Abstract
Background:
Cannabidiol (CBD) is being investigated as a novel antipsychotic treatment, but its effects on psychosis are mainly drawn from pre-clinical studies, leading to uncertainty about its clinical impact.
Aims:
To evaluate the efficacy and tolerability of CBD in patients with schizophrenia spectrum disorders.
Methods:
PubMed, Scopus, Embase, PsycInfo, Cumulative Index to Nursing and Allied Health Literature (CINAHL), and clinicaltrials.gov were searched up to July 2025. Randomized controlled trials (RCTs) of CBD in adults with schizophrenia spectrum disorders were included. Pairwise meta-analyses were conducted using random-effects models. The primary outcome was the standardized mean difference (SMD), with 95% confidence interval (95% CI), between CBD and controls at post-treatment on psychosis symptom severity. Tolerability assessment considered pooled odds ratio (OR, with 95% CI) of trial withdrawal and side effects across treatment groups.
Results:
A total of eight trials (six published and two unpublished), accounting for 288 participants diagnosed with psychotic disorders, were included. CBD was administered orally, at a median daily dose of 800 mg. Follow-up times ranged from 20 minutes to 12 weeks. Effect size for CBD on psychosis symptom severity was not statistically significant (SMD: −0.194; 95%CI: −0.444 to 0.056), similarly on cognitive, and psychosis positive and negative symptoms. Tolerability assessments were comparable across CBD and controls. Quality of the evidence ranged from low to very low.
Conclusions:
Despite the limited number of outcomes assessed at the current state of the evidence, CBD did not show a clear benefit for psychosis symptoms in RCTs but was generally well tolerated. Larger, high-quality trials are needed to reach more robust conclusions about CBD efficacy in these disorders.
Introduction
Cannabis has long been linked with psychosis (Osborne et al., 2025). Exposure to high-potency delta-9-tetrahydrocannabinol (THC) cannabis has shown to increase the risk of developing and relapsing psychotic disorders (Vaucher et al., 2018). In contrast, cannabidiol (CBD), a non-intoxicating cannabis constituent, has been proposed to alleviate psychotic symptoms and to attenuate some of the acute psychotomimetic and cognitive effects of THC (Schubart et al., 2014). Preclinical and experimental pharmacology research suggested CBD to modulate the endocannabinoid system, particularly by enhancing anandamide signaling, via inhibition of its intracellular degradation by fatty acid amide hydrolase (Leweke et al., 2012). Furthermore, CBD was reported to activate N-methyl-d-aspartate and serotonin 1A (5-HT1A) receptors, and to antagonize G protein-coupled receptor 55, involved in neuroinflammation and neuronal excitability, which together may contribute to antipsychotic, cognitive-facilitating, and anxiolytic effects (Devinsky et al., 2014; Linge et al., 2016; Rodrigues Da Silva et al., 2020). Early clinical trials of cannabinoids in psychiatric disorders have highlighted CBD as a promising, but still unproven, treatment option for schizophrenia spectrum disorders and underscored the need for adequately powered randomized controlled trials (RCT; Black et al., 2019; Dyck et al., 2022).
Trials of CBD in individuals with psychosis have been listed among the most impactful research of 2025 (Webster and Healey, 2024).
Therefore, we conducted a systematic review and meta-analysis of published and unpublished trials of CBD in psychosis to summarize the current state of the evidence.
Methods
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Page et al., 2021). The protocol was registered with the International prospective register of systematic reviews (PROSPERO, CRD420250653493).
Selection strategy and data extraction
We searched PubMed, Scopus, Embase, PsycInfo, CINAHL, and clinicaltrials.gov until July 2025 using the search strings reported in the Supplemental Table 1.
We included any RCTs comparing CBD (any formulation/dose) to placebo or active control in adults with schizophrenia spectrum or related psychotic disorders. To maximize coverage of the available evidence, we also included unpublished RCTs with summary results posted on clinicaltrials.gov. Non-RCT designs were excluded. We performed deduplication by checking trial identification number and date/place of enrollment to make sure each sample rather than publication was the unit of interest.
Two reviewers independently screened studies and extracted data on study design, participant characteristics (diagnosis, illness stage, concomitant treatments), interventions (CBD dose, comparator), and outcomes of interest. The primary outcome was post-treatment psychotic symptom severity. Secondary outcomes were any other outcome reported by ⩾3 studies. Along these, we assessed the safety/tolerability of the treatment. For each outcome, we extracted data from the time point closest to the median duration across trials.
Statistical analysis
We conducted pairwise meta-analyses (CBD vs controls) with random-effects models (DerSimonian and Laird, 1986). Continuous outcomes were pooled as Hedges’ g standardized mean differences (SMDs) and dichotomous outcomes as odds ratios (ORs), each with 95% confidence intervals (95% CIs). Standard Q tests and the I2 statistic were used to assess between-study heterogeneity, considering values of I2 ⩾ 75% indicative of high heterogeneity. Subgroup comparisons were conducted on the primary outcome and were based on (1) published/unpublished trial status; (2) inclusion/exclusion of cannabis users in the sample; (3) psychosis stage; (4) timing of response assessment. Analyses used the meta package in R (Balduzzi et al., 2019; Posit Team, 2025). Statistical tests were two-sided and used a significance threshold of p-value < 0.05.
Evidence quality assessment
Bias risk in the included studies was independently assessed by two reviewers using the Cochrane risk of bias tool, and the GRADE tool was used to assess the overall certainty of evidence (Higgins et al., 2011; Schünemann et al., 2013). At the study level, overall risk of bias was classified as low (all domains low risk), moderate (⩾1 domain unclear risk and no domain high risk) or high (⩾1 domain high risk). Further information is available in the Supplemental Appendix.
Statement of ethics
This systematic review is exempt from ethics approval because it collected and synthesized data from previous studies in which ethical approval has already been obtained by the trial investigators at their respective local sites.
Results
Study characteristics
As shown in Figure 1, from 925 records identified in the primary search, 8 RCTs (6 published, 2 unpublished of which 1 terminated before completion based on a business decision of the Sponsor, without additional specifications) were included in the final selection (Boggs et al., 2018; Chesney et al., 2025; Jazz Pharmaceuticals, 2023; Köck et al., 2021; Leweke et al., 2012; McGuire et al., 2018; O’Neill et al., 2021; Ranganathan, 2022), accounting for 288 participants. Of these, 116 treated with CBD, 117 controls, and 55 participated in a crossover trial and contributed data to both conditions.
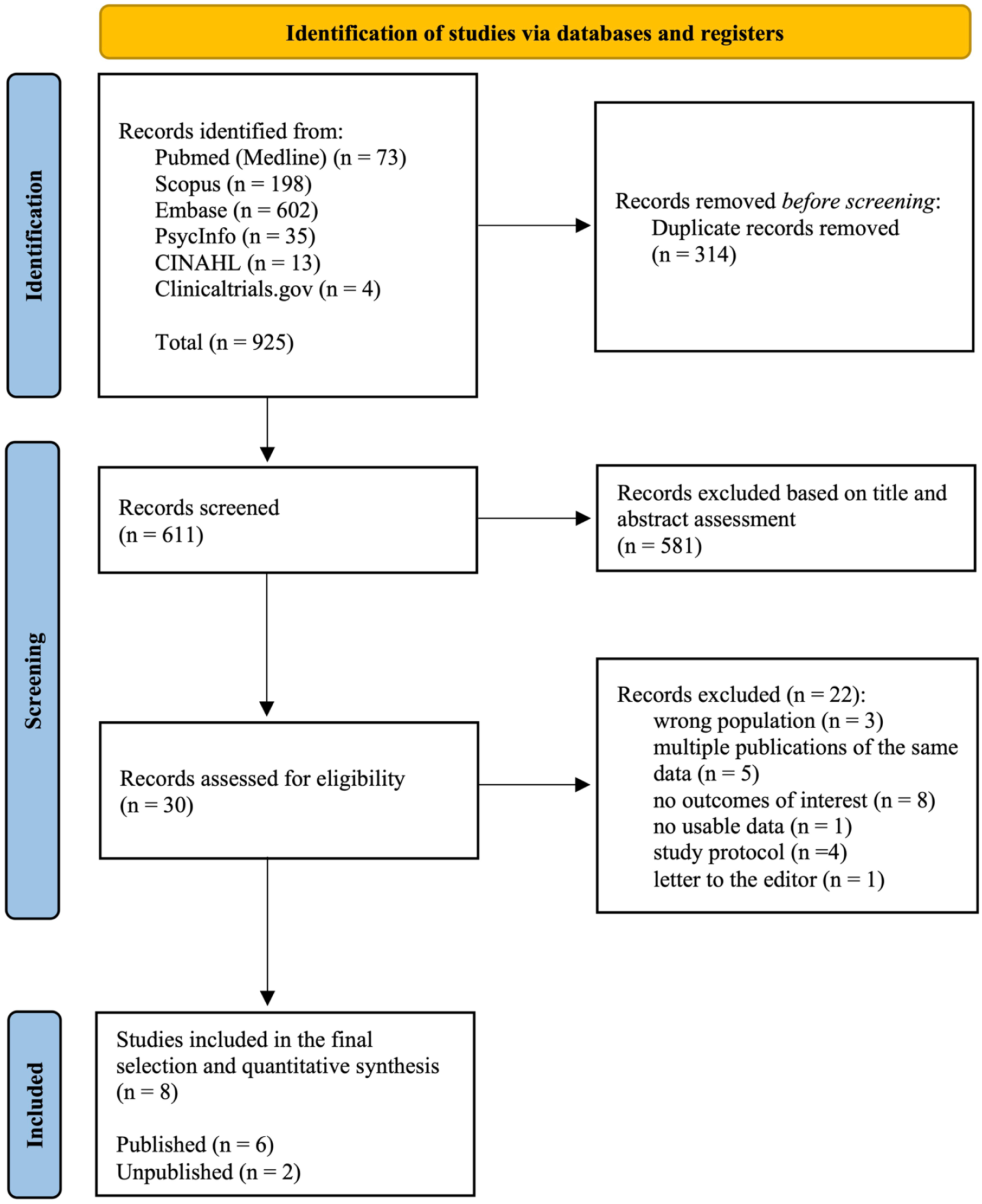
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
The full characteristics of the included studies are reported in Table 1.
Characteristics of the included studies.
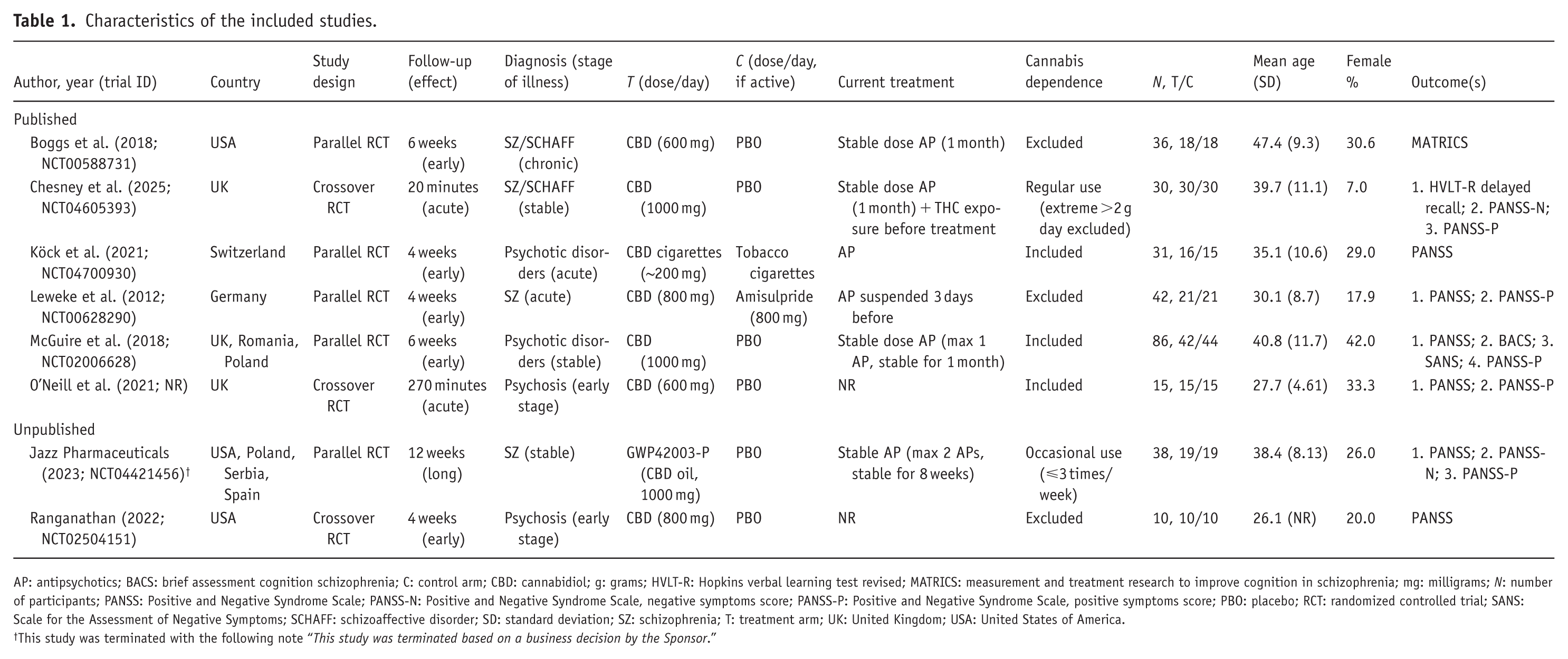
AP: antipsychotics; BACS: brief assessment cognition schizophrenia; C: control arm; CBD: cannabidiol; g: grams; HVLT-R: Hopkins verbal learning test revised; MATRICS: measurement and treatment research to improve cognition in schizophrenia; mg: milligrams; N: number of participants; PANSS: Positive and Negative Syndrome Scale; PANSS-N: Positive and Negative Syndrome Scale, negative symptoms score; PANSS-P: Positive and Negative Syndrome Scale, positive symptoms score; PBO: placebo; RCT: randomized controlled trial; SANS: Scale for the Assessment of Negative Symptoms; SCHAFF: schizoaffective disorder; SD: standard deviation; SZ: schizophrenia; T: treatment arm; UK: United Kingdom; USA: United States of America.
This study was terminated with the following note “This study was terminated based on a business decision by the Sponsor.”
Meta-analysis of efficacy
We did not find evidence supporting the efficacy of CBD for any outcome considered. The pooled effect size for psychosis symptom severity indicated a small effect magnitude (SMD = −0.194; 95% CI: −0.444 to 0.056). Given the low number of studies contributing to this estimate (n = 6), we performed a post-hoc power analysis to assess the ability of the meta-analysis to detect clinically relevant effects. Based on the observed standard error of the pooled estimate, the meta-analysis had approximately 65% power to detect a true effect of SMD = 0.30 and 88% power to detect SMD = 0.40, at a two-sided alpha of 0.05. The smallest absolute effect size that the present meta-analysis could have detected with 80% power and alpha 5% was approximately SMD = 0.358.
Concerning safety, one trial reported the death of one participant randomized to the CBD arm, however, the authors reported that CBD intoxication was ruled out as the cause of death (Köck et al., 2021). No statistically significant difference in tolerability was found between CBD and controls. The results are shown in Table 2 (see also Supplemental Figures 1–2 displaying the forest plot of tolerability assessment).
Results of meta-analysis of CBD efficacy and tolerability for individuals with psychosis.
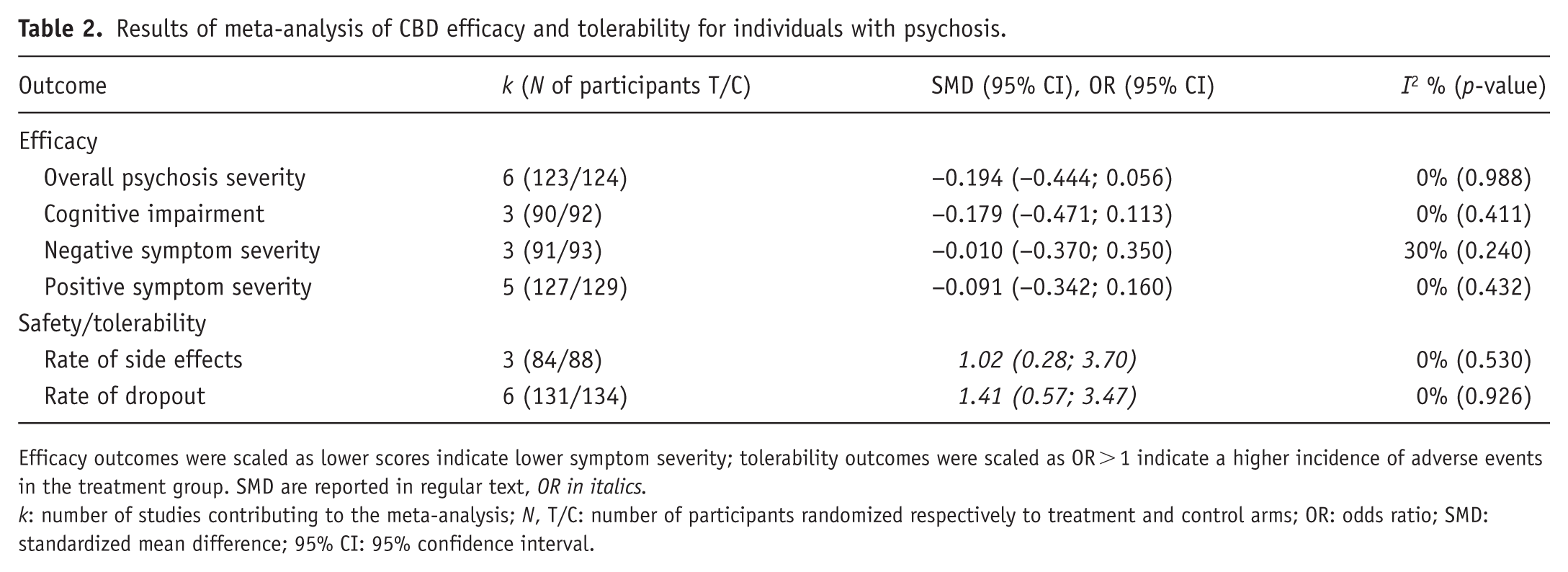
Efficacy outcomes were scaled as lower scores indicate lower symptom severity; tolerability outcomes were scaled as OR > 1 indicate a higher incidence of adverse events in the treatment group. SMD are reported in regular text, OR in italics.
k: number of studies contributing to the meta-analysis; N, T/C: number of participants randomized respectively to treatment and control arms; OR: odds ratio; SMD: standardized mean difference; 95% CI: 95% confidence interval.
Between-study heterogeneity estimated through I2 was low in all meta-analyses, ranging from 0% for psychosis severity to 39% for the rate of any adverse event. However, there were marked differences across the studies in terms of CBD dose, trial duration, participants’ illness stage, and cannabis use status. Subgroup meta-analyses on the primary outcome by trial duration showed similar pooled estimates and largely overlapping confidence intervals among studies assessing CBD acute effect (SMD: −0.200; 95% CI: −0.917 to 0.518, based on one study, duration 270 minutes), early effect (SMD: −0.182; 95% CI: −0.476 to 0.112, based on four studies, mean duration 4.5 weeks), and long-term effect (SMD: −0.248; 95% CI: −0.887 to 0.391, based on one study, duration 12 weeks). The results are presented in Figure 2.
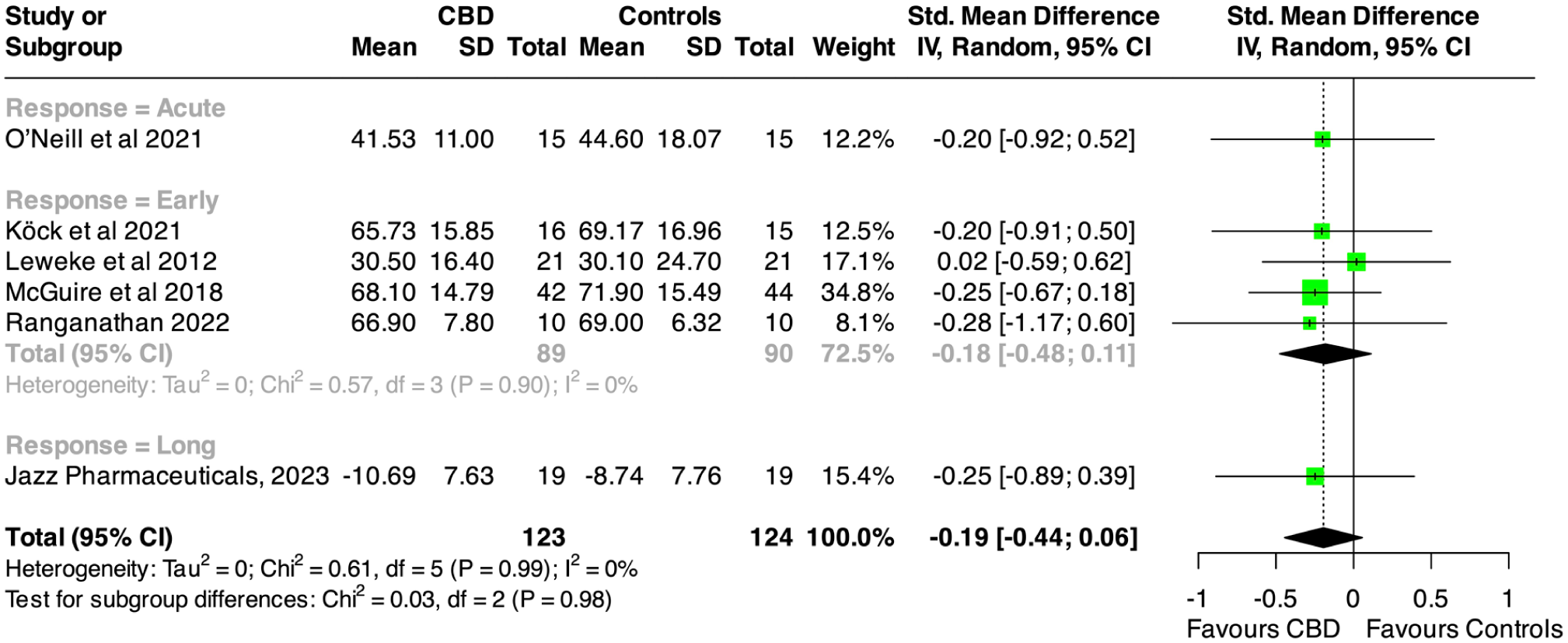
Forest plot of psychosis severity among CBD and control groups: overall and by response time.
Interestingly, trials including cannabis users showed numerically larger effect size favoring CBD, as displayed in Table 3.
Results of subgroup meta-analysis of CBD efficacy and tolerability for individuals with psychosis by cannabis use.
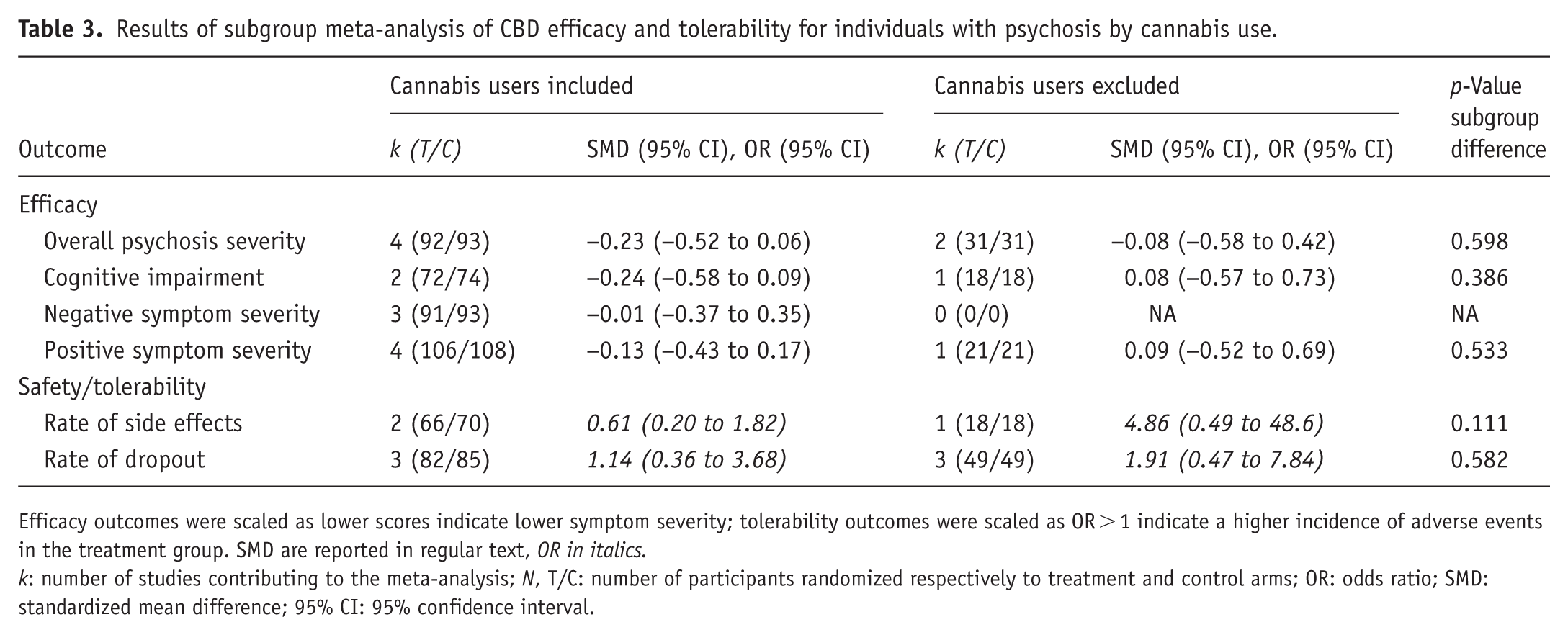
Efficacy outcomes were scaled as lower scores indicate lower symptom severity; tolerability outcomes were scaled as OR > 1 indicate a higher incidence of adverse events in the treatment group. SMD are reported in regular text, OR in italics.
k: number of studies contributing to the meta-analysis; N, T/C: number of participants randomized respectively to treatment and control arms; OR: odds ratio; SMD: standardized mean difference; 95% CI: 95% confidence interval.
Formal test for subgroup differences was not statistically significant for all comparisons, although these analyses were limited by the small number of studies available (see Supplemental Figures 3–5). Leave-one-out meta-analyses of efficacy on psychosis severity and tolerability measured as drop-out rate showed that by excluding the trial applying amisulpride as active comparator (Leweke et al., 2012) increased the pooled estimate favoring CBD efficacy, although the effect remained non-significant (SMD: −0.238; 95% CI: −0.513 to 0.037). The exclusion of any other single trial produced negligible changes in the pooled effect size (⩽20%, see Supplemental Table 2). Cumulative meta-analysis showed that the five most recent trials have not materially changed the pooled treatment effect (see Supplemental Figure 6).
At the individual-trial level, two studies (Leweke et al., 2012; McGuire et al., 2018) were rated as low risk of bias across all domains. By contrast, the remaining trials showed some concerns (unclear or high risk) in at least one domain, mostly related to blinding integrity, use of per-protocol rather than intention-to-treat analyses, and incomplete reporting, although this last assessment was largely influenced by the inclusion of a terminated trial. When these trial-level assessments were combined with imprecision and the small number and size of studies, the overall certainty of evidence for most outcomes was rated as low to very low according to GRADE (see Supplemental Figures 7–8 and Supplemental Table 3).
Discussion
In the current meta-analysis, CBD did not show greater effectiveness than placebo or amisulpride in improving clinical outcomes of psychotic disorders. The included study using amisulpride as an active comparator showed no difference in efficacy, but since it was not designed as a noninferiority trial, considerations about equivalence cannot be made. Moreover, no difference between CBD and controls emerged for tolerability, aligning with previous findings in other psychiatric populations (Black et al., 2019).
The precision of effect estimates was limited by the small number and size of available trials, reducing statistical power to detect small-to-moderate effects. Meta-analyses of established antipsychotics in schizophrenia spectrum disorders generally reported effect sizes for total symptom reduction versus placebo around 0.3–0.5, which are typically regarded as clinically meaningful in this population (Haddad and Correll, 2018; Huhn et al., 2019; Leucht et al., 2022; Schneider-Thoma et al., 2022). In contrast, our confidence interval, while excluding large effects, still included values in the lower part of this range (up to SMD of 0.444). Our post-hoc precision analysis suggested that the current data are reasonably informative against a large, robust antipsychotic-like effect of CBD on overall psychosis severity, but they do not definitively exclude smaller effects in the range of many adjunctive pharmacological treatments yielding SMD around 0.1–0.3 (Fond et al., 2023). This implies that the modest benefits of CBD cannot be definitively excluded based on the findings of this review. However, the pooled point estimates were consistently close to the null, suggesting that any true effect, if present, is likely small.
Overall, the current findings temper expectations regarding CBD as a therapeutic option for psychosis. While additional large, well-powered RCTs may be necessary to definitively rule in or out a clinically meaningful benefit, current evidence suggests that CBD is unlikely to yield a robust antipsychotic effect. Notably, larger, multicenter trials are currently underway—such as the Oxford Stratification and Treatment in Early Psychosis trials (University of Oxford, 2025)—and hopefully will provide higher-quality evidence to establish CBD role in psychosis treatment.
Although subgroup analyses were underpowered due to the limited number of included studies and did not identify statistically significant interactions, trials including participants with concurrent recreational cannabis use showed numerically larger effects of CBD. This observation may inform treatment personalization and may reflect a reduction in cannabis-related effects rather than a direct impact on psychosis itself (Freeman et al., 2020; Malik et al., 2025). Given the high prevalence of cannabis use among individuals with psychotic disorders and its association with relapse and poorer outcomes (Osborne et al., 2025), a preferential effect of CBD in patients with cannabis-related presentations would be clinically and mechanistically plausible. Future RCTs could therefore target patients whose psychosis is related to cannabis use, for instance by including biomarkers of recent cannabis use as stratification variables, to clarify whether CBD is particularly beneficial in this subgroup.
Finally, the pharmaceutical-grade CBD formulations tested in current trials are substantially more expensive than commonly used antipsychotics, with some health technology assessments suggesting up to ten-times higher drug acquisition costs (CADTH, 2024; Doshi et al., 2024). At the same time, cheaper CBD preparations are widely available in retail outlets and online, but these products usually differ in dose, purity, and regulation from trial formulations and cannot be assumed to be comparable. Although a formal assessment of the cost-effectiveness of CBD in psychosis was beyond the scope of the present review, economic evaluations of CBD in other psychiatric or chronic conditions have suggested potential benefits (Doucette et al., 2025; Erku et al., 2021). Future, well-designed clinical trials and health economic evaluations are needed to generate adequate clinical and cost-effectiveness evidence to assist in resource allocation.
Limitations
This meta-analysis is limited by the small number and size of trials, which reduces statistical power. The range of outcomes that could be assessed was also restricted and did not include other symptom domains where CBD might have effects, such as anxiety, depression, or sleep. Second, although statistical heterogeneity was low in all meta-analyses, there was still substantial variability between trials. Some studies were short-term or single-dose, which may not capture longer-term treatment effects, and two trials used non-placebo comparators (amisulpride and tobacco cigarettes). Single-dose designs and the trial comparing CBD with tobacco cigarettes challenged interpretation as conventional treatment outcome trials and contributed to increasing clinical heterogeneity in the current meta-analyses. Third, some items of risk of bias scored high or unclear, reflecting methodological limitations in the included studies. Fourth, our analyses were based on published and registry summary data rather than individual participant data. As a result, subgroup analyses (particularly those related to cannabis-use status) were performed at the study level and provide only a relatively raw, hypothesis-generating test of differential effects. Finally, the limited number of trials prevented formal assessment of publication bias and treatment moderators.
Conclusions
Until more robust data are available, CBD should be considered experimental for psychosis, as current pooling RCT evidence does not support its clinical use.
Supplemental Material
sj-docx-1-jop-10.1177_02698811261430501 – Supplemental material for Cannabidiol for psychotic disorders: A meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-jop-10.1177_02698811261430501 for Cannabidiol for psychotic disorders: A meta-analysis of randomized controlled trials by Mattia Marchi, Karim Rachedi, Alessia Bof, Giulio Mele, Luca Pingani, Silvia Ferrari and Gian Maria Galeazzi in Journal of Psychopharmacology
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
