Abstract
The use of insects and their hosts is essential in Mesoamerican bio-culture. Preserving this rich heritage and associated biodiversity is a multifaceted process. The first steps involve correctly identifying the used species, documenting its basic ecology, and the collecting and consumption methods. In the Monte Albán archeological site, Oaxaca, Mexico, spanning 13 centuries of pre-Hispanic occupation, local people still practice traditional native species utilization. One of the most used insect species is the cigarras. We sequenced a 648 bp region of this edible cicada's cytochrome oxidase gene. These DNA sequences and morphological studies concur that the cicada used is Quesada gigas (Olivier 1905) (Homoptera). People collect cicadas from April to May and cook them for self-consumption. Based on archeological and etymological evidence of one of their major hosts, the guaje, Leucaena esculenta (DC.) Benth (Fabaceae), the cicada consumption likely has pre-Hispanic roots. The cicada and the guaje pods are rich in proteins, probably due to the guaje's symbiotic association with nitrogen-fixing bacteria. The pods are collected for self-consumption during winter. Thus, the guaje–cigarra system provides humans with two protein sources during the dry season at different times. Q. gigas is considered a pest of some crops, illustrating different people's contrasting perceptions of the same insect. The Quesada consumption in Oaxaca might be part of a pre-Hispanic symbiosis between the guaje and humans and an example of how a potential insect pest can be used as an alternative protein source. Monte Albán is a biodiversity and pre-Hispanic traditions reservoir worth preserving.
Introduction
Mesoamerican people have a long tradition of consuming wild species for survival. Such traditions likely have pre-Hispanic roots. Contrary to the belief that maize was their sole protein source, pre-Hispanic Mesoamericans relied on many species for protein intake (e.g., Feinman and Nicholas 2020; Flannery 2009). Among such species, insects were probably an important source (Ramos-Elorduy and Pino-Moreno 1989). However, evidence of insect consumption by this group of people is needed. Not only in Mesoamerica but in other parts of the globe, edible insects are virtually invisible in studies exploring the dietary habits of the early settlers in most archeological studies. There is, however, a significant probability that even our closest human ancestors consumed insects for subsistence: insects are abundant and rich in protein (see Lesnik 2019). In southern Mexico's relatively well-studied Oaxacan Central Valleys archeological areas, insects were not considered a significant protein source for the pre-Hispanic inhabitants. This is the case of the studies conducted in the Guilá Naquitz Cave, which shows the vestiges of early domestication of maize and other plant crops (Flannery 2009). Similarly, insects are not included as possible food sources in the nearby Monte Albán archeological site, one of the most important Mesoamerican pre-Hispanic archeological sites (Feinman and Nicholas 2020; Marcus 2008). This probable omission can be explained more by the difficulty in gathering evidence of insect usage through paleozoological studies in archaeological sites than by the lack of insect consumption in pre-Hispanic times.
Studying insect consumption and other alternative food sources by a particular ethnic group is essential for historical reasons and because of the increasing demands of reconciling healthy protein ingesting with sustainable food production (Weindl et al. 2020). Maintaining a healthy human protein balance requires protein consumption (Phillips 2017; Watford and Wu 2018). However, the increasing human demand for food is generally achieved by disrupting essential ecosystem functions that eventually compromise human welfare and biodiversity (Forssell and Lankoski 2015; Weindl et al. 2020). One way to mitigate such problems could be using alternative foods (Michel-Villarreal et al. 2019). There are at least two ways to achieve this goal. One is the de novo design of food production systems. The other is recovering and fomenting ancient ways of utilizing nonconventional food sources.
Correctly identifying the species used when searching for alternative protein sources through biocultural heritage inspection is mandatory. Vernacular names of the same species may change in a few kilometers (e.g., del Castillo and Acosta 2002). Thus, a uniformizing method for referring to the studied species is required. Consequently, taxonomy is the most effective solution and the starting point in applied biological research (de Moraes 1987). This task requires reliable methods for species identification. The taxonomy of species such as insects is complicated for several reasons. Insect groups are the most diverse on the planet. Hemiptera, for instance, has roughly 82,000 described species (references in Forero 2008). Morphologically similar species are relatively common and can be taken as a single species without adequate identification techniques (e.g., Hebert et al. 2004; Kanturski et al. 2018). Furthermore, the paucity of taxonomic specialists exacerbates this problem for several insect groups (Floyd, Wilson, and Hebert 2009).
DNA barcoding is an increasingly widely used technique that can solve the identification problem in many cases. It allows the taxonomic identification of virtually all species using a single method requiring a small amount of tissue. The identification can be performed on dead organisms or preserved specimens from scientific collections. It has helped detect cryptic insect species (e.g., Kanturski et al. 2018). Once the barcoding sequence is published in open-access worldwide databases such as GenBank and BOLD, they remain permanently accessible to any people. This technique might not require taxonomic specialists for identification once DNA barcoding is available in such databases. The published sequences in BOLD are accompanied by metadata such as the geographic coordinates, specimen photographs, and names of the collectors and taxonomists identifying the specimen (see Floyd, Wilson, and Hebert 2009). Finally, this information can help further identification processes and taxonomic refinements.
In addition to taxonomy, the ecological context, such as the hosts, of the focus species is another basic requirement in the search for alternative food sources. This knowledge lies the foundation for any task aimed at bioconservation and increasing progress toward sustainable development (see Gerique 2006). Also, ethnoecology, the study of the relationship between plants, animals, and human groups, could be a promising starting point for achieving a better understanding of nature, by looking through the eyes of people who have strong ties with natural ecosystems (see Molnár et al. 2024; Posey 2001). Because of such ties, ethnic groups inhabiting their natural habitats have ample knowledge of their environment and apply it to generate direct benefits such as food procurement (see Toledo, Boege, and Barrera-Bassols 2010). Moreover, documenting the ecological knowledge and cultural value of native species by local cultures facilitates the preservation of biological and cultural diversity (Boege 2008; Maffi and Woodley 2012).
Regions with high biocultural heritage, such as Mesoamerica, are rich in nonconventional food sources. In particular, the consumption of alternative protein sources, such as insects, remains among peasants and people with tight Indian backgrounds in Mesoamerica (Ramos-Elorduy and Pino-Moreno 2004). However, due to the overwhelming acculturation, and with few notable exceptions (e.g., grasshoppers, “chapulines,” Sphenarium spp., and leaf-cutter ants, “chicatanas,” Atta sp.) widely consumed in southern Mexico, the use of other insects for food often are localized, limited to few groups of people, and rapidly vanishing.
The cicadas in southern Mexico are one example of an alternative food source. Collecting cicadas for food is still practiced in areas with a high historical and cultural heritage, such as the Central Valleys of Oaxaca. One case is Monte Albán, an outstanding pre-Hispanic center due to its long historical persistence, among other things (Feinman et al. 2022). Such persistence suggests enduring efficient methods for food procurement. Remarkably, the Zapotec descendants of the Monte Albán settlers still inhabit the towns near this archaeological site (Marcus 2008). Some of them collect different native species for food in the vegetation remnants that surround Monte Albán. Here, we identify the cicada they consume using DNA barcoding and morphological information and provide photographic documentation and ethnoecological data of this insect as well as of one of its hosts, which is also edible. Finally, with such information, we discuss the probable implications and benefits that the consumption of the cicada and its hosts might have in pre-Hispanic times and contrast such benefits with the problems this insect is causing to tree plantations.
Materials and Methods
The Cicadas
The cicadas (Cicadidae: Hempitera) are large insects with three ocelli, a characteristic shape, and the ability to produce sound. Their life cycle is distinctive, with an aerial and an underground phase. During the aerial phase, the males produce a loud noise to attract females. Once mated, the females lay their eggs on branches of trees or shrubs. The resulting nymphs go to the ground, reach the roots of their host plant, and feed on its roots. After several nymphal instars, which are species-dependent, the nymph makes its way out of the ground and climbs a nearby object, usually the tree host, to which it fastens with its claws. The last molt results in the emergence of an adult individual (see below and Triplehorn and Johnson 2005). For instance, in the cicada Quesada gigas, nymphs from the fifth instar emerge from the soil, and their aerial phase lasts approximately 21 months, after which time the adult arises and can fly (Kubota 2013; Sánchez-García et al. 2017, 2020). During oviposition and the nymphal stage, some cicada species may inflict severe damage to the plants they feed upon, being pests of commercial plantations (González-Ruiz et al. 2024). The taxonomy of the cicada taxa can be confusing (Sanborn 2023) and often requires molecular and phenotypic analyses (Nunes et al. 2013).
The Study Site
We conducted our field study on the seminatural landscape of the Monte Alban slopes and nearby mountains of San Bartolo Coyotepec at 1500–1800 m elevation in the Central Valleys of Oaxaca state, southern Mexico. This landscape is a unique and diverse ecosystem resulting from the interaction between natural landscape elements and human activities such as terracing in pre-Hispanic times (Marcus 2008) and agriculture and native species collecting until the present. Thus, this peculiar landscape plays a crucial role in the local bio-culture and is of significant interest to anthropology, archaeology, botany, ecology, and entomology researchers. Monte Alban is in the Central Valleys, one of the physiographic provinces of the Oaxaca state in southern Mexico. Elevated cordilleras with some peaks higher than 3000 m surround these valleys (Ortíz-Pérez, Hernández Santana, and Figueroa Mah-Eng 2004). This high elevation explains the relatively low annual precipitation in the valleys caused by rain-shadow (800 mm; UNAM 2024). The average annual temperature is semi-warm (26°C; UNAM 2024).
The flat highlands of the Central Valleys of Oaxaca, currently covered by crop fields, human settlements, and remnants of disturbed vegetation, surround scattered mountains. Monte Albán, an archeological site built atop one of these mountains, holds significant historical importance. It was an extended Mesoamerican center during pre-Hispanic times lasting 1300 years (Feinman et al. 2022). The city began to decline between 800 to 900 AD (Robles García 2004). When the Spaniards arrived in America, Monte Albán was uninhabited and covered by vegetation before archeologists discovered it.
Currently, the hills surrounding the Monte Albán and adjacent areas contain elements of native vegetation, such as grasslands, dry forests, and scrublands dominated by a wide variety of grasses intermixed with succulents, shrubs, or small trees of diverse species but dominated by native leguminous shrubs or short trees (Acaciella angustissima (Mill.) Britton & Rose, Acacia spp., Calliandra spp., Leucaena esculenta (DC.) Benth, Pithecellobium dulce (Roxb.) Benth, and Senna septemtrionalis (Viviani) Irwin & Barnebi), and planted trees such as Eucalyptus globulus Labill., Myrtaceae (R.F. del Castillo, personal observation).
Fieldwork and Ethnoecological Surveys
From 2008 to 2024, during the last molting period (March to June), we collected cicadas directly from the host trees for entomological collections and morphological and molecular analyses with three methods: the traditional employed by the locals, with a long piece of reed with a bottle on top as described in the Results section, with entomological nets, or manually, mostly in cases when the adult emerges from the last molt and the cicada is fastened to a tree trunk. Some of the insects collected were preserved in 70% alcohol or mounted for the CIIDIR Oaxaca, Instituto Politécnico Nacional Insect Entomological Collection. We also took notes and photographs of the host trees in the field and estimated the number of emerging cicadas on 10 P. dulce host trees at the CIIDIR Oaxaca, Santa Cruz Xoxocotlán campus.
The field observations related to collecting cicadas for food consumption were conducted on the slopes near the archaeological site of Monte Albán from April 2021 to May 2023. During that period, we also found people collecting pods of one of the hosts of this cicada. A total of 15 groups of people were seen collecting cicadas. We reported the collecting methods, and when possible, we also took photographs of the process. We also conducted a total of 50 informal interviews (sensu; Gerique 2006) about how people collect or prepare the cicadas in the towns of San Baltazar Yatzachi el Bajo, San Miguel Piedras, Zaachila, Santa Cruz Xoxocotlán, San Francisco Infiernillo, and Monte Albán. The hills surrounding Monte Albán are open to people, including locals and tourists. The information we collected is not exclusive or the legal property of any group of people, and the informants agree to disclose the information we gathered from them. However, their names and other personal data are not revealed to preserve their privacy.
DNA Methods
A batch of 11 specimens were collected for DNA analysis. The DNA of 10 specimens could be successfully amplified. Genomic DNA was extracted from cicadas using three or four legs. We employed the FastPrep24® homogenizer and FastPrep Kit for DNA extraction. Universal DNA barcoding primers for insects LepF1 (ATTCAACCAATCATAAAGATATTGG) and LepR1(TAAACTTCTGGATGTCCAAAAAATCA) were used to amplify partial cytochrome oxidase 1 gen (648 bp) following Primer Sets of the Canadian Centre for DNA Barcoding (CCDB) Protocols (Ivanova and Grainer 2006). We used the kit MyTaqDNA Polymerase (Bioline) for PCR with a volume reaction mixture of 25 µL that contained 15.8 μL of nuclease-free water, 1 μL (10 μM) LepF15 forward primer, 1 µl (10 μM) LepR1 reverse primer, 5 μL MyTaq Buffer reaction, 0.2 μL of MyTaq polymerase, and 2 μL of genomic DNA template (10–20 ng/μL) DNA amplification was carried out in a Veriti® thermocycler (Applied Biosystems). PCR temperature cycling programs followed Wilson (2012): initial denaturation at 94 °C for 1 min; five cycles of 94 °C for 30 s, annealing at 47 °C for 40 s, and extension at 72 °C for 1 min; followed by 34 cycles of 94 °C for 30 s, 52 °C for 40 s, and 72 °C for 1 min; with a final extension at 72 °C for 10 min, followed by indefinite hold at 4 °C. PCR products were run on 1.2% TBE agarose gels to ensure accurate visualization of the amplicon size. The observed amplicons were cleaned with the EZ-10 Spin Column PCR Products Purification Kit (Biobasic). Capillary DNA sequencing was performed in an ABI 3730xl System at the Macrogen sequencing facility (Macrogen Inc., Seoul, Korea).
BLAST Analysis
The DNA sequences were analyzed with the CodonCode Aligner v. 10.0.2 software. The alignment of individual contigs was carefully checked by eye, and Clustal Omega® was selected for DNA sequence alignment of all contigs together. A project was registered under the name “Edible cicadas” project code CIGAR (http://www.boldsystems.org) at The Barcode of Life Data System (BOLD), which is a computational workbench focused on the collection, storage, analysis, and dissemination of DNA barcode sequences (Ratnasingham and Hebert 2007). Three files were submitted to BOLD before DNA sequences. First, the Specimen data file (detailed voucher information, scientific names, and collection dates), an image file (high-quality specimen images), and finally, a trace file along with the direction and DNA sequence of the used primers. Sequences in FASTA format were uploaded to BOLD and GenBank systems. The obtained DNA sequences were analyzed with the Basic Local Alignment Search Tool for Nucleotide (BLASTN) method, which searches against the sequence database available online at the National Center for Biotechnology Information (NCBI): https://www.ncbi.nlm.nih.gov, and the BOLD database.
Results
Ecology and Ethnobiology
The cicadas are locally known as “cigarras” or “chicharras” in Central Valleys Oaxaca. These insects are collected and eaten just before the beginning of the rainy season, roughly from the last two weeks of April and the first two weeks of May. At that time, the males produced a strident noise to call for the females, the most distinctive sound of the slopes of Central Valleys
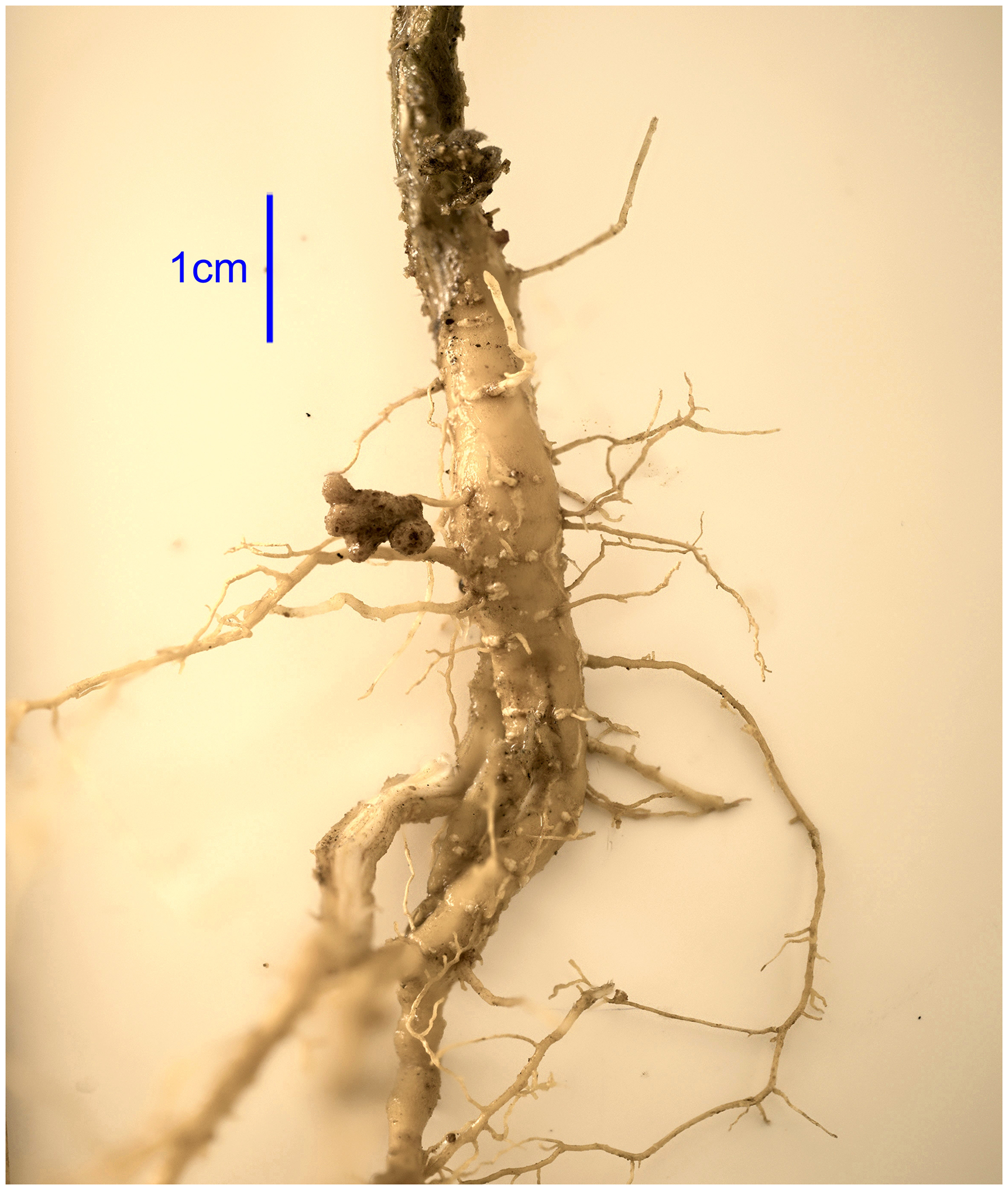
Nitrogen-Fixing Nodules in the Roots of an Unearthed guamúchil, Pithecellobium dulce Sapling, at 5 km from Monte Albán. At the Adult Stage, the guamúchil is one of the Host Trees of Quesada gigas. The Reddish Color of the Nodules is Owing to the Protein Leghemoglobin, which Helps Trap Oxygen Molecules that Inhibit the Nitrogen-Fixing Functions of Nitrogenase, the Protein that Carries out Nitrogen Fixation (Voet and Voet 2004).
The insects are usually collected by families or groups of young people (Figure 2). For this purpose, the collectors use a long piece of “carrizo” 2.5 m long or longer. The carrizo is a piece of the stem usually of the reed Phragmites australis, Cav., Trin. Ex Steud, Poaceae, a cosmopolitan plant that dominates the wetlands of North America. This species is common in brooks and streams of Central Valleys. Although reported as a native species, recent studies have suggested that the observed increase in this species in North America is due to an invasion of an introduced genotype (Saltonstall 2002).

Cicada Collecting near Monte Albán area, Central Valleys, Oaxaca, Mexico, April 30, 2023. Collecting Cicadas is a Common Activity in the Area Before the Rainy Season. Usually, this Activity is Conducted by Groups of People Including Young Children, Displaying a Labor Division. The Insert Shows a Cicada on the Trunk of the Host Leucaena esculenta. Note the Smooth and Silvery Cortex Distinctive of this Legume Tree.
A PET soda bottle, whose bottom was cut off, is attached upside down to one extreme of the reed. The bottle helps trap insects high on the trees’ trunks or branches. Although less common, other variants for trapping cicadas can also be used. One is using a reed stem again but with chewing gum put on top instead of the bottle. Chewing gum adhesivity immobilizes the insect. Cigarra collecting involves at least one person helping to localize the insect on the trees and another collecting the insect using the reed stem. Thus, collecting cigarras helps promote cooperation among the locals even at a young age and maintains the tradition of consuming local insects (Figure 2).
We found that cicadas are traditionally cooked in four different ways. (a) Toasted and fried. The cicadas are first toasted and when acquiring a golden color, the wings are easily removed in a kitchen strainer. Then, they are fried with garlic. (b) Fried. After removing the legs and the wings, the cicadas are washed with water, rinsed for 10 min, and fried in a pan with garlic and chili tree pepper (a small chili pepper that is very spicy). (c) Battered. Alive cicadas are boiled in water to keep their body soft. Then, after removing the wings and legs, the cicadas are coated with battered eggs, fried, and served with rice and beans. (d) “Salsa de chicharra” (cicada sauce). Alive cicadas are wrapped in aluminum foil and roasted with a chili tree pepper and a garlic clove, then the legs and wings are removed, and ground with a traditional basalt mortar (molcajete) with a little bit of water, salt, the chili, and the garlic clove.
We also detected that during the winter in the Monte Albán area, some locals collect the edible pods of the guaje for self-consumption, either raw or cooked (Figure 3). The guaje pods are also sold in the Central Valley markets, but we did not detect that such pods came from our study site.

Collecting Pods of the guaje, Leucaena esculenta, Near Monte Albán Area, Central Valleys, Oaxaca, Mexico, February 21, 2023.
BLAST Analysis
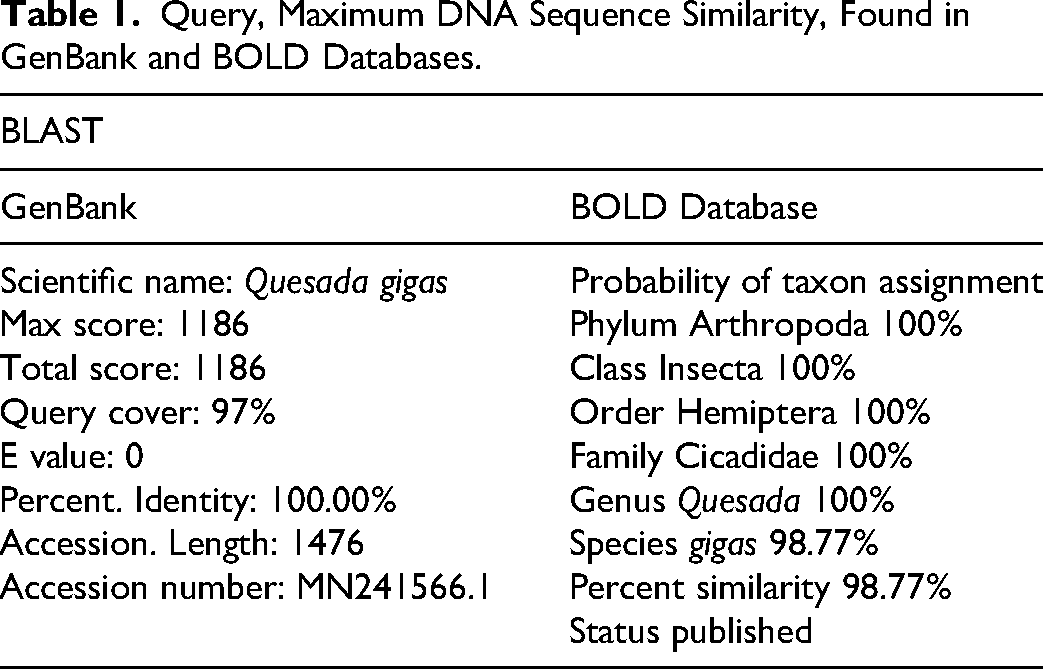
All 10 DNA sequences obtained in this study were genetically identical. Table 1 shows the detailed BLAST results observed in GenBank and BOLD. The BOLD Process ID number and GenBank accession numbers obtained are shown in Table 2. The similarities derived from comparing our DNA sequences with those published in the GenBank and BOLD indicate that Q. gigas (Oliv. 1905), is the taxon with the highest match. Indeed, GenBank data rendered 100% similar with a specimen from this taxon collected in Corrientes, Argentina, roughly 6100 km south of our collecting sites. Interestingly, our sequences gave a 100% match with the published subunits 1 and 2 of cytochrome oxidase.
Query, Maximum DNA Sequence Similarity, Found in GenBank and BOLD Databases.
DNA Libraries Formed With CO1 Sequences Obtained From 10 Cicadas of Quesada gigas from Central Valleys in Oaxaca State, México, Along With Their BOLD and GenBank Registration Numbers.

Our BOLD results displayed a 98.77% similarity from a sequence of a specimen of Q. gigas collected in Texas. A neighbor-joining tree (NL) using a distance model Kimura 2 parameter, generated at BOLD after BLAST analysis, shows our query sequence in a clade along with the morphospecies cicadidJanzen Janzen01 and 03, from Costa Rica, and Q. gigas from Venezuela.
The GenBank has four Q. gigas DNA sequences obtained with CO1 gen from Argentina (2), the USA, and Venezuela. Similarly, BOLD displays three sequences from Argentina, the USA, and Venezuela. No published DNA sequences of cicadas from Mexico were available (both databases were accessed on 27 May 2024).
Morphological Identification of Q. gigas
The original description of Q. gigas by Distant as Tympanoterpes gigas (Distant and Fowler 2005) and those of Martinelli and Zucchi (1997) in the tribe Fidicinini Amyot & Audinet-Serville, 1843 (see Marshall et al. 2018) morphologically confirm the molecular identification of the used cicada in Central Valleys. No other possible insect species in Central Valleys could match the description. Here, we provide photographs that show features of its external morphology (Figure 4). Apart from the male genitalia, which requires dissection, the lateral shape of the pronotum and the wing venation are distinctive features of this taxon (cf. Distant and Fowler 2005, Figure 9; Martinelli and Zucchi 1997, Figures 3 and 4).

Quesada gigas near Monte Albán area, Central Valleys, Oaxaca, Mexico. (A) Lateral view. (B) Dorsal View Showing Wing Venation. (C) Detail of the Dorsal Side Showing Part of the Head with the Eyes and Ocelli, the Pronotum, and Mesocutellum. (D) Anterior View of the Head Showing Compound Eyes, Ocelli, the Antenna, and Clypeus.
Discussion
A previous study of edible insects in the state of Oaxaca by Ramos-Elorduy and Pino-Moreno (2004) reports two species of Hemiptera (Homoptera): Proarna sp. and Hoplophorium monogram. Here, we document the uses and confirm the identification of another hemipteran insect, Q. gigas, in this state, particularly in the surroundings of the archaeological site of Monte Albán, using DNA barcoding and morphology. We contributed to ten new Q. gigas CO1 DNA sequences, the first published from Mexico. This species is collected in the field mainly for self-consumption and cooked in several ways. We also detected that Q. gigas is abundant; their host trees are mostly legume tree species, also common in the Oaxacan Central Valleys. We find evidence that the hosts are associated with nitrogen-fixing bacteria. With this information, we examine its significant historical and contemporary transcendence
At least four reasons can explain its popularity. First, this species is abundant when they reproduce on the days before the rainy season and is the most common cicada in the Oaxacan Central Valleys. Also, the loud noise of the males makes it easy to spot them. Second, Q. gigas has a high protein (12.6%) and lipid (8.4%) dry weight content (Aoki et al. 2011), which is significant in a region where protein sources may be limited. Thirdly, Q. gigas is one of the biggest insect species in Central Valleys (45 mm long) and the largest among other sympatric cicadas. Thus, its collection likely renders the highest protein content per trapped unit relative to other edible insects.
Finally, local people consume the seeds of one of the primary hosts of Q. gigas, the guaje, Leucaena esculenta. While Q. gigas, in its edible adult stage, is available mainly during the spring, the pods of the guaje are primarily available during the winter. Thus, in the same place, the collectors can find different protein sources at different times during the dry season. In contrast to the seeds of other legume species, the seeds of the guaje can be eaten uncooked. Human consumption of guaje seeds is still widespread in other regions with traditional Indian backgrounds (e.g., Peralta Juárez et al. 2017).
The high protein content and their ability to have symbiosis with nitrogen-fixing bacteria make legumes a system with a high potential to increase protein production sustainably (Weindl et al. 2020). Indeed, the pods of Leucaena are rich in protein (Ortiz-Domínguez et al. 2017; Zapata Campos et al. 2020). Also, we could verify the presence of nodules of N-fixing bacteria in a guamúchil, another cicada host, near the Monte Albán area (Figure 1). The nitrogen-fixing symbiosis of the leguminous host trees likely enhances protein levels in these plants, making them more suitable for the cicadas. This bacterial association might also account for the high protein levels found in the cicada. Consequently, the dietary use of cicadas and the guaje by local human communities around Monte Albán likely has strong biological foundations tied to the nutritional benefits of nitrogen-fixing bacteria symbiosis in Oaxaca's leguminous plants.
The Quesada–Leucaena System in the Past
We cannot attest that the pre-Hispanic inhabitants of Oaxaca consumed cicadas and Leucaena seeds. However, it is very likely. First, guaje trees abound in the Central Valleys of Oaxaca and probably were more abundant in the past. Indeed, the most accepted hypothesis for the etymological origin of the Oaxaca voice is from the Nahuatl language: “Huaxyácac,” which literarily means “guajes in the point of your nose,” or “wherever you turn your head, you find a guaje tree.” This interpretation was taken from a Mendocino Codex glyph, which refers to the foundation of Oaxaca city, at ∼5 km from Monte Albán, on a guaje forest (Pérez 2015). Indeed, Oaxaca City was built on a settlement named “Lushu’lá” in the Zapotec language, which means “Valley of guajes” (Pérez 2015). Furthermore, Zárate (1999) provides a detailed ethnobotanical study of the guaje and suggests that this species is in an ongoing domestication process due to its great variety of uses and cultivation practices. This process is likely pre-Hispanic, given the evidence provided by historical documents and archaeological remains found of this species in the nearby Tehuacán Valley (Zárate 1997, 2000). Thus, etymological and ethnobotanical evidence suggest close and long-lasting tights between the pre-Hispanic inhabitants of Oaxaca and the guaje.
Second, there is a close association between the guaje and the cigarra. Indeed, we found that the cigarra collection in the Monte Albán area was predominately on guaje trees (Figure 2). The abundance of these insects and their male's loudness during their mating period makes it almost impossible that the cigarras were undetected by pre-Hispanic people. The fact that the local people, many of which are of Zapotec origin, regularly collect cicadas also supports the pre-Hispanic roots of this practice and the ancient ties of contemporary Zapotecs with this insect.
Third, the domestication of maize and agricultural development undoubtedly account for the long-lasting duration of Monte Albán (Feinman et al. 2022). However, even after the agricultural phase started, the consumption of wild plants and animals should have been critical for the pre-Hispanic settlers in the Oaxacan Central valleys. These valleys are considered “marginal” for agriculture due to the relatively low rainfall and high seasonality (Marcus 2008). With elevated biological activity, obtaining food should not have been problematic during the rainy season. However, protein procurement should have been uneasy when most biological activity is halted. Thus, obtaining proteins from the Quesada–Leucaena system at different times during the dry season should have been very valuable.
The Quesada–Leucaena System: Present Times
Q. gigas, in other countries of Latin America, such as Brazil, is a severe pest of commercially cultivated species, such as the parica, Schizolobium amazonicum Huber ex Ducke (Soares et al. 2008), a planted tree used for the wood industry. Also, in this country, Q. gigas has been reported on 30 host tree species (Martinelli and Zucchi 1997), affecting economically important species, including the coffee tree (Maccagnan and Martinelli 2004; Decaro Jr. et al. 2012). In Mexico, Q. gigas is also a plague in coffee and lemon plantations (references in Sosa-Álvarez et al. 2023).
Combining these findings with our study suggests that an insect species regarded as a pest in some situations can serve as a valuable protein source for certain human populations in others. In this regard, Mexico has a long tradition of using noncultivated plants in crop fields as a food source or controlling insects (e.g., Altieri 2016). Why Q. gigas is a severe plague in some croplands remains to be explored. However, cicadas are widely consumed by various bird species (Pons 2020). Thus, we hypothesize that agricultural malpractices causing over-simplification of plantation ecosystems significantly reduce the abundance of potential predators that could control the cicadas’ population size. Contrastingly, the preservation of natural ecosystems and the human consumption of cicadas may have mitigated the insect's negative impact on its host plants, while simultaneously providing a valuable source of protein. This relationship could reflect a pre-Hispanic symbiosis between the guaje and humans, offering an example of how an insect and its host plant can be used as alternative protein sources.
Conclusions
Using DNA barcoding protocols and morphological features, we could adequately identify the edible cicada in Central Valleys of Oaxaca as Q. gigas. We also detected that several leguminous trees are the chief hosts. Q. gigas is abundant and regularly collected before the rainy season as food for self-consumption by local people in Monte Albán. The abundance of one of the primary hosts, the edible guaje, L. esculenta, its presence in archeological remains in nearby sites, and pre-Hispanic references to its abundance in the Oaxacan Central Valleys suggest that both this host and the cicadas were known and consumed before the Spaniard’s arrival. This finding once more suggests that the protein diet of pre-Hispanic people in the Oaxaca Central Valleys included a variety of sources, including insects, and points to the importance of the Quesada–Leucaena system as a high protein temporal food sources during two times of the year, probably associated with the symbiosis of the hosts with nitrogen-fixing bacteria. In some crops in Mexico and Brazil, Q. gigas is a plague. Consuming cicadas may help explain why this species is not a plague in Oaxaca and provides an alternative for controlling this insect in plantations while providing a nonconventional protein food source. The archeological site of Monte Albán and its surroundings remain a rich biocultural reservoir due to valuable native species and the practice of pre-Hispanic traditions, a heritage that we should preserve.
Footnotes
Acknowledgments
We would like to thank the informants for their patience and for sharing their valuable knowledge. Two anonymous reviewers provided useful comments that helped to improve the manuscript.
Ethical Considerations
The informants consulted verbally agreed to share their knowledge, which is considered part of the traditions but does not belong to any person or group. In all cases, the informants were kept anonymous. The fieldwork did not affect or put the studied species at risk nor adversely affected the native collectors.
Author Contributions
RFDC, STA, and JASG conceived the research. Ethnobiological surveys in the field were conducted by RFDC and STA, and those in towns by RJL. JASG collected and curated the insect collection. JASG did the morphological identification of the cicadas. STA conducted the DNA lab work and bioinformatic studies. RFDC wrote the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CONAHCyT project CBF2023-2024-3006, and Instituto Politécnico Nacional (SIP20241916, 2023164, and 20211341).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
