Abstract
Metamodels can be used to reduce the computational burden associated with computationally demanding analyses of simulation models, although applications within health economics are still scarce. Besides a lack of awareness of their potential within health economics, the absence of guidance on the conceivably complex and time-consuming process of developing and validating metamodels may contribute to their limited uptake. To address these issues, this article introduces metamodeling to the wider health economic audience and presents a process for applying metamodeling in this context, including suitable methods and directions for their selection and use. General (i.e., non–health economic specific) metamodeling literature, clinical prediction modeling literature, and a previously published literature review were exploited to consolidate a process and to identify candidate metamodeling methods. Methods were considered applicable to health economics if they are able to account for mixed (i.e., continuous and discrete) input parameters and continuous outcomes. Six steps were identified as relevant for applying metamodeling methods within health economics: 1) the identification of a suitable metamodeling technique, 2) simulation of data sets according to a design of experiments, 3) fitting of the metamodel, 4) assessment of metamodel performance, 5) conducting the required analysis using the metamodel, and 6) verification of the results. Different methods are discussed to support each step, including their characteristics, directions for use, key references, and relevant R and Python packages. To address challenges regarding metamodeling methods selection, a first guide was developed toward using metamodels to reduce the computational burden of analyses of health economic models. This guidance may increase applications of metamodeling in health economics, enabling increased use of state-of-the-art analyses (e.g., value of information analysis) with computationally burdensome simulation models.
Decision analytic models are valuable tools to inform health policy decisions by estimating the health and economic impact of health care technologies. When decision-analytic models take the form of simulation models, and particularly if they incorporate patient-level heterogeneity and stochasticity, the computational power of standard desktop computers may be insufficient to perform computationally demanding analysis within feasible time horizons.1–3 Although it is typically feasible to perform traditional analyses, such as probabilistic analysis to reflect parameter uncertainty, 4 performing more advanced analyses, such as value of information analysis, 5 may not be possible within a feasible time frame unless simulations are executed in parallel using high-performance computing clusters. Similarly, if we wish to optimize some specific model outcome, for example, to identify a screening or treatment strategy that maximizes patient outcomes subject to some set of constraints, we may find that this is infeasible using only desktop computing resources. 6
Performing these more advanced analyses may be computationally challenging, because they can require a large number of model evaluations (i.e., simulation runs). For example, suppose a discrete event simulation model has been developed to estimate the health economic impact of a novel cancer drug compared with an existing drug. Now assume that running this simulation model with 10,000 hypothetical patients for each of the 2 treatment strategies is sufficient to obtain stable outcomes over model runs and takes approximately 1 min. If an expected value of perfect parameter information analysis is to be performed for only 1 group of parameters using an inner probabilistic analysis simulation loop of 5000 runs and outer simulation loop of 2500 runs, 12.5 million simulation runs would be required in total. Even if it only requires 1 min to perform a simulation run, performing this analysis using a brute force approach on a desktop computer with 8 central processing unit cores working in parallel would take more than 1000 days.
Metamodeling methods can be applied to reduce the computational burden of computationally demanding analyses with simulation models.7,8 A metamodel, also known as a surrogate model or emulator, in general can be thought of as a function that approximates an outcome (i.e., response variable or dependent variable) of a simulator (i.e., the original simulation model) based on input that would otherwise have been provided to that simulator. 9 Metamodels are typically defined over the same (constrained) input parameter range as the corresponding simulator, as caution is needed when extrapolating input parameter values beyond their simulator range. Since metamodels are computationally cheap to evaluate, requiring only a fraction of the time that it takes to evaluate the simulator, they can be used as a substitute for the simulator to substantially reduce the analysis runtime. In the example above, and as illustrated in Figure 1, metamodels can be used to replace a health economic simulation model. Although this will still require 12.5 million evaluations to be performed, this can be done in very limited time. For example, if a metamodel would require approximately 0.1 s to evaluate, performing the analysis using the metamodel would take 2 instead of more than 1000 days. However, metamodels themselves take time to build and validate,10,11 but this will not take 1000 days.
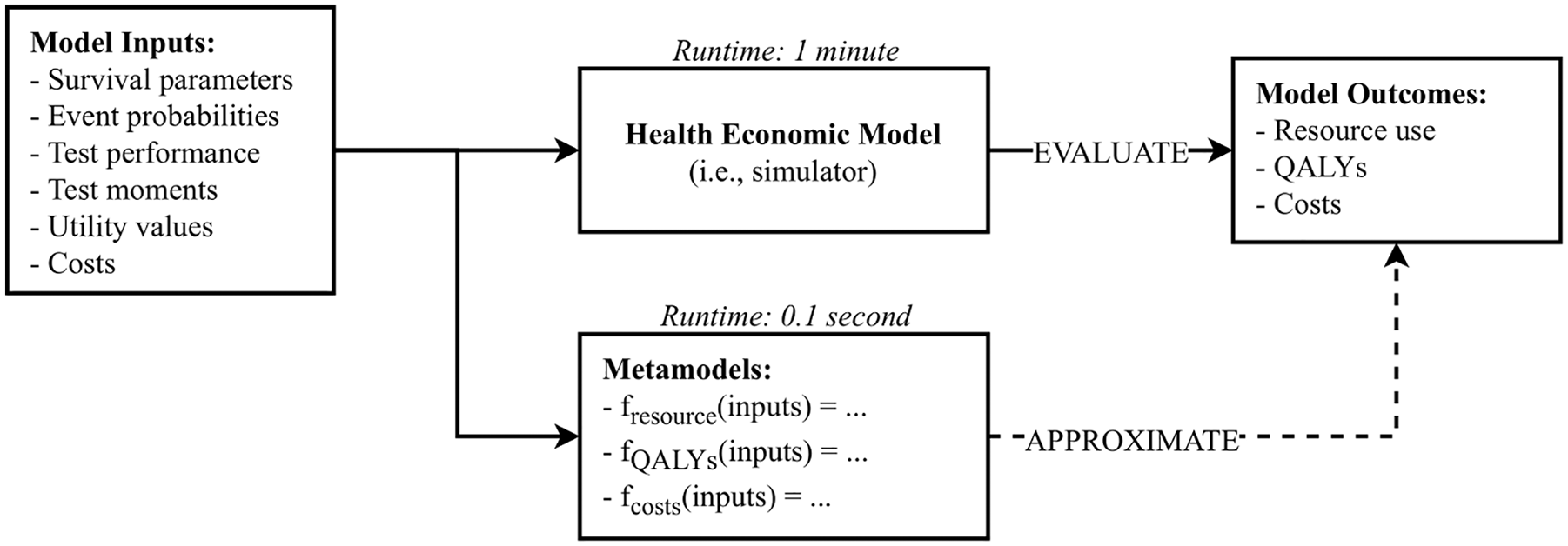
Illustration of how metamodels can be used in a health economic context to approximate the outcomes of the original health economic simulation model.
Figure 2, which will be discussed in detail throughout this article, includes an overview of how metamodels are developed. After identifying candidate metamodeling techniques, a set of experiments is to be generated. An experiment refers to a single sample of values of the model input parameters (thus, if there are
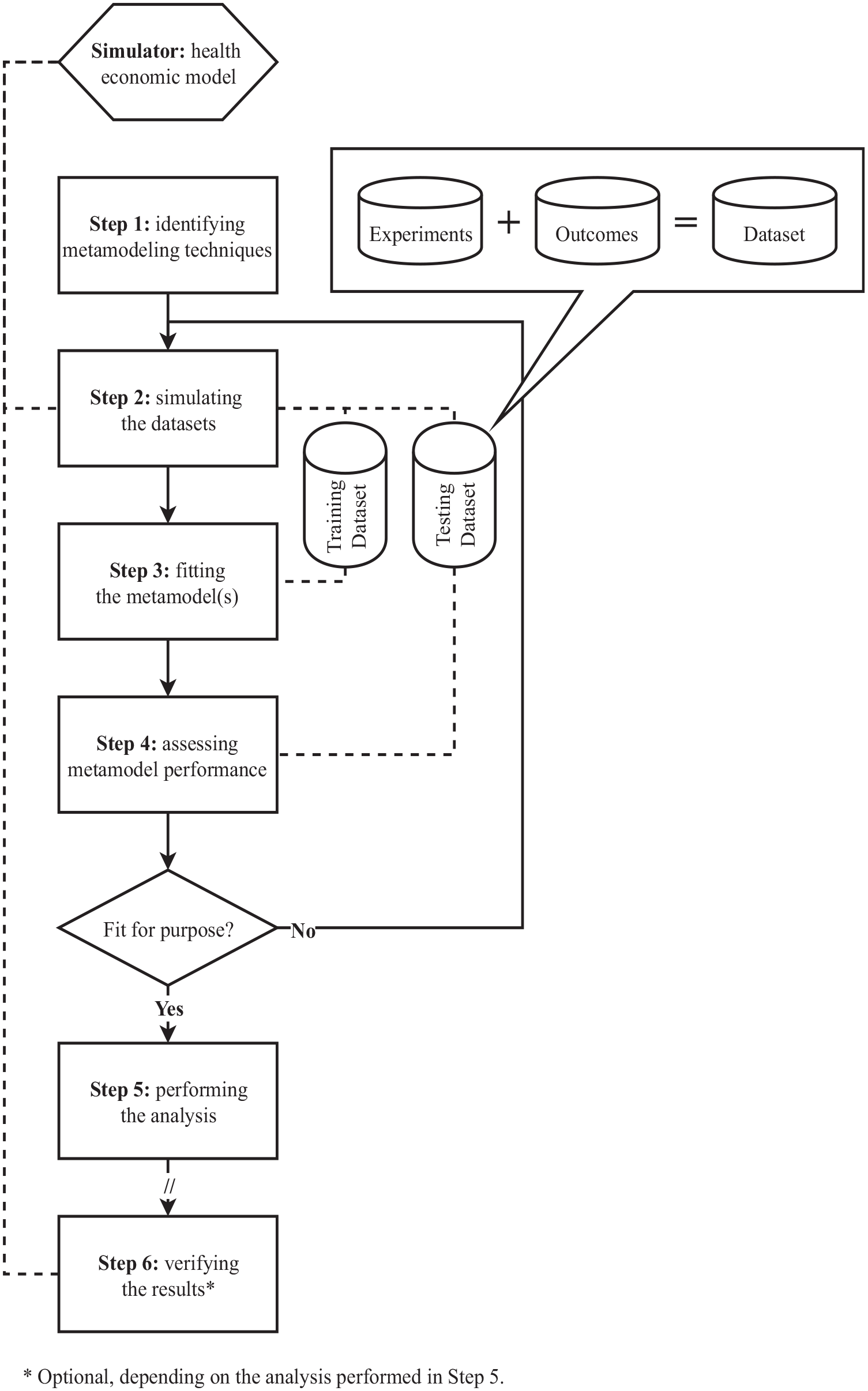
Process for developing, validating, and applying metamodeling methods in health economics.
Metamodeling methods are used widely across different fields of science and engineering, for example, to optimize designs of coronary stents, 12 high-speed trains, 13 and groundwater remediation, 14 as well as to estimate future water temperatures. 15 In health economics, de Carvalho et al. 16 recently demonstrated that a metamodel can be used to perform probabilistic analysis, which was not possible in a feasible time frame using their original model. 16 A previous literature review identified only 13 additional applications of metamodeling methods in health economics, mostly aiming to perform value of information analysis and applying various, relatively basic metamodeling methods compared with those used in other fields of research, suggesting the field of metamodeling within health economics to be in its infancy. 3 An important reason for the limited uptake of metamodeling methods within health economics may be that most health economic models and applied analyses have, until recently, been relatively simple and could often be performed within acceptable time frames. Other potential reasons include a lack of awareness of the potential of metamodeling methods to reduce runtime and a lack of guidance on how to apply these methods in a health economic context, which would explain the diversity in methods applied.
To increase awareness of the potential for applying metamodels within health economics, and to provide guidance for doing so, this study introduces the concepts of metamodeling to the wider health economic audience and presents a comprehensive, structured overview of metamodeling methods deemed suitable for use in a health economic context. Points of consideration for selecting and applying metamodeling methods are discussed, including directions specific to health economics.
Identification of Metamodeling Methods
Metamodeling methods (and the steps to be taken when applying them) were identified by a scoping literature search that was performed by K.D. This involved online searches, searches in Scopus and PubMed, and cross-referencing. Several publications that provide information on steps taken when applying metamodeling methods in health economics, identified in a recent review, 3 were used as a starting point.17-20 Method-specific information, other candidate metamodeling methods, and potentially relevant process steps were identified by iterative searches on methods and process steps introduced in these publications and by cross-referencing. For example, if the impact of different experimental designs on metamodel performance was discussed in an article found from a search on a specific metamodeling technique (i.e., structure of the metamodel), additional searches on these designs of experiments were performed to identify further information on these experimental designs and other designs of experiments. The iterative search process terminated when additionally found literature did not result in further inclusion of methods (i.e., until theoretical saturation was reached).
Metamodeling methods were included only if they are considered appropriate for use in health economics and have been commonly used in other fields of research, in line with the objective of the study. Methods were considered applicable to health economics if they are able to account for mixed (i.e., continuous and discrete) input parameters and continuous outcomes (i.e., response variables). Typical continuous input parameters of health economic models are, for example, costs and utilities, whereas the number of hospital days after a surgical intervention can be included as a discrete parameter. Similarly, typical continuous outcomes of interest are the net health or monetary benefit, total cost, and QALYs. Relevant steps to be taken when applying metamodeling methods in health economics were not prespecified but extracted from the literature as described above and structured in a process. Metamodeling methods and their characteristics were described according to this process and presented in a table or graphs when appropriate. In addition, examples of packages available to implement methods in R Statistical Software 21 and Python 22 were identified via an online search and introduced along with the corresponding methods. These 2 software environments were selected because they can be used to develop both the health economic simulation model and metamodel in a single script and are commonly used by academics, although other software environments such as SAS, Stata, and C++ can also be used to develop metamodels.
A Process for Metamodeling in Health Economics
A 6-step process for metamodeling in health economics was consolidated, covering methods from selecting suitable metamodeling techniques up to validating metamodel outputs against simulator outputs (Figure 2). A validated health economic simulation model (i.e., simulator) that is considered appropriate to perform the analysis of interest is a prerequisite, because although metamodels can theoretically be as accurate as their corresponding simulators, they cannot compensate for inaccuracies or bias in these simulators. Here, the analysis refers to what is to be analyzed using the original health economic model but is considered infeasible because of the associated computational burden. Depending on the analysis to be performed, the sixth step is facultative, as will be discussed. As for any type of modeling study, the process of metamodeling is iterative, since new insights may question prior decisions. Next, each process step will be described, including an overview of corresponding methods. An illustration of how this process would be applied to perform value of information analysis is presented in Appendix A.
Step 1: Identifying Candidate Metamodeling Techniques
Identification of theoretically suitable metamodeling techniques is based on study characteristics, including the analysis to be performed, type of input parameters (continuous, discrete, or mixed, i.e., both continuous and discrete), number of input parameters, and type of outcome (continuous or discrete). As discussed previously, the focus here is on techniques capable of handling mixed input parameters and continuous outcomes. In the presence of time or budget constraints for metamodel development, and when multiple techniques are considered appropriate for use, modelers can start by selecting and applying one of these techniques and only select and apply another technique if the resulting metamodel does not yield acceptable performance (see step 4).
Tappenden et al
18
identified 5 metamodeling techniques for application in value of information analysis: linear regression, response surface methodology, multivariate adaptive regression splines, Gaussian processes, and neural networks. These techniques are complemented with symbolic regression, which was also identified from the review,
19
and generalized additive models, which have been used previously for performing value of information analysis.23,24 In Table 1, an overview of techniques and their characteristics is provided. For each metamodeling technique, this overview includes the typically required number of experiments (which we have defined as low:
Overview of Candidate Metamodeling Techniques for Application in Health Economics, All of Which Are Able to Account for Mixed Input Parameters and Continuous Outcomes
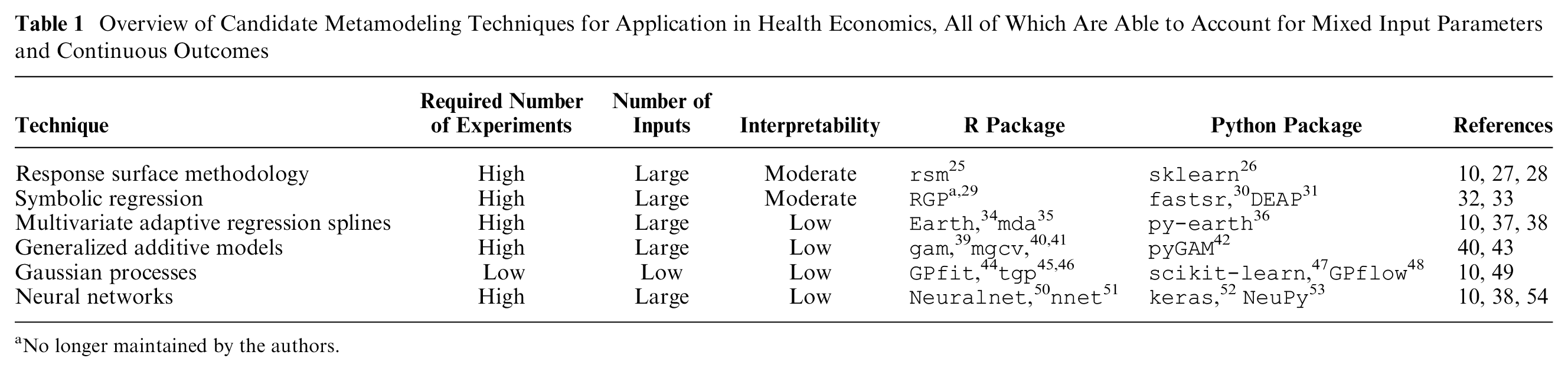
No longer maintained by the authors.
Simple linear regression is a statistical modeling technique well known in health economics and, theoretically, suitable for metamodeling. This assumes a linear relationship between independent variables (i.e., input parameters) and the dependent variable (i.e., outcome of interest) and is linear in the regression model parameters. 55 These models can easily be fitted to data sets of all sizes, including data sets with large numbers of experiments and input parameters, while allowing for both continuous and categorical input parameters. Although fitting linear regression models and interpreting their structure can be considered relatively easy, they are unlikely to be useful as metamodels of health economic simulation models, as the latter typically induce complex and nonlinear parameter interactions. More advanced techniques, allowing for more flexible model structures, are often better suited to represent such simulation models.
Response surface methodology is also linear in the regression model parameters but does not assume a linear input-output relationship, and it fits polynomial regression models to predict responses (i.e., outcomes).10,27,28 Both continuous and categorical input parameters can be considered in response surface models, and data sets including large numbers of experiments and input parameters can be used. However, high nonlinearity will require higher-order polynomials, which will require larger numbers of experiments; hence, it will require larger up-front simulator runtime. Although polynomial models are more difficult to interpret compared with linear models, if desired, general trends of model parameter influence can still be extracted from their model structures.
Symbolic regression uses genetic programming to construct a mathematical expression from elementary operators (e.g., “+” and “×”) and elementary functions (e.g., “log”), accurately describing the relation between input parameters and the outcome of interest, without making any priori assumption about this relationship.32,33 Fitting an accurate symbolic regression model may take substantial time, because of a potentially large number of candidate metamodels (i.e., large solution space). However, symbolic regression is capable of handling large data sets, including a large number of mixed input parameters. Symbolic regression models can be difficult to interpret unless the final expression is relatively simple or is simplified.
Multivariate adaptive regression splines were developed to model input-outcome relations that may not be constant across input space.10,37,38,56 Regression spline modeling divides the outcome domain into intervals and then estimates an equation, typically a low-order polynomial, for each interval. Different types of splines can be distinguished, based on how the number of intervals and level of smoothness are defined. Fitting multivariate adaptive regression splines includes an automated input parameter importance analysis (see step 2). Although capable of handling large data sets of mixed input parameters, regression splines are prone to overfitting. In contrast to the previously discussed metamodeling techniques, the interpretability of multivariate adaptive regression splines is limited.
Generalized additive models assume that the dependent variable is a smooth, but unknown, function of the independent variables.40,43 This unknown underlying smooth function is usually represented using splines, with the cubic spline as a common choice. In its simplest case, a univariate cubic spline represents an arbitrary smooth single-input function as a series of short cubic polynomials joined piecewise such that the function is twice differentiable at the “knots” (i.e., join points). The same spline can also be represented as the weighted sum of a series of predetermined “basis functions” that extend over the whole range of the function input. Simple univariate cubic splines have natural extensions to higher dimensions and to a metamodeling framework, in which the spline parameters (i.e., the basis function weights) are estimated from noisy data. Generalized additive models can handle large data sets and high numbers of input parameters, but their structure is difficult to interpret.
Gaussian process regression is a nonparametric regression method also known as Kriging.10,49 Gaussian processes use information on neighbor experiments for new predictions while directly providing information on the uncertainty in these predictions. This is unique for metamodeling techniques, since other techniques require additional effort to obtain information on prediction uncertainty. Although Gaussian processes are capable of considering mixed input parameters, 57 Treed Gaussian processes have been developed specifically for this type of data. 58 The interpretability of Gaussian processes is low. A disadvantage of Gaussian processes is that computational burden, both in terms of fitting and predicting, increases dramatically with increasing numbers of experiments and parameters, limiting their applicability. Hence, Gaussian processes are often well suited for optimization problems, which are typically defined by limited numbers of decision parameters. Furthermore, input parameter importance analysis can be performed to reduce the number of parameters (see step 2) and, thereby, computational burden.
Neural networks are nonparametric models that are commonly found in machine learning applications. These models consist of networks of nodes (called neurons) and layers, which learn about relationships between inputs, either continuous or categorical, and outputs, typically using large data sets.10,38,54 Although neural networks are commonly used for classification, they are also able to predict continuous outcomes. 59 Since no assumption regarding simulator structure is made, neural networks may well represent complex (i.e., nonlinear) health economic models. Developing large neural networks typically requires large numbers of experiments, which may pose challenges regarding obtaining sufficient simulator samples. Similar to multivariate adaptive regression splines, generalized additive models, and Gaussian processes, neural networks are “black boxes” that are hard to interpret.
In conclusion, as illustrated in the selection flowchart (Figure 3), Gaussian processes are particularly useful when obtaining sufficient simulator samples to apply the other techniques is infeasible. Response surface methodology, symbolic regression, multivariate adaptive regression splines, generalized additive models, and neural networks are typically useful when sufficient samples can be obtained from the simulator (i.e., original health economic model). If metamodel interpretation is important, response surface methodology and symbolic regression can be used to develop metamodels that may be interpretable to some extent.
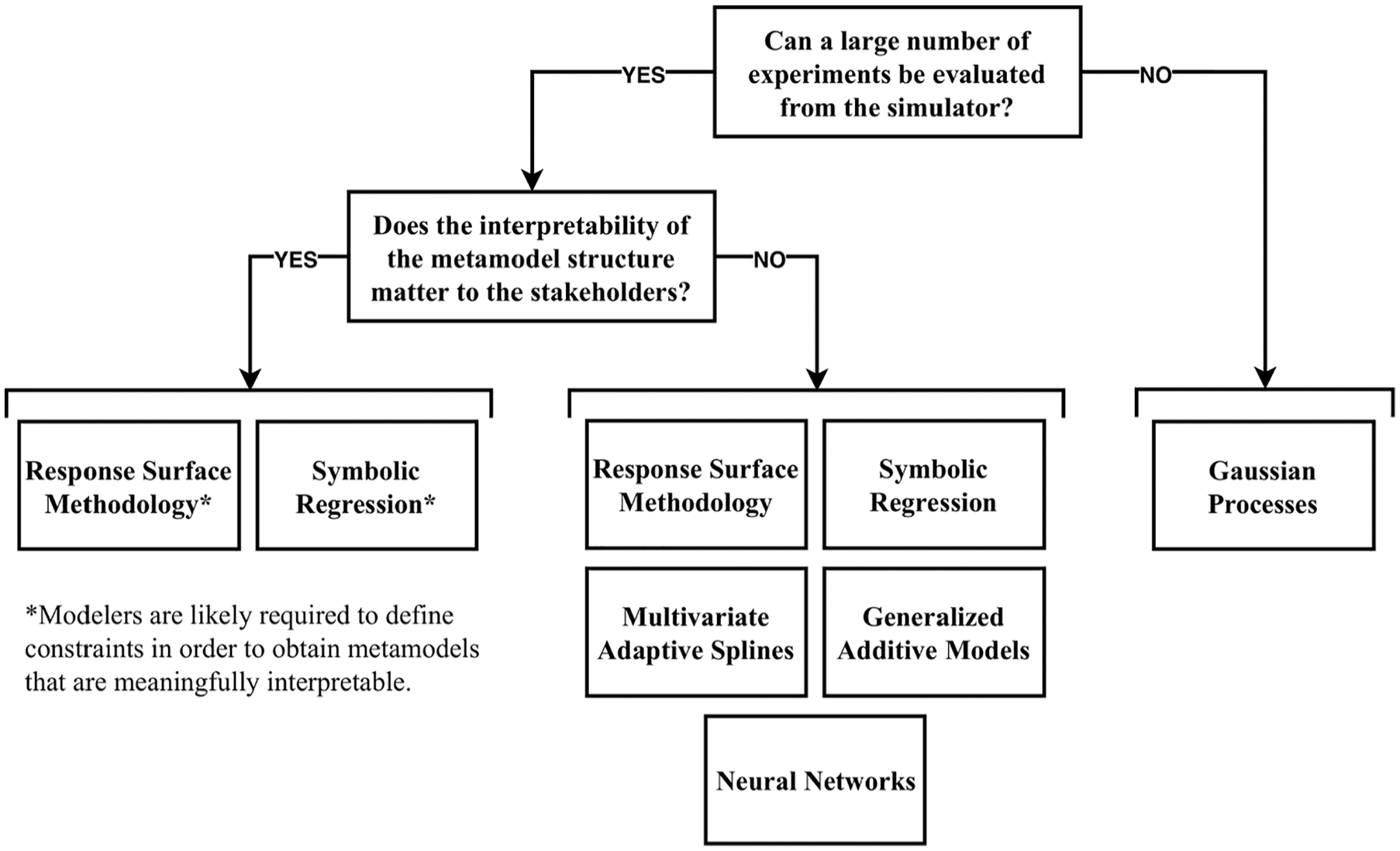
Flowchart for the selection of appropriate metamodeling techniques for a specific case study.
Step 2: Simulating Data Sets
Simulating data from the simulator is crucial in metamodeling studies, as metamodel performance is highly dependent on the data used for fitting. 9 Modelers control the number and definition of experiments used for fitting metamodels, which is fundamentally different from prediction modeling studies, for which data are typically observed from clinical studies or registries. 55 Furthermore, challenges regarding handling missing data, reversed causality, omitted variables, and measurement error are not applicable to metamodeling. There are 5 key aspects to simulating data sets for metamodeling: 1) the number of data sets, 2) parameter ranges, 3) design of experiments, 4) number of experiments, and 5) analysis used for obtaining simulator outcomes. As explained previously, an experiment refers to the generation of a single sample of model input parameter values in a metamodeling context. Hence, the number of experiments does not refer to a number of (hypothetical) patients but to the number of sets of model input parameter values for which the simulation model is evaluated to create data sets for metamodel fitting and validation.
As in prediction modeling, 2 distinct data sets are preferred for metamodeling studies: one for fitting (i.e., training or development data set) and one for validation (i.e., testing or validation data set). In prediction modeling studies, validation data sets would typically be obtained by isolating a proportion of the data from a single cohort for internal validation or by gathering additional data from another “plausibly related” cohort for external validation. 55 In metamodeling, however, it is preferable to obtain 2 separate data sets from the simulator, each having a prespecified design with comprehensive coverage. Obtaining 1 large data set and separating it in 2 data sets for training and validation may compromise the coverage of these data sets: either data set may lack the structure and properties induced by the design of experiments used to generate the single large data set. By obtaining 2 separate data sets, their structure and properties according to the design of experiments used will be maintained, as will be discussed.
The range of values that is to be covered in the data sets requires careful consideration for each input parameter separately. Although metamodels are theoretically capable of extrapolating beyond the parameter ranges covered by the data set on which they were fitted, such extrapolations are not preferable. The ranges that need to be covered are determined by the ranges of interest in the analysis that is to be performed using the metamodel. For example, if a metamodel is developed to optimize a cancer screening strategy, the ranges that are considered feasible in the optimization should be the same as those in the data sets used for fitting and validating the used metamodel(s). If the screening interval in years is a parameter of interest and any value between 1 and 10 is considered feasible, the parameter range for this parameter in the training and testing data set should also range from 1 to 10.
Design of experiments methods determine how sets of samples of parameter values are selected, which are to be evaluated from the simulator in order to obtain data sets for fitting and validation. 60 The objective of these methods is to cover parameter spaces and parameter interactions as effectively and efficiently as possible (i.e., with the least number of experiments). Failing to represent the full parameter spaces and parameter interactions will decrease metamodel performance. Most common designs of experiments are so-called single-pass methods that first define a complete set of experiments, all of which are subsequently evaluated using the simulator. 11 Commonly used designs are random designs, full factorial designs, and Latin hypercube designs.
Random designs, also known as Monte Carlo sampling methods, obtain
Full factorial designs fully enumerate possible combinations of discrete parameter values.
11
More specifically, for
Latin hypercube designs have been used often for designing computer experiments, as they efficiently cover the full parameter space.60,62,63 In its simplest form, Latin hypercube samples represent random combinations of values for each parameter, which are equally spaced between their minimum and maximum value for each parameter. More often, Latin hypercube samples represent random combinations of random values from equally sized bins that cover the parameters’ domains. Over the years, more advanced versions have been developed, such as the maximin Latin hypercube design, 64 which maximizes the minimum distance between design points, and orthogonal Latin hypercube designs. 65
Figure 4 illustrates how random, full factorial, and maximin Latin hypercube designs may define 9 experiments for 2 continuous parameters
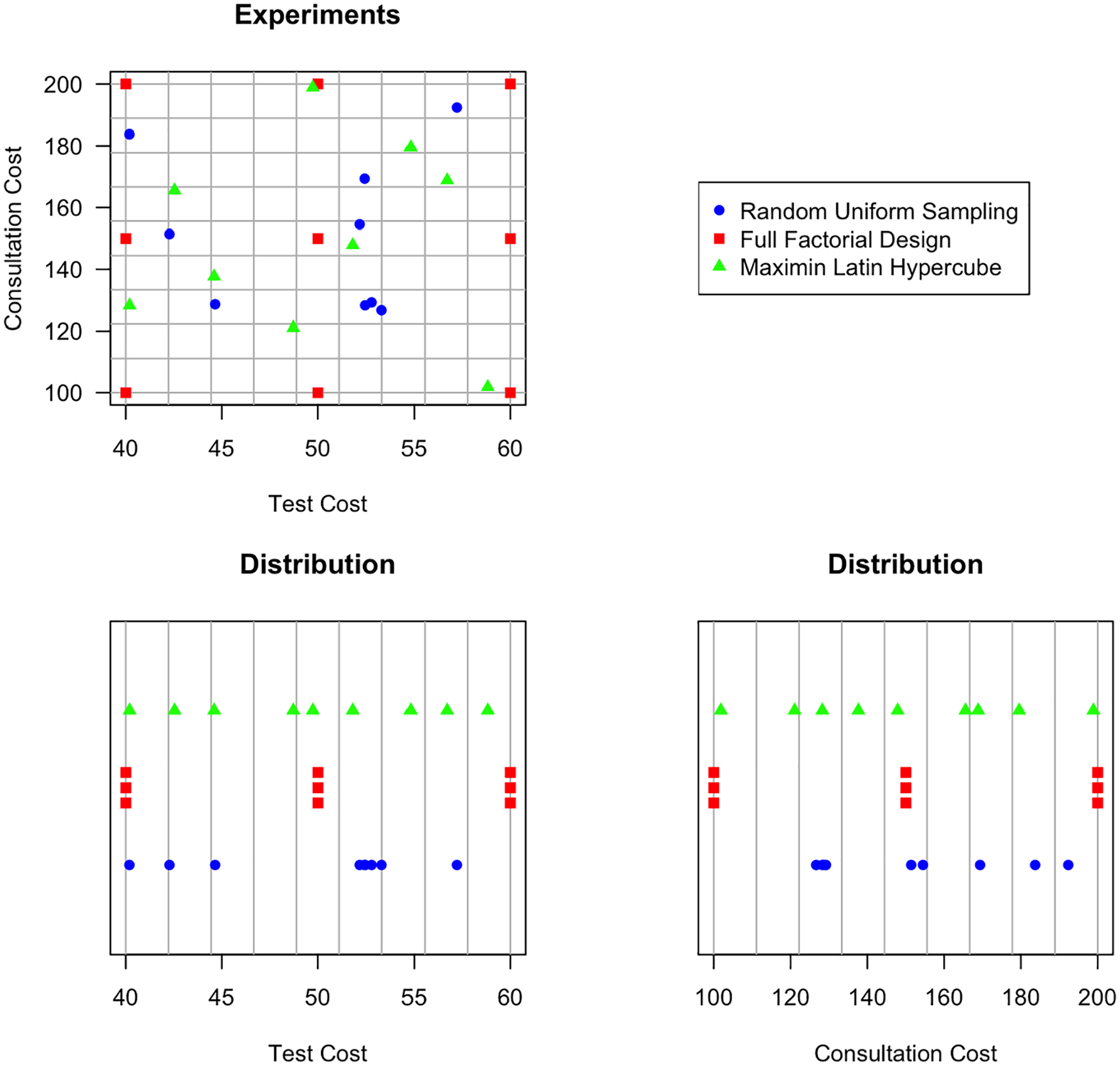
Illustration of how a random uniform sample, full factorial design, and maximin Latin hypercube sample may define 9 experiments for 2 continuous parameters,
In general, Latin hypercube designs are preferable for both training and testing data sets, especially when only a limited number of experiments can be evaluated from the simulator in the available time. Optimized Latin hypercube designs can easily be generated in most software environments, for example, using the
How many experiments are required (i.e., how large the
Whether a deterministic or probabilistic analysis needs to be performed to evaluate experiments from the simulator depends on the analysis to be performed with the metamodel. In a deterministic analysis, the simulator is evaluated once for the expected values of the input parameters. 4 In a probabilistic analysis, the simulator is evaluated numerous times, typically thousands of times, based on parameter values sampled from distributions that reflect the uncertainty in the parameter values (i.e., second-order uncertainty). If a model is nonlinear, which most health economic models are, health economic outcomes from a deterministic analysis are not equal to those of a probabilistic analysis. 74 If metamodels are being used to perform model probabilistic analysis or value of information, simulator outcomes based on a deterministic analysis should be used. If the aim is to perform calibration or optimization, simulator outcomes based on probabilistic analyses are preferred, because these are the values expected to be observed in reality given the current information. However, performing a probabilistic analysis for each experiment might not be feasible because of the required simulator runtime. In that case, outcomes from a deterministic analysis may be used to approximate the outcomes of a probabilistic analysis, although this should be clearly noted as a limitation when reporting the results.
The stability of outcome estimates is another important aspect. If stochastic uncertainty, also referred to as uncertainty on the patient level or first-order uncertainty, is reflected in a patient-level simulation model, sufficient hypothetical patients need to be simulated to obtain stable outcomes. Similarly, regardless of whether first-order uncertainty is reflected, sufficient probabilistic analysis runs need to be performed to obtain stable point estimates. When insufficient hypothetical patients are simulated, or probabilistic analysis runs performed, the subsequent noise in the data used for fitting metamodels may have a pernicious effect on metamodel performance. Outcomes can be considered stable if the outcomes obtained from simulations with different random numbers, but with the same input parameter values, are sufficiently similar. What defines “sufficiently similar” differs among case studies and should be discussed with all relevant stakeholders (e.g., care providers, decision makers, and modelers). Obtaining stable outcomes may require a substantial number of patients to be simulated or simulation runs to be performed and may not be feasible in practice. However, to reduce the number of patients to be simulated or the number of runs to be performed to obtain stable outcomes, variance reduction techniques may be applied, such as using common random numbers when comparing strategies.75,76
Step 3: Fitting Metamodels
After evaluating an initial set of experiments from the simulator, this training data set can be used to fit selected metamodeling techniques. Steps involved in the fitting processes differ between techniques as well as any settings to be provided. We refer to the corresponding literature and software documentation to learn about the steps to be taken and settings to be provided (see step 1 and Table 1). As a basic example, some metamodeling techniques or software packages require input parameters to be rescaled. Fitting metamodels is an iterative process, in which settings may be adapted, or more experiments may be evaluated from the simulator, after assessing model performance (see step 4).
Step 4: Assessing Metamodel Performance
Assessing the performance of fitted metamodels is essential to further improve that performance, by iteratively improving (extending) the design of the training data set used or adapting the settings for fitting these models. In addition, an initially selected metamodeling technique may be deemed inappropriate if performance does not reach an acceptable level, resulting in exclusion of this technique from the list of potential candidates (step 1). Performance can be assessed using the testing data set evaluated from the simulator in step 2. Since metamodels of health economics models will typically predict continuous scale outcomes, of main interest is to quantify how close predictions are to actual simulator outcomes. Assessing accuracy and comparing different metamodels can be done graphically and using quantitative performance criteria. A validation plot, with predicted values on the
It is important to be aware of performance criteria characteristics when selecting one, or several, to compare metamodels or to assess whether model performance is acceptable. For example, compared with mean absolute errors, mean squared errors place more weight on outliers. In addition, compared with squared errors, setting a desired level of accuracy is more straightforward for absolute (relative) errors, as these can be set by answering questions such as, “What is the maximum mean deviation in predicted life-years the metamodel is allowed to have compared with the simulator outcomes?” The performance that can be considered acceptable for deciding to apply a metamodel for performing analyses differs among case studies and should be based on input from all stakeholders. For example, when the point estimate for the incremental QALYs is 0.18 QALYs, an absolute error of 0.01 QALY may be considered appropriate by stakeholders. Since different performance criteria and definitions of acceptable performance may yield alternative conclusions, these should be decided upon prior to metamodel development.
Step 5: Applying the Metamodel
Once a metamodel has been developed and validated, it can be used to perform analyses that could not be performed in a feasible time period with the original health economic model. Previous applications of metamodels in health economics include value of information analysis, model calibration, optimization, probabilistic analysis, and obtaining stable outcomes over multiple runs with the same input values. 3 In addition, metamodels can be used in online tools for which limited computer resources are available. For example, see de Carvalho et al. 16 for a demonstration of metamodels used for probabilistic analysis or Appendix A for an illustration of the presented 6-step process for performing value of information analysis. Another example may be to use metamodels for evaluating a large set of (thousands of) screening strategies, for example, to identify the starting age, screening interval, and number of screening rounds that optimize health and economic outcomes.
Step 6: Verifying Results (Optional)
If metamodels are used for optimization purposes, it is recommended to reevaluate a certain number of best strategies identified by the metamodel using the original health economic model, to assess whether their outcome and ordering meaningfully differ. By providing these results, decision makers are better informed about the expected impact of choosing a good but not optimal candidate strategy for implementation, which may be favored over the optimal strategy for practical reasons. For other types of analyses, such as probabilistic or value of information analysis, additional verification will not add to the validation of step 4, because reevaluating a number of strategies using the simulator will yield approximately the same error values as those obtained in step 4. Although this will also be the case for several best-performing strategies when optimization is performed, knowing the true outcomes and ordering of the strategies according the simulator is informative, whereas knowing the true outcome for a specific probabilistic analysis run is not of any value.
Discussion
This study provides an introduction to metamodeling methods that can be used to reduce the computational burden of advanced analyses with health economic models and addresses challenges regarding the selection and application of these methods. Similar to ordinary statistical regression modeling, different methods, which are discussed, are available with their own advantages, disadvantages, and underlying assumptions, and directions for selecting and implementing these methods are provided. Selected methods are structured in a comprehensive 6-step process that can be followed to ensure essential modeling steps are covered, as it includes all relevant design choices. In addition, the process discussed can be used as a structure to effectively and efficiently communicate metamodeling studies, to increase modeling transparency and reproducibility.
Given that tools and packages are available to generate experiments according to specific designs and to fit different types of metamodels, for example, in R and Python, applying metamodeling methods is feasible for health economic analysts. Currently available software and results from this study enable analysts to perform computationally demanding analyses with their models, such as value of information analysis, model calibration, and optimization. The benefits of developing metamodels are relevant to analyses using patient-level simulation methods, such as microsimulation state-transition modeling and discrete event simulation, but also to cohort models used to perform analyses that require a large number of model evaluations.
Applying metamodeling methods can reduce computational burden, but this usually comes at the price of introducing additional uncertainty in the model outputs. Consequently, checking whether underlying assumptions are met and checking metamodel performance are crucial to success and essential to build confidence in the metamodel. Since modelers typically have access to the original health economic model, validation of the metamodel is often not a problem, although it is likely to be more demanding in terms of effort compared with developing the metamodel itself. The starting point for building any metamodel, however, should be a realistic and validated health economic model, since metamodels can theoretically be as accurate as their corresponding simulators but will not compensate for inaccuracies in these simulators. Moreover, when metamodels are used for optimization, the strategies considered, and possibly identified as optimal, may not be supported by (the data underlying) the simulator. Caution is required when such extrapolation is (automatically) performed, and such optimization results should serve only to initiate discussion on the appropriateness and validity of the simulator and the data supporting it. In addition, the application of metamodeling methods requires communication of metamodeling design choices made in publications, for which space is typically already limited. Hence, metamodeling studies may be published separately from their simulator to ensure the metamodeling process can be appropriately described. Furthermore, there is a “sweet spot” for metamodeling: sufficient experiments need to be evaluated using the simulator to develop an accurate metamodel, but evaluating all experiments of interest should not be feasible.
Several technical challenges remain regarding the application of metamodeling methods in health economics. Simulators in health economics may include complex behavior, such as rigid cutoffs due to clinical decision rules, which may be complex for metamodeling techniques to capture. In addition, (combinations of) model input parameters may be subject to constraints, which are difficult to incorporate in efficient designs of experiments, such as Latin hypercube sampling. If sufficient samples can be evaluated from the simulator to use random or full factorial designs in which constraints can be accounted for more easily, however, this might not be an issue. Alternatively, more advanced adaptive sampling strategies may need to be applied.
Not all metamodeling methods are directly suitable for application in health economics and hence have not been discussed. However, it is important to note that some techniques, such as those that can be used for categorical outcomes (i.e., classification), could theoretically be applied in health economics after discretizing continuous outcomes. Such an approach has been taken previously by using a binary outcome to reflect whether one treatment was preferred over another in a logistic regression model. 73 Similarly, examples of packages for R and Python were discussed, whereas additional packages are likely to be available and other software environments can also be used to develop metamodels, such as Stata, SAS, and C++. In addition, alternative performance criteria for metamodel validation can be found, or may be developed, based on study-specific needs. With regard to sampling methods, only single-pass methods have been discussed, whereas iterative methods, also known as adaptive sampling or active learning methods, also exist.79,80 Iterative methods use an initial data set for fitting an initial metamodel, which is subsequently used in an iterative process to identify additional experiments to be added to the data set, to update the initial metamodel, and to check the updated metamodel performance, until this performance is in accordance with a predefined threshold. 71 The additional experiments are sampled in the area in which performance needs to be improved. Although iterative methods are more efficient compared with single-pass methods, they are substantially more complicated to implement and require simulators to be available in the same software environment used for generating experiments and fitting the metamodel. Nevertheless, these methods may be useful if insufficient experiments according to a single-pass design can be obtained to develop an accurate metamodel. Also, alternative designs of experiments are available, such as D-optimal designs, which are efficient and can account for constraints, but for which an linear or quadratic model simulator model structure should be known, 81 or for so-called Sobol sequences, which may be more efficient compared with Latin hypercube designs with low-dimension (i.e., number of input parameters) problems.82,83
Future metamodeling applications should further illustrate the potential and use of these research methods and also identify common challenges. Once the field of metamodeling in health economics has evolved, good research practices (i.e., consensus guidance) can be identified to further improve the quality of metamodeling studies.
Supplemental Material
appendix.rjf_online_supp – Supplemental material for Introduction to Metamodeling for Reducing Computational Burden of Advanced Analyses with Health Economic Models: A Structured Overview of Metamodeling Methods in a 6-Step Application Process
Supplemental material, appendix.rjf_online_supp for Introduction to Metamodeling for Reducing Computational Burden of Advanced Analyses with Health Economic Models: A Structured Overview of Metamodeling Methods in a 6-Step Application Process by Koen Degeling, Maarten J. IJzerman, Mariel S. Lavieri, Mark Strong and Hendrik Koffijberg in Medical Decision Making
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
