Abstract
Past research with North American participants has demonstrated a naturalness bias in the medical context: people prefer natural drugs to synthetic drugs under a variety of situations. Does such a bias exist in other countries (such as China) where cultural values and practices are quite different from those in the United States? We conducted 3 studies (N = 1,927) to investigate the naturalness bias with drugs and vaccines across cultures with American, Canadian, and Chinese participants. In studies 1A and 1B, participants chose or rated drugs (natural v. synthetic) for a hypothetical medical issue. The drugs were presented as having identical effectiveness and side effect profiles. Study 2 focused on a different medical context, vaccines, and required participants to rate their likelihood of taking vaccines (made from either more natural or more synthetic ingredients) for a harmful virus. The naturalness bias occurred across cultures in studies 1A and 1B, although it was not significant among Chinese participants in study 1B. In study 2, Chinese participants showed a stronger naturalness bias than Americans did, and safety concerns mediated the effect. Perceived safety accounted for the naturalness bias among Americans and Canadians, but did so only among Chinese in study 2. Overall, the results suggest that the naturalness bias in drug and vaccine decision making occurs across cultures, but Chinese participants may be more sensitive to the medical context.
Highlights
The naturalness bias — preferring natural to synthetic drugs or vaccines — occurred across cultures (Americans, Canadians, and Chinese).
Chinese participants showed a stronger naturalness bias than Americans did when the medical context was focused on vaccination, and safety concerns mediated this effect.
The naturalness bias may influence medical decision making across cultures, but Chinese participants may be more sensitive to naturalness in a vaccine context.
Items that are labeled “natural” are commonplace (e.g., natural beauty products, natural vitamins, natural drugs, etc.). The term can reflect the ingredients or processes involved in the creation of an item. Yet, the use of the term might also be motivated by attempts to benefit from people’s belief that natural things are better, healthier, or safer than unnatural, synthetic, or artificial things. These types of beliefs have been called the “naturalness bias” or the “natural is better” bias, defined as people’s tendency to prefer natural things even when they are chemically identical to synthetic alternatives.1,2
To the best of our knowledge, Baron et al. 3 was the first to formally examine the preference for naturalness. They found that obstetricians preferred a natural to synthetic hormone replacement therapy for a hypothetical patient. Since then, research has shown this bias in a host of areas such as with food, 4 drugs,5–7 beauty products, 8 and physical environments. 9 Furthermore, some work reveals that the preference extends to children aged 5 to 10 y when considering foods and drinks. 10
Researchers have proposed a couple of reasons for the naturalness bias.2,11 One is the general belief people hold that natural things are inherently more positive than nonnatural or synthetic things. A second, related hypothesis is that people may perceive greater safety in natural things than in synthetic things. The idea that “natural” has positive connotations and that safety is important in medical decision making coincides with some reasons why people use complementary and alternative medicines (CAM) such as yoga, meditation, prayer, and natural remedies.12,13 For example, concerns about the side effects of traditional medicine and treatments were reported to be one reason why cancer patients used CAM procedures such as natural medicines. 14
Several studies have examined the naturalness bias with drugs. For example, when told explicitly that natural and synthetic drugs are identical in effectiveness and safety, the majority of participants preferred natural to synthetic drugs to treat hypothetical medical issues and in behavioral choices when offered “free samples.”5,6 This naturalness preference, even in the context of having a synthetic alternative with identical safety and effectiveness, might have negative consequences for medical decision making. For example, people who preferred natural to synthetic drugs had more negative attitudes toward vaccines—which are synthetized—and were less likely to actually get a flu vaccine.1,15 Furthermore, physicians have been shown to exhibit the bias in hypothetical situations, 16 and some people show such a bias even when told that a natural drug option was less safe or less effective than a synthetic drug option. 5
The naturalness bias with drugs may affect decision making in the medical realm, with potential positive or negative outcomes. For example, some people may be less likely to critically evaluate safety data for drugs described as natural, which could lead to detrimental decisions if a safer synthetic alternative is available. However, if a drug, which is described as natural, has a similar safety and efficacy profile as a synthetic drug, people might be more amenable to engaging in a pharmacologic regimen involving the natural drug because they perceive there to be a reduced risk of side effects.
Cross-Cultural Comparisons
Given the crucial implications of a naturalness bias in the drug domain and its broad applications in various other domains, it is important to examine its robustness across cultural groups. Yet, to the best of our knowledge, there has been little cross-cultural research on the naturalness bias. Continental Europeans (including participants from the United Kingdom, France, Germany, Italy, and Switzerland) and Americans had a similar understanding of the meaning of the word natural, although the former had a stronger opposition to genetic engineering food than Americans did. 17 This study involved Western participants only and examined definitions around the word natural. We are interested in examining the impact of the naturalness bias on drug and vaccine decision making by Chinese and North American participants.
There are reasons to expect cultural differences in the naturalness bias with drugs and vaccines. Safety concern appears to be a reason for the naturalness bias. 2 Past research has shown that Chinese participants have a stronger prevention focus—a motivational state of being cautious and concerned about safety—compared with North Americans.18–20 Thus, Chinese participants may be more driven by the safety concern than Americans, which could result in a stronger naturalness bias. Traditional Chinese medicine, which is still widely practiced in China, encourages the acceptance and value of natural herbs as potential treatments (i.e., similar to some CAM procedures). Such practice may also foster a stronger preference for natural options among Chinese people. Although the naturalness bias in drug choices has been observed among Chinese participants,21–23 we are not aware of any existing cross-cultural studies comparing the naturalness bias between Chinese and North Americans. Furthermore, examinations of preferences for more natural versus more synthetic vaccines is scarce (although see DiBonaventura and Chapman 1 ). To the best of our knowledge, the present research will be the first to examine such preferences for vaccines across cultures.
Present Research
We conducted 3 studies to investigate the naturalness bias in the context of drugs and vaccines among Chinese and North American participants. Studies 1A and 1B involved natural versus synthetic drugs in hypothetical medical scenarios. Participants in study 1A were asked to choose 1 of the 2 drugs to treat the hypothetical medical issue. Participants in study 1B had the choice of only 1 drug, randomly assigned, and had to indicate their likelihood of taking it. Participants in study 2 rated their likelihood of taking a natural or synthetic vaccine for a hypothetical virus. We expected to observe the naturalness bias in all 3 studies, even though the research paradigm and medical context varied across studies. Importantly, the studies explored how cultures may differ in the naturalness bias when considering drugs (treatments for medical conditions) versus vaccines (prophylactic, or preventive, measures). We included safety and effectiveness ratings as potential mechanisms given that past work has routinely examined these factors in this domain. 2 Participants in North America completed the studies in English, and Chinese participants completed the studies in Chinese. The study materials, developed in English first, were translated into Chinese independently by 2 English-Chinese bilingual researchers and then checked by other bilingual researchers to ensure cross-linguistic and cross-cultural equivalence. This procedure has been used in cross-cultural research. 24
Data Statement and Sample Size Considerations
We have reported all measures, conditions, data exclusions, and the way we determined sample sizes. Study materials and data are available by contacting the first author. A priori power analysis with Gpower 3.1 25 shows that a total sample of at least 349 participants would be required to detect a small effect (w = 0.15, α = 0.05, power = 0.80) in a 2-group chi-square design (study 1A), and at least 690 participants in total would be required for a 2 × 3 between-participant design (study 1B) to detect a small effect (f = 0.15, α = 0.05, power = 0.95). For study 2 with a 2 × 2 mixed design, a minimum sample of 148 participants would be required to detect a small effect (f = 0.15, α = 0.05, power = 0.95). Based on these power analyses while considering the resources available, we attempted to collect a minimum of 200 participants in each culture for each study.
Data were not analyzed until data collection was complete for each study. The studies received approval from the Institutional Review Boards of the corresponding author for each study.
Study 1A: Choice between Synthetic and Natural Drugs
Method
Participants
A total of 249 Chinese (180 women, 55 men, 14 no response; age:
Procedure
We asked participants to imagine that they had a medical condition and needed to take a drug to treat it. 5 They had to choose 1 drug from the following 2 options:
The order of the options was counterbalanced. Due to differences across countries in income levels and health care systems, we controlled for the drug cost by having participants assume that cost was not a concern in their choice. After making their selection, they indicated how safe and effective they thought each of the drugs was on a 9-point scale (1 = not at all, 5 = moderately, 9 = very).
Participants also completed other items that were not examined for this current article: a nature relatedness scale and a description of the reasoning for their drug choice. Finally, participants completed demographic questions (age, gender, and ethnicity) and were debriefed. In all of the US samples, ethnicity was assessed by asking people to “select your race”: Asian or Pacific Islander, Black or African American, Hispanic or Latino, Native American or Alaskan Native, White or Caucasian, multiracial or biracial, or a category not listed here.
Results
The descriptive statistics and statistical analyses for the main results of each study are shown in Table 1. 85.5% (213 of 249) of Chinese participants and 90.4% of American participants (235 of 260) chose the natural drug over the synthetic drug. These ratios were significantly greater than 50%, χ2s >125, Ps < 0.001. The cultural difference was not significant, χ2(1)=2.83, P = 0.093. i
Choices and Means (Standard Deviations) across Culture and Types of Drugs or Vaccines and Statistical Tests for Studies 1–3
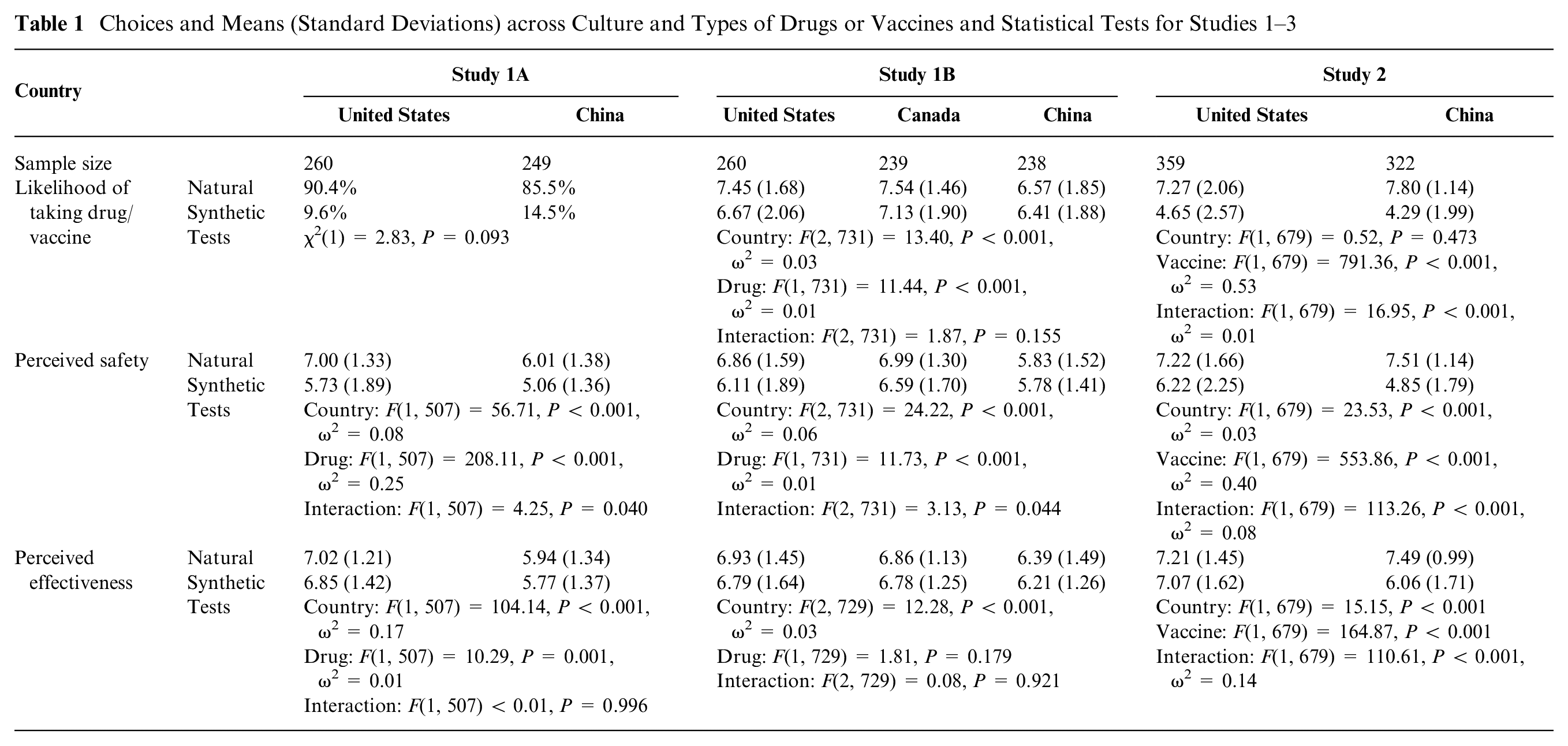
A 2 (country) × 2 (drug) mixed analysis of variance (ANOVA) test on perceived safety showed significant main effects of country and drug, as well as a significant interaction effect. ii Overall, participants perceived the natural drug to be safer than the synthetic drug, and this tendency was stronger among Americans than among Chinese.
A 2 (country) × 2 (drug) mixed ANOVA test on perceived effectiveness showed significant main effects of country and drug but no interaction effect. iii Overall, both Americans and Chinese perceived the natural drug to be more effective than the synthetic drug, and Americans perceived higher drug effectiveness than did Chinese in general.
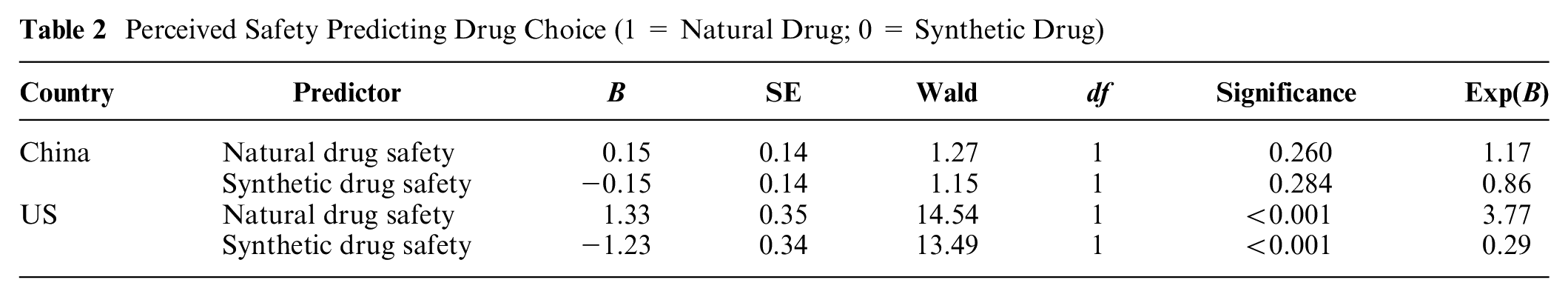
A logistic regression analysis with country and perceived safety and their interaction terms as the predictors showed that safety ratings of the natural versus synthetic drugs significantly predicted Americans’ choice of the natural (versus synthetic) drug, but drug safety ratings did not predict Chinese participants’ choice (see Table 2). These cultural differences (i.e., predictor × culture interactions) were statistically significant, Wald’s > 8.79, Ps≤0.003. A similar logistic regression analysis with perceived drug effectiveness revealed only 1 significant effect: perceived effectiveness of the natural drug predicted the drug choice, b = 0.42, Wald = 6.00, P = 0.014, Exp(B) = 1.53.
Perceived Safety Predicting Drug Choice (1 = Natural Drug; 0 = Synthetic Drug)
Overall, participants from both the US and China exhibited a natural drug bias. The size of the bias did not vary significantly across cultures. Furthermore, both American and Chinese participants perceived the natural drug as safer and more effective than the synthetic drug. Yet, the safety scores predicted drug choice for Americans only, but not Chinese. We attempted to replicate these effects in study 1B using a between-participants paradigm.
Study 1B: Ratings of Synthetic or Natural Drugs
Method
Participants
A total of 238 Chinese participants (184 women, 45 men, 9 no response; age:
Procedure
Participants were asked to imagine that they had a medical condition and could take a drug to treat it. They were then randomly assigned to 1 of the 2 drugs shown in study 1A. Participants indicated on a 9-point scale (1 = not at all likely, 5 = moderately likely, 9 = very likely) how likely they would be to take the drug, assuming cost was not a concern. They then rated the drug safety and effectiveness using the same scale as in study 1A.
Participants also completed other items that were not part of this current project: a nature relatedness scale and a description of the reasons for their choice. Finally, participants completed demographic questions in the same way as in study 1A and were debriefed.
Results iv
A 3 (country) × 2 (drug) ANOVA test on the likelihood of taking the drug revealed a significant main effects of country and drug. Specifically, Chinese participants indicated a lower likelihood to take any drug than the Canadian or American participants, Ps≤ 0.001. Overall, people preferred natural to synthetic drugs. The interaction effect of drug and country was not significant (see Table 1). Although the interaction was not significant, within-country simple effects revealed no significant preference among Chinese, F(1, 731) = 0.46, P = 0.496; a marginally significant preference for a natural drug among Canadians, F(1, 731) = 3.10, P = 0.079; and a significant preference for a natural drug among Americans, F(1, 731) = 12.13, P = 0.001.
Perceived safety followed a similar pattern. A 3 (country) × 2 (drug) ANOVA revealed significant main effects of country and drug. Chinese participants perceived lower safety in both drugs than the American and Canadian participants, Ps < 0.001. Overall, all participants perceived higher safety in natural than synthetic drugs. The interaction effect of drug and country was significant: Americans (P < 0.001) and Canadians (P = 0.050) perceived higher safety in natural than in synthetic drugs, but Chinese participants did not show any difference (P = 0.833).
For perceived effectiveness, the only significant effect in a 3 (country) × 2 (drug) ANOVA was the main effect of country. Again, the Chinese reported lower perceived effectiveness than the Canadians or Americans, Ps < 0.001. No other effect approached statistical significance, Ps > 0.170.
In addition, as seen in Figures 1 and 2, Hayes’s process macro (model 4) with 10,000 bootstrapping samples showed that safety mediated the effect of drug on likelihood of taking it among Americans, b = 0.39, 95% confidence interval [CI; 0.15, 0.69] and Canadians, b = 0.32, 95% CI [0.01, 0.65] but not among Chinese, b = 0.03, 95% CI [−0.22, 0.28]. 26 Effectiveness did not have any significant indirect effects in any of the groups: among Americans, b = 0.05, 95% CI [−0.08, 0.19]; among Canadians, b = 0.01, 95% CI [−0.04, 0.08]; and among Chinese, b = 0.06, 95% CI [−0.05, 0.20].
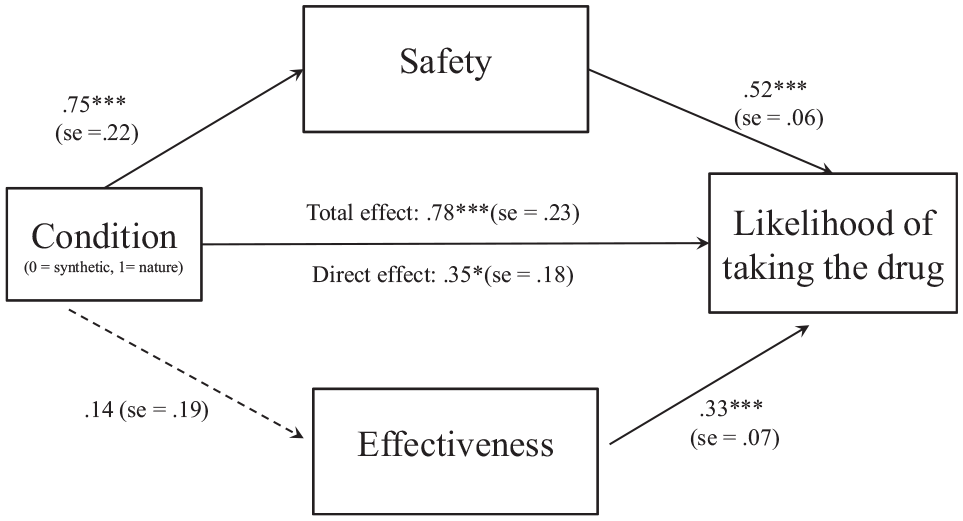
Perceived drug safety mediated the naturalness bias among American participants. ***P < 0.001; **P < 0.01; *P < 0.05 (study 1B).
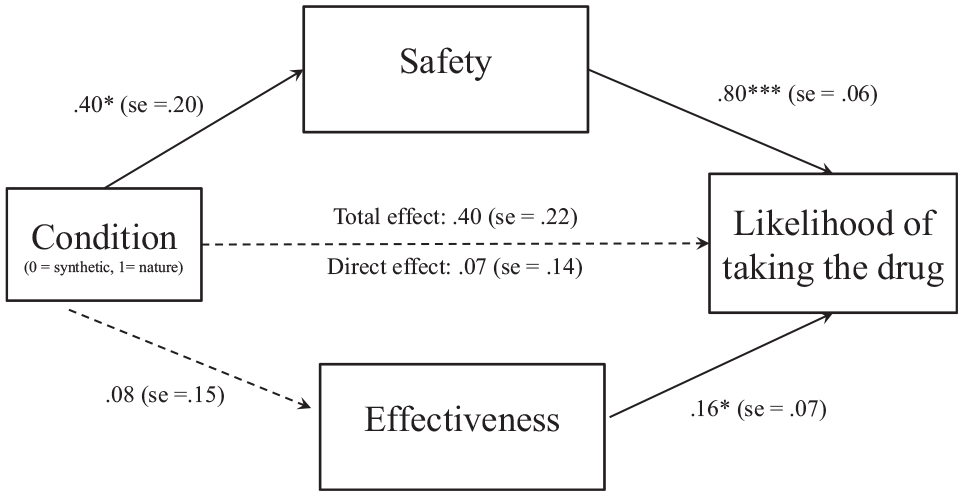
Perceived drug safety mediated the naturalness bias among Canadian participants. ***P < 0.001; **P < 0.01; *P < 0.05 (study 1B).
As in study 1A, participants from the United States, Canada, and China exhibited a natural drug bias, although the bias was not statistically significant among Chinese participants. The size of the bias did not vary across cultures in a statistically significant manner. However, as shown in Table 1, the bias among Chinese participants appears much smaller. Again, as in study 1A, participants perceived the natural drug as safer and more effective than the synthetic drug. Furthermore, drug safety ratings mediated the link between drug condition and the likelihood of taking the drug in the Canadian and American samples but not in the Chinese sample. These results again suggest that safety perceptions seem to partially drive the natural drug bias among Canadians and Americans but not among Chinese participants (similar to study 1A).
Study 2: Ratings of Natural versus Synthetic Vaccines, within Participant Design
In study 2, we changed the medical context from drugs to vaccines. Unlike drugs, which in our studies were presented as being used to treat an existing medical condition, vaccines are prophylactic measures used to prevent the development of a medical condition. We sought to determine if this distinction would affect the cross-cultural comparison of the naturalness bias. Furthermore, vaccine hesitancy has been a great concern in the fight against the COVID-19 pandemic, and investigating people’s vaccine decisions may shed light on the problem. We also attempted to recruit participants who were similar in age, education level, and gender to facilitate cross-cultural comparisons.
Method
Participants
We recruited 359 American adults (175 women, 177 men, 6 other, 1 missing; age:
Procedure
Participants were given the following information about a hypothetical virus and a vaccine for it: Imagine that there is a virus that causes a very painful rash. It most often appears as a single stripe of blisters that wraps around either the left or the right side of your torso, but it can also appear on your face or around your eyes. It’s not life-threatening, but it can be very painful. Vaccines can help reduce the risk. Doctors have recommended that you should take a vaccine for it. There are two options:
Option 1 is a vaccine made from mostly synthetic ingredients NOT FOUND in nature.
Option 2 is a vaccine made from mostly natural ingredients FOUND in nature.
Both vaccines were presented with the following information: Studies have been conducted on this vaccine for 20 years. It has been shown to be effective in 85% of users. The vaccine has also been shown to cause mild side effects on rare occasions and serious side effects in 0.5% of users.
We used the terms “mostly natural” and “mostly synthetic” because vaccines are not entirely synthetic or natural in practice. Participants indicated how likely (1 = not at all likely; 5 = moderately likely; 9 = very likely) they would be to choose each vaccine, assuming cost was not a concern. They then reported how safe and effective they thought each vaccine was using the same scale as in study 1A. The order of vaccines was counterbalanced across participants.
We also asked participants 5 additional questions designed for this study to address factors of general concerns when considering vaccines. Participants were asked to indicate to what extent (1 = not at all, 5 = moderately, 9 = very much) each of the following factors influenced their decision of whether or not to take a vaccine in general: 1) the effectiveness of the vaccine, 2) the severity of the possible side effects, 3) the likelihood of serious side effects, 4) the quality of the research on the vaccine, and 5) the ingredients in the vaccine.
The US participants also completed other unrelated items that were not part of this current project. Both US and Chinese participants completed demographic questions (including age, gender, education level, and ethnicity). Education level was assessed by asking participants to “select your level of education” from the following options: high school or less, 2-y college (including current college student), 4-y university (including current university student), master’s degree (including current master’s student), or doctoral degree (including current doctoral student).
Results
Likelihood of taking the vaccines
A 2 (country) × 2 (vaccine) mixed ANOVA test with vaccine as a within-participant variable was conducted on the likelihood of taking the vaccine. The country main effect was not significant, while the vaccine main effect was significant, in that participants overall were more likely to choose the natural vaccine than the synthetic vaccine. The interaction between country and vaccine was significant. As seen in Table 1, the naturalness bias (i.e., difference in the likelihood of taking natural v. synthetic vaccines) was stronger among Chinese individuals than among Americans. v
Perceived safety of vaccines
Perceived vaccine safety showed a similar pattern as the likelihood of taking the vaccines. The vaccine main effect was significant; participants in general perceived the natural vaccine to be safer than the synthetic vaccine. The country main effect was significant. As seen in Table 1, the interaction between vaccine and country was significant; the naturalness effect was stronger among Chinese than among Americans. vi
Perceived vaccine effectiveness
The main effects of vaccine and country, as well as their interaction effect, were significant. As seen in Table 1, Chinese participants perceived the natural vaccine to be more effective than the synthetic vaccine, F(1, 679) = 258.73, P < 0.001, while Americans perceived no difference in effectiveness between the 2 vaccines, F(1, 679) = 2.85, P = 0.092. vii
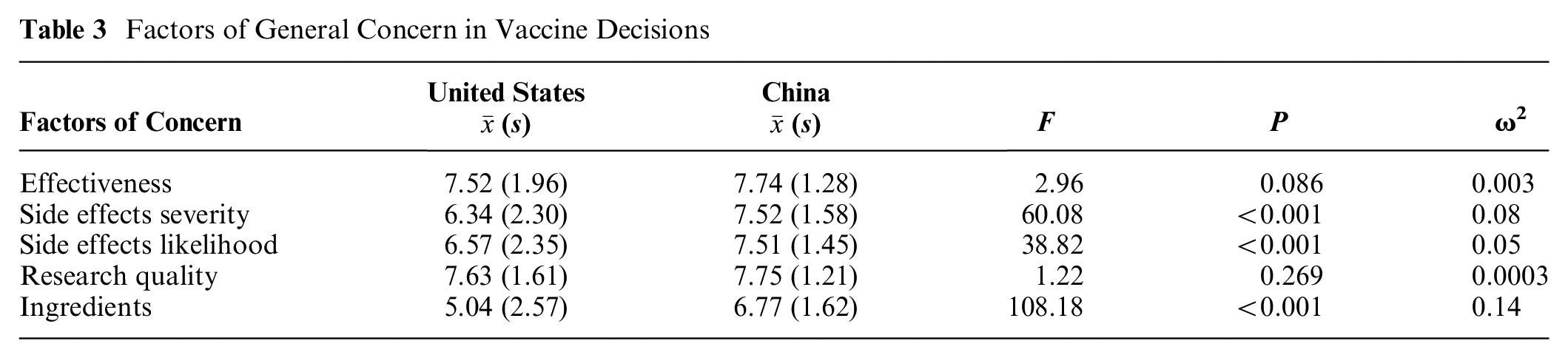
Factors of concern when making vaccination decisions in general
We next examined participants’ ratings of general concerns when deciding to take a vaccination in their everyday life. As seen in Table 3, concerns in terms of severity of side effects, likelihood of side effects, and vaccine ingredients were significantly higher among Chinese than among Americans. There was no significant cultural difference in concerns about the effectiveness of the vaccines or the quality of vaccine research.
Factors of General Concern in Vaccine Decisions
Indirect effects of culture on the naturalness bias
Perceived safety and effectiveness
We ran model 4 in Hayes’s process macro with 10,000 bootstrapping samples to explore the mediating factors for cultural differences in the naturalness bias. A naturalness bias was computed by subtracting the likelihood of taking the synthetic vaccine from that of taking the natural vaccine. As seen in Figure 3, differences in perceived safety associated with the 2 vaccines mediated the effect of culture on the naturalness bias, b = 1.34, 95% CI [1.06, 1.63], but not differences in perceived effectiveness, b = 0.13, 95% CI [−0.02, 0.30]. 25
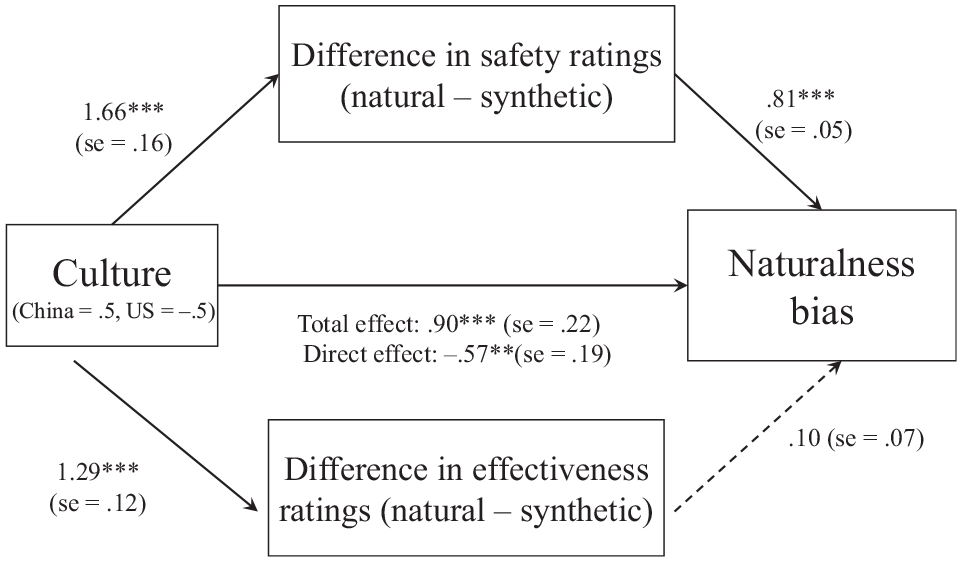
Indirect effect of culture on naturalness bias via perceived safety (unstandardized regression coefficient). ***P < 0.001; **P < 0.01; *P < 0.05 (study 2).
Factors of concern in vaccine decisions
We also examined mediation with the factors of general concerns in vaccine decisions. We first conducted a factor analysis to determine if the items form a factor or factors. We found that 3 items (concern about the severity of side effects, concern about the likelihood of side effects, and concern about the ingredients) loaded onto what we labeled a “safety” factor (all loadings >0.459). The other 2 items (concern about the effectiveness of the vaccine and concern about the quality of research) loaded onto another factor that we labeled an “effectiveness” factor (all loadings >0.434). The “safety” factor mediated cultural differences in the naturalness bias (Figure 4), b = 0.73, 95% CI [0.51, 0.99], but concerns about “effectiveness” issues did not, b = −0.04, 95% CI [−0.11, 0.005]. Note that some of the factor loadings were lower than optimal, and therefore, results should be interpreted with caution.
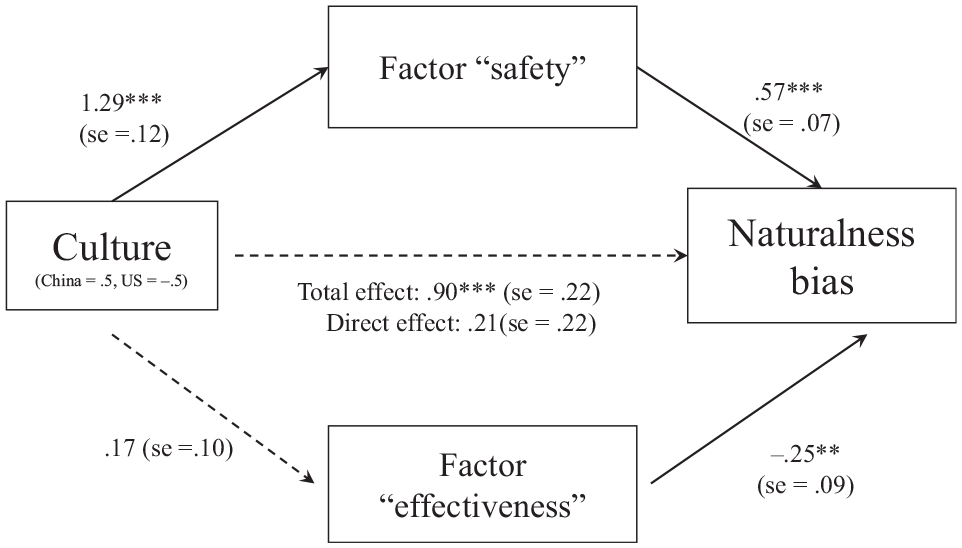
Indirect effect of culture on naturalness bias via perceived “safety” concerns (unstandardized regression coefficients). ***P < 0.001; **P < 0.01; *P < 0.05 (study 2).
Study 2 was perhaps the strongest test of the naturalness bias across cultures. Both samples were from online sources, and age, gender, and education level were more similar than in studies 1A and 1B. We found a stronger naturalness bias for the Chinese than American participants when considering more natural versus more synthetic vaccines. Furthermore, perceived safety of the vaccines in the scenarios and more general safety concerns about vaccines in general mediated this effect.
General Discussion
We conducted 3 studies to examine the naturalness bias in drug and vaccine decision making across cultures. Overall, Americans, Canadians, and Chinese participants consistently demonstrated the naturalness bias across various medical contexts and research designs. When using a vaccination context, Chinese participants showed a stronger naturalness bias than Americans did, and safety concerns mediated this effect. Although perceived safety predicted the naturalness bias among Americans and Canadians in all studies, it predicted the bias among Chinese participants only in study 2. Below, we discuss the implications of these effects.
Cultural Differences
The Chinese samples had results similar to the American and Canadian samples in studies 1A and 1B but not in study 2. The major difference between studies 1A/1B and study 2 was the medical context: drugs versus vaccines. It appears that Chinese participants are more concerned about synthetic substances when the context is vaccines versus drugs as compared with North American participants. Indeed, Chinese participants were significantly more concerned about ingredients, likelihood of side effects, and the severity of side effects of vaccines in general compared with US participants. Furthermore, a factor composed of these safety concerns about vaccines in general mediated the link between country and vaccine choice. In other words, Chinese participants had higher safety-related concerns for more synthetic versus more natural vaccines, which partially drove their heightened choice for the more natural vaccine. These findings suggest that at least in the context of vaccines, Chinese participants seem to be more concerned about safety-related issues with the synthetic aspects of vaccines. These findings appear to coincide with some research on the perceptions and acceptance of the COVID-19 vaccines, which revealed that Chinese participants were more concerned about adverse effects than American participants were. 27
We believe study 2 is the most informative in terms of examining cultural differences given that the two samples were more similar in demographics than the samples in studies 1A/1B. Chinese participants in studies 1A/1B were university students, while American participants were adults recruited via Prolific.co. Thus, they were not entirely comparable in age, gender, and education levels, although controlling for age and gender statistically did not alter the main patterns of results. To address the issue of comparability, we recruited university students from Canada in study 1B, who ultimately did demonstrate a naturalness bias similar to that observed in the American adult sample from Prolific.co. This finding underscores the robustness of the naturalness bias in the drug context. To more explicitly address these issues in study 2, we recruited Chinese adult participants through an online platform, making them more comparable with the American participants in terms of age, gender, and education level.
Although we believe the context change from studies 1A/1B to study 2 impacted the cultural effect, it is possible that methodological changes affected the results as well. Study 2 used a within-participants rating paradigm, whereas studies 1A/1B used a forced-choice paradigm or a between-participants rating paradigm. We used slightly different paradigms in each study to examine the robustness of the effects, but such changes may have affected the results as well. Past research has shown that Chinese participants are more sensitive to the context, 28 and this includes the question context. 29 Thus, it is possible that Chinese participants were sensitive to the presence or absence of another alternative drug/vaccine (within-participant v. between-participant design), as well as whether they had to make a binary choice or they could indicate their preferences for both alternatives. Additional research that manipulates the medical context or the methodological factors will be necessary to fully examine these ideas.
Clinical and Medical Decision-Making Implications
The current findings can be useful when applied to clinical or health decision-making situations. Overall, participants from multiple cultures exhibited a naturalness bias when thinking about taking drugs or vaccines for hypothetical medical issues. In situations in which a natural drug or a more natural vaccine is available as a medical option, people in different cultures may prefer that option and be more likely to follow a treatment plan to its completion. Such effects could be beneficial or detrimental. If a drug or vaccine obtained from more natural sources is equally, or more, safe and effective than a synthetic one, the bias might be beneficial because people might be more likely to take it. Furthermore, safety concerns appear to partially drive people’s preferences for natural drugs and vaccines. Therefore, highlighting the natural components of a drug or vaccine, even if the drug or vaccine is primarily of synthetic origins, might help reduce these concerns and enhance usage.
If, however, a synthetic drug or vaccine has a more favorable safety and/or efficacy profile, the bias found in the current studies might be detrimental because it could cause people to bypass synthetic drugs or vaccines because of the naturalness bias. Such behavior could have considerable negative health consequences, especially when considering the life-saving impact of some drugs (e.g., cholesterol medications) and vaccinations (e.g., in the context of COVID-19). Therefore, it might be helpful in such cases to educate people about the naturalness bias given its somewhat inherent aspects. Indeed, some research has shown that educating people about the naturalness bias in the drug domain and pointing out that both natural and synthetic drugs can be positive or negative, depending upon the context, reduced people’s naturalness bias. 6
Limitations
The current studies are not without limitations. First, as with similar research of the present type, the participants in all studies do not represent random samples across North America or China. The samples were convenience samples; therefore, responses may not represent people in general. Second, the responses made were hypothetical in nature and may not represent actual behavior. The scenarios used examined the cognitive decision making of participants, but the decisions were based on self-report. Past work has demonstrated that the naturalness bias influences drug and vaccine behavioral choices,1,6,23 which suggests that the effects in the current work are predictive of actual behavior.
Conclusion
The results of the 3 studies are informative in revealing a natural drug and vaccine bias across cultures. Chinese participants exhibited a similar natural versus synthetic drug bias compared with North American participants for the most part, but they exhibited a stronger bias for more natural versus more synthetic vaccines compared with North American participants. Safety concerns appeared to be a significant mediator. Overall, the results suggest that the naturalness bias in medical decision making occurs across cultures, but Chinese participants may be more sensitive to naturalness in a vaccine context.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this article was provided in part by funding from Social Science and Humanities Research Council of Canada (435-2018-0061) and from Gettysburg College. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
