Abstract
Background
Economic evaluations of treatments increasingly employ price-threshold analyses. When a treatment has multiple indications, standard price-threshold analyses can be overly simplistic. We examine how rules governing indication-specific prices and reimbursement decisions affect value-based price analyses.
Methods
We analyze a 2-stage game between 2 players: the therapy’s manufacturer and the payer purchasing it for patients. First, the manufacturer selects a price(s) that may be indication specific. Then, the payer decides whether to provide reimbursement at the offered price(s). We assume known indication-specific demand. The manufacturer seeks to maximize profit. The payer seeks to maximize total population incremental net monetary benefit and will not pay more than their willingness-to-pay threshold. We consider game variants defined by constraints on the manufacturer’s ability to price and payer’s ability to provide reimbursement differentially by indication.
Results
When both the manufacturer and payer can make indication-specific decisions, the problem simplifies to multiple single-indication price-threshold analyses, and the manufacturer captures all the consumer surplus. When the manufacturer is restricted to one price and the payer must make an all-or-nothing reimbursement decision, the selected price is a weighted average of indication-specific threshold prices such that reimbursement of more valuable indications subsidizes reimbursement of less valuable indications. With a single price and indication-specific coverage decisions, the manufacturer may select a high price where fewer patients receive treatment because the payer restricts reimbursement to the set of indications providing value commensurate with the high price. However, the manufacturer may select a low price, resulting in reimbursement for more indications and positive consumer surplus.
Conclusions
When treatments have multiple indications, economic evaluations including price-threshold analyses should carefully consider jurisdiction-specific rules regarding pricing and reimbursement decisions.
Highlights
With treatment prices rising, economic evaluations increasingly employ price-threshold analyses to identify value-based prices. Standard price-threshold analyses can be overly simplistic when treatments have multiple indications.
Jurisdiction-specific rules governing indication-specific prices and reimbursement decisions affect value-based price analyses.
When the manufacturer is restricted to one price for all indications and the payer must make an all-or-nothing reimbursement decision, the selected price is a weighted average of indication-specific threshold prices such that reimbursement of the more valuable indications subsidize reimbursement of the less valuable indications.
With a single price and indication-specific coverage decisions, the manufacturer may select a high price with fewer patients treated than in the first-best solution. There are also cases in which the manufacturer selects a lower price, resulting in reimbursement for more indications and positive consumer surplus.
Keywords
Prices of new treatments developed for conditions from cancer to heart disease are rising.1,2 For payers and health systems interested in efficiently achieving better health for their populations, price negotiation and value-based pricing are essential. To support such negotiations, evidence in the form of price-threshold analyses is an important feature of economic evaluations, especially those conducted to assess treatments with potentially large budget impacts.
For treatments with a single indication (i.e., disease, condition, or patient population), the threshold analysis is straightforward: the price of treatment is set such that the incremental cost-effectiveness ratio is equal to the payer’s willingness-to-pay threshold. 3 When there are multiple indications but separate prices can be charged for each, the analysis is again straightforward, as it is separable by indication. Such separate threshold analyses will identify higher threshold prices for indications with more per-patient value (i.e., those with higher incremental net monetary benefit at any given treatment price), and for each indication, there is no consumer surplus for the payer. While indication-specific pricing requires a straightforward analysis, it is potentially difficult and costly in terms of administrative infrastructure to implement in practice.4–6
Price-threshold analyses can be substantially more complicated for treatments with multiple indications considered for reimbursement in systems or situations in which it is not feasible or permissible to pay separate indication-specific prices.4,5,7 In such cases, a single price is required for all indications, raising the question of what single price should be charged.
Similarly, payers may be able to use preauthorized reimbursement or restricted formularies to enforce indication-specific reimbursement decisions. If indication-specific reimbursement decisions are possible, payers may be able to obtain consumer surplus. 3 However, there may also be situations in which the financial or political costs of implementing such mechanisms for differentiating indications may be too high or where it may be unethical to differentiate subpopulations, resulting in an all-or-nothing reimbursement decision. A single price based on the average incremental cost-effectiveness of each indication generates no consumer surplus and requires both the payer and the manufacturer to have confident and shared estimates of the number of people who will access each indication.3,4,8,9
There are multiple real-world examples of situations where a single therapy has multiple indications but where differential pricing and reimbursement by indication may be more or less feasible. These range from a therapy used for the same disease with patients of different characteristics or severity, a therapy used for the same pathogen that causes different diseases across patients groups, or a therapy used to treat distinct diseases across distinct patient populations. For example, human papillomavirus (HPV) vaccination can prevent a variety of cancers for both females and males. However, given higher rates of cancers, specifically uterine cervical cancer, HPV vaccination costs less per quality-adjusted life-year (QALY) gained for girls than for boys.10–12 Direct-acting antiviral therapies for chronic hepatitis C virus (HCV) can arrest the progression of liver fibrosis, preventing advanced liver diseases (ALD). However, given that ALD will occur more rapidly and with higher likelihood for chronic HCV-infected 50-y-olds with moderate liver fibrosis than those without liver fibrosis, the cost per QALY gained is lower for the former group than the latter. 13 Chimeric antigen receptor T-cell (CAR-T) therapies are effective in treating a variety of otherwise refractory cancers but can cost hundreds of thousands of dollars for a single dose. The cost per QALY gained is lower for CAR-T treatment of relapsed B-cell acute lymphoblastic leukemia in children than for relapsed diffuse large B-cell lymphoma in adults in part because of the larger number of QALYs that can be gained by preventing a cancer death in a child than in an older adult.14,15
Whether a payer can and should negotiate separate prices by indication or make separate reimbursement decisions in these examples and other cases is relevant for population health gains, the efficient use of limited health care resources, and fairness to patient populations. We focus on a setting with a single public payer using health economics to inform coverage decisions. Our study uses game theory to analyze the optimal pricing and reimbursement decisions for settings in which the pricing and/or reimbursement decisions may be required to be the same across all indications. Furthermore, we evaluate the welfare implications of these restrictions as well as their effects on manufacturer profit and consumer surplus.
Methods
We analyze a 2-stage game between 2 players: the pharmaceutical manufacturer and the payer who purchases treatment(s) on behalf of patients. In the first stage, the manufacturer selects a price or indication-specific prices for the treatment. In the second stage, the payer decides whether to reimburse patients for the treatment at the offered price(s) for 1 or more possible treatment indications. We index the indications with
Decisions
The game involves a sequence of decisions: pricing decisions made by the manufacturer and then reimbursement decisions made by the payer.
The manufacturer chooses the price for each indication,
The payer then chooses whether or not to reimburse the treatment at the manufacturer’s offered price(s). We denote the payer’s decision for each indication with
There are 3 variations of this game that may occur in practice (Table 1). First, both the manufacturer and the payer may be able to make indication-specific decisions. The manufacturer may be able to offer different prices because of very different dosing or different means of delivery, and the payer may similarly be able to easily differentiate how the product is being used on that same basis. Second, the manufacturer is restricted to a single price, and the payer is restricted or uninterested in making an indication-specific decision due to difficulty or cost associated with verifying the specific indication or due to the ethical dimensions of providing differential access. Third, the manufacturer is restricted to a single price, potentially because of a common formulation and dose range, but it may be possible for the payer to use preauthorized approval to impose different reimbursement decisions by indication. We do not study the fourth possible case, in which the manufacturer may use indication-specific pricing and the payer is limited to a single reimbursement decision. We do not believe this case is realistic as the payer could use the same basis as the manufacturer for making an indication-specific decision. Furthermore, if the same reimbursement decision for all indications is the optimal action, it will be identified as the solution to the more general problem.
Policy-Relevant Scenarios in which the Manufacturer May Have the Ability to Price Differently by Indication and the Payer May Have the Ability to Make Different Decisions by Indication

Objective Functions
The manufacturer seeks to maximize profit
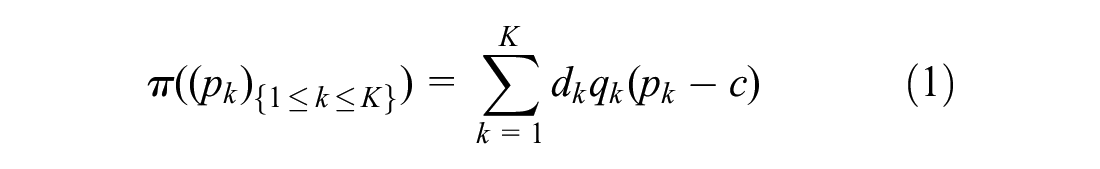
For simplicity, we use a linear profit function of the per-patient prices charged for each indication, the quantities of each treatment sold
The payer seeks to maximize total population incremental net monetary benefit
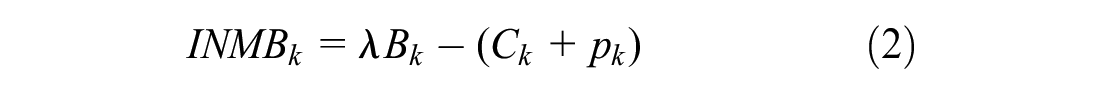
where
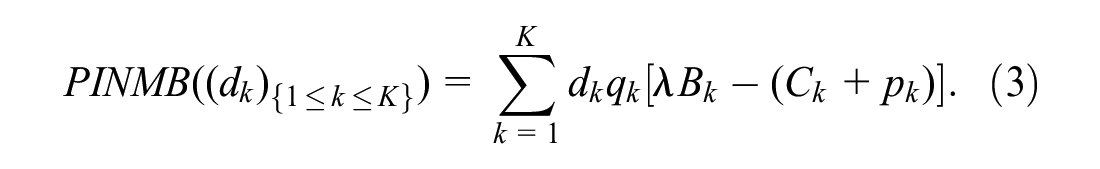
Note that the
The total social welfare,
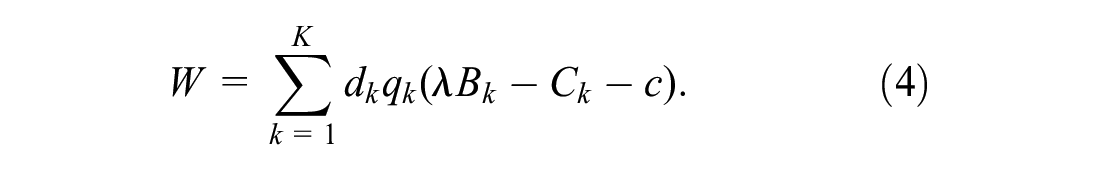
Participation Constraints
The participation constraints for the manufacturer and the payer respectively are
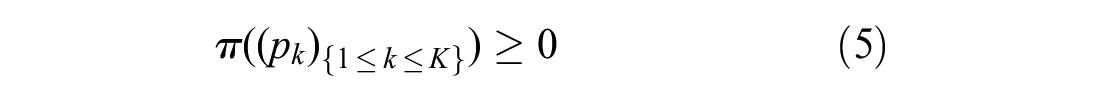
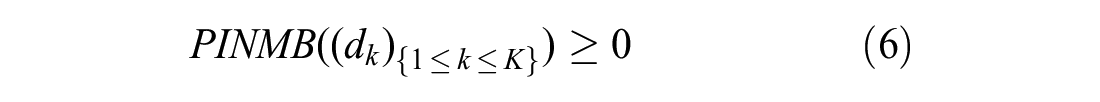
If the participation constraints are not satisfied, then either the manufacturer will opt not to sell the treatment as it is unprofitable to do so or the payer will opt not to reimburse use of the treatment as its overall incremental costs are larger than the value of its overall incremental benefits at the payer’s willingness-to-pay threshold.
Numerical Analysis
To assist in the visualization of our results, we graphically present a numerical analysis for a case with 2 indications, that is,
Results
Social Welfare Maximizing Policy
We first identify the reimbursement policy decision that will maximize social welfare,
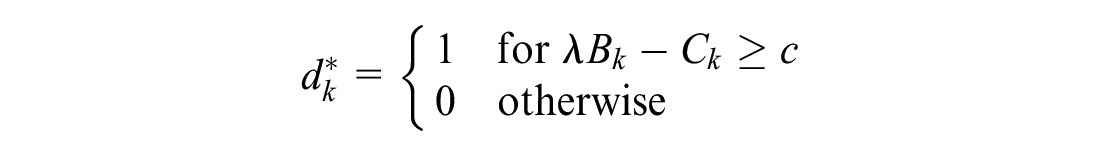
This set of decisions provides patients access to all indications for which there is potential for positive net benefit. We refer to this policy as the “first-best,” and we compare other policies resulting from the 2-stage game between manufacturers and payers to this policy outcome in later sections.
Indication-Specific Prices and Reimbursement Decisions
In this section, we first focus on the interesting case in which the incremental net benefit of treatment, excluding the cost of treatment, is at least equal to the marginal cost of producing the treatment, that is,
When the offered prices can be indication specific, the range of feasible prices at which both the manufacturer and payer wish to participate can be evaluated separately and independently for each indication. Based on participation constraints, feasible price ranges exist, for each indication
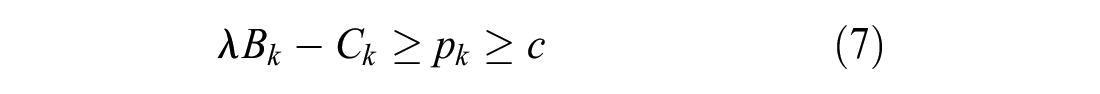
Because the manufacturer’s profit function is linear in each
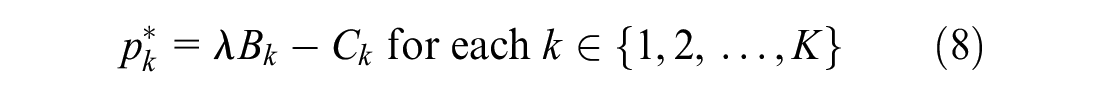
For any indication for which the incremental net benefit of treatment, excluding the cost of treatment, is not at least the marginal cost of production, that is,
Ultimately, this situation provides the first-best policy, achieving the highest total welfare, and providing patients access to all indications for which there is potential for positive net benefit, that is,
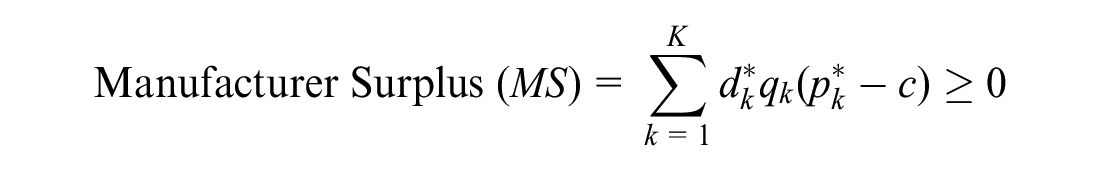
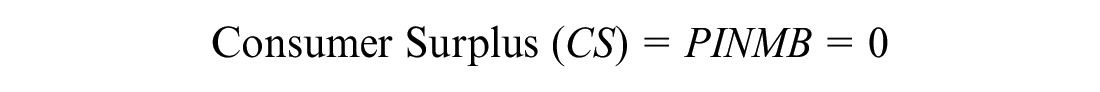
This solution is illustrated when
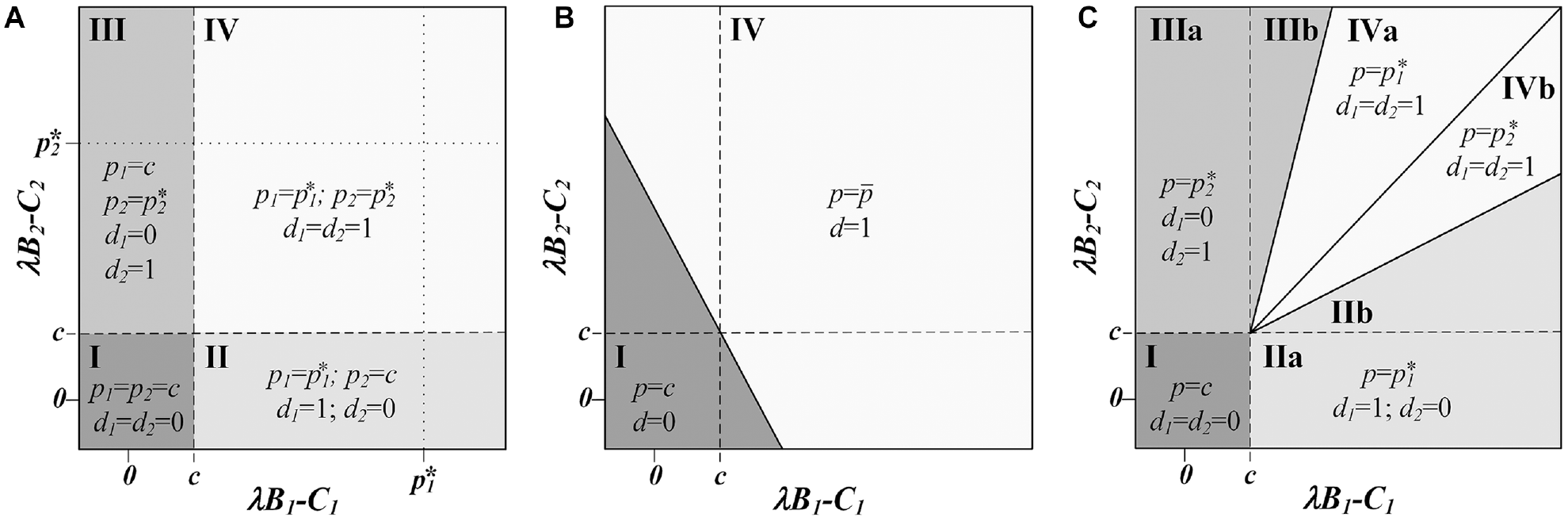
(a) Optimal pricing decisions by the manufacturer, (p1, p2), and optimal reimbursement decisions by the payer,
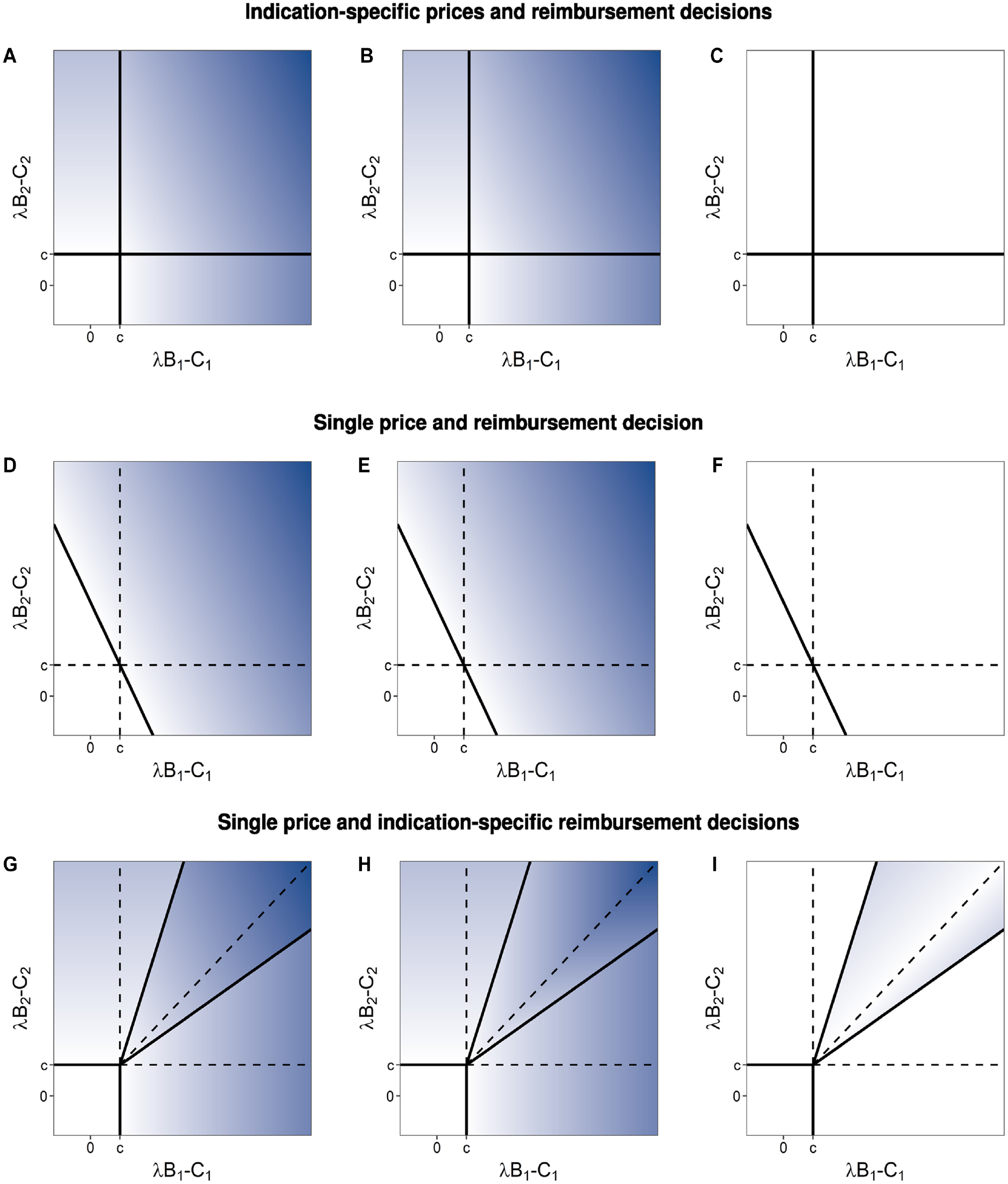
Total welfare (column 1, panels a, d, and g), manufacturer profit (column 2, panels b, e, and h), and consumer surplus (column 3, panels c, f, and i) for indication-specific prices and reimbursement decisions (row 1, panels a–c), single price and reimbursement decision (row 2, panels d–f), and single price and indication-specific reimbursement decisions (row 3, panels g–i). Color scale is set such that white is zero and a greater intensity of blue represents increasingly positive values.
Single Price and Single Reimbursement Decision for Multiple Indications
For multiple indications, the single treatment price and single reimbursement decision constraints can be formalized as


To be feasible from the perspective of the manufacturer,
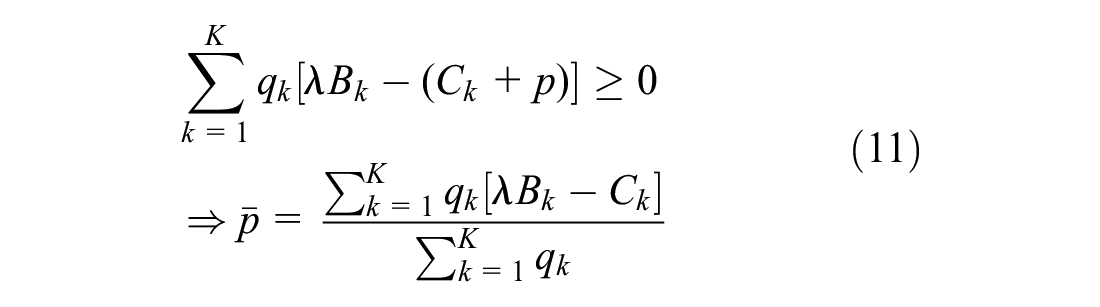
Equation 11 identifies that the maximum price the payer is willing to accept is a weighted average of the value of each indication, with weights determined by the market size for each indication.
As in the previous setting, because the manufacturer’s profit function is linear in
Whenever there is a mutually acceptable price, because it is always the payer’s maximum acceptable price, the manufacturer captures all of the surplus, and there is no consumer surplus.
This solution is illustrated when
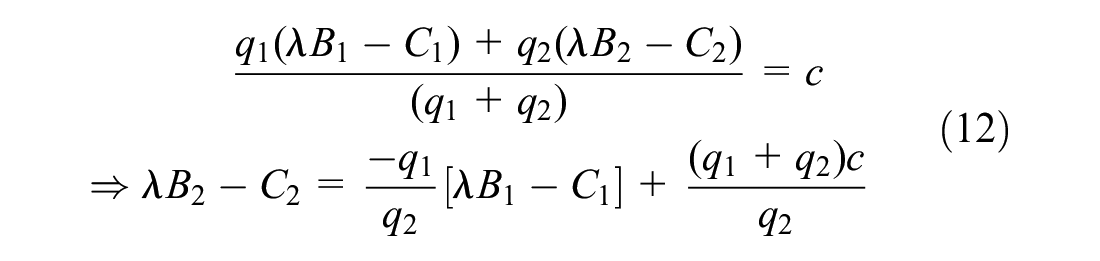
Comparing this setting to that of indication-specific prices and reimbursement decisions, the additional constraints of a single price and a single reimbursement decision affect outcomes for both the manufacturer and the payer (Figure 3a–c). Regions II and III from Figure 1a are now each split into 2 parts. One of these parts joins region IV, in which the payer reimburses both indications, providing patients access to the less valuable indication, decreasing social welfare, and decreasing the profits of the manufacturer even as it increases the units sold. The second part joins region I, in which the payer now decides not to reimburse for either indication, removing patient access to the more valuable indication and thereby decreasing social welfare and manufacturer profits. In both altered regions, the payer is not better or worse off, as they do not obtain any consumer surplus in either situation, but the total social welfare is reduced relative to that obtained in the corresponding parts of regions II and III of Figure 1a.
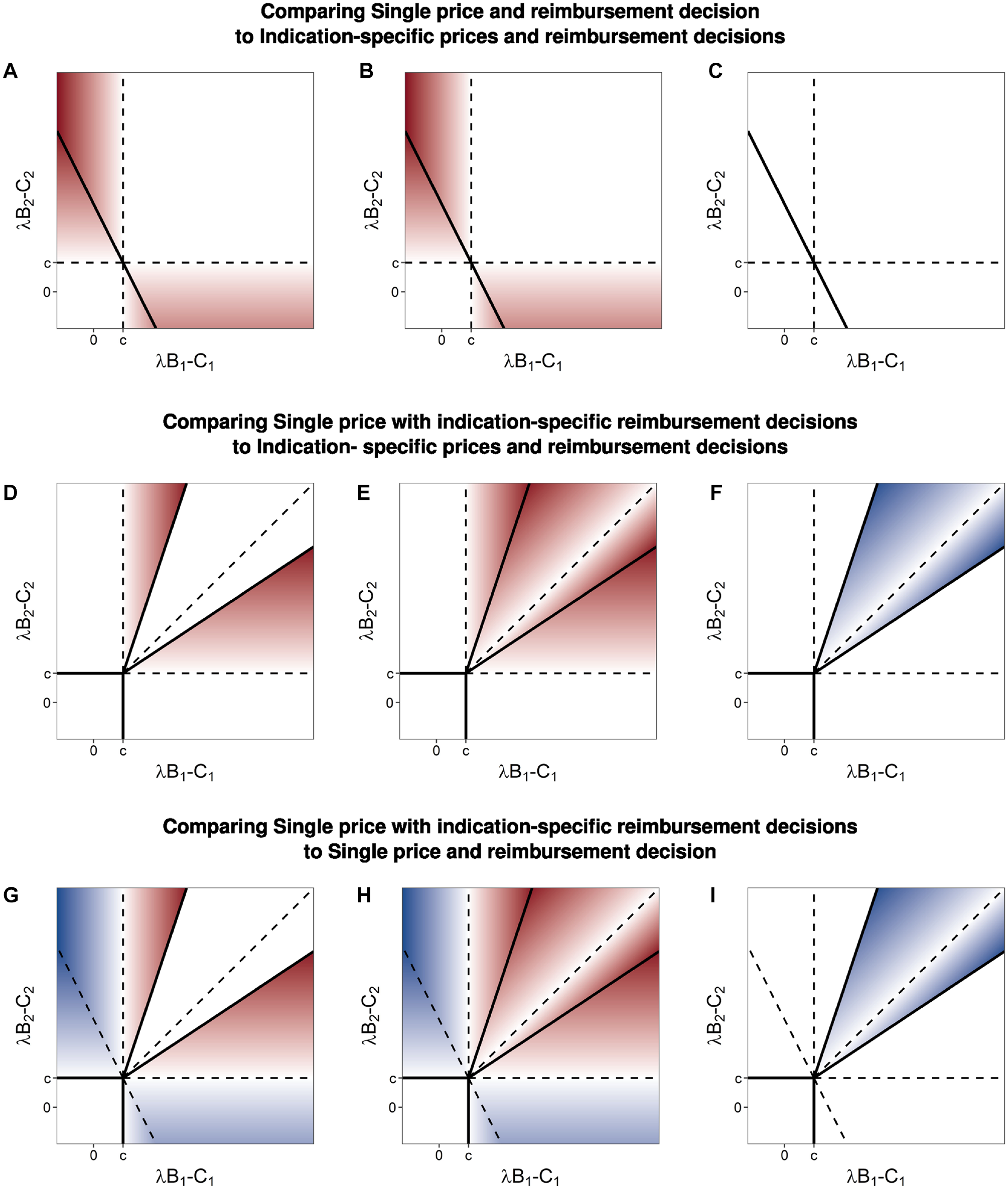
Differences in: (a, d, and g) total welfare (column 1), (b, e, and h) manufacturer profit (column 2), and (c, f, and i) consumer surplus (column 3) comparing games with single and indication-specific pricing and/or reimbursement decisions. Color scale is set such that white is zero, greater intensity of blue represents increasingly positive values, and greater intensity of red represents increasingly negative values.
Single Price with Indication-Specific Reimbursement Decisions
We now focus on the situation in which there is a single price for all indications and indication-specific reimbursement decisions. In this section, it is useful to rank potential indications based on their incremental net monetary benefit excluding the cost of treatment, that is,
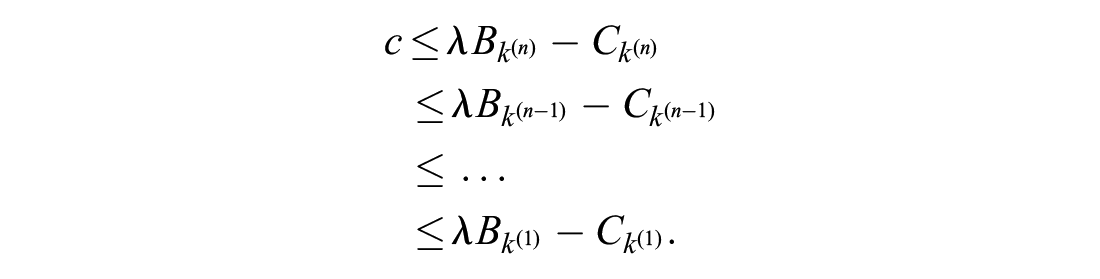
With a ranked list of the treatment indications, it is clear to see that the payer has
The manufacturer may prefer to select a price lower than the threshold price for indication
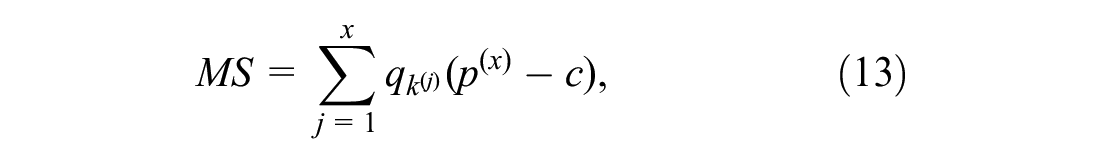
and the consumer surplus is
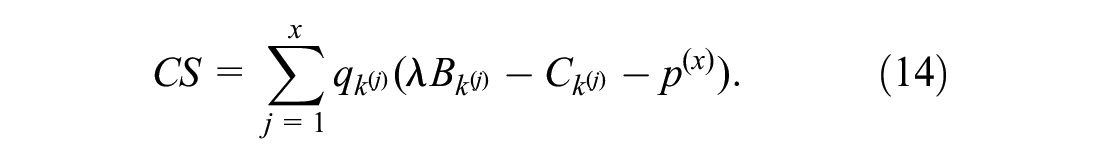
Focusing on 2 possible prices,
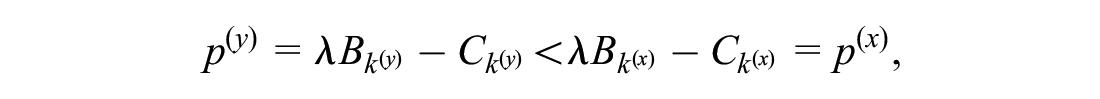
the manufacturer prefers the lower price
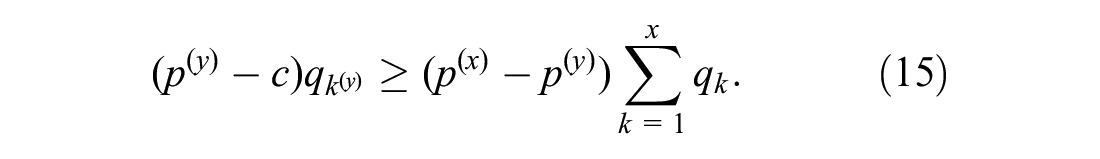
The left side of the inequality represents additional profit from an increase of
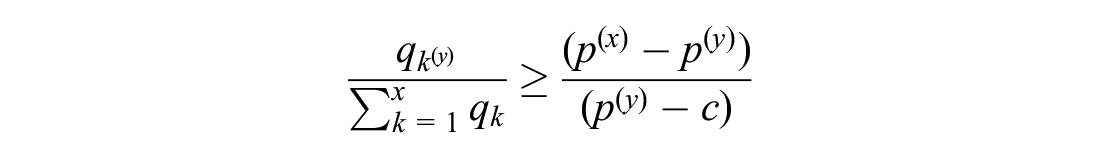
Therefore, in their effort to maximize profit, the manufacturer prefers the lower price if the percentage increase in quantity sold when selecting the lower price exceeds the percentage change in profit margin when selecting the higher price.
This solution is illustrated when K = 2 in Figures 1c and 2g to i with 4 policy regions: (I) in which the manufacturer’s minimum price is greater than the incremental net monetary benefit for both indications, (II and III) in which the manufacturer secures a positive reimbursement decision for only 1 indication for which it can charge the threshold price of that indication, and (IV) in which the manufacturer secures a positive reimbursement decision for both indications. Region IV is further divided into 2 sections, each in which the price selected to secure the “reimburse all” decision is the threshold price for the lower-valued indication.
Comparing this setting to that of indication-specific prices and reimbursement decisions, there are several differences for the manufacturer and the payer (Figure 3d–f). First, regions II and III, in which only 1 indication is reimbursed, are larger. In the regions of Figure 1c labeled IIb and IIIb, the payer selects the threshold price of the higher-valued indication, forgoing market access to the lower-valued indication, because the incremental sales that would come from the lower-valued indication do not offset the lost revenue from the reduced price charged for the higher-valued indication. In these regions, the outcome of the game sequence is no longer the first-best solution for society (Figure 3d). Compared with the first-best solution, total welfare is lower because there is no reimbursement for an indication that has the potential to provide positive net benefit (at some price satisfying the participation constraints). Compared with the setting in which there are both indication-specific prices and decisions, manufacturer profits are reduced. In both settings, the payer receives no consumer surplus in these regions.
In the regions labeled IVa and IVb in Figure 1c, the manufacturer offers the threshold price associated with the lower-valued indication maximizing profits through a larger total volume of sales. In these regions, the policy outcome of the game sequence is the first-best solution for society. Compared with the setting in which there are both indication-specific prices and decisions, manufacturer profits are reduced, with the surplus now shared between the manufacturer and the payer (Figure 3e and f).
The line separating regions IIb and IVb (and similarly regions IIIb and IVa) is identified by solving for the manufacturer’s indifference point where they make the same profit from selling only the higher-valued indication at the higher price as they do selling both indications at the lower-valued indication’s threshold price.
Line separating region IIb and IVb:
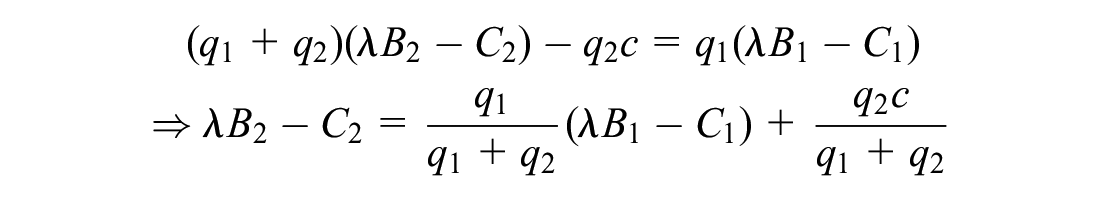
Line separating region IIIb and IVa:
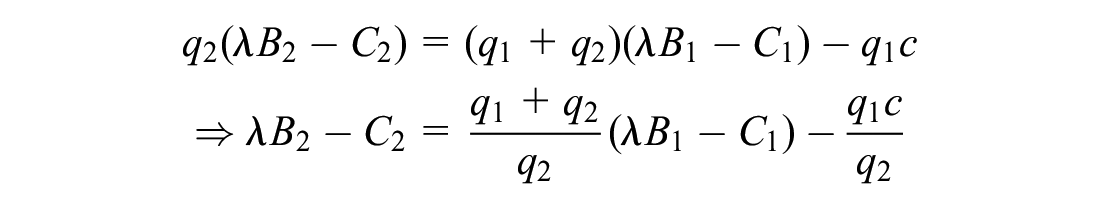
These equations reveal the importance of the relative size of each indication’s treatment population. If the higher-valued indication has a relatively large treatment population, the manufacturer will offer the higher price, expanding region IIb (or IIIb). If the higher-valued indication has a relatively small treatment population or difference in value between the 2 indications is relatively small, the manufacturer is willing to forgo some otherwise obtainable revenue for the higher-value indication in favor of sales of both indications, enlarging regions IVa (or IVb).
Comparing Outcomes with a Single versus Indication-Specific Reimbursement Decisions
Comparing outcomes with a single price and indication-specific reimbursement decisions to that of a single decision for all indications provides insight into which game is preferred by manufacturers and payers (Figure 3h–i). In regions labeled IIa and IIIa in Figure 1c, indication-specific decisions are preferred by the manufacturer because otherwise the lower-valued indication negatively affects the weighted-average single price, lowering manufacturer profits. The payer has the same consumer surplus in both settings, but when there are indication-specific decisions, all indications for which there is positive net monetary benefit, excluding the cost of treatment, are reimbursed, allowing patients from more indications access to effective treatments and increasing overall welfare.
When both indications have positive incremental net monetary benefit, excluding the cost of treatment, the manufacturer always prefers a single decision. In regions labeled IIb and IIIb on Figure 1c, this is because the manufacturer optimizes their profit by choosing the higher-value indication’s threshold price, forgoing market access to the lower-value indications sales and, overall, having less profit than would occur with a single decision. In regions labeled IVa and IVb on Figure 1c, the manufacturer prefers a single decision because the manufacturer maximizes their profit by choosing the lower-valued indication’s threshold price, and while this results in market access for both products, it does so at a lower price than the weighted average price that occurs when the payer must make a single decision.
In regions IIb and IIIb, the payer has the same consumer surplus (none) regardless of whether they make indication-specific decisions or not. However, in these regions, social welfare is higher because both indications are ultimately reimbursed when there is a single decision. In contrast, in regions IVa and IVb, the payer strictly prefers indication-specific decisions. In these regions, the same policy decision is made in both settings, leading to the same overall welfare. However, in the setting of indication-specific decisions, the manufacturer selects a lower price to ensure that both indications are given market access.
Discussion
Our study analyzes 3 different 2-stage pricing/reimbursement games, in which the pricing and/or reimbursement decisions may be required to be the same across all indications. Our study shows that value-based reimbursement decisions are substantially more complex than a simple price-threshold analysis when a given treatment has more than 1 indication and if either indication-specific pricing or indication-specific reimbursement decisions are disallowed. The implication is that, when a treatment has more than 1 indication, analysts should be cautious in claiming that because a treatment becomes cost-effective for a specific indication at an identified price threshold, the manufacturer should, therefore, alter the price accordingly and that, likewise, the payer should not reimburse the treatment for that indication unless this is done. Instead, caution and nuance are warranted when separate indication-specific prices and indication-specific reimbursement decisions are not permitted.
Our analysis shows that the socially optimal outcome, in which patients have access to all treatments for which the incremental net monetary benefit, excluding the marginal cost of production, can be guaranteed only when every treatment, or treatment-indication pair, is priced commensurate with its own incremental net monetary benefit. Practical, logistical, or ethical considerations can effectively impose limits on whether a manufacturer or a payer can make indication-specific decisions (Table 1). In situations in which a manufacturer is effectively restricted to a single price because its product has the same formulation, a similar dosing range, and the same routes of administration across indications, payers may be able to choose between making a single reimbursement decision or indication-specific decisions using mechanisms such as preauthorized reimbursement schemes or restricted formularies. However, there may also be situations in which the financial or political costs of implementing such mechanisms for differentiating indications may be too high or in which it may be unethical to differentiate subpopulations.
In the standard, single-indication value-based pricing analysis, the price is set, irrespective of the size of the patient population, such that the manufacturer absorbs all of the gains, and there is no consumer surplus. The extension of these separate, indication-specific pricing and reimbursement decisions yields the same results for each indication. However, when indication-specific pricing or reimbursement decisions are not possible, our study shows that using a price representing the average value across indications, weighted by the relative size of each indication’s patient population, can achieve the first-best reimbursement policy when all indications contribute positive net incremental benefit excluding the cost of treatment.
However, the feasibility of successful implementation of such a weighted-average single price is potentially vulnerable to asymmetries of information between the manufacturer and the payer, to uncertainty in the treatment effectiveness as well as the size of the treatment population, to the possibility of new entrants into the marketplace, or to gaming by the manufacturer with respect to strategic sequential introduction of indications. For example, the manufacturer may strategically launch higher value indications with relatively smaller patient populations to market first, yielding a higher price, then release clinical evidence and seek market access for lower value indications treating relatively larger patient populations. 21 Reimbursement decisions are notoriously difficult to reverse, and so manufacturers may maintain market access, once obtained, even when the price is inefficient based on the realized patient mix across indications. 22
One key challenge is in the estimation of the quantity demanded for each indication,
A single price and reimbursement decision scheme is also susceptible to innovation, affecting the indication mix over time. Changes to the indication mix can happen in numerous ways. First, through additional study, evidence may emerge that the treatment is less effective for 1 or more indications than initially estimated or that there is a previously unknown lower-value indication. In some situations, it may be possible for a manufacturer to strategically lead with market introduction of higher-valued indications, garnering a high price, and only then introduce evidence supporting use of the product in lower-valued indications. Second, a new product can enter the market replacing the optimal treatment or influencing the incremental value for 1 or more indication. Because pricing in this setting is linked across indications, the entrance of a new treatment relevant to a subset of the indications in the original game can disrupt the price it determined. This disruption can have surprising and unintended effects, such as leading to the loss of reimbursement for other (lower-value) indications, that perhaps have no direct relationship to the new treatment.
For example, bevacizumab was first approved for the treatment of advanced colorectal cancer in 2004 with new indications identified over time, 26 which could have destabilized other prices or market access in a strict single price and single reimbursement decision situation. Regardless of how the indication mix changes over time, the payer may end up overpaying for the remaining indications for which the treatment remains in use unless there is an ongoing, value-based market access and price negotiation. However, reimbursement decisions are notoriously difficult to reverse, giving payers little power to effectively renegotiate prices. 27 As a result, even when our analysis indicates payers may be indifferent between indication-specific and single reimbursement decisions scenarios, other practical considerations not captured in our model may lead payers to strictly prefer indication-specific decisions.
While our study analyzes each of the 3 price/reimbursement games separately, in principle, decisions by either party or negotiations between them could determine which of these games to engage in, effectively generating a unified 3- or 4-stage game or a series of sequential games. For example, the manufacturer may choose indication-specific pricing and present the decision problems as unrelated in an effort to secure a higher price for a higher-valued indication (e.g., bevacizumab and ranibizumab for neovascular macular degeneration28,29). Similarly, manufacturers may present a single decision problem when the payer is better off making indication-specific decisions to select only indications with positive incremental net benefit (e.g., tumor-agnostic chemotherapies 30 ). Ultimately, when the manufacturer is restricted to a single price, there is no guarantee of achieving the socially optimal policy. When the payer can make indication-specific reimbursement decisions, we have shown that the manufacturer may select a price to maximize profit that will lead to some potentially good-value indications being excluded from reimbursement. In contrast, when the indications have relatively similar value, the selection of a lower price aimed at ensuring a positive reimbursement decision for all indications leads to the first-best policy outcome, albeit at a lower profit for the manufacturer as the payer accrues some consumer surplus. In this latter case, it is easy to see that the manufacturer may prefer 1 of the 2-stage games over the others. In addition to the considerations discussed in the previous paragraphs, our analysis has a number of other limitations. It makes a number of simplifying assumptions that avoid complicating the mathematical exposition but do not affect the implications of our findings. First, we consider only linear profit functions. Second, we assume the marginal cost of production is the same for all indications. Third, we assume that all parameters in our analysis are known and constant. Uncertainty in the effectiveness of 1 or more indication may exceed the payers’ willingness to accept risk; more information may be warranted. 31 This is another setting in which indication-specific decisions may be preferred by both manufacturers and payers, as to not have uncertainty in some indications affect the timeline for access to treatment for other indications. We do not consider strength of negotiating power, the role of international markets, competition from similar treatments (e.g., drugs in the same class), or decision uncertainty as factors influencing the offered price.3,32 We do not consider pay-for-performance/risk-based reimbursement schemes or temporary coverage decisions conditional on additional evidence.22,33–35 We do not consider the possibility of the manufacturer influencing the number of units sold using marketing effort (e.g., physician detailing or direct-to-consumer advertising). 9 Finally, we limit our focus to a setting with a single payer using health economics to inform coverage decisions and do not consider concurrent and sequential price negotiations with multiple competing insurance providers.36,37
Our analysis and findings apply to a broad range of decisions beyond those regarding drugs or other treatments that are effective across numerous diseases. It also applies to cases in which indications are not separate diseases but rather subgroups of individuals defined by their risk level or severity of illness within a given disease or condition. For example, treatment with a drug for stage 2 hypertension may yield greater
Our analysis also contributes to the literature on value-based pharmaceutical pricing, indication-specific pricing,4,5,20 and applying strategic game theory to value-based pharmaceutical pricing. Coyle et al. 38 presented patient population stratification and limited use criteria to restrict access to care to those populations in whom treatment is cost-effective. Zaric 39 considered the ability of a manufacturer to select both the price and the patient populations with access via limited use criteria. Zaric and O’brien 40 considered optimal pricing using a risk-sharing agreement with total budget constraint, Zaric and Xie 41 evaluated the impact of 2 different types of risk-sharing agreements on pricing and access decisions, and Zhang et al. 8 identified optimal prices for a price-volume risk-sharing agreement with asymmetric information about market size. Critchley and Zaric 9 used a game theoretic approach to study pricing and access policies incorporating the manufacturer’s marketing effort to increase sale volume. Extending the multiple-indication pricing framework we presented in this analysis to include the strategic dynamics of risk-sharing agreements, uncertain market size and marketing effort, uncertainty in treatment effectiveness, and strategic sequencing of market entry by manufacturers may constitute fruitful directions for future research.
Conclusions
For economic evaluations, price-threshold analyses are simple only when treatments have a single indication or where pricing and reimbursement by indication are permitted and feasible. However, this is infeasible in many jurisdictions and settings. Furthermore, many treatments have multiple indications in terms of providing benefits to people with different diseases or in terms of providing benefits to several subgroups of patients with the same disease whose expected magnitude of benefit and/or costs differ. The result is that at any given price, the treatment has a different incremental cost-effectiveness ratio for each of these groups. In such situations, the inability to price by indication or to reimburse by indication makes the analysis much more complex and nuanced. Hence, analysts should be cautious when conducting economic evaluations that include treatments with multiple indications. Likewise, policy makers should be cautious in implementing rules that link pricing or reimbursement decisions across indications as they can have unintended, negative implications for social welfare and health equity.
Supplemental Material
sj-pdf-1-mdm-10.1177_0272989X231197772 – Supplemental material for Pricing Treatments Cost-Effectively when They Have Multiple Indications: Not Just a Simple Threshold Analysis
Supplemental material, sj-pdf-1-mdm-10.1177_0272989X231197772 for Pricing Treatments Cost-Effectively when They Have Multiple Indications: Not Just a Simple Threshold Analysis by Jeremy D. Goldhaber-Fiebert and Lauren E. Cipriano in Medical Decision Making
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lauren Cipriano is supported by a Canada Research Chair in Healthcare Analytics, Management, and Policy (Tier 2) (CRC-950-233070) and a Discovery Grant from the Natural Sciences and Engineering Research Council of Canada (RGPIN-2020-06665).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
