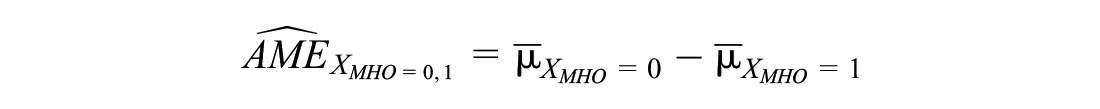Abstract
Objectives
This study aims to characterize the spillover effects of selected mental health disorders (episodic mood disorder (EMD), anxiety, substance use disorder (SUD), schizophrenia, attention-deficit/hyperactivity disorder (ADHD), and dementia) on family members’ health-related quality of life and to compare the magnitude of spillover effects across these types.
Methods
Using the 2000–2015 Medical Expenditure Panel Survey, households having individuals with mental health disorders were identified. The SF-12 and EQ-5D surveys were used to acquire utility and health status scores for household members. The outcomes in households including an individual with a mental health disorder were compared to those of the control group (absence of individuals with mental health disorders in the household). We also compared a total of 15 pairs of diseases based on the SF-6D scores. A beta generalized estimating equation model was employed.
Results
Average scores of utility and health status among individuals living with a member with a mental health disorder in the household were statistically lower than those of the control group and; for the SF-6D, met the minimally important difference for SUD, schizophrenia, and dementia. Differences in the SF-6D scores were statistically significant for 5 pairs of the mental health disorders: EMD–anxiety, EMD–ADHD, dementia–anxiety, dementia–ADHD, and schizophrenia–ADHD.
Conclusions
This study provides evidence of family spillover effects in mental illness using both utility and health status measures from a US representative sample. Integrating this evidence into clinical and policy decision making as well as economic evaluations would allow for a more comprehensive valuation of the societal benefits of mental and behavioral health interventions.
The impact of illness on health and well-being extends beyond the affected individuals to their family members. Family caregiving often imposes considerable physical and emotional tolls not only on the ill individual but also on other family members, adversely affecting the health outcomes and quality of life of the latter.1–6 As suggested by the growing body of evidence in the literature, considering the spillover effects of illness in clinical decision making may provide immediate and long-term benefits to patients and family members, resulting in improved physical health and psychological well-being for all.7–9
Incorporating spillover effects into economic evaluations facilitates more efficient resource allocation. Experts in cost-effectiveness analysis (CEA) and decision makers in health technology assessment agencies recommend the inclusion of spillover effects on the caregiver’s quality of life in the reference case (United Kingdom and Canada) or scenario analysis (Ireland, Scotland, and Australia) when estimating the cost-effectiveness of an intervention program.10–14 This approach may enhance the value of an intervention under evaluation, specifically, in a way that the incremental cost-effectiveness ratio (ICER) will be lower when the health benefits for family caregivers are included in the denominator.15,16 For example, Lavelle et al. 15 and Lin et al. 16 recalculated ICERs of interventions targeted for pediatric patients and patients with Alzheimer’s disease, respectively, with and without spillover effects and found that incorporating health spillover effects of family caregivers (i.e., caregiving effect) decreased ICERs. However, none of the studies included in their analysis addressed spillover effects on noncaregiving family members who may also be affected (i.e., family effect)—the value of the interventions was still underestimated without the inclusion of both types of spillover effects within a family.17,18 Therefore, more accurate estimates of the value of medical interventions fully accounting for caregiving and family effects can yield better informed decision making among patients, clinicians, and policymakers, thereby ensuring optimal resource allocation.19,20
The spillover effects of mental health disorders on the health-related quality of life (HRQoL) of family members have been well documented. 21 Research findings are consistent among different types of mental health disorders, such as depression, alcohol and opioid use disorders, schizophrenia, autism, and dementia/Alzheimer’s disease, further demonstrating that negative spillover effects must be considered when estimating the cost-effectiveness.8,9,22–25 However, most studies estimate the HRQoL of a single mental health condition. Direct estimates comparing the size of spillover effects across several mental health conditions are lacking. In addition, previous results originated from varying sample populations, weakening comparisons of spillover effects between mental health disorders.
This study aims to address these critical gaps. Study objectives include 1) characterizing the spillover effects of selected mental health disorders (i.e., episodic mood disorder (EMD), anxiety, substance use disorder (SUD), schizophrenia, attention-deficit/hyperactivity disorder (ADHD), and dementia) on family members’ utility (SF-6D and EQ-5D) and health status (i.e., physical and mental composite scores (PCS and MCS)) of the 12-item Short Form Health Survey (SF-12), from a US nationally representative sample, and 2) comparing the magnitude of spillover effects across these types.
Methods
Data
We pooled 16 years of data from the 2000–2015 Medical Expenditure Panel Survey (MEPS) in which all the SF-12 survey (SF-12v1 [2000–2002] and SF-12v2 [2003–2015]), EQ-5D (3L; time trade-off valuation) survey (2000–2003), and International Classification of Diseases, Ninth Revision (ICD-9) codes were employed.26–28 MEPS is a set of extensive interview surveys of the noninstitutionalized US population, collected from individuals and their families via 5 rounds over a 2-y period. Particularly, among the general population surveys in the United States, the 2000–2003 MEPS provide a unique opportunity to obtain information of 2 different types of preference-based HRQoL scores (i.e., EQ-5D and SF-6D). Each interview round solicits data relevant to a specific period. Rounds 1 and 2 take place in year 1, while Rounds 3 and 4 take place in year 2. Full-Year Consolidated Data files and Medical Conditions files were used in this analysis. The Full-Year Consolidated Data files contain responses from the SF-12 and EQ-5D surveys administered in Rounds 2 and 4 to individuals aged 18 y and older, as well as the sociodemographic information from each respondent. The Medical Conditions files consist of self-reported medical conditions classified by the ICD-9 coding system. In every round, a primary respondent is asked to report on the medical conditions experienced by household members over the period between each round. MEPS is a publicly available and deidentified data set; thus, institutional review board approval was not required for this research.
Measures
We first identified households having individuals with mental health disorders using the Medical Conditions files. With the operational definitions based on ICD-9 codes (see Suppl. Table S1), we selected 7 types of mental health disorders: 1) EMD, 2) anxiety, 3) SUD, 4) schizophrenia, 5) ADHD, 6) dementia, and 7) a combination of all 6. The rationale for choosing the 7 different categories is presented in Appendix.
We then subgrouped these types in 2 ways. First, we subgrouped by age (i.e., <18 y [child] and ≥18 y [adult]) to measure the difference in spillover effects by age groups. Second, we subgrouped by time of disease occurrence, defining existing disease as being reported in both Round 1 or 2 (i.e., year 1) and Round 3 or 4 (i.e., year 2), and new disease as first being reported only in Round 3 or 4 (i.e., year 2). We hypothesized that individuals from the households including a child diagnosed with a mental health disorder or a patient experiencing a mental health disorder over at least 2 consecutive Y would report lower HRQoL scores compared to their counterparts. Finally, we made pairwise comparisons among all 6 mental health disorders.
We used the SF-12 and EQ-5D surveys to acquire health status and utility scores for household members. PCS and MCS are summary measures of health status from the SF-12 survey for the physical and mental health domains, respectively. 27 Scores range from 0 to 100, with higher scores indicating better health status. To produce the SF-6D, we converted SF-12 survey data of individuals using a validated algorithm. 29 The SF-6D is a preference-based utility measure that yields a single index score ranging from 0 (death) to 1 (full health). The measure is composed of 6 dimensions of health (i.e., physical functioning, role limitations, social functioning, pain, mental health, and vitality). 30 In addition, the EQ-5D, another preference-based utility measure, was selected to compare between different utility measures. 31 The EQ-5D comprises 5 dimensions of health (mobility, self-care, usual activities, pain/discomfort, and depression/anxiety). When multiplied by the time spent in each health state, the SF-6D and EQ-5D scores are used to produce quality-adjusted life years (QALYs) and thus can be applied in cost-utility analysis. In MEPS, both the SF-12 and EQ-5D surveys are conducted twice for individuals aged 18 y and older, in Rounds 2 and 4, respectively. We used the scores from Round 4 for the analysis.
Study Population
We established 2 separate sets of study populations, one each for outcome measures derived from the SF-12 (Figure 1) and the EQ-5D surveys (Suppl. Figure S1). The study population for the analysis of PCS, MCS, and SF-6D is composed of adults who completed all items on the SF-12 surveys administered in Rounds 2 and 4. The study population for EQ-5D included only those who answered both SF-12 and EQ-5D surveys administered in Rounds 2 and 4. Because our primary interest was in determining the spillover effects for someone without a mental health disorder living with someone with a mental health disorder, we restricted the data set to MEPS survey respondents who did not have a mental health disorder. In addition, we excluded individuals from the households where 1) the member with mental health disorder left the household prior to Round 4, 2) the member only used mental health services at Round 1 or 2, 3) the member experienced more than 1 mental health disorder from the selected diseases, and 4) families where a child and adult with mental health disorder live together. With the first 2 exclusion criteria, we can determine the time of disease occurrence as an existing or new disease using the Round where the disease is first reported. The third criterion allows us to estimate a spillover effect attributable solely to a single mental health disorder. Last, using the fourth criterion, we can dichotomize the household status into child or adult based on age of members with a mental health disorder.
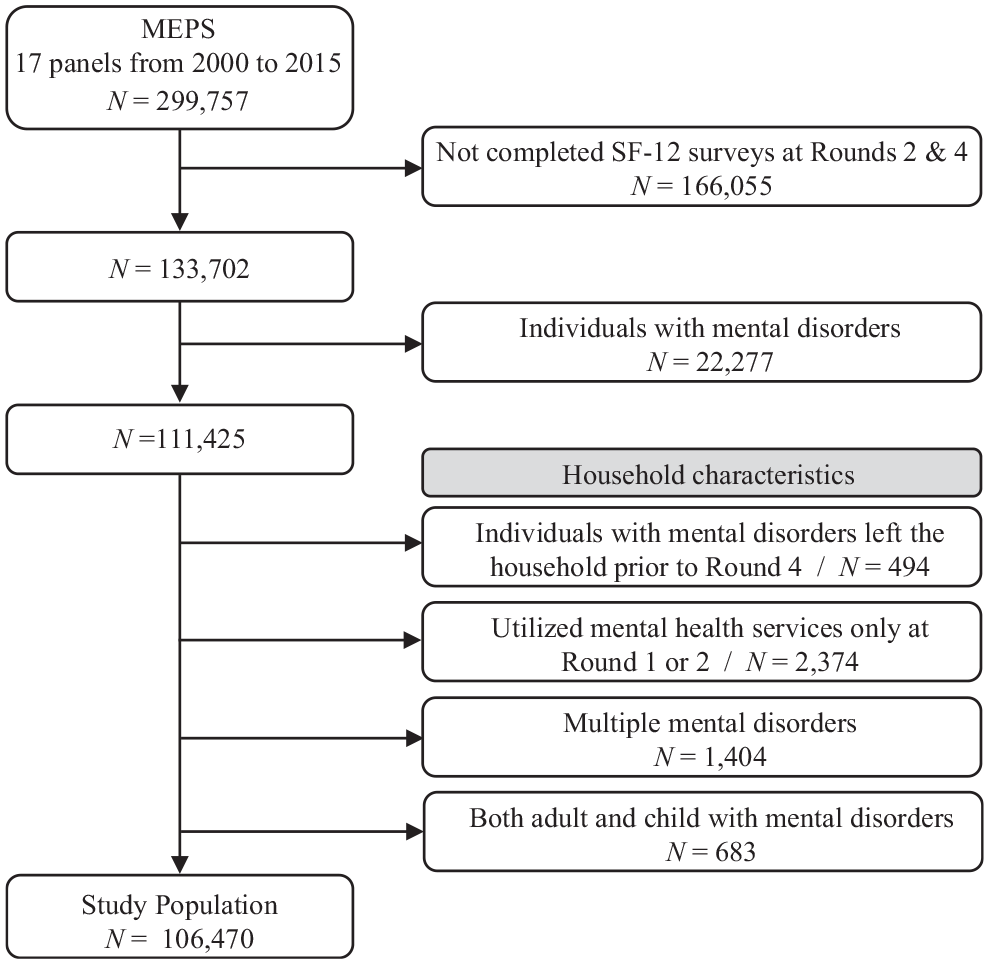
Flow diagram of the selection procedure for the study population (SF-6D, PCS, and MCS)
Statistical Analysis
Beta generalized estimating equations
We employed a beta generalized estimating equation (GEE) model to determine spillover effects of mental health disorders on household members’ HRQoL scores (i.e., SF-6D, EQ-5D, and converted PCS and MCS (
We examined the primary objective in 3 ways to identify any joint effects of the mental health status and disease occurrence by age (child/adult) and time (existing/new). Specifically, for the ith individual in the jth household
Model 1:
Model 2:
Model 3:
where g(ċ) is a logit link function.
The rationale for the 3 models for estimating the spillover effects is as follows:
Model 1 compares the outcomes of the control group to those of the group living with a patient with a mental health disorder.
Model 2 examines differences in the outcomes across the 3 mutually exclusive categories based on age of the patients: 1) the control group, 2) the group living with the adult patient, and 3) the group living with the child patient. We conducted the pairwise comparison with these categories: the control group v. the adult patient group, the control group v. the child patient group, and the adult patient group v. the child patient group.
Model 3 investigates differences in the outcomes across the 3 mutually exclusive categories based on disease onset timing of the patients: 1) the control group, 2) the group living with a patient with an existing mental health disorder, and 3) the group living with a patient with a newly occurring mental health disorder. Three pairwise comparison groups were also created: the control group v. the existing patient group, the control group v. the new patient group, and the existing patient group v. the new patient group.
To compare the magnitude of the spillover effects across the 6 mental health disorders, we adopted a similar model to those described in Models 1–3 above but included
Average marginal effect and standard error of beta GEE
Consider only Model 1 as an example. We calculated the AME using the following formula: 36
where
We applied the Sidak correction for the formal evaluation of significance for individual mental health disorders to adjust for multiple comparisons for each outcome (i.e., a familywise alpha level =
Results
The study population for the analysis of PCS, MCS, and SF-6D consisted of 106,470 adults from 65,389 households (Table 1). Among these adults, 13,926 (13.1%) belonged to households with members experiencing mental health disorders: specifically, 6,629 (6.2%) for EMD, 4,311 (4%) for anxiety, 167 (0.2%) for SUD, 157 (0.1%) for schizophrenia, 2,276 (2.1%) for ADHD, and 386 (0.4%) for dementia. The mean age of the study population was 45 y. The proportion of non-Hispanic whites was 67%, 53% were female, 55% were married, and 37% lived in households with incomes lower than 200% of the federal poverty level. The average score of the PCS, MCS, and SF-6D was 50, 53, and 0.8, respectively. The sample population for analysis incorporating EQ-5D data reduced to 17,776 adults from 11,725 households. Demographic characteristics of the EQ-5D sample were similar to those from the SF-12 sample, with the exception that the proportion of non-Hispanic whites was 15% higher in the EQ-5D sample. The mean EQ-5D score was 0.84.
Summary statistics of the study population
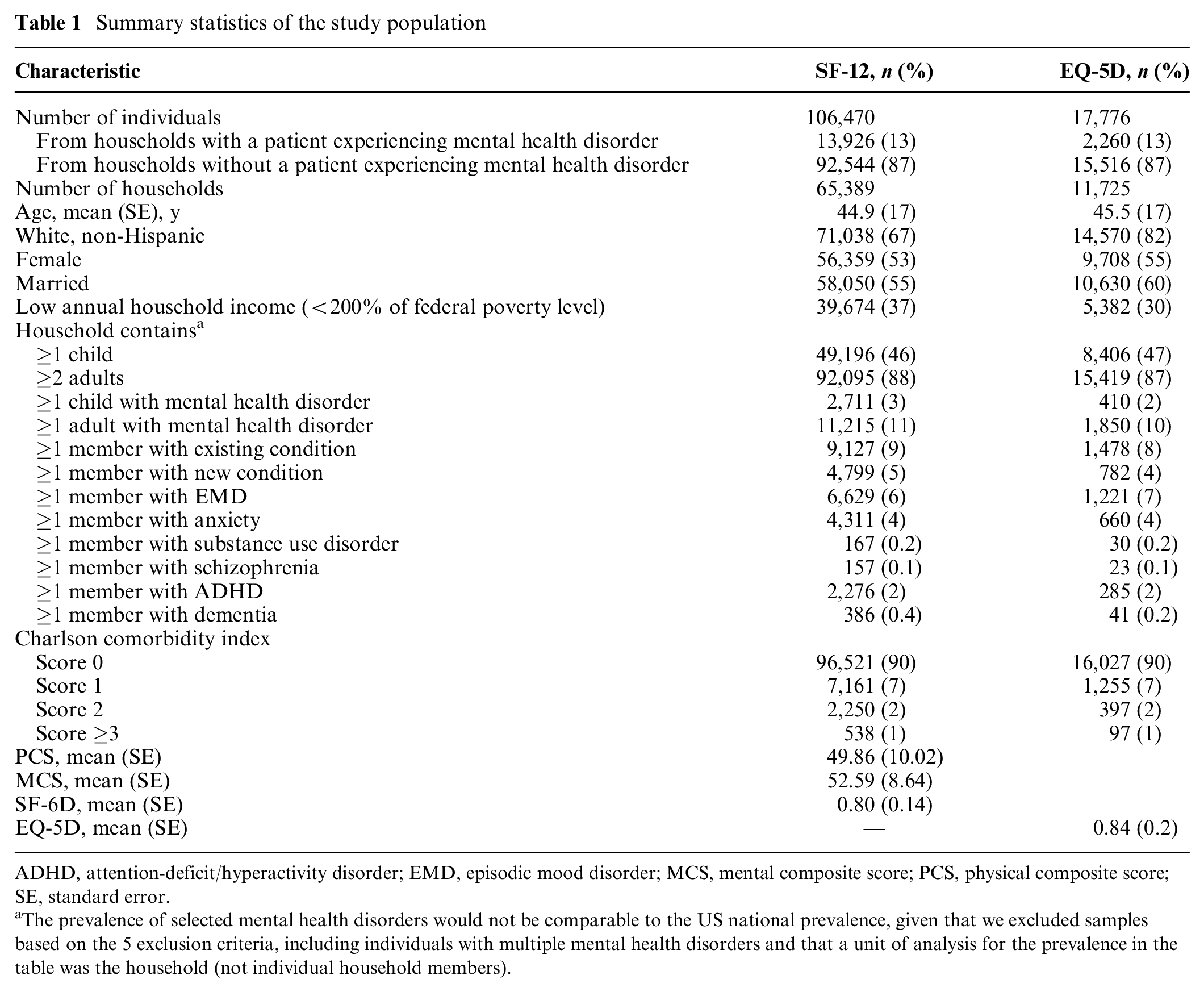
ADHD, attention-deficit/hyperactivity disorder; EMD, episodic mood disorder; MCS, mental composite score; PCS, physical composite score; SE, standard error.
The prevalence of selected mental health disorders would not be comparable to the US national prevalence, given that we excluded samples based on the 5 exclusion criteria, including individuals with multiple mental health disorders and that a unit of analysis for the prevalence in the table was the household (not individual household members).
Our models show that for some mental health disorders, there are statistically significant associations between the presence of a patient with a mental health disorder in the household and the HRQoL of the survey respondent (Tables 2 and 3, Figures 2 and 3). Specifically, in all 7 types of mental health disorders, the results of SF-6D demonstrate that average scores among individuals living with a family member diagnosed with a mental health disorder are statistically lower than those of the control group. The differences in the scores range from −0.013 to −0.052, with individual scores as follows: –0.024 (95% CI; –0.028, –0.020) for the combination category, –0.029 (–0.035, –0.024) for EMD, –0.017 (–0.024, –0.009) for anxiety, –0.044 (–0.080, –0.008) for SUD, –0.052 (–0.092, –0.012) for schizophrenia, –0.013 (–0.023, –0.002) for ADHD, and –0.051 (–0.075, –0.026) for dementia. Regarding the EQ-5D, however, we only found statistically significant associations from the combination category and EMD with the differences of –0.02 (–0.035, –0.005) and –0.022 (–0.042, –0.002), respectively.
Results of the beta generalized estimating equation models for SF-6D and EQ-5D
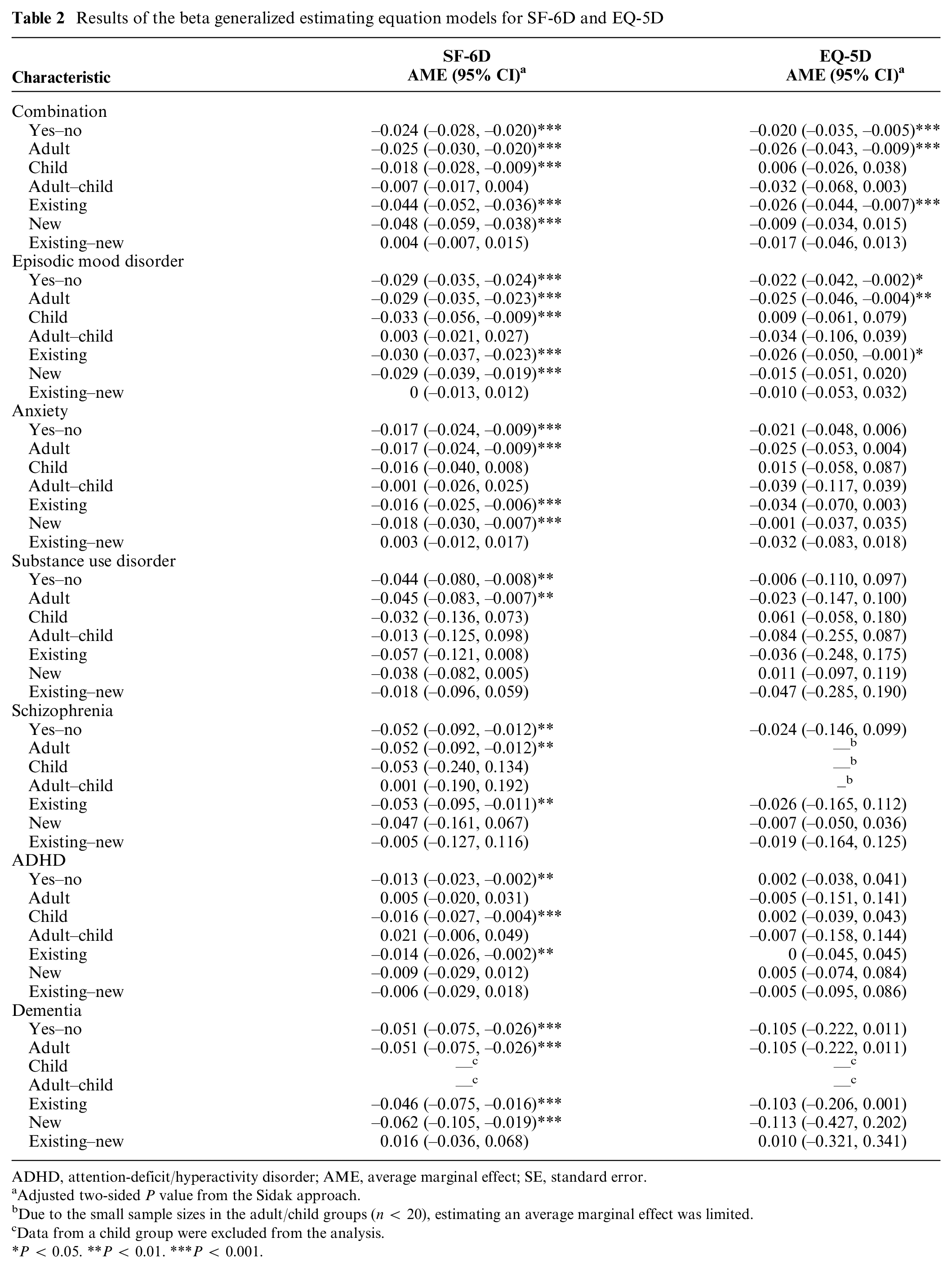
ADHD, attention-deficit/hyperactivity disorder; AME, average marginal effect; SE, standard error.
Adjusted two-sided P value from the Sidak approach.
Due to the small sample sizes in the adult/child groups (n < 20), estimating an average marginal effect was limited.
Data from a child group were excluded from the analysis.
P < 0.05. **P < 0.01. ***P < 0.001.
Results of the beta generalized estimating equation models for PCS/100 and MCS/100
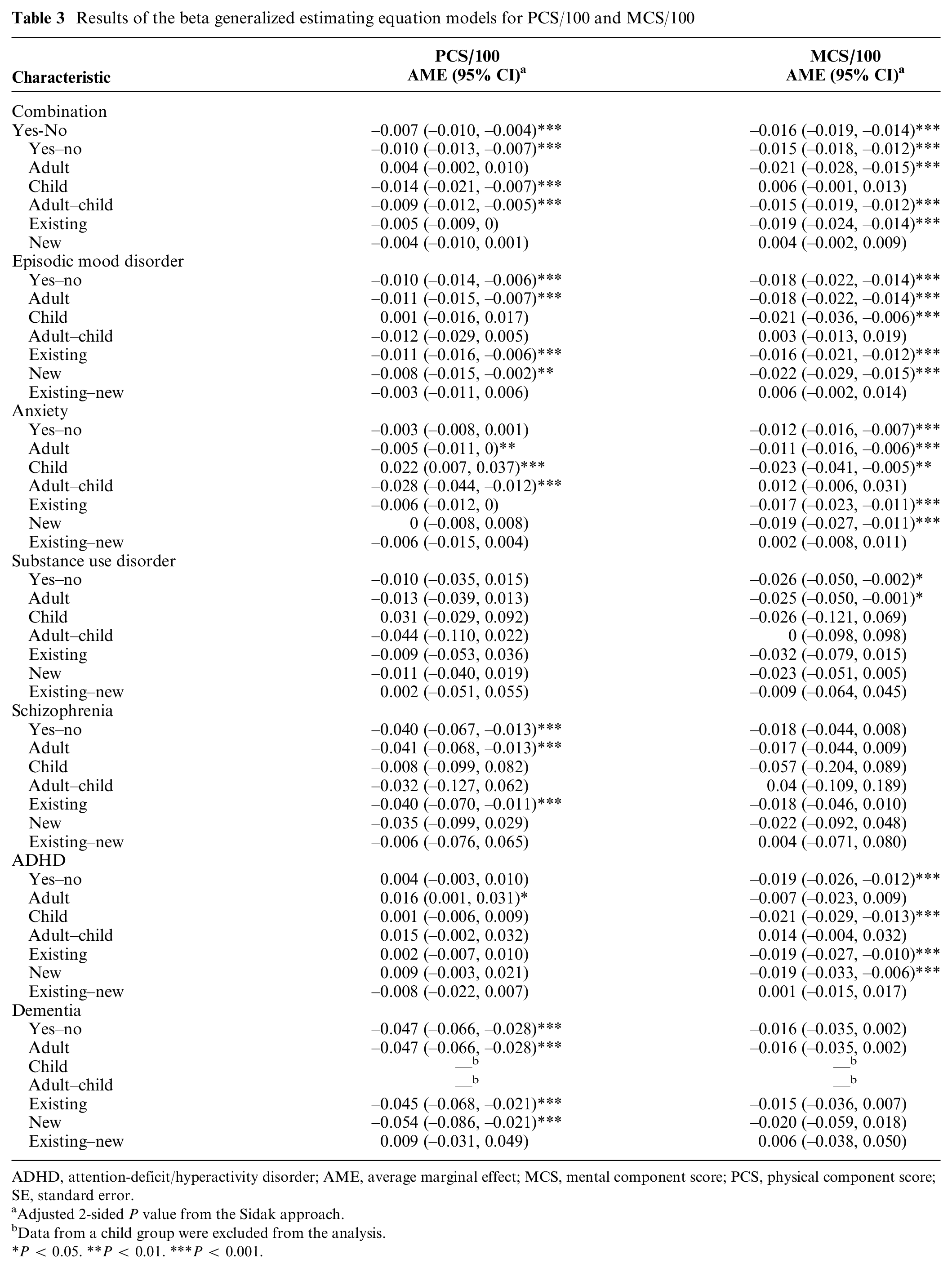
ADHD, attention-deficit/hyperactivity disorder; AME, average marginal effect; MCS, mental component score; PCS, physical component score; SE, standard error.
Adjusted 2-sided P value from the Sidak approach.
Data from a child group were excluded from the analysis.
P < 0.05. **P < 0.01. ***P < 0.001.
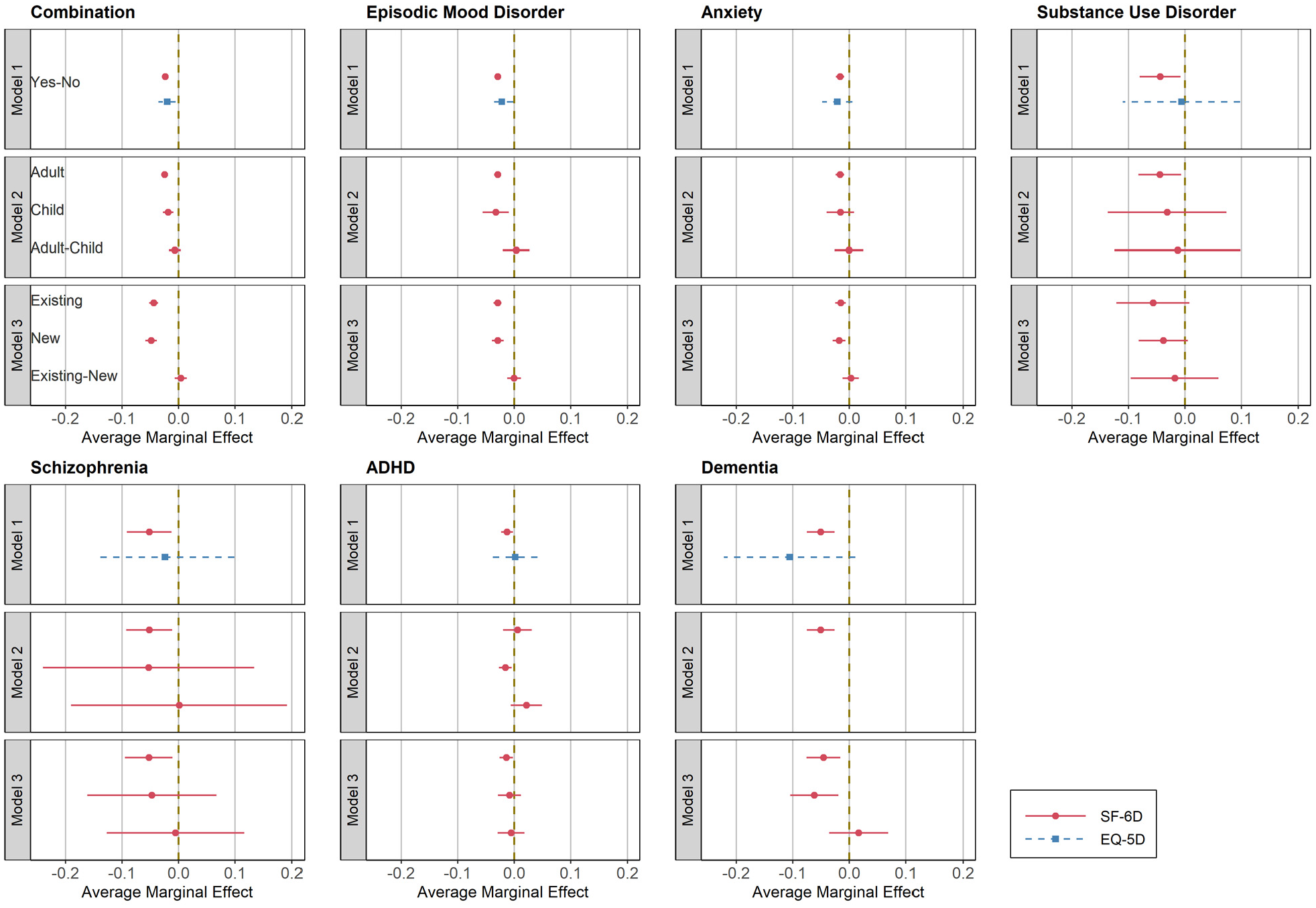
Results of spillover effects by SF-6D and EQ-5D. Simultaneous confidence intervals using Sidak approach are applied for each outcome. ADHD, attention-deficit/hyperactivity disorder.
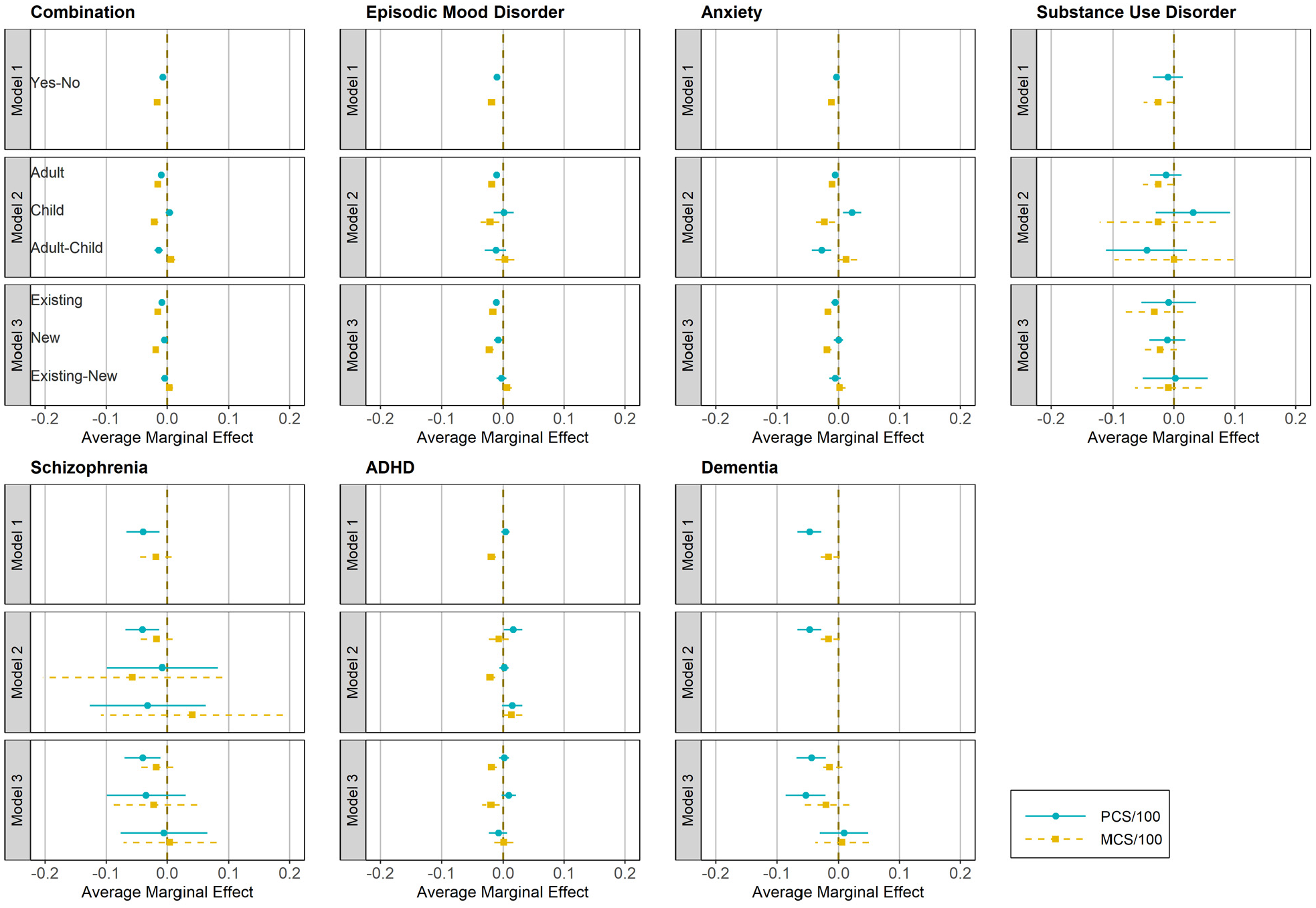
Results of spillover effects by PCS/100 and MCS/100. Simultaneous confidence intervals using the Sidak approach are applied for each outcome. ADHD, attention-deficit/hyperactivity disorder; MCS, mental component score; PCS, physical component score.
Moreover, the results of
Differences in the SF-6D were statistically significant for 5 of the 15 pairs of the mental health disorders: 1) EMD–anxiety, 2) EMD–ADHD, 3) dementia–anxiety, 4) dementia–ADHD, and 5) schizophrenia–ADHD (Table 4 and Figure 4). For example, the average SF-6D among individuals living with a family member experiencing EMD is 0.013 (–0.021, –0.005) points lower than that of those living with a family member experiencing anxiety.
Results of comparison across 15 disease pairs by SF-6D
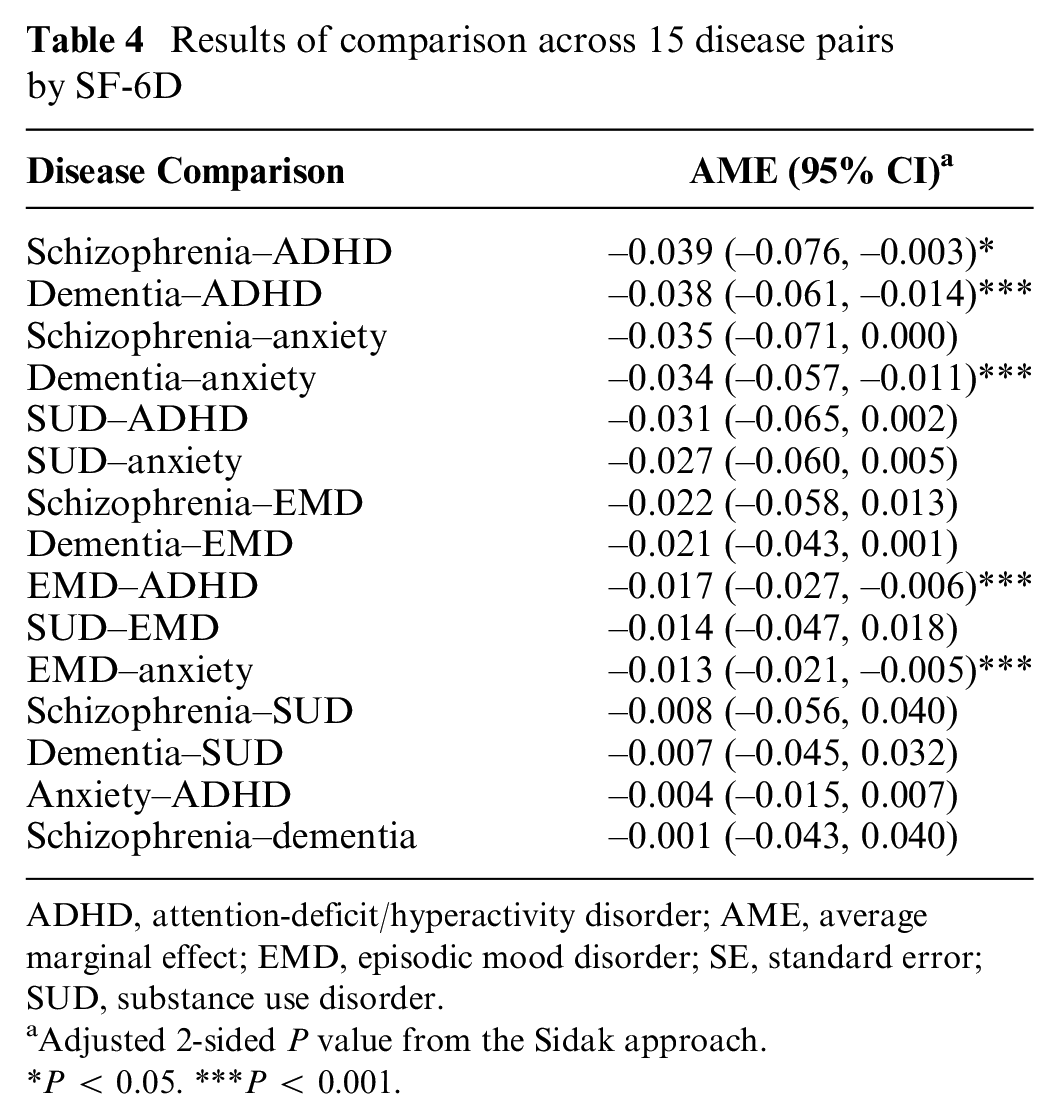
ADHD, attention-deficit/hyperactivity disorder; AME, average marginal effect; EMD, episodic mood disorder; SE, standard error; SUD, substance use disorder.
Adjusted 2-sided P value from the Sidak approach.
P < 0.05. ***P < 0.001.
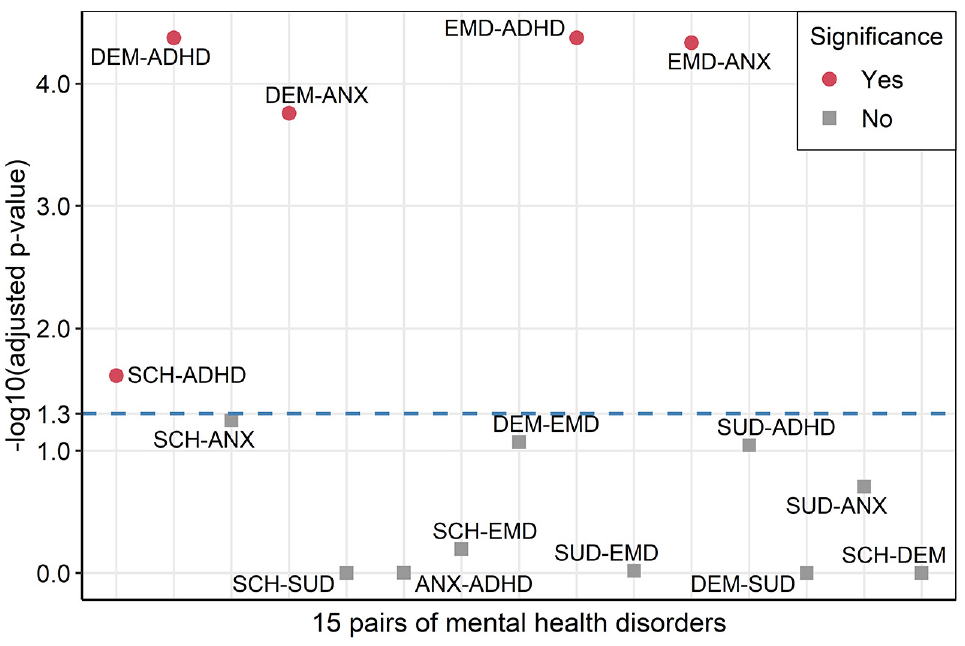
Results of comparison across 15 disease pairs by SF-6D. Adjusted 2-sided P values by Sidak approach are presented with –log10 transformation. This transformation changes lower P values as larger values. The dashed line indicates a significance threshold at α = 0.05 (1.3 = −log10 (0.05)). ADHD, attention-deficit/hyperactivity disorder; ANX, anxiety; DEM, dementia; EMD, episodic mood disorder; SCH, schizophrenia; SUD, substance use disorder.
Discussion
Our findings provide evidence of negative spillover effects on HRQoL among individuals living with a family member diagnosed with a mental health disorder. That is, the presence of a family member with a mental health disorder in the household is associated with lower utility and health status scores among household members who do not experience any mental health disorders. The magnitude of spillover disutility between the SF-6D and EQ-5D is comparable from 2 types, given that the spillover disutility amounts to –0.024 and –0.02 for the combination category, as well as –0.029 and –0.022 for EMD in the SF-6D and EQ-5D, respectively. We did not observe evidence of spillover disutility from the EQ-5D in the other 5 types of mental health disorders, which may be attributable to the small sample sizes within these types in the EQ-5D data set. We also found that the magnitude of spillover disutility for the SF-6D was heterogeneous among the selected mental health disorders. However, ranking the order of magnitude across the mental health disorders may be infeasible since differences in the SF-6D for 10 of the 15 pairs of diseases are not statistically significant. In addition, we recognize that PCS and MCS scores do not show a parallel pattern in some types. In particular, the negative spillover effects are demonstrated in schizophrenia and dementia for the PCS scores, whereas the same effects are identified only in anxiety, SUD, and ADHD for the MCS scores. Considering that the evidence of spillover effects on physical health is relatively rare in the literature compared to the effects on mental health, this finding warrants further research to understand a potential role of mental health deterioration as a mediator for the decline of physical health functioning among caregiving and noncaregiving family members.42–44
Using the 3 different models, we attempted to characterize heterogeneity of the spillover effects according to the age of patients (child/adult) and time of disease occurrence (existing/new). For example, the study conducted by Lavelle et al. 45 investigated the variation of spillover effects depending on familial relationship with a patient (e.g., parents, spouse, and child), suggesting that a child patient would induce a greater degree of spillover effects. Moreover, it seems plausible that the effects may increase with an additional year of caring for the member who suffers from the disease. However, no prominent patterns of spillover effects were identified by the age and time from the selected mental health disorders. To further understand the heterogeneity derived from the age and time, it may be worth investigating our hypotheses in the context of different health conditions.
Although having an appropriate comparator is crucial to better infer spillover effects, only a handful of studies have attempted to use matched comparison groups to analyze spillover effects of mental illnesses. 21 For instance, Wittenberg et al. 7 investigated spillover effects of chronic conditions, including mental health disorders, on household members’ utility measured by the EQ-5D in MEPS, comparing between household members living with a family member diagnosed with a mental health disorder and those without the family member diagnosed with a mental health disorder. They found that the EQ-5D scores for individuals living with an adult patient with a mental health disorder were 0.013 and 0.021 points lower than for those not living with an adult patient with a mental health disorder, according to the patient’s disease onset timing (i.e., existing/new), respectively. Regarding specific mental health disorders, for example, Gupta et al. 24 estimated the spillover effects of informal schizophrenia caregiving using samples obtained from 5 European Union countries (France, Germany, Italy, Spain, and United Kingdom). Their results showed that the average utility score of the caregivers was –0.07 points lower than that of noncaregivers in terms of SF-6D. Moreover, Gupta et al. 46 and Laks et al. 47 examined the spillover effects of dementia on caregivers’ utility measured by the SF-6D based on sample populations in the United States and Brazil. Their estimates of the spillover effects comparing caregivers to noncaregivers were similar in that the former study reported –0.04, and the latter was –0.033. Also, the study conducted by Serrano-Aguilar et al., 48 which analyzed EuroQol visual analogue scale (EQ-VAS) of informal caregivers of individuals with Alzheimer’s disease in Spain, reported the value of spillover disutility as –0.034 (i.e., VAS scores were converted to utilities by Wittenberg et al. 49 ). Our results support these findings as the spillover estimates are within the range of 95% confidence intervals for our calculations in the combination category, schizophrenia and dementia.
To provide clinically meaningful interpretations, it is imperative to ascertain the magnitude of these differences based on a minimally important difference (MID). The MID refers to the smallest change in a patient-reported outcome score that is perceived as important from the patient’s or clinician’s perspective. 50 According to the study synthesizing the MID for the SF-6D and EQ-5D scores from various patient groups, the mean MID is 0.041 (range, 0.011, 0.097) and 0.074 (–0.011, 0.140) for the SF-6D and EQ-5D, respectively. 51 Although no patient group with mental health disorders was included in this analysis, a direct comparison of the mean MIDs with our results may provide useful information for clinical decision making, given that the study populations of our study are the family members without mental health disorders. Regarding clinical significance, we found that the estimates of differences in the SF-6D scores for SUD (–0.044), schizophrenia (–0.052), and dementia (–0.051) are higher than the corresponding mean MID from Walters and Brazier’s publication. 51 In contrast, none of the statistically significant differences in our EQ-5D scores reached the mean MID.
A lack of guidance for the inclusion of family spillover effects in economic evaluations may pose a challenge to systematic consideration of the effects in practice. To fully incorporate health effects in family members, 2 distinctive sources, the caregiving effects and the family effects, should be considered.17,18 The former refers to the effects of providing informal care on caregivers’ health, whereas the latter refers to the effects on health of family members who care about the well-being of the others. Regarding the current practice of economic evaluations, however, most studies still incorporate the effects partially, omitting the family effects of noncaregiving family members from their calculation of the ICER.15,16 This may lead to suboptimal decision making; if the burden on the primary informal caregiver is only valued without distinguishing between the caregiving effects and the family effects (i.e., the primary caregiver would be affected by both effects), medical interventions in which informal care is not present would have a lower value because of the inconsistency of incorporating family effects across the interventions. 18
Our findings may improve the practice of economic evaluations in measuring family spillover effects in the field of mental and behavioral health. Specifically, the issue of incorporating QALY loss in family members attributable to both caregiving and family effects can be addressed by combining the following methods. Adding the QALY loss of primary caregivers to that of patients in the denominator of the ICER is the most common method of incorporating the caregiving effects. 15 In addition to this, the estimates of QALY loss from our study, which represent the family effects, need to be added to the denominator to fully integrate the family spillover effects. It should be noted that an assumption about the number of family members affected needs to be made when doing this. This combined approach may better inform the policy and decision making process with a more accurate valuation of medical interventions. A caveat is that mental and behavioral health interventions generally lessen the severity of illness, rather than completely prevent or cure it; thus, information on utility impact by severity is needed to reflect the characteristics of the interventions.
This study has several limitations. First, since MEPS does not provide the information about disease severity, the selected types of mental health disorders in this study included the whole range of mental health conditions. This heterogeneity may weaken our ability to estimate the spillover effects precisely in that spillover disutility may only be detected if the severity is above a certain threshold. Given that our study population only consists of those living with an individual diagnosed with a single mental health disorder, the findings of this study can be interpreted as conservative estimates of spillover effects because disease severity may increase with the number of comorbid mental health disorders. Second, we did not consider a comorbidity status of individuals with mental health disorders. As they are more likely to experience chronic illnesses or comorbid mental health disorders than those without mental health disorders, caution should be exercised when interpreting our results.52,53 Specifically, the negative spillover effects in this study may not be solely derived from the single mental health disorder itself but partly from other comorbid conditions (e.g., chronic respiratory diseases) as well. Third, we could not completely disentangle 2 different components of family spillover effects, the caregiving effects and family effects, from our analysis due to lack of information on caregiver status in MEPS. This may lead to overestimation of caregiving effects when incorporating our estimates into CEAs as family effects. Fourth, since the negative relationship between multimorbidity and HRQoL is well established in the literature, we tried to adjust this trend using the CCI. However, the CCI, a useful measure for predicting mortality, may not be the most relevant comorbidity index because some conditions, such as migraine, are associated with significantly lower HRQoL scores yet are not incorporated into the algorithm of CCI.37,54,55 Fifth, although use of beta regression may be superior to ordinary least squares regression for analyzing HRQoL data, its application to individuals with negative utility values may be restricted. 56 Specifically, since the range of beta distribution lies between 0 and 1, negative values (i.e., health state worse than death) of EQ-5D need to be excluded from analysis. Sixth, since our estimates are averaged values from the 16y of pooled data, application of the value to CEAs may be restricted if pharmaceutical innovations for any of selected mental health disorders occurred in a certain years between 2000 and 2015. Last, compared to the SF-12 data set, the EQ-5D data set is small in sample size because the EQ-5D questionnaire was only administered in MEPS from 2000 to 2003. This limited our ability to identify the spillover effects from the EQ-5D among the types of mental health disorders. However, the SF-6D was reportedly more sensitive than the EQ-5D in detecting health spillovers because the former contains more socially oriented dimensions (e.g., social functioning) better reflecting characteristics of the caring situation and provides more response options in the questionnaire.25,57 Our findings that the EQ-5D had a much lower percentage of significant findings than the SF-6D align with the observations from previous studies.
Despite these limitations, our results contribute to the literature to inform CEAs, providing detailed information about the spillover effects from mental health disorders. We captured a heterogeneous trend of family spillovers not only across all 7 types of mental health disorders but also when distinguishing between adult and child patients with a mental health disorder, as well as patients with an existing and newly occurring condition. This evidence may stimulate more rigorous evaluation of health interventions, enabling clinicians and policymakers to appreciate their potential impact that goes beyond the patient, especially in the field of mental and behavioral health. Considering that the role of family caregiving for individuals with mental health disorders may be critical in the successful management of such conditions, this evidence can provide better guidance for clinicians and policymakers. Moreover, using our results as a catalog of estimates for spillover disutility of mental health disorders may assist the inclusion of family spillover effects when parameterizing QALYs in economic evaluations. Leveraging this work to systematize the degree of family spillover effects among other diseases would be advantageous, and thus future research may prioritize this process.
Conclusions
This study provides evidence of family spillover effects in mental illness using both utility and health status measures from a US representative sample. Integrating this evidence into clinical and policy decision making as well as economic evaluation practice would allow for a more comprehensive valuation of the societal benefits of mental and behavioral health interventions.
Supplemental Material
sj-docx-1-mdm-10.1177_0272989X211027146 – Supplemental material for Spillover Effects of Mental Health Disorders on Family Members’ Health-Related Quality of Life: Evidence from a US Sample
Supplemental material, sj-docx-1-mdm-10.1177_0272989X211027146 for Spillover Effects of Mental Health Disorders on Family Members’ Health-Related Quality of Life: Evidence from a US Sample by Donghoon Lee, Yeonil Kim and Beth Devine in Medical Decision Making
Footnotes
Acknowledgements
We are grateful to Lars Almquist and 3 anonymous reviewers for their helpful comments on earlier versions of the manuscript.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.