both adaptation and constraint. Traditional tools, such as clay fermentation vessels, are increasingly being abandoned and replaced by plastic containers due to the fragility, cost, and limited availability of traditional materials. While some producers cite environmental awareness, such as conservation concerns about harvesting barrel cacti, as a reason for changing practices, practical limitations often prove more decisive across the regions studied. In this sense, plastic containers offer producers a practical, accessible fermentation material.
Physicochemical Characteristics of Traditional Fermented Cactus Beverages and Yeast Diversity
Traditional fermentation practices maintain complex ecological relationships among humans, plants, and microbes. Yeasts play a central role in shaping the sensory profiles, nutritional value, and resilience of these beverages. Our analysis revealed differences in yeast diversity in both colonche and imám hamaax, highlighting the biocultural and ecological value of these fermentation systems. Although the pH values of all samples are similar, the sugar content of the open-fermented colonche samples differed from that of the other colonche samples (Supplementary Table 1).
Forty-three yeast isolates representing 12 species in 10 genera were obtained (Supplementary Table 1). Notably, no operational taxonomic units (OTUs) were shared between the colonche and imám hamaax samples, reflecting the distinct yeast communities associated with these beverages. Although there was similar α diversity (species richness and abundance) among producers (Supplementary Table 2), β diversity (species turnover between samples) was higher in colonche (2.6) than in imám hamaax (1.5; Figure 5A), suggesting greater heterogeneity in colonche fermentations and differences in sample size. The Shannon index confirmed these patterns, indicating higher richness and evenness in the colonche samples (Supplementary Figure 1).
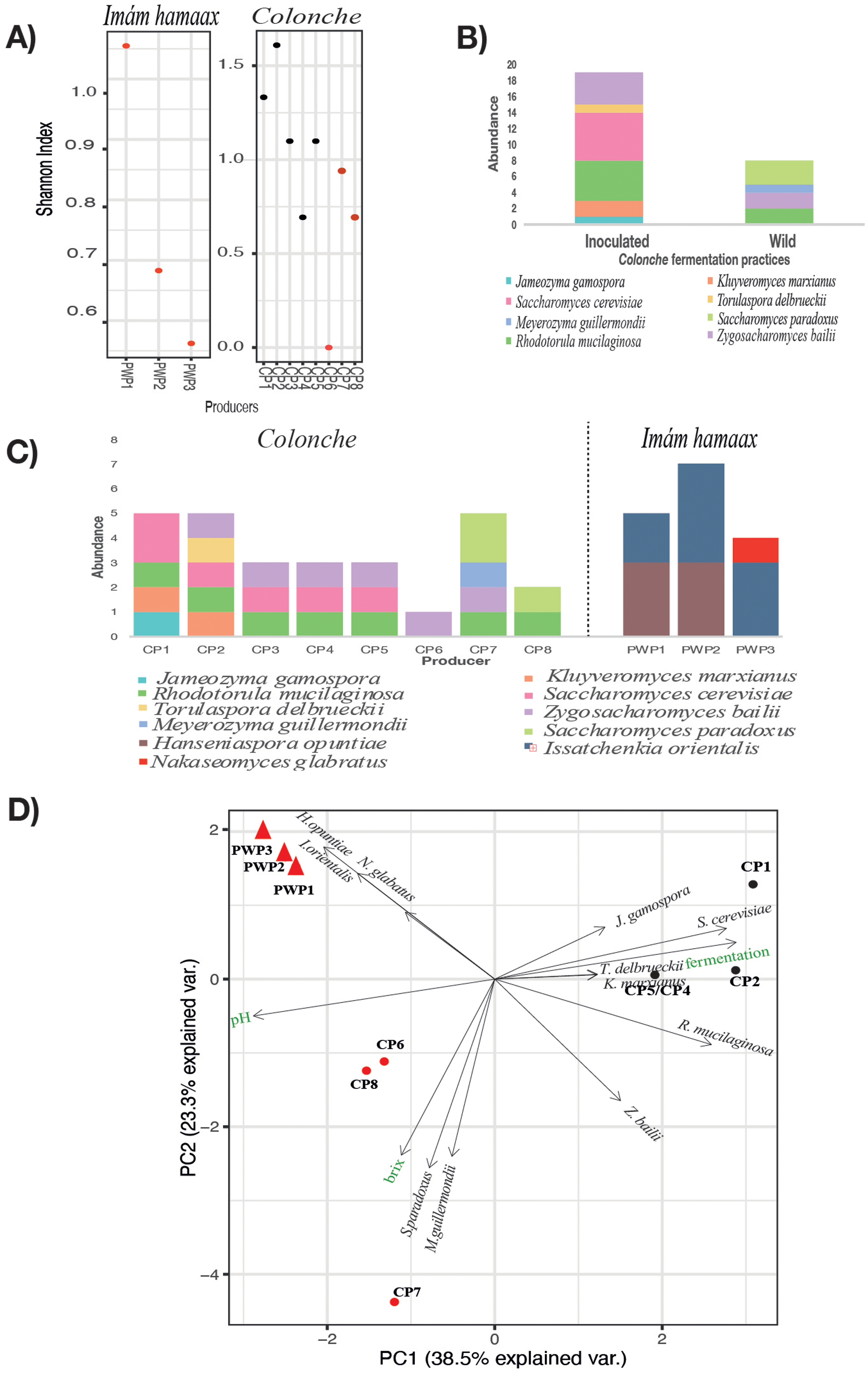
Diversity and community structure of yeast isolates from traditional fermented cactus fruit beverages. (A) Shannon diversity index values comparing yeast richness within colonche and imám hamaax samples by producer (red dots = open fermentation; black dots = inoculated samples). (B) Stacked bar plot of community structure in colonche samples grouped by fermentation practice (inoculated vs. open fermentation); taxa abundances are shown in different colors. (C) Stacked bar plot of community structure and taxa abundance across colonche and imám hamaax samples, organized by producer. (D) Principal Coordinates Analysis (PCoA) of yeast community composition, incorporating chemical characteristics (pH and °Brix). Triangles = imám hamaax; circles = colonche. Black markers = inoculated samples; red markers = open fermentation.
The yeast species varied between beverages. In imám hamaax, Issatchenkia orientalis was consistently present in all producers (Supplementary Table 1), while Nakaseomyces glabratus appeared in only one sample. Colonche fermentations showed greater taxonomic diversity. Rhodotorula mucilaginosa was present in nearly all samples, while Torulaspora delbrueckii and Jamesozyma gamospora were isolated only from certain producers (Figure 5A). The greater yeast diversity observed in colonche samples may be partly attributed to the larger number of samples analyzed (Figure 5C). Specifically, Saccharomyces cerevisiae was abundant in inoculated colonche fermentations, yet absent or present at very low levels in open fermentations (Supplementary Table 1).
Fermentation practices also influenced the composition of the yeast community. Open fermentations included species that were absent from the inoculated samples, such as Meyerozyma guilliermondii and Saccharomyces paradoxus (Figure 5D). These yeasts were associated with slightly higher pH and Brix levels (Figure 5E), which suggests potential differences in metabolic activity or sugar consumption. Notably, two colonche producers (PC3 and PC7) from Los Pinos in Zacatecas and Mineral de Pozos in Guanajuato exhibited nearly identical yeast profiles and chemical parameters. However, we cannot currently attribute this similarity to a specific factor.
Phylogenetic reconstruction based on ITS-5.8S sequences using maximum likelihood (ML) methods grouped the yeast isolates into well-supported monophyletic clades (Figure 6). Nevertheless, low bootstrap values for species such as M. guilliermondii, Jamesozyma gamospora, and Nakaseomyces glabratus highlight the limitations of single-locus markers in capturing the complete evolutionary complexity of these taxa. This finding is consistent with previous studies using whole-genome data (Hittinger et al. 2015; Shen et al. 2016; Li et al. 2021). Although ITS-5.8S-based reconstructions offer a broad view of genus-level relationships, our primary objective was to evaluate community structure within genera rather than to determine the phylogenetic relationships among the discovered taxa. The presence of under-characterized yeast taxa, along with beverage-specific communities, illustrates the importance of characterizing the ecological and cultural co-evolution of microbial communities in fermented beverages, as well as human-guided technological preferences.
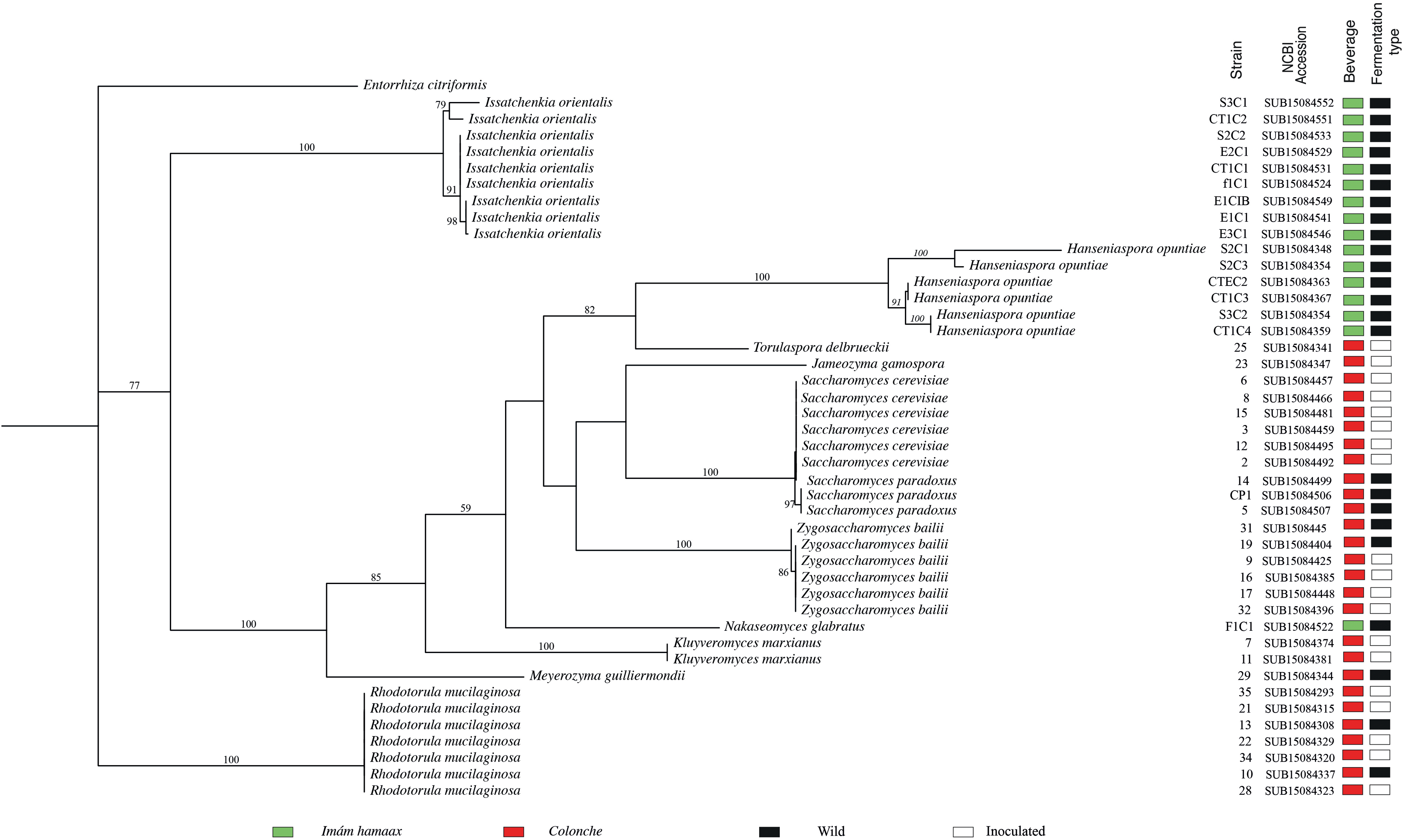
Phylogenetic tree of yeast species isolated from colonche and imám hamaax, inferred using maximum likelihood (ML) analysis based on ITS-5.8S sequences. Panel colors on the right indicate beverage source (red = colonche, light green = imám hamaax). Black boxes represent samples from open fermentations, and white boxes represent samples from inoculated fermentations. For each strain isolated, the corresponding NCBI accession number is shown. Bootstrap support values (>50%) are indicated at major nodes. The tree highlights genus-level clustering and taxonomic relationships among isolates from both beverages.
Discussion
Traditional Fermentation Practices as Biocultural Heritage
Fermented beverages have gained global attention for their probiotic properties and health benefits (Katz 2012). However, many traditional fermentation practices, especially in arid regions such as the Chihuahuan and Sonoran Deserts, remain undocumented. This invisibility endangers both microbial and cultural diversity (Sirakova 2023) because much of this knowledge is transmitted orally and is vulnerable to erosion due to modernization and changing cultural values (Flachs and Orkin 2019; Sirakova 2023). Documenting these practices through recipes, videos, or interviews is essential because it preserves the practices and the local biodiversity and knowledge systems they embody. Similarly, characterizing the microbial communities associated with these beverages is crucial, as they are part of the local agrobiodiversity that is often overlooked (Ojeda-Linares et al. 2024).
In harsh and variable environments, fermentation serves as a strategy for preserving food and as a repository for local ecological knowledge and microbial diversity (Nabhan, Colunga-GarcíaMarín and Zizumbo-Villarreal 2021; Ojeda-Linares et al. 2024). It is also used for rituals and festivities. Our findings on colonche and imám hamaax, for example, demonstrate how microbial variation correlates with distinct fermentation methods and local fruit ecologies. This highlights the interplay between human knowledge and the environment. These traditional fermentation practices are not uniform across the Aridomericas. Indigenous groups such as the N'dee (Apache), O’ob (Pima), Opata, and Tohono O’odham have historically fermented cactus fruits, including those of Carnegiea gigantea and various Opuntia species. Ethnobotanical studies, such as Fontana's Ethnobotany of the Saguaro (1980) and earlier work on Papago ceramics (Fontana 1962), demonstrate how fermentation techniques vary according to vessel design and other culturally specific practices. Although studies on the use of different fermentation containers are scarce, clay vessels may facilitate microbial population enrichment due to their porous structure, which allows microbes to accumulate and persist. In contrast, plastic containers are disposable, non-porous, and offer limited microbial adhesion. The relevance of the material in supporting microbial communities should be addressed in future studies. These localized practices reflect a broader, pan-Aridamerican tradition of fermentation shaped by ecological diversity and sociocultural innovation.
Examining these traditions through the lens of biocultural diversity (Maffi 2005) and food sovereignty provides insight into how communities maintain resilience in the face of ecological stress and colonial disruption. Our comparative approach reveals that fermentation knowledge is both culturally situated and microbially distinct, providing insight into microbial terroir, ecological adaptation, and food heritage.
The Disruption of Fermented Beverage Landscapes
The fragmentation and privatization of communal lands have profoundly disrupted the socioecological foundations of traditional fermentation practices. As noted by De Grammont and Mackinlay (2009) and Martínez Navarrete and Stahler-Sholk (2024), the shift from collective to individualized land tenure has eroded the traditional governance systems that once supported the sustainable harvesting of ecologically and culturally significant species, including wild cactus fruits and agaves. These changes limit access to key fermentation ingredients and weaken spatial and social relationships, such as seasonal gatherings, communal labor, and ritual exchange, which enabled the transmission of traditional knowledge across generations (Toledo 2001; Barkin 2020).
As communal lands become more fragmented or integrated into market-based systems, often through land sales, tourism development, or extractive industries, biocultural practices become more vulnerable to neglect, displacement, or co-optation. The weakening of territorial autonomy not only threatens agrobiodiversity and local food systems but also reduces the resilience of traditional ecological knowledge networks, which are central to sustaining these systems (Robson and Berkes 2011; Koechlin and Eguren 2016).
These changes are exacerbated by long-standing patterns of internal and international migration, which alter local labor structures and family dynamics. For example, migration has affected colonche producers. Migration often disrupts the intergenerational continuity of traditional ecological knowledge, including fermentation expertise (Koechlin and Eguren 2016). Conversely, including fermentation expertise. At the same time, diasporic communities may reconfigure or revive fermentation practices in new contexts, influencing how beverages like colonche are prepared and perceived in their places of origin (Nabhan 2008). Recognizing the role of mobility in shaping and reconfiguring tradition deepens our understanding of the evolving nature of these beverages and the cultural resilience they represent. Nevertheless, future studies could examine changes in sensory attributes resulting from new techniques and differences in the historical taste memories among producers and consumers.
Migration, Changes in Consumption, and Climate Threats
The decline of traditional fermented beverages, such as colonche and imám hamaax, is also closely linked to the expansion of industrialized food systems and changing consumption patterns. Commercial beverages are proliferating in rural and indigenous markets and increasingly displacing local alternatives, contributing to the erosion of biocultural heritage (Hernández-Santana and Narchi 2018; Narchi, Marlett and Hernández-Santana 2020; Ojeda-Linares et al. 2021; Ojeda-Linares, Vallejo and Casas 2023). This marginalization is not only economic but also epistemic: fermentation practices are largely excluded from dominant public health and sustainability narratives, despite their nutritional, ecological, and cultural importance (Ojeda-Linares et al. 2021; Ojeda-Linares, Vallejo and Casas 2023). These converging pressures underscore the urgency of documenting, supporting, and revitalizing fermentation knowledge as part of broader efforts to defend food sovereignty, agrobiodiversity, and cultural resilience in arid and semi-arid regions.
As food systems face pressures from land privatization, migration, and industrial food regimes (De Grammont and Mackinlay 2009; Hernández-Santana and Narchi 2018), another equally urgent and interconnected challenge emerges: climate change. The erosion of cultural practices weakens adaptive capacity (Toledo 2001; Robson and Berkes 2011), and climate variability directly threatens ecological resources and environmental conditions. This also threatens the production of fermented beverages. Understanding the future of traditional fermented beverages requires examining how microbial communities and desert-adapted crops intersect with resilience strategies in the face of increasingly unpredictable environmental conditions (Tengö et al. 2017). This highlights the role of fermentation as both a cultural repository and a biotechnological ally in navigating ecological uncertainty (van der Heijden and Hartmann 2016).
Climate change is likely to exacerbate the instability of crop yields and the frequency with which conventional annual and perennial crops fail (Lobell and Gourdji 2012), forcing us to rethink the design of our food systems, especially in arid and semi-arid regions (Chivenge et al. 2015). In this context, desert-adapted perennial polycultures, which are deeply embedded in Indigenous food systems, are a strategic pool for selecting candidate crops to develop fermented foods with prebiotic and probiotic potential (Nabhan and Mabry 2016; Ojeda-Linares, Solís-García and Casas 2022).
Opportunities for Policy and Community-Led Revitalization
Despite the above-outlined challenges, recent policy efforts suggest that there may be opportunities to support the revitalization of traditional foodways through community-led, culturally grounded development frameworks. National policy frameworks, such as the Planes de Justicia, have emerged in recent years to address historical injustices by restoring territorial and cultural rights to indigenous communities in Mexico (INPI, 2022). These initiatives are particularly important for indigenous groups in the Sonoran Desert, including the Yaqui, Seri, and Yoreme peoples. Their lands and livelihoods have long been affected by extractive development and state neglect (Valencia 2021; Robles 2023).
Current government efforts focus on improving the economic well-being of Seri families by strengthening traditional livelihoods such as fishing and ecotourism, promoting productive and commercial activities, and sustainably managing local ecosystems and biodiversity (SEMARNAT, 2021; INPI, 2022). Within this framework, the revitalization of traditional foodways, including fermentation practices, is expected to become more important, as these practices often play a central role in community-led initiatives for cultural resilience and ecological restoration (Ojeda-Linares, Vallejo and Casas 2023). However, without targeted, community-informed interventions, such as protecting land and developing access, developing climate adaptation strategies, and providing culturally sensitive economic incentives, these beverages may face an irreversible decline. This would not only reduce microbial and plant biodiversity but also erode cultural heritage, weaken food sovereignty, and sever connections between people, place, and ecological knowledge systems.
Microbial Terroir and Ecological and Biocultural Patterns in the Yeast Diversity of Cactus Beverages
The microbial composition of traditional fermented beverages exemplifies the interconnected relationships between humans, microbes, plants, and the environment. Current research highlights the co-production of knowledge, demonstrating that the microbial diversity of beverages such as colonche and imám hamaax is shaped by ecological and cultural factors. These practices generate distinct microbial communities that influence the beverages’ sensory profiles.
The analysis of yeast diversity among different producers revealed variations in both species composition and abundance, consistent with previous reports showing higher yeast richness and abundance in inoculated samples (Ojeda-Linares, Solís-García and Casas 2022). These patterns reflect the influence of specific fermentation practices, local fruit ecologies, and community-based methods. Similar trends have been observed in studies of open-fermented agave sap (pulque), where differences in yeast composition were associated with ecological variation and fermentation techniques (Chacón-Vargas et al. 2020a, 2020b; Gallegos-Casillas et al. 2024; Colón-González et al. 2025).
Open-fermented beverages, such as colonche and imám hamaax, serve as reservoirs for diverse yeast species, many of which are influenced by the specific ecological conditions of arid and semi-arid landscapes. The microbial communities in these fermentations reflect not only cultural practices and fermentation techniques but also local environmental conditions and substrate composition.
Phylogenetic reconstruction based on ITS-5.8S is limited to a single-locus marker and cannot fully capture the evolutionary complexity of these taxa. Previous studies have reported incongruences when reconciling fungal taxonomic circumscription with true evolutionary divergence, highlighting the broader challenges of fungal systematics (Hittinger et al. 2015; Shen et al. 2016; Li et al. 2021). In this context, our ITS-based analysis should not be interpreted as an attempt to resolve the deeper evolutionary history of these lineages. Rather, it serves as a practical tool to depict the composition and exclusivity of yeast communities in traditional fermented beverages. This analysis highlights community-level patterns and genus-level specificity, which are shaped by ecological conditions and human-guided selection processes. ITS-based reconstructions may show inconsistencies compared with whole-genome phylogenies. Low branch support for sparsely sampled genera, such as Jamesozyma gamospora, Nakaseomyces glabratus, and Torulaspora delbrueckii, underscores the central message of our work. Microbial communities in fermented beverages are under-characterized biotic assemblages whose diversity and organization cannot yet be fully explained by systematics alone. Instead, these communities must be understood in the context of the ecological interactions, cultural traditions, and technological practices that have shaped them over time.
Our results show that Saccharomyces cerevisiae was consistently present in all inoculated colonche samples but conspicuously absent from open fermentations and imám hamaax. Although S. cerevisiae is not always dominant in spontaneous fermentations, its presence alongside other yeasts, such as Kluyveromyces marxianus, has been reported in agave-based fermentations (Gschaedler 2017; Chacón-Vargas et al. 2020a, 2020b; Jara-Servín et al. 2024; Colón-González et al. 2025; Lopez-Gallegos et al. 2025). Differences in microbial competition, geographic origin, and substrate physicochemistry strongly influence yeast community composition and succession (Barata, Malfeito-Ferreira and Loureiro 2012; Bokulich et al. 2014), creating ecological dynamics that may inhibit the S. cerevisiae establishment (Bagheri, Bauer and Setati 2017).
The presence of S. cerevisiae in inoculated colonche fermentations is most likely linked to the pulque inoculum used by producers, rather than to a lineage specifically adapted to colonche. It is important to note that domestication status cannot be generalized across S. cerevisiae, because the species comprises multiple populations that may be wild, domesticated, or of undetermined status (De Chiara et al. 2022; Sampaio and Pontes 2025). While some domesticated populations are indeed associated with traditional fermentations, their presence does not necessarily imply deliberate introduction. In our case, pulque-derived inocula may carry domesticated populations adapted to agave substrates; however, it is unclear whether such strains can be considered domesticated in colonche. Future studies, particularly those analyzing colonche produced with reused fermentations (e.g., zinaiste), could help determine if S. cerevisiae populations undergo domestication under cactus fruit fermentation. Additionally, the biochemical properties of cactus substrates, such as sugar content, pH, and secondary metabolites, may influence the growth and persistence of S. cerevisiae (Fleet 2007; Ciani et al. 2010).
Notably, colonche is produced in regions where pulque and mezcal are produced, raising the possibility that S. cerevisiae strains migrate between agave and cactus substrates. In contrast, the production region of imám hamaax (Punta Chueca) is geographically isolated from agave cultivation areas, which may limit such microbial exchange. This remains a hypothesis, but future comparative studies on colonche and agave fermentations in the Altiplano region could help determine if yeast strains are shared between these substrates. Thus, the absence of S. cerevisiae in imám hamaax likely results from a combination of biogeographic constraints, substrate-specific limitations, and microbial competition. This observation underscores the role of local ecological conditions in shaping fermentation outcomes and highlights that S. cerevisiae is a domesticated yeast species that is typically introduced deliberately by humans (Gallone et al. 2016).
Saccharomyces paradoxus, which was found in open-fermented colonche, belongs to a lineage of Saccharomyces that is more commonly associated with forest ecosystems and arboreal habitats than with human-managed fermentations (Boynton and Greig 2014; Liti 2015). Unlike certain populations of S. cerevisiae that have undergone domestication and adaptation to anthropogenic environments such as baking, brewing, and winemaking, S. paradoxus is typically found in association with tree bark, soil, and insects. While historically not considered a major contributor to industrial fermentations, studies show that S. paradoxus can perform competitively in high-sugar environments such as grape juice (Dashko et al. 2016). More recent work has demonstrated its ability to efficiently consume sugar in wine and beer fermentations (Nikulin et al. 2020). Its presence in traditional beverages, such as pulque (Chacón-Vargas et al. 2020a, 2020b;), suggests that the microbial communities in these systems are shaped by more than just intentional inoculation with starter cultures; they are also shaped by local ecological reservoirs, such as the fermentation vat. The detection of S. paradoxus in these contexts may underscore the permeable boundaries between wild and managed microbial ecologies in traditional fermentation systems, especially since it was only found in inoculated samples (Lopez-Gallegos et al. 2025).
Similarly, Hanseniaspora opuntiae, which was identified in imám hamaax, is a common apiculate yeast found on fruit surfaces and insect vectors (Lopez-Gallegos et al. 2025). It is typically found during the early stages of open fermentation due to its tolerance of oxidative environments, rapid growth rate, and ability to compete under low-ethanol conditions (Ciani et al. 2010; Padilla, Gil and Manzanares 2016). Its dominance in imám hamaax may be related to the beverage's short fermentation time and relatively low alcohol content. These conditions create an environment in which H. opuntiae can persist and influence the structure of the initial microbial community. H. opuntiae plays a crucial sensory role in fermentation. It contributes fruity and floral aroma compounds, such as esters and higher alcohols, which enhance the organoleptic complexity of fermented beverages (Seguinot, Ortiz-Julien and Camarasa 2020). Although H. opuntiae is eventually outcompeted by more ethanol-tolerant yeasts in longer fermentations, its metabolic contributions in the initial stages are critical for flavor development and microbial succession.
Interestingly, local producers have reported fermentation delays or failures when pollinating insects are scarce, suggesting these insects may play an ecological role as vectors of key fermentative yeasts associated with cactus fruits. This observation aligns with studies demonstrating the importance of insect-mediated dispersal in maintaining wild yeast populations and promoting microbial diversity in natural fermentations (Lam and Howell 2015; Madden et al. 2018; Stefanini et al. 2018). Together, these findings highlight the role of H. opuntiae in fermentation systems linked to arid-land ecologies and cactus fruits, and they underscore the need for further ecological and microbiological research on insect-yeast-plant interactions in traditional fermentation practices.
Arid fermentation environments support yeast communities with exceptional metabolic flexibility and tolerance to environmental stressors. Rhodotorula species, which are identified by their distinctive orange-salmon pigmentation due to carotenoid production, were present in all colonche samples. These pigmented basidiomycetous yeasts are commonly found in phyllospheres, soils, and extreme environments. They are known for their ability to assimilate a wide range of carbon and nitrogen sources, tolerate oxidative stress, and grow under fluctuating pH levels, osmotic conditions, and temperature regimes (Kot et al. 2016; Lopes et al. 2023). Their metabolic plasticity, antioxidant potential, and ability to accumulate lipids may explain their ecological success in the fluctuating and sugar-rich environment of colonche fermentation. Rhodotorula species were diverse throughout the colonche samples. These results are consistent with those of a previous study, in which this yeast group was present in both clay vessel and inoculated fermentation samples, as well as in both types of fermentation (Ojeda-Linares, Solís-García and Casas 2022).
In contrast, Issatchenkia orientalis was exclusively found in imám hamaax fermentations. This species is gaining recognition in traditional fermentation and biotechnological processes thanks to its exceptional tolerance of high temperatures (up to 45°C), low pH levels (below 3.0), ethanol, and fermentation inhibitors such as acetic acid and furfural (Chan et al. 2012; Mukherjee et al. 2017). Its metabolic activities contribute to flavor development by releasing organic acids (especially succinic and acetic acids) and volatile esters. Additionally, I. orientalis may have probiotic effects, and researchers are investigating its ability to survive gastrointestinal transit and modulate host metabolism (Endo, Okada and Uchimura 2011). The presence of I. orientalis in imám hamaax suggests its adaptation to the rapid, low-alcohol, and acidic fermentation environment characteristic of this beverage.
Zygosaccharomyces bailii is present in open and inoculated fermentations of colonche samples and is often referred to as a spoilage yeast due to its persistence in preserved foods. However, recent research has highlighted its resilience under extreme osmotic pressure, low pH, and the presence of weak acids. It can also produce killer toxins with antifungal properties that may influence microbial succession and diversity in mixed fermentations (Stratford et al. 2013; Solieri 2021). These species were found in different inoculated samples, and these yeast strains were also characterized in pulque fermentations (Chacón-Vargas et al. 2020b). Thus, the presence of these species may be related to the use of pulque as a starter culture for colonche fermentation.
Other yeasts found in colonche and imám hamaax demonstrate the functional and ecological diversity of these traditional fermentations. Nakaseomyces glabratus, found in imám hamaax, is phylogenetically close to S. cerevisiae and has similar ethanol tolerance and fermentation potential. However, it is better known for its opportunistic pathogenicity in clinical contexts. In traditional fermentation systems, its role may be limited to the early stages, where it contributes to initial sugar metabolism and the exclusion of pathogens through competition. Nevertheless, future studies should aim to confirm its potential as a fermentative yeast and to characterize the microbial succession processes in both beverages.
Kluyveromyces marxianus was detected in two inoculated colonche samples. It is a thermotolerant, fermentative yeast that can metabolize a variety of sugars, including lactose, inulin, and pentoses. This yeast can switch between respiration and fermentation, grows rapidly, and produces aroma-active compounds such as ethyl acetate and isoamyl alcohol. These characteristics make it increasingly valuable in the food and bioethanol industries (Fonseca et al. 2008; Lane and Morrissey 2010). K. marxianus was found in samples from a previous colonche study (Ojeda-Linares, Solís-García and Casas 2022). Similarly, Jamesozyma gamospora species, which were also isolated from a sample of inoculated colonche, are not efficient fermenters on their own, but they are known to enhance aroma complexity in mixed fermentations. When co-inoculated with S. cerevisiae, they contribute to the development of fruity, floral, and vinegary notes typical of traditional wines and some spontaneously fermented beverages. They play a key role in the sensory expression of the final product (Jood, Hoff and Setati 2017; Hranilovic et al. 2018). This sensory expression has been recently characterized in colonche samples, where fruity, floral, and vinegary notes were detected by a panel of specialists and are markedly different in inoculated samples (Ojeda-Linares et al. 2025).
Meyerozyma guilliermondii was detected in a single open-fermented colonche sample. It is a metabolically versatile yeast with remarkable biotechnological potential. It can produce lipids, organic acids, and riboflavin (vitamin B2) under nutrient-limited and stressful conditions, especially in iron or other micronutrient-deficient media (Abbas and Sibirny 2011; Yan et al. 2021). In addition to its nutritional contributions, M. guilliermondii exhibits tolerance to osmotic and oxidative stress. This tolerance may explain its survival under the fluctuating conditions of colonche fermentation, including varying sugar concentrations, low water activity, and intermittent heat exposure, which are typical of arid environments. M. guilliermondii is also known for its antagonistic activity against certain fungal pathogens and has been studied in postharvest biocontrol applications (Cordero-Bueso et al. 2017). Its rare occurrence in colonche underscores the potential of traditional, low-intervention fermentation processes to harbor understudied microbial taxa of both ecological and applied importance.
As previously discussed, traditional food practices involve deep and complex interconnections among humans, microbes, plants, and the environment. These relationships not only shape cultural identity and food systems, but they also allow the preservation of the hidden microbial diversity. Our findings reveal the distinct microbial signatures of each beverage and the strong influence of local environments and practices, such as fermentation techniques, on microbial composition. Both beverages exhibited comparable α-diversity across producers, although species-level differences drove these patterns. In contrast, β-diversity revealed greater variation among Colonche producers (Fig. 5D), likely due to their more open fermentation systems and diverse environmental reservoirs (Wu et al. 2021). Additionally, previous studies have described similar patterns, showing that inoculated samples support higher fungal richness and community structure compared to open-fermented colonche samples (Ojeda-Linares, Solís-García and Casas 2022).
However, unresolved questions remain about the primary ecological drivers of yeast community assembly in cactus-based fermentations. While our findings highlight the influence of production techniques and ingredients, further research is needed to understand the role of sugar concentrations in shaping microbial dominance (Zhang et al. 2024), especially given the selective pressure they impose on osmotolerant yeasts. Environmental variables, including temperature, humidity, and exposure to ambient microbes, also play significant roles in influencing microbial metabolism, growth rates, and community composition across seasons (Zhang et al. 2022).
Perspectives
The diversity of yeast communities in colonche and imám hamaax reflects the deep intertwining of microbial life, traditional practices, and local ecologies. These communities contribute not only to the flavors and textures of fermented beverages but also to the cultural identity and environmental resilience of the communities that produce them. However, microbial diversity, particularly in arid regions, is largely absent from biodiversity assessments (Al-Atrash, Khadur and Khadim 2021). As traditional beverages face mounting pressure from industrialization and climate change, recognizing and safeguarding microbial ecologies becomes a scientific, cultural, and ethical imperative. These yeasts are more than biochemical agents; they cohabit our landscapes, shaped by human history and ecological specificity.
Global conservation efforts have long prioritized visible biodiversity such as plants, animals, and landscapes. Meanwhile, the microbial dimension of biocultural heritage remains largely invisible and undervalued. This oversight is particularly critical in the context of traditional fermentation systems, where microbial communities are central not only to food production but also to place-based knowledge, ecological adaptation, and cultural continuity. Traditional fermented beverages, such as colonche and imám hamaax, face pressures from industrialization, climate change, and sociopolitical marginalization, as do the microbial ecologies that sustain them. Their decline signals an underappreciated erosion of microbial biodiversity (Tamang et al. 2010; Escobar-Zepeda, Sánchez-Flores and Quirasco 2016).
Fermentation is more than a biochemical process; it is a multispecies collaboration shaped by human knowledge, local ecologies, plant substrates, and microbial interactions (Ojeda-Linares et al. 2024). The production of fermented cactus beverages in arid regions exemplifies this relationality. It is a practice that encodes ecological memory, adapts to extreme environments, and fosters cultural resilience. Microbes are not passive agents but co-producers of flavor, nutrition, identity, and adaptation (Flachs and Orkin 2019; Quave and Pieroni 2014; Sõukand et al. 2015; Flachs and Orkin 2019; Sirakova 2023). However, their role remains largely absent from biodiversity policy and conservation frameworks. Microorganisms are notably underrepresented in international agreements such as the Convention on Biological Diversity (CBD). The institutional support for the in situ or ex situ conservation of microbial strains associated with traditional food systems is minimal (Langarica-Fuentes, Woodward and Haider 2023).
The conservation of microbial diversity in fermented beverages from arid regions calls for rethinking how biodiversity is conceptualized and managed. To truly protect biocultural heritage, we must recognize microbes not only for their industrial or nutritional value but also for their entanglement with landscapes, practices, and peoples. Fermentation is a compelling site of resistance and regeneration, where microbial life supports both ecological and cultural futures in the face of accelerating change (Quave and Pieroni 2014; Sõukand et al. 2015; Flachs and Orkin 2019; Sirakova 2023; Ojeda-Linares et al. 2024).
Further conservation efforts should prioritize protecting and maintaining the landscapes where these fermented beverages are traditionally produced, recognizing these areas as biocultural heritage zones. This includes in situ conservation of the native cacti and the ecological and cultural practices associated with them. Initiatives such as gastronomic festivals can play a key role in promoting both the consumption and continued production of these beverages, strengthening local economies and cultural identity. In parallel, ex situ conservation measures, such as the preservation and documentation of associated microbial communities, are essential for safeguarding the microbial diversity that underpins these unique fermentation traditions.
Conclusions
Fermented beverages such as colonche and imám hamaax embody a confluence of ecological adaptation, microbial diversity, and cultural resilience in arid regions. Their continued, if increasingly fragile, production reflects the enduring value of biocultural knowledge systems that have evolved over centuries to thrive in dryland ecologies. These beverages not only protect microbial communities uniquely adapted to desert environments but also sustain cultural identities, social networks, and food sovereignty in Indigenous and mestizo communities. Our findings show that fermentation, often dismissed as a marginal or disappearing practice, actually persists as a dynamic strategy for resilience amidst intersecting pressures: land privatization, migration, the expansion of industrial food systems, and climate change. The microbial ecologies involved in fermentation, shaped by traditional ecological knowledge and desert-adapted crops, offer powerful insights into how food systems can remain adaptive, decentralized, and biodiverse under increasingly unpredictable conditions. Rather than treating these beverages as static remnants of the past, we argue for recognizing them as biocultural refugia, living archives of co-evolutionary relationships between humans, plants, and microbes. Preserving and revitalizing these fermentation practices is not only a matter of heritage but also of global relevance, pointing toward food system models that are locally grounded and capable of meeting future challenges in climate-vulnerable regions. Future studies should aim to characterize the full microbial communities present in both beverages using advanced techniques such as metagenomics, which can reveal the taxonomic and functional diversity of these complex fermentation systems. This detailed microbial profiling will be crucial for identifying biogeographical patterns and understanding the ecological dynamics that shape microbial agrobiodiversity across arid regions. Moreover, these insights can inform the development of culturally grounded and community-based conservation strategies that recognize the role of local knowledge and practices in sustaining microbial diversity. Establishing protocols for the conservation, management, and promotion of these microbial communities, both in situ and ex situ, will be essential to preserve the integrity and resilience of these traditional fermentation systems in the face of environmental and socio-economic change.
Supplemental Material
sj-docx-1-ebi-10.1177_02780771251411948 - Supplemental material for Yeast Communities and Traditional Knowledge in Fermented Cactus Fruit Beverages: A Microbial Heritage at Risk
Supplemental material, sj-docx-1-ebi-10.1177_02780771251411948 for Yeast Communities and Traditional Knowledge in Fermented Cactus Fruit Beverages: A Microbial Heritage at Risk by César Iván Ojeda-Linares, Laura Figueroa-Corona, Patricia Lappe-Oliveras, Sylvie Le Borgne, Rodrigo Arredondo-Fernández, Laura González-Resendiz, Erika Adriana Barnett, Gary Paul Nabhan and Alejandro Casas Fernandez in Journal of Ethnobiology
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Programa de Becarios Posdoctorales por México of the Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti) through grant support (CVU_632166). As well, the projects PAPIIT—UNAM IN209024 and IN224023.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
