Abstract
Background
Patient-centered care includes matching treatments to patient values and preferences. This assumes clarity and consistency of values and preferences relevant to major medical decisions. We sought to describe stability of patient-reported values regarding aggressiveness of care and preferences for left ventricular assist devices (LVADs) for advanced heart failure.
Methods and Results
We conducted a secondary analysis of patients undergoing LVAD evaluation at 6 US centers. Surveys at baseline, 1 month, and 6 months included a single 10-point scale on the value of aggressive care (score 1 = “do everything,” 10 = “live with whatever time I have left”) and treatment preference (LVAD, unsure, no LVAD). Data were captured for 232 patients, of whom 196 were ultimately deemed medically eligible for LVAD, and 161 were surgically implanted by 1 month. Values at baseline favored aggressive care (mean [SD], 2.49 [2.63]), trending toward less aggressive over time (1 month, 2.63 [2.05]; 6 months, 3.22 [2.70]). Between baseline and 1 month, values scores changed by ≥2 points in 28% (50/176), as did treatment preferences for 18% (29/161) of patients. Values score changes over time were associated with lower illness acceptance, depression, and eventual LVAD ineligibility. Treatment preference change was associated with values score change.
Conclusion
Most patients considering LVAD were stable in their values and treatment preferences. This stability, as well as the association between unstable treatment preferences and changes to stated values, highlighted the clinical utility of the values scale of aggressiveness. However, a substantial minority reported significant changes over time that may complicate the process of shared decision making. Improved methods to elicit and clarify values, including support to those with depression and low illness acceptance, is critical for patient-centered care.
Self-reported values and preferences change significantly over time in about a quarter of patients actively considering left ventricular assist device implantation. Instability in stated values and preferences challenges clinicians who want to maximally match patient preferences to the treatments they receive. For most patients, clinicians can normalize the desire to maximize survival and empathize with the difficulty of making the decision. For others, clinicians may want to help patients explore the benefits and tradeoffs of therapy, including whether values other than the ones being asked about dominate their consideration.Highlights
Keywords
Introduction
Decision making for invasive treatments with complex tradeoffs relies on patients being able to prioritize competing values. In these circumstances, a patient’s treatment decision is viewed as “good” when 2 criteria are met: first, that patients are adequately informed of the risks and benefits of each medically reasonable option available (including to forego curative treatments) and, second, that the decision made is most consistent with their values (i.e., values-choice concordance). 1 Patient decision aids are used to facilitate shared decision making and are distinguished from general educational materials in part by their use of values clarification methods, which seek to highlight patient values and treatment preferences and integrate them into the conversations occurring between patients and their clinicans.2,3
Defining values (characteristics of treatment options found to be individually desirable or undesirable) and preferences for care (specific treatment(s) a patient wishes to pursue in light of these values) 2 can present substantial challenges for patients. To define and elucidate these preferences, patients use a combination of intuition and deliberate problem solving while deciding on courses of treatment. Emphasis of one mode of thought over another may lead to different expressions of values (e.g., short-term satisfaction or avoidance of undesirable outcomes v. long-term planning). 4 Meanwhile, patients’ medical and nonmedical situations can change dramatically and rapidly, challenging these values and associated treatment preferences. 5 For example, advanced care directives, 6 resuscitation preferences, 7 and treatment choice among patients with metastatic cancer 8 have been shown to change over time among different groups of patients, even when the predominant preference for treatment across the population is to pursue aggressive care.
One such complex decision with major tradeoffs is left ventricular assist device (LVAD) for patients with advanced heart failure. Currently available LVADs are electrical rotary pumps surgically implanted in the chest and attached to the heart to pump blood out of a weakened left ventricle to the aorta. Of the nearly 6 million US heart failure patients, 5% to 10% will eventually develop end-stage disease that no longer responds to medication, requiring consideration of advanced heart replacement therapies (transplant or LVAD) v. symptom-focused care. 9 Many of these patients will not be eligible for transplant owing to advanced age or comorbidity, referred to as receiving “destination therapy” (DT). LVADs can dramatically increase survival, from approximately 50% mortality at 1 year to an average survival of 5 years postimplant, and also improve quality of life through reduced symptoms of heart failure. 10 However, they also come with substantial risks of complications, such as stroke and major bleeding, and lifestyle burdens, including invasive surgery and recovery, the need for a dedicated caregiver, frequent and expensive follow-up care, and constant access to reliable electricity.11–13 Patients with DT LVADs cannot have devices removed once implanted, and deactivating the device typically results in immediate death.
Previous studies have highlighted medical aggressiveness as the dominant value relevant to decision making around LVAD: survival as the primary value for LVAD acceptors v. consideration of quality of time for those who declined LVAD. 13 To align patients’ personal values with the choice of whether or not to accept LVAD therapy, patients must situate the decision along a continuum of care preferences. On one end of that continuum is aggressive care to maximize chance of survival with an LVAD, accompanied by risk of major complications and burdens. On the other end is electing to forgo a potentially life-prolonging device to focus on comfort care without burdens associated with an LVAD. This scale of aggressiveness, when compared with the patient’s treatment preferences, can help show a patient’s values-choice concordance.
The DECIDE-LVAD trial was a multisite randomized study of patients enrolled at the time an LVAD evaluation was initiated that serially collected patient-reported 1) values assessed on an aggressiveness of care scale and 2) preferences for treatment. The main findings from DECIDE-LVAD have been reported elsewhere,14,15 showing that use of formal patient decision aids improved patient understanding of the decision, improved concordance between patients’ self-reported values and treatment choice, and reduced overall LVAD placement. The data from this study provide a rare opportunity to characterize the stability of patients’ reported values and preferences over time when considering a life-altering medical decision, which has implications for values-choice concordance measures in the assessment of patient-centered decision making. We hypothesized that a significant number of patients would vary in their reported value of aggressive care and stated preferences for LVAD and that such instability could be partially anticipated.
Methods
Patients
The DECIDE-LVAD trial, which has been previously described, 14 enrolled patients from 6 participating sites across the United States from June 2015 through January 2017. Patient eligibility criteria included ≥18 years of age, end-stage heart failure, and active consideration for destination therapy LVAD (DT LVAD) placement (meaning that the patient would expect to continue on LVAD therapy until death). Written informed consent was obtained from all patient participants.
Data Collection
Values and treatment preference were assessed through participant surveys at baseline (at enrollment, prior to formal LVAD education), 1 month after enrollment, and 6 months after enrollment. Demographics and other secondary measures were collected as well, including validated measures of PEACE illness acceptance, 16 perceived stress, 17 depression (Patient Health Questionnaire–2), 18 and quality of life (EuroQol visual analog scale). 19 Final treatment received was recorded from the patient’s medical record in the 6 months after enrollment.
Values and Preferences
Patient values were assessed with a single-item, the 10-tier Likert scale of aggressiveness of care. This scale was modeled after a well-accepted values evaluation tool 14 and using findings from earlier needs assessment work.20–22 Anchor scores on this item were “Do everything I can to live longer, even if that means having major surgery and being dependent on a machine” (score = 1) v. “Live with whatever time I have left, without going through major surgery or being dependent on a machine” (score = 10; Figure 1). In this scale, participants were presented with only a line with end anchor points and asked to place an “x” where they felt they landed. This item was then scored by superimposing a 10-point scale along that line and finding the number that was closest to the “x” marking. Treatment preference assessment was modeled from the validated Decision Conflict Scale using a single item asking participants, “Which treatment option do you prefer?” with categorical response options including “Getting a DT LVAD,”“Not getting a DT LVAD,” and “Unsure.”
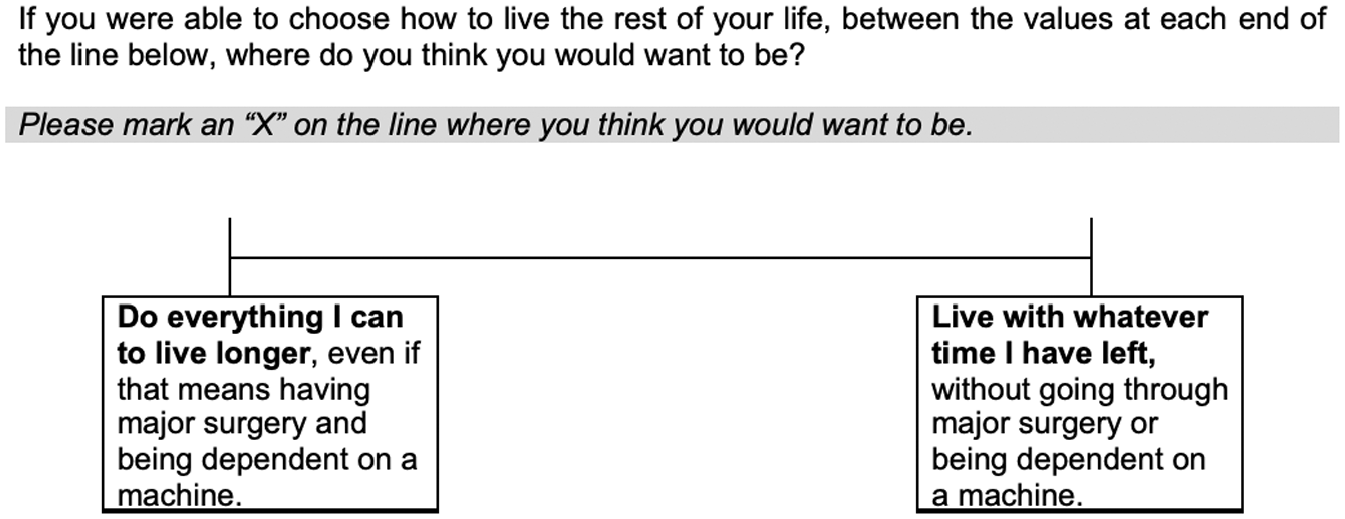
Patient-expressed values visual analog scale. Participants were asked to place an “x” on the line, and responses were scored according to a 10-point scale superimposed onto the line.
Changes in Values or Treatment Preference
To investigate the degree that stated values remained constant between observations, we used several different markers for change. Our primary definition for a change in values was a difference of ≥2 points between baseline and 1 month. Changes between 1 to 6 months and baseline to 6 months were secondarily examined. Secondary definitions of change included a difference of ≥5 points (50% of the scale) or shifting from one side of the scale to the other (providing a predominantly aggressive score of 1–5 followed by a predominantly nonaggressive score of 6–10 or vice versa). Any change in expressed preference for treatment (i.e., LVAD, no LVAD, or unsure) was characterized as a change.
Statistical Analysis
We described the distribution of stable and changing values and treatment preferences between each pair of time points. We fit generalized linear mixed models to assess the associations of participant characteristics with the values score and treatment preference change binary outcomes. These models used a logit link and included a random effect for site and fixed effects for intervention period and time period in the stepped-wedge design in the original trial. Individual participant characteristics of interest were modeled using fixed effects. We computed adjusted mean values scores at each time point stratified by receipt of LVAD using linear mixed models, also adjusting for site, intervention, and time period in the stepped-wedge design. We accounted for within-subject correlation with an unstructured covariance matrix. Finally, we assessed associations of participant characteristics with missing outcome data at 1-month follow-up using χ2 tests and t tests.
Within our data set, having had an LVAD implanted at 1 month and determination that the patient was not eligible for LVAD therapy were associated with missingness for both patient-reported values and treatment preference at 1-month follow-up. We employed multiple strategies to evaluate continuity between models including all participants and only those eligible for LVAD therapy. First, our primary models estimating values over time adjusted for LVAD implantation status. In models of values and treatment preference instability, we report results from models both adjusted and unadjusted for LVAD eligibility. Second, in a post hoc sensitivity analysis, we refit our generalized linear mixed models only on the subset of participants who were deemed medically eligible for LVAD to analyze whether or not eligibility for LVAD independently influenced changes to values or treatment preferences (i.e., to test the potential effect of limited clinical options on preferences for care or expressed values).
Results
In the DECIDE-LVAD trial, 385 patients were screened across 6 participating sites during the study period (June 2015–January 2017), 248 of whom enrolled. Enrolled patients were more likely to be white non-Hispanic when compared to those who were screened but not enrolled (75.8% v. 63.9%, P = 0.03) but did not differ along any other demographic or clinical status category assessed. Of these 248 participants, 232 completed the baseline values assessment, and 226 did so for treatment preference. Participants who did not complete the baseline surveys were more likely to have completed their first assessment in an intensive care unit (ICU) setting than in an outpatient or non-ICU inpatient setting (P = 0.07) but did not differ according to any individual demographic measure. Baseline characteristics as well as 1- and 6-month follow-up data are displayed in Table 1.
Baseline Characteristics and 1- and 6-Month Follow-up of Study Sample a
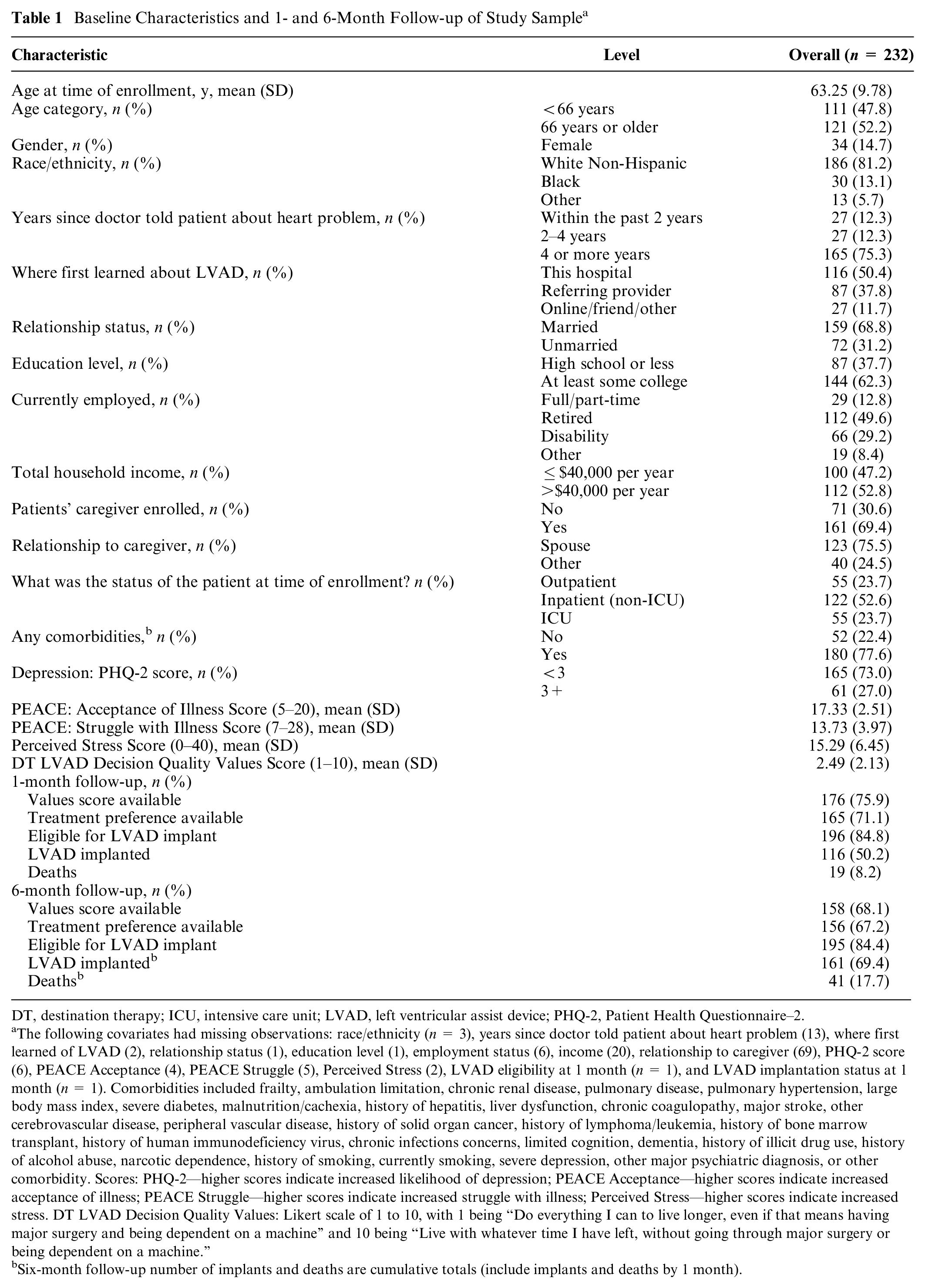
DT, destination therapy; ICU, intensive care unit; LVAD, left ventricular assist device; PHQ-2, Patient Health Questionnaire–2.
The following covariates had missing observations: race/ethnicity (n = 3), years since doctor told patient about heart problem (13), where first learned of LVAD (2), relationship status (1), education level (1), employment status (6), income (20), relationship to caregiver (69), PHQ-2 score (6), PEACE Acceptance (4), PEACE Struggle (5), Perceived Stress (2), LVAD eligibility at 1 month (n = 1), and LVAD implantation status at 1 month (n = 1). Comorbidities included frailty, ambulation limitation, chronic renal disease, pulmonary disease, pulmonary hypertension, large body mass index, severe diabetes, malnutrition/cachexia, history of hepatitis, liver dysfunction, chronic coagulopathy, major stroke, other cerebrovascular disease, peripheral vascular disease, history of solid organ cancer, history of lymphoma/leukemia, history of bone marrow transplant, history of human immunodeficiency virus, chronic infections concerns, limited cognition, dementia, history of illicit drug use, history of alcohol abuse, narcotic dependence, history of smoking, currently smoking, severe depression, other major psychiatric diagnosis, or other comorbidity. Scores: PHQ-2—higher scores indicate increased likelihood of depression; PEACE Acceptance—higher scores indicate increased acceptance of illness; PEACE Struggle—higher scores indicate increased struggle with illness; Perceived Stress—higher scores indicate increased stress. DT LVAD Decision Quality Values: Likert scale of 1 to 10, with 1 being “Do everything I can to live longer, even if that means having major surgery and being dependent on a machine” and 10 being “Live with whatever time I have left, without going through major surgery or being dependent on a machine.”
Six-month follow-up number of implants and deaths are cumulative totals (include implants and deaths by 1 month).
Value of Aggressive Care and Values Stability
Average scores on the single-item values measure favored aggressive care but shifted toward valuing less aggressive care over time: baseline mean (SD), 2.49 (2.13), 1-month mean (SD), 2.64 (2.06); and 6-month mean (SD), 3.22 (2.71) (Figure 2A). This change was driven in large part by a subset of participants whose values changed substantially across observations. Between baseline and 1 month, values scores changed by ≥2 points in 28% (50/176). Between baseline and 6 months, 8.3% of scores changed by ≥5 points, and 21.5% shifted from one side of the value spectrum to the other (score 1–5 to 6–10 or vice versa). However, most respondents had values scores that remained stable (no more than 1-point change) over time: 71.6% from baseline to 1 month and 67.1% from baseline to 6 months (see Table 2).
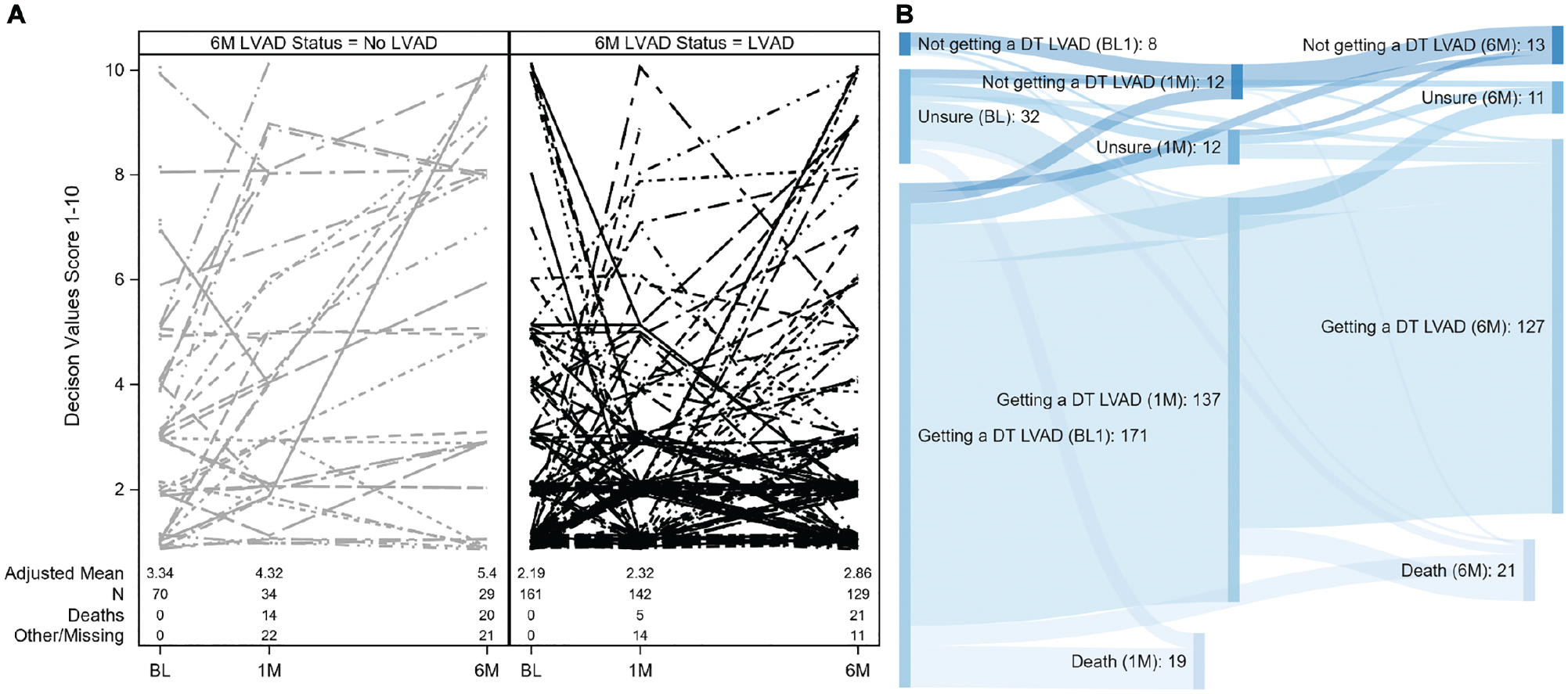
(A) Values scale responses for 232 patients over time (1 = “Do everything I can to live longer” to 10 = “Live with what time I have left without going through major surgery or being dependent on a machine”), stratified by therapy received 6 months after LVAD evaluation initiated. Dots are mean values for each time period. (B) Sankey diagram depicting proportional flow of participant treatment preference responses across observations. DT, destination therapy; LVAD, left ventricular assist device.
Patient Values Score at Baseline, 1 Month, and 6 Months from Initiation of DT LVAD Evaluation and Changes in Values Score between Pairs of Time Points
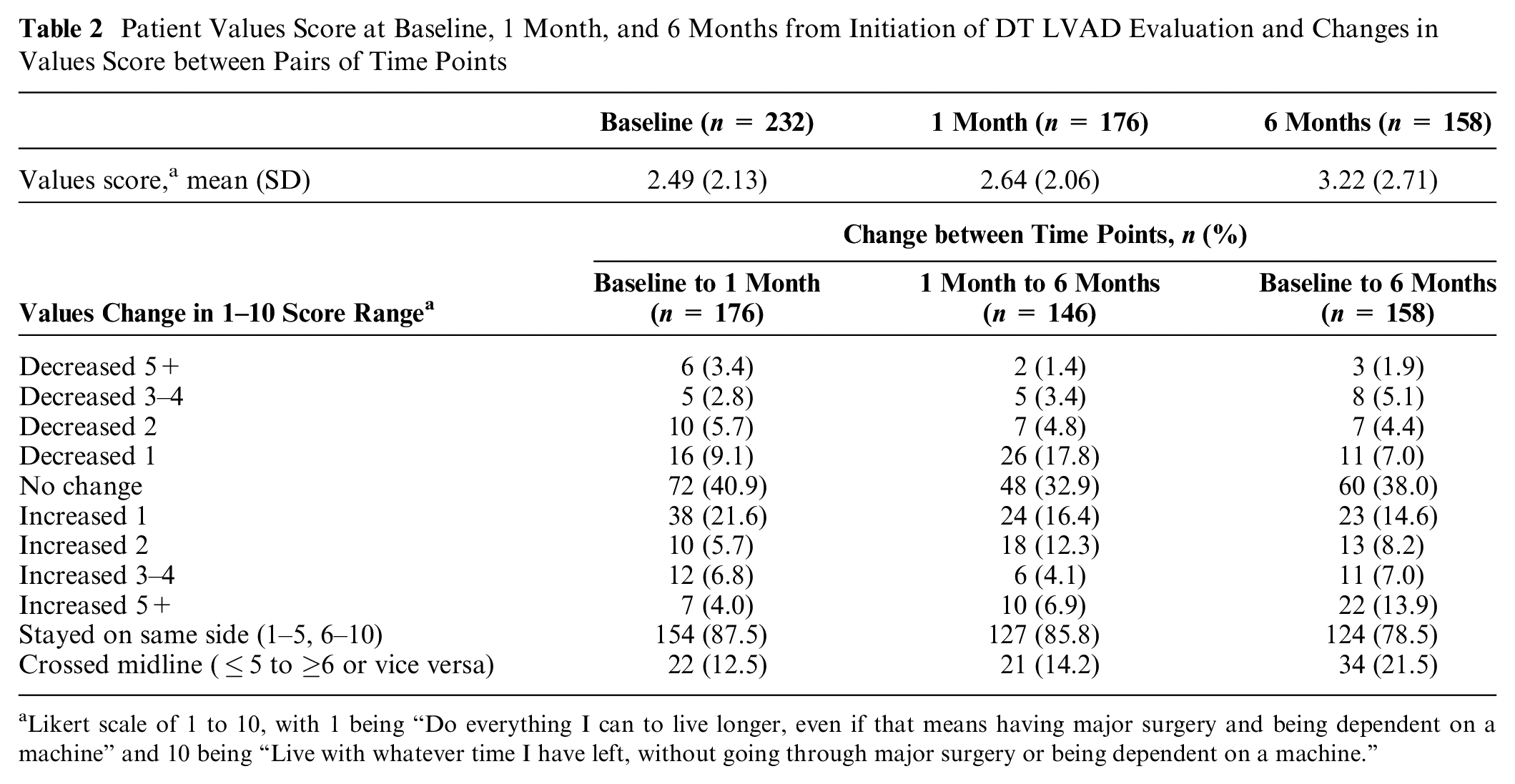
Likert scale of 1 to 10, with 1 being “Do everything I can to live longer, even if that means having major surgery and being dependent on a machine” and 10 being “Live with whatever time I have left, without going through major surgery or being dependent on a machine.”
Stated Treatment Preference and Preference Stability
At baseline, 226 participants responded to the treatment preference item on the survey. LVAD treatment was preferred by 79.2% (n = 179), while 4.0% (n = 9) preferred no LVAD, and 16.8% (n = 38) reported that they were unsure. Most patients (82.0%; n = 132) provided the same response at 1 month, and of these, 93.2% (n = 123) preferred the LVAD treatment (Table 3). In the cases where treatment preferences changed, the most common change was for patients who initially responded “unsure” but later reported a preference for LVAD placement, shown in Figure 2B.
Frequency with Which Treatment Preference Scores Change or Are Stable
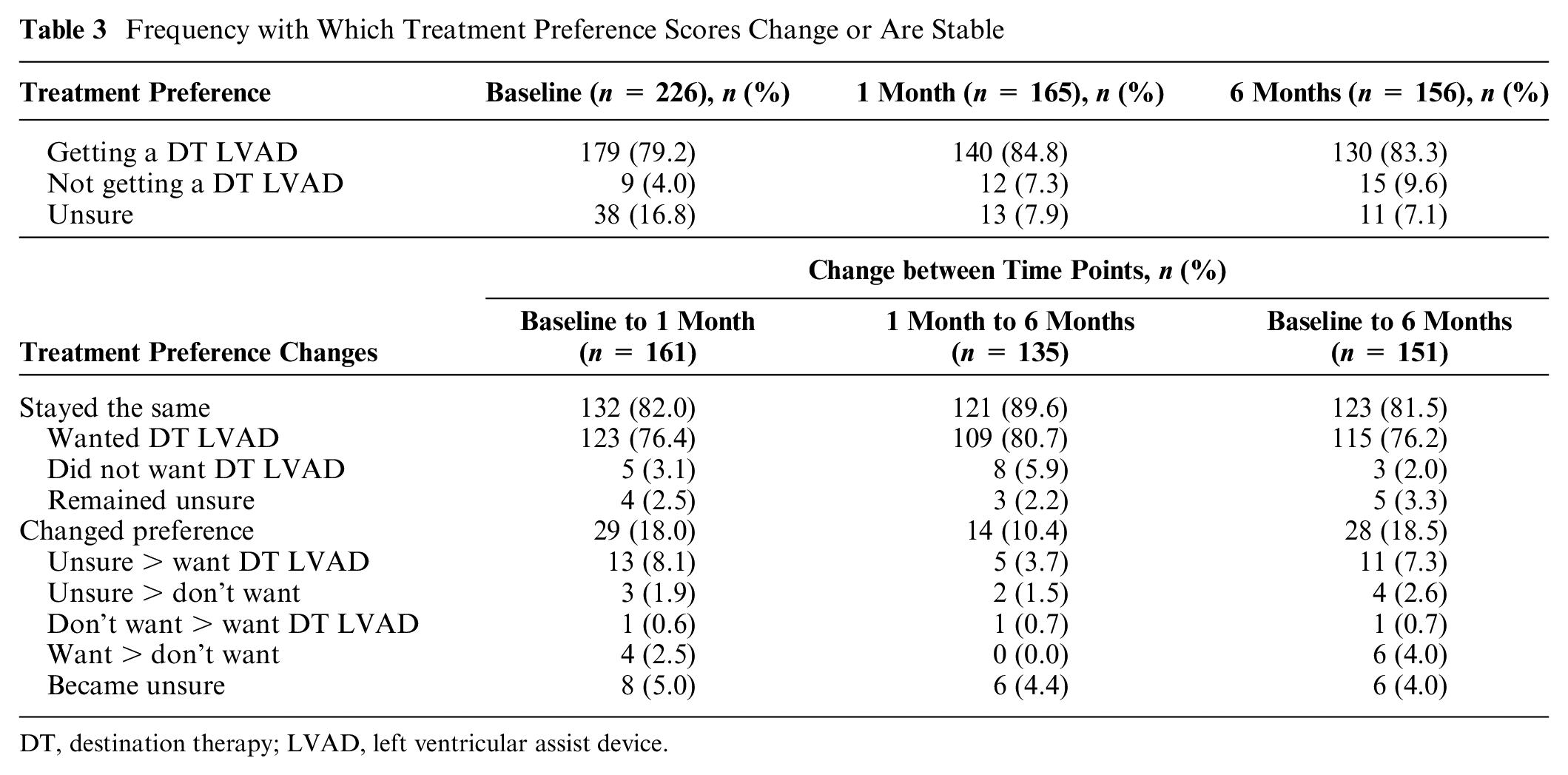
DT, destination therapy; LVAD, left ventricular assist device.
Relationship of Values Instability to Treatment Preference Instability
Participants whose values scores changed by ≥2 points from baseline to 1 month were more likely to have a treatment preference change than those whose scores remained stable (29.5% [13/44] v. 13.9% [16/115], P = 0.02, χ2 test).
Demographic and Experiential Predictors of Instability
In multivariable analysis, participants who were deemed ineligible for LVAD by the medical team were more likely to report changes to their stated values than participants who were eligible for LVAD (odds ratio [OR], 3.47; 95% confidence interval [CI], 1.07–11.30; P = 0.039; Table 4). Participants with higher illness acceptance scores, signifying more acceptance, were less likely to have values score changes than participants with lower illness acceptance scores (OR, 0.82; 95% CI, 0.71–0.95; P = 0.009). Participants with PHQ-2 scores 3 and higher, signifying greater depression, were more likely to have a change in their values score than those with lower scores (OR, 2.20; 95% CI, 1.02–4.72; P = 0.044). Both LVAD eligibility and illness acceptance score remained statistically significantly associated with changing values score when included in the same model. Of note, while the original trial included an experimental arm in which patients were exposed to a decision aid, including a values clarification exercise, these patients were not more likely to exhibit changes in either their stated values or treatment preferences across observations.
Experiential and Treatment Variables’ Association with Changes in Stated Values and Treatment Preference a
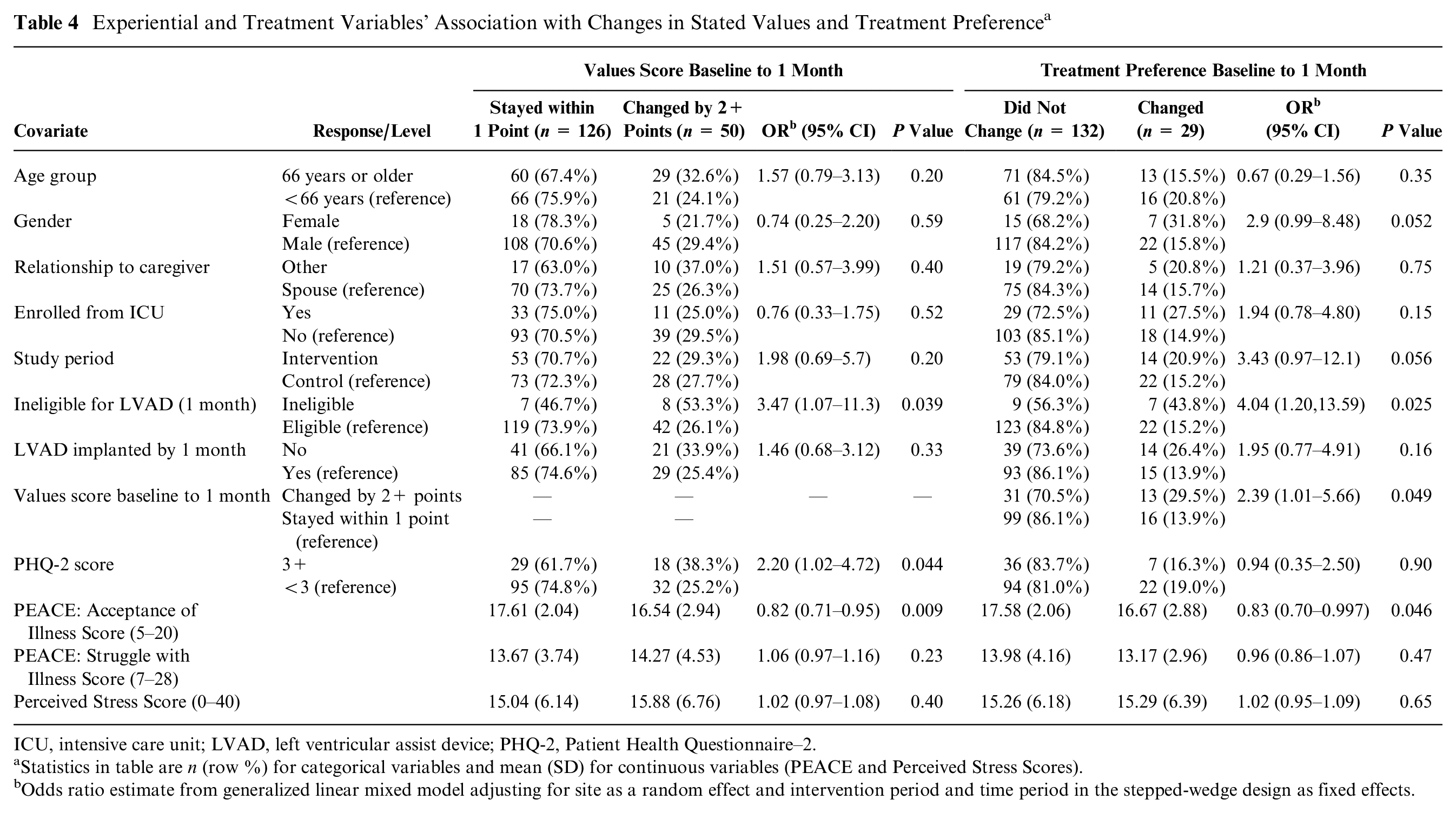
ICU, intensive care unit; LVAD, left ventricular assist device; PHQ-2, Patient Health Questionnaire–2.
Statistics in table are n (row %) for categorical variables and mean (SD) for continuous variables (PEACE and Perceived Stress Scores).
Odds ratio estimate from generalized linear mixed model adjusting for site as a random effect and intervention period and time period in the stepped-wedge design as fixed effects.
Similarly, participants who were deemed ineligible for LVAD were more likely to change their treatment preferences than participants who were eligible for LVAD (OR, 4.04; 95% CI, 1.20–13.59; P = 0.008). Higher illness acceptance score was associated with lower likelihood of changing treatment preferences in this subset as well (OR, 0.83; 95% CI, 0.70–0.997; P = 0.046).
In our analyses on the subset of participants who were found to be medically eligible for LVAD implantation by the advanced heart failure team, we observed a similar relationship between illness acceptance and both outcomes of values change and treatment preference change: higher illness acceptance scores were associated with less likelihood of change (supplementary tables). As in the total sample, participants in this subset with higher PHQ-2 scores were more likely to have a change in values score.
Discussion
When facing the life-altering decision of whether to pursue LVAD treatment, most patients remain stable in their values and preferences for treatment. Virtually all such patients continued to value an aggressive approach that maximized chances of survival and thus remained steadfast in their stated preference to get an LVAD. Conversely, about a quarter of patients reported significant changes to their values and associated preferences in the 6 months after initiating LVAD evaluation, highlighting a clinical need to identify and support these patients as they wrestle with this choice. Within the group of patients who were deemed medically eligible for an LVAD, those with depression and lower acceptance of illness were more likely to report significant changes to values and preferences, highlighting the need for improved methods to elicit and clarify values in certain populations if we are to achieve high-quality medical decision making and patient-centered care.
The consistently high value placed on aggressive care and preferences for LVAD for the majority of this population is not surprising (Figure 2A). Participants were enrolled at the time they agreed to undergo formal evaluation for LVAD therapy, often following their agreement for referral to highly specialized medical centers. Many of these patients may have engaged in consideration of whether or not they wanted to pursue aggressive therapy prior to presenting for evaluation, thereby removing an unknown number of patients who may not have been interested in LVAD therapy prior to referral. Therefore, the minority of patients whose reported values and preferences changed (Figure 2A,B) and without a discernable pattern for how values scores changed should receive particular clinical attention. Notably, the major shift observed in terms of treatment preference was from “Unsure” to “Get a DT LVAD” (Figure 2B).
Our findings may contextualize values and treatment preference instability seen in other studies. Previous studies in similarly complex patient populations have focused on desired outcomes of treatment (e.g., amelioration of symptoms, preservation of functioning)21,22 in addition to the development of taxonomies of values23–25 specific to certain decision contexts. However, in a recent trial, 68% of metastatic cancer patients’ treatment preferences remained stable over the course of treatment. 8 Changes in preferences, by contrast, have been associated with severity of current illness,5,6 which led us to incorrectly hypothesize that stability of values and preferences among the critically ill LVAD candidate population would be higher than what we found.
Helping patients with values clarification is challenging for clinicians in their existing clinical workflow, although doing so more effectively may help care teams to target appropriate interventions to help patients align their decisions to what they “actually” want.26,27 A substantial proportion of our sample illustrated consistency in their values and treatment preferences, suggesting that single-item measures collected longitudinally may be an effective way to not only identify values but also flag individuals whose values or preferences may be comparatively labile. For most patients, this would encourage clinicians to normalize the desire to maximize survival, while simultaneously empathizing with the stress, anxiety, and uncertainty associated with the decision itself. If the subset of patients struggling to clarify their values and associated preferences is easily identified, as our data suggest, clinicians can adjust the medical decision making process to better explore values and therapeutic tradeoffs—including the possible dominance of different value domains for that specific patient28,29—or address other underlying psychosocial needs. Such a process should thus ideally reduce decisional conflict and better match treatments to those the patient actually prefers.
Efforts to measure and improve important outcomes of decision quality—especially value-choice concordance—are challenged by a number of psychological and practical issues. First, there is the labile nature of values and preferences among any subset of patients. The fluid nature of patient values and preferences is less problematic for treatments that are reversible (e.g., preventive medications, placement of devices that could be deactivated or removed). More fateful and irreversible decisions, including whether or not to pursue LVAD therapy, demand a higher degree of certainty in matching treatments to patients’ core values and true preferences. 5 This level of certainty about what is “right” for a patient requires a high degree of stability in those values and preferences. Second, methods by which to assess patient values struggle with a tradeoff between psychometric validity and practicality in clinical environments. Our single-item method—while certainly feasible within even the most hectic clinical workflow—leaves little in the way of traditional psychometric validation, limiting our ability to further assess validity and reliability of the personal values construct as it was assessed. However, when one considers the association between changes in reported values and PEACE Acceptance of Illness Score alongside the instability of treatment preferences and the proportion of participants for whom reported values did not change, this item appears to adequately capture this construct. Further research is needed to evaluate how this item, with or without the PEACE Acceptance scale, would perform when administered alongside items similarly assessing the complex array of psychosocial considerations that weigh on patients with end-stage heart failure considering LVAD therapy.
Reconceptualizing values instability as a common aspect of decision making in a significant minority of patients, rather than as an indicator of weak psychometric properties of patient-reported outcome measures themselves, has potentially wide-reaching impact on the manner in which patients are supported while making major medical decisions.
Limitations
This analysis has several limitations. First, missing data were common, most frequently due to functional decline and death, but also associated with a medical determination of ineligibility for LVAD. Second, it is possible that being denied LVAD candidacy affects the manner in which patients consider their values, such that they are responding to the menu of available options (which no longer includes LVAD), rather than what they might have desired if other options were viable. We attempted to account for this difference both by including a term for LVAD eligibility in our primary analysis and by conducting a post hoc sensitivity analysis including only LVAD-eligible patients, and our findings for values appeared consistent across these subgroups. It may be possible that missingness among patients not eligible for a device in terms of changes to treatment preferences may have underestimated the actual effect of no longer having device therapy as an option. Nevertheless, we may not have captured actual changes to patient values among medically ineligible patients using a single-item measure. Third, the sample size would not allow for the increased explanatory value a more highly specified model might provide. Fourth, the educational process that coincides with LVAD evaluation may be partially responsible for changes in participants’ expression of values and what preferences they have regarding LVAD therapy. Finally, we elected not to place a valence on the types of changes observed with respect to treatment preference (i.e., whether a change from being unsure to having a preference was desirable). It may be the case that patients clarifying their preferences over time are characteristically different from those whose preferences changed in other ways. This analysis did not have sufficient sample size to meaningfully analyze these differences, but future research should consider whether specific preference changes are advantageous.
Conclusion
In the setting of difficult tradeoffs created by the option of LVAD, a substantial minority of patients reported changing prioritization of competing values. Understanding how to improve values clarification and preference elicitation with patient–provider communication is critical in the context of advanced therapy options for life-threatening illnesses.
Supplemental Material
sj-docx-1-mdm-10.1177_0272989X211028234 – Supplemental material for Changes over Time in Patient Stated Values and Treatment Preferences Regarding Aggressive Therapies: Insights from the DECIDE-LVAD Trial
Supplemental material, sj-docx-1-mdm-10.1177_0272989X211028234 for Changes over Time in Patient Stated Values and Treatment Preferences Regarding Aggressive Therapies: Insights from the DECIDE-LVAD Trial by Christopher E. Knoepke, Erin L. Chaussee, Daniel D. Matlock, Jocelyn S. Thompson, Colleen K. McIlvennan, Amrut V. Ambardekar, Elisabeth M. Schaffer, Prateeti Khazanie, Laura Scherer, Robert M. Arnold and Larry A. Allen in Medical Decision Making
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through a Patient-Centered Outcomes Research Institute (PCORI) Program Award (CDR-1310-06998). All statements in this report, including its findings and conclusions, are solely those of the authors and do not necessarily represent the views of PCORI, its Board of Governors, or Methodology Committee.
This work was also supported in part by the National Heart, Lung, and Blood Institute (1K23HL105896, LAA; 1K23HL153892, CEK), the American Heart Association (18CDA34110026, CEK), the Heart Failure Society of America (CKM), the National Institute on Aging (1K23AG040696, DDM), and REDCap database hosting through University of Colorado supported by NIH/NCRR Colorado CTSI (grant UL1 TR001082). This trial was registered at clinicaltrials.gov as NCT02344576.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
