Abstract
Objective
Shared decision making (SDM) tools can help implement guideline recommendations for patients with atrial fibrillation (AF) considering stroke prevention strategies. We sought to characterize all available SDM tools for this purpose and examine their quality and clinical impact.
Methods
We searched through multiple bibliographic databases, social media, and an SDM tool repository from inception to May 2020 and contacted authors of identified SDM tools. Eligible tools had to offer information about warfarin and ≥1 direct oral anticoagulant. We extracted tool characteristics, assessed their adherence to the International Patient Decision Aids Standards, and obtained information about their efficacy in promoting SDM.
Results
We found 14 SDM tools. Most tools provided up-to-date information about the options, but very few included practical considerations (e.g., out-of-pocket cost). Five of these SDM tools, all used by patients prior to the encounter, were tested in trials at high risk of bias and were found to produce small improvements in patient knowledge and reductions in decisional conflict.
Conclusion
Several SDM tools for stroke prevention in AF are available, but whether they promote high-quality SDM is yet to be known. The implementation of guidelines for SDM in this context requires user-centered development and evaluation of SDM tools that can effectively promote high-quality SDM and improve stroke prevention in patients with AF.
Keywords
Atrial fibrillation (AF) is a heart arrhythmia associated with a 5-fold increase in the risk of stroke. It is estimated that 30% of people with AF develop at least 1 cerebrovascular event in their life time1–3; this event is more likely to be fatal in patients with AF (19%–35%) compared to patients without AF (5%–14%). 4 Stroke survivors live with physical and cognitive disabilities, and their families and caregivers often experience social, physical, emotional, and financial difficulties.5–7
Large randomized trials have demonstrated the benefits of anticoagulation in reducing the risk of AF-related strokes, 8 yet many at-risk patients do not receive these benefits9–11 as less than 50% of high-risk patients are treated with anticoagulation therapy 12 and more than 40% discontinue therapy within 12 months.13–18 There are multiple patient- and clinician-associated factors that may lead to underuse of anticoagulants within this population such as inadequate patient/caregiver resources, lack of understanding about risks and benefits, and difficulties with effective communication.19,20
In response to these challenges, and to realize the full benefits of anticoagulation, the 2014 and 2019 guidelines from the American Heart Association, American College of Cardiology, and The Heart Rhythm Society for the management of patients with AF recommended that shared decision making (SDM) be used to individualize antithrombotic care.9,21 This call for SDM emphasizes its role as a patient-centered strategy in forming plans of care that respond well to the threat of stroke in each patient’s clinical and personal contexts.22,23
SDM tools could support the implementation of these guideline recommendations. Effective tools should be feasible to implement in busy clinical practices and could help 1) share tailored information about the available options, 2) clarify the different attributes of the options in patients’ lives and develop preferences about these, 3) support patient-clinician conversations in which these options are considered in the lives of patients, and 4) arrive at an implementable decision. A systematic search conducted in 2016 identified 6 SDM pertinent tools. 24 Since then, direct oral anticoagulants (DOACs), included in only 1 of the 6 tools, have increased in use, and the Centers for Medicare & Medicaid Services (CMS) tied reimbursement to performance and documentation of SDM for patients with AF considering a left atrial appendage closure (LAAC) device. 25
These events have significantly affected SDM surrounding stroke prevention among AF patients. We, therefore, determined that an updated scan of the published record and online resources would be beneficial. The goal of this review was to identify available SDM tools designed to support SDM about stroke prevention for patients with AF and assess their quality and impact on SDM outcomes.
Methods
We conducted a systematic review of academic databases and environmental scanning to collect SDM tools and associated literature about their development and efficacy. The current report follows the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA). 26 The protocol of this study can be accessed by request.
Eligibility Criteria
Eligible SDM tools were developed to support SDM about pharmacological and nonpharmacological strategies (e.g., LAAC device) for stroke prevention in patients with AF. These tools were either patient decision aids (supporting the preparation of patients for SDM) or encounter tools (supporting both patients and clinicians participating in SDM). They were required to include warfarin and ≥1 DOAC as stroke prevention options. We also included any study assessing the impact of any eligible SDM tool v. usual care or other active control on SDM.
Data Sources and Search Strategy
Literature search
An experienced librarian (L.P.) designed a search strategy that was carried out in Ovid MEDLINE and Epub Ahead of Print, In-Process & Other Non-Indexed Citations, and Daily, Ovid EMBASE, Ovid PsycINFO, Ovid Cochrane Central Register of Controlled Trials, Ovid Cochrane Database of Systematic Reviews, Web of Science, and Scopus. The search was conducted from each database’s inception to May 19, 2020 (Supplemental Material 1). There were no restrictions on study design, language, or date of publication.
Environmental scan
A systematic search of social media platforms Facebook and Twitter was conducted and updated as of July 10, 2020, by introducing different combinations of the words atrial fibrillation and shared decision making in their search bars (Supplementary Material 2). In addition, during the data extraction for the systematic review, we extracted all author names and emails. Each author was emailed up to 2 times and asked to verify the information collected about their SDM tool, to identify missed SDM tools, and to provide access to the content of their tools when not otherwise freely available (Supplementary Material 3). Finally, we conducted a search of the Ottawa Health Research Institute SDM tool inventory, 27 using the terms atrial fibrillation, anticoagulation, and stroke.
Study and SDM Tool Selection
Nine reviewers (V.T.R., O.J.P., N.E.S., T.B., F.B., A.D.T., P.W.O., F.B., and S.J.) working independently and in duplicate assessed each report for eligible SDM tools. To ensure quality and consistency, we performed multiple pilots and teaching rounds until we reached at least 90% of agreement before each phase. Disagreements resulting from full-text screening were resolved by a third author (J.P.B.). Three reviewers (V.T.R., O.J.P., and J.P.B.), working independently and in duplicate, assessed the eligibility of the SDM tools identified through the environmental scan.
Data Extraction
Five reviewers (V.T.R., M.U.-S., N.E.S., C.L.-S., and O.J.P.) extracted features of each SDM tool and each efficacy study. For risk-of-bias assessment, we used the Cochrane Collaboration’s tool 28 on randomized clinical trials and the Newcastle-Ottawa tool 29 on nonrandomized studies.
SDM Tool Features
Two reviewers (J.P.B. and V.T.R.) checked each SDM tool against the International Patient Decision Aids Standards instrument (IPDAS) version 4.0. 30 All conflicts were resolved by discussion. This 35-item tool (Supplementary Material 4) groups standards into 9 domains: information (8 items), outcome probabilities (6 items), values (2 items), decision guidance (2 items), development (6 items), evidence (6 items), disclosure (2 items), plain language (1 item), and evaluation (2 items).
The funding source had no role in the study conception, design, analysis, or interpretation.
Results
Figure 1 describes the results of our search. Table 1 and Supplementary Material 5 describe the 14 included SDM tools.31–55 All but 2 were in English; the mAF app 42 was in Chinese and MATCh AFib43,44 in Portuguese. When examining their intended use, 3 were patient decision aids, 5 were encounter tools, 4 had features of both, and 2 were not classifiable because of either lack of information or access to the tool itself. Most tools offered information about the available treatment options, mostly warfarin and DOACs, and the probabilities of specific outcomes. All the tools included tailorable risks of stroke and bleeding (mostly using CHA2DS2-VASc and HASBLED calculators) and compared different options of anticoagulation based on dosing, frequency of laboratory testing, drug side effects/interactions, and costs.
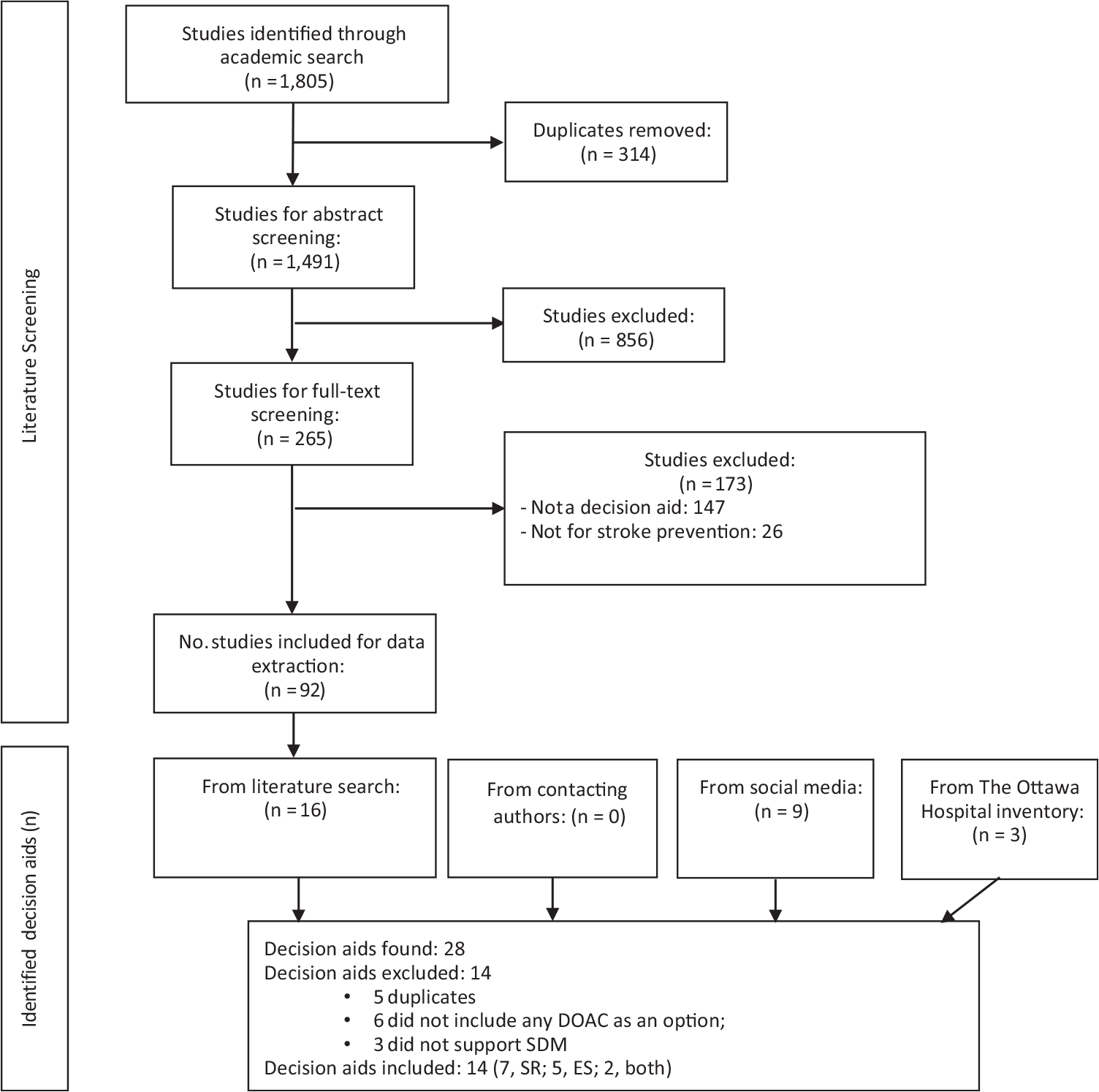
Eligibility of decision aids.
List and Overall Characteristics of Decision Aids

NA, not available.
All but these 2 decision aids are available in English: the content of mAF app and MATCh AFib are in Chinese and Portuguese, respectively.
SDM Tool Quality Assessment
Twelve decision aids met more than 50% of the IPDAS items (Figure 2). The top-rated tools were PtDA,51–53 Anticoagulation Choice,45–48 Don’t Wait to Anticoagulate, 38 and PDA, 55 which met >70% of all IPDAS items. PtDA was the only tool that assessed for readability. Only 2 tools, Anticoagulation Choice and Don’t Wait to Anticoagulate, reported field testing with patients and clinicians.
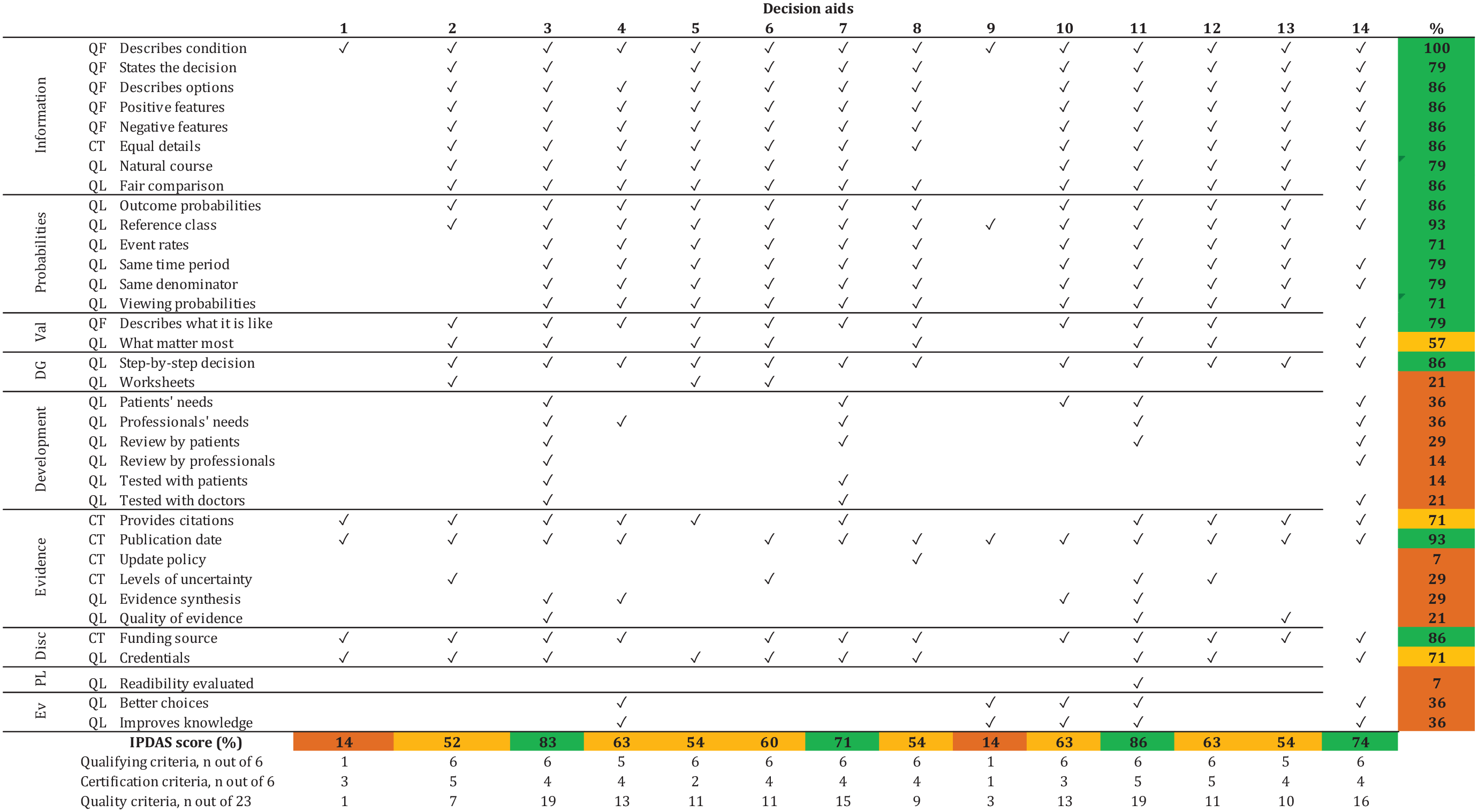
Quality of decision aids: IPDAS checklist.
SDM Tools’ Effectiveness and Risk-of-Bias Assessment
Six studies, including 2 randomized trials42,48 and 4 nonrandomized studies,34,43,53,55 at high risk of bias reported the effect of SDM tools on SDM outcomes (Table 2 and Supplementary Material 6).
Characteristics of Studies Evaluating Effectiveness

AFSDM, Atrial Fibrillation Shared Decision Making; NA, not available; PDA, patient decision aid.
The outcomes evaluated included knowledge, decisional conflict, quality of life, and medication adherence. These results are further described in Table 3. In summary, knowledge was evaluated and found significantly improved with the use of SDM tools in 5 studies. One of the trials 48 reported minimal change in knowledge probably due to nearly optimal levels at baseline. Five studies reported low decisional conflict immediately postintervention (9–19 out of 100 points).34,43,48,53,55 The only study that reported preintervention scores demonstrated a large effect associated with the intervention. 34 Quality of life was evaluated in only 1 randomized trial, which had substantial between-arm imbalance at baseline. 42 Two studies measured and reported statistically significant improvements in adherence to anticoagulants with the use of the SDM tool when compared to adherence at baseline and in the control group.34,42
Summary of Findings

AF, atrial fibrillation; AFKA, AF knowledge assessment; CI, confidence interval; DCS, decisional conflict score; NA, not avaialble; QoL, quality of life.
Discussion
We found 14 SDM tools for patients with AF considering stroke prevention strategies. Most were patient decision aids that offered information about the available treatment options, described probabilities of specific outcomes, included some type of value clarification activity, and included information about cost, required lab tests, dosing, potential changes in diet, and potential side effects; very few included information about other lifestyle changes and the burden of treatment (e.g., what it means to take a pill daily or what it takes to attend periodic clinic appointments). Patient decision aids improve patient knowledge and decisional conflict. Encounter SDM tools have not been evaluated. None of the 14 tools met all IPDAS certification criteria, 30 although most met 50% to 75% of them. Finally, in light of the CMS statement about the mandatory use of SDM when considering percutaneous LAAC, 25 we found only 1 SDM tool (CardioSmart) that included LAAC as an option.
One possible limitation of this study might have been not including government or nongovernmental organizations’ websites in our search strategy. We believe, however, that our search strategy ensured the inclusion of the SDM tools more available to clinicians and patients. In addition, the data on the development of the SDM tools were scarce. Most authors did not publish a article explaining the development process or included this information on their websites. Lack of reporting was considered as unmet IPDAS criteria by our group because we considered that the information of the development process should have been available to users in their published manuscripts, websites, or tools themselves. This decision could have led to lower IPDAS scores across all tools included in this analysis. The current study updates the database of existing SDM tools about anticoagulation for patients with AF. Compared to the review by O’Neill et al., 24 we found 5 additional tools, including the PtDA,51–53 which met the largest number of IPDAS standards. Our review also draws attention to the lack of participation of patients and clinicians in the content, design, and implementation of the tools and the lack of development of the tools within the context of their use. 56 If we expect tools to be applied within the clinical setting, they must be developed in a way that places the patient at the center of the development process. This can best be done through early and frequent testing of prototypes within actual clinical encounters of clinicians and AF patients facing the decision about whether and how to anticoagulate. Furthermore, for SDM tools to be ready for use and implementation, they should undergo rigorous efficacy testing. Yet, our review found that only a small subset of the tools underwent any type of testing. These studies, at high risk of bias, showed that the tools improve outcomes such as knowledge and decisional conflict, which may be useful to achieve SDM but at the same time might not be enough by themselves. None of studies directly tested whether the tools facilitated SDM. Some studies measured long-term, yet still indirect, consequences of SDM such as adherence and quality of life, but the results were inconclusive.
Conclusions
Several SDM tools are available, but their efficacy in promoting high-quality SDM is unknown. SDM tools should be rigorously evaluated in terms of their ability to support SDM and affect patient care.
Supplemental Material
sj-doc-1-mdm-10.1177_0272989X211005655 – Supplemental material for Shared Decision Making Tools for People Facing Stroke Prevention Strategies in Atrial Fibrillation: A Systematic Review and Environmental Scan
Supplemental material, sj-doc-1-mdm-10.1177_0272989X211005655 for Shared Decision Making Tools for People Facing Stroke Prevention Strategies in Atrial Fibrillation: A Systematic Review and Environmental Scan by Victor D. Torres Roldan, Sarah R. Brand-McCarthy, Oscar J. Ponce, Tereza Belluzzo, Meritxell Urtecho, Nataly R. Espinoza Suarez, Freddy J. K. Toloza, Anjali D. Thota, Paige W. Organick, Francisco Barrera, Carolina Liu-Sanchez, Soumya Jaladi, Larry Prokop, Elissa M. Ozanne, Angela Fagerlin, Ian G. Hargraves, Peter A. Noseworthy, Victor M. Montori and Juan P. Brito in Medical Decision Making
Supplemental Material
sj-docx-2-mdm-10.1177_0272989X211005655 – Supplemental material for Shared Decision Making Tools for People Facing Stroke Prevention Strategies in Atrial Fibrillation: A Systematic Review and Environmental Scan
Supplemental material, sj-docx-2-mdm-10.1177_0272989X211005655 for Shared Decision Making Tools for People Facing Stroke Prevention Strategies in Atrial Fibrillation: A Systematic Review and Environmental Scan by Victor D. Torres Roldan, Sarah R. Brand-McCarthy, Oscar J. Ponce, Tereza Belluzzo, Meritxell Urtecho, Nataly R. Espinoza Suarez, Freddy J. K. Toloza, Anjali D. Thota, Paige W. Organick, Francisco Barrera, Carolina Liu-Sanchez, Soumya Jaladi, Larry Prokop, Elissa M. Ozanne, Angela Fagerlin, Ian G. Hargraves, Peter A. Noseworthy, Victor M. Montori and Juan P. Brito in Medical Decision Making
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The investigators Juan P. Brito, Ian G. Hargraves, Victor M. Montori, and Peter A. Noseworthy participated in the development of the Anticoagulation Choice, which is one of the shared decision making tools included in this review. The rest of the authors declare no conflicts of interest regarding this work.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided entirely by a grant from American Heart Association and the Patient-Centered Outcomes Research Institute. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
