Abstract
Background:
The continual emergence of novel synthetic cathinones poses significant public health concerns due to their unpredictable pharmacological profiles and potential for abuse. Among these, 4-F-3-Me-α-PVP—a newly identified analogue of α-PVP—has recently surfaced on the illicit drug market, yet its biological effects remain uncharacterized.
Aim:
To provide the first comprehensive pharmacological evaluation of 4-F-3-Me-α-PVP.
Methods:
In vitro transporter inhibition was assessed using HEK293 cells expressing human dopamine (DA), norepinephrine, and serotonin transporter (DAT, NET, or SERT). In male rodents, locomotor activity was measured after i.p. (mice) or s.c. (rats) administration. In vivo microdialysis in rats quantified extracellular DA in the nucleus accumbens. Rewarding and reinforcing effects were evaluated using conditioned place preference (CPP) in mice and intravenous self-administration (IVSA) in rats under fixed-ratio and progressive-ratio schedules.
Results:
In vitro assays revealed that 4-F-3-Me-α-PVP acts as a potent DAT and NET inhibitor, with additional, though weaker, activity at SERT. In vivo, 4-F-3-Me-α-PVP significantly increased locomotor activity in male rodents (10 and 30 mg/kg in mice; 3 mg/kg in rats). Importantly, 4-F-3-Me-α-PVP also increased extracellular DA levels in the rat nucleus accumbens (3 mg/kg, s.c.), pointing to its potential for abuse. Behavioral assays further demonstrated rewarding and reinforcing effects in rodents, with significant CPP in mice at all doses tested and dose-dependent IVSA in rats observed under both fixed-ratio and progressive-ratio schedules.
Conclusions:
Collectively, these findings indicate that 4-F-3-Me-α-PVP possesses substantial psychostimulant and abuse-related effects in rodents, underscoring the need for regulatory vigilance and continued investigation into emerging synthetic cathinones.
Introduction
Synthetic cathinones are a class of novel psychoactive substances that have become increasingly popular as recreational drugs in recent years (United Nations Office on Drugs and Crime (UNODC), 2024). These substances are chemically derived from the naturally occurring stimulant cathinone, which is the primary active constituent of the khat plant. Moreover, these compounds are structurally similar to psychostimulants such as amphetamine, methamphetamine and 3,4-methylenedioxymethamphetamine (MDMA; Sitte and Freissmuth, 2015).
Among synthetic cathinones, one prominent subgroup includes pyrovalerone analogs, such as 3,4-methylenedioxypyrovalerone (MDPV) and α-pyrrolidinopentiophenone (α-PVP). Structurally, MDPV contains a methylenedioxy group, similar to MDMA, while α-PVP lacks this group, similar to methamphetamine. Both compounds, MDPV and α-PVP, were first synthesized in the mid-late 1960s as potential central nervous system stimulants, though neither substance ever reached the pharmaceutical market (Koppe et al., 1969; Seeger, 1966). Nearly 40 years later, MDPV emerged on the European illicit drug market, rapidly gaining popularity (EMCDDA-Europol, 2014; Spiller et al., 2011), leading to a sharp rise in intoxication cases and even fatalities (Beck et al., 2015; Marinetti and Antonides, 2013; Spiller et al., 2011; Wright et al., 2013). Shortly after, α-PVP—often sold under the street name “flakka”—rose to prominence in the early 2010s, drawing media attention for reports of severe agitation, paranoia, bizarre, and sometimes, violent behavior as well as fatal intoxications (Beck et al., 2016; EMCDDA-Europol, 2015; Wright and Harris, 2016).
Both MDPV and α-PVP have a typical psychostimulant mode of action involving the inhibition of the reuptake of dopamine (DA) and norepinephrine (NE), leading to an increase in their synaptic concentrations (Baumann et al., 2013; Glennon and Young, 2016). Recent structural and functional studies have highlighted that pyrrolidinophenones, such as MDPV and α-PVP, distinguish themselves from other stimulants by their interaction with the DAT. Specifically, molecular modeling have identified key non-conserved residues in the human DAT binding pocket that dictate the high selectivity and potency of these synthetic cathinones compared to classical stimulants like cocaine (Steele et al., 2021). Furthermore, the pyrrolidine ring contributes to a distinct binding orientation that results in a more efficient inhibition of DA uptake (Duart-Castells et al., 2021; Steele et al., 2021). Moreover, some studies have also demonstrated their abuse potential in preclinical models of addiction (Aarde et al., 2015; Baumann et al., 2013; Spiller et al., 2011). Particularly, α-PVP has been shown to induce significant behavioral changes in rodent models, including hyperactivity, stereotypies, increased locomotor activity, and agitation, closely resembling the effects seen with other potent stimulants (Giannotti et al., 2017; Nelson et al., 2019; Taffe et al., 2021).
4-F-3-Me-α-PVP, also referred to as “MFPVP”, is an α-PVP analog that has recently emerged in the illicit drug market (Hobbs et al., 2022; Kuropka et al., 2023; NMS Labs, 2020). In fact, the first fatality solely attributed to this compound was reported in 2022 (Hobbs et al., 2022). Given that 4-F-3-Me-α-PVP is a hybrid of 4-F-α-PVP and 3-Me-α-PVP, insights from these parent compounds are also particularly relevant. Previous studies, including work from our research group, have shown that 4-F-α-PVP induces hyperlocomotion and rewarding effects in mice, along with potent inhibition of DA and NE uptake but weaker effects at 5-HT transporter (SERT; Meltzer et al., 2006; Nadal-Gratacós et al., 2022; Wojcieszak et al., 2020). In addition, 3-Me-α-PVP has also been characterized as a highly potent and selective inhibitor of DA and NE transporters (DAT and NET, respectively; Meltzer et al., 2006). In this sense, the DAT/SERT inhibition ratio is a common pharmacological benchmark used to predict the behavioral profile of synthetic cathinones. High DAT/SERT ratios typically correlate with pure psychostimulant effects and high abuse liability, whereas lower ratios—due to increased SERT activity—often shift the profile towards entactogenic effects, similar to MDMA (Baumann et al., 2018; Simmler et al., 2013).
The history of this class of compounds illustrates the cat-and-mouse dynamic between drug manufacturers and regulators, as evidenced by the continual banning of molecules that differ only by minor structural modifications (i.e., MDPV and α-PVP; DEA, 2015, 2022). Even slight chemical alterations can enable new derivatives to circumvent existing drug laws, posing an ongoing challenge for forensic toxicologists and public health authorities. Thus, further research into the pharmacology, toxicity, and behavioral effects of these new substances is crucial to better understand their risks and guide harm-reduction strategies. To our knowledge, investigations of 4-F-3-Me-α-PVP have so far been limited to its physical and chemical properties (Rojkiewicz et al., 2023; Wang et al., 2023), while its pharmacological and toxicological effects remain unexamined. Therefore, the aims of this study were: (i) to assess in vitro its potential as a DA, NE, and 5-HT uptake inhibitor; (ii) to investigate its psychostimulant and rewarding effects in mice, and (iii) its reinforcing effects and its impact on extracellular levels of DA and its metabolites in the nucleus accumbens (NAcc) of rats.
Methods
Subjects
All animal care and experimental protocols were approved by the Animal Ethics Committee of the University of Barcelona, supervised by the Autonomous Government of Catalonia, COMETHEA or Stockholms Norra djurförsöksetiska nämn following the directives of the Swedish Animal Welfare Act 1988: 534). The procedures adhered to the guidelines of the European Community Council Directive 2010/63/EU, as amended by Regulation (EU) 2019/1010, and complied with the ARRIVE guidelines for reporting animal experiments (Percie du Sert et al., 2020). Male Swiss CD-1 mice (Janvier, Le Genest, France), aged 6–8 weeks and weighing 30–35 g, were used for horizontal locomotor activity (HLA) and conditioned place preference (CPP) experiments. Male Sprague-Dawley rats (Janvier, Le Genest, France) weighing 300–400 g (8–12 weeks old), were used for microdialysis and self-administration experiments. All animals were housed in temperature-controlled conditions (22 ± 1°C) under a 12-hour light/dark cycle with free access to food and water (standard laboratory diet, Panlab SL, Barcelona, Spain). Mice and rats were randomly assigned to experimental groups, and all efforts were made to minimize their use and suffering.
Drugs and materials
4-F-3-Me-α-PVP was synthesized in racemic form as a hydrochloride salt and identified as described in Supplemental Material. Solutions for injection were prepared daily in isotonic saline solution. Cocaine was provided by the Spanish National Institute of Toxicology. [3H]1-Methyl-4-phenylpyridinium ([3H]MPP+) and [3H]5-HT were purchased from Revvity Inc. (Boston, MA, USA). Cell culture media (Dulbecco’s Modified Eagle’s medium (DMEM) high-glucose), DA hydrochloride, 3,4-dihydroxyphenylacetic acid (DOPAC), and 4-hydroxy-3-methoxyphenylacetic acid (HVA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). The stable isotope deuterium labeled DA-d4, DOPAC-d5, and HVA-d3 were purchased from Toronto Research Chemicals (Toronto, ON, Canada). Acetonitrile (ACN, Liquid Chromatography-Mass Spectrometry (LC-MS) grade) and methanol (MeOH, LC-MS grade) were purchased from Thermo Fisher Scientific (Gothenburg, Sweden). All other reagents were of analytical grade and purchased from several commercial sources.
Uptake inhibition assays in HEK293 cells
Cell culture
Human embryonic kidney cells (HEK293) stably transfected with the human isoforms of the dopamine, norepinephrine, and serotonin transporters (DAT, NET, and SERT, respectively) were used for uptake inhibition assays. Cells were maintained in DMEM supplemented with heat-inactivated 10% fetal bovine serum, 1 µg/mL streptomycin, 100 IU/mL penicillin, and geneticin (G418; 50 µg/mL) at a subconfluent state and cultured in a humidified atmosphere (5% CO2, 37°C). For uptake inhibition assays, cells were seeded 24 hours prior to the experiment at a density of 0.36 million cells/well onto 96-well plates previously coated with poly-D-lysine.
Uptake inhibition assays
On the day of the experiment, the culture media were removed and replaced immediately with 200 µL per well of Krebs-HEPES buffer (KHB; 10 mM HEPES, 120 mM NaCl, 3 mM KCl, 2 mM CaCl2·2H2O, 2 mM MgCl2·6H2O, 20 mM D-glucose; pH 7.3). Cells underwent a preincubation step, where they were exposed to varying concentrations of the test drug in KHB (50 µL per well) for 5 minutes to achieve equilibrium. After preincubation, the solution was removed, and cells were incubated with either 0.02 µM [3H]MPP+ for 3 minutes (DAT and NET) or 0.1 µM [3H]5-HT for 1 minute (SERT) in the presence of different drug concentrations in KHB.
Following incubation, the tritiated substrate was removed, and cells were washed with ice-cold KHB and lysed using 1% sodium dodecyl sulfate. The lysate was mixed with scintillation fluid, and radioactivity was measured using a beta-scintillation counter (PerkinElmer, Waltham, MA, USA). Non-specific uptake was determined by the presence of 100 µM cocaine for DAT or 3 µM paroxetine for SERT and was subtracted from all values. Non-specific uptake accounted for less than 10% of total uptake. Uptake without the drug was normalized to 100%, and uptake in the presence of varying drug concentrations was expressed as a percentage of the control.
Horizontal locomotor activity
Mice
A habituation phase was conducted to minimize the novelty and stress associated with handling and injection. During this phase, all mice received an intraperitoneal (i.p.) injection of saline and were placed in a black Plexiglass open-field arena (25 cm × 25 cm × 40 cm), with sufficient height to prevent escape, for 20 minutes. The experiment was performed under low-light conditions. On the test day, the animals received an i.p. injection (saline at 5 mL/kg or 4-F-3-Me-α-PVP at 3, 10, or 30 mg/kg) and were immediately placed in the open-field arena for 60 minutes. The total traveled distance was measured using a specific tracking software (Smart 3.0, Panlab, Barcelona, Spain).
Rats
In rats, locomotor activity was monitored concurrently with brain microdialysis. Each rat was placed in a transparent polycarbonate cage (48 cm × 38 cm, height 40 cm) and allowed to acclimate for 60 minutes. Following habituation, the rats received subcutaneous (s.c.) administrations of 4-F-3-Me-α-PVP (3 mg/kg) or vehicle and were then returned to their respective cages. HLA was video-recorded using the Smart 3.0 computerized video system (Panlab, Harvard Apparatus, USA), and the distance traveled in 5-minute intervals was measured during 120 minutes. Upon completion of the experiments, the rats were euthanized with an overdose of isoflurane, followed by cervical dislocation.
Thigmotaxis (open-field test)
Thigmotaxis, the tendency to remain near the walls of the arena, was used as an indicator of anxiety-like behavior, with increased time in the periphery reflecting heightened anxiety (Simon et al., 1994). For this experiment, mice received an acute i.p. administration of saline or 4-F-3-Me-α-PVP (3, 10, or 30 mg/kg) and were immediately placed in the center of an open-field arena (25 × 25 × 40). The arena was subdivided into two areas, a center (8 cm × 8 cm) and its periphery, using a specific tracking software (Smart 3.0, Panlab, Barcelona, Spain), and the time spent, in seconds, in each area was monitored for 60 minutes.
Conditioned place preference
The rewarding effects of 4-F-3-Me-α-PVP were investigated using the CPP paradigm in mice. This approach employed a two-compartment chamber separated by a corridor, with distinct visual and tactile cues in each compartment. The CPP procedure consisted of three phases: Preconditioning, where mice had free access to both compartments for 15 minutes, and their time spent in each compartment was recorded using a tracking software (Smart 3-0, Panlab, Barcelona, Spain). Conditioning, with two sessions performed per day separated by a 5-hour period, where mice received an intraperitoneal injection of the drug (saline at 5 mL/kg or 4-F-3-Me-α-PVP at 3, 10, or 30 mg/kg) and were placed in the drug-paired compartment for 20 minutes, while on alternative sessions they received saline and were placed in the other compartment for 20 minutes; and post-conditioning, where the same conditions as the preconditioning phase were applied. A preference score, expressed in seconds, was calculated as the difference between the time spent in the drug-paired compartment during the post-conditioning test and the preconditioning phase. Mice with an initial preference for one of the compartments (>70% preference) in the preconditioning test were excluded from the analysis.
Microdialysis
Microdialysis experiments were performed on awake rats between 8 AM and 6 PM, adhering to a previously described protocol (Nadal-Gratacós et al., 2025).
Surgery
Microdialysis experiments were conducted under aseptic conditions following established guidelines for rodent surgery and analgesia (Flecknell, 2018; Hoogstraten-Miller and Brown, 2008). Rats received pre-operative analgesia 30–36 minutes prior to surgery (buprenorphine 0.05 mg/kg s.c. and carprofen 5 mg/kg s.c.) and were anesthetized with isoflurane using a Univentor 40 anesthesia unit (AgnThos, Lidingö, Sweden). Surgery was performed in a stereotaxic frame, with a guide cannula implanted into the NAcc based on Paxinos and Watson’s atlas (Paxinos and Watson, 2014), as described in (Nadal-Gratacós et al., 2025). Body temperature was maintained at 37°C using a CMA/105 temperature controller (CMA Microdialysis, Stockholm, Sweden). Post-surgical care included temperature maintenance, ocular protection, and analgesia, with a 7-day recovery period before experiments.
Microdialysis experiments
On the day of the experiment, awake rats were implanted with microdialysis probes and connected to the perfusion system. Two CMA/12 probes (0.5 mm o.d., 2 mm membrane length, 20 kDa cut-off) were inserted into the guide cannulae targeting the NAcc and perfused with artificial cerebrospinal fluid (aCSF). The aCSF solution (148 mM NaCl, 4 mM KCl, 0.8 mM MgCl2, 1.4 mM CaCl2, 1.2 mM Na2HPO4, and 0.3 mM NaH2PO4, pH 7.2) was sterilized through a 0.2 μm disc filter and loaded into a 1 ml gas-tight glass syringe (CMA Microdialysis). Each rat was placed into a Rotating Animal Cage System (RACS, Microbiotech/se, Årsta, Sweden), which enables a swivel-free connection between the syringe pump and fraction collector. After a 120-minute stabilization period, three baseline samples were collected to determine extracellular neurotransmitter levels. The rats then received a s.c. injection of either saline (5 mL/kg) or 4-F-3-Me-α-PVP (3 mg/kg), followed by an additional 4 hours of sample collection. The dose and administration method were based on prior microdialysis research on synthetic cathinones conducted in our laboratory (Kehr et al., 2011). Moreover, s.c. administration was preferred in this specie to reduce stress and avoid potential displacement or interference with the microdialysis tethering system during drug delivery.
Quantification of neurotransmitters and metabolites by UHPLC-MS/MS
The quantification of neurotransmitter and metabolites levels was carried out as previously described (Nadal-Gratacós et al., 2025). In short, DA, DOPAC, and HVA levels in microdialysates were analyzed using an ultra high-performance liquid chromatography with electrospray tandem mass spectroscopy (UHPLC-MS/MS) after benzoyl chloride derivatization. The system included an EVOQ Elite triple quadrupole mass spectrometer with electrospray ionization in positive mode, coupled with an Elute HTG UHPLC pump and an Elute column oven (all from Bruker Daltonik GmbH, Bremen, Germany). For separation, a Cortecs UHPLC C18 column (100 mm × 2.1 mm, 1.6 μm, Waters, USA) was used, with a gradient mobile phase of ammonium formate (10 mM) and formic acid (0.15% v/v) in water (mobile phase A) and acetonitrile (mobile phase B). For specific source parameters, mobile phase gradient and flow rate see (Nadal-Gratacós et al., 2025). Samples were injected via a PAL autosampler and maintained at 6°C. The system was operated using Compass 2.0/HyStar 4.0 software (Bruker, USA), and data acquisition and processing were performed with TASQ 2.2 (Bruker). Nitrogen and air for the mass spectrometer were supplied by a Genius 3045 nitrogen/air generator (Peak Scientific Instruments, Inchinnan, Scotland, UK). Samples were processed with internal standards, derivatized, centrifuged, and injected into the UHPLC-MS/MS system for quantification. Calibration curves ranged from 0.1 to 1000 nM.
Self-administration
Catheter implantation
Rats were anesthetized with isoflurane (5% for induction, 2.5% for maintenance) in O2 and administered ketoprofen (2.5 mg/kg, s.c.) for analgesia before undergoing surgical catheterization of the right jugular vein for 4-F-3-Me-α-PVP self-administration. To maintain body temperature, they were placed on a heating pad during the procedure. A custom-made silastic catheter was inserted into the jugular vein, with its distal end routed to the back between the scapulae. Following surgery, rats were given a 7-day recovery period and received daily flushing with 0.1 mL of sterile saline (0.9%) containing heparin (100 IU/mL) to prevent catheter occlusion.
Self-administration experiment
Self-administration experiments were conducted in Imetronic experimental chambers equipped with nose-pokes as operanda (Imetronic, Pessac, France). The dose range for self-administration (40–320 µg/kg/infusion) was determined based on the internal pharmacological consistency of the study. Specifically, the potency of 4-F-3-Me-α-PVP in the locomotor activity assay and its DAT inhibition profile (IC50) were found to be comparable to those of α-pyrrolidino-2-phenylacetophenone (α-D2PV) and 3-fluoro-N-ethyl-buphedrone (3-F-NEB), studies in which the same dose range was used (Nadal-Gratacós et al., 2025; Pain et al., 2026). Rats were first allowed to self-administer 4-F-3-Me-α-PVP at a dose of 160 µg/kg/injection for nine 2-hour sessions under a fixed-ratio 1 (FR1) schedule, where each nose-poke resulted in one drug infusion. On the 10th session, the reinforcing efficacy of this dose was assessed using a progressive ratio (PR) schedule, where the required number of nose-pokes to get a drug infusion increased exponentially according to the equation: response ratio = (5e(0.2 × reinforcer number)) − 5, rounded to the nearest integer. The required number of responses followed the sequence: 1, 2, 4, 6, 9, 12, 15, 20, 25, 32, 40, 50, 62, 77, 95, 118, 145, 178, 219, 268, 328, 402, 492, 603, and 737 (Richardson and Roberts, 1996). The PR session ended if a rat failed to complete a step within 1 hour or after 3 hours of testing. Afterward, the dose was changed, and rats were allowed to self-administer 4-F-3-Me-α-PVP at the new dose for 3–5 days under FR1, followed by another PR session to evaluate reinforcing efficacy at the new dose. A total of four-unit doses were tested: 40, 80, 160, and 320 μg/kg/injection. The dose-response characterization was conducted using a fixed-order sequence (160, 80, 40, and 320 µg/kg/infusion). This descending-then-ascending sequence was designed to prioritize the acquisition of self-administration behavior at a dose (160 µg/kg) with established reinforcing efficacy, thereby providing a stable behavioral baseline for subsequent testing of lower (40–80 µg/kg) and higher (320 µg/kg) doses. Dose-response relationships were established by averaging the last 3 days of FR1 responding and the value obtained in the corresponding PR session, at which point behavior had stabilized, thus minimizing potential carryover effects from the previous dose.
Data and statistical analysis
Nonlinear regression was applied to fit the plotted competition curves, with the data best fitted to a sigmoidal dose-response curve to obtain an IC50 value. To assess the effects of 4-F-3-Me-α-PVP on HLA and thigmotaxis, one-way or two-way analysis of varianc (ANOVA) of repeated measures was conducted, followed by Tukey’s post hoc test if the F-value was significant. Statistical differences in the CPP experiment were determined using Kruskall–Wallis followed by Dunn’s test. The comparison of mean basal levels of DA, DOPAC, and HVA in the control and treated groups was performed using a two-way ANOVA mixed-effects model followed by Tukey’s multiple comparison test. Differences in relative Area Under the Curve (AUC) (0–240 min) values between groups were analyzed using an unpaired Student’s t-test. For HLA in rats, a two-way repeated-measures ANOVA was applied, followed by Tukey’s multiple comparison test. Acquisition of 4-F-3-Me-α-PVP self-administration was analyzed using a two-way ANOVA of repeated-measures, with session and active/inactive nose-poke as factors, followed by Tukey’s multiple comparison test. Dose-response data under the FR1 schedule (number of responses and total intake) and the PR schedule (number of responses and injections and last ratio completed) were analyzed using a mixed-effects model of repeated measures, followed by Tukey’s multiple comparison test. For all experiments, data are presented as mean ± SEM. The α error probability was set at 0.05 (p < 0.05). The sample size for each experiment is provided in the corresponding figure legends. All statistical calculations were performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA).
Results
4-F-3-Me-α-PVP has high DAT/SERT inhibition ratios
The IC50 values obtained in the monoamine uptake inhibition assay, the corresponding DAT/SERT ratios are presented in Table 1, and concentration-response curves are depicted in Figure 1. Data for α-PVP and cocaine, obtained in previous experiments performed in our lab (Nadal-Gratacos et al., 2023; Nadal-Gratacós et al., 2025), are also shown for comparison. 4-F-3-Me-α-PVP exhibited similar potency in inhibiting DAT but showed greater potency in inhibiting SERT compared to α-PVP. This led to a lower DAT/SERT ratio than α-PVP, although it remained significantly higher than that observed for cocaine. Regarding NET, 4-F-3-Me-α-PVP demonstrated a lower inhibitory potential for this transporter compared to α-PVP, but higher than that of cocaine.
Monoamine uptake inhibition at hDAT, hNET and hSERT of 4-F-3-Me-α-PVP, α-PVP and cocaine. Values are IC50 given as µM (mean ± SEM). hDAT/hSERT ratio = 1/DAT IC50: 1/SERT IC50.
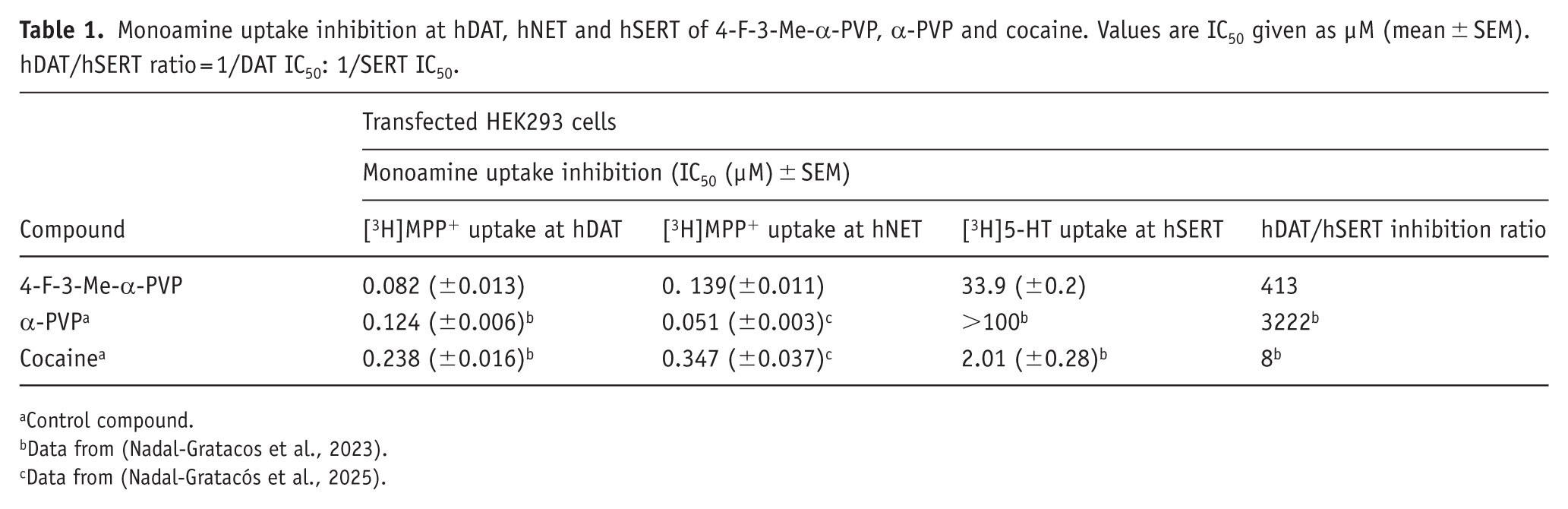
Control compound.
Data from (Nadal-Gratacos et al., 2023).
Data from (Nadal-Gratacós et al., 2025).
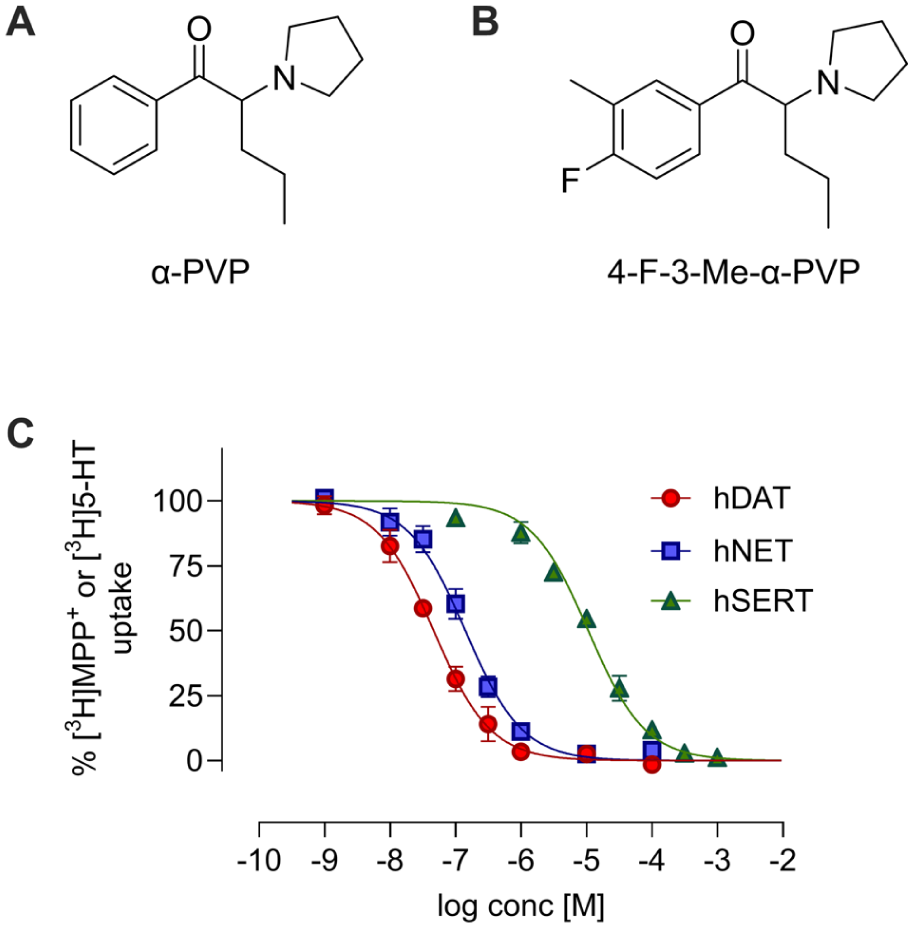
Chemical structure of α-PVP (a) and 4-F-3-Me-α-PVP (b). Concentration-effect curves of 4-F-3-Me-α-PVP on [3H]MPP+ uptake at DAT and NET, and [3H]5-HT uptake at SERT (c). Data are expressed as a percentage of control uptake (mean ± SEM) of >3 independent experiments performed per triplicate.
Dose-dependent effects of 4-F-3-Me-α-PVP on locomotor activity in mice
4-F-3-Me-α-PVP increased HLA in a dose-dependent manner in mice, as shown in Figure 2(a). One-way ANOVA of the total distance traveled revealed a significant effect of the variable Dose (F(3, 52) = 43.97; p < 0.001). A significant increase in HLA was observed after an acute i.p. injection of 10 and 30 mg/kg compared to the saline group, but not at the lowest dose tested (3 mg/kg). HLA time courses are presented in Figure 2(b). Two-way ANOVA of repeated measures yielded the following results: Dose (F(3, 52) = 43.97; p < 0.001), Time (F(11, 572) = 129.1; p < 0.001) and Interaction variable (F(33, 572) = 8.568; p < 0.001). HLA profile showed a rapid onset of action (5–10 minutes) following i.p. injections of 10 and 30 mg/kg of 4-F-3-Me-α-PVP. Additionally, at the medium dose tested, the enhanced locomotor activity lasted for 60 minutes. Regarding thigmotaxis, none of the tested doses induced this behavior when compared to the saline group, as shown in Figure 2(c). Statistical analysis using one-way ANOVA revealed a significant effect of the variable Dose (F(3, 46) = 6.851; p < 0.001). Notably, animals that received an acute administration of 30 mg/kg spent significantly less time in the center of the arena compared to those that received 3 or 10 mg/kg of 4-F-3-Me-α-PVP (Figure 2(c)), though this reduction was not significant when compared to the saline group. Figure 2(d) illustrates the tracking of ambulation during 10 minutes in one representative mouse from each experimental group.
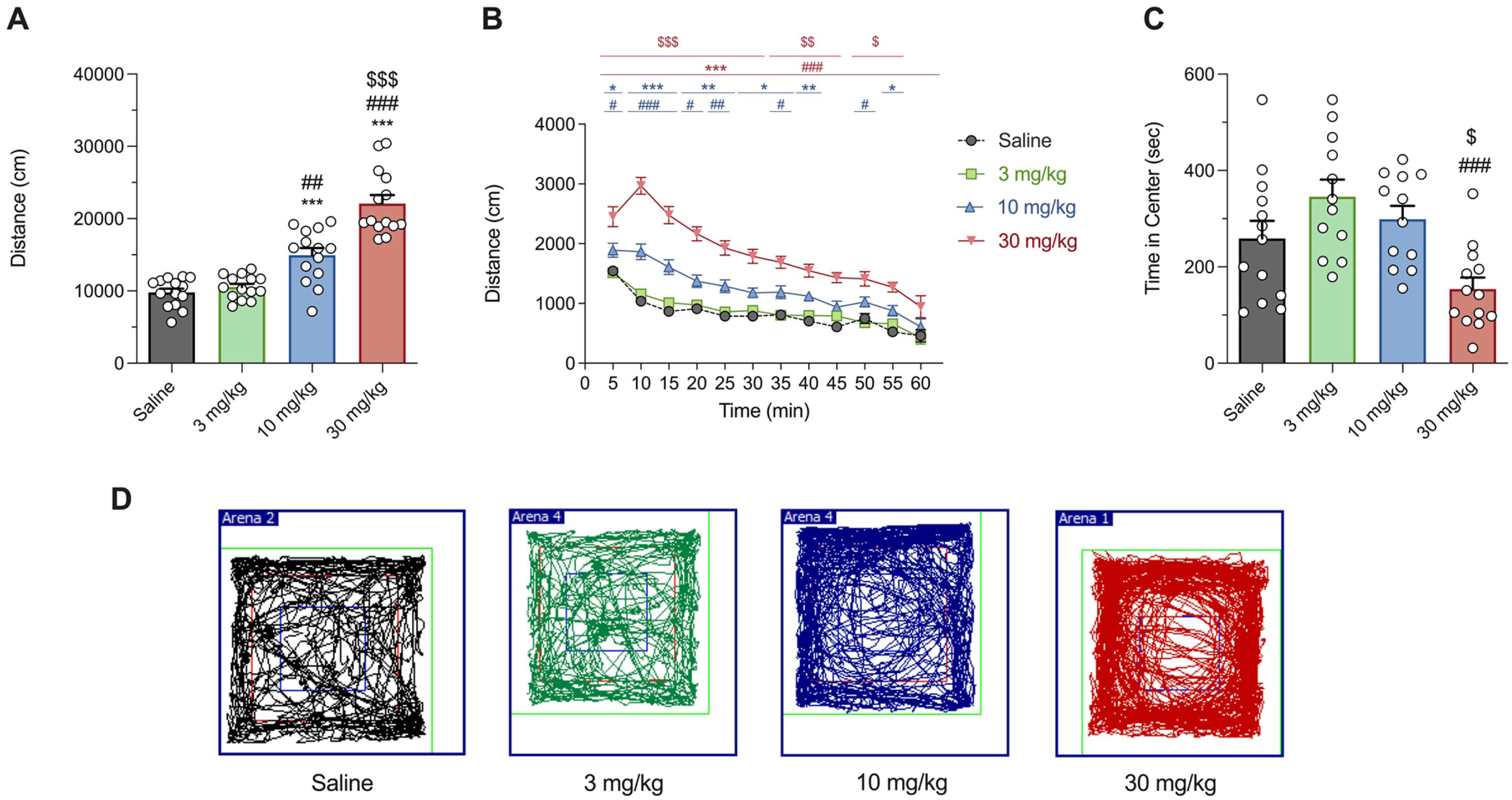
Effect of 4-F-3-Me-α-PVP on cumulative HLA in mice (a). Tukey’s multiple comparison test: Bars represent mean ± SEM of total distance (cm) traveled in 60 minutes. n = 14/group. Tukey’s multiple comparison test: ***p < 0.001 versus saline, ##p < 0.01 and ###p < 0.001 versus 3 mg/kg, and $$$p < 0.001 versus 10 mg/kg. Time course profile of HLA induced by 4-F-3-Me-α-PVP in mice (b). Each time point represents the mean ± SEM of the distance (in cm) traveled in 5 min blocks. n = 14/group. Tukey’s multiple comparison test: *p < 0.05, **p < 0.01, and ***p < 0.001 vs saline, #p < 0.05, ##p < 0.01, and ###p < 0.001 versus 3 mg/kg, and $p < 0.05, $$p < 0.01, and $$$p < 0.001 versus 10 mg/kg. Effects of 4-F-3-Me-α-PVP on thigmotaxis (c). Bars represent mean ± SEM of time in the center (seconds). n = 12–13/group. Tukey’s multiple comparison test: ###p < 0.001 versus 3 mg/kg and $p < 0.05 versus 10 mg/kg. Tracking of the ambulation for 10 minutes of one representative mouse for each experimental group (d). The colored squares indicate the regions used for the analysis of center and peripheral zones, with the blue square corresponding to the center area.
Acute administration of 4-F-3-Me-α-PVP enhances extracellular DA while reducing its metabolite levels in the rat nucleus accumbens
In contrast with the results observed in mice, a single s.c. administration of 3 mg/kg of 4-F-3-Me-α-PVP in rats resulted in a statistically significant increase in locomotion (t(10) = 5.769; p < 0.001), as depicted in Figure 3(a) and (b). The effects of 4-F-3-Me-α-PVP on locomotor activation of habituated rats, measured as distance travelled in 5-min bins is shown in Figure 3(a). Two-way ANOVA of repeated measures yielded the following results: Dose (F(1, 10) = 30.22; p < 0.001), Time (F(3.840, 38.40) = 7.917; p < 0.001), Interaction (F(35, 350) = 9.384; p < 0.001). Since 3 mg/kg was enough to induce hyperlocomotion in rats, this dose was deemed appropriate to perform microdialysis experiments. Our results demonstrate that acute administration of 4-F-3-Me-α-PVP (3 mg/kg, s.c.) is able to induce a rapid increase in extracellular DA levels in the NAcc (Figure 3(c) and (d)). Two-way ANOVA mixed-effects model of repeated measures yielded the following results: DA: time: F(1.797, 17.71) = 9.521, p < 0.01; treatment: F(1, 10) = 12.41, p < 0.01; and interaction variable: F(14, 138) = 9.506, p < 0.001. Subsequent Tukey’s multiple comparison test revealed a significant difference in the extracellular levels of accumbal DA in rats treated with 4-F-3-Me-α-PVP compared to saline-treated rats 20–160 minutes after administration. Moreover, when analyzing the AUC relative to the saline group, Student’s t-test revealed a significant increase in extracellular accumbal DA levels in rats treated with 4-F-3-Me-α-PVP compared to the control group (t(10) = 3.604, p < 0.001). On the other hand, a decrease in extracellular DA metabolites was also seen following 4-F-3-Me-α-PVP administration (Figure 3(e)–(h)). Two-way ANOVA mixed effects model of repeated measures yielded the following results: DOPAC: time: F(5.002, 49.31) = 1.689, p > 0.05; treatment: F(1, 10) = 10.82, p < 0.01; and interaction variable: F(14, 138) = 2.981, p < 0.001; HVA: time: F(4.246, 42.46) = 2.463, p > 0.05; treatment: F(1, 10) = 23.78, p < 0.001; interaction: F(14, 140) = 6.112, p < 0.001. Particularly, subsequent Tukey’s post hoc test revealed a decrease in extracellular accumbal DOPAC 40 minutes after administration of 4-F-3-Me-α-PVP, with decreased levels still apparent 200 minutes after administration. Regarding HVA, decreased levels were apparent 60 minutes after administration, and remained decreased even 220 minutes post-administration. In addition, Student’s t-test revealed a significant decrease in the AUC relative to the saline group of both accumbal DOPAC and HVA of 4-F-3-Me-α-PVP-treaded rats compared to the saline-treated control group (AUC DOPAC: t(10) = 2.518, p < 0.05; AUC HVA: t(10) = 4.933, p < 0.001).
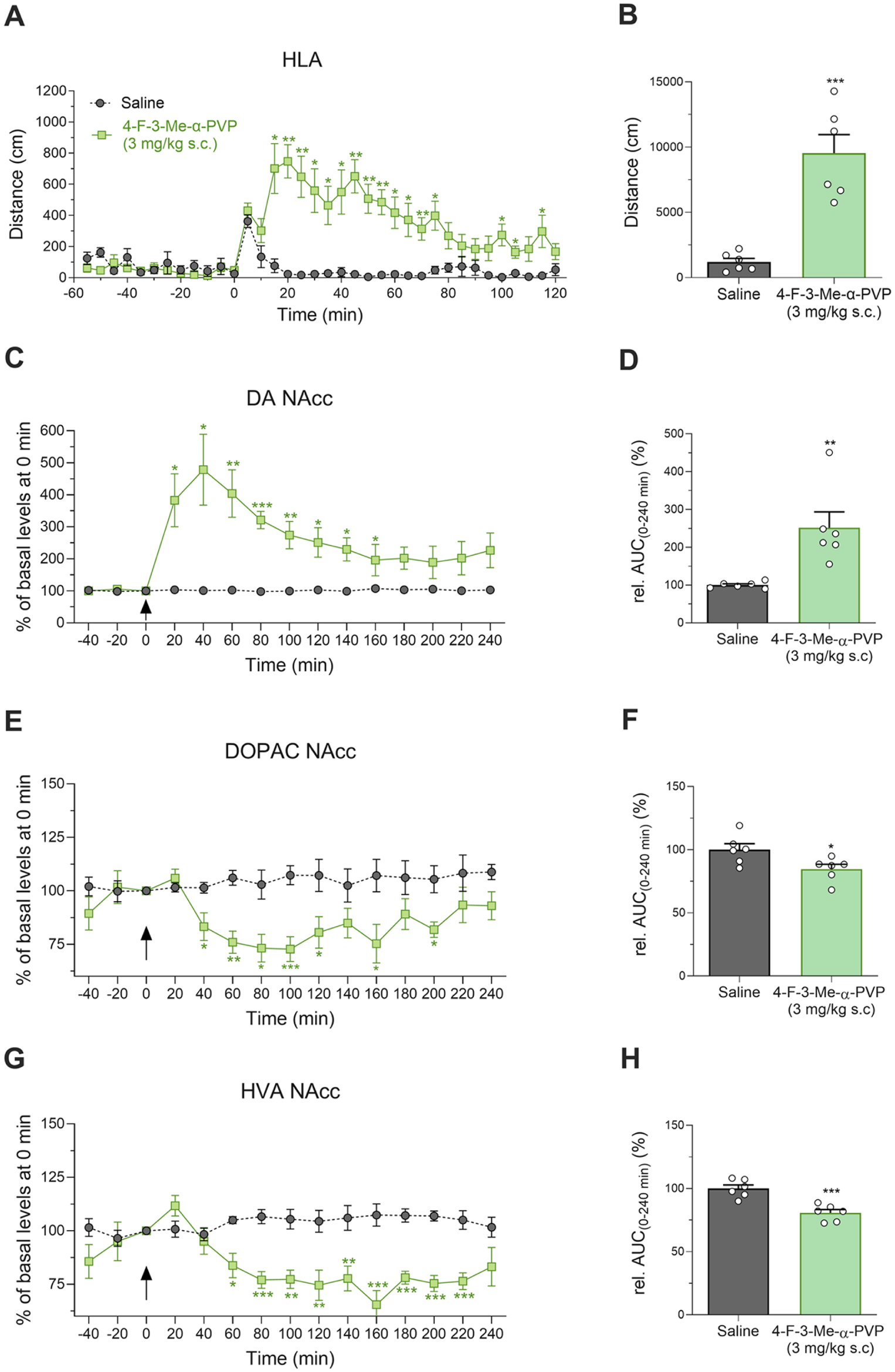
Time course profile of HLA induced by 4-F-3-Me-α-PVP in rats (a). Each time point represents the mean ± SEM of the distance (in cm) traveled in 5-minute blocks. n = 6/group. Tukey’s multiple comparison test: *p < 0.05 and **p < 0.01 versus saline. Effect of 4-F-3-Me-α-PVP on cumulative HLA in rats (b). Bars represent mean ± SEM of total distance (cm) traveled in 120 minutes. n = 6/group. Student’s t-test: ***p < 0.001 versus saline. Extracellular levels of DA (c), DOPAC (e), and HVA (g) in the NAcc of awake rats treated with 4-F-3-Me-α-PVP (3 mg/kg s.c.; mixed effects model) and its respective AUC relative to the saline group (d, f, and h), (student’s t-test). Bars represent mean ± SEM of % of basal levels. n = 6/group. *p < 0.05, **p < 0.01, and ***p < 0.001 versus saline.
4-F-3-Me-α-PVP has rewarding and reinforcing effects in rodents
The rewarding effects of 4-F-3-Me-α-PVP were assessed using the CPP paradigm. During the preconditioning phase, no significant differences were observed in the percentages of time spent in each compartment, with animals spending approximately 50% of the session time in each, indicating no inherent preference. Two animals were excluded from the study due to an initial preference for one of the compartments (>70% of the total session time). Kruskal–Wallis test yielded a significant effect of the variable Dose: H(3) = 14.86; p < 0.01. As seen in Figure 4(a), a significant preference score was observed at all doses tested.
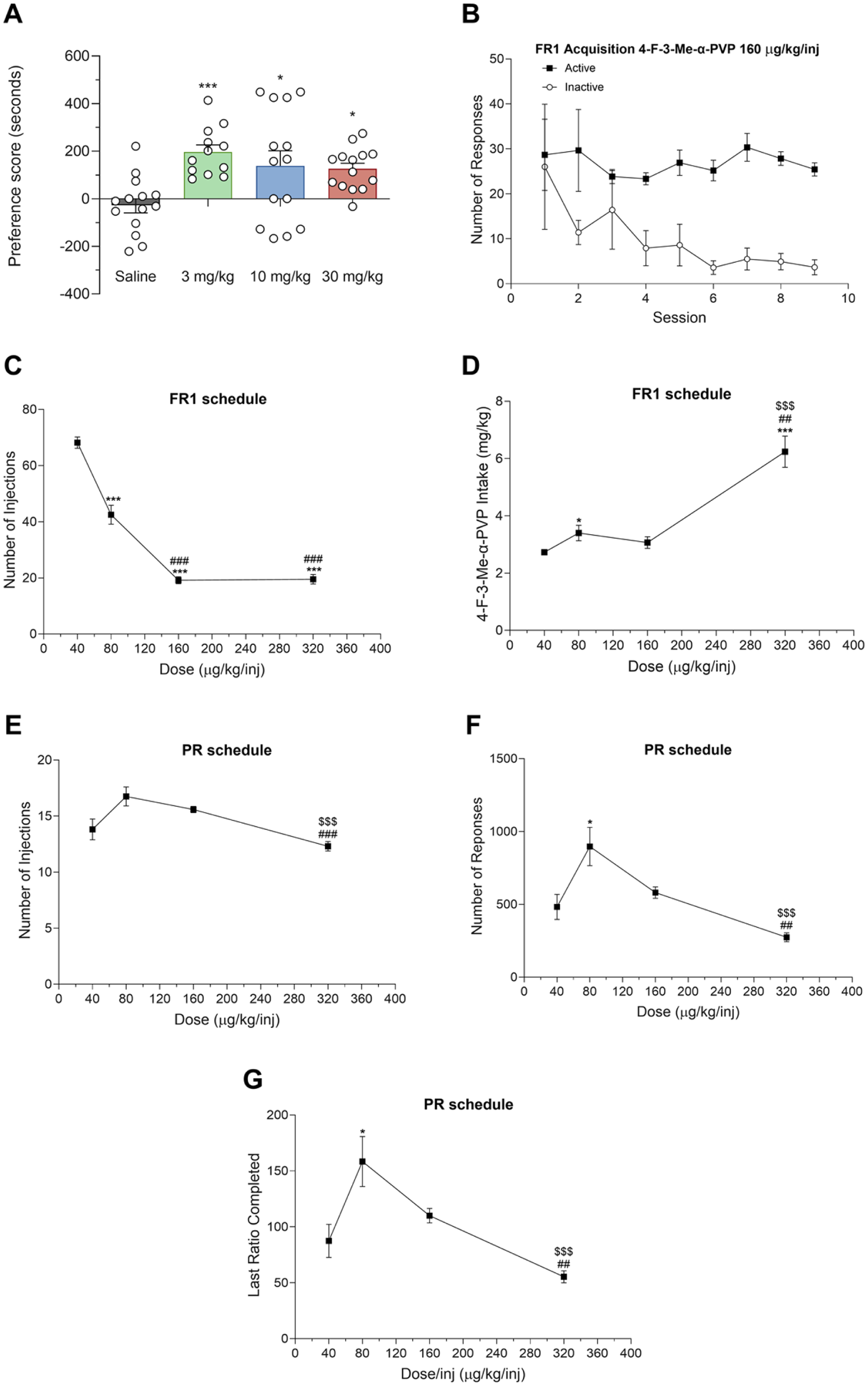
Effect of 4-F-3-Me-α-PVP on the CPP test in mice (a). Bars represent the mean ± SEM of the preference score (difference between the time spent in the drug-paired compartment on the test day and the preconditioning day). n = 12–14/group. Dunn’s multiple-comparisons test: ***p < 0.001 and *p < 0.05 versus saline. Acquisition of self-administration of 4-F-3-Me-α-PVP (160 µg/kg/inj) in rats under the FR1 schedule (b). n = 12/group. Dose-response of the number of injections (c) and total 4-F-3-Me-α-PVP intake (d) under FR1 schedule. n = 11–12/group. Tukey’s multiple comparisons test *p < 0.05 and ***p < 0.001 versus 40 µg/kg/inj, ##p < 0.01 and ###p < 0.001 versus 80 µg/kg/inj and $$$p < 0.001 versus 160 µg/kg/inj. Dose-response of the number of injections (e), responses (f), and last ratio completed (g) under the PR schedule. n = 10–12/group. Tukey’s multiple comparisons test: *p < 0.05 versus 40 µg/kg/inj, ##p < 0.01 and ###p < 0.001 versus 80 µg/kg/inj and $$$p < 0.001 versus 160 µg/kg/inj. All data are represented as mean ± SEM.
Acquisition of self-administration behavior of 4-F-3-Me-α-PVP (160 µg/kg/inj) under an FR1 schedule is seen in Figure 4(b). All 12 rats quickly learned to self-administer 4-F-3-Me-α-PVP and reliably discriminated between active vs inactive nose-pokes. Statistical analysis revealed a significant effect of session active/inactive nose poke (F(1, 22) = 17.70, p < 0.001), but no significant effect of time (F(2.055, 45.20) = 1.457, p > 0.05) and no significant time × session active/inactive nose-poke interaction (F(8, 176) = 1.234, p > 0.05). When 4-F-3-Me-α-PVP was tested at varying doses under the FR1 schedule, statistical analysis revealed a significant effect of the dose on both the number of injections (F(1.709, 18.23) = 116.2, p < 0.001) and total drug intake (F(1.458, 15.55) = 27.44, p < 0.001). Tukey’s multiple comparison test indicated that the number of injections was higher at the lowest dose tested, and progressively decreased as the dose increased (Figure 4(c)). On the other hand, total intake of 4-F-3-Me-α-PVP remained consistent across 40, 80, and 160 µg/kg/inj doses and peaked at the highest dose tested (320 µg/kg/inj; Figure 4(d)). Under the PR schedule, statistical analyses also indicated a significant effect of the dose on both the number of injections (F(2.084, 20.84) = 9.492, p < 0.01) and responses (F(1.622, 16.22) = 10.63, p < 0.01; Figure 4(e) and (f). Self-administration of the four doses under the PR schedule yielded a similar number of injections across doses, though it was slightly lower at the highest dose tested. The 80 µg/kg/inj dose elicited the highest number of responses, and corresponded to the highest breaking point, referred to as the maximum number of responses an animal is willing to make to obtain a drug infusion under a PR schedule (Figure 4(f)–(g)).
Discussion
Given the rapid emergence of novel synthetic cathinones and their increasing prevalence in the illicit drug market, research into their pharmacological and behavioral effects is essential. These compounds, often developed as structural analogs of controlled substances, pose significant challenges for public health and regulatory agencies due to their unpredictable potency, toxicity, and abuse potential. Despite legislative efforts to control the distribution of synthetic cathinones, new derivatives continue to surface, often with uncharacterized biological effects. Among them, 4-F-3-Me-α-PVP, a newly identified analog of α-PVP, has recently appeared in the illicit market, yet its pharmacological profile remains largely unknown. Thus, the present study provides the first insights into the mechanism of action of 4-F-3-Me-α-PVP, its effects on DA and related metabolites in the NAcc, as well as evidence of its psychostimulant, rewarding, and reinforcing effects in rodents.
Pyrovalerone analogs are known to act as monoamine transporter blockers, primarily inhibiting DAT, NET, and, to a lesser extent, SERT (Baumann et al., 2018; Kolaczynska et al., 2021). Therefore, we first evaluated the ability of 4-F-3-Me-α-PVP to inhibit these transporters. Our findings indicate that 4-F-3-Me-α-PVP exhibits similar potency to α-PVP in inhibiting the DAT. In contrast, although both compounds are low-potency inhibitors at SERT, 4-F-3-Me-α-PVP displays slightly greater inhibitory activity at this transporter compared to α-PVP. This results in a lower DAT/SERT ratio compared to α-PVP, yet it remains higher than that observed for cocaine. The use of cocaine as the reference psychostimulant, instead of methamphetamine, was based on the fact that pyrrolidinopentiophenones act as potent reuptake inhibitors (blockers) of monoamine transporters (Baumann et al., 2018; Simmler et al., 2013), a mechanism that is shared with cocaine. Moreover, a high DAT/SERT ratio has been correlated with an increased potential for addiction, as drugs that strongly inhibit DAT while sparing SERT tend to produce more pronounced stimulant and reinforcing effects (Dolan et al., 2018; Nadal-Gratacós et al., 2024; Negus and Banks, 2017). In our study, 4-F-3-Me-α-PVP showed a high DAT/SERT ratio, consistent with its robust psychostimulant profile. However, it is important to note that for transporter blockers, the DAT/SERT selectivity may be more correlative than determinative of abuse liability (Negus and Miller, 2014). Unlike transporter substrates, where SERT activity can significantly blunt the reinforcing effects of DAT-mediated dopamine increases, for inhibitors such as the α-PVP analogues, DAT potency remains the primary predictor of reinforcement (Baumann et al., 2013). Therefore, while the slight increase in SERT potency of 4-F-3-Me-α-PVP compared to its parent compound is noteworthy for its pharmacological characterization, its impact on the overall abuse potential is likely secondary to its potent DAT blockade. On the other hand, the increased potency at SERT seen in 4-F-3-Me-α-PVP may be explained by the addition of substituents in the aromatic region—a structural modification that our research group, along with others, has previously shown to shift selectivity toward SERT (Bonano et al., 2015; Nadal-Gratacós et al., 2021, 2022; Rickli et al., 2015). In particular, we previously demonstrated that introducing a fluorine at the para-position of α-PVP resulted in a decreased DAT/SERT ratio due to enhanced SERT inhibition (Nadal-Gratacós et al., 2022). Similarly, Meltzer et al. (2006) also demonstrated that the addition of a methyl group at the meta-position (3-Me-α-PVP) also increases the interaction with SERT when compared to α-PVP, although still maintaining a high selectivity toward DAT (Meltzer et al., 2006). In the present study, we observed that the addition of both a fluorine at the para-position and a methyl at the meta-position further shifts the selectivity towards SERT, reinforcing the impact of ring substitution on monoamine transporters, especially SERT. On the other hand, 4-F-3-Me-α-PVP is less potent at inhibiting NET than α-PVP but more potent than cocaine. The pharmacological profile of 4-F-3-Me-α-PVP reported here provides crucial data for refining predictive frameworks of synthetic cathinone toxicity. Within the SAR of α-pyrrolidinopentiophenones, the pyrrolidine moiety serves as a structural ‘switch’ that ensures potent DA reuptake inhibition, regardless of phenyl ring substitutions. Our findings demonstrate that the combination of a para-halogen (4-F) and a meta-alkyl group (3-Me) results in a compound that retains the high DAT/SERT selectivity typical of the α-PVP family, albeit with a slight increase in SERT activity compared to the parent compound. This confirms that emerging ‘multi-substituted’ analogues follow a predictable pattern: they may maintain high abuse liability as long as the α-pyrrolidino-structure is preserved. This insight is vital for early warning systems to anticipate the risk of novel analogues appearing on the market with similar aryl substitution patterns.
In vivo, the psychostimulant effects of 4-F-3-Me-α-PVP were evaluated using a motor performance test. This compound produced dose-dependent increases in locomotor activity in mice, with significant hyperlocomotion effects observed after 10 and 30 mg/kg acute i.p. administration. These results are consistent with findings for other synthetic cathinones, such as the structurally related α-PVP and MDPV, which are also known to induce psychostimulant effects (Duart-Castells et al., 2021; Giannotti et al., 2017). Interestingly, an increase in HLA was already observed in rats at 3 mg/kg. This apparent difference between species is, however, attributable to dose scaling, as 3 mg/kg in rats corresponds approximately to 10 mg/kg in mice when adjusted for interspecies differences in body surface area (Food and Drug Administration (FDA), 2005; Nair and Jacob, 2016; Reagan-Shaw et al., 2008). In the same experimental setup, we also assessed whether acute administration of 4-F-3-Me-α-PVP elicited anxiety-like effects through the evaluation of thigmotaxis. Mice treated with the highest dose of 4-F-3-Me-α-PVP spent less time in the center of the arena compared to those that received 3 or 10 mg/kg. However, these differences were not statistically significant when compared to the saline-treated group. While this preliminary observation suggests a limited anxiogenic effect, further studies are warranted to confirm this finding.
The dopaminergic system has been defined to play a central role in the regulation of locomotor activity and abuse liability, commonly associated with increased DA signaling in the mesolimbic pathway (Izawa et al., 2006; Miyamoto et al., 2014). Particularly, enhanced dopaminergic transmission within the NAcc has also been strongly associated with the addictive effects of drugs of abuse and is considered a key neurochemical correlate of abuse liability (Di Chiara and Imperato, 1988; Volkow et al., 2017). In line with this, our study not only shows that 4-F-3-Me-α-PVP is a potent DAT inhibitor in vitro, but also demonstrates that a single dose of the compound significantly increases extracellular DA levels in the NAcc of rats. In our study, we also report decreased levels of the DA metabolites DOPAC and HVA in the NAcc after an acute administration of 4-F-3-Me-α-PVP. This inverse relationship likely reflects reduced intracellular DA catabolism due to DAT blockade (Hernandez et al., 1987); by preventing DA reuptake into presynaptic terminals, 4-F-3-Me-α-PVP limits the availability of intracellular DA for enzymatic degradation by monoamine oxidase, thereby decreasing the formation of its primary metabolites.
Given the observed increase in DA levels in the NAcc, we sought to explore whether 4-F-3-Me-α-PVP elicits rewarding and reinforcing effects in rodents. Thus, we first assessed the compound’s potential for abuse by examining its ability to induce CPP, a widely used assay for evaluating the rewarding effects of psychoactive substances. Our results suggest that 4-F-3-Me-α-PVP induces a significant preference for the compartment associated with drug administration at all doses tested, demonstrating that this compound produces rewarding effects in mice. In the CPP experiments, a plateau was observed across all doses that produced a significant effect, suggesting a ceiling effect for conditioned reward. It is important to note that the CPP paradigm is not a primary method for establishing graded dose-response curves, but rather a screening tool for identifying rewarding or aversive properties (Bardo and Bevins, 2000; Tzschentke, 2007). Moreover, to assess the reinforcing effects of the compound, we conducted a self-administration experiment, where rats were allowed to self-administer 4-F-3-Me-α-PVP. Our results demonstrate that, under the FR1 schedule, rats quickly acquired self-administration behavior, demonstrating that they could discriminate between active and inactive nose-pokes. This rapid acquisition of behavior is a hallmark of substances with reinforcing effects (Creehan et al., 2015; Valadez and Schenk, 1994), supporting the hypothesis that 4-F-3-Me-α-PVP has abuse potential. Further examination of 4-F-3-Me-α-PVP’s dose-dependent effects on self-administration under the FR1 schedule revealed a decrease in the number of injections as the dose increased, similar to other structurally related synthetic cathinones (Aarde et al., 2015; Nadal-Gratacós et al., 2025).
When 4-F-3-Me-α-PVP was tested under the PR schedule, which provides a measure of the relative reinforcing value of the drug, a significant dose-dependent effect was also observed with peak PR break points at the 80 µg/kg/inj dose. The behavioral profile of 4-F-3-Me-α-PVP revealed a non-prototypical dose-response relationship in the self-administration PR schedule. While an ascending limb was identified between the 40 and 80 µg/kg doses, higher doses led to a decline in breakpoints. This “inverted U-shaped” curve differs from the profile of prototypical stimulants, such as cocaine, which typically shows a more stable ascending or plateaued response at higher doses (Richardson and Roberts, 1996). However, the descending limb observed here is consistent with reports for other highly potent pyrrolidinopentiophenones, such as MDPV and α-PVP (Aarde et al., 2013, 2015). This pattern may be attributed to the characteristic long-lasting effects of these pyrrolidinic derivatives, which can lead to a state of satiety during the session (Baumann et al., 2013; Gatch et al., 2017). In this sense, as demonstrated by (Niello et al., 2023), the binding kinetics—specifically a slow dissociation rate from the DAT—is a critical factor that predicts the sustained psychostimulant effects of this class of compounds. The persistent behavioral activation and the “inverted-U” shape observed in our PR experiments could also be partially explained by such slow-binding kinetics, which would lead to a prolonged occupation of the transporter and a more intense, long-lasting dopaminergic tone compared to shorter-acting stimulants such as cocaine. In addition, for these extremely potent DAT inhibitors, high doses often trigger intense stereotypic behaviors (Aarde et al., 2013; Lopez-Arnau et al., 2018) that may compete with the operant response, thereby artificially lowering the breakpoints despite the high reinforcing value of the drug. These findings underscore that 4-F-3-Me-α-PVP is a potent stimulant with a narrow pharmacological window between optimal reinforcing effects and behavioral interference.
In conclusion, the present study provides the first detailed pharmacological and behavioral characterization of 4-F-3-Me-α-PVP, a recently identified synthetic cathinone that has emerged on the illicit drug market. Through a combination of in vitro assays, in vivo neurochemical analyses, and preclinical behavioral paradigms, we demonstrate that 4-F-3-Me-α-PVP acts as a potent DAT inhibitor, leading to increased extracellular DA levels in the NAcc and concomitant reductions in DA metabolites, indicative of impaired reuptake and reduced intracellular metabolism. These neurochemical alterations are accompanied by robust psychostimulant, rewarding, and reinforcing effects in rodent models, including increased locomotor activity, significant place preference, and sustained self-administration under both FR and PR schedules. Finally, a limitation of this study is the exclusive use of male rodents. While this choice aimed to reduce biological variability for this first characterization of 4-F-3-Me-α-PVP, it limits the generalizability of the findings. Given the well-documented sex differences in the response to psychostimulants, future research including female subjects is necessary to confirm these results across both sexes. In light of the ongoing emergence of structurally related substances, a thorough understanding of the neurochemical profiles, reinforcing effects, and potential risks of newly emerged synthetic cathinones is essential to guide evidence-based regulatory decisions and inform effective public health strategies.
Supplemental Material
sj-docx-1-jop-10.1177_02698811261430492 – Supplemental material for Pharmacological characterization of 4-F-3-Me-α-PVP: A novel synthetic cathinone with psychostimulant effects and abuse liability in rodents
Supplemental material, sj-docx-1-jop-10.1177_02698811261430492 for Pharmacological characterization of 4-F-3-Me-α-PVP: A novel synthetic cathinone with psychostimulant effects and abuse liability in rodents by Núria Nadal-Gratacós, Stephanie Pain, Sandra Mata, Pol Puigseslloses, Morgane De Macedo, Virginie Lardeux, Fu-Hua Wang, Liselott Källsten, David Pubill, Xavier Berzosa, Jan Kehr, Marcello Solinas, Elena Escubedo, Jordi Camarasa and Raul López-Arnau in Journal of Psychopharmacology
Footnotes
Ethical considerations
All animal care and experimental protocols were approved by the Animal Ethics Committee of the University of Barcelona, supervised by the Autonomous Government of Catalonia, COMETHEA or Stockholms Norra djurförsöksetiska nämn following the directives of the Swedish Animal Welfare Act 1988:534). The procedures adhered to the guidelines of the European Community Council Directive 2010/63/EU, as amended by Regulation (EU) 2019/1010, and complied with the ARRIVE guidelines for reporting animal experiments (Percie du Sert et al., 2020).
Author contributions
Núria Nadal-Gratacós: Writing – review & editing, Writing – original draft, Methodology, Investigation, Formal analysis, Data curation. Stephanie Pain: Methodology, Investigation, Formal analysis, Data curation. Sandra Mata: Methodology, Investigation. Pol Puigseslloses: Methodology, Investigation. Morgane De Macedo: Methodology, Investigation, Formal analysis, Data curation. Virginie Lardeux: Methodology, Investigation, Formal analysis, Data curation. Fu-Hua Wang: Methodology, Investigation. Liselott Källsten: Methodology, Investigation. David Pubill: Writing – review & editing, Methodology. Xavier Berzosa: Writing – review & editing, Resources, Methodology, Investigation, Funding acquisition. Jan Kehr: Writing – review & editing, Funding acquisition, Formal analysis, Supervision, Data curation, Conceptualization. Marcello Solinas: Writing – review & editing, Funding acquisition, Formal analysis, Supervision, Conceptualization. Elena Escubedo: Writing – review & editing, Writing – original draft, Visualization, Methodology, Investigation, Funding acquisition, Conceptualization. Jordi Camarasa: Writing – review & editing, Supervision, Project administration, Funding acquisition, Conceptualization. Raul López-Arnau: Writing – review & editing, Writing – original draft, Visualization, Supervision, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by European Union (EU) Home Affairs Funds, NextGenPS project (101045825), Ministerio de Ciencia e Innovación/Agencia Estatal de Investigación /MICIU/AEI/10.13039/501100011033 (PID2022-137541OB-I00) and Plan Nacional Sobre Drogas (2024I057). DP, JC, EE, and RLA belong to 2021SGR0090 from Generalitat de Catalunya. NNG and XBR belong to 2021SGR00520 from Generalitat de Catalunya. PP received a doctoral scholarship grant from Generalitat de Catalunya (AGAUR), 2022 FISDU 00004. This study has benefited from the facilities and expertise of the PREBIOS platform (Université de Poitiers) and the technical help of Catherine Le Goff.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available on request.
Supplemental material
Supplemental material for this article is available online.
