Abstract
Contemporary efforts to strengthen Indigenous food sovereignty can take many forms, some of which may depart from traditional food gathering practices. Common camas (Camassia quamash) is a wild-gathered geophyte that served as a staple food for Indigenous Peoples prior to colonialism. Because the abundance of camas has declined and gathering areas are difficult to access, this project sought to increase Shoshone-Bannock Tribal citizen access to camas by experimenting with ways to grow camas locally. We also held events that provided an opportunity for Tribal citizens to interact with camas. Strengthening relationships with camas in these ways not only can enhance the health of Indigenous communities because of camas’ nutritional value, but also because renewing practices and values associated with camas brings Indigenous Peoples together to redefine and assert their nationhood. Our team of authors includes Tribal- and university-affiliated professionals and students who worked collaboratively to develop this project. We evaluated how different growing conditions and seed sources influence camas cultivation. We found that soil type, stratification setting, and seed source significantly affected seed percent germination. While cold-moist stratifying seeds in a controlled setting in potting soil generally led to the highest percent germination, soil type interacted with seed source, suggesting some degree of local adaptation across gathering areas. Transplanted bulbs had high survival rates, suggesting both seeds and bulbs as viable options to increase local access to camas. This project contributes to our understanding of camas ecology while also demonstrating one approach to help strengthen traditional foodways through Tribal-university partnerships.
Introduction
Indigenous food systems play an essential role in the operation and maintenance of cultural, social, and political structures. For example, particular bands of the Northern Shoshone in western North America were named after the foods they ate such as the Agaideka’a or “eater of salmon,” or the Pasigo’deka’a or “camas eaters,” which reinforces language and social connections for the Tribe (Liljeblad 1962; Sturtevant and D’Azevedo 1986). Because of this, various tribal communities are making efforts to create stronger relationships with and improve access to traditional foods to promote the health and well-being of their members and cultures (Budowle, Arthur and Porter 2019; Coté 2016; First Nations Development Institute 2018; Kamal et al. 2015; Timler and Brown 2019). Current efforts to maintain connection to traditional foods exercise the ancestral knowledge that upholds culture, demonstrating the substantial value that strengthening access to them may have in a tribal community (Whyte 2016; Coté 2016). For example, Daigle (2019) reports that Anishinaabe women and Elders in Ontario, Canada are teaching tribal youth their ancestral knowledge of food processing and preparation that demonstrate caretaking of the land and waterways, which reinforces the continuation of the community's cultural teachings. Because many Tribes have lost access to their traditional gathering lands through colonial dispossession, contemporary efforts to strengthen Indigenous food sovereignty may depart from traditional food gathering practices and locations. For example, Tribes may need to establish new ways of growing and/or gathering traditional foods that require developing new ecological knowledge about those foods, as well as how to grow and distribute them. Because of this, cultivating traditional foods in new places is one way to renew access to them.
Study Design
The goal of the work presented in this paper is to identify the best cultivation methods for common camas (Camassia quamash) in Fort Hall, Idaho, to increase access to this culturally significant plant among Tribal citizens. The focus of the project on C. quamash, and the research methods and protocols, were jointly determined and carried out by the authors who include a Tribal citizen and employee of the Shoshone-Bannock Language and Culture Preservation Department, Nolan Brown, a Tribal citizen and student, Sidney Fellows, postdoctoral researcher, Georgia Hart-Fredeluces, and faculty member at Idaho State University, Morey Burnham. The goals of the project were identified through conversations of the authors with advising representatives from the Tribe's Department of Language and Culture Preservation, Department of Fish and Wildlife, Planning Program, and Culture Committee. Key to the formation of this research partnership was the fact that this project fit within the priorities of the Tribal Practices for Wellness in Indian Country grant and with the interests of the broader tribal community, while also fulfilling the interests and undergraduate research requirements for the lead author. The authors and advising representatives have a shared interest in helping to develop protocols for growing camas in Fort Hall, and in understanding how and why the C. quamash plants at two frequently-visited gathering areas varied in terms of ecological and morphological characteristics. Despite being classified as the same subspecies of C. quamash by Gould (1942), characteristics such as bulb size and ease of harvesting are different among camas at these two traditional gathering locations. Given that no recent genetic or taxonomic work has addressed differences in the camas populations at these two gathering areas, and that genetic work on Camassia spp. is very limited (Uyeda and Kephart 2006), it is unclear how relocating plants from these locations to Fort Hall may influence the genetic diversity of the species or change the genetic composition of the few plants that do already occur on the Fort Hall Indian Reservation. Understanding basic differences in performance of seeds and bulb transplants is therefore a first step towards understanding these potential differences as well as the conservation implications of a cultivation and distribution program. To address these needs, we developed a set of seed germination and bulb transplant experiments with treatments that reflected accessible growing conditions to Tribal citizens, and which also could compare and contrast seeds and bulbs sourced from two key traditional gathering areas.
Background
Camas Ecology and Distribution
Camassia quamash (Asparagaceae), or common camas, is an ecologically important perennial herb that occurs from British Columbia to northwestern California, and eastward to Montana and Nevada (Stevens, Darris and Lambert 2000; Turner and Kuhnlein 1983). Camas populations grow in seasonally wet areas, such as floodplains and wet meadows, that usually have a dry period (Gritzner 1994; Leffingwell 1930). The plant may reproduce sexually through seed as well as asexually by offsets around the mother bulb (Genders 1973; Thoms 1989). Thoms (1989) reports that the annual growth cycle of a mature C. quamash represents three generations: a decaying mother bulb, a growing daughter bulb, and a new terminal inflorescence. Camas seeds require cold-moist stratification, or subjecting seeds to cold and moist conditions over a period of time, in order to germinate (Beckwith 2004; Guerrant and Raven 1995). Though published work on camas ecology is sparse, Camassia spp. bulbs are consumed by bears and rodents (Thoms 1989), while leaves are an important source of food for deer, elk, small mammals, and herbivorous insects (Stevens, Darris and Lambert 2000; Stanley, Dunwiddie and Kaye 2011; Thoms 1989). Camas flowers provide resources for native bees including bumblebees (Parachnowitsch and Elle 2005; pers. observ. GHF 2020 and 2021), and the endangered Fender's butterfly (Schultz 2001). Camas seeds are also consumed by water birds (T. Gregory, pers. comm. 2020) while other birds may build their nests among the plant leaves (GHF person. observes.).
Traditional Camas use
Camassia spp., or camas, is perhaps one of the most important and widely consumed traditional foods among Indigenous Peoples in western North America and is commonly gathered, stewarded, and consumed by Indigenous Peoples today (Evans 2017; Lyons and Ritchie 2017; Whyte 2018). The Shoshone and Bannock, who call themselves the Newe, lived and migrated throughout much of the Great Basin, the Columbia Plateau region, Idaho Snake River Plain and the central Idaho mountains and eastward over the Rocky Mountains for thousands of years prior to European settlement (Evans 2017; Tuell 2013). Since time immemorial, the Newe have traveled seasonally to Camas Prairie Centennial Marsh in south-central Idaho and surrounding areas to harvest from abundant fields of common camas, Camassia quamash, a tradition that is upheld by tribal people today (Anderson 2005; Evans 2017; Thoms 1989; Statham 1981) (Figure 1). Common camas served as a staple food or a principal source of carbohydrates for the Newe (Statham 1981), as it did for other Indigenous Peoples in western North America (Kramer 2000, Gritzner 1994).

Derek No-Sun Brown, Sweet Breeze Through the Camas, 2020, oil on canvas, courtesy of the artist.
Using traditional digging sticks and modern tools, Indigenous groups gather Camassia spp. bulbs from late spring to late fall depending on Tribal or family practice, soil moisture, and/or when the flowers begin to senesce (Gritzner 1994; Beckwith 2004; Thoms 1989). The Shoshone-Bannock in central Idaho currently harvest in early summer. Bulbs were historically cooked in earth ovens for one to three days and often pounded into cakes to be dried and stored (Beckwith 2004; pers. comm. N. Brown, 2023). Today, a wider range of cooking approaches are used to bake camas, including slow cookers. In addition to gathering bulbs, Indigenous Peoples cared for, and continue to care for, camas patches through harvest itself, replanting of small bulbs, weeding of surrounding vegetation, applying natural fertilizers, and burning (Gritzner 1994; Sultany, Kephart and Eilers 2007; Weddell n.d.), and such practices have been shown to have positive impacts on camas populations (Stucki, Rodhouse and Reuter 2021). Historical camas management strategies, such as selective harvesting and replanting, speak to Indigenous peoples’ cultivation of the plant for millennia (Carney et al. 2021; Lepofsky and Lertzman 2008).
Postcolonial Suppression
The plant's time-honored traditional use and caretaking, along with the general use of a variety of traditional food practices among Indigenous Peoples, began drastically declining at the onset of settler colonialism (Anderson 2005; Evans 2017; Pevar 2004; Renick and Henthorne 2019). Colonial policy established numerous treaties that directly affected Indigenous peoples, and, in particular, their use of culturally important plants, including camas. The 1868 Treaty of Fort Bridger established today's Fort Hall Indian Reservation and forcibly relocated Shoshone and Bannock peoples within its boundaries. The government did not honor the treaty's guarantee of reserving Camas Prairie for the Tribe's permanent homeland by failing to include it as part of the reservation (Smoak 2004), and by failing to ensure the Camas Prairie was protected from White settlement. Tribal rights and access to the prairie were hindered by White settlement and related agricultural practices that degraded and reduced the camas plant population (Smoak 2004, 2008). In the following years, the government continually failed and refused to uphold the various treaty obligations that ensured Fort Hall Indian Reservation residents’ food supplies and other necessities. The unlivable circumstances on the reservation, including the lack of possibilities for hunting or gathering, caused rampant starvation (Gould and Loether 2002; Madsen 1980). Lack of food was a major cause for the Bannock War of 1878 (Gould and Loether 2002; Madsen 1980). In response to their access to hunting and gathering areas being removed and disrupted by European agriculture and the government's failure to provide alternative food sources, the Bannock initiated the war by killing cattle ranchers. The war continued between the Tribe and their Paiute allies, and the U.S. Army and volunteer troops through the summer. U.S. government officials at the time determined that food deprivation caused by the Tribes’ displacement and denial of access to their regular food sources, namely Camas Prairie and its bulbs, caused the war, stating the situation was “war path or starvation” (Madsen 1980:85).
Colonial tactics, such as restricting access to traditional harvesting areas as described above, worked to break Indigenous food systems and replace Indigenous ways of life through what Grey and Patel refer to as the “civilizing forces of modernity” (Grey and Patel 2015; Shanks et al. 2016; Vantrease 2013; Whyte 2016). These tactics led many into a dependence on U.S. government food rations. Some Tribal people undertook Euro-American agriculture as such work was promoted for newly placed Tribal members on the Fort Hall Indian Reservation. And although the U.S. government often broke their treaty obligations and provided inadequate supplies, a portion of families successfully fed themselves (Madsen 1980). These early days of food rations and assimilation into Euro-American agriculture marked a significant transition in the Indigenous diet: one primarily made up of culturally inappropriate foods, often with low nutritional value (Chino, Haff and Francis 2009; Shanks et al. 2016; Vantrease 2013; Wishart 2011). This transition into a diet of foreign foods also marked an interference in or degradation of the peoples’ quality of cultural integrity, such as loss of language that was directly tied to the use of traditional foods (Whyte 2018).
Camas Food Sovereignty
Indigenous food sovereignty is a strategic process whereby certain foods are revived to motivate a group of people to come together to strengthen or uphold communal structures (Whyte 2016). Many tribal nations across the U.S. and Canada have developed programs to revitalize their food system traditions by growing their own foods (traditional and non-traditional), thereby reestablishing intergenerational sharing (e.g., of nutritive and medicinal plant knowledge) and related food practices (Budowle, Arthur and Porter 2019; Coté 2016; First Nations Development Institute 2018; Kamal et al. 2015; Timler and Brown 2019). Convening via traditional foods, Indigenous communities may be motivated to take an action toward increasing individual and collective well-being by addressing multiple, intersecting challenges related to health, culture, education, and politics (Whyte 2016). The renewal of traditional foods also encourages an Indigenous collective's capacity to negotiate imposed colonialism (e.g., forced dependence on governmental rations) by creating an opportunity to return to the ancestral food practices, and related practices, that some argue to be the very fabric of Indigenous community (Coté 2016; Grey and Patel 2015; Kamal et al. 2015; Vantrease 2013; Whyte 2016).
Contemporary Food Revitalization
By identifying camas cultivation methods for Fort Hall, Idaho through this project, Tribal citizens may have increased access to the plant and to the cultural practices associated with it. The Shoshone-Bannock Language and Culture Preservation Department (LCPD) supports the development of Tribal scientists and projects intended for the restoration and enhancement of available plants and the associated natural and cultural resource areas. Today, multiple efforts are underway, and have been for decades, to revive and strengthen connections to traditional foodways as a means to revitalize Tribal community health and support self-determination. For example, the LCPD recently received a three-year grant in 2018 from the U.S Center For Disease Control and Prevention (CDC), titled “Tribal Practices for Wellness in Indian Country” (TPWIC). The LCPD works to establish healthy living practices in the Fort Hall community through cultural education, cultural preservation, cultural research, and language preservation efforts (Shoshone-Bannock Tribes 2020). In the TPWIC grant, both traditional and contemporary Shoshone and Bannock cultural practices were the framework from which the grant strategies for activities and events were structured. TPWIC funds allowed the LCPD to consult with tribal cultural experts, hire grant event staff, create teaching aides & publications, host new cultural education classes, conduct community discussion forums, take community members on seasonal trips for hunting wild game and gathering traditional plants, distribute meals including traditional foods, assist community gardening, and distribute traditional foods seedlings or seeds and garden bed boxes. The grant supplemented the yearly LCPD and SBT Culture Committee cultural events, including the Camas Prairie Homecoming, an event begun by Tribal Elders to return community members to the Camas Prairie area to harvest camas and celebrate homecoming. In doing so, the Shoshone-Bannock Tribes are exercising their tribal treaty-reserved rights to hunt and gather upon the unoccupied lands of the United States.
In addition to supporting the specific needs of the Fort Hall community in developing a camas cultivation program, this project also contributes to a broader understanding of trait diversity across camas populations. Several studies on the camas plant explore the plant's importance as a staple and cultural food to various Indigenous tribes (Anderson 1997, 2005; Beckwith 2004; Gritzner 1994; Thoms 1989; Turner and Kuhnlein 1983). Others have investigated Indigenous management of the plant in order to identify practices that increase plant survival and abundance (Anderson 1997, 2005; Beckwith 2004; Matthews 2020; Stucki 2018). However, few studies have looked at the plant's general ecology or how to cultivate it. If camas is to be grown at Fort Hall to help support food sovereignty strategies within the community, a basic understanding of the best practices for cultivation are needed, as well as a focus on experimental treatments accessible to Tribal citizens for the plant's cultivation, including soil types and cold-moist stratification settings.
Therefore, in this paper, we ask the following questions necessary to strengthen access to camas for the Shoshone-Bannock Tribes:
How does seed percent germination vary across locally-accessible soil types and stratification approaches, and how does performance vary for seeds sourced from two gathering locations? What differences do we find in the survival and germination of seeds and bulbs sourced from two different gathering locations?
Methods
Partnership Approach
The research questions, focal species, and goals of this project were decided based on ongoing communication and interaction between the authors, the LCPD, and broader Fort Hall departments and employees. Various meetings and events were conducted throughout the project between the authors and advising representatives to continuously align the project's objectives to that of the Tribe's TPWIC grant. Prior to beginning our project, we sought and obtained formal approval from the Fort Hall Business Council through a resolution (FHBC-2020-0194) and informal approval from the LCPD's Culture Committee through a presentation that outlined the project. Toward the end of the project, the LCPD reflected on the importance of the first author reaching out to them for partnership prior to establishing the aims of the project, and for following the formal and informal processes necessary in any research project taking place at Fort Hall or with the Tribal community. Such processes represent the proper protocols necessary for the researchers to be responsive to the Tribe's needs. By following these protocols, the research authors and advising representatives are positioned to create and/or strengthen their relationship to one another.
Study Sites
We conducted our research at two study sites chosen because they are both traditional and current gathering areas for the Shoshone and Bannock Peoples that have notable differences in the phenology, bulb size, and ease of harvest (Figure 2). The first, Grays Lake National Wildlife Refuge in southeastern Idaho, managed by the U.S. Fish and Wildlife Service, is a bulrush marsh dominated by Scirpus americanus at an elevation of 1950 meters. It is an approximately 1.5 h drive east from Fort Hall, Idaho. The annual average precipitation is 38 centimeters with average summer temperatures at 13°C and average winter temperatures at 1.5°C (Walker 2022). Soils are primarily of the Grayslake-Tepete complex, which are loamy peat soils. Camas plants here have a later phenology compared to our second site and bulbs are generally smaller. Gathering bulbs here is somewhat more challenging due to a thicker root mass of surrounding vegetation at the harvest locations, including non-native Alopecurus spp.
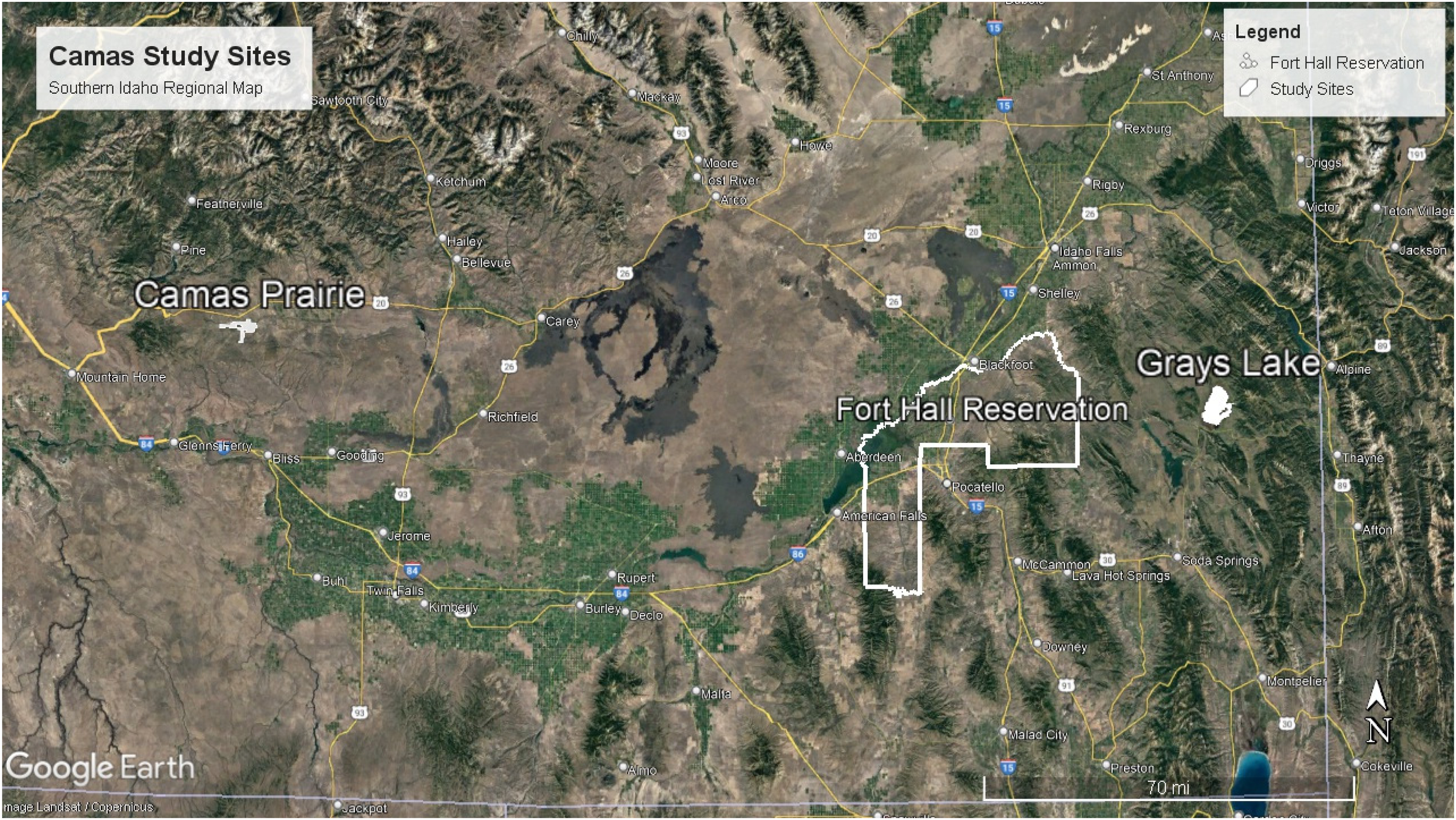
Map of study sites, Camas Prairie Centennial Marsh and Grays Lake Wildlife Refuge, in relation to the Fort Hall Indian Reservation.
The second site, Camas Prairie Centennial Marsh in southern Idaho, is a seasonally-flooded wet prairie at 1535 meters elevation (Idaho Fish and Game n.d.). It is managed by Idaho Fish and Game and is an approximately 3-h drive west from Fort Hall, Idaho. The Prairies’ average temperatures are −6°C in winter and 17°C in the summer with an annual precipitation of 40 centimeters (Gregory 2014). Soils are primarily Houk silty clay loam. Camas Prairie is primarily classified as palustrine, persistent, emergent, seasonally flooded wetland (Cowardin et al. 1979) dominated by common spikerush (Eleocharis palustris), Nebraska sedge (Carex nebrascensis), Baltic rush (Juncus balticus), common camas (Camassia quamash) and nodding groundsel (Senecio biglovii) (Gregory 2014). Non-native Alopecurus arundinaceus is common at the site, but at the gathering area grows in isolated patches rather than intermixed with the C. quamash plants as occurs with non-native Alopecurus spp. at Grays Lake.
Seed Collection
To assess the percent germination of camas seeds across two soil types and by two types of cold moist stratification, we collected seeds from haphazardly-selected plants spaced at least 20 feet apart at each study site. We collected seeds from nine plants within each of two subsites at each study site for a total of 18 plants per site. We collected seeds at Camas Prairie Centennial Marsh on July 27, 2020 and at Grays Lake Wildlife Refuge on August 7, 2020, as Grays Lake camas plants typically set seed a few weeks later than those at Camas Prairie (Figure 3A and B). We selected plants for collections if their seed capsules were dry and partially dehisced. A minimum of 60 seeds per plant were harvested and placed into a coin envelope. We only collected apparently good seeds by avoiding discolored or misshapen seeds, and then stored seeds in coin envelopes in a refrigerator until we used them in experiments. Individual plants were included in our logistic regression models as random effects (see Data analysis below). Maternal factors including plant size are known to impact seed size (Sundaresan 2005, Li and Li 2015; Biere 1991) and therefore emergence and germination rates (Andersson 1996). Therefore, to account for variance in seed germination explained by differences in individual plant size, for each plant that we collected seed from, we measured its basal diameter and flower stalk basal diameter.

(A) Camas seeds from Camas Prairie Centennial Marsh on July 27th, 2020. (B) Camas seeds from Grays Lake Wildlife Refuge on August 7th, 2020. (C) A harvested camas bulb at Camas Prairie Centennial Marsh on June 6th, 2020. (D) Shoshone-Bannock Tribes’ Language and Culture Preservation Department staff and tribal members prepare to harvest pasigo (camas) and pihaguyu (sweet tobacco root) at Grays Lake Wildlife Refuge, July 2021. Photo credit: Nolan Brown.
Seed Germination Experiments
We assessed seed germination in two soil types: commercially-available potting soil (Fertilome Seed and Cutting Mix) and soil collected in Fort Hall, Idaho because these are the two soil types most accessible for a camas cultivation program in Fort Hall, as they can be purchased or sourced locally (Figure 4). The Fort Hall soil type used for this experiment was Declo loam (USDA NRCS, n.d.). The subsites at Camas Prairie Centennial Marsh and Grays Lake National Wildlife Refuge where camas seeds for this experiment were harvested have soils characterized as loam and silty clay loam or silt loam (USDA NRCS, n.d.; Table 1). We also assessed seed germination between two cold-moist stratification methods: outdoor stratification over the winter (“outdoor” method) and artificial stratification in a refrigerator (“indoor” method) (Figure 5). These experimental conditions were chosen to reflect accessible and realistic conditions that might be used to grow camas in Fort Hall to increase access to camas among the Tribal community. Seeds collected from a given plant were distributed evenly across all experimental treatments and were also distributed evenly across potting containers to allow us to disentangle plant-specific or container-specific effects on germination from those resulting from our experimental treatments. Given limited refrigerator space, seeds from Camas Prairie (n = 432) were sown for indoor stratification on August 4, 2020, while those from Grays Lake (n = 432) were subsequently sown indoors on October 17, 2020 and outdoors on October 31, 2020. While we replicated our experimental design for seeds from each study site, due to wind and animal disturbances, as we explain below, we did not include outdoor-stratified Camas Prairie seeds in our analyses.

Experimental design for seed germination experiment, corresponding to Table 2, showing the number of seeds from each seed source in each treatment combination. Seeds collected from a given plant were distributed evenly across treatments and across potting containers within treatments. Local soil is Declo loam sourced from Fort Hall, Idaho.
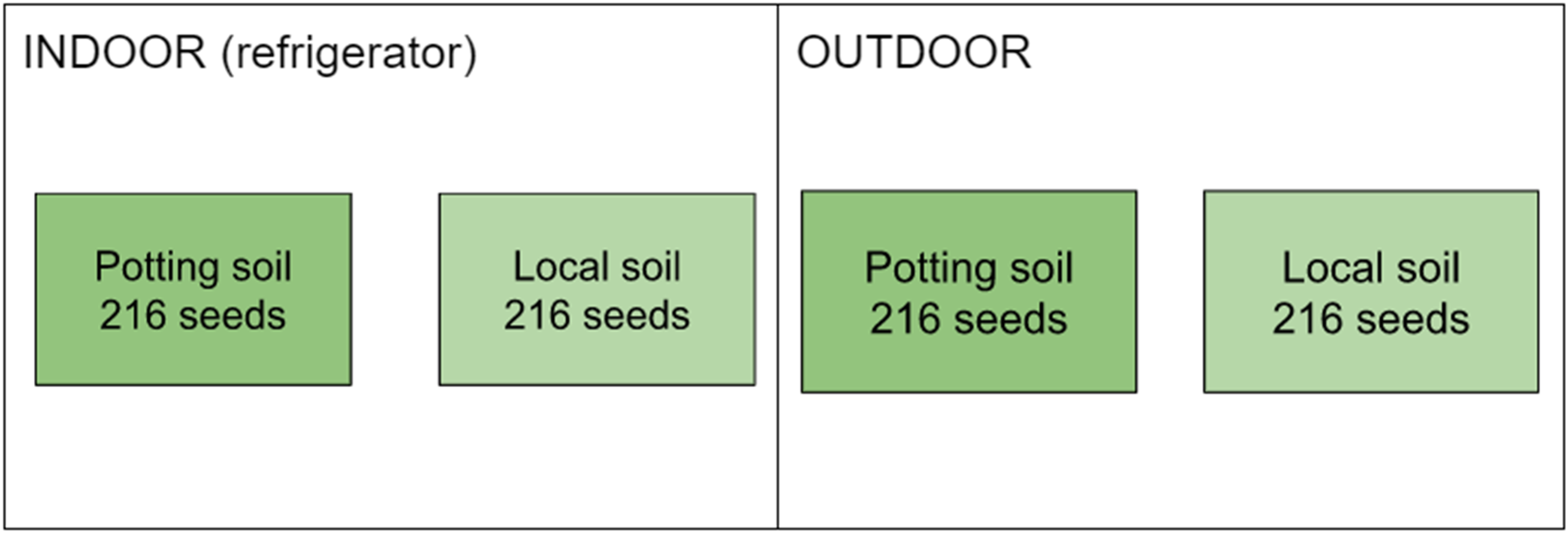
Experimental design for seed germination experiment, corresponding to Table 3, showing the number of seeds from Grays Lake in each treatment combination. Seeds collected from a given plant were distributed evenly across treatments and across potting containers within treatments. Local soil is Declo loam sourced from Fort Hall, Idaho.
Soil Types at Seed Collection Locations and for the Soil Collection Location in Fort Hall, Idaho.

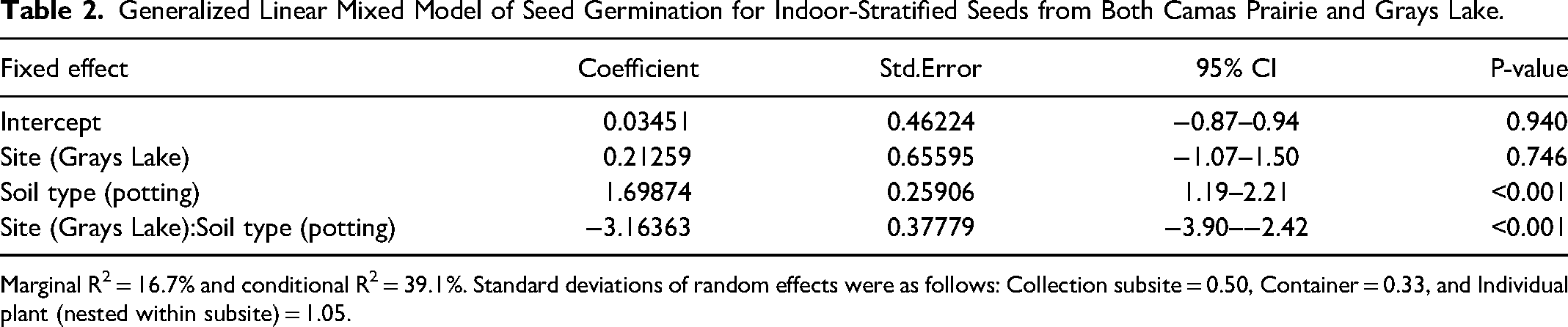
Generalized Linear Mixed Model of Seed Germination for Indoor-Stratified Seeds from Both Camas Prairie and Grays Lake.
Marginal R2 = 16.7% and conditional R2 = 39.1%. Standard deviations of random effects were as follows: Collection subsite = 0.50, Container = 0.33, and Individual plant (nested within subsite) = 1.05.
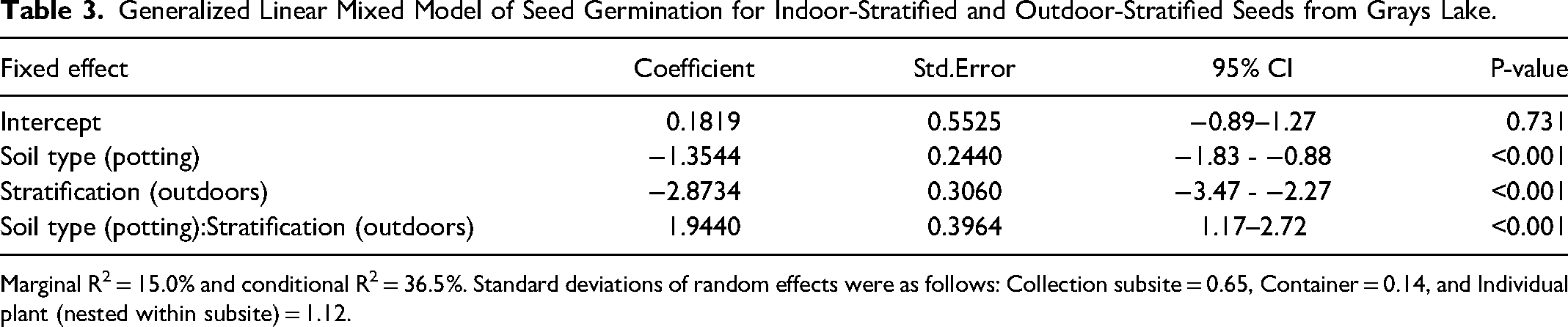
Generalized Linear Mixed Model of Seed Germination for Indoor-Stratified and Outdoor-Stratified Seeds from Grays Lake.
Marginal R2 = 15.0% and conditional R2 = 36.5%. Standard deviations of random effects were as follows: Collection subsite = 0.65, Container = 0.14, and Individual plant (nested within subsite) = 1.12.
For all experiments, seeds were sown at 1/8th inch depth in 2.5 × 2.5-inch containers with drainage holes. Each container received nine seeds. Each seed was evenly spaced within the container with three rows of three seeds each. The corner of the container was marked with a cocktail pick to track germination of individual seeds. The containers were placed in 10 × 10 × 2.44-inch trays to facilitate maintenance of a wet environment. For refrigerator-stratified seeds, cold moist stratification was accomplished by maintaining a half-inch of water in the bottom of the trays while in the refrigerator. The trays were also loosely covered in a plastic bag to help maintain a moist environment. The soil surface was wetted with a spray bottle whenever dry. Refrigerator temperatures fluctuated between 34 and 40oF during the stratification period. Seeds were removed from the refrigerator after 76 days (Camas Prairie seed) or 90 days (Grays Lake seed), both near the upper range of time under cold-moist stratification known to be required by C. quamash for germination, which is reported to be 42–100 days (Beckwith 2004; Guerrant and Raven 1995). The locations of each container in the refrigerator were rotated weekly. After the refrigerator cold-moist stratification period, the containers were transferred to a countertop approximately 60–70oF near a south-facing window. Their germination was assessed until it ceased, at approximately one week, one month, and two-months post removal from the refrigerator. Germination was defined as emergence of the seedling from the soil. Seedlings were transplanted and placed in a greenhouse to support their further propagation for eventual distribution to the community.
For the outdoor-stratified seeds, which only included seeds from Grays Lake because Camas Prairie outdoor seeds were lost due to wind and animal disturbance to the potting containers, the soil was watered at their initial planting on October 31st 2020 and again, beginning in April, when the snow melted and soil began drying out. Watering was intended to match seasonal soil water characteristics known to support camas persistence (Matthews 2020). The containers were kept moist through the spring until germination ceased, although it is possible that sporadic drying-out may have occurred. The potting containers were placed in a sunny location in the Bannock Creek District of the Fort Hall Indian Reservation. In response to the disturbance to the Camas Prairie outdoor seed experiment, a chicken-wire fence with a removable top that allowed sun exposure was built to enclose the seed area for the Grays Lake seeds to prevent animal disturbance to the experiment.
Bulb Transplants
On June 9th, 2020, 30 mature camas bulbs were harvested with in-situ soil from Camas Prairie Centennial Marsh and placed in large buckets for transport to Fort Hall, ID (Figure 3C). On June 24th, 2020, 30 mature camas bulbs were harvested from Grays Lake Wildlife Refuge. The plants were harvested after they had flowered using contemporary digging sticks made of metal and we selected plants for harvest that had either greater stem thickness or greater leaf number compared to surrounding plants.
We prepared two plastic kiddie pools with 45-inch diameters for bulb transplants by creating drainage holes in their bottom surface. We placed the pools on top of 2 inches of gravel to support water drainage. One pool was filled with SunGro Black Gold Natural & Organic Potting Soil + Fertilizer and the other was filled with local Fort Hall soil (Declo loam). Thirty camas bulbs from each of the study sites were divided between two treatment pools and each plant was tagged with a metal tag and stake. We recorded the number of bulbs that produced aboveground shoots the following spring of 2021. In the spring, the pools were watered each time the soil surface became dry until the plant leaves senesced in mid-July. Of the 60 original bulb transplants, 11 tags were lost, leading to a lower sample size for some treatments as described in the results.
Data Analysis
We analyzed the germination data from this study using two separate generalized linear mixed models that included different sections of our data and different predictors. The first model included only the indoor-stratified seeds and explored the effects of soil type, seed source location, and their interaction on seed germination. Given the loss of outdoor-stratified seeds from Camas Prairie to wind and animal disturbance, in our second model, we analyzed indoor and outdoor-stratified seeds from Grays Lake only. For this second model, we explored the influence of soil type (potting or Fort Hall), stratification approach (indoor or outdoor), and their interaction on seed germination. Both sets of data were analyzed using a generalized linear mixed effects model with a binomial error structure built with the lme4 package (Bates et al. 2015) in R (R Core Team 2020). Germination success was the dependent variable for both models. Mixed models contain both fixed (treatment) and random (random variable) effects. A mixed model approach is appropriate for this study to account for the non-independence or hierarchical structure of the samples. For example, seeds collected from a particular subsite might germinate at a higher or lower rate based on the conditions of that subsite in addition to being influenced by our experimental conditions (i.e., stratification type, soil type). The mixed model approach allowed us to account for such random effects (subsite, container, individual plant), while simultaneously estimating our fixed effects of interest: soil type, stratification approach, and seed source. As seeds from each plant were spread evenly across treatments and containers, random effects for both models included container crossed with plants nested within subsites. We assessed the pseudo R2 of our models using the MuMIn package and delta estimates were reported when available (Barton 2020). We report Wald 95% confidence intervals for fixed effects. We also explored the relationship of plant basal diameter and flower stalk basal diameter to percent germination by building linear models using the ‘stats’ package in base R, to predict average percent germination at the plant level from plant basal diameter or plant flower stalk basal diameter.
Results
Seed Source Location by Soil Type
For indoor-stratified seeds, the effect of soil type on germination depended on seed source (P < 0.0001, Table 2, Figure 6). Camas Prairie seeds achieved a higher percent germination in potting soil, while Grays Lake seed had a higher percent germination in the local Fort Hall soil (Declo loam). More specifically, of the seeds that were indoor-stratified, 82.4% from Camas Prairie germinated in potting soil compared to 50.9% in local Fort Hall soil. In contrast, 30.2% from Grays Lake germinated in potting soil, while 54.2% germinated in Fort Hall soil. The fixed effects in this model explained 16.7% of variation in percent germination, while fixed and random effects combined explained 39.1%. In terms of random effects, germination varied about twice as much by individual plant as by collection subsite, and was much less impacted by the container where a seed was planted (Table 2).
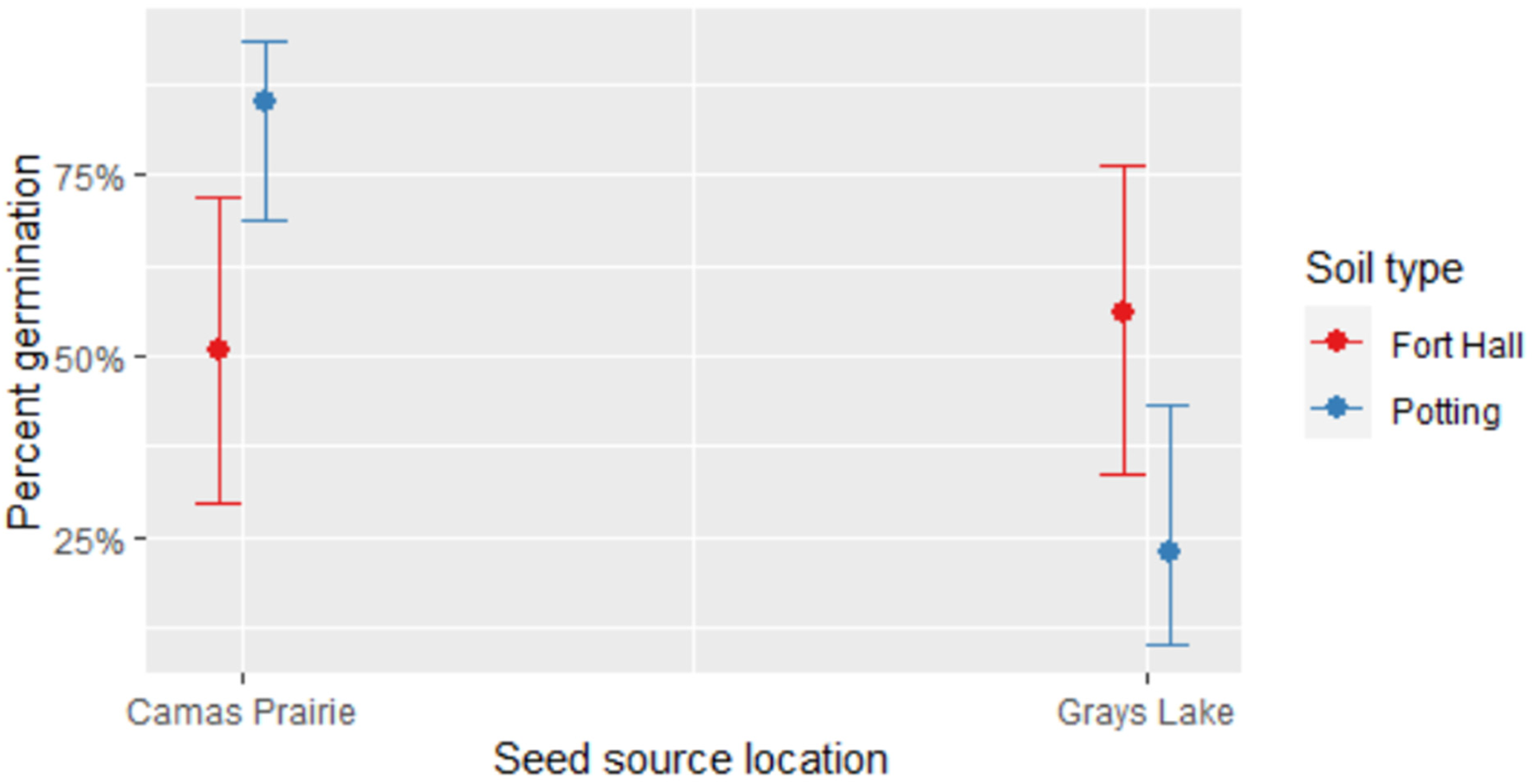
Comparison of percent germination for indoor-stratified C. quamash seed in Fort Hall or potting soil sourced from Camas Prairie or Grays Lake. Shown are 95% confidence intervals.
Stratification Approach by Soil Type
For seeds sourced from Grays Lake, we found that indoor stratification led to higher percent germination than outdoor stratification, and that the difference in the effectiveness of these two stratification approaches was more pronounced for Fort Hall soil compared to potting soil (P < 0.0001, Table 3, Figure 7). More specifically, 54.2% of indoor-stratified seeds in Fort Hall soil germinated, while 30.1% of those in potting soil germinated. For outdoor stratified seed, 10.7% germinated in Fort Hall soil and 16.7% in potting soil. When seeds from Gray's Lake in both soil types were combined for analysis, 41.7% of seed germinated after indoor stratification compared to 13.7% after outdoor stratification. When seeds from both stratification approaches were combined for analysis, 32.4% of seeds germinated in Fort Hall soil compared to 23.3% in potting soil. The fixed effects in our model explained 14.9% of the variation in percent germination, while fixed and random effects together explained 36.9% of the variation. In terms of random effects, germination varied almost twice as much by individual plant as by collection subsite, and was much less impacted by the container where a seed was planted (Table 3).
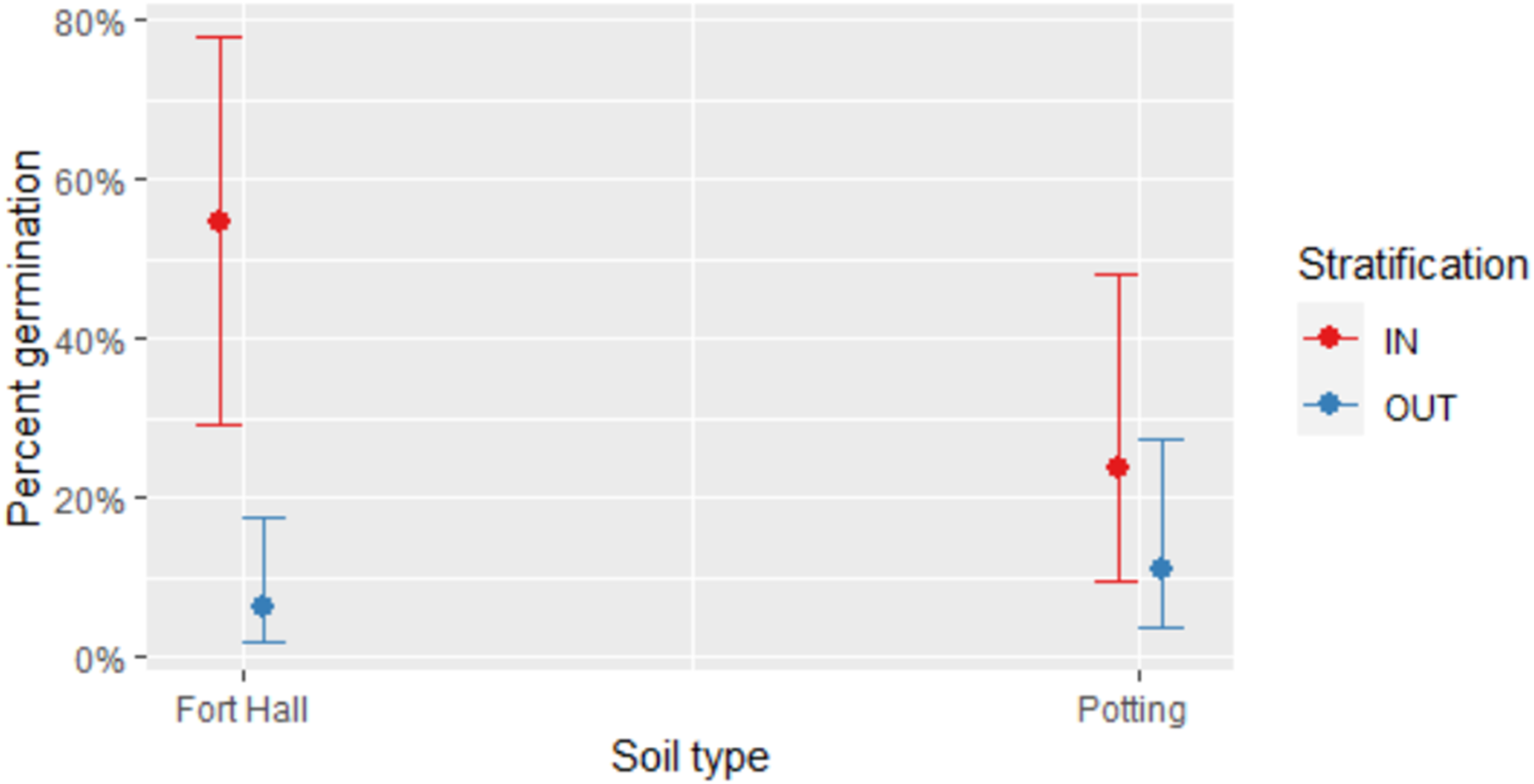
Comparison of percent germination for C. quamash seed sourced from Grays Lake in Fort Hall or potting soil sourced and after indoor or outdoor stratification. Shown are 95% confidence intervals.
Plant Size and Percent Germination
Percent germination at the plant level (36 plants, 24 seeds per plant) for indoor-stratified seeds was not related to the basal diameter of the plant (p = 0.49), nor to the basal diameter of the flower stalk (p = 0.14). Percent germination at the plant level for Grays Lake seeds (18 plants, 24 seeds per plant) was not explained by the basal diameter of the plant (p = 0.31), but was related to the basal diameter of the flower stalk (P < 0.001). For plants at Grays Lake, those with larger flower stalks had significantly lower percent germination.
Bulb Transplants
Of the 30 Grays Lake camas bulb transplants, 93.3% of the bulbs produced new leaf growth during the spring that followed the transplant. In potting soil, 86.6% (13 out of 15) of bulbs produced new leaves while 100% (15 out of 15) of bulbs in Fort Hall soil produced new leaves. For Camas Prairie, 94.7% of bulbs produced new leaf growth the following spring. Transplants in potting soil produced new above-ground growth at 92.9% (13 out of 14) and Fort Hall soil had 100% (5 out of 5) new growth.
Discussion
To increase access to camas (Camassia quamash) on the Fort Hall Indian Reservation, our project compared various methods for camas cultivation that are both accessible and practical approaches for a food sovereignty program on the reservation. We tested seed germination and bulb transplant survival in local Fort Hall soil and commercial potting soil because these are accessible options for a cultivation program in Fort Hall. We found that soil type, stratification approach, and seed source all led to significantly different rates of camas seed germination and that the effects of soil type on germination varied by the location where seeds were gathered, as well as by stratification approach. After indoor (refrigerator) stratification, seeds collected from Camas Prairie exhibited a higher percent germination in potting soil while Grays Lake seeds had higher percent germination in Fort Hall soil. These differences could be due to local adaptation of plants across these two sites, or may be attributed to annual variation in seed quality, or other factors and further study would be needed to investigate potential local adaptation. Compared with outdoor stratification, seeds from Grays Lake germinated at significantly higher rates after indoor stratification, and this effect was much more pronounced for seeds in Fort Hall soil compared to potting soil. Bulb transplants from both study sites survived and emerged at a high rate in the spring following translocation into both Fort Hall and potting soil.
Seed Source Location and Soil Type
Camas Prairie seeds generally had a higher average percent germination than Grays Lake seed, which could be attributed to their different seed traits. At the time of collection, Camas Prairie seeds were larger than Grays Lake seeds and more wrinkled (Figure 3A and B). Seed mass is an important plant trait with implications for plant community ecological and evolution. In general, and consistent with our results, smaller seeds tend not to germinate as well as larger seeds (Gamiely et al. 1990; Hendrix 1984; Leishman and Westoby 1994). Further, some seeds require a drying out period before germinating (Deno 1993) and the more wrinkled appearance of Camas Prairie seed may indicate that those seeds had a better opportunity to meet this requirement than Grays Lake seed. Finally, the fact that Grays Lake seeds were stored longer (71 days for indoor planting and 85 days for outdoor planting) than Camas Prairie seeds (8 days) before being sown into experimental treatments may also explain why Grays Lake seeds germinated at significantly lower rates. All seeds lose viability over time in storage, but the rate of this decline is highly variable (Deno 1993). We are not aware of any studies that have tested the change in seed viability of Camassia spp. with storage, though Davis (2018) indicates that Camassia spp. seed can maintain viability over several years of proper storage. Our approach of collecting only apparently “good” seeds, avoiding overly wrinkled or discolored seeds, likely increased germination rates.
Seeds from the Camas Prairie study site and Grays Lake study site germinated at their highest percentage in different soil types. This interaction of seed source with soil type is difficult to explain, but could be due to local adaptations of the seeds at each site to different germination conditions, or to other factors. Different soil types have been shown to affect seed percent germination (Baskin and Baskin 1998) in part because of differences in soil moisture (Fay and Schultz 2009; Wenk and Dawson 2007). However, the soils at the two Grays Lake locations and two Camas Prairie locations where seeds were collected are very similar in texture and therefore water-holding capacity and other physical properties (Table 1), meaning that it is unlikely that seeds from either location were more adapted to Fort Hall Declo loam soil based on texture. However, different soils also contain different microorganisms, some of which produce plant hormones that regulate seed germination, such as gibberellins (Deno 1993; Miransari and Smith 2014). It is possible that microorganisms in Fort Hall soil differentially influenced seeds from each location based on genetic or other differences between the seeds from each study site. In other words, plant hormones or other substances in the Fort Hall soil may have been more beneficial to the germination of Grays Lake seeds than Camas Prairie seeds. Reciprocal transplant experiments would be necessary to test for the possibility of local adaptation (e.g., Nagy and Rice 1997). Other studies have pointed to the importance of soil pH (Johnson 2000; Statham 1981) and soil drainage (Statham 1981) in relation to camas occurrence, meaning these factors could be investigated in future cultivation-related studies.
In support of the idea that possible local adaptation of plants at Grays Lake and Camas Prairie caused different germination responses in different soils, we noticed that the seedlings from each location looked different and that seeds germinated in a visibly different way. Seedlings from Grays Lake had narrower leaves with a more twisted appearance compared to Camas Prairie whose leaves were relatively stout and untwisted. While we stratified our seeds for this study in soil, we also noticed differences in the physical appearance of seeds upon germinating because we cold-moist stratified seeds in a paper towel inside of the same refrigerator for distribution to the community. On these paper towels, Grays Lake seeds had multiple dark-colored bulges of tissue emerging from multiple locations upon the seed's germination. In contrast, Camas Prairie seeds produced elongated white tissue from a single, consistent part of the seed (Figure 8). This difference in germination suggests that there may be genetic differences between the seed sources, or that seeds were degraded from environmental conditions at Grays Lake prior to being harvested. The negative relationship we observed between flower stalk basal diameter and seed germination might indicate that abnormal seed germination is more common on older or larger individuals at Grays Lake. Future studies are needed to determine the cause of these observed differences, which may be attributable to local adaptation, contaminant exposure, environmental stress, or differences in stewardship practices across the two sites.

Germinating C. quamash seeds from Camas Prairie (A) and Grays Lake (B).
For Grays Lake seeds, there was a significant negative relationship between flower stalk basal diameter and percent germination. This relationship was not found with the plant basal diameter. This finding was surprising because we would expect that larger plants would have larger seeds (Hendrix 1984), leading to a higher germination percentage (Uddin et al. 2015; Yalamalle 2016). We are unsure why the opposite relationship was observed in our case for this location. It is possible that larger plants had an earlier phenology and/or were older and that this may have exposed them for a longer time period to an environmental toxin or pathogen that negatively influenced seed viability (Biere and Honders 1996; Devi and Kumar 2020). Marks and Truscott (1985) also found plants with greater reproductive investment had less viable seed, but they weren’t sure why this occurred. Further investigation is needed to understand this trend and why it occurred at only one of the two study sites.
Stratification Approach and Soil Type
The significantly higher percent germination of Grays Lake seed after indoor stratification compared to outdoor stratification suggests that the indoor refrigerator conditions were more favorable for breaking seed dormancy (Luna, Wilkinson and Dumroese 1949). This is consistent with previous findings by Guerrant and Raven (1995) where C. quamash seeds germinated at a mean rate of 90% when cold stratified indoors. The difference in percent germination between indoor stratification and outdoor stratification of Grays Lake seed could be due to different moisture regimes. The moisture level for the indoor stratified seeds was controlled and more consistent throughout the 90 days of cold moist stratification. In contrast, the outdoor stratified seeds received moisture upon their summer planting, from precipitation (i.e., rain and snow), and when manual watering began in the spring. This less consistent moisture level could have resulted in lower percent germination, as some seeds need constant moisture during stratification for the best germination outcomes (Baskin and Baskin 1998; Swain et al. 2006).
Implications for the Cultivation of Camas in Fort Hall
Near the start of this project, the Language and Culture Preservation Department expressed interest in the future camas cultivation program using greenhouses located on the reservation. While we did not attempt to germinate seeds within the greenhouses, we did transfer germinated seeds to the greenhouse to support their continued growth. Here we briefly reflect on the pros and cons of indoor (i.e., refrigerator and greenhouse) vs. outdoor settings for seed germination and compare these to bulb transplants. Based on our experiences, we see the main benefits of indoor cultivation are the ability to control moisture levels, which we suggest may lead to higher germination rates, while the main drawback is the amount of labor required to support the indoor germination process, such as watering and monitoring temperature fluctuations. Outdoor cultivation is beneficial in that it is less labor intensive and provides seeds with naturally fluctuating temperatures and moisture levels, however there is potential for disturbances from animals, wind, and weeds that could suppress germination rate. Further, even if germinating seeds outdoors, it is still necessary to provide supplemental watering in spring given the local Fort Hall climate.
In comparison to growing plants from seed, given the high survival rates of bulb transplants across soil types, transplanting mature camas plants to Fort Hall as the best method to quickly establish camas populations and this can be done successfully in potting soil or directly into local soils. However, we recommend that the harvesting of camas plants should be done with the guidance of people who have cultural connections to the plant in order to maintain gathering practices that support existing camas populations.
Tribal-University Partnerships
This project contributes to the broader, long-term goal of reducing morbidity and mortality from heart disease, obesity, and other illnesses through the strengthening of relationships of Shoshone-Bannock Tribal citizens to traditional foods. Without taking a co-produced approach that included building relationships through the work and carefully following Tribal protocols, it is unlikely that the project could have contributed to the LCPD's and to the broader community's priorities. Our co-produced approach, as described earlier in the article, included jointly identifying research questions, focal species, and methods with the LCPD and other Tribal departments and programs and making sure that Tribal priorities drove project decision making. Our approach also included navigating formal and informal approval processes that may not have been known to the authors without having first meet with the LCPD to identify research priorities and plan the project. As noted by other authors (Matson et al. 2021), we found that a co-produced approach for this Tribal-university partnership was essential to meaningful research outcomes.
Indigenous research methodologies generally place a strong emphasis on relationships and on supporting Indigenous self-determination (Brayboy et al. 2012, Smith 2012; Wilson 2008), and this project revealed to the authors how taking a co-produced approach that parallels aspects of Indigenous research methodologies allowed for those relationships to grow and strengthen, as well as how being responsive to Tribal priorities allowed the project to support Indigenous self-determination. Specifically, we saw that this project supported self-determination by exploring opportunities for adjusting ancestral food practices to contemporary circumstances, including adaptations to redress limited access to traditional harvesting areas.
Just as this project sought to strengthen relationships of Tribal citizens to camas, it also contributed to a strengthening of relationships among the authors, among the authors and other community members, and among community members and other traditional food plants. For example, as a result of this project, Tribal youth in two different programs had the opportunity to learn about camas, and to plant camas bulbs at public locations in their community where they can continue to visit and monitor the plants. Further, while gathering bulbs at Grays Lake for one of these programs through the Tribes’ wellness grant (Figure 3D), which seeks to revive Tribal cultural practices especially with regards to traditional foods, it was fortuitous that we also became attentive to a harvestable population of sweet tobacco root (Valeriana edulis). As a result, information about sweet tobacco root, including the Shoshone band name Kuyudeka’a, “sweet tobacco root eaters” was revived and included on an interpretive sign for a local cultural site (Language and Culture Preservation Department, Shoshone-Bannock Tribes and Bureau of Land Management 2021). When taking the view of relationships of people to plants, the benefits of this project extended far beyond the immediate goals of learning how best to cultivate camas.
A white paper and brochure were created from this research project for both tribal and non-tribal citizens. These documents shared information on camas ecology, suggestions for camas cultivation from this study, and the historical relationship between the Shoshone-Bannock and other tribes to camas. These documents are meant to serve as accessible educational resources to those interested in camas cultivation or historical relationships to the plant.
Conclusion
This project sought to strengthen relationships of Shoshone-Bannock Tribal citizens to camas by providing information that could support the cultivation of camas on the reservation, with particular emphasis on comparing seeds and bulbs sourced from two key traditional gathering areas. While the highest percent germination was achieved with seeds collected from Camas Prairie stratified indoors in potting soil, seeds from both gathering sites germinated at a moderate high level after indoor stratification, suggesting both sites as reasonable sources of seed for a cultivation program on the reservation. While outdoor stratification led to lower germination percentages, the benefit of the lower time investment for this approach would need to be weighed against the drawback of the lower percent germination. Bulb transplants appear highly effective for establishment of new populations, at least in the short-term, but we suggest that gathering bulbs should be guided by Indigenous cultural practices and knowledge that sustain populations. In addition, either seed or bulb ex-situ cultivation and distribution may have impacts on the genetic diversity of Camassia quamash populations in the local Fort Hall area, and it would be important to investigate their genetic differences in future studies. Finally, as a partnership between a Tribal Nation and university, this study demonstrates that when approached with respect, such partnerships open up the possibility for the tools of western science to contribute to solving contemporary food sovereignty issues, while also creating a space for the strengthening of relationships between people, camas, and other plants that are central to Indigenous food sovereignty.
Footnotes
Acknowledgments
We thank Susan Filkins and Brenda Beckwith for their insight on camas seed germination and transplanting and Karen Trebitz for sharing her extensive camas literature. We give our deep gratitude to the Shoshone-Bannock Tribal community for sharing their knowledge and guidance with us.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This publication was made possible by the NSF Idaho EPSCoR Program and by the National Science Foundation under award number OIA-1757324, as well as by the Centers for Disease Control National Center for Chronic Disease Prevention and Health Promotion Tribal Practices for Wellness In Indian Country grant award number CDC-RFA-DP18-1812PPHF18, and the TRIO McNair Scholars Program at Idaho State University
Permissions
This project received approval from the Fort Hall Business Council per resolution FHBC-2020-0194 on March 10th, 2020 and the Shoshone-Bannock Tribe's Language and Culture Preservation Department's Culture Committee on March 10th, 2020.
