Abstract
Highlights
We identified 30 unique HPV vaccination models that incorporated marginalized populations, including populations living with HIV, low-income or rural populations, and individuals of a marginalized race, ethnicity, or sexual behavior.
Methods for incorporating these populations, as well as the assumptions inherent in the modeling structure and parameter selections, varied substantially, with models explicitly or implicitly attributing prevaccine differences to alternative combinations of biological, behavioral, and societal mechanisms.
Modelers seeking to incorporate marginalized populations should be transparent about assumptions underlying model structure and data and examine these assumptions in sensitivity analysis when possible.
Keywords
The burden of human papillomavirus (HPV) is disproportionately felt by individuals from historically and contemporarily marginalized populations, including those who are low income; minoritized based on race, ethnicity, or sexual identity; live in a rural area; or are living with HIV.1–4 To reduce the disproportionate burden of HPV and HPV-related cancers, researchers can incorporate data on differences in disease epidemiology and existing disease burden for these groups within HPV decision-support models. This ensures that models more accurately reflect HPV burden and transmission dynamics and enables the consideration of differential effects on marginalized groups in policy and programmatic decision making.
Including marginalized populations in decision models introduces distinct challenges. Researchers developing any HPV vaccination model must make numerous decisions about model structure and the source, granularity, and stratification of input data, each of which suggests key assumptions about the nature of disease and populations under study. 5 For those additionally incorporating stratification for marginalized populations, these decisions also reflect underlying theoretical understandings or assumptions about the root causes of different patterns of disease by choosing to incorporate potential differences in exposure to HPV, biological response to HPV, access to primary or secondary prevention, or combinations of these. 6
To better understand how marginalized populations have been incorporated in HPV vaccination simulation models, we conducted a systematic review of studies reporting on HPV vaccination simulation models that explicitly incorporated 1 or more marginalized populations (through stratification of any input variables). We sought to describe key analytic decisions in model structure, input data sources and granularity, calibration approaches, and presentation of results.
Methods
Literature Search
We conducted a systematic review of studies published from January 2006 through June 2022 using PubMed, CINAHL, Scopus, and Embase databases and identified articles describing HPV modeling and incorporating 1 or more marginalized populations in model structure and parameter values. Search terms included synonyms for modeling (i.e., “mathematical model,” “cost-effectiveness,” “economic evaluations,” “simulation”), HPV (“HPV,” “Human Papillomavirus”) and vaccination (“vaccine,” “immunization”). Our full search strategy for each database is available on PROSPERO (CRD42022314552) and has been described previously. 7
Study titles and abstracts were screened in parallel by 2 reviewer pairs (A.A.I./J.C.S. or O.O.O./C.B.B.) to exclude those that (1) were not available in English, (2) did not involve original research, (3) did not present a simulation model, or (4) did not evaluate the impact or cost-effectiveness of prophylactic HPV vaccination. The same reviewer pairs evaluated the full texts in duplicate to determine whether the model specifically delineated (i.e., through model structure or parameter values) 1 or more marginalized populations. We defined “marginalized population” as populations experiencing interpersonal or structural barriers to health care access or the resources needed for overall well-being due to social, political, or economic discrimination.8,9 Populations meeting the study team’s definition of marginalized were included, regardless of the stated intention for their inclusion in the original study. This included rural populations, those of minoritized race or ethnicity, individuals living with HIV, individuals living in poverty, those of minoritized sexual identity or behavior, and individuals employed in sex work. 10 We did not include studies evaluating only disparities between countries (e.g., high- versus low-income countries) or stratifying only by sex (e.g., male-only or female-only vaccination).
Data Extraction
Study characteristics, including author, year, included marginalized population, model type, and time horizon were extracted from each study. Because our goal was to analyze model structure/design, we grouped studies that reported analyses conducted on the same underlying model and reported on the earliest available study that met our inclusion criteria (additional studies reporting on each model are noted in Supplemental Table 1).
To characterize model structure and stratification, we identified whether models incorporated inputs across several structural categories6,11 and whether the inputs in those categories were stratified or selected specifically to reflect the marginalized population. Input categories included all-cause (or noncancer) mortality, sexual behavior (including number of partners, frequency of partner change, and partnership selection criteria, also called “assortivity” parameters), HPV prevalence, HPV vaccine uptake, cervical cancer screening use, health care access by any measure other than screening use (including primary care or sexual health care, cancer screening follow-up, and availability of treatment for cancer or precancer), HPV cancer incidence, and HPV cancer survival or mortality. Inputs were considered “stratified” if they were generated using data sources specific to the marginalized population, regardless of whether the data reflected statistically significant differences relative to a population estimate or comparison group. Our study team then iteratively developed a categorization scheme for the included HPV models using an inductive approach to describe key differences in data and assumptions used to incorporate the marginalized population(s) across models.
Finally we evaluated the use of model calibration, model validation, and sensitivity analysis. For sensitivity analysis, we coded for analyses that specifically tested parameter values or assumptions related to the included marginalized populations. Similarly, we identified whether calibration or validation was reported for the included marginalized population group or groups. Model calibration is the process of estimating uncertain model parameter values to maximize accuracy between data on an observed “target” and simulated equivalents, using either a deterministic or, more commonly for HPV models, a probabilistic approach. 12 We extracted the estimated parameters (calibrated parameters) and what data were used as targets (calibration targets). We also recorded which models performed calibration informed by existing data (for example, starting with literature estimates of HPV clearance for men who have sex with men [MSM] and using calibration to refine this by age to fit prevalence targets) compared with those conducting calibration with broad or arbitrary bounds (for example, varying a partner age-assortment constant from 0 to 1) or set based on population-level data (i.e., varying natural history inputs for both urban and rural settings across the identical ranges but with different population-specific targets). When a probabilistic approach was used, we noted whether a single best-fitting value was used to generate model outcomes or whether multiple sets of calibrated inputs were obtained to produce a range of plausible outcomes.
In contrast to calibration, external model validation compares the modeled estimate of a given outcome to an observed estimate of the same outcome using a source of data that was not incorporated into the model itself (or used for model calibration). 13 We extracted whether models conducted validation of outcomes specific to included marginalized populations and, if so, what outcomes were used (validation targets).
Results
Characteristics of Included Models
We identified 30 unique models (Table 1; Supplemental Table 1). Marginalized populations included MSM (k = 9),14–22 MSM living with HIV (k = 4),23–26 women living with HIV (k = 3),27–29 rural populations (k = 3),30–32 minoritized race or ethnicities (k = 5),33–37 low-income populations or populations living in areas of high poverty (k = 4),7,38–40 a combination of race and income (k = 1), 41 and female sex workers (k = 1). 42
Study and Model Characteristics

MSM, men who have sex with men.
A notable difference across models was how they approached structural representation of HPV transmission or acquisition. We found that 14 models (47%) used static HPV infection or HPV cancer-prediction models; this is defined as using a predefined (exogenous) risk of HPV infection or HPV cancer incidence as a model input. This included 5 Markov models (static compartment) models,24,26,29,32,41 6 microsimulation models,33–35,37,39,40 and 3 other simulation approaches that did not directly model transmission.14,17,38 The remaining 16 models (53%) incorporated explicit and dynamic (i.e., time-varying and model-driven) sexual transmission of HPV, including 1 network, 20 3 agent-based,16,23,42 and 12 using a differential equations/dynamic compartmental approach. ** These approaches capture both the direct and indirect impacts (herd immunity) of HPV vaccination.
Model Structure and Incorporation of Group-Specific Inputs
We categorized identified models into 5 categories based on their primary approaches for incorporating marginalized population-specific data into the model (Figure 1) and describe each below, including the populations or scenarios most often reflected in this approach, the inputs and assumptions required, and the key advantages and disadvantages of each approach.
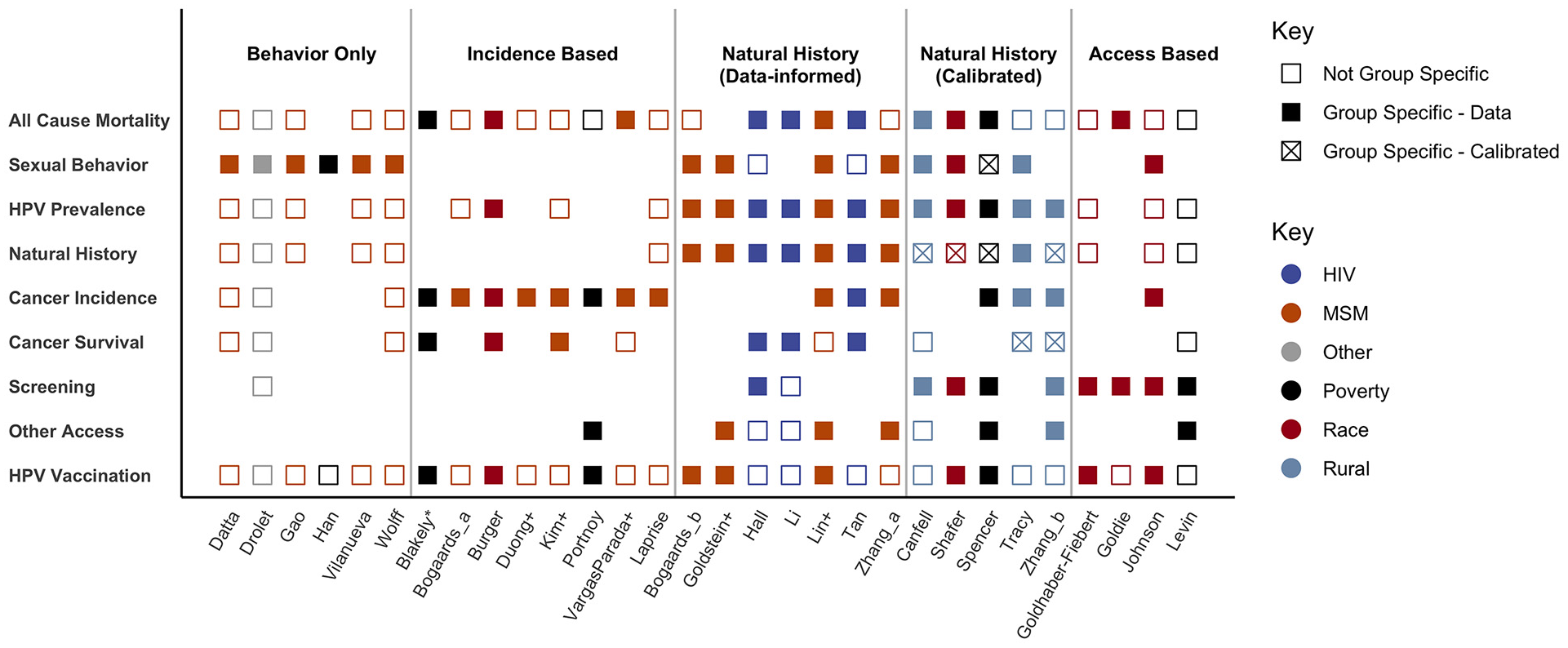
Included parameters by study and marginalized group. Each row indicates a category of inputs; a filled square indicates at least 1 input in this category incorporated group-specific data, and an empty square indicates at least 1 input in this category was incorporated but no group-specific data were used. An x indicates this input was calibrated to reflect a group-specific value.
Sexual behavior only
We classified this modeling approach as those delineating marginalized populations only through differences in sexual behavior. Typically, this involved a detailed sexual transmission model that incorporated group-specific data on behaviors (partner selection and frequency of partner change). These models inherently assumed the only relevant differences affecting outcomes for the marginalized population were in patterns of sexual behavior; therefore, models were commonly used to understand changes in HPV transmission and prevalence at a population level and generally presented more proximal outcomes (i.e., HPV prevalence) rather than distal (i.e., HPV cancer cases/deaths). We identified 6 models using a sexual behavior–only approach,16,18,20,21,38,42 most of which also defined the marginalized population itself by sexual behavior: either sex workers or MSM.
Drolet and colleagues 42 incorporated data on female sex workers as a separate sexual activity category but did not present stratified outcomes, conceptualizing this population as a “high exposure” group and important for capturing herd immunity dynamics. A similar approach was used by 4 models that incorporated MSM sexual behavior in HPV transmission while reporting results primarily for the overall population.16,18,20,21 This was primarily done to understand the impact of MSM on herd immunity benefits when examining gender-neutral vaccination. Although 2 of these studies presented changes in HPV prevalence specifically among MSM,18,20 neither examined stratified outcomes further downstream such as cancers prevented or quality-adjusted life-years gained for MSM.
By contrast, Han et al. 38 estimated the incidence of cervical cancer using a sexual behavior–only approach in a model of “socially vulnerable individuals” in Korea (defined as individuals with low incomes or from single-parent households). Using data from a national survey, researchers estimated poverty-specific outcomes by assuming the prevalence of HPV was linearly related to “risky” sexual behavior (not defined but implied to be younger age at onset of sexual behavior). These assumptions resulted in an estimated baseline burden of cervical cancer for the socially vulnerable that was 3 to 4 times higher than that of the general population. This resulted in HPV vaccination having a lower “realized efficacy” in low-income populations (as earlier onset of sexual behavior results in a larger share of individuals being exposed prior to vaccination), although the study found vaccination likely to be cost-effective for the socially vulnerable (due to the higher prevalence of disease).
A key advantage of the sexual behavior approach is the use of an explicit and clearly definable mechanism underlying group differences, for which data can generally be obtained from population-based surveys. This approach is likely a good fit when interested primarily in the impact of sexual behavior on population outcomes or a better understanding of HPV transmission dynamics. However, caution is needed, as this approach may further stigmatize marginalized groups if used without careful consideration of the larger context when interpreting findings or if evaluating more distal outcomes without inclusion of other systemic factors influencing disease burden.
Incidence based
This modeling approach was defined as delineating the marginalized population through data on the incidence of HPV cancers or genital warts. These models did not typically include explicit sexual transmission of HPV nor the natural history of HPV progression or clearance. Rather, models began with data on group-specific disease burden and assumed vaccination would result in a proportional reduction in HPV disease across groups. As a result, researchers did not have to attribute these differences to any specific biological or societal cause; rather, they assumed that any underlying differences would not influence the realized effectiveness of HPV vaccine on the modeled outcome(s). We identified 8 models that used an incidence-based approach to incorporate marginalized populations.14,17,19,24,26,37,40,41
An incidence-based approach was used in 5 models of HPV sequalae in MSM,14,17,19,24,26 with 2 also incorporating HIV status.24,26 These models relied on literature-based estimation of the relative risk (v. heterosexual men) of anal cancer,14,17,19,24,26 oropharyngeal cancer,14,19 penile cancer, 19 and anogenital warts17,24 in MSM. Researchers then projected the reduction in disease following HPV vaccination targeting either young-adult MSM24,26 or all adolescent boys.15,17,19 One model incorporated this method for MSM alongside a detailed sexual behavior model for heterosexual individuals to estimate the benefits of gender-neutral vaccination. 19
The other 3 incidence-based models evaluated the impact of HPV vaccination on disparities for individuals of a marginalized race and ethnicity, 37 individuals with low-incomes, 40 or a combination of these 41 relative to a more advantaged group. These models incorporated group-specific data on cancer incidence (informed by data from cancer registries or literature estimates) along with group-specific data on vaccine uptake (from vaccination registries or survey data) to estimate the potential reduction in HPV cancers. One model additionally incorporated data on racial and ethnic differences in HPV type distribution by cancer site, 37 whereas the others assumed identical type distribution (and therefore an identical percent reduction in disease) for all groups.
Incidence-based models offer a practical and potentially more tractable approach to incorporating marginalized populations. These models generally require only data on disease burden by group and the overall or type-specific reduction in disease at a given vaccination rate. As a result of lower data needs and computational burden, studies using this approach may compare multiple groups more easily.37,40,41 However, as this approach does not model either sexual behavior or HPV natural history, it may not be suitable for more complex policy questions. In addition, this method inherently assumes either that there are no differences in natural history pathways for an included marginalized group or that any differences would not affect the efficacy of HPV vaccination, assumptions that should be carefully considered and explicitly stated by researchers.
Data-informed natural history models
We defined the approach as a “data-informed natural history” when models differentiated a marginalized population through incorporating specific biological differences in the natural history of HPV, with parameters informed by available literature. This approach makes explicit the biological mechanisms by which disease burden is assumed to differ for a certain population by modifying HPV transmission, clearance, progression to cancer, or a combination of these. As a result, these models all included a detailed natural history of HPV, with or without a sexual transmission model. We identified 7 models using this approach, which incorporated HIV status, MSM, or a combination.15,22,23,25,27–29
Three models of women living with HIV27–29 accounted for differences in susceptibility to HPV infection and increased risk of HPV progression to cancer in the presence of a co-occurring HIV infection, both of which are well-described in epidemiologic studies.1,44 This allowed studies to examine the differential impact of HPV vaccination for women living with HIV27,29 as well as understand the interaction between HIV prevention and treatment, HPV vaccination, and cervical cancer incidence. 28
While it is not necessarily true that MSM experience different trajectories of HPV disease than heterosexual men do, we found a natural history–focused approach was used in several studies of MSM (regardless of HIV status) due to epidemiological and clinical studies reporting disease outcomes specifically for this population. Four models of MSM used this approach to examine the cost-effectiveness of targeted MSM vaccination in the United States, 23 Australia, 22 England, 25 and the Netherlands, 15 incorporating data from epidemiologic and cohort studies on HPV clearance and progression for HIV+ and HIV− MSM.4,45–47 Of note, all 74 MSM models refined some aspect of natural history through calibration.15,22,23,25 For instance, Bogaards et al. 15 calibrated 20 different versions of transmission probabilities using different assumptions around natural immunity and latent HPV infections. However, all studies used data on observed natural history specific to MSM (typically acquisition and clearance of infections) as starting points for this calibration process.
The advantage of a data-informed approach is through the explicit incorporation of differences in the mechanisms of disease susceptibility, progression, or severity, which allows for a granular understanding of the implications of these differences on future HPV outcomes in different HPV vaccination scenarios. For populations with potentially altered natural history, incorporation of these differences is paramount in evaluating vaccine impact. However, an extensive amount of epidemiological and clinical data are required to inform these models effectively, which is not available for many populations and settings.
Calibrated natural history models
We identified a calibrated natural history approach as using model structures similar to those of data-informed natural history models but without available data on differences in HPV progression, clearance, or acquisition. Instead, natural history parameter values were calibrated to fit estimates of HPV outcomes. In this way, calibrated natural history approaches were similar to those used for incidence-based models, starting with observed differences in measurable outcomes (cancer incidence/mortality or HPV prevalence) without clear evidence as to the cause of these differences. However, through fitting either single or multiple sets of parameter values, 1 or more explicit pathway for differences was constructed. This approach was most commonly used for populations in which data on HPV outcomes is reported but epidemiological data are not available, including rural populations and those living in poverty.
We identified 5 models that distinguished marginalized populations using a calibration-based natural history approach.30–32,36,43 Two of the calibrated natural history models presented multiple sets of calibration outcomes,30,43 demonstrating a range of outcomes across alternative combinations of parameter values, while the remaining 3 models presented only the single best-fitting calibrated parameter set.31,32,36
Of the 3 calibrated natural history models that examined rural populations, 2 were conducted in China30,32 and 1 in Mali. 31 Both studies in China started with a previously developed natural history model of HPV and used setting-specific data on HPV prevalence,30,31 cervical cancer incidence, 32 or cervical cancer mortality. 32 Both varied parameter values related to HPV natural history (transmission, clearance, natural immunity) across a broad range to fit these outcomes in the absence of setting-specific data to inform initial values for model calibration. The models based in Mali fit transmissibility and progression of HPV to cancer using urban data, holding both constant for an identically structured rural model. The incidence of cervical cancer for the rural setting subsequently differed as a result of differences in HPV prevalence, but only HPV cancer survival varied independently to fit cancer mortality data for the rural versus urban model. 31
A US-based model independently calibrated parameter values for sexual behavior and natural history of HPV in high- and low-poverty counties to stratified data on HPV prevalence and cervical cancer incidence in these settings. 43 Using a similar approach, a Canada-based model calibrated sexual behavior and natural history by ethnicity parameters to fit data on HPV prevalence and cervical cancer incidence. 36 Both of these studies suggested in their methods that higher smoking rates (in high-poverty US counties and First Nations populations in Canada) may be an underlying mechanism for differences in natural history but did not incorporate this directly.
The advantage of a calibration-based natural history model is that it allows for examination of differences across multiple components of natural history but without the extensive data requirements of a fully data-informed model. However, the disadvantage of this approach is computational and logistical: variations across natural history can be nonidentifiable, meaning multiple combinations of parameters fit the data equally well in calibration but could have important differences in model outcomes. A thorough analysis of a calibration approach may require not only the computational effort for probabilistic model calibration but also evaluation of outcomes incorporating both parameter uncertainty and nonidentifiability of calibrated inputs, creating challenges for the presentation and interpretability of findings.
Health care access
Our final categorization of model approaches is those that defined the marginalized population through assuming a fixed natural history of disease and relying solely on differences in care access to drive differences in outcomes. These models typically did not incorporate sexual transmission and sometimes included detailed natural history models. Regardless of the model structure, this approach clearly defined an assumption that differences are driven by social inequities that result in poor access to preventive, diagnostic, or curative care. We identified 4 models using this approach, 3 of which were focused on differences in care access by race or ethnicity33–35 and 1 on differences in care access by income. 39
Levin et al. 39 evaluated the impact of HPV vaccination by income quintile in China and assumed that the only prevaccine differences in cervical cancer were a result of differential cervical cancer screening rates and follow-up to care. Two models examined the impact of HPV vaccination by race and ethnicity in the United States, both of which incorporated racial differences in cervical cancer screening use as the key driver of disparities, evaluating the impact of HPV vaccination with or without improving screening usage.33,34 Johnson and colleagues 35 evaluated differences in cervical cancer by race and ethnicity in the United Kingdom, incorporating ethnicity-specific patterns of sexual behavior and cervical cancer screening usage based on available survey data but keeping natural history fixed for all populations.
The chief advantage of an access-focused model is, similar to sexual behavior or natural history models, offering an explicit driver of existing disparities. In addition, for cervical cancer screening and treatment, inputs may be easy to identify from existing data for many groups. This approach focuses on the role of structural inequality for driving differences in health care outcomes that may produce more interpretable findings compared with methods that are agnostic to the underlying causes of differences. However, the impact of structural inequity is complex and therefore could also affect upstream aspects of the disease pathway (HPV exposure or progression) in ways that are hard to measure or incorporate explicitly.
In addition, although the models that we identified using an access-only approach were focused on race and income, we note that, by definition, all marginalized populations may face societal barriers that reduce access to appropriate care, making this a common consideration in many studies, even when evidence was also available for behavioral and natural history pathways. To this end, 9 models that incorporated differences primarily through incidence or natural history also accounted for some differences in access to or use of care for sexual health,22,25 cancer screening,28,30,32,36,43 or appropriate precancer or cancer treatment.32,40,43
HPV Vaccine Uptake
Generally, models incorporated HPV vaccination using an arbitrary value (such as 80% or 100% coverage) with the goal of exploring the impact or cost-effectiveness of introducing vaccination. However, 8 studies incorporated marginalized population–specific uptake of HPV vaccination into their model using survey data,23,37,43 evidence from uptake of other vaccines,15,25,40 clinical data, 35 or registry data. 41 An additional 3 studies examined the impact of a hypothetically unequal vaccine uptake for the marginalized population relative to a more advantaged population as either a primary or secondary analysis.33,36,41
Calibration, Validation, and Sensitivity Analysis
Eleven of the included models (37%) calibrated parameter values specific to a marginalized population (Table 2; Supplemental Table 2). † Parameters related to per-sex-act or per-partnership transmission rates and the duration or extent of natural immunity after clearance of infection were most commonly calibrated. Data on sexual behavior, HPV clearance, and cancer progression were also sometimes refined in calibration, either from a starting estimate15,16,22,23,25,27,28 or calibrated starting from data not specific to the marginalized group.30–32,36,43
Calibration and Validation of HPV Vaccination Models

HPV, human papillomavirus.
Calibration targets included HPV prevalence, ‡ incidence or prevalence of HPV-associated diseases including genital warts,22,25 cervical cancer,28,31,32,36,43 and anal cancer.22,25 Several models were also calibrated to fit cervical cancer mortality.31,32 Five of the models presented results from only a single best-fitting parameter set,23,27,31,32,48 while 6 presented results across a range of top-fitting parameter sets.14,16,22,25,30,43
Only 5 models (17%) provided external validation of outcomes for included marginalized populations (some studies refer to “validation” of findings but are referring to the fit of calibrated data, rather than an external validation). One model of women living with HIV was calibrated to HIV prevalence and then validated against the diagnosis of AIDS and HPV prevalence by HIV status. 28 Another model had natural history parameters calibrated to fit 2002 HPV prevalence for high- and low-poverty US counties and validated short-term model estimates to HPV prevalence from 2014. 43 The remainder of the studies, including one of women living with HIV, 27 one among rural-dwelling populations, 30 and one of ethnic disparities in the United Kingdom, 36 used group-specific cervical cancer incidence or mortality to validate model outcomes.
While nearly all studies performed sensitivity analysis around the cost or efficacy of HPV vaccination itself, few performed sensitivity analysis focused on testing assumptions around the included marginalized population or drivers of existing disparities (Table 2; Supplemental Table 3). As noted above, 6 models included calibration data across multiple parameter sets, reflecting sensitivity across combinations of sexual behavior or natural history parameters.14,16,22,25,30,43 Three studies conducted a 1-way analysis on the impact of removing differential all-cause mortality for a marginalized group (therefore not “penalizing” estimates of benefits through a shorter life expectancy).29,37,41 Three studies specifically assessed uncertainty around a partnership assortivity parameter by ethnicity, 36 age, 31 or positional preference for MSM (insertive v. receptive). 15 Several studies on populations living with HIV assessed sensitivity around efforts for HIV control (scale-up of prevention, detection, or treatment).23,27,29 Similarly, several studies evaluated the scale-up or targeting of cervical cancer screening for the marginalized group.28,33–35,39
Discussion
While there is no singular correct approach for incorporating marginalized populations into simulation models of HPV, researchers both developing and interpreting models should seek to understand the strengths, weaknesses, and assumptions inherent in alternative approaches. Simulation studies incorporating a marginalized population should be explicit, when possible, in defining the nature of the increased HPV-related risk in the model structure and inputs, as these differences can influence model results and interpretation. Models incorporating individuals with low incomes or living in high-poverty areas serve as an excellent example of the impact of these differences in structure. The models we identified attributed existing disparities through differences in sexual behavior, 38 access to screening and treatment, 39 natural history of disease, 43 or through cancer incidence. 40 Each of these structural choices could have implications for the estimated impact or cost-effectiveness of HPV vaccination.
Although the best approach may vary by target populations and research questions, we identify several best practices for model development. First, when sufficient data are available, models specifying full transmission and natural history pathways may offer the most transparent and comprehensive modeling approach by incorporating data on disease acquisition, progression, diagnosis, and treatment. These more complex models can incorporate differences driven by biological, behavioral, or societal factors as well as combinations of these, depending on the evidence best supported by existing data on these factors. Importantly, the direct incorporation of societal factors, including access to and quality of primary and secondary care are underrepresented in existing models and merit consideration. Overall, models should fit a theoretical understanding of the setting(s) and population(s) included; therefore, a thorough review to understand the evidence for alternative drivers of disparities is important for conceptualizing the model and guiding data generation or acquisition.
In the absence of robust data to characterize the nature of disparities, less data-intensive methods may still provide valuable insights. Incidence-based approaches may be sufficient for understanding the impact of vaccination where there are not likely to be differences that affect HPV type distribution or the progression or severity of disease. Under these circumstances, assuming a proportional reduction in HPV disease for a given vaccination rate may be a fair approximation of the direct benefits. Similarly, several of the models we identified used complex model structures and model calibration to generate alternative “drivers” of observed differences in HPV outcomes. Under ideal circumstances, this approach can relax the assumptions of the incidence-based method through testing multiple scenarios with alternative underlying causes of differences in HPV cancer outcomes, some of which could affect realized HPV vaccine efficacy. But, in a worst-case scenario, this approach may convey overconfidence in a single set of outcomes while the calibration process obscures the fundamental assumptions underlying the differences described by the model. Therefore, it is important that researchers are transparent about calibration processes and set plausible search ranges, guided by best available evidence. Further, as calibrating multiple natural history parameters simultaneously may result in “nonidentifiability,” researchers using this approach should evaluate both parameter uncertainty and calibration uncertainty, ideally presenting data across multiple good-fitting parameter sets, alongside other deterministic or probabilistic sensitivity analysis.12,49
In all cases, it is important that researchers are explicit, both internally and externally, about the assumptions being made and how those reflect their conceptual understanding of the marginalized population being studied. Included populations were defined by combinations of sexual behavior, health conditions, geography, socioeconomic status, and social constructs of race or ethnicity, none of which are discrete or homogenous categories and many of which can vary for an individual over time. Few of the included studies were explicit about how they conceptualized the included marginalized populations nor how their theoretical understanding might contribute to the data used and variables included and excluded in their model. Perhaps clearest in this regard was the study by Goldie and Daniels, 34 which both created and used a framework for conceptualizing racial disparities and incorporated this into model estimates.
We encourage researchers to consider in sensitivity analysis whether and how gaps in data can be tested or better understood. In particular, as the landscape of HPV vaccination continues to change,50–52 studies including marginalized populations should evaluate the impact of differential HPV vaccine access or uptake across groups53,54 as well as consider the potential for differential impact of policy and intervention on these groups.55,56 Finally, model validation was rare in our studies, with only 5 studies externally validating outcomes for marginalized populations against data on disease prevalence or incidence. In addition to employing sensitivity analysis to test modeling assumptions and data uncertainty, performing external validation may provide additional confidence that the model is accurately reflecting the populations the researchers are seeking to include.
Many areas of health research seek to describe or address health inequities, but often studies remain agnostic to the underlying causes of these differences. Decision modeling, by its nature, requires the researcher to clearly define drivers of inequities, providing additional challenges but also important opportunities to advance the science and improve health equity in unique ways. HPV researchers have an opportunity to use decision modeling to better guide HPV policy and program planning through addressing the disproportionate impact of HPV on marginalized populations. Decision modelers hoping to incorporate marginalized populations into their models, especially their HPV vaccine models, should seek to carefully understand existing literature on the structural, behavioral, and clinical drivers of existing inequity and be clear, discerning, and transparent with their target audiences about the assumptions underlying their models and should work toward improvements in data collection for these populations to better inform future work.
Supplemental Material
sj-docx-1-mdm-10.1177_0272989X251325509 – Supplemental material for Methodological Approaches for Incorporating Marginalized Populations into HPV Vaccine Modeling: A Systematic Review
Supplemental material, sj-docx-1-mdm-10.1177_0272989X251325509 for Methodological Approaches for Incorporating Marginalized Populations into HPV Vaccine Modeling: A Systematic Review by Jennifer C. Spencer, Juan Yanguela, Lisa P. Spees, Olufeyisayo O. Odebunmi, Anna A. Ilyasova, Caitlin B. Biddell, Kristen Hassmiller Lich, Sarah D. Mills, Colleen R. Higgins, Sachiko Ozawa and Stephanie B. Wheeler in Medical Decision Making
Footnotes
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SBW receives unrelated grant funding paid to her institution from Pfizer Foundation and Astra Zeneca. LPS has received unrelated grant funding paid to her institution from AstraZeneca. SO receives unrelated grant funding paid to her institution from Merck. The other authors have no disclosures to report. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by grants with the National Institutes of Health (5P01 CA250989-03 and 1K01MD0176 33-02). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Authors’ Note
This work was presented in part at the 2023 SMDM European Meeting in Berlin, Germany. A related publication has been previously published in Preventive Medicine that uses the same systematic review but reports only study outcomes: Spencer JC, Spees LP, Biddell CB, Odebunmi OO, Ilyasova AA, Yanguela J, Lich KH, Mills SD, Higgins CR, Ozawa S, Wheeler SB. Inclusion of marginalized populations in HPV vaccine modeling: a systematic review. Prev Med. 2024;182:107941. DOI: 10.1016/j.ypmed.2024.107941. Epub 2024 Mar 24. PMID: 38522627; PMCID: PMC11194695
Ethical Considerations
This study was a systematic review involving no human subjects and was exempt from review by the Institutional Review Board at the University of North Carolina at Chapel Hill.
Consent to Participate
This study was a systematic review involving no human subjects and was exempt from review by the Institutional Review Board at the University of North Carolina at Chapel Hill.
Consent for Publication
Not applicable.
Data Availability
**
References 15, 18, 19, 21, 22, 25, 27, 28, 30, 31, 36, and 43.
†
References 15, 16, 22, 25, 27, 28, 30–32, 36, 43.
‡
References 15, 16, 22, 25, 27, 30, 31, 36, and 43.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
