Abstract
Background
Decades of effectiveness research has established the benefits of using patient decision aids (PtDAs), yet broad clinical implementation has not yet occurred. Evidence to date is mainly derived from highly controlled settings; if clinicians and health care organizations are expected to embed PtDAs as a means to support person-centered care, we need to better understand what this might look like outside of a research setting.
Aim
This review was conducted in response to the IPDAS Collaboration’s evidence update process, which informs their published standards for PtDA quality and effectiveness. The aim was to develop context-specific program theories that explain why and how PtDAs are successfully implemented in routine healthcare settings.
Methods
Rapid realist review methodology was used to identify articles that could contribute to theory development. We engaged key experts and stakeholders to identify key sources; this was supplemented by electronic database (Medline and CINAHL), gray literature, and forward/backward search strategies. Initial theories were refined to develop realist context-mechanism-outcome configurations, and these were mapped to the Consolidated Framework for Implementation Research.
Results
We developed 8 refined theories, using data from 23 implementation studies (29 articles), to describe the mechanisms by which PtDAs become successfully implemented into routine clinical settings. Recommended implementation strategies derived from the program theory include 1) co-production of PtDA content and processes (or local adaptation), 2) training the entire team, 3) preparing and prompting patients to engage, 4) senior-level buy-in, and 5) measuring to improve.
Conclusions
We recommend key strategies that organizations and individuals intending to embed PtDAs routinely can use as a practical guide. Further work is needed to understand the importance of context in the success of different implementation studies.
Keywords
Decades of effectiveness research has firmly established the patient-level benefits of using patient decision aids (PtDAs).1,2 More work is needed to assess the true impact of routine PtDA implementation on health care users and providers, but the promising benefits and lack of harms identified by controlled studies has led to strong international policy support for more person-centered health care systems underpinned, in part, by increasing implementation of PtDAs.3–9 However, broad clinical implementation has not yet occurred, and there is a notable intention-behavior gap when PtDAs are used outside experimental studies in routine clinical settings. 10
PtDAs support patients’ participation in shared decision making (SDM) with health care professionals by making options explicit, providing evidence-based information about the associated benefits/harms, and helping patients to consider what matters most to them in relation to the possible outcomes. 1 Formats vary (e.g., paper based, DVD, website), and distribution methods can be tailored to the condition and setting, with PtDAs being delivered either as part of the clinical pathway (e.g., made available to patients before or during consultation) or via direct-to-consumer approaches (e.g., for population-level cancer screening programs, access provided via screening invitations). Various studies have examined and described key factors that influence successful implementation of SDM more broadly.11–15 Interventions studied include PtDAs and other approaches that encourage SDM behaviors, including patient activation materials and clinician SDM skills training.
The International Patient Decision Aid Standards (IPDAS) Collaboration review published in 2013 16 explored the success levels of different implementation strategies and included findings from controlled trial settings. Key barriers identified in the 2013 review included health care professionals’ (HCPs) attitudes toward SDM, lack of understanding in how to use PtDAs and undertake SDM, HCPs’ lack of trust in PtDA content, lack of clarity among HCPs regarding the purpose of PtDAs in relation to other information available for patients, HCPs believing that patients do not want decisional responsibility, competing clinical demands, and the time it would take to distribute and use the PtDA. Key facilitators included system-level approaches (e.g., systematic identification of patients ahead of appointments via electronic health records and distribution methods that did not rely on HCPs to initiate access), SDM and/or PtDA training and skills development, and dedicated clinical leadership (e.g., clinical champion).
Despite their benefits and various policies mandating their dissemination and use,3–9 widespread adoption of PtDAs has not occurred, and significant gaps exist in understanding factors contributing to adoption, implementation, and sustainability of these interventions in routine clinical settings. Strong foundational research has examined the implementation of SDM in health care, typically through large-scale demonstration projects (e.g., Informed Medical Decisions Foundation/Healthwise),17,18 and excellent examples of local adoption also exist (e.g., Dartmouth Hitchcok Medical Center, Lebanon, NH, USA). 19 The literature listing barriers and facilitators of PtDA dissemination and implementation, as perceived by HCPs and patients, is also well established. 16 However, despite the valuable learning, much of it is derived from highly controlled settings, which might not be representative of day-to-day processes and resources (human or financial) in routine clinical settings. Further, although lists of barriers and facilitators are useful markers to guide efforts to embed PtDAs, they provide less insight into why and how these factors influence implementation and can overlook the relations between different factors. 20 PtDAs are not “magic bullets” that will always deliver the intended benefits to patients; their usefulness will ultimately depend on context and implementation. 21 If clinical teams and organizations are being encouraged or mandated (e.g., clinical guidelines) by national health agencies to embed PtDA as a means to support person-centered care, we need to better understand what this might look like outside of a research setting, which contexts are likely to be more successful, and which might face additional challenges.
This current review was conducted in response to the IPDAS Collaboration’s evidence update process, which informs their published standards for PtDA quality and effectiveness.22–24 It updates the theory and evidence provided in the 2013 review16,25 through the sole inclusion of real-world data, exclusion of data from highly controlled settings, an understanding of the contexts that enable or hinder implementation, and the mechanisms (i.e., changes in people’s reasonings and actions) through which implementation is achieved. The main aim of this current review is to develop context-specific program theories that explain why and how PtDAs are successfully implemented in routine health care settings, providing a framework that will be useful to various stakeholders committed to embedding these tools routinely.
Method
We used rapid realist review (RRR) methodology 26 and the RAMESES publication standards for realist reviews. 27 RRR methodology moves beyond traditional reviews by allowing researchers to answer questions about why interventions in complex social contexts, such as routine health care, work or do not work. 28 We chose this method as it allowed us to look beyond the overall success of a PtDA intervention to generate explanations about what works for whom, in what contexts, to what extent, and, most importantly, how and why? 28
The resulting knowledge synthesis highlighted possible interventions (I) that could be implemented in a specific context (C) that in turn interact with various mechanisms (M) and produce outcomes (O) of interest, 28 in this case, implementation of PtDAs in routine health care settings (see Box 1 for definitions of specific terminology). Two reviewers (N.J.-W. and T.v.d.W.) conducted a scoping exercise of existing literature examining barriers and facilitators to implementing PtDAs and SDM9,11–13,16,25,29,30 to agree on the review questions and scope and to generate initial theories. The a priori proposal was reviewed and approved by the IPDAS Steering Committee and registered with PROSPERO (CRD42019153334).
Glossary of Key Terms and Abbreviations

We followed the key stages of an RRR: identifying scope/research questions, identifying literature for inclusion, quality appraisal, data extraction, and data synthesis. 26 Our specific foci were to engage key experts and stakeholders to streamline the review process, produce useful results for those planning to implement PtDAs, and to create a set of recommendations for the IPDAS Collaboration’s updated evidence document for PtDA implementation. We convened a review team (named co-authors, led by N.J.-W. and T.v.d.W.) identified via the IPDAS Collaboration’s call for evidence update chapter authors to support the review process in the areas of literature identification, data extraction, and theory development. Typically, RRRs involve consultation with a broader expert panel to identify literature and corroborate theory development; however, as the review team consisted of a number of key international experts in the field of PtDA development, evaluation, and implementation, representing a range of disciplines and backgrounds, the review team also fulfilled this role.
Identifying and Selecting Literature for Inclusion
The review team identified an initial list of potential articles that could contribute to theory development and a list of known organizations and individuals involved in the implementation of PtDAs. The lead authors (N.J.-W./T.v.d.W.) screened articles to determine if they could likely contribute to understanding what facilitates and/or hinders PtDA implementation in routine clinical settings. Articles were included if the study reported implementation of a PtDA (as defined by the IPDAS Collaboration) 1 in a routine health care setting (defined as daily situations without significant additional resources, in which clinicians and/or providers had been encouraged to integrate the PtDA into usual care routines) and if PtDA and dissemination/implementation strategies were described. Articles were excluded if the study used an intervention not classified as a PtDA 1 (e.g., education resource, information leaflet), if the PtDA supported decisions about health insurance/provider options, or if the PtDA was implemented in highly controlled settings, such as randomized controlled trials or process evaluations conducted as a sibling study assessing implementation in a controlled research setting. Although secondary analysis of experimental studies has its relevance, we chose to exclude sibling studies associated with experimental studies that focus on measuring the efficacy or effectiveness of PtDAs. These studies likely bypassed routine clinical procedures to enlarge the effect of the PtDA, thus being less representative of everyday clinical settings, and would have limited bearing on our program theory. Studies exploring routine implementation of SDM outside controlled settings are relatively new, and our aim was to build on the 2013 review; thus, articles were restricted to a 10-y period (2009–2019). There were no restrictions regarding PtDA format (e.g., web based, paper based), type of decision, healthcare settings, or population/participants.
Using the initial set of articles, a combination of free-text and MeSH headings related to “decision aids,”“shared decision making,” and “implementation” were used to develop a Medline search strategy, which was adapted for use in CINAHL (see Supplementary File 1 for the Medline search strategy). Relevant websites (e.g., databases of funders who support PtDA implementation programs), policy documents, and known individuals and organizations were consulted to determine whether any unpublished works relating to the review questions were available. Citations were exported to EndNote; titles and abstracts of all papers identified via electronic searches were screened (by T.v.d.W.) to determine if they could answer the review question. Potentially relevant articles were obtained, and full texts were screened (N.J.-W. and T.v.d.W.) against our inclusion/exclusion criteria (noted above). Reference lists of included studies were consulted for forward and backward searching, and a clear audit trail of all data sources was maintained.
Data Extraction
A data extraction team was convened from members of the broader review team, and a data extraction template was developed, piloted, and streamlined to increase emphasis on context-mechanism-outcome (CMO) configurations. In the final version, qualitative, quantitative, and contextual data that could answer our review questions were extracted under the following broad categories: study/participant characteristics, PtDA characteristics, dissemination and implementation, implementation evaluation data (e.g., reach, dose, feasibility), and data supporting emerging theories about what works, how, and in what circumstances, for implementation of PtDAs (if-then statements). N.J.-W. coordinated extractions completed by the data extraction team members, checked the accuracy and consistency of data extracted, and consulted with individuals when necessary to clarify information or resolve discrepancies.
Data Synthesis
Explanatory data in the results sections of included studies relating to “what works in implementing PtDAs?” were initially extracted as “if-then” statements that described links between elements of contexts, mechanisms, and outcomes. As the synthesis progressed, comparable if-then statements were grouped by N.J.-W., while ensuring linkage to the original data and source of the individual if-then statements. We applied Pawson’s reasoning processes 31 to generate refined CMO configurations based on the grouped if-then statements (see Supplementary File 2 for example process of theory development). Realist reviewers typically make use of existing theories to make sense of the evidence generated during their review. We chose the Consolidated Framework for Implementation Research (CFIR) 32 to help us interpret the findings emerging from the data, as it is designed to guide systematic assessment of multilevel implementation contexts to identify factors that might influence intervention implementation and effectiveness. It is composed of 5 major domains, each made up of several constructs (see Box 2 for domain descriptions). The initial draft of CMOs was presented back to the review team, who were asked to assess validity, relevance to the research questions, and importance of the inferences made. Feedback from the review team was used to refine the program theory (3 iterations), exclude theories viewed as less important and relevant, and inform further data searches for elements that were perceived as missing, based on prior knowledge and experience.
Definitions of the 5 Major Domains of the Consolidated Framework for Implementation Research 32

Results
A total of 29 articles from 23 distinct studies contributed data to the developing theories. Figure 1 outlines the review process, including data sources and exclusions, and Table 1 presents the key characteristics of included studies. Most studies were from the United States (
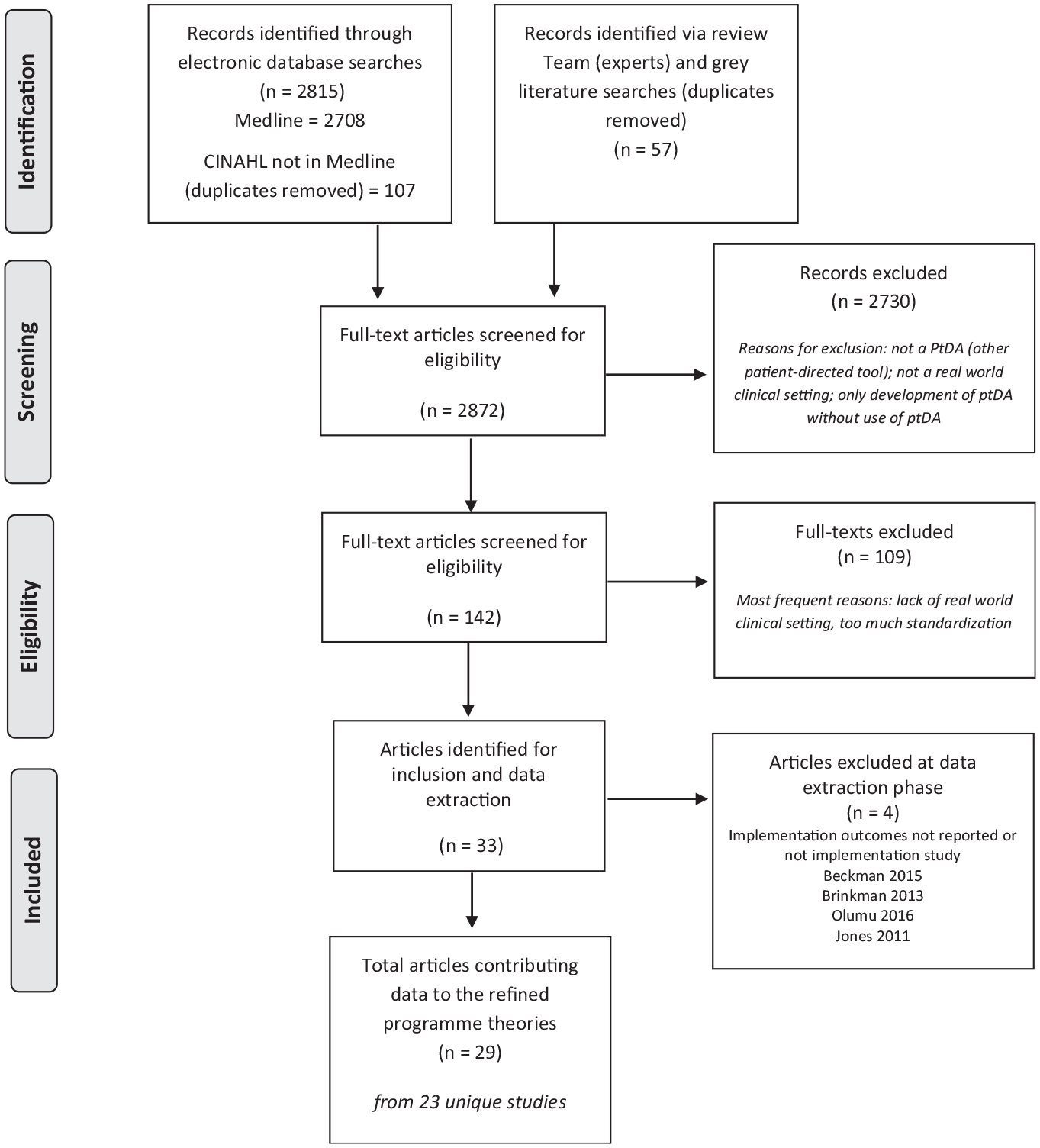
Data searches and sources of included articles.
Characteristics of Included Studies (
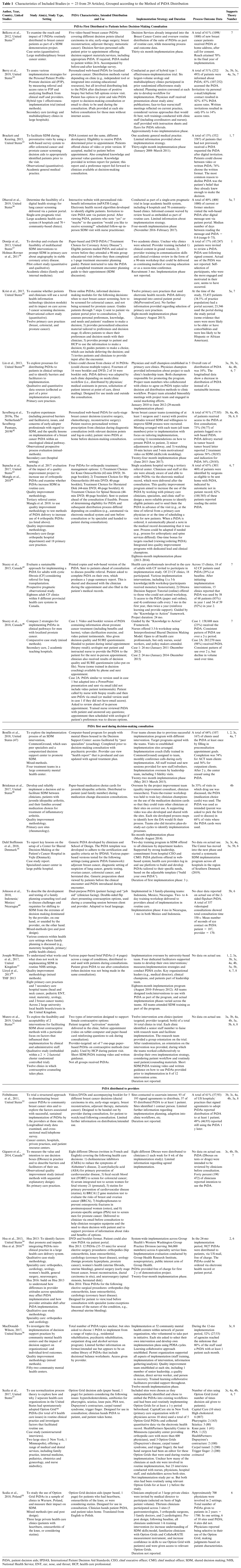
PtDA, patient decision aids; IPDAS, International Patient Decision Aid Standards; CEO, chief executive officer; CMO, chief medical officer; SDM, shared decision making; NHS, National Health Service; ENT, ear, nose, and throat; HCP, health care professional.
Program Theories: What Works in Implementing PtDAs into Routine Clinical Settings?
A total of 124 explanatory if-then statements were extracted from the included articles. Using the CFIR 32 to help understand factors that might influence intervention implementation and effectiveness, these statements were refined into 8 program theories (CMO configurations). The program theories are described below, organized under relevant domains of the CFIR, 32 and are summarized in Table 2 with supporting data (“I” is used in the results to denote features of the implementation strategy, e.g., skills training session, automated electronic delivery of PtDA). None of our theories mapped to domain II of the CFIR. Domains are not mutually exclusive, and some CMO configurations could map to more than 1 of the CFIR domains; however, for brevity and clarity, we mapped the 8 program theories to the most relevant domain. Because of the limited number of included studies, CMOs have been presented generically, with limited contextual reference to specific diseases or decisions, with the exception of theory 2, which is specific to crisis-driven and life-threatening situations.
Implementing PtDAs in Routine Clinical Settings Program Theories: Context-Mechanism-Outcome Configurations Identified from 29 Articles (23 Studies)

PtDA, patient decision aids; HCP, health care professional; SDM, shared decision making; IT, information technology.
I. Intervention Characteristics
Theory 1: PtDA complexity: simple tools for busy settings
Five articles contribute data to the theory that less complex tools are more likely to be integrated into routine care.35,37,47,50,59 When PtDAs were perceived as more complex by the clinical team, especially those PtDAs that required technical knowledge and support, and required an increased number of PtDA-related tasks and personnel time, the teams felt that they competed with existing practice, were too resource intensive, and were more difficult to embed.35,37,47 Some HCPs reported that the shorter and less complex tools (e.g., brief in-consultation paper-based tools) were preferable as they fit better with existing practices and required limited additional resources.35,59,50
III: Inner Setting
Theory 2: Crisis-driven and life-threatening situations—urgent care needs prioritized over decision support needs
Five articles support the theory that PtDAs are less likely to be embedded by HCPs in teams that are typically crisis driven or deal with life-threatening issues.47,54,55,59,50 When exploring implementation of a PtDA within a community mental health setting, Bonfils et al. 47 found that staff would often need to prioritize immediate patient needs over PtDA distribution; for example, “we’re a crisis-driven clinic and you could use this [resource], and you could use that [resource], but then they’re like ‘well they don’t have a house,’ so some of that stuff gets in the way.” Life-threatening situations 54 also present challenging contexts to embed PtDAs, where HCPs tend to prioritize supporting the immediate health care needs, and sometimes the more emotional needs, of the patient.
Theory 3: Bringing the whole team on board: establishing a common goal, senior-level buy-in, and distributing PtDA tasks appropriately
Six articles provide data for this theory.14,34,40,46,51,59 Various studies reported that PtDA integration was more successful when all members of the clinical team had been introduced to the PtDA and not only the HCPs who would be using the tool. When administrative staff understood the purpose of the PtDA and how it fit into the patient pathway, they were more supportive of its use and motivated to support the distribution processes as part of their administrative role. Joseph-Williams et al. 13 reported the “shared understanding” that emerged when all team members were involved and how reception staff played a key role in introducing the concept of choice at the very start of the patient journey as well as in distributing materials. Other studies also found that administrative staff played a critical role in integrating the PtDA into workflows40,51; they were responsible for 73% of PtDA distribution in the study by Lin et al. 40 Berry et al. 34 further highlighted the importance of “coherence” about purpose and use across the entire team (clinical and administrative); when the administrative staff members knew very little about why a PtDA was important or being used, this acted as a barrier to implementation.
Eight articles support the theory that distributing PtDA tasks among a multidisciplinary team (clinical and nonclinical) is more likely to lead to the PtDA being distributed and used as planned.34,40,41,44,46,51,52,59 Lin et al. 40 reported how a “team-based practice model,” in which clinic staff were empowered to distribute PtDAs, was more successful than a model that relied on physicians alone; however, they also noted that this model can only support, not substitute, HCP involvement in patient engagement. Whole-team involvement, particularly senior clinical staff, led to perceptions such as “I’m not in this by myself” and “this won’t be seen as my ‘little’ project,” 13 which motivated individual team members to continue use of PtDAs. Conversely, Fiebleman et al. 52 showed that when service physicians were not supportive of the PtDA, the remaining staff were less likely to use it. Omission of certain team members from the process (e.g., nurses not involved after use of PtDA) 41 or inappropriate allocation of PtDA tasks to the wrong team member (e.g., reliance on physicians to order PtDAs) 44 can lead to reduced fidelity in the way the PtDA is used and the subsequent SDM discussion and reduced distribution.
Twelve articles support this theory.13,14,34,40,46,47,50,52,53,56,57,59 Leadership from senior clinicians and managers plays an important role in determining whether teams use and continue to use the PtDA. Several studies found that a clinical lead, or “champion” played a significant role in making training available, prioritizing and keeping SDM and the use of the PtDA high on the agenda, conveying seriousness of intent, and ensuring evaluation data were being fed back to the team13,14,46,56—all which results in greater motivation and an improved skill set among the team, making it more likely that the PtDA use will be sustained. As one member of the obstetrics team said in the study by Joseph-Williams et al 13 : “once you use the big names, the well-respected consultants, people sit up and listen . . . that’s needed.” Scalia et al. 58 reported how a champion orthopedic surgeon influenced colleagues by playing a significant role in PtDA development and demonstrating the benefit of using the tool. On the other hand, Berry et al. 34 found that even when a designated lead was appointed, the absence of a clinical lead who is physically present in the clinic and seeing patients acted as a barrier to PtDA use.
IV: Characteristics of Individuals
Theory 4: Activating and supporting HCPs to deliver PtDAs: HCPs aware, trained, and motivated to change practice
Ten articles contributed data to support this theory.14,34,40,41,47,51,50,52,57,59 Implementation is unlikely to occur when teams are not familiar with PtDAs or lack awareness of the PtDA’s purpose and intended use. This was an important barrier to routine implementation. For example, one staff member in the study by Bonfils et al. 47 of a mental health PtDA noted, “I think it’s underutilized because people don’t understand the richness of it . . . I don’t think they realize how much is on there [the PtDA].” When team members lack knowledge on why or how the PtDA should be used, they do not understand the benefits for patients or the role it plays in the decision-making process, which results in the PtDA being underused or misused.40,41,47 Conversely, when they are clear about the purpose and intended use, PtDA adoption is higher.50–52,59
Twelve articles contributed to this theory.13,40,41,44,45,48,50–53,57,59 PtDAs are sometimes implemented in settings where the HCPs lack knowledge of SDM skills and therefore do not understand how SDM differs from their current practice and thus the additional benefit of using PtDAs (over other educational resources). For instance, Joseph-Williams et al. 13 found that when asked about SDM approaches, many HCPs report “we do it already.” On the other hand, if HCPs lack knowledge of SDM skills, they often lack confidence in their SDM/PtDA delivery skills, thus holding back from enacting the skills. SDM training workshops that incorporate methods for practicing skills (e.g., role-play scenarios) can help HCPs better understand how SDM builds on existing good health care communication practices, that SDM is an approach rather than another communication model, and enhance SDM skills. Lloyd et al. 50 reported a significant change in attitudes among HCPs following the workshops; for example, as one nurse stated, “Initially when we started, like many of us, I thought ‘we do that anyway.’ I think the biggest difference is, well actually, we didn’t do it well.” These training opportunities encourage HCPs to reflect on their current practice and understand and agree on the role PtDAs can play in that, making it more likely that the PtDA will be used as intended.
Theory 5: Preparing and encouraging patients to use PtDAs—explicit invitations from clinical team to use PtDA before and during decision-making consultations
Eight articles provide support for this theory.13,14,34,36,39,51,52,59 Many patients are unfamiliar with PtDAs and have no experience of using them to support their health care decisions. When explicit invitations to engage with the PtDA are sent to patients
Six articles support the theory that explicit invitations from HCPs for the patient to engage with the PtDA during decision-making consultations is important to ensuring the PtDA is used in the way intended (e.g., helping patients to understand their options and encouraging them to share their preferences with the HCP, ask questions, and engage in the decision-making process).13,14,41,49,51,53 Patients in the study by Joseph-Williams et al. 13 reported that this explicit encouragement from HCPs during consultations provided “permission” for them to share their preferences and become involved, and the handover of the PtDA during the consultation meant it was used in the way intended: to guide questions for the clinician and to prompt them to share their preferences. Conversely, when opportunities to share preferences were not provided after receipt of a PtDA, it was not self-evident to patients that they could and should express their preferences. 41
V: Process
Theory 6: Collaborative PtDA development and implementation planning: early and meaningful involvement of the clinical team and providers
Thirteen articles support this theory, which indicates that early involvement of those affected by change or the intended knowledge users was important in integrating a new PtDA into routine care.13,14,41,43,49,51,54,55,57–60 PtDAs were distributed in teams where HCPs had preestablished ways of communicating treatment and management options to patients, whether that be via verbal communication or existing educational resources, which HCPs often believed to be effective. When the team/providers collaboratively contributed to plans regarding PtDA focus, content, design, and proposed use from inception, this led to greater ownership and buy-in to the new approach,14,50,59 sometimes helping HCPs to understand how their existing approaches might not fully support SDM50,59; greater trust and legitimacy in the content of the tool50,59; and development of a tool that best fit within their setting, addressing concerns head on.49,50,51,55,59 The opportunity to adapt care pathways meant that the PtDA could fit better with ongoing and simultaneous processes. For example, one breast cancer clinic felt that their current “one-stop-shop,” whereby a patient received diagnosis and was asked to make a decision, would not support PtDA use. Instead, they adapted the pathway so that the patient could take the PtDA home following diagnosis and then discuss at a follow-up appointment. 59 Conversely, lack of involvement of those affected by change resulted in less positive attitudes. 54 These mechanisms were important in helping teams to adapt existing practices to integrate PtDAs into routine care.
Theory 7: Earlier distribution of PtDAs: systematizing delivery of PtDAs to eligible patients before decision-making consultations
Fourteen articles contributed data to this theory.33–36,39,40,43–47,53,57,59 PtDAs are typically being implemented in settings characterized by limited staff resources, multiple competing demands and priorities, short appointment times, and, sometimes, short time frames within which to deliver the PtDA, so that it is feasible for the patient to use as intended and relevant. When teams were able to embed processes that could preidentify eligible patients and standardize (ideally automate) the delivery of the PtDA before the decision-making consultations, this resulted in improved reach of the PtDA to the right patient at the right time.34,39 Processes that support and standardize the 2-way delivery of information (i.e., returning patient preferences/questions to the HCP prior to the decision-making consultation) act as a prompt for the HCP to use the PtDA with the patient, meaning HCPs are more likely to integrate it into their consultation. Online delivery of PtDAs prior to the consultation also helps to overcome time limitations (i.e., if there is limited time available between identifying eligible patients and the decision-making consultation taking place).33,46 HCPs also perceived such processes would alleviate concerns regarding the time it would take to deliver the PtDA (e.g., “if you really want to use these kinds of Dboxes, and you want to make it work, I think you could organize it so you would have a pre-visit”). 53
Theory 8: Linking with “learning health care systems”: using measurement to show how PtDA outcomes link with and improve key organizational priorities
Seven articles contribute data to this theory.13,41,50,51,52,56,59 Some studies were implementing PtDAs in contexts in which organizational priorities were aligned with principles of SDM and PtDA outcomes, and a learning health care system existed, whereby ‘science, informatics, incentives and culture are aligned for continuous improvement and innovation, with best practices seamlessly embedded in the delivery process and new knowledge captured as an integral by-product of the delivery experience.” 18 When such a context exists, integration of routinely collected measures alongside the PtDA encouraged PtDA use and made it more likely to be sustained in routine care. Clinical team members frequently wanted to know whether SDM/PtDA use aligned with existing organizational priorities; as a head and neck consultant commented in the study by Lloyd et al., 50 “if you haven’t got Board buy-in, if you haven’t got support from that level of management, then everything is an uphill struggle.” By implementing measures alongside PtDAs (tying in with existing routinely collected data or developing new measurements), the teams felt that an important part of PtDA use was to drive change and improvements, which motivated them to sustain implementation.13,41,50,56,59 The fact that these measures aligned with key organizational priorities showed HCPs that SDM was important, and they started to view it as something the organization does, rather than as another initiative being imposed on them and competing with other demands. 13
Discussion
Principal Findings
We developed 8 refined theories using data from 23 implementation studies to describe the mechanisms by which PtDAs become successfully implemented into routine clinical settings and the implementation strategies and contexts that enable these mechanisms. The combined program theory shows that PtDAs are more successfully embedded in health care contexts that make it clear that SDM is an organizational priority, take ownership of PtDA implementation by appointing accountable leadership, coproduce PtDA implementation strategies with end-users, engage and inform the entire team (clinical and administrative) about the PtDAs’ purpose and intended use, recognize the significance every team member plays in PtDA implementation and distributes tasks appropriately, provides adequate SDM skills training for those delivering PtDAs, uses simpler tools that integrate into clinic workflows, and prepares and prompts patients to engage in the SDM process so they understand their role in the process and feel comfortable with having open and honest discussions. Using this program theory as a basis, we recommend key strategies that organizations and individuals intending to embed PtDAs routinely can use as a practical guide (see Table 3).
Strategies for Routine PtDA Implementation, Derived from Our Program Theory of Successful PtDA Implementation in Routine Health Care Settings

PtDA, patient decision aid; HCP, health care professional.
The fifth key strategy, “measuring to improve,” will not be sufficient if it focuses solely on measuring the uptake of PtDAs by patients, as this may result in tokenistic use of PtDAs and a focus on distribution rather than actual and meaningful engagement with the tool. Only a few benchmarks for PtDA uptake have been reported in the included studies (e.g., “80% of the staff uses a PtDA with at least 1 individual each month”), and so we lack insight into reasonable benchmarks for PtDA use. Most studies reported actual PtDA distribution (see Table 1); very few focused on how many patients actually used the PtDA. An additional challenge of focusing on uptake is that it fails to take into account legitimate reasons for the PtDA not being distributed. For example, it might be that the HCP has already informed the patient at an earlier consultation, or they have sought an alternative source of information given the patient’s low health literacy. Likewise, patients might also have several good reasons for not using the PtDA that has been offered to them.
Most of the contributing factors we identified relate to the “inner setting” (e.g., how the organization and the team view SDM and PtDAs, appropriate division of work, dedicated leadership), “characteristics of individuals” (e.g., do the team have the necessary skills and self-efficacy to use PtDAs, and are patients aware of their role and feel comfortable being involved?), and “process” CFIR domains (e.g., collaborative development and planning, earlier distribution of PtDA via automated systems). 32 Other than complexity of PtDAs (theory 1), few CMOs mapped to the “intervention characteristics” domain. This might indicate that PtDAs as interventions are relatively well accepted in routine clinical settings, and thus, challenges with implementation are less likely to do with the tool itself but more the processes we use to embed the tools (e.g., timing of delivery, collaborative agreement on how they are introduced) and who they are used by (e.g., do they have the skills to introduce and use the PtDA effectively?). No CMOs mapped to the “outer setting” domain, or the external context within which the organization resides. This most likely reflects the recent emergence of such guidelines and processes and awareness of these, rather than them not playing a significant role in successful PtDA implementation. Indeed, elsewhere, the emergence of national governance and guidelines has acted as a key driver for SDM implementation (e.g., several NICE guidelines in the United Kingdom now recommend SDM supported by PtDAs). 65 However, another core construct of the “outer setting” domain, “patient needs and resources,” has not been adequately addressed by this review. This signifies that the focus should move from describing “needs assessments” during the development of PtDA, and we should seek to understand patients’ needs and resources in relation to implementation (e.g., delivery modes, readiness for implementation, time, etc.).
Comparison with Other Literature
This review builds on and aligns with a body of work examining SDM implementation.6,30,66–69 More specifically, several contextual factors identified in this review align with factors described in the 2013 review. 16 Adequately trained clinicians with the skills and confidence to deliver PtDAs proved to be significant facilitators in both reviews (theory 4), as did processes of systematizing earlier delivery of PtDAs to patients before health care consultations, when feasible (theory 7). The previous review also reported how distrust in content and lack of clarity of the PtDA’s purpose in relation to other sources of information acted as barriers. This review builds on these ideas by showing how contexts that use a collaborative and co-produced approach to PtDA development and implementation can overcome these barriers; by increasing a sense of ownership and buy-in, legitimizing content, and ensuring the PtDA content and delivery can fit with current practice (theory 6). Competing demands, time pressures, and poor teamwork also featured as barriers in the 2013 review. 16 We build on these themes by showing how a whole-team approach, with appropriate distribution of PtDA tasks between clinical and administrative staff (theory 3), can help to overcome the dissociation of ownership of PtDA tasks and result in more successful integration of the tool. Further, we show how contexts with adequate and ongoing leadership for PtDA implementation help to overcome the tendency for busy clinicians to relegate the priority of PtDA distribution, by demonstrating how PtDA outcomes align with organizational priorities, monitoring progress and improvement, and providing motivation and the skill set needed to use the PtDA (theory 3c).
Significant contributions of the current review not covered previously include the important role of both preparing (either explicitly via earlier distribution of PtDA or implicitly through organizational messaging that patient involvement is valued) and prompting patients to engage with PtDAs (theory 5). This moves beyond the preparedness and engagement of clinicians covered previously 16 and focuses more on the engagement of the main end user, the patient. The current review also highlights the importance of simpler PtDAs for busy and time-limited settings, likely demonstrating further support for brief in-consultation tools (theory 1), the challenge of balancing PtDA tasks with more immediate patient needs in crisis-driven and life-threatening situations (theory 2), and the importance of learning health care system contexts and linking PtDA outcomes with organizational priorities, thus improving integration (theory 8).
Waldron et al. 70 have recently published a program theory of SDM. Although the focus of our realist review is on the implementation of one specific intervention (i.e., PtDAs) that can support the SDM process, there are some consistent observations across the 2 program theories. Notably, both reviews demonstrate that contexts that provide system-level support such as training, senior-level leadership, and organizational support are facilitative (theories 3c, 4, 8). We also found that self-efficacy and recognition of the decision were important mechanisms in play when contexts that provided adequate training (theory 4) and preparation and patient engagement opportunities (theory 5) existed. The perception of time mechanism identified by Waldron et al. 70 also featured as a prominent mechanism in our theories and can be alleviated somewhat by whole-team approaches (theory 3) and systematization of PtDA delivery before clinical consultations (theory 7).
Strengths and Limitations
The RRR allowed us to understand the mechanisms by which PtDAs become routinely implemented into routine clinical settings and to draw on the expertise of a large international and multidisciplinary team of experts. Inclusivity of this review approach does remain an issue because of the rapid nature, 26 but supplementary electronic searches did not identify significant additional papers for consideration, and the large author group was in agreement that no key studies have been missed. Given that exclusive implementation studies in this field are still relatively sparse, our exclusion of highly controlled trials and associated sibling studies might result in data that could contribute to our theories being missed. However, our inclusion of real-world implementation studies should make these findings more relevant to policy makers, organizations, and HCPs looking to implement PtDA in routine settings. Despite this, we acknowledge that these implementation studies typically involved willing volunteers, who were selected, for example, for their commitment to embed person-centered care approaches or prior success in a trial setting. As such, although the core strategies identified are likely to be valid in new implementation settings, their level of success might vary depending on preexisting attitudes and behaviors. Most of the included studies were also from high-income countries with well-established and well-resourced health care services, and so routine implementation in lower- or middle-income countries might look different. We were limited by the number of overall studies and the number of different contexts that examined PtDA implementation, and this impeded our ability to make more specific recommendations of which strategies worked best in which settings (e.g., in line with theory 3). We have limited or no information about what implementation looks like in emergency, pediatric, or end-of-life settings, for example, or for surrogate decision-making processes. We also have limited understanding of supporting patients with low health literacy and a general lack of inclusivity in PtDA approaches. As more implementation studies are conducted, researchers should pay attention to report the contexts and mechanism supportive of implementation; it would then be prudent to assess what works and does not work in those settings, to broaden our understanding of appropriate strategies that can be tailored according to specific contexts.
Practice Implications
Globally, health organizations are developing policies that encourage or mandate person-centered health care approaches when patients are faced with decisions about their health and care. Despite these efforts, limited guidance exists regarding the types of strategies that are likely to be the most effective in routine health care settings. Through this program theory development, we have been able to recommend key strategies that can support successful integration of PtDAs into routine clinical settings (Table 3). Building on existing work, 16 this framework emphasizes the importance of training for entire teams, of better preparing patients to engage with SDM and PtDAs, and of linking PtDA outcomes with key organizational priorities and data collection (e.g., PROMS and PREMS). The strategies chosen by PtDA implementers should still ultimately depend on context and the key barriers anticipated in each setting; for example, the simplicity of PtDA design and delivery method would be more significant in settings with very limited time/human resources or limited flexibility in pathway design. This review was also not able to explore the added benefit of the more complex and harder-to-achieve strategies (e.g., strategy 1, co-production of interventions) over relatively more straightforward strategies (e.g., strategy 2, skills training for teams). Unit we have further outcome data reported by implementation studies, considerations of the feasibility and effort versus expected benefit are still needed when choosing strategies. We also fully acknowledge that PtDAs are only one means to improve SDM and that true SDM implementation would require a multifaceted user-centered plan, with interventions/approaches targeting attitudes, knowledge, skills, and self-efficacy of all end users, while also addressing key meso- and macro-level barriers.13,29 However, the proposed strategies may inform an initial framework and then be supplemented by more specific strategies depending on context and to also address the broader SDM goals.
Conclusions
The goal for this review was to identify why and how PtDAs become successfully implemented in routine health care settings. This study presents a program theory derived from implementation studies across a range of routine health care settings and recommended strategies that could be used as a practical guide by organizations and individuals attempting to embed PtDAs routinely. Further work is needed to understand the importance of context in the success of different implementation studies, as these studies become available.
Supplemental Material
sj-doc-1-mdm-10.1177_0272989X20978208 – Supplemental material for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration
Supplemental material, sj-doc-1-mdm-10.1177_0272989X20978208 for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration by Natalie Joseph-Williams, Purva Abhyankar, Laura Boland, Paulina Bravo, Alison T. Brenner, Suzanne Brodney, Angela Coulter, Anik Giguere, Aubri Hoffman, Mirjam Körner, Aisha Langford, France Légaré, Daniel Matlock, Nora Moumjid, Sarah Munro, Karina Dahl Steffensen, Christine Stirling and Trudy van der Weijden in Medical Decision Making
Supplemental Material
sj-doc-2-mdm-10.1177_0272989X20978208 – Supplemental material for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration
Supplemental material, sj-doc-2-mdm-10.1177_0272989X20978208 for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration by Natalie Joseph-Williams, Purva Abhyankar, Laura Boland, Paulina Bravo, Alison T. Brenner, Suzanne Brodney, Angela Coulter, Anik Giguere, Aubri Hoffman, Mirjam Körner, Aisha Langford, France Légaré, Daniel Matlock, Nora Moumjid, Sarah Munro, Karina Dahl Steffensen, Christine Stirling and Trudy van der Weijden in Medical Decision Making
Supplemental Material
sj-doc-3-mdm-10.1177_0272989X20978208 – Supplemental material for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration
Supplemental material, sj-doc-3-mdm-10.1177_0272989X20978208 for What Works in Implementing Patient Decision Aids in Routine Clinical Settings? A Rapid Realist Review and Update from the International Patient Decision Aid Standards Collaboration by Natalie Joseph-Williams, Purva Abhyankar, Laura Boland, Paulina Bravo, Alison T. Brenner, Suzanne Brodney, Angela Coulter, Anik Giguere, Aubri Hoffman, Mirjam Körner, Aisha Langford, France Légaré, Daniel Matlock, Nora Moumjid, Sarah Munro, Karina Dahl Steffensen, Christine Stirling and Trudy van der Weijden in Medical Decision Making
Footnotes
Acknowledgements
We are grateful for the assistance of the information specialist, Gregor Franssen Maastricht University, who supported the development of the electronic search strategies and to Hélène Elidor and Inheldia Cossou-Gbet (Université Laval) for their input into the data extraction phase. We also thank Dr. Freya Davies and Dr. Alison Cooper (Cardiff University) for their guidance on the realist review methodology.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Brodney is currently supported in part by a grant from Healthwise, Inc., a nonprofit organization, to Massachusetts General Hospital.
The grant funding agreement ensured the author’s independence in designing this study, interpreting the data, writing, and publishing the report. All other authors declare that there are no conflicts of interest. The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
