Abstract
Background
Patient decision aids (PtDAs) are effective interventions to help people participate in health care decisions. Although there are quality standards, PtDAs are complex interventions with variability in their attributes.
Purpose
To determine and compare the effects of PtDA attributes (e.g., content elements, delivery timing, development) on primary outcomes for adults facing health care decisions.
Data Sources
A systematic review of randomized controlled trials (RCTs) comparing PtDAs to usual care.
Study Selection
Eligible RCTs measured at least 1 primary outcome: informed values choice, knowledge, accurate risk perception, decisional conflict subscales, and undecided.
Data Analysis
A network meta-analysis evaluated direct and indirect effects of PtDA attributes on primary outcomes.
Data Synthesis
Of 209 RCTs, 149 reported eligible outcomes. There was no difference in outcomes for PtDAs using implicit compared with explicit values clarification. Compared with PtDAs with probabilities, PtDAs without probabilities were associated with poorer patient knowledge (mean difference [MD] −3.86; 95% credible interval [CrI] −7.67, −0.03); there were no difference for other outcomes. There was no difference in outcomes when PtDAs presented information in ways that decrease cognitive demand and mixed results when PtDAs used strategies to enhance communication. Compared with PtDAs delivered in preparation for consultations, PtDAs used during consultations were associated with poorer knowledge (MD −4.34; 95% CrI −7.24, −1.43) and patients feeling more uninformed (MD 5.07; 95% CrI 1.06, 9.11). Involving patients in PtDA development was associated with greater knowledge (MD 6.56; 95% CrI 1.10, 12.03) compared with involving health care professionals alone.
Limitations
There were no direct comparisons between PtDAs with/without attributes.
Conclusions
Improvements in knowledge were influenced by some PtDA content elements, using PtDA content before the consultation, and involving patients in development. There were few or no differences on other outcomes.
Highlights
This is the first known network meta-analysis conducted to determine the contributions of the different attributes of patient decision aids (PtDAs) on patient outcomes.
There was no difference in outcomes when PtDAs used implicit compared with explicit values clarification.
There were greater improvements in knowledge when PtDAs included information on probabilities, PtDAs were used in preparation for the consultation or development included patients on the research team.
There was no difference in outcomes when PtDAs presented information in ways that decrease cognitive demand and mixed results when PtDAs used strategies to enhance communication.
Keywords
Background
Patient decision aids (PtDAs) are effective interventions designed to support patient and public involvement in health care decisions. According to a Cochrane systematic review, PtDAs are defined as interventions that, at a minimum, focus on a specific decision, provide information on the options and outcomes (e.g., benefits/harms) relevant to a person’s health status, and help patients clarify the values they place on benefits and harms of options using implicit or explicit methods. 1 PtDAs can also include other nonessential content elements: 88% presented probabilities of outcomes, 73% provided guidance in decision making, and 67% used explicit values clarification exercises. 1 The most recent Cochrane review of 209 randomized controlled trials (RCTs) evaluated PtDAs across a wide variety of decisions with 107,698 participants. 1 Compared with usual care, PtDAs demonstrated higher informed values choice, improved knowledge, more accurate risk perceptions, an increased active role in decision making, lower elements of decisional conflict (uninformed, unclear values), and fewer undecided. Since the original systematic review was published in 1999, 2 the evidence base supporting PtDAs has grown significantly, and the 2024 findings, appraised using GRADE, provide moderate to high certainty and confidence in the evidence for the primary outcomes.
PtDAs are complex interventions 3 given they allow for flexibility in the ways the content elements are presented, timing of their delivery in relation to the consultation, and the processes used for their development. To appreciate the contributions of their different attributes, they need to be theoretically deconstructed.3,4
The International Patient Decision Aid Standards Collaboration (IPDAS) provides a theoretical structure for deconstructing their attributes and has embraced differences in the ways they can be fulfilled. 5 For example, the values clarification attribute in PtDAs includes implicit approaches by providing a rich description of the options so users can imagine what it is like to experience them and can weigh up the desirability of the different options on their own to arrive at a preferred option or explicit exercises such as rating scales, rankings, or conjoint analysis, so users can actively engage in a task to compare the relative importance of features of each option to reach their preferred option. 6 Probabilities may be communicated using numbers (e.g., x of 100) and/or figures using pictograms or bar charts.7,8 Other variations between PtDAs are strategies to decrease cognitive demand (e.g., presenting information on options side by side, step-by-step guidance in decision making) and strategies to facilitate discussion with others (e.g., personal summaries, list of questions, encouraging discussion with others). For timing of delivery, PtDAs can be given to the patient for use in preparation for the consultation or used by the patient and clinician together during the consultation. Finally, there are variations in the processes by which PtDAs are developed, with a shift in the last 10 y to involving patients and other potential users of PtDAs on the development team. 9 Although PtDAs are effective complex interventions, it is not clear whether there are relative advantages to ways in which the content elements are presented (or whether they are presented at all), timing of their delivery, or processes for development.
Our overall aim was to use a network meta-analysis (NMA) model to compare the effects of different attributes of PtDAs from a comprehensive systematic review of RCTs. The specific research question was as follows: What effects do PtDAs with different attributes (specify the content element, development process, or delivery mechanism) have on informed values choice, knowledge, accurate risk perception, decisional conflict related to feeling uninformed and unclear about personal values, and undecided?
Methods
Methods for this exploratory NMA were guided by the undertaking NMA chapter in the Cochrane Handbook. 10 Our data source was the 209 RCTs included in the 2024 updated Cochrane systematic review of decision aids for people considering health care treatment or screening decisions. 1 Detailed methods for the systematic review are reported elsewhere 1 and in the NMA protocol published in open science. Results for this NMA were reported following the PRISMA extension statement for reporting of systematic reviews incorporating NMA of health care interventions. 11
Included Studies
Eligible RCTs compared PtDA to usual care and reported on at least 1 primary outcome of interest with data suitable for NMA.
Primary Outcomes
Of 10 primary outcomes in the Cochrane review of PtDAs, 1 6 were considered for this NMA given the higher certainty of evidence based on Grading of Recommendations Assessment, Development and Evaluation (GRADE) ratings and the volume of RCTs that measured these outcomes. The outcomes were the participants’ 1) informed values-based health care choice indicated the match between the chosen option and the features that mattered most to the informed participant (moderate certainty), 2) knowledge of the options and their features using standardized scores out of 100 (high certainty), 3) accurate risk perception measured the accuracy of participants’ perceived probabilities of an outcome (high certainty), 4) decisional conflict subscale for feeling uninformed using standardized scores out of 100 (high certainty), 5) decisional conflict subscale for unclear values or unclear about the features that mattered most to them using standardized scores out of 100 (high certainty), and 6) proportion undecided (high certainty). Too few RCTs measured the participants’ participation in decision making, decision regret, communication with their clinician, satisfaction with the decision-making process, and preparation for making decisions to include them in this NMA.
Attributes of PtDAs
In the process of revising the IPDAS using an informed modified Delphi consensus process, several questions emerged about the influence of various attributes on the primary outcomes of PtDAs, and we asked IPDAS review teams to submit potential NMA questions.5,12 For each of the following PtDA attributes, we considered and compared the following differences:
Specific content elements: a) type of values clarification method (comparisons: implicit, explicit); b) presenting probabilities (comparisons: yes, no); c) presenting information using a side-by-side format (comparisons: yes, no); d) providing step-by-step guidance in decision making (comparisons: yes, no); e) use of a personal summary to facilitate communication (comparisons: automated summary, worksheet, no); f) provided a list of questions to ask about the decision (comparisons: yes, no); and g) written encouragement to discuss the decision with others (yes, no). A personal summary is a report of patients’ answers to questions posed in the PtDA, which include, for example, values clarification exercises, their preferred option, and their questions to discuss with the clinician. The patient can share their personal summary with the clinician or others involved in the decision. An automated personal summary was generated after patients completed an online PtDA and a worksheet was completed within a paper-based PtDA.
Timing of delivery: timing of PtDA administration (comparisons: preconsultation, during consultation).
Developmental processes: PtDA user involvement in development (comparisons: health care professional [HCP], patient/consumer, HCP and patient/consumer, none); PtDA user involvement in testing (comparisons: HCP, patient/consumer, HCP and patient/consumer, none); use of a theory, conceptual model, or framework (comparisons: IPDAS, other theory, conceptual model, or framework with or without IPDAS, none). Theories provide concepts, relationships between them, and specific predictions that can be used to guide observation, understanding, and explanation of a phenomena.13,14 Conceptual models are usually a more simplified description of a phenomenon without being explanatory. Frameworks are an overview or outline consisting of descriptive categories (e.g., concepts, constructs, variables) and relationships between them that are relevant to a phenomenon without being explanatory.
NMA
A Bayesian NMA was conducted using WinBUGS software version 1.4.3 (MRC Biostatistics Unit, Cambridge, UK) using data from the 2024 Cochrane systematic review comparing PtDAs to usual care. An initial analysis comparing PtDA to usual care was done to verify the results from this subset of studies that were included in the 2024 Cochrane review of PtDAs. Dichotomous outcomes were assessed using a binomial likelihood model with a logit link function, and continuous outcomes were assessed using a normal likelihood model with an identity link function. 15 The reference group for the NMAs was usual care, and the nodes in the network consisted of PtDAs with different options for content elements, delivery mechanisms, or development strategies. Effect estimates were risk ratio (RR) for dichotomous outcomes and mean difference (MD) for continuous outcomes. Point estimates and 95% credible intervals (95% CrIs) were determined using Markov Chain Monte Carlo methods, and basic parameters of the treatment effects in the model were assigned vague prior distribution, N (0, 1002). The NMA model used random effects, and informative priors 16 for the between-study variance parameters were considered. Continuity correction when needed was applied to adjust for zero events reported in dichotomous outcomes by adding a fraction of the reciprocal of the size of the opposite treatment arm to the event. 17 Goodness of model fit was assessed using the deviance information criterion and a comparison of the residual deviance to the number of unconstrained data points, and model convergence was assessed trace plots and the Brooks–Gelman–Rubin statistic. Three chains to fit the model were used, each with more than 10,000 iterations, with a burn-in of more than 10,000 iterations.18–20
Results
Of 209 RCTs included in the Cochrane Review, 149 (71.3%) reported relevant data for primary outcomes (see Figure 1; Supplementary Table S1). The attributes of PtDAs were obtained from available PtDAs (n = 114). For missing PtDAs, the attributes were obtained only if authors provided these PtDA details in their publication(s) (n = 35) (see Supplementary Table S1). The 149 included RCTs involved 45,249 participants from 16 countries. The number of RCTs and total number of participants involved for each outcome by content elements, timing of delivery, and development are provided in Table 1.
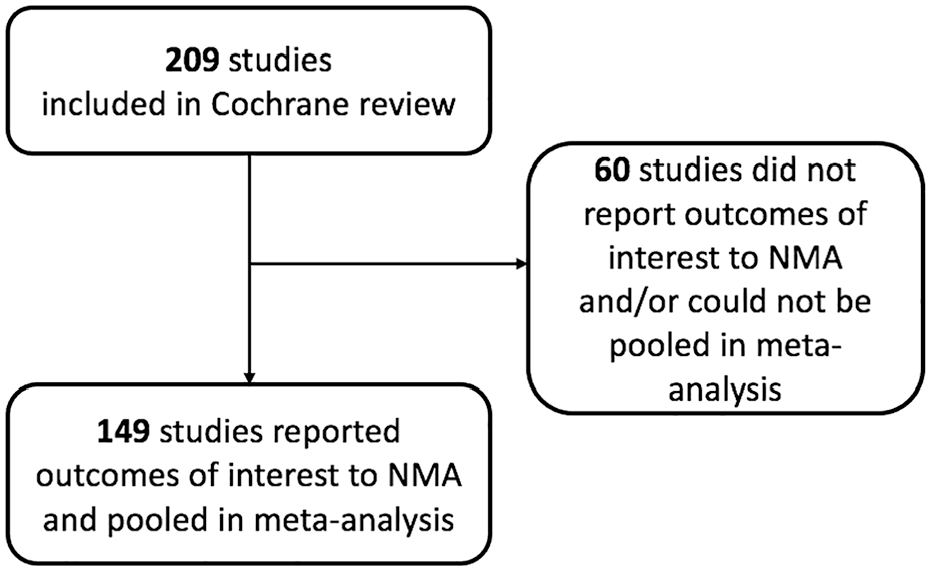
Flow diagram of study inclusion.
Number of Randomized Controlled Trials (RCTs) and Total Participants for Each Outcome by Attribute of the Patient Decision Aid a
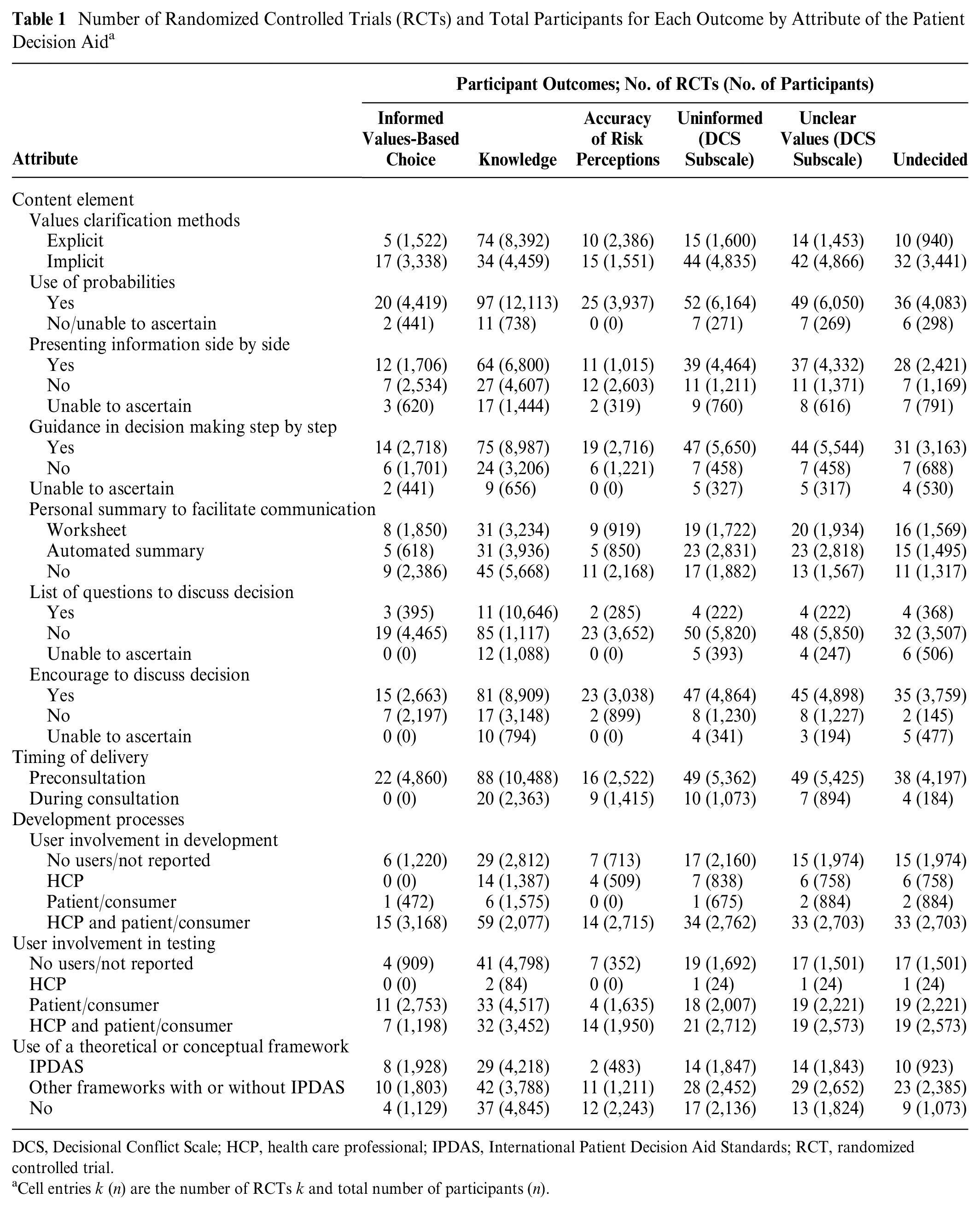
DCS, Decisional Conflict Scale; HCP, health care professional; IPDAS, International Patient Decision Aid Standards; RCT, randomized controlled trial.
Cell entries k (n) are the number of RCTs k and total number of participants (n).
There were 123 PtDAs used ahead of the consultation and 26 PtDAs used in the consultation. All met the minimal definition of being a PtDA but had differences in some content elements (Table 2). For example, an explicit values clarification exercise was used in 76% preconsultation PtDAs and 31% during consultation. Both types included probabilities (87%; 96%), presented information side by side (57%; 58%), and encouraged discussion of the decision (72%; 100%). Only preconsultation PtDAs provided lists of questions to discuss (12%).
Content Elements for PtDA used Preconsultation versus During Consultation
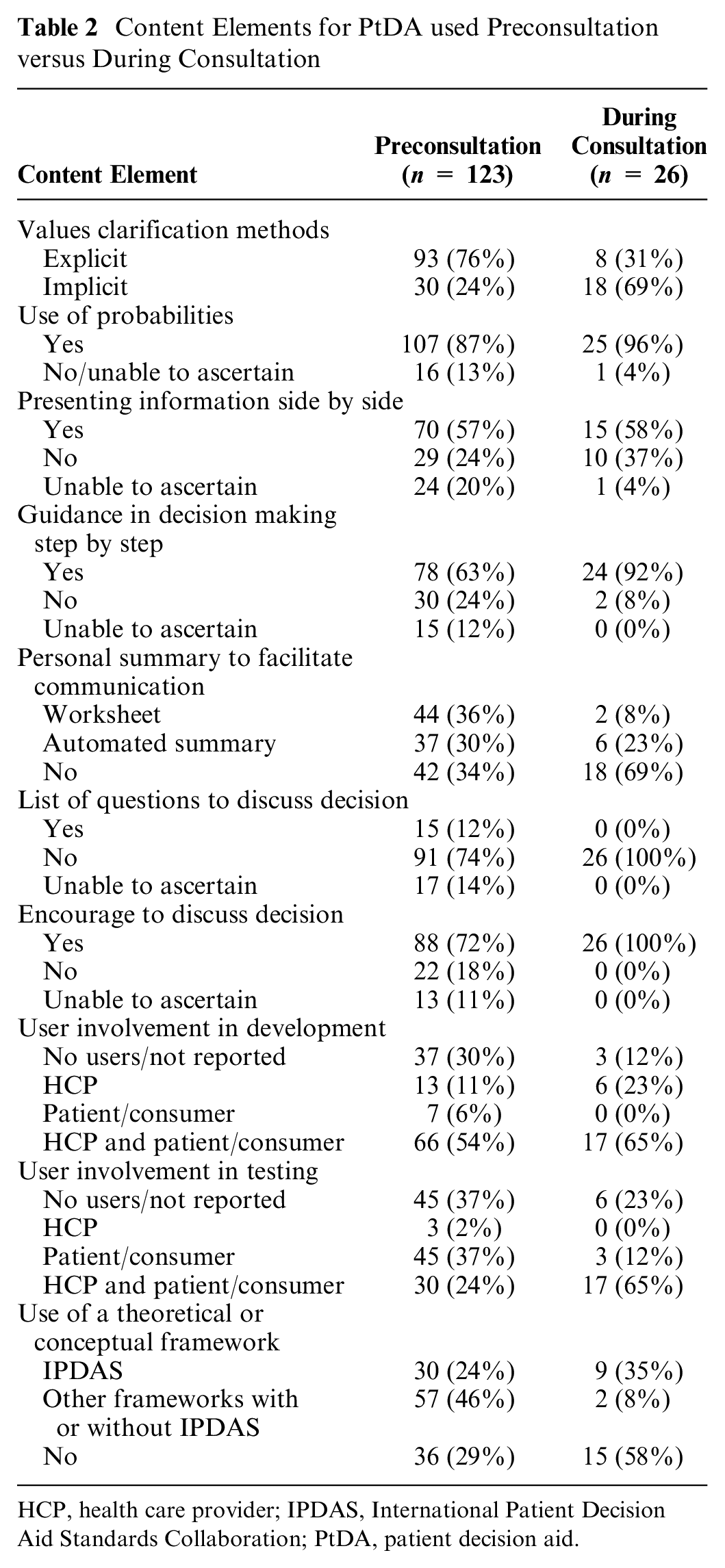
HCP, health care provider; IPDAS, International Patient Decision Aid Standards Collaboration; PtDA, patient decision aid.
Is There an Effect of Explicit or Implicit Values Clarification in PtDAs on Outcomes?
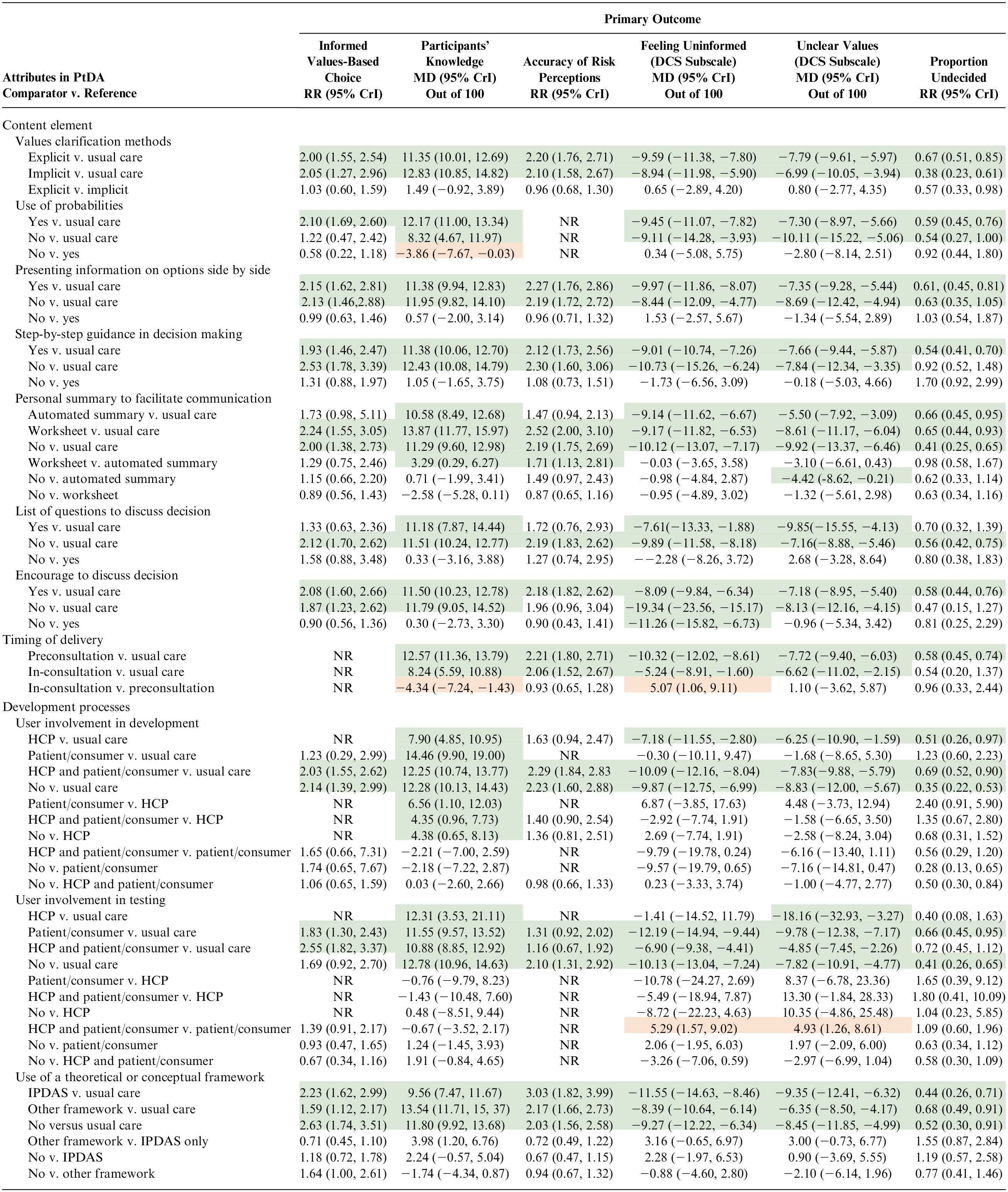
Of 149 RCTs, 48 PtDAs used implicit values clarification and 101 had an explicit values clarification exercise. The most common values clarification exercises were rating scales (n = 62), importance of pros and cons (n = 16), and decision analysis (n = 7). Compared with usual care, PtDAs with implicit or explicit values clarification were significantly better for all outcomes (see Table 3). PtDAs that used implicit values clarification had a significantly larger reduction in the proportion undecided compared with explicit values clarification (RR 0.57; 95% CrI 0.33 to 0.98). There was no significant difference in outcomes when PtDAs that used implicit values clarification were compared with PtDAs that used explicit values clarification.
Results of the Network Meta-analysis a
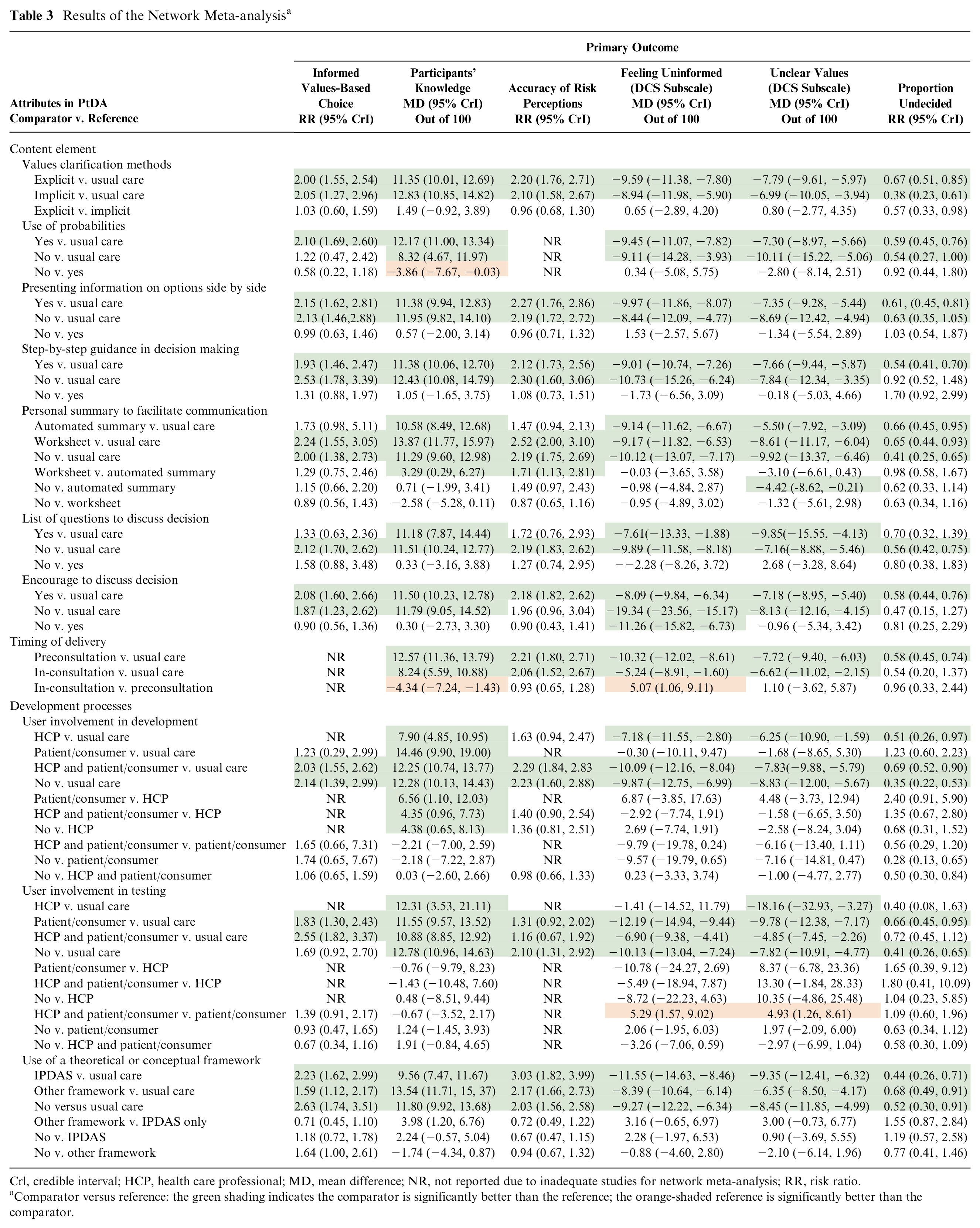
Crl, credible interval; HCP, health care professional; MD, mean difference; NR, not reported due to inadequate studies for network meta-analysis; RR, risk ratio.
Comparator versus reference: the green shading indicates the comparator is significantly better than the reference; the orange-shaded reference is significantly better than the comparator.
Is There an Effect of PtDAs with or without Probabilities on Outcomes?
Of 149 RCTs, 132 PtDAs presented probabilities, 13 did not, and 4 were unable to ascertain. The most common types of probabilities were numeric information plus pictograms (n = 60) and numeric information only (n = 28). There were too few RCTs to evaluate the effect on accurate risk perception. Compared with usual care, PtDAs with probabilities were significantly better for all of the other outcomes (see Table 3). Compared with usual care, PtDAs without probabilities were also associated with significantly improved knowledge, patients feeling more informed, and fewer patients having unclear values except for informed values choice and proportion undecided. Compared with PtDAs with probabilities, PtDAs without probabilities had significantly lower improvements in knowledge (MD −3.86; 95% CrI −7.67 to −0.03). There was no significant difference in the other outcomes.
Is There an Effect of Presenting Information Side by Side in PtDAs on Outcomes?
Of 149 RCTs, 85 PtDAs presented information on options side by side, 39 did not, and 25 were unable to ascertain. Compared with usual care, a PtDA with information on options presented side by side was significantly better for all outcomes, and a PtDA that did not present information side by side was significantly better for all outcomes except for proportion undecided (see Table 3). There was no statistically significant difference when PtDAs with information side by side was compared with PtDAs without this attribute for any outcomes.
Is There an Effect of Step-by-Step Guidance in Decision Making in PtDAs on Outcomes?
Of 149 RCTs, 102 PtDAs presented step-by-step guidance in decision making, 32 did not, and 15 were unable to ascertain. Compared with usual care, a PtDA with step-by-step guidance in decision making was significantly better for all outcomes, and a PtDA that did not have step-by-step guidance in decision making was significantly better for all outcomes except for proportion undecided (see Table 3). There was no statistically significant difference when PtDAs presented step-by-step guidance compared with PtDAs without this attribute for any outcomes.
Is There an Effect of a Personal Summary in PtDAs on Outcomes?
Of 149 RCTs, a personal summary of patients’ responses within the PtDA was provided in the PtDAs using a worksheet (n = 46), an automated summary (n = 43), or unable to ascertain (n = 1). Compared with usual care, PtDAs without a personal summary improved all outcomes (see Table 3). Compared with usual care, PtDAs with an automated summary improved knowledge, feeling uninformed, unclear values, and proportion undecided but showed no difference for informed values choice and accurate risk perceptions. Compared with PtDAs with automated summaries, those with worksheets improved knowledge (MD 3.29; 95% CrI 0.29, 6.27) and improved accurate risk perceptions (RR 1.71; 95% CrI 1.13, 2.81). Compared with PtDAs with automated summary, PtDAs without a personal summary resulted in fewer with unclear values (MD −4.42; 95% CrI −8.62, −0.21). There were no statistically significant differences for other outcomes.
Is There an Effect of Having a List of Questions or Encouragement to Discuss the Decision in PtDAs on Outcomes?
Of 149 RCTs, 15 provided a list of questions to consider or ask the clinician, 117 did not provide a list of questions, and 17 were unable to ascertain. Compared with usual care, PtDAs without questions were significantly better for all outcomes, and PtDAs that provided questions were significantly better for some outcomes (e.g., knowledge, feeling uninformed, unclear personal values). There was no difference between PtDAs providing questions compared with PtDAs without questions for any outcomes.
There were 114 RCTs of PtDAs that encouraged patients to discuss the decision, 22 did not encourage discussion, and 13 were unable to ascertain. Compared with usual care, PtDAs encouraging discussion were significantly better for all outcomes, and PtDAs that did not encourage discussion were significantly better for some outcomes (e.g., knowledge, feeling uninformed, and unclear values). There was no difference between PtDAs that encouraged discussion as compared with PtDAs that did not encourage discussion for any outcome; however, when PtDAs did not encourage discussion, fewer people felt uninformed (MD −11.26; −15.82, −6.73).
Is There an Effect of the Timing of PtDA Delivery on Outcomes?
Of 149 RCTs, 123 PtDAs were delivered in preparation for consultations, and 26 were used by the patient and clinician in consultations. There were too few RCTs to evaluate the effect on informed values choice. Compared with usual care, PtDAs delivered before consultations or during consultations were significantly better for all outcomes except undecided when used in the consultation (see Table 3). PtDAs used during consultations showed significantly lower improvements in knowledge (MD −4.34; 95% CrI −7.24, −1.43) and more patients felt uninformed (MD 5.07; 95% CrI 1.06, 9.11) compared with PtDAs used in preparation for consultations. There was no difference for other outcomes.
Is There an Effect of User Involvement in the PtDA Development or Testing on Outcomes?
Of 149 RCTs, 40 reported no users involved in PtDA development, 7 involved patients, 19 involved HCPs, and 83 involved patients and HCPs. Compared with usual care, a PtDA developed with the involvement of HCPs and patients or with no user involvement was better for all outcomes (Table 3). Findings were variable when HCP alone or patient alone was compared with usual care. Compared with involvement of HCPs alone, there were statistically significant improvements when patients were involved alone (MD 6.56; 95% CrI 1.10, 12.03), patients and HCPs were involved (MD 4.35; 95% CrI 0.96, 7.73), or no users were involved (MD 4.38; 95% CrI 0.65, 8.13). There were no statistically significant differences between the types of users involved for other outcomes.
Of 149 RCTs, 51 reported no users involved in PtDA testing, 48 involved patients only, 3 involved HCPs only, and 47 involved both patients and HCPs. Compared with usual care, when patients were involved alone or with HCPs, there were statistically significant improvements in all outcomes except undecided in PtDA testing involving patients and the HCPs on the team (Table 3). Compared with the involvement of patients alone, more felt uninformed (MD 5.29; 95% CrI 1.57, 9.02) and had unclear values (MD 4.93; 95% CrI 1.26, 8.61) when patients and HCPs were both involved. There were no statistically significant differences between users involved for other outcomes.
Is There an Effect of Using a Framework or Conceptual Model for PtDA Development on Outcomes?
Of 149 RCTs, the development of 98 PtDAs was informed by a theoretical framework or conceptual model. The most used frameworks were IPDAS only (n = 39), the Ottawa Decision Support Framework (ODSF) only (n = 20), ODSF plus IPDAS (n = 19), Informed Medical Decisions Foundation (IMDF) only (n = 9), and IMDF plus IPDAS (n = 2). Compared with usual care, the use of a framework/model or not was significantly better for any outcomes (see Table 3). When development was informed by a framework with or without IPDAS compared with IPDAS alone, there was improved knowledge (MD 3.98, 95% CrI 1.20, 6.76). There were no statistically significant differences for other outcomes.
Discussion
Our NMA used data from 149 RCTs that evaluated PtDAs compared with usual care. There was no difference in outcomes for PtDAs using implicit compared with explicit values clarification. There were greater improvements in knowledge when PtDAs included information on probabilities, PtDAs were used in preparation for the consultation, or development included patients on the research team. There was no difference in outcomes when PtDAs presented information in ways that decrease cognitive demand and mixed results when PtDAs used strategies to enhance communications. Our findings lead us to the following points of discussion.
Content Elements
The magnitude of improvement for knowledge changed depending on the content element being compared with usual care. When probabilities were used, there were greater improvements in knowledge but no difference in other outcomes. Given the variability in how probabilities were reported, further investigation is required to determine best practices. We were surprised to observe no difference between the use of implicit compared with explicit values clarification, given findings from Witteman’s IPDAS Values Clarification group that favored explicit values clarification. 6 An important difference between Witteman’s meta-analysis and our NMA was they included findings from RCTs evaluating explicit values-clarification methods and acknowledged their findings were heavily influenced by participants making hypothetical decisions. There was no difference in the use of strategies expected to decrease cognitive demand (e.g., presenting information side by side or guidance step by step) or the use of strategies to improve communication with others (e.g., personal summaries, list of questions, encourage discussion with others).
Timing of Delivery
When PtDAs were used in preparation for consultations versus during consultations, there were significantly greater improvements in knowledge and fewer people felt uninformed. This finding is not entirely surprising because patients who use a PtDA in preparation for the consultation have time to learn about their options and consider questions to ask in the consultation. In fact, when worksheets, typically used in preconsult PtDAs, were used to summarize personal information compared with automated summaries (used in a quarter of PtDAs), patients had improvements in their knowledge and accurate risk perceptions. However, previous RCTs showed variability in the degree to which patients used PtDAs in preparation for consultations. 21 Patients are more likely to use the PtDAs in preparation for consultations when they are given an explicit invitation and encouraged to use the PtDA. 22 When the PtDA is presented in the consultation, patients are often being informed of their options for the first time. This finding is concerning as patients may not have had the opportunity to fully process the information and hence be fully informed about their choices when PtDAs are used solely during a clinical encounter. Use during the consultation, often intended to facilitate shared decision making,23,24 was shown to be better than usual care for significantly improving patient-clinician communication using the observer OPTION-12. 1 Hence, the timing of PtDA delivery in relationship to the clinical consultation has the potential to influence outcomes differently.
Development Process
It is important to involve both patients and HCPs together on the PtDA development team, and reliance solely on HCPs leads to poorer patient knowledge. These findings are consistent with the guidance from the IPDAS best practices for development. 9 PtDA developers are encouraged to apply principles of user-centred design, which requires involvement of patients (including caregivers, family, surrogates) and HCPs in steps to understand users’ needs, develop the PtDA, and test the PtDA. 9 However, a realist review on the implementation of PtDAs discussed only the coproduction of PtDA content with HCPs. 22 Our findings showed that coproduction should, at a minimum, include patients on the PtDA development team. Interestingly, our findings for involvement of PtDA users (patients, HCPs, both) in the testing PtDAs conducted prior to the RCT did not show significant differences in the outcomes. These findings need to be interpreted cautiously given they may be influenced by inadequate reporting on user involvement in testing PtDAs or may not be relevant outcomes that were measured. 9
The use of a theoretical framework or conceptual model in the development of PtDAs showed mixed results. There were greater improvements in knowledge when development was based on a theoretical framework or conceptual model with or without IPDAS compared with IPDAS alone. For the included RCTs, it was unclear if IPDAS guided the PtDA development or if it was used as a checklist at the end of the study. The most commonly used theoretical framework was the Ottawa Decision Support Framework, 25 and authors of this framework were influential in the establishment of IPDAS. 26 Further research is required to determine the influence of theoretical frameworks or conceptual models on the effect of PtDAs on patient outcomes.
Strengths and Limitations
Our NMA was the first conducted to determine the contributions of the different attributes of PtDAs on patient outcomes given variations in PtDA content elements, timing of delivery, and the development processes. Included RCTs were evaluated across a large range of health care treatment and screening decisions. An important limitation in relying on the Cochrane systematic review of PtDAs was that eligible RCTs compared PtDAs to usual care and excluded RCTs comparing 2 types of PtDAs. Another limitation was the challenges of ascertaining the presence or absence of these attributes when they were not adequately described in the published articles and if we did not have access to the PtDAs. We extracted the PtDA attributes from PtDAs when available and detailed descriptions were included in the published articles. There may also have been nder-reporting of PtDA attributes, including patient involvement in their development, as indicated in previous RCTs.9,27 To advance the knowledge on the merits of different attributes of PtDAs, we recommend that authors of PtDA studies follow the SUNDAE guidelines to ensure the content and development of PtDAs are reported in adequate detail. 28 Finally, there were too few RCTs measuring some of the primary outcomes from the original systematic review to conduct the NMA on all primary outcomes, and it would be helpful to determine if there were intersecting features (e.g., if PtDA developers used several features compared with those that provided few or no additional features).
Conclusions
Our NMA of 149 RCTs evaluating PtDAs compared with usual care showed that some content elements of PtDAs, the timing of delivery preconsultation, and the involvement of patients in development were associated with higher patient knowledge. Although there were little or no differences on other outcomes, these are exploratory findings based on indirect comparisons, and the NMA findings could be strengthened by including findings from RCTs that directly compared 2 or more PtDAs with different attributes. Our findings support growing evidence on the importance of considering various attributes of PtDAs and the need for more robust NMA to evaluate the contribution of individual attributes.
Supplemental Material
sj-docx-1-mdm-10.1177_0272989X251318640 – Supplemental material for The Effect of Patient Decision Aid Attributes on Patient Outcomes: A Network Meta-Analysis of a Systematic Review
Supplemental material, sj-docx-1-mdm-10.1177_0272989X251318640 for The Effect of Patient Decision Aid Attributes on Patient Outcomes: A Network Meta-Analysis of a Systematic Review by Dawn Stacey, Meg Carley, Janet Gunderson, Shu-Ching Hsieh, Shannon E. Kelly, Krystina B. Lewis, Maureen Smith, Robert J. Volk and George Wells in Medical Decision Making
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely by a Project Grant from the Canadian Institutes of Health Research (PJT - 180385). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Ethical Considerations
This study was based on a systematic review of 209 RCTs included in the 2024 updated Cochrane systematic review of decision aids for people considering health care treatment or screening decisions. The study did not involve human participants and therefore did not require any review by an ethics board. This study was registered with Open Science Framework on July 1, 2024 (osf.io/64j52).
ORCID iDs
Data Availability
Data will be made available upon reasonable request to the authors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.