Abstract
In shared decision making (SDM), patients and clinicians engage in a conversation and work together to make decisions about health and care that fit best individual patients and their lives.1,2 SDM is considered particularly pertinent when there is more than one reasonable approach available to manage the patient’s situation and when these approaches differ in ways that matter to patients.2,3 Although SDM models differ to some extent, 4 many prominent models distinguish 4 key elements: 1) fostering choice awareness, 2) discussing relevant options and corresponding pros and cons, 3) discussing patient views and preferences, and 4) making the final decision.3,5
As the interest in implementing SDM in routine care is growing, the need to evaluate its occurrence and the effects of SDM interventions is growing as well. In a recent systematic review, Gärtner et al. 6 identified 40 different instruments that assess SDM. Some of these are self-report instruments, providing insight in the experiences of patients or clinicians. These self-report instruments tend to show ceiling effects (i.e., scores are generally high without much variance),7,8 possibly due to halo effects (i.e., SDM is difficult to disentangle from other qualities attributed to the care received or from overall satisfaction). 9 Other SDM instruments are observer-based coding schemes, requiring a trained assessor to observe and code the patient-clinician conversation. This is time and resource consuming, but raters are trained in the evaluation of SDM and thus can be expected to apply stricter criteria and avoid ceiling effects. Indeed, previous research has shown results from observer-based instruments to reveal lower levels of SDM compared to results based on self-report instruments.7,10
In their review, Gärtner et al. 7 found an overall lack of evidence for the psychometric quality of SDM instruments. The authors therefore recommend to select SDM instruments for studies or evaluation based on the content of these instruments. The aim of this study was to determine whether the items of extant SDM observer-based coding schemes assess the 4 key elements of SDM.
Methods
Selection of Instruments
For this content analysis, we selected SDM instruments identified in the review by Gärtner et al. 6 These authors searched 7 databases for studies investigating instruments measuring the process of SDM and identified 51 articles describing 40 instruments, of which 20 were observer-based schemes. For our content analysis, we used these coding schemes; we excluded translated versions of the same scheme (n = 4),11–14 schemes of which a revised version was available (n = 1), 15 and schemes that contained the same items and only used a different scoring method or rating scale (n = 3),16–18 thus leaving 12 schemes for analysis.19–30
Analysis
We extracted all items of selected schemes and classified them into categories of SDM behaviors. In addition to the 4 key elements of SDM,3,5 we inductively created (sub)categories based on the data (Table 1). Items were categorized independently and in duplicate by 2 researchers (MK, IH). Each item was attributed to 1 category only (Table 2). Discrepancies were discussed and solved in consensus. A third researcher (FRG) checked all final categorizations, and again, discrepancies were solved in consensus.
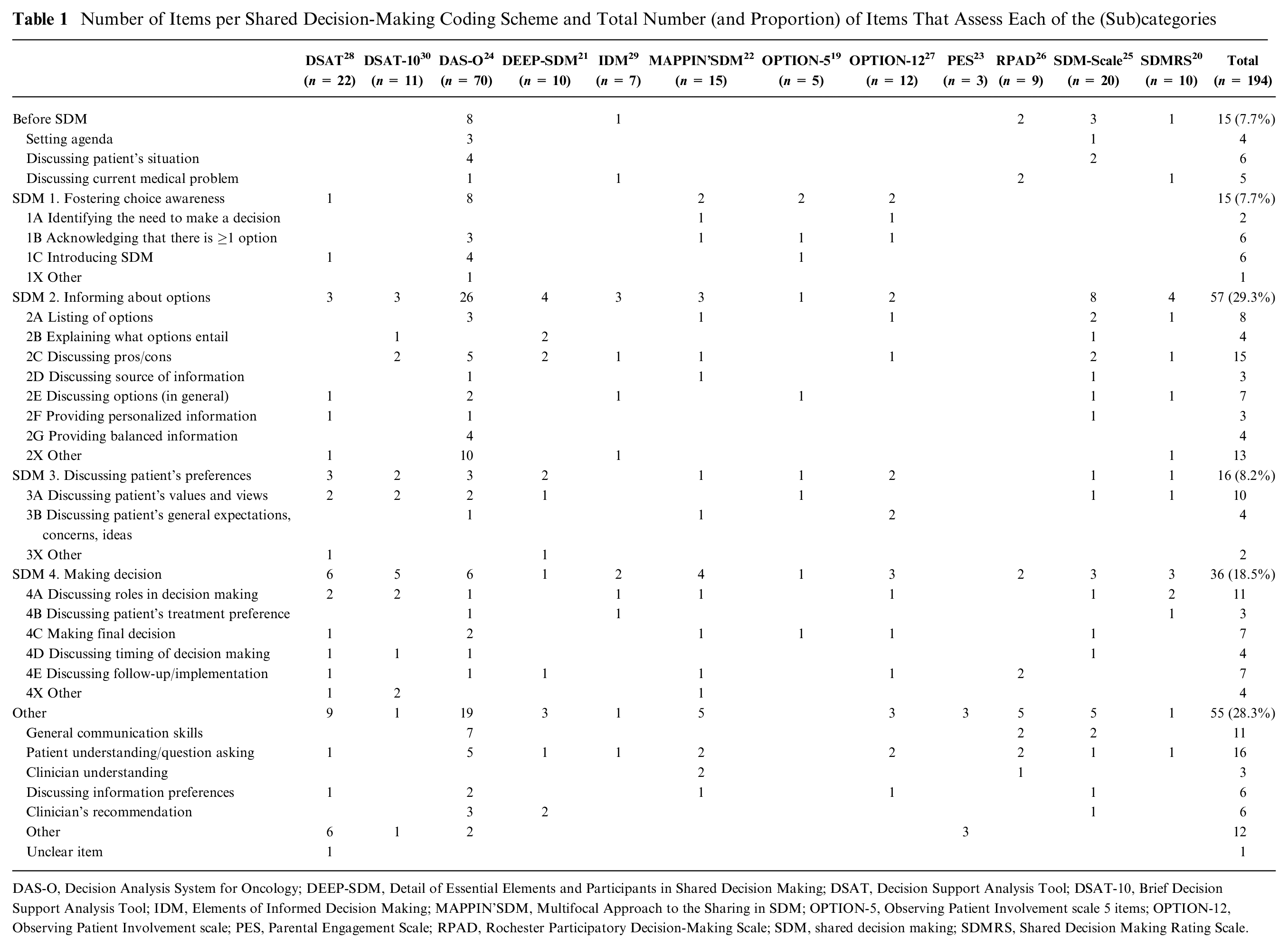
Number of Items per Shared Decision-Making Coding Scheme and Total Number (and Proportion) of Items That Assess Each of the (Sub)categories
DAS-O, Decision Analysis System for Oncology; DEEP-SDM, Detail of Essential Elements and Participants in Shared Decision Making; DSAT, Decision Support Analysis Tool; DSAT-10, Brief Decision Support Analysis Tool; IDM, Elements of Informed Decision Making; MAPPIN’SDM, Multifocal Approach to the Sharing in SDM; OPTION-5, Observing Patient Involvement scale 5 items; OPTION-12, Observing Patient Involvement scale; PES, Parental Engagement Scale; RPAD, Rochester Participatory Decision-Making Scale; SDM, shared decision making; SDMRS, Shared Decision Making Rating Scale.
Examples of Items of Shared Decision-Making Measurement Instruments Classified in Each (Sub)category.

DAS-O, Decision Analysis System for Oncology; DEEP-SDM, Detail of Essential Elements and Participants in Shared Decision Making; DSAT, Decision Support Analysis Tool; DSAT-10, Brief Decision Support Analysis Tool; IDM, Elements of Informed Decision Making; MAPPIN’SDM, Multifocal Approach to the Sharing in SDM; OPTION-5, Observing Patient Involvement scale 5 items; OPTION-12, Observing Patient Involvement scale; PES, Parental Engagement Scale; RPAD, Rochester Participatory Decision-Making Scale; SDM, shared decision making; SDMRS, Shared Decision Making Rating Scale.
Results
The 12 included observer-based SDM coding schemes contained a total of 194 items (median = 10, range 3–70; see Table 1).
Five of 12 schemes (DSAT (Decision Support Analysis Tool), DAS-O (Decision Analysis System for Oncology), OPTION-5 (Observing Patient Involvement scale 5 items), OPTION-12 (Observing Patient Involvement scale), Mapping’SDM (Multifocal Approach to the sharing in SDM)) contained at least 1 item for all 4 SDM key elements. The schemes with the highest percentage of their items classified in 1 of the 4 SDM key elements were the OPTION-5 (n = 5/5; 100%) and the DSAT-10 (Brief Decision Support Analysis Tool) (n = 10/11; 90%).The schemes with the lowest percentage were the PES (Parental Engagement Scale) (n = 0/3; 0%) and the RPAD (Rochester Participatory Decision-Making Scale) (n = 2/9; 22%). Seven schemes did not assess the key element “fostering choice awareness,” and 3 did not assess “discussing patient’s preferences” (Table 1).
Of the 194 items, 124 (64%) could be classified into 1 of the 4 SDM key elements. Almost half of these (n = 57/124, 46%) assessed “informing about options” (SDM element 2; see Figure 1), and almost one-third (n = 36/124, 29%) assessed “making a decision” (SDM element 4). The remaining items assessed “discussing patient’s preferences” (SDM element 3, n = 16/124, 13%) or “fostering choice awareness” (SDM element 1, n = 15/124, 12%). The SDM subcategories with the most items were “discussing pros and cons” (n = 15/124, 12%), and “discussing roles in decision making” (n = 11/124, 9%).
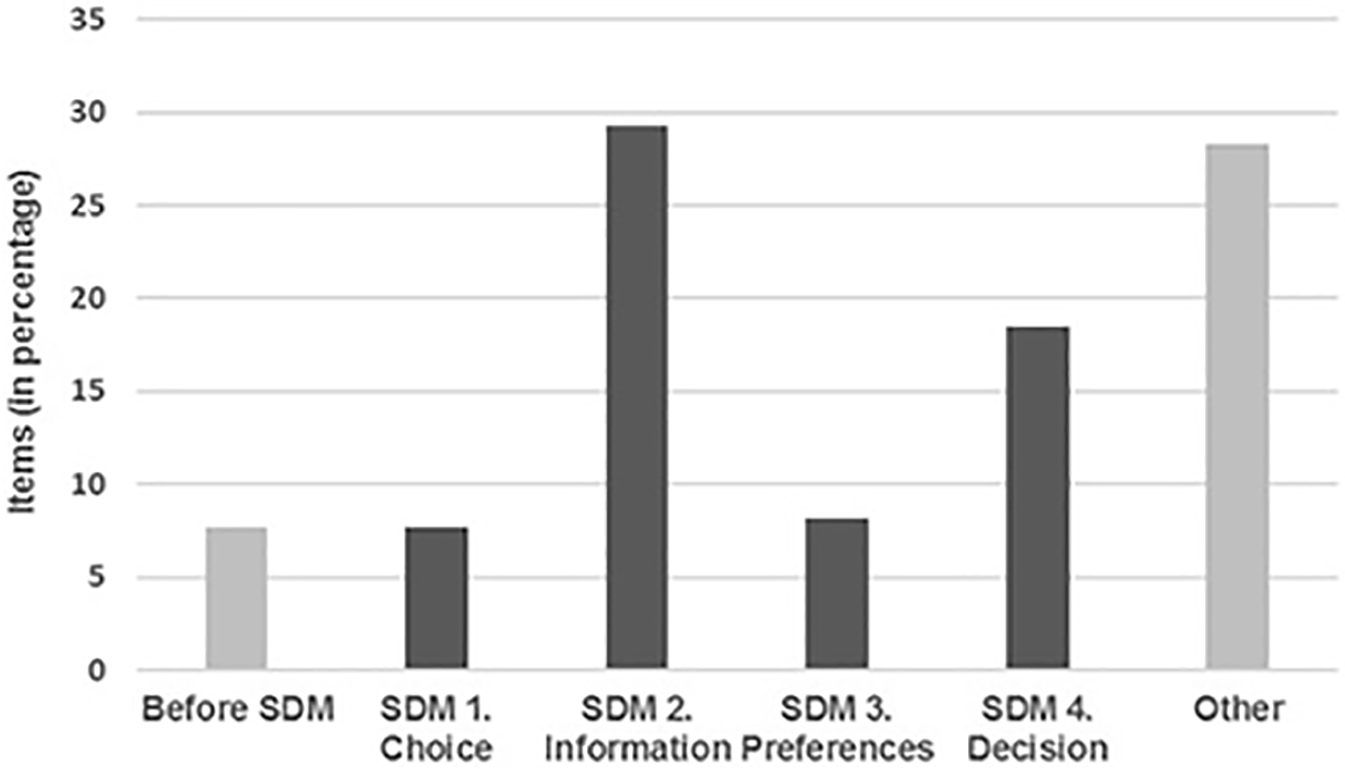
Distribution of items from shared decision-making (SDM) coding instruments.
Seventy of 194 items (36%) could not be classified into 1 of the 4 SDM key elements and assessed, for example, the discussion of the current medical problem (n = 5/70, 7%), general communication skills (n = 11/70, 16%), or checking patients’ understanding or allowing patients to ask questions (n = 16/70, 23%).
Discussion
This study aimed to determine the extent to which extant SDM coding schemes assess the key elements of SDM. We showed that less than half of the coding schemes assess all 4 key elements of SDM. The coding schemes mainly focus on information provision, while items to assess other key elements such as fostering choice awareness or discussing patient’s preferences are less often included. Especially the latter is surprising, as discussing what matters to patients seems the drive for SDM. The importance of fostering choice awareness was only recently incorporated in SDM models, which may explain why this element is absent in most coding schemes (Bomhof-Roordink H, Gärtner FR, Stiggelbout AM and Pieterse AH; unpublished data). Moreover, the schemes assess more than the 4 elements considered key to SDM alone; over one-third of items assess, for example, general communication and consulting skills, such as agenda setting and checking patient or clinician understanding. These are relevant behaviors for SDM but may not be specific to SDM. 31
A strength of our study is that it could build on a recent systematic review on SDM instruments. 6 The categorization of items was performed independently and involved SDM experts from different backgrounds and institutes. However, we need to put in perspective that the diversity in the content of different extant coding schemes reflects the reported lack of consensus on what SDM entails (Bomhof-Roordink H, Gärtner FR, Stiggelbout AM and Pieterse AH; unpublished data). 4 As further efforts are made to conceptualize SDM, what authors of SDM models consider to be “key elements” may shift as well.4,32
Our study showed that observer-based SDM coding schemes have a strong focus on assessing information provision. This is not surprising; providing patients with information has received great attention in SDM research and implementation, and providing information is crucial to come to an informed decision. 33 Also, information provision encompasses a broad range of behaviors, such as explaining the options at hand, as well as their pros and cons and respective probabilities, which can all be relevant to evaluate when assessing SDM. Surprisingly, the SDM coding schemes hardly or not at all assessed some of these behaviors. We did not find any item specifically focusing on whether or how probabilities are discussed, despite the importance of clear and understandable risk communication to consider and weigh pros and cons of available approaches.34–36 Also, whether information is presented in a balanced and nonsteering way 37 cannot be assessed by any of the SDM coding schemes except the DAS-O. This is striking, as these behaviors are relevant to identify whether information provision supports the SDM process.
SDM elements such as the fostering of choice awareness or discussing the patient’s preferences were underrepresented in the coding schemes. Hence, the schemes are less likely to pick up behaviors or behavior changes in these areas. In addition, the inclusion of many items that are not specific to SDM, such as general communication skills, may limit the size of the effects of interventions that specifically focus on SDM. Since our review suggests that there is no dominant “best option” among the SDM instruments to evaluate SDM or the effects of SDM interventions, the decision on which SDM instrument to use should depend on its content and the focal elements aimed to assess. Similarly, in presenting and interpreting (total) scores of SDM instruments, we must bear in mind the focus of their content.
To move forward in studying and implementing SDM, we need to pay attention to scores on individual items or (sum) scores per SDM element 38 to discover effects that are diluted in the total scores. As an example, information interventions, such as some decision aids, may not necessarily improve the involvement of patients in other ways than by informing them more thoroughly, thus increasing the total score in ways that do not reflect an encompassing effort to promote SDM. In addition, providing more information does not necessarily mean better information provision, nor does it mean that the information provided actually helps to advance the situation of the patient. Still, total scores of SDM instruments may suggest “more shared decision making.” Consequently, we may be “checking the SDM boxes” without improving the quality of SDM. 39 Previously, Kunneman et al. 40 warned for such “measurement with a wink” in which we may be getting higher scores on the instruments but not necessarily higher-quality SDM.
Conclusion
Our study shows large variation in the content of observer-based SDM coding schemes, with a strong focus on information provision and absence of other elements considered key to SDM. We need to be cautious in interpreting scores of SDM instruments, as high scores do not necessarily mean more SDM. We recommend researchers to be mindful and transparent about their motivation for using particular SDM measurement instruments and to present scores of individual items or SDM elements when reporting and interpreting the findings of their studies.
Footnotes
Acknowledgements
We thank Meltem Tokat for her help in data collection and analysis.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by a personal grant from the Dutch Cancer Society (UVA 2013-5949). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The following author is funded by the sponsor: Inge Henselmans, PhD.
