Abstract
Background:
Negative allosteric modulators (NAMs) of gamma-aminobutyric acid (GABAA) receptors targeting the α5-subunit, primarily expressed on glutamate pyramidal neurons in the hippocampus and cortex, reduce inhibitory tone and enhance α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor throughput. The α5-GABAA-NAM, L-655,708, has shown rapid and sustained antidepressant-like effects in rodents.
Aim:
This study aimed to investigate the effects of L-655,708 on the monoamine and glutamate systems in rats in relation to the antidepressant-like response.
Methods:
Male Sprague-Dawley rats received a single dose of L-655,708 (3 mg/kg, i.p.) or vehicle. In vivo extracellular recordings were conducted in the dorsal raphe nucleus (DRN), locus coeruleus (LC), ventral tegmental area (VTA), and medial prefrontal cortex (mPFC) acutely, 1 day, 1 week, and 2 weeks post-injection. AMPA and N-methyl-D-aspartate (NMDA) responsiveness in the hippocampal CA1 region was assessed using microiontophoresis.
Results:
The NMDA-induced response in the CA1 hippocampus was significantly reduced 1 day post-injection of L655,708, while the AMPA response remained unchanged. Although mPFC pyramidal neurons’ firing was not changed, there was a two-fold increase in population activity of VTA dopamine (DA) neurons 1 day post-injection, lasting up to 1 week. Flumazenil, the benzodiazepine site antagonist, and 2,3-dioxo-6-nitro-7-sulfamoyl-benzo[f]quinoxaline (NBQX), an AMPA receptor antagonist, blocked this effect. L-655,708 had no acute effect on firing activity of serotonin or norepinephrine neurons.
Conclusion:
L-655,708 enhanced VTA DA neuron population activity for up to 1 week after a single injection. This effect was dependent on AMPA and took place through action on the benzodiazepine site.
Introduction
Selective serotonin (5-HT) reuptake inhibitors (SSRIs) have traditionally been the first line of treatment for patients with major depressive disorder (MDD). They have, however, a slow rate of onset of action, numerous side effects and a third of patients fail to respond to an initial trial (Hamon and Blier, 2013). While SSRIs increase 5-HT transmission, they concurrently dampen norepinephrine (NE) and dopamine (DA) neuronal activity (Dremencov et al., 2009; Ghanbari et al., 2010).
Although there has been a shift towards treatments primarily targeting the glutamate system, monoamines remain implicated in the antidepressant response due to the well-established reciprocal interaction between the two systems (Millan, 2006; Pralong et al., 2002). For instance ketamine, which acts by blocking N-methyl-D-aspartate (NMDA) receptors on gamma-aminobutyric acid (GABA) neurons and increasing α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor throughput (Gerhard et al., 2020; Li et al., 2011; Miller et al., 2016; Widman and McMahon, 2018), does also affect the monoamine systems. Administration of ketamine increases DA neuron population activity and NE neuron firing activity without any action on 5-HT neuronal activity (Daniels et al., 2023; El Iskandrani et al., 2015; Witkin et al., 2016). Nevertheless, microdialysis studies show elevated extracellular concentrations of 5-HT, DA, and NE in the medial prefrontal cortex (mPFC) and ventral hippocampus, following a single injection of ketamine (López-Gil et al., 2019; Lorrain et al., 2003; Moghaddam et al., 1997).
While ketamine indirectly enhances pyramidal neuron activity through GABAergic inhibition, similar increases in excitatory glutamate transmission can be directly induced by targeting α5-subunits on pyramidal neurons (Brünig et al., 2002) using negative allosteric modulators (NAMs) of GABAA receptors (α5-GABAAR-NAMs; Glykys and Mody, 2006; Prenosil et al., 2006; Zorrilla de San Martin et al., 2020).
GABAARs are pentameric ligand-gated ion channels located on glutamate neurons, highly concentrated in hippocampal CA1 and CA3 regions and, to a lesser extent, in the PFC (Jacob, 2019; Serwanski et al., 2006; Sur et al., 1999). α5-GABAAR-NAMs are a class of drugs that specifically target the benzodiazepine site of α5-GABAARs. Their binding causes a decrease in the frequency of ion channel opening, limiting the influx of Cl− ions and thereby leading to depolarization and a subsequent release of glutamate that allows for an increase in AMPA receptor throughput (Jacob, 2019; Sigel and Steinmann, 2012). L-655,708 was reported to be a selective α5 GABAAR-NAM exhibiting 50- to 100-fold selectivity for the α5-subunit (Quirk et al.,1996). It was also shown to exert antidepressant-like effects in the forced swim test (FST) and sucrose preference test (SPT) through the blockade of the α5-subunit on GABAA Rs (Bugay et al., 2020; Carreno et al., 2017; Troppoli et al., 2022; Xiong et al., 2018; Zanos et al., 2017).
Given the predominant expression of α5 GABAA receptors in the hippocampus, the present study aimed at determining whether their blockade by L-655,708 alters AMPA- and NMDA- responses in this brain region. Since α5-GABAA receptors are also present, albeit in moderate density in the mPFC, it was deemed important to determine the effect of L-655,708 on the firing activity of pyramidal neurons in this structure. Given the results in behavioural studies, and taking into account the fact that FST (swimming and climbing) and SPT, respectively inform on 5-HT, NE and DA systems, the present study aimed at determining whether: (1) acute administration of L-655,708 has any effect on the firing and burst activity of 5-HT, NE and DA neurons; (2) potential L-655,708-induced changes in monoamines neurons activity are mediated through a) the benzodiazepine site and b) by AMPA receptors, as ketamine does; (3) AMPA is involved in sustaining any changes in neuronal activity induced by a single injection of L655,708.
Methods
Animals
In vivo electrophysiological recordings were conducted during the light phase in male Sprague-Dawley rats obtained from Charles River (St. Constant, Quebec, Canada) weighing 250–350 g at the time of the experiment. Upon arrival, rats were housed in pairs under standard laboratory conditions (12 hour dark/light cycle) with access to food and water. Rats were acclimated to the novel environment for 7 days prior to the experiments. All animals were handled in accordance with the Canadian Council on Animal Care guidelines, and study protocols were approved by the local Animal Care Committee (University of Ottawa Animal Care Committee, Ottawa, Canada).
Drug administration
L-655,708 was purchased from Cayman Chemical (MI, USA) and dissolved in 75% dimethyl sulfoxide (DMSO) and 25% water. A single 3 mg/kg dose of L-655,708 was administered intraperitoneally (i.p.) 30 minutes, 1 day, 1 week and 2 weeks prior to electrophysiological recordings, which lasted for up to 2 hours. Control rats were injected with a vehicle solution. The dose of L655,708 used in the present study is based on previous work by Atack et al. (2006) showing that a dose of 3 mg/kg results in approximately 75% occupancy at α5-GABAARs and 33% at α1, α2, and α3 receptor subtypes; this indicates an optimal balance for achieving maximal α5-selective engagement while minimizing off-target effects.
NBQX (10 mg/kg) was purchased from Cayman Chemical (MI, USA) and dissolved in 0.9% aqueous saline solution. Flumazenil (3 mg/kg) was purchased from Cayman Chemical (MI, USA) and dissolved in 80% DMSO and 20% water. Both NBQX and flumazenil were administered i.p. 30–60 minutes prior to recording in treated and control rats. Rats were not randomized, and the experimenter was not blinded.
In vivo electrophysiological recordings
In vivo extracellular recordings were conducted in rats anaesthetized using chloral hydrate (400 mg/kg, i.p) and mounted on a stereotaxic apparatus (David Kopf, CA, USA). Periodic administration of supplemental chloral hydrate (100 mg/kg, i.p) was carried out during the experiment to maintain anaesthesia and body temperature of 37°C was maintained using a heating pad. For each brain structure of interest, a burr hole was drilled in the skull according to standard rat brain stereotaxic coordinates, using the atlas by Paxinos and Watson (1987). Neuronal activity was captured in real time using the Spike2 program (Cambridge Electronic Design, Cambridge, UK).
Extracellular recordings and microiontophoresis in hippocampus CA1 pyramidal neurons
Microiontophoresis and extracellular recordings of pyramidal neurons in the dorsal CA1 region of the hippocampus were conducted using five-barrelled glass micropipettes and positioned in accordance to the following coordinates: 4.0–4.2 mm anterior to lambda and 4.0–4.2 mm lateral to the midline. Pyramidal neurons were typically encountered at a depth of 2.5–3.0 mm below the surface of the brain. The central barrel used for electrophysiological recordings contained 2 M NaCl solution, while the peripheral barrels contained AMPA (5 mM in 200 mM NaCl, pH 8), NMDA (10 mM in 200 mM NaCl, pH 8) and 2 M NaCl for current balancing. Neurons were identified based on the following properties: (1) a large amplitude (0.5–1.2 mV), (2) long-duration (0.8–1.2 ms) action potentials alternating with complex spike discharges (Kandel and Spencer, 1961). Ejection currents of AMPA (−1 and −2 nA) and NMDA (−3, −5, and −8 nA) were used to activate pyramidal neurons, which are not firing spontaneously in the hippocampal CA1 region of rats under chloral hydrate anaesthesia. The currents of AMPA and NMDA for microiontophoretic ejections of 40 seconds in duration were kept constant for both vehicle and rats that received L-655,708. A retention current of +15 nA was used between ejections that lasted 40 seconds. AMPA and NMDA responses were assessed in an average of six neurons per rat.
Electrophysiological recordings of mPFC pyramidal neurons
Neurons in the mPFC were recorded by positioning a single-barrel glass micropipette at 3.2–3.4 mm anterior to Bregma and 0.6–0.8 mm lateral to the midline suture at a depth of 2.5–5.0 mm from the surface of the brain. mPFC pyramidal neurons were identified in accordance with the following electrophysiological criteria: (1) irregular firing with rates ranging between 0.01 and 3 Hz, (2) long duration (0.8–1.2 ms), and (3) biphasic or triphasic waveforms (Riga et al., 2017).
Electrophysiological recordings of ventral tegmental area DA neurons
For DA neuron recordings, a single-barrel glass micropipette was positioned at 3.0–3.6 mm anterior to lambda and 0.6–1.0 mm lateral to the midline suture. DA neurons were typically encountered at a depth of 7.5–8.5 mm from the surface of the brain. DA neurons were classified based on well-established characteristic features including: (1) regular and irregular, slow firing rates (2–10 Hz), (2) biphasic or triphasic waveforms, (3) the distinctive presence of a notch on the depolarization phase of the spike, (4) the distance from the spike initiation to the trough is equal or greater than 1.1 ms, and (5) a characteristic low-pitch sound on the audio monitor (Ungless and Grace, 2012). The number of spontaneously active DA neurons found per track, referred to as population activity, was determined by recording multiple trajectories in a grid of 6–9 tracks per rat.
Electrophysiological recordings of dorsal raphe nucleus 5-HT neurons
Serotonin neurons were recorded using single-barrel glass micropipettes positioned on the midline at 0.9–1.2 mm anterior to lambda and ±0.2 mm lateral to the midline suture. Neurons were typically encountered at a depth of 4.5–5.5 mm from the surface of the brain. Serotonin neurons were identified based on the following electrophysiological criteria: (1) regular and slow firing rates (0.5–2.5 Hz), (2) long duration (2–5 ms), and (3) biphasic or triphasic waveforms (Vandermaelen and Aghajanian, 1983).
Electrophysiological recordings of locus coeruleus NE neurons
Norepinephrine neurons were recorded by positioning a single barrel glass micropipette 0.8–1.1 mm posterior to lambda and 0.9–1.3 mm lateral from the midline suture at a depth of 4.5–6.0 mm from the surface of the brain. Norepinephrine neurons were identified by the presence of: (1) regular firing rates (0.5–5 Hz), (2) long-duration (>2 ms), and (3) a volley of discharge of action potentials in response to pinching of the contralateral paw, followed by a transient silent period (Marwaha and Aghajanian, 1982).
Burst analysis
The beginning of a burst was determined by the occurrence of two spikes with interspike intervals (ISI) < 0.08 seconds for NE and DA neurons and <0.01 seconds for 5-HT neurons. Burst termination was defined as an ISI > 0.16 seconds for NE and DA (Grace and Bunney, 1983) and ISI > 0.01 seconds for 5-HT neurons (Hajós and Sharp, 1996). For mPFC pyramidal neurons, burst identification was based on the following criteria: a series of two or more spikes, with ISI < 0.045 seconds from the initiation and >0.045 seconds for termination of burst (Laviolette et al., 2005).
Statistical analyses
Data are presented as mean values ± standard error of the mean. Statistical comparisons between control and L-655,708-treated groups were carried out using the two-tailed t-test when normality passed with the Shapiro–Wilk test. When normality failed, the nonparametric Mann–Whitney test was utilized and is based on comparing medians of the groups. One-way and two-way analysis of variance (ANOVA) with a post-hoc analysis were also used when appropriate. Statistical comparisons were conducted using the software SigmaPlot 12.5 (Systat Software Inc, CA, USA).
Results
Effects of a single administration of L-655,708 on AMPA- and NMDA-induced responses in CA1 region of the hippocampus
The AMPA- and NMDA-evoked response of CA1 pyramidal neurons was assessed by determining the number of spikes generated when ejecting AMPA at currents of −1 and −2 nA, and NMDA at currents −3, −5, and −8 nA, in controls and rats that received L-655,708. There was no significant effect of treatment (two-way repeated measures ANOVA F[1,10] = 0.2; p = 0.6), current (F[1,10] = 4; p = 0.1) and interaction (F[1,10] = 1.4; p = 0.3) on AMPA-evoked responses (Figure 1(a) and (c)). In contrast, a two-way repeated measures ANOVA on NMDA-induced response revealed a robust decrease in responsiveness of these neurons (F[1,20] = 17; p = 0.002) and current (F[2,20] = 8; p = 0.003). There was, however, no significant interaction between treatment and current intensity (F[2, 20] = 1.4; p = 0.3). Holm–Sidak post hoc analysis showed that in the L-655,708-administered group, there was a statistically significant decrease in the NMDA-induced response that was independent of intensity of current (p = 0.002; Figure 1(d)).
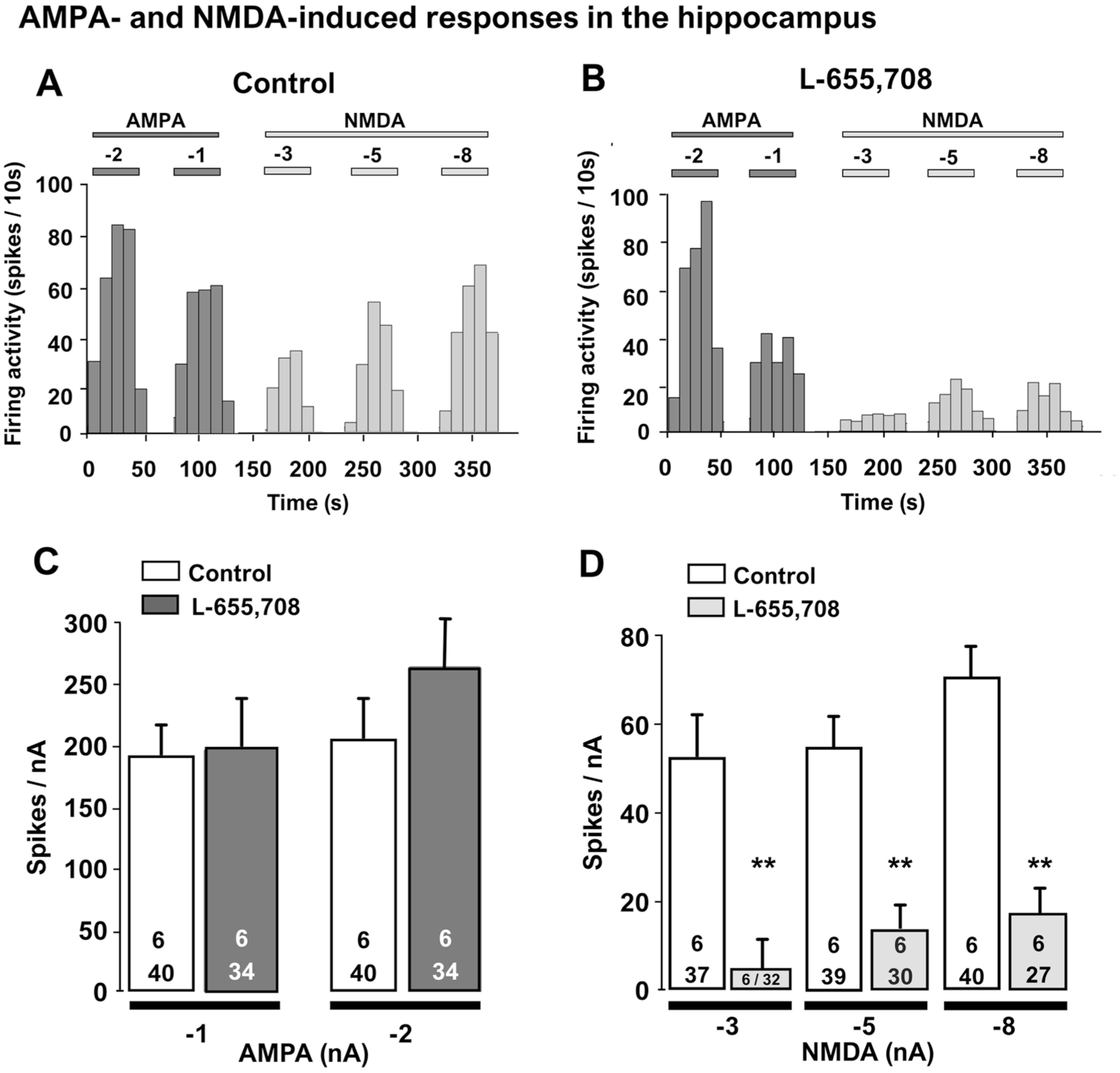
Representative integrated firing rate histograms illustrating the effects of L-655,708 (3 mg/kg, i.p) on AMPA- and NMDA-induced response in CA1 region of the hippocampus, 1-day after administration (a, b). Overall change in the number of spikes generated by nA by AMPA and NMDA in hippocampal CA1 pyramidal neurons following injection of L-655,708 compared to controls (c, d).
Effects of a single administration of L-655,708 on the activity of mPFC pyramidal neurons
The mean firing rate and the percentage of spikes in bursts of a population of mPFC pyramidal neurons were determined before and at least 30 minutes after L-655,708 injection. One day after a single administration of L-655,708, there was no significant alteration of pyramidal neuronal firing (two-tailed t-test, t[8] = −0.5; p = 0.7; Figure 2(a)). There was also no change in the percentage of spikes occurring in bursts (two-tailed t-test, t[8] = 0.8; p = 0.4; Figure 2(b)).
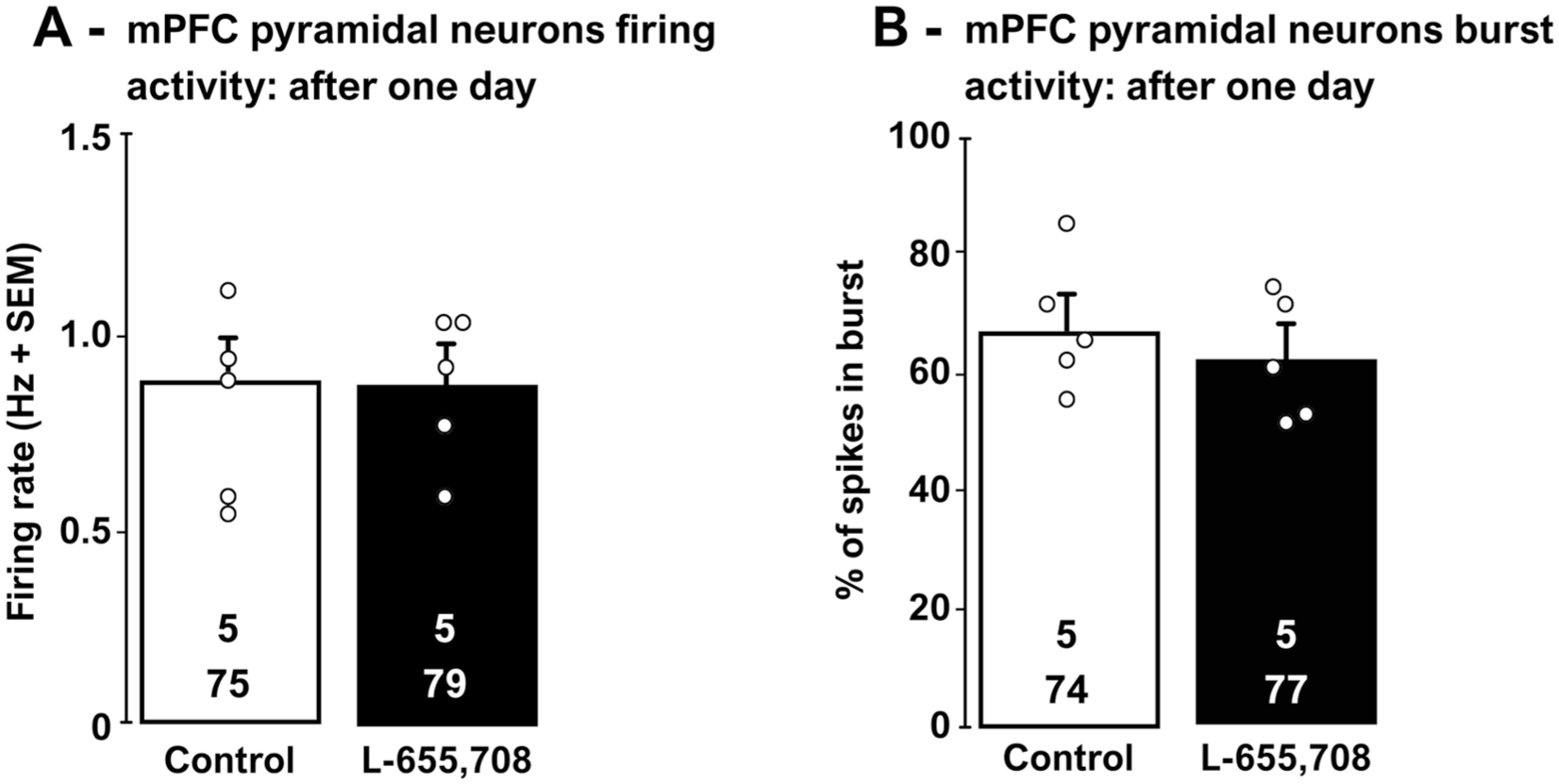
The effects of L-655,708 (3 mg/kg, i.p) administration on mPFC pyramidal neurons assessed 1-day after administration.
Effects of a single administration of L-655,708 on the activity of ventral tegmental area DA neurons
When injected acutely, L-655,708 had no effect on the firing rate of DA neurons (two-tailed t-test, t[8] = −0.6; p = 0.6; Supplemental Table 1), the percentage of spikes in burst (control: 18 ± 3.7%, L-655,708: 21 ± 6.1%; two-tailed t-test, t[8] = −0.4; p = 0.7; Supplemental Table 1), and population activity of DA neurons (control: 1.4 ± 0.2 neurons/tract, L-655,708: 1.9 ± 0.2 neurons/tract; two-tailed t-test, t[8] = −1.8; p = 0.1; Figure 3(a)). One day following the administration of L-655,708 injection, however, there was more than a two-fold increase in the number of spontaneously active DA neurons compared to rats administered with the vehicle (control: 1.3 ± 0.1 neurons/tract, L-655,708: 3.3 ± 0.1 neurons/tract, one-way ANOVA F[4,24] = 37; p = 0.001; Supplementary Table 1, Figure 3(a)). However, following Kruskal–Wallis test on ranks the firing activity (H[4] = 8; p = 0.1), the percentage of spikes occurring in bursts was not significantly altered by L655,708 (one-way ANOVA F[4,24] = 0.4; p = 0.8; Supplemental Table 1).
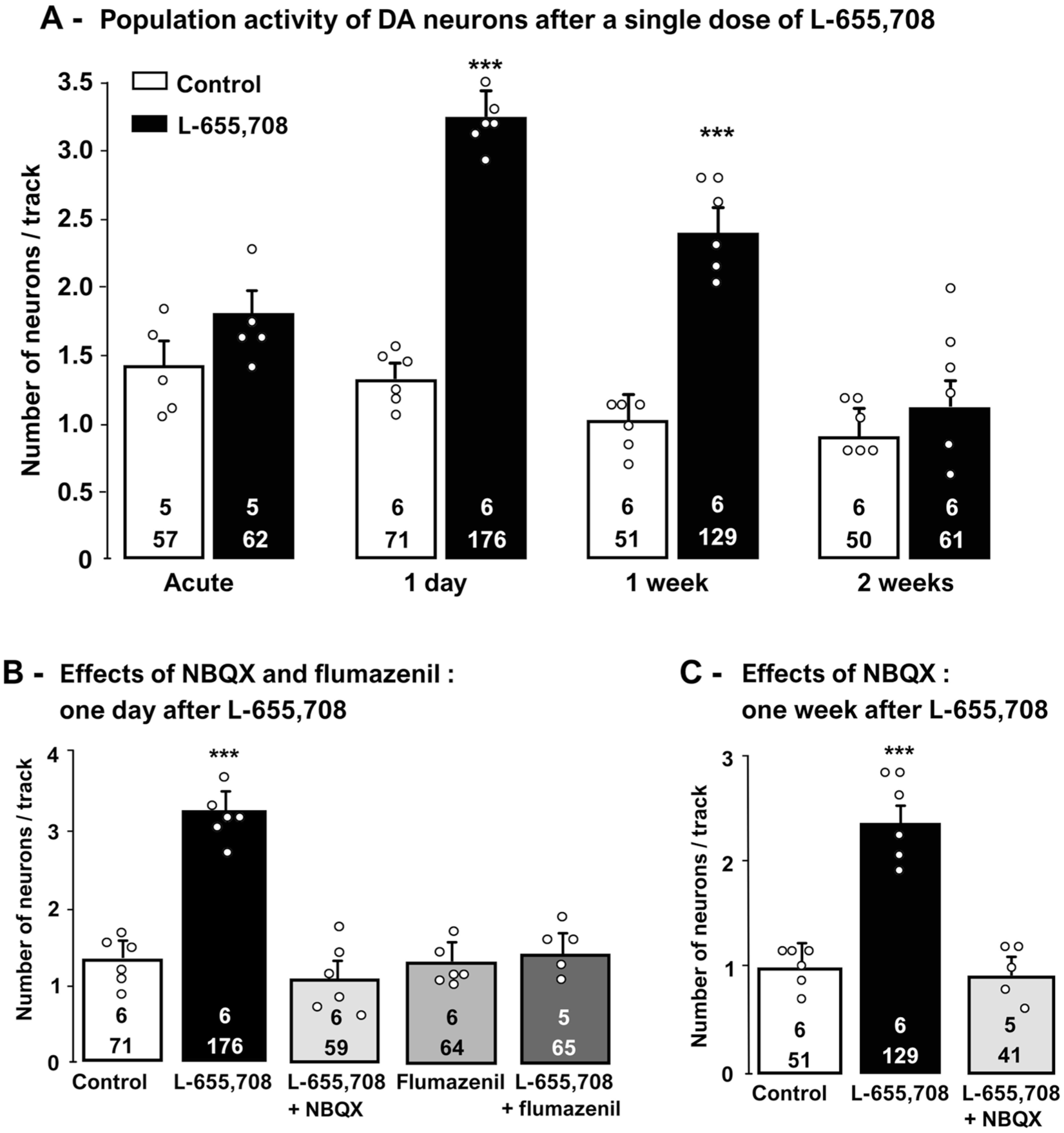
Effects of a single dose of L-655,708 (3 mg/kg, i.p.) on VTA DA neurons, 30 minutes, 1-day, 1-week and 2-weeks following its administration (a). Histograms show data as mean values ± SEM.
The Holm–Sidak post-hoc test revealed that the increase in DA neuron population activity was abolished when the AMPA receptor antagonist NBQX (10 mg/kg) was administered 30 minutes before L-655,708 (p = 0.7; Figure 3(b)). It is important to note that at this dose, NBQX on its own was previously shown to be devoid of action on this parameter (El Iskandrani et al., 2015).
To ascertain that L-655,708 was acting through α5-GABAARs to increase DA neuron population activity, the competitive benzodiazepine site antagonist flumazenil (3 mg/kg) was injected 30 minutes prior to recordings. Indeed, administration of the latter drug, which has no effect on its own, abolished the increase in DA neuron population activity induced by injection of L-655,708 (Holm–Sidak post-hoc, p = 0.9; Figure 3(b)).
To evaluate whether the effects of a single L655,708 administration were prolonged beyond the elimination of the drug, its effects were examined 1 week after injection. Indeed, a significant increase in the number of spontaneously active DA neurons in the ventral tegmental area (VTA) was still present 1-week post-injection, when compared to control (one way ANOVA F[2,14] = 44; p = 0.001; Supplemental Table 1, Figure 3(a)). In order to assess if AMPA receptors are involved in maintaining this effect, NBQX was administered 30 minutes before the recordings. In fact, the increase of DA neuron population activity induced by L-655,708 was abolished by NBQX (Holm–Sidak method, p = 0.5; Figure 3(c)). No changes from baseline were observed in DA neurons’ firing activity (one-way ANOVA F[2,15] = 2; p = 0.2; Supplemental Table 1) and the percentage of spikes occurring in burst 1 week after administration of L-655,708 (Kruskal–Wallis test H[2] = 0.8; p = 0.6; Supplemental Table 1).
To determine for how long the effects of L-655,708 on DA neurons were sustained, VTA DA neuron population activity was assessed 2 weeks after a single injection. The enhancement in the number of spontaneously active DA neurons produced by L-655,708 was no longer present (control: 0.9 ± 0.1 neurons/tract; L-655,708: 1.1 ± 0.2 neurons/tract; two-tailed t-test, t[10] = 1; p = 0.3; Supplemental Table 1; Figure 3(a)). Firing and burst activity were unchanged 2 weeks following a single injection of L655,708.
Effects of a single administration of L-655,708 on the activity of dorsal raphe nucleus 5-HT neurons
When injected 30 minutes prior to electrophysiological recordings, a single dose of L-655,708 did not significantly change the firing activity of 5-HT neurons during the 2 hour recording session (Mann–Whitney test U = 7; p = 0.2; Figure 4(a)). Additionally, there was no alteration in the number of neurons firing in bursts (two-tailed t-test, t[10] = 1; p = 0.3; Supplemental Table 1). Similarly, 1 day after its administration, L-655,708 did not induce any significant changes in the firing rate (two-tailed t-test, t[11] = 1.4; p = 0.2; Figure 4(b)) and burst activity (two-tailed t-test, t[11] = 0.4; p = 0.7; Supplemental Table 1).
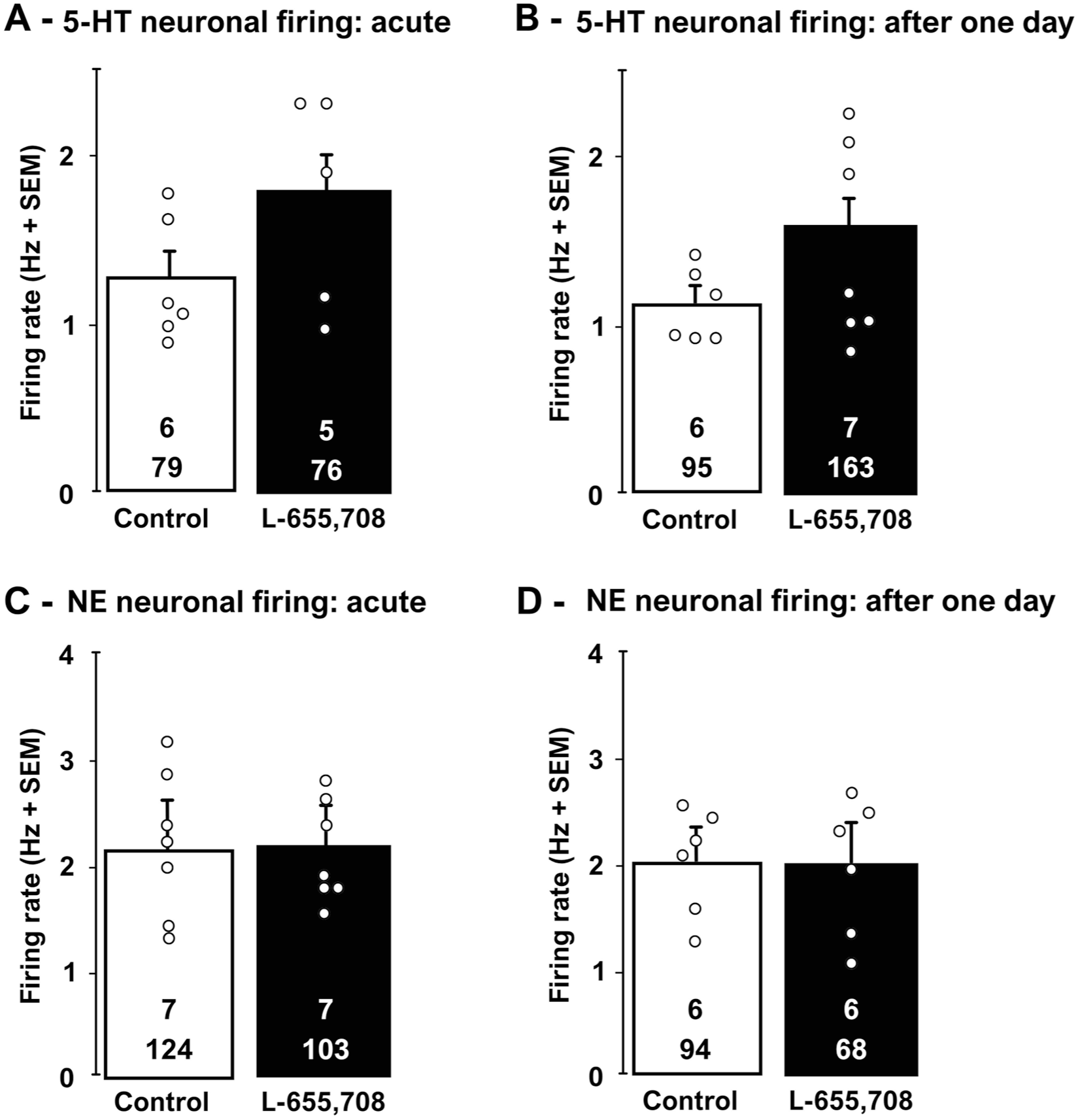
Effects of a single dose of L-655,708 (3 mg/kg, i.p) on the firing activity of DRN 5-HT (a, b) and LC NE neurons (c, d), 30 minutes and 1-day following its injection.
Effects of a single administration of L-655,708 on the activity of locus coeruleus NE neurons
As depicted in Figure 4(c), there were no significant changes in NE neurons firing activity (two-tailed t-test, t[12] = 0.2; p = 0.8; Figure 4(c)) and the number of NE neurons with burst activity (two-tailed t-test, t[12] = −0.4; p = 0.7; Supplemental Table 1), 30 minutes after the administration of L-655,708, for up to 2 hours. Additionally, no alterations in neuronal firing activity were observed 1 day post administration (two-tailed t-test, t[10] = 0.07; p = 0.9; Figure 4(d)), or in the bursting activity of neurons (two-tailed t-test, t[10] = 0.2; p = 0.9; Supplemental Table 1).
Discussion
The present study documented that L-655,708 significantly reduced NMDA but not AMPA receptor responsiveness in the hippocampus. Furthermore, a striking two-fold enhancement in VTA DA neuron population activity was observed 1 day following a single injection, which persisted for up to 1 week before returning to baseline by week 2. The reversal of these effects by flumazenil and NBQX confirmed the involvement of the benzodiazepine site of α5-GABAARs and an AMPA receptor-dependent mechanism, respectively. Notably, either acutely or 1 day post administration, L-655,708 had no effect on the firing activity of 5-HT and NE neurons, nor glutamate neurons in the mPFC.
α5-subunit-containing GABAA receptors, the primary target of L-655,708, are highly expressed in the hippocampus and moderately in the frontal cortex (Fritschy and Mohler, 1995; Sotiriou et al., 2005; Sur et al., 1999). Indeed, local application of L-655,708 in the ventral hippocampus decreased immobility and increased climbing in the FST (Bugay et al., 2020), while these effects were blocked when lidocaine was directly injected into the hippocampus (Carreno et al., 2017). Together, this suggests that the antidepressant-like effects of L-655,708 originate in the hippocampus. While NMDA receptor responsiveness was reduced and AMPA activity remained unchanged in the present study, previous work in animal models of depression reported that L-655,708 significantly increased the hippocampal AMPA/NMDA ratio—a key measure of synaptic strength (Fischell et al., 2015). In the same model, L-655,708 rapidly restored weakened AMPA (GluA1)-mediated excitatory transmission, an effect associated with the reversal of depressive-like behavioural phenotypes (Fischell et al., 2015). Despite these synaptic changes in the hippocampal-mPFC pathway, L-655,708 did not alter the firing activity of pyramidal neurons in the mPFC 1 day post-administration (Bugay et al., 2020). Given that modulation of α5-GABAARs is known to relieve inhibitory control over mPFC pyramidal neurons (Sur et al., 1999), an increase in firing activity might have been expected. Contrarily, the direct disinhibition of GABAergic interneurons by the enantiomer S-ketamine has been shown to enhance pyramidal firing, an effect not observed with either ketamine or L-655,708 (Blier and El Mansari, 2025). The mechanisms underlying these differential effects on mPFC pyramidal neuron activity remain to be elucidated. Nonetheless, L-655,708 elevated levels of the AMPA receptor GluA1 subunit in mPFC synaptosomal fractions and altered the AMPA-to-NMDA ratio at temporo-ammonic–CA1 synapses, suggesting the induction of glutamatergic plasticity within the mPFC, possibly originating from hippocampal inputs (Bugay et al., 2020; Troppoli et al., 2022). The critical role of mPFC AMPA receptor signalling to the antidepressant-like effects of L-655,708 is further supported by findings that local infusion of the AMPA receptor antagonist NBQX blocked both AMPA-mediated excitatory transmission and behavioural antidepressant-like effects in the FST (Bugay et al., 2020). Similarly, inactivation of the infralimbic (IL) region of the PFC completely abolished the antidepressant and anxiolytic effects of systemic ketamine in rodents, whereas direct microinfusion of ketamine into the IL-PFC reproduced these effects and enhanced the number and function of layer V pyramidal neuron spines (Fuchikami et al., 2015; Li et al., 2011; Moda-Sava et al., 2019). Collectively, these findings highlight the central role of mPFC circuitry—particularly AMPA receptor–mediated plasticity—in both initiating and sustaining the antidepressant-like responses induced by L-655,708 and related other compounds such as ketamine.
In addition to regulating mPFC activity, the ventral subregions of the hippocampus also modulate DA neurotransmission through polysynaptic circuitry (Floresco et al., 2001; Grace et al., 2007). In the VTA, it is assumed that half of the DA neurons are tonically inhibited through ventral hippocampus-accumbens (NAc)-ventral pallidal (VP)-VTA feedback pathway (Grace et al., 2007). Since intra-ventral hippocampus injection of L655,708 increases DA neuron population activity (Perez et al., 2022), it is reasonable to assume that the ability of L-655,708 to increase DA neuron population activity in the present study is mediated via its induced increase in neuron excitability in the ventral hippocampus (Bugay et al., 2020). This in turn enhanced neuronal firing in the NAc, which yielded a GABAergic inhibition of firing of the VP, relieving DA neurons from inhibition and increasing DA neuron population activity herein (Floresco et al., 2001; Floresco and Grace, 2003). This constitutes a stable baseline level of extrasynaptic DA in projection areas, hence allowing the hippocampus to modulate novelty-gated information storage (Lisman and Grace, 2005). Remarkably, the increase in the population activity of DA neurons persisted for up to 1 week after a single administration of L-655,708 but returned to baseline levels 2 weeks after a single injection. This enhancement both after 1 day and 1 week took place through AMPA activation because their antagonism with NBQX blocked this effect. In light of these results, and although the mechanisms involved in maintaining this response remain to be elucidated, two explanations can be proposed based on the hippocampus-VTA loop (Lisman and Grace, 2005). First, an increase in the AMPA/NMDA ratio and AMPA-dependent excitatory synaptic strength in the hippocampus (Fischell et al., 2015) may underlie the sustained augmentation observed in the VTA, as this effect was abolished by local hippocampal lidocaine injection (Carreno et al., 2017). Second, the sustained response may reflect the involvement of long-term potentiation within the VTA, as a similar and long-lasting form of synaptic plasticity induced for instance by a single cocaine injection has previously been reported in the VTA, lasting for up to 5 days (Ungless et al., 2001; see also Argilli et al., 2008). The increase in population activity, along with behavioural data showing the reversal of DA-related hedonic deficits in the SPT by L-655,708 (Fischell et al., 2015), emphasizes the important role of DA modulation though AMPA receptors in triggering and maintaining the antidepressant response of this drug. Despite a DA transmission enhancement, L655,708 does not induce any addiction in rats, in paradigms of self-administration, which are highly predictive of drug potential for abuse (Carreno et al., 2017). Interestingly in the clinic, the enantiomer of ketamine, S-ketamine, did not yield any addictive tendencies for addiction in patients with MDD over a 5-year period (Zaki et al., 2025).
Flumazenil abolished the L-655,708-induced enhancement in DA neuron population activity herein, which is in line with its blunting of improvement seen in preference for sucrose in the SPT and the female urine sniff test following injection of the NAM MRK-016 (Troppoli et al., 2022). Altogether, these data show that, in addition to allosteric modulation of the benzodiazepine site on the effect on anhedonia, it also mediates the L655,708-induced action on DA neuron population activity.
The present study showed that L-655,708 had no impact on 5-HT neuronal firing after 30 minutes and 24 hours post-administration. The FST results support these findings as L-655,708 showed no effect on 5-HT-dependent swimming at those time points. A significant increase in swimming was, however, observed 1 week after the direct injection of L-655,708 into the ventral hippocampus (Bugay et al., 2020; Carreno et al., 2017). Since α5-GABAA receptors are not expressed in the DRN and LC, direct effects of L-655,708 on the firing activity of 5-HT and LC neurons can be excluded (Corteen et al., 2011; Fritschy and Mohler, 1995; Sur et al., 1999). As the hippocampus has no known projections to the DRN and LC, indirect modulation of the mPFC by L-655,708 may still influence 5-HT and NE neuronal firing through blockade of α5-GABAARs, although these represent only 13% of receptor in this region (Lee et al., 2003; Peyron et al., 1997; Sur et al., 1999). While stimulation of the mPFC inhibits most 5-HT neurons, up to 17% can be activated (Celada et al., 2001; Hajós et al., 1998). Although the overall mean firing activity of 5-HT neurons recorded following L-655,708 was unchanged, it is possible that its administration induced an increase in a subpopulation of neurons that can be activated by mPFC stimulation. Nevertheless, whether mPFC neurons containing α5-GABAARs innervate 5-HT neurons that are inhibited or activated by stimulation of PFC remains to be elucidated. Similarly, while it is unknown whether mPFC neurons containing α5-GABAARs innervate LC NE neurons, the present findings show no effect of L-655,708 on the average firing rate of NE neurons. This is in line with results showing that NE-dependent climbing in FST was also unchanged 30 minutes after administration of L-655,708, although an increase in climbing behaviour was found 24 hours following the direct administration of L-655,708 into the ventral hippocampus (Bugay et al., 2020). Nonetheless, while the current study showed no change in mean firing activity of 5-HT and NE neurons, alterations in 5-HT and NE receptor functions in postsynaptic areas may contribute to the antidepressant-like effects and warrant further investigation.
While both L-655,708 and ketamine can increase DA population activity, ketamine does so acutely, and its action dissipates after 24 hours, whereas the effects of L-655,708 become apparent after 24 hours. While the enhancing effect of L-655,708 is sustained for up to 1 week with a single injection, that of ketamine requires 2 weeks of repeated injections to produce a sustained increase, and it lasts only 3 days (El Iskandrani et al., 2015; Iro et al., 2021). The delay in onset of L-655,708 compared to ketamine remains to be determined. However, although rapid synthesis of hippocampal BDNF/TrkB is suggested to be involved in the rapid antidepressant response to ketamine (Autry et al., 2011; Adachi et al., 2008), these proteins did not increase in the hippocampus 30 minutes and 1 day after L655,708 administration (Bugay et al., 2020). It is important to note also that in the clinic, some patients respond to ketamine only after hours or days, and this is considered as a rapid therapeutic response (Phillips et al., 2019).
In summary, the present study demonstrated that a single injection of L-655,708 produced a sustained increase in the population activity of VTA DA neurons that lasted for up to 1 week. These results indicate that modulation of α5-GABAA-NAM could exert a rapid and sustained antidepressant action, in part, by prolonging enhanced DA transmission through AMPA activation.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251405691 – Supplemental material for α5-GABAA allosteric modulation triggers and prolongs dopamine neuronal activity via AMPA receptors
Supplemental material, sj-docx-1-jop-10.1177_02698811251405691 for α5-GABAA allosteric modulation triggers and prolongs dopamine neuronal activity via AMPA receptors by Areebah Ahmed, Mostafa El Mansari and Pierre Blier in Journal of Psychopharmacology
Footnotes
Author contributions
M.E.M. and P.B. designed the study and wrote the protocol. A.A. carried out the experiments and undertook the statistical analysis. A.A., M.E.M., and P.B. wrote the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Canadian Institutes of Health Research (grant PJT-169078 to P.B.).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.A. and M.E.M. declare no conflict of interest. P.B. received grant funding and/or honoraria for lectures, expert testimony, and/or participation in advisory boards for Allergan, Eisai, Idorsia, Janssen, Lundbeck, Otsuka, Pfizer, Valeant, and Takeda.
Supplemental material
Supplemental material for this article is available online.
