Abstract
Introduction:
This review provides an overview of Roland R. Griffiths’ history of research, and his mentoring and collaborating approach to science that contributed to his impact in behavioral and neuropsychopharmacology and psychedelic medicines development.
Approach:
The approach was to summarize studies in his major domains of research, including preclinical and clinical abuse liability assessment science, alcohol, benzodiazepines, caffeine, tobacco, and psychedelics. All the authors of this review were mentored by and collaborated with Griffiths—some over several decades—and were able to provide personal perspectives and insights into Griffiths’ approach to science and scientific collaborations, including insights into how major research initiatives were conceived and evolved with personal anecdotes and quotes.
Overview:
Roland Griffiths is widely described as a “scientist’s scientist,” driven by his powerful curiosity to explore new frontiers in behavioral biology and neuropharmacology, with a passion to pursue humanity-serving science. His methodical approach to research development and then systematic extension and assessment of the generalizability of findings contributed to the evolution of thinking and scientific methods for abuse liability assessment, policy, and regulation of alcohol and other sedatives, tobacco and nicotine, caffeinated products and other stimulants, and in his last 2 decades, psychedelics. His inclusive and collegial approach to science, mentoring, and collaborating fueled his creativity and productivity and a fountain of innovation and research that will go on in perpetuity. Nowhere is this more evident than at the Johns Hopkins Center for Psychedelic and Consciousness Research established in the last few years of his life, in part because of his remarkable scientific life.
Introduction
The Johns Hopkins University Center for Psychedelic and Consciousness Research (CPCR) was established in 2019 with Professor Roland R. Griffiths as its founding director. It was the first such research center in the United States and is currently the largest in the world. Its short history has had three endowed professorships established in psychedelic- and consciousness-related research. How this came about was unquestionably only possible due to the innovative, systematic, and rigorous science that Dr. Griffiths brought to a field of research that was fraught with more than half a century of controversy. This review traces the path from his training and through several areas of his pioneering research that laid the foundation for his approach to psychedelic research.
At the Johns Hopkins University School of Medicine, Griffiths was Professor of Behavioral Biology in the Department of Psychiatry and Behavioral Sciences and Professor of Neuroscience in the Department of Neuroscience. In his last year of life, he was honored as the inaugural recipient of the Oliver Lee McCabe, III, PhD Professor in the Neuropsychopharmacology of Consciousness.
Griffiths led the development of a psychedelic and consciousness research program that was distinguished by scientific innovation and excellence, with a strong commitment to the safety of participants. This approach contributed to the 21st-century global renaissance in psychedelic research. It also demonstrated to the leadership of the National Institutes of Health (NIH), including its National Institute on Drug Abuse (NIDA), that psychedelic research could be conducted safely and yield important neuropsychopharmacology science with potential applications to major brain-related medical disorders including major depression and addiction (Henningfield and Belouin, 2024; Moghaddam, 2023; Xi et al., 2023). The Johns Hopkins psychedelic research program contributed to the global renaissance in psychedelic research, but Griffiths was always first to say that his research program was built on the foundation of earlier research and in collaboration with many other researchers at Johns Hopkins and beyond.
However, it is less known that before psychedelic research, Griffiths was also a pioneer in several other classes of psychoactive drugs and contributed substantially to the science of abuse liability assessment of central nervous system-acting drugs in general. Understanding this history and recognizing his rigorous approach to research helps make clear the powerful impact of his first published clinical psychedelic study in 2006, and follow-on studies that occurred over the next few years (Griffiths et al., 2006, 2008, 2011; Griffiths and Grob, 2010).
Griffiths’ impact was magnified by his approach to mentoring and collaboration, which he attributed in part to several of his own mentors. At his core, his deep sense of curiosity and interest in exploring the unknown led to his evolution in thinking and research that spanned a variety of domains.
He was an intense listener, and that included asking his colleagues about their families, which colleagues came to realize, then gave him the opportunity to brag about his own children—perhaps the only bragging that anyone ever heard from him, except when it was to promote his own mentees in lectures and for awards and promotions. Such sharing of personal life stories contributed to lasting bonds and deep connections with many of the more than 200 fellows and collaborators through the Johns Hopkins Behavioral Pharmacology Research Unit (BPRU) post-doctoral training program.
With respect to novel or unexpected findings, Griffiths’ approach was almost always characterized by measured skepticism. This was demonstrated by his frequent questions about the reliability and generalizability of the findings and the need for further investigation. This was sometimes expressed by questions such as “Is this real?” “How robust are the findings?” and “What is the most important next step?” Thus, in each area of his scientific focus, there emerged a cohesive series of studies systematically planned to assess the reliability and generalizability of the findings. To him, psychedelic research was to be approached not only as a systematic path that included four decades of training, research, mentoring, and collaboration in other areas of neuropsychopharmacology science but also fueled by his passionate curiosity and love of humanity-serving discovery that came before his psychedelic research. This, “what,” “how,” and “why” in thinking and planning studies with colleagues about a new wonder of nature to take on was often preceded and followed by “this is going to be fun,” in a manner that he attributed to his mentor, George Bigelow.
This sentiment was carried through to his last collaborations, with the emphasis that when approaching questions with the utmost rigor and seriousness, research should fundamentally be driven by the joy of discovery, and we should not lose sight of the immense excitement of generating new knowledge and contributing to new discoveries.
As a scientist, Dr. Griffiths epitomized the scientific approach that examines the unknown in pursuit of understanding and characterizing phenomena, then furthers and follows the science and not theoretical or preconceived expectations (Brady, 1958, 1992; Sidman, 1960; Thompson and Schuster, 1968). He seemed to take as much delight in findings that were contrary to initial hypotheses as in those that seemed consistent with what may have been expected. For example, this was evident in his leading work to better understand the behavioral neuropsychopharmacology of caffeine and nicotine, neither of which was widely accepted as carrying the potential for dependence, as were the prototypic addictive substances such as opioids, stimulants, depressants, and other drugs that were widely accepted as addictive and regulated as controlled substances. As discussed further in this review, initial findings suggesting similarities with prototypic dependence-producing substances were followed by many additional studies, and his research supporting the conclusions that both caffeine and nicotine could be understood as addictive by the same standards as many controlled substances.
Approach and scope of this review
This narrative review aims to provide a sense of the scope and scale of Dr. Griffiths’ work, and includes a discussion of his various overlapping interests, the evolution of his research, and his major contributions and accomplishments. While not comprehensively summarized here, his contributions and various interests have been described in more than 400 mostly peer-reviewed articles published over nearly six decades. In addition, this review provides a brief overview of Griffiths’ training, early mentors, and their influence on his approach to science, mentoring, and collaborating.
All authors of this review worked directly with and learned from Griffiths for at least several years, and in some cases, decades, in what were then new areas of exploration in various research fields. The authors also learned from and appreciated Dr. Griffiths’ efforts to find balance between science, family, friends, and other aspects of life, which seems to be a constant struggle among many who are highly committed to research and discovery. So, it is important to note that Dr. Griffiths was very personable and caring, engaging in social events, and eager to share his activities with and love for his family, especially his children. Several of the authors remember Dr. Griffiths’ summertime reminders during his children’s early years that he would be largely out of touch for up to two weeks on Winnebago camper van trips across the United States and also recall his sharing of experiences post-trip.
Although the recognition of his role as a major scientific pioneer and leader in the 21st-century renaissance of psychedelics is well-deserved, part of the purpose of this review is to remember and learn more from his other domains of research, which provided the foundation for much of his thinking and scientific approach to deciphering the mysteries underlying the neuropsychopharmacology of psychedelics and consciousness.
His research domains are characterized by a cohesive series of studies in each of several areas of focus, often fueling ideas for subsequent areas of focus from the 1960s through the early 2020s. Each of these research domains has been impactful in advancing the science and applications to medicine, public health, policy, and education.
The studies conducted in each domain varied in size by the numbers of publications and years of focus. For example, most of his alcoholism research was conducted in the 1970s and was captured in only six articles but had a substantial impact on the field of study. They contributed to the understanding of alcohol use disorder as an orderly neurobehavioral process involving a substance that, with reinforcing and other stimulus effects, could be modulated by behavioral contingencies, social, and other factors (Bigelow, 2001; Griffiths et al., 1975a, 1976a, 1976b, 1981). His research conducted under the umbrella of drug abuse liability (i.e., methods development and application) spanned his entire career and continues to influence new methods that are under development for the application of psychedelics and other novel acting substances. His focus on psychedelic research occurred over more than two decades, and at his death, the influence of his work was and remains on an accelerating trajectory.
Given the volume and breadth of published articles across Dr. Griffiths’ various domains of interest, the authors have distilled what they feel are the most impactful within these domains into summary tables, culminating with an overview of his psychedelic and consciousness research. Specifically, (1) animal abuse liability research with a focus on benzodiazepines; (2) human abuse liability research and methods development; (3) behavioral pharmacology of alcoholism; (4) behavioral pharmacology of cigarette smoking and nicotine; (5) abuse liability and physical dependence potential of caffeine; and (6) psychedelic research and medicines development and beyond.
Neuropsychopharmacology training, mentors, and mentoring
Roland Griffiths was born in Glen Cove, New York, on July 19, 1946. In his second year, his family moved to the Oakland-San Francisco Bay area of California. His father was a psychologist, health educator, and faculty member at the University of California, Berkeley. Griffiths had an early interest in science and was drawn to small, intimate colleges rather than large universities. He enrolled in Occidental College in Los Angeles, in 1964, initially planning to major in engineering but later shifting to psychology (Stutz, 2023).
Occidental College, Los Angeles, California
Psychology classes by Professors David Cole and Luther Jennings sparked his interest in behavioral science and “changed everything” (Stutz, 2023). In a profile article in Occidental Magazine, he commented on the impact of his first psychology course. “I became fascinated and got pulled into psychology” and “I had always been interested in the physical sciences and figuring out how things work,” but in those psychology courses, “I was immediately drawn to experimental psychology.” Years later, he took pride in mentioning his father’s involvement in public health that included advising on one or more of the 1970s U.S. Surgeon General’s reports on the health consequences of smoking. He clearly appreciated the public health impact that psychologists could have.
His first published paper with Dr. Jennings and his Occidental College roommate Barry Sears was titled “Specificity of transfer of a learned response by intracisternal injection of brain extract from trained rats: negative findings” (Griffiths et al., 1969). It helped resolve a mid-1960s claim with mixed findings across studies that specific learned behaviors could be transferred by the injection of RNA from trained rats to naïve rats. Their well-controlled study did not find performance differences between rats that were injected with extract from trained rats as compared to rats injected with extract from untrained rats or saline.
He graduated from Occidental College in 1968 with a Bachelor of Science in Psychology, with honors. He applied to the Peace Corps but was recruited to the Psychopharmacology Training Program at the University of Minnesota and was awarded a United States Public Health Service fellowship.
University of Minnesota Psychopharmacology Training Program
The University of Minnesota Psychopharmacology Training Program (later renamed the Neuro-Behavioral Pharmacology Training Program) was the preeminent program established to train generations of researchers in addiction science. It was established with initial funding from the National Institute of Mental Health of a grant application led by Travis Thompson and Gordon Heistad and was expanded as part of President Nixon’s efforts to address drug abuse and addiction through a multidisciplinary education approach, as well as research in the fields of pharmacological and behavioral sciences. It was based in the Psychiatry Research Unit of the Department of Psychiatry in the Medical School. The program’s leadership, and Griffiths’ advisor, included the prominent behavioral pharmacologist Travis Thompson, who previously developed the primate intravenous drug self-administration model for assessing drug reinforcement with Charles R. Schuster in the laboratory of Joseph V. Brady at the University of Maryland (Thompson and Schuster, 1964).
The psychopharmacology fellows in this program were required to take courses and participate in seminars in pharmacology, psychology, and psychiatric diagnosis. Most of those directed by Thompson, including Griffiths, Bigelow, and Henningfield, also participated in behavioral analysis and modification efforts at various state mental institutions. Working with seriously disadvantaged patients with mental illness and experiencing the joy of helping to make their lives better, through behavioral counseling and contingency management, provided lessons relevant to the conduct of clinical research in general, including the importance of rigorous monitoring and careful manipulation of experimental variables. It also made clear to Griffiths the real-world humanitarian significance of science that could contribute to the treatment of behavioral- and brain-related disorders and a dedication to pursue research that could enhance health and well-being (e.g., Bigelow and Griffiths, 1972, 1974; Pickens et al., 1973; Thompson et al., 1973).
The main animal behavioral pharmacology laboratory in which Griffiths completed his PhD thesis research focused on methods that would become the mainstay of preclinical abuse potential assessment to guide drug scheduling, as required by the 1970 U.S. Controlled Substances Act (CSA), and contribute to the development of new medicines. The methods incorporated in this laboratory included intravenous drug self-administration studies, pioneered by Thompson and Schuster, as well as the emerging oral drug self-administration model led by Richard Meisch and Thompson. Additional methods included the drug discrimination model for comparing the interoceptive perceptions of substances, the persistence of drug-seeking behavior after the drug was no longer available (Griffiths and Thompson, 1973a, 1973b), and a variety of other methods then referred to as behavioral pharmacology (Thompson and Schuster, 1968).
The programs at the University of Minnesota and Johns Hopkins fostered and nourished Griffiths’ approach to neuropsychopharmacology science involving animals and humans. Primary outcome variables of his human research often included self-reports of interoceptive states produced by drug administration—states that were modulated by a broad range of individual, genetic, and environmental factors that included social, cultural, and other influences. Griffiths’ insatiable curiosity, along with his measured skepticism of new findings and theories by other researchers and his own studies, motivated much of his continuously advancing and evolving research.
Most studies were accompanied by questions about the reliability and generalizability of the findings and the need for further investigation in the traditions of his own mentors and earlier pioneers in behavioral neurobiology research (e.g., Hodos et al., 2012; Sidman, 1960; Thompson and Schuster, 1968). This may have contributed to Griffiths’ remarkable record for high levels of persistent research funding by federal agencies as well as philanthropists, and sometimes pharmaceutical developers over the decades.
The empirically driven experimental approach taken by the Minnesota program was an ideal match for Griffiths’ curiosity and love of discovery. He was as enamored by the philosophy of science as the science itself. As espoused by senior faculty in their training and writings (e.g., MacCorquodale and Meehl, 1948; Skinner, 1953; Thompson and Schuster, 1968), he loved to discuss the distinctions between hypothetical constructs with little empirical basis from intervening variables that helped explain the relationships between interventions (e.g., drugs and behavioral contingencies) and their effects on observed behavior and interoceptive responses (e.g., mood and feeling). This may have contributed to his later interest and efforts in applying neurobiological approaches to the study of consciousness, spirituality, and well-being. As a practical matter, he also applied the same principles to increasing his own productivity, tracking target behaviors such as studying and later writing, with timers, and recording his performance and trends, as did some of the other psychopharmacology program fellows.
Over the decades, it became increasingly clear that his early training and diligent approach to science brought him success and credibility. The aphorism “success breeds success” described him well. Griffiths understood that in science, success is not usually measured by “what” the findings are, but whether they are replicable, valid, credible, and lead to a better understanding of nature. In the biomedical sciences, if the findings also contribute to advancing public health, well-being, disease prevention and treatment, and questions of broad interest, then funding organizations and peer reviewers of grant applications are often more willing to provide further support.
As discussed further in the summaries of his research domains addressing tobacco, caffeine, benzodiazepines, and most recently, psychedelics, many of Griffiths’ research programs persisted with support for multiple 3- to 5-year grant cycles, and the postdoctoral training program is now in its fifth decade. From these perspectives, much of Griffiths’ work was considered “science-advancing” and “humanity-serving” and therefore successful. The fact that he was a pleasure to work with fueled further opportunities for collaboration and further expansion of his research with diverse and younger generations of scientists eager to work with him.
Although Thompson was Griffiths’ PhD program mentor, their time spent together at the University of Minnesota also gave birth to a life-long friendship and highly productive collaborative relationship with George Bigelow, whom Griffiths also referred to as his mentor. When Griffiths arrived at the University of Minnesota, George Bigelow was two years into his fellowship in the same program, also under the direction of Thompson. Griffiths often referred to Bigelow as not only a dear friend and colleague but also an important mentor, and he liked to think of himself as Bigelow’s first mentee. In March 2018, following a ceremony that recognized Bigelow with The Johns Hopkins University School of Medicine Dean’s Distinguished Mentoring Award, Griffiths made the following comment in an email to Henningfield and several other Hopkins faculty: In the Fall of 1968, I arrived in Minneapolis to begin my graduate training in psychopharmacology at the University of Minnesota. In the first few days of that program, senior students gave presentations of their ongoing research to all of the incoming students so we could decide what projects and with whom we wanted to get our first research experience. George [Bigelow] was conducting a fascinating project examining drug self-injection behavior in free-roaming rhesus monkeys housed in a social environment in which they could interact. Immediately after the presentations, I rushed over to George expressing my interest, and we have been working together in adjacent offices for the past 50 years. He has been my mentor ever since and I still, not infrequently, drop by his office seeking his advice on research.
In 1970, Bigelow was awarded his PhD and joined The Johns Hopkins Department of Psychiatry and Behavioral Sciences, Division of Behavioral Biology (DBB), under the Division founder and director, Joseph V. Brady. Upon Griffiths’ completion of his PhD in 1972, he was recruited to the same program by Bigelow, with support from Brady. He and Bigelow collaborated on research and trained new generations of scientists over five decades with Maxine Stitzer, another founder and co-leader of the BPRU as discussed below.
Johns Hopkins University School of Medicine, Department of Psychiatry and Behavioral Sciences, DBB, and the BPRU
Griffiths’ arrival at Johns Hopkins in 1972 was just a few years after Brady had joined Hopkins, in which his initial efforts included contributing to adding “and Behavioral Sciences” to the name of the Department of Psychiatry. Within this department, he established the DBB Program, which included an animal laboratory on the downtown Hopkins Hospital-centered campus, and a human research program on the Baltimore City Hospitals campus in east Baltimore (later assimilated into Johns Hopkins University as the Bayview Medical Center). Brady also established the Behavioral Biology Professorship tract, which Bigelow and Griffiths, as well as some authors of this article, came to hold.
The human research program eventually came to include Maxine Stitzer and Ira Liebson, the latter serving as its medical director as well as research collaborator. In 1977, the Bigelow, Griffiths, Liebson, and Stitzer foursome developed an application to NIH for a postdoctoral training program (T32), and this was approved and funded in 1978. The application required a name for the section or unit in which it would be based and a director. They decided it would be called the BPRU with Bigelow as its director. The founders collaborated across decades and by 2020 had more than 200 postdoctoral fellows, most remaining for 2–4 years. Many of the fellows went on to become leading scientists in their own right, primarily at other institutions. Some, however, remained at Johns Hopkins for several decades, including some of the co-authors of this review. Other BPRU fellows took leading roles in federal agencies, including the Centers for Disease Control and Prevention (CDC), Drug Enforcement Administration (DEA), Food and Drug Administration (FDA), NIDA, and other institutions, and some in private sector pharmaceutical development.
The enduring legacy of Brady and his mentees in behavioral biology and behavioral pharmacology was furthered by the establishment of The George E. Bigelow PhD Professorship in 2014. Its inaugural recipient was BPRU member and present BPRU Director, Eric Stain. As will be discussed further on in this review, part of the key to Griffiths’ impact in the 21st-century psychedelic renaissance was his rigorous application of behavioral pharmacology and neuroscience methods to the study of psychedelic substances and their effects on behavior and consciousness, as well as therapeutic applications.
From his arrival in the 1970s until the 1990s, Griffiths split his time between the downtown and Bayview campuses, allowing him to be actively involved with his postdoctoral fellows and collaborators in both animal and human research. During the 1990s, most of the DBB was consolidated into a temporary building on the Bayview Campus and later relocated to a building dedicated to and named in honor of Brady, as the Joseph Vincent Brady Behavioral Biology Research Center Building at Johns Hopkins Bayview Medical Center, on June 2, 2017.
The CPCR was officially launched by and administratively based in the BPRU, and at the time of this writing, both are housed in the Joseph Vincent Brady Behavioral Biology Center Building.
Griffiths’ mentees. Fostering diversity, equity, and inclusion
Griffiths’ mentees came from diverse backgrounds and often went on to achieve scientific prominence. His mentorship style was attentive and empathetic. For example, he loved to hear about the interests of his mentees, resulting in frequent hours-long and wide-ranging conversations. Griffiths’ leadership approach included one-on-one discussions of the research purpose and approach, as much to get the thoughts of his mentees as to make clear what was expected in pursuit of safe, ethical, and reliable research. Griffiths’ mentees and collaborators, including the coauthors of this review, knew that they could count on him for advice and support, including supporting letters as their own careers developed.
He appreciated diversity in thinking and the innovations that this approach sometimes afforded research and the interpretation of findings. He was also active in the establishment of programs to enhance excellence, as well as diversity, equity, and inclusion in scientific organizations, including the American College of Neuropsychopharmacology and College on Problems of Drug Dependence, where he was active in the establishment of its Under Represented Populations committee in the early 1990s (Henningfield et al., 1994). He was vocal in making clear that he viewed such efforts as not only justifiable from a humanitarian perspective but also as vital to fostering innovation in research, including the relevance of this research to both local and global human diversity. This focus on diversity was indeed part of the culture of the Bigelow, Griffiths, Stitzer-led BPRU, in which numerous fellows made important contributions to the diversity, equity, and inclusion pipeline of public health-serving scientists and clinicians.
Such individuals included Albert Garcia-Romeu, who remains especially active in efforts to ensure that psychedelic research embodies the spirit of diversity and acknowledges indigenous traditions across the scientific pipeline of development (Garcia-Romeu and Richards, 2018; Hughes and Garcia-Romeu, 2024; Jones et al., 2025; Ortiz et al.2022; Thrul and Garcia-Romeu, 2021). Other individuals included Bridgett Garrett (who headed a Minority Health program at the CDC); Edward Singleton (whose efforts led to diversity programs at the NIDA Intramural Research Program (IRP), CPDD, and the American College of Neuropharmacology); as well as Jean Lud Cadet and Beny Primm, who were frequent collaborators in these efforts (Henningfield et al., 1994, 2020).
Griffiths’ diversity and inclusion efforts included global research collaborations and efforts to foster more equitable and reciprocally beneficial efforts with Indigenous Peoples with respect to traditional medicine practices such as those involving psychedelic substances. This is exemplified in his several-year collaboration on the topic with Veronica Magar and others (Urrutia et al., 2023). Magar et al. (2023, 2025) continued this endeavor in subsequent efforts and publications, including in this special issue of the Journal of Psychopharmacology.
Others whom Griffiths frequently acknowledged included his own life-changing mentors who greatly contributed to his thinking, career development, and innovations. There were many, but George Bigelow and the late Travis Thompson and Joseph Brady were clearly prominent in their influence. Their approaches were unique but shared common threads, which is not surprising given that Brady was an important mentor to Thompson in the early 1960s, and Thompson to Bigelow in the same decade.
To those of us who have had the good fortune to have been mentored by two or even all three of these pioneers, it is clear that they all shared the traits of kind, thoughtful, and empowering mentors who stimulated and reinforced curiosity, set high standards for excellence, and a frankly “fun” approach to research design and planning which Griffiths claimed to have “inherited” from Bigelow. It is an approach that has been embodied by many of the mentees of Griffiths and his colleagues. 1 This approach included the importance of following the data as they emerged and evolved, and taking as much, if not more, delight in unexpected discoveries than research that reinforced earlier thinking.
Like his mentors, Griffiths was generous in offering opportunities to coauthor articles in which authorship order was generally determined by level of contribution and not independent stature and other factors. He also encouraged his mentees to collaborate with others without any explicit or implicit expectation that he should be included. This is in contrast to many research centers in which the founder(s) and principal investigators of grants routinely co-author most, if not all, of the articles in their programs regardless of their personal level of input. That was not the approach of Drs. Brady, Bigelow, and Thompson, and it was not the approach of Griffiths. All of the authors of this article published much of our initial work with Griffiths in areas that he had initiated with us, but all of us also collaborated with others and co-authored less with Griffiths in those areas over time. This was in part due to Griffiths’ philosophy that authorship should reflect a greater level of tangible collaboration and input that he felt he could provide or had already provided. Griffiths was the prototype of “the good colleague” described in the book by the prominent neuroscientist, Michael J. Kuhar titled, The Art and Ethics of Being a Good Colleague (Kuhar, 2020).
Animal abuse liability with a focus on benzodiazepines
Among Griffiths’ most far-reaching and enduring contributions were his studies that advanced the scientific basis of preclinical and human abuse potential assessments. 2 This work began in response to the 1970 U.S. CSA requirements for evidence-based drug scheduling, which also guides drug scheduling under the international drug control treaties (United Nations Office on Drugs and Crime, 2020). The CSA provides a framework, including evaluation of eight factors, 3 that are determinative of whether medicines recognized by the FDA for therapeutic use should be placed in the CSA, and if so, which schedule is most appropriate based on the scientific evidence. 4
For new chemical entity based medicines, Factors 1–3 provide a basis for comparing the relative abuse potential to that of previously scheduled drugs based on behavioral and pharmacological studies; Factor 7 is focused on “psychic” or psychological and physiological dependence potential in humans; Factors 4–6 are focused on the potential public health risks based on the relevant history of the substance under evaluation and similar substances, with consideration given to projected patterns of abuse, misuse, addiction, overdose, and other potential abuse-related risks. Potential public health benefits of a given schedule may also be considered, for example, less restrictive scheduling of lower abuse potential drugs for pain, sleep, and daytime sleepiness that may incentivize prescribing of these drugs instead of drugs with higher abuse-related risks.
For new chemical entities, in which there is no history of real-world use, the estimation of abuse potential in Factor 1 (“Its actual or relative potential for abuse.”) is guided strongly by dedicated animal and human abuse potential studies; most prominently and generally initially, by various intravenous self-administration approaches to assess reinforcing or rewarding effects, and drug discrimination approaches to characterize the interoceptive effects—both in comparison with already scheduled substances of abuse. Griffiths’ studies, in collaboration with Brady, Hienz, and then Ator, Lukas, and others, systematically advanced the approaches that emerged during the 1960s, including the primate self-administration model of Thompson and Schuster (1964) and the rodent self-administration model of Weeks and Collins (1964).
Table 1 provides the first authors, year of publication, and titles for some of Griffiths’ preclinical abuse potential studies that accelerated progress in the field. It is noteworthy that this table covers the 25-year span that preceded his first involvement with psychedelic research, which relied in part on his approach to human studies of abuse potential assessment.
Preclinical abuse liability assessment studies.
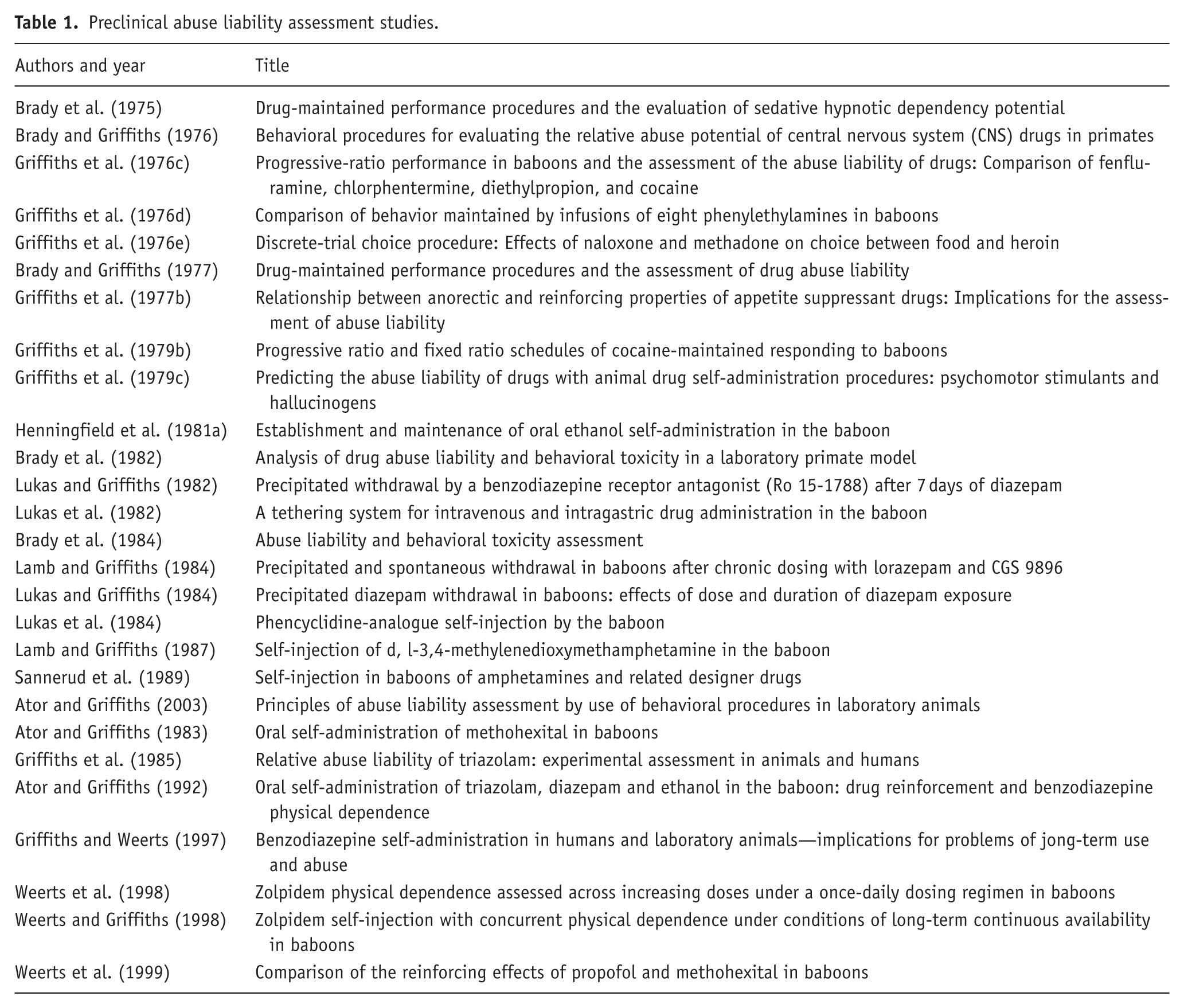
In addition to controlling access to medicines commensurate with their potential for abuse and public health consequences, the CSA provides an incentive for developing and prescribing potentially safer and less likely to be abused medicines than legacy medicines in the same therapeutic categories (e.g., for treating anxiety, epilepsy, insomnia, obesity, pain, and substance use disorders). All of this assumed a well-established, reliable scientific methodology that was predictive of real-world potential for abuse and addiction. However, in the early 1970s, abuse potential science was in its infancy, with much of the earlier abuse potential assessment research focused on opioids.
The practical needs of developing the science to guide CSA drug scheduling, along with funding from both NIMH and the DEA to conduct such research, provided an important context for Griffiths’ early research at Johns Hopkins. Support by NIDA began in the mid-1970s following NIDA’s establishment in 1974. Griffiths was well prepared by his University of Minnesota Psychopharmacology Training Program experience, which had been rich in methods addressing the reinforcing and other effects of drugs that contributed to their abuse potential. Fortuitously, when Griffiths arrived at Johns Hopkins, Brady was in the process of securing funding from both the DEA and NIMH, focusing on the development of abuse potential assessment methodology. Thus, as interested and committed as Griffiths was to collaborate with Bigelow and others in clinical behavioral pharmacology at the Bayview campus, he was also eager to continue his preclinical animal behavioral pharmacology research in the Brady-directed program.
DEA funding to the DBB was initially focused on the further development of abuse potential assessment methods, such as evaluating the reinforcing effects of stimulants and hallucinogens that were part of the focus of the CSA, as well as its international counterpart, the 1971 United Nations Convention on Psychotropic Substances (e.g., Griffiths, et al., 1979c; United Nations, 1971). In addition to its interest in validating methods for preclinical assessment of stimulants, sedatives, and hallucinogens, the DEA also provided funding for evaluating the “side-effects of drugs of abuse” including evaluation of their physical and behavioral toxicities (e.g., altered reaction time, risk taking, and auditory and visual thresholds; Brady et al., 1979; Hienz et al., 1981). These approaches came together with studies that evaluated the relationship between reinforcing effects as well as their sensory/motor toxicity to develop ratios of what these investigators were increasingly referring to as their liabilities for use and abuse, that is, their abuse liability (e.g., Brady and Griffiths, 1983; Brady et al., 1983, 1984). The titles of these and other animal behavioral pharmacology studies related to abuse liability are included in Table 1.
Establishment of oral drug self-administration model in baboons
In 1978, Griffiths et al. received grant funding from NIDA to evaluate benzodiazepines and newer generations of drugs for the treatment of anxiety and epilepsy. For these drugs, there was interest in establishing an oral model of self-administration in baboons because both therapeutic and nonmedical use of these drugs was overwhelmingly by the oral route. Ator and Henningfield joined the DBB faculty at Johns Hopkins in mid-1978 and formed a three-way collaboration with Griffiths to establish the oral primate (i.e., baboon) drug self-administration model previously developed by Meisch and Thompson in the Minnesota behavioral pharmacology laboratory in rhesus monkeys (Meisch et al., 1975). Following similar protocols used at Minnesota, the first study at Johns Hopkins involved ethanol as the prototype sedative (Henningfield and Meisch, 1976; Meisch et al., 1975). The protocol was readily adapted to baboons and formed the backbone for yet another behavioral assay that served several decades of research in the DBB (Henningfield et al., 1981a). Drs. Ator, Griffiths, Weerts and other colleagues developed a prolific program for assessing the abuse liability of a variety of sedative hypnotics in the oral self-administration baboon model as well as drug discrimination methods (Ator and Griffiths, 1983, 1985, 1987; Griffiths and Weerts, 1997; Weerts et al., 1998, 1999). Whereas the oral self-administration model was ideal for some studies, the mainstay preclinical model for most of the dozens of substances evaluated for abuse liability since the late 1970s has been intravenous drug self-administration, and this approach was employed in baboons, with the baboon research ending at Johns Hopkins in 2025, leaving the rodent model as the mainstay approach for assessing reinforcement potential.
By the 1980s, abuse liability studies were being used by Griffiths and colleagues to better understand the reinforcing potential of so-called “club drugs,” such as methylenedioxymethamphetamine (MDMA; Lamb and Griffiths, 1987); occasionally abused over-the-counter drugs such as phenylpropanolamine (Lamb et al., 1987), and nicotine (Ator and Griffiths, 1983). In addition to studies of MDMA, which has hallucinogenic effects, the abuse liability and behavioral pharmacology of novel phenethylamines (Sannerud et al., 1996) and several phencyclidine analogues were studied (Lukas et al., 1984).
Elise Weerts was accepted into the DBB as a postdoctoral fellow under the supervision of Griffiths in 1993, focusing on animal abuse liability and related research. Weerts and Griffiths continued to collaborate on a series focused on gamma-Hydroxybutyric (GHB) acid and prodrugs gamma-butyrolactone (GBL) and 1,4-butanediol (1,4-BD) in the intravenous baboon paradigm (e.g., Goodwin et al., 2011). Over the past nearly three decades, she helped to expand the benzodiazepine and related drug program, as well as research on GABAergic modulators (acts on gamma-aminiobutyric acid), GHB, cannabinoids, and more. In the typical fashion that is characteristic of Dr. Griffiths’ mentees, this work was in collaboration with a variety of other DBB and BPRU postdoctoral fellows, including Goodwin, Sannerud, and others.
Drug self-administration studies remain vital because this is generally the most reliable model for assessing the likelihood of nonmedical recreational substance use in novel CNS-active medicines in development (Heal et al., 2023; Henningfield et al., 2025; Weerts et al., 1998, 1999). With continued technological development, drug self-administration methods in rodents have continued to evolve. In keeping with Dr. Griffiths’ tradition of leading research on novel drugs, the DBB research has expanded to include oral dosing studies and vapor self-administration in rodents to increase understanding the behavioral pharmacology and reinforcing effects of cannabis constituents (Moore and Weerts, 2022, 2025; Moore et al., 2021, 2022, 2023a, 2023b; Jenkins et al., 2023, 2025; Weerts et al., 2024). In fact, the rodent intravenous drug self-administration model may be more reliable than human abuse potential studies for many of the novel acting substances increasingly in the drug development pipeline, as discussed in another article in this special issue (Henningfield et al., 2025).
Establishment of physical dependence and withdrawal in baboons and the seminal characterization of the diazepam withdrawal syndrome
An important abuse liability issue related to the growing trend towards chronic benzodiazepine use was related to emerging concerns in the 1970s and 80s that drugs like diazepam and possibly others had greater physical dependence and withdrawal potential than was assumed when they were approved by the FDA. In fact, their initial approved labeling did not warn about potential withdrawal symptoms upon discontinuation of dosing (see review by Lerner and Klein, 2019).
In 1979, Scott Lukas joined the Behavioral Biology Division at Johns Hopkins under the joint mentorship of Brady and Griffiths to focus on abuse liability assessment development. As recounted by Lukas, the development of the baboon withdrawal model came about in a similar way as many of Griffiths’ new initiatives—driven by his curiosity and desire to explore emerging issues of interest. According to Lukas, it was a dark and stormy evening in July 1981, and Lukas and Griffiths were in the hallway outside the baboon laboratory discussing research. Lukas told Griffiths that when baboons that had been chronically self-administering diazepam were switched to intravenous cocaine, they were displaying greater agitation and “hyperactivity” than was expected in baboons that were well accustomed to cocaine. Moreover, the agitation was even greater when they were switched from a benzodiazepine to a vehicle control, as was done when it was time for a full body health evaluation check that was regularly performed on the animals for health and safety reasons.
These observations bore similarities in clinical reports of agitation and distress in some patients with anxiety disorders who discontinued their benzodiazepines. Still, it was generally assumed that this was the re-emergence of their anxiety disorder symptoms and not a rebound withdrawal syndrome.
Griffiths and Lukas agreed that such an explanation seemed unlikely in baboons that were healthy and without evidence of anxiety disorders. They wondered if diazepam might produce such a sufficiently strong physical dependence that discontinuation of drug administration would precipitate a true withdrawal syndrome. According to Lukas, Brady came upon them in the hallway and joined the discussion. Within a few minutes, they all agreed that this merited exploration. For additional context, it had only been about 6 years earlier that the opiate receptor was discovered (Pert and Snyder, 1973) and only about 4 years earlier that a high-affinity binding site for benzodiazepines had been discovered (Möhler and Okada, 1977; Squires and Braestrup, 1977). Thus, the concept of agonists, antagonists, partial agonists, etc., was still in its infancy, but potential parallels of opioid and benzodiazepine pharmacology were under exploration in other laboratories. It was also known that the pharmaceutical developer, Hoffman La Roche was developing novel benzodiazepines and that some seemed to have antagonist-like effects.
A few days later, Griffiths announced that he was in touch with a representative from the Swiss pharmaceutical company Hoffman La Roche, who expressed a willingness to provide a novel benzodiazepine receptor antagonist for evaluation of its abuse liability and related pharmacology. It was an experimental drug called Ro 15-1788 that would later be known as flumazenil for use in benzodiazepine overdose.
Their first study evaluated its potential to precipitate withdrawal in diazepam-treated baboons, modeling the experiments after those that used naloxone to precipitate opiate withdrawal. Diazepam was delivered intragastrically by an approach invented by Griffiths and Lukas for the purposes of this study using a modified drug reservoir (Lukas et al., 1982). The drug reservoir was modified by the addition of an infusion pump that could steadily deliver diazepam at a dose of 20 mg/kg, intragastrically (i.g.) for 45 consecutive days.
Ro 15-177 was administered intragastrically via catheter, as Griffiths and Lukas observed the baboon (subsequent experiments involved blinded observer monitoring). They watched intently for any changes in the animal’s behavior. Initially, there was no observable change in the behavior; the baboon was very quiet and ignored them for 5, 10, and 15 minutes. At the 18th minute, the baboon began to retch, assumed a bent-over posture, and started to shake and then vomit (see Figure 1 for a visual characterization). They were stunned. At that moment, according to Lukas, Griffiths leaned over his shoulder and with a quirky tone in his voice said, “Scott, this is a Science paper.”
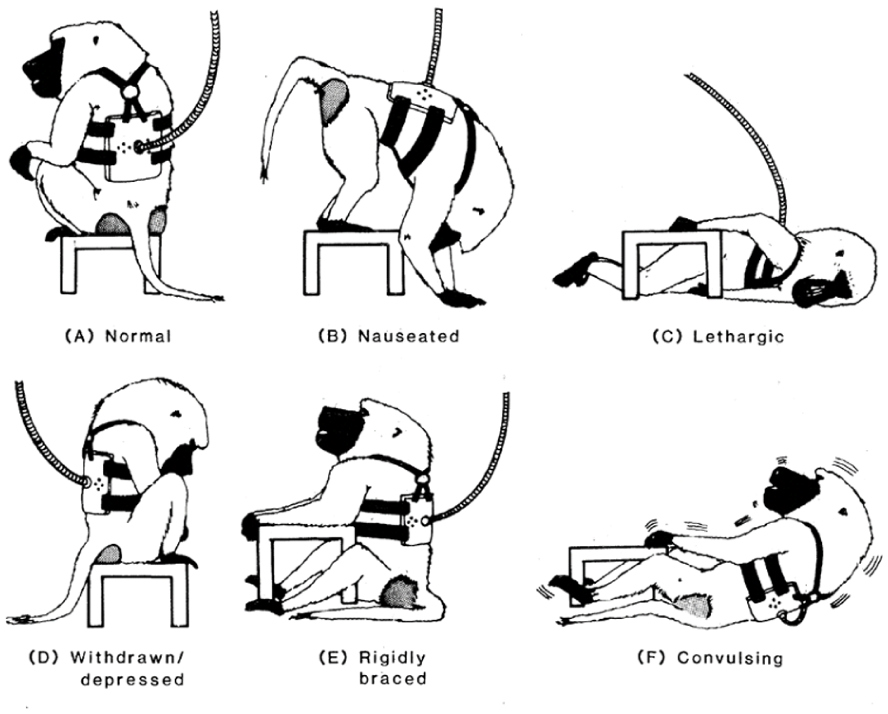
Baboon body postures observed during control conditions and during benzodiazepine withdrawal. The ratings in this figure were conducted on the basis of the observation of body postures and not on the basis of the interpretative labels that appear below each posture sketch. The baboons are wearing harness-tether vests. The rigidly braced posture may be a pre-convulsive state. Figure art was by Lukas with the original in pencil on a graph paper and then hand-traced in ink for publication. (a) Normal, (B) Nauseated, (c) Lethargic, (d) Withdrawn/depressed, (e) Rigidly braced, and (f) Convulsing.
The baboon recovered within a few minutes, possibly because the antagonist had been regurgitated. In the rest of the studies, Ro 15-177 was administered intramuscularly, and the withdrawal signs lasted a few hours.
The experiment was repeated multiple times after both 7 and 35 days of diazepam treatment. The observed physiological and behavioral reactions are shown in Figure 1, including a few episodes of pre-convulsive agitation, tremors, and shaking. Then, diazepam was administered for another 10 days, and the pumps turned off, allowing for observation of potential spontaneous withdrawal. Under that condition, few signs of withdrawal were evident until about day 9 when Griffiths and Lukas observed limb tremors and other depressed body postures. By day 14, the signs of spontaneous withdrawal had subsided. The initial study, including the figure shown below, was indeed accepted and published in Science (Lukas and Griffiths, 1982).
Follow-up studies were conducted replicating the first study across a range of doses that demonstrated withdrawal at lower doses (Lukas and Griffiths, 1984), as well as the importance of prior exposure to benzodiazepines on subsequent drug-taking behavior—a relationship that was earlier demonstrated in humans (Griffiths et al., 1979a, 1980b).
The Lukas and Griffiths findings were generally similar to findings reported in initial and subsequent clinical studies and had implications for understanding that some reports of anxiety and other symptoms upon discontinuation of dosing were not simply reemergence of disease symptoms but were better understood as withdrawal symptoms that might be prevented by gradual discontinuation of dosing (e.g., Busto and Sellers, 1991; Lerner and Klein, 2019; Mellor and Jain, 1982). The FDA eventually modified the labeling of approved benzodiazepines to include a warning of potential withdrawal signs following abrupt discontinuation, along with guidance for dose tapering to minimize withdrawal. Their findings also contributed to drug developers and the FDA requiring drug developers to more carefully evaluate most CNS-active drugs for physical dependence and withdrawal, regardless of whether they appeared to have sufficient abuse potential to warrant scheduling in the CSA (Lerner and Klein, 2019; U.S. FDA, 2017).
As discussed by FDA staff, even in cases in which the approved drug does not show meaningful evidence of abuse and psychological dependence potential that might warrant CSA scheduling or discussion in Section 9 of the approved FDA drug label, it is important for patient safety and to guide patients and their health care providers as to whether discontinuation of the drug for whatever reason (e.g., lost drug on vacation or the medical determination that continuation of therapy may not be needed) that withdrawal may emerge and if possible, discontinuation of dosing should gradual (i.e., “tapered” or “weaned off gradually”; Lerner and Klein, 2019; U.S. FDA, 2017).
Other drugs evaluated by Weerts, Griffiths, and colleagues for physical dependence and withdrawal potential in baboons included GBL (Goodwin et al., 2006), GHB (Weerts et al., 2005) and 1,4-BD (Goodwin et al., 2013), each of which had emerged as club drugs at that time
Increasingly broad application of the baboon self-administration and physical dependence model
After its initial demonstration as a valid model for preclinical abuse potential assessment in baboon self-administration studies (i.e., intragastric and intravenous) by Griffiths and Lukas, abuse liability assessments included increasingly diverse substances. These studies included a creative evaluation of the reinforcing effects of zolpidem in baboons. Further, an intravenous self-administration paradigm involving flumazenil-precipitated withdrawal and spontaneous withdrawal following long-term zolpidem self-administration demonstrated withdrawal signs that were similar to those documented by Lukas and Griffiths (Weerts and Griffiths, 1998).
Human abuse liability research and methods development
Methods of human abuse liability assessment have evolved considerably since the approaches pioneered at the predecessor to the NIDA IRP, the Addiction Research Center, in Lexington Kentucky 5 , and later in Baltimore on the Johns Hopkins Bayview Campus. Early studies relied heavily on various versions of the Addiction Research Center Inventory (ARCI) and participant- and observer-completed forms with similar questions (“Single Dose Questionnaire”) that included a 5-point drug-liking scale (0 described as “not at all” to 4 described as “an awful lot”; see description and forms at Jasinski, 1977; Jasinski et al., 1984; Jasinski and Henningfield, 1989).
The Griffiths-led studies beginning in the late 1970s not only generally employed similar approaches as the NIDA IRP but also generally included psychometric scales, such as the Profile of Mood States, to more fully characterize the potential abuse-related effects (e.g., Henningfield and Griffiths, 1981).
Table 2 lists the year of publication and titles of some of Griffiths’ human abuse liability studies, published by Griffiths and his mentees, several of which involved collaborations with NIDA IRP researchers.
Human abuse liability assessment studies.

From the mid-1970s until his death, Griffiths and his colleagues continued to advance the science of human abuse potential assessment through BPRU studies. These included evaluation of alternative outcome measures to assess the potential for recreational use, potential adverse effects, and in some studies associated with pharmacokinetics. For example, Mumford et al. (1995) compared an experimental drug under development for anxiety disorder (abecarnil) to a prototypic well studied benzodiazepine with standard measures and additional measures that employed behavioral economics approaches (e.g., “willingness to pay on the street,” “worth on the street,” next day assessment of “take [the drug] again”) and an indirect measure of drug reinforcement in which the participants were offered a choice between the drug or money. Behavioral effects included a variety of measures of cognitive and psychomotor performance and, most recently, driving simulators. This broad range of measures, including behavioral economic estimates of the “value” of the drug, its potential detrimental cognitive effects, and abuse-related subjective effects, provided a more nuanced comparison of the neuropsychopharmacology of the drugs under study.
Griffiths and colleagues were also interested in how novel delivery systems and speed of drug delivery affected abuse liability. For example, Reissig et al. (2015) compared the effects of alprazolam administered orally and by an inhalation device with a broad spectrum of measures related to potential for recreational use and liability of abuse. Their study showed that onset of effects was more rapid for inhaled versus oral alprazolam (about 2 vs 49 minutes) with greater potency for the inhaled route; however, the routes were approximately equipotent across routes and by measures of cognitive and psychomotor performance (Figure 2) and measures of sedation. Taken together, the results suggested that overall abuse potential was “modestly increased.” At this writing, the FDA has not yet approved an inhaled alprazolam product. If the product is approved by the FDA, such data will provide a more nuanced evidence base for the FDA to consider in its recommendation for CSA scheduling (which must accompany approval of the new drug application) than would be provided by the typical primary outcome measure of peak liking scores.
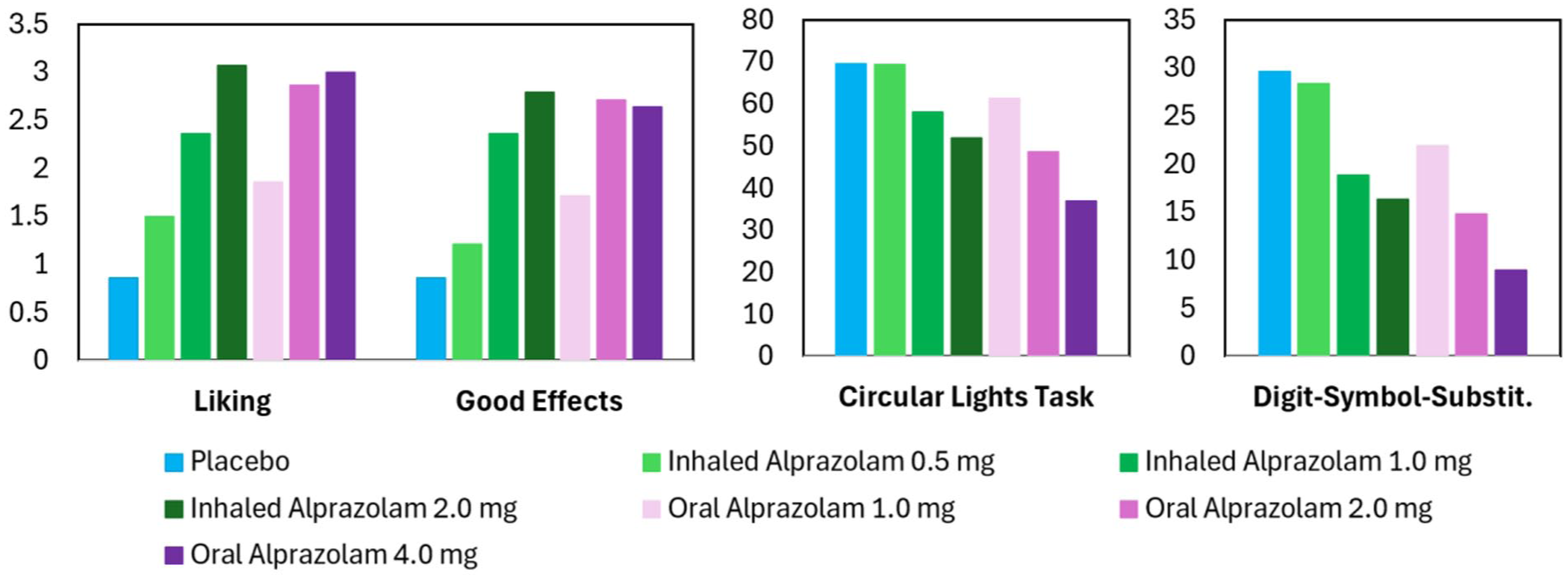
Peak effects on measures directly related to euphoria and abuse potential (subject ratings of Liking and Good), and the liability for abuse related to potential cognitive or psychomotor disruption (means scores on Circular Lights tracking task and mean correct trial scores on Digit Symbol Substitution). (This figure has been simplified and revised from Reissig et al., 2015).
By the early 2000s, the 5-point Likert-style liking scales developed in the 1960s (Jasinski et al., 1984) were increasingly replaced with visual line analog scales, including several questions potentially related to abuse potential in most laboratories and later with encouragement by the FDA (e.g., U.S. FDA, 2017). For example, a comparison of intravenous nicotine to intravenous caffeine studies included the following 100-point visual line analog scales: “Do you feel a drug effect?” “Does the drug have any good effects?” “Does the drug have any bad effects?” and “Do you feel relaxed” (Garrett and Griffiths, 2001). The main findings were an overall higher abuse-related effect of nicotine as compared to caffeine.
These and additional approaches were presented by Griffiths in a 2002 “Abuse Liability Assessment of Drugs” conference that was coordinated with FDA and DEA staff and leading experts to provide the foundation for what would become the FDA’s first formal abuse potential assessment guidance for industry (Griffiths et al., 2003; Schuster and Henningfield, 2003). Although the guidance was not quick to come, the conference was cited in the 2011 draft guidance and the 2017 finalized guidance (U.S. FDA, 2017). The same conference included principles of assessment of abuse liability in animals by Ator and Griffiths (2003), which also contributed to the FDA’s 2017 abuse potential assessment guidance. Drawing from these guidances and reviews and more recent research on psychedelics, the FDA has released a guidance document for psychedelic medicines development (including abuse potential assessment) and has also published a commentary providing additional perspectives for evaluating psychedelics and their abuse potential (Calderon et al., 2023; U.S. FDA, 2023).
The FDA 2017 abuse potential guidance has been important in drug development and stressed the need to provide the premarket data necessary to guide the FDA’s CSA drug scheduling recommendations. As medicines continue to evolve in chemical structure, mechanisms of action, and delivery systems, abuse liability science also must continue to evolve. This includes the applications of newer behavioral economics approaches across a range of novel-acting new medicines, some derived from marijuana and hallucinogenic plants, as well as other plants used as dietary ingredients (e.g., Henningfield et al., 2025; Strickland and Lacy, 2020; Strickland et al., 2023).
In the CPCR, the assessment of psychedelics includes an evaluation of motivations for nonmedical use and their therapeutic potential, and it also incorporates instruments designed to assess effects that include awe, spiritual experiences, and human connection that can be occasioned or evoked by these substances (Barrett and Griffiths, 2018; Barrett et al., 2015; Carbonaro et al., 2018, 2020; Johnson et al., 2018a, 2018b).
For example, in one such study, Carbonaro et al. (2020) compared psilocybin and dextromethorphan using standard drug-liking related human abuse potential assessment measures, along with four additional instruments designed to more fully characterize a broad range of potential altered states of consciousness and effects. 6 The scales were administered sevenhours post drug administration on each test day, and one week and one month following the last test session.
Figure 3 summarizes the outcomes across nine domains at the highest psilocybin dose compared to the prototypic euphoria-inducing dextromethorphan dose, assessed seven hours post drug administration. As shown in Figure 3, there were substantial differences between psilocybin and dextromethorphan-related emotional and other subjective states associated with well-being and considered “meaningful” by many people who have used psychedelics non-medically. Liking-related measures were elevated for both substances.
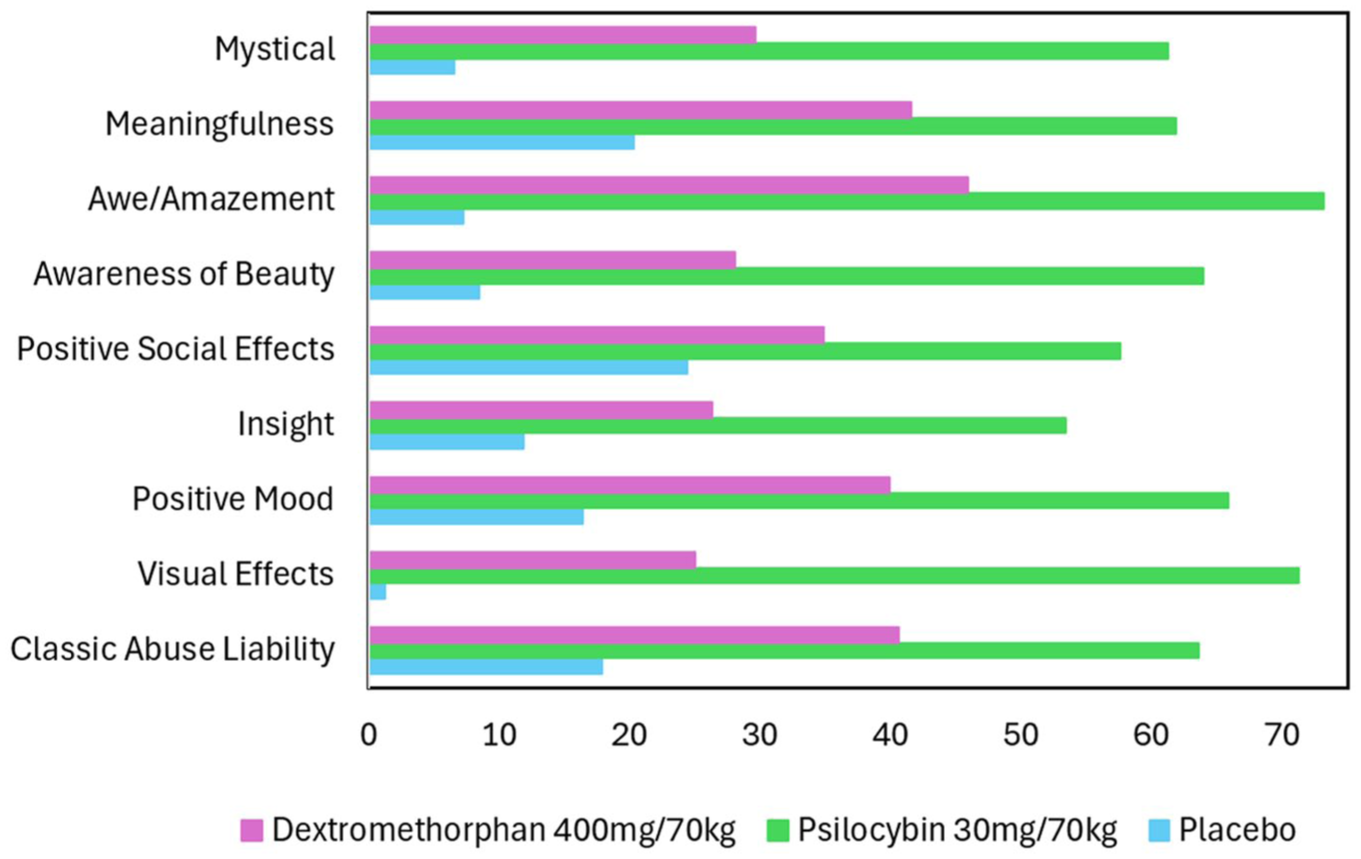
This figure shows mean participant scores expressed as percentages of the maximum possible score on the rating scales for the nine effect domains, each of which included measures based on data presented in Carbonaro et al. (2020), Table 1. For example, the classic drug abuse liability domain includes measures of Liking, Euphoria, and Satisfaction; Awe/Amazement included measures of sense of awe and experience of amazement; positive social effects include feelings of emotional closeness, and understanding others’ feelings.
At one week post drug administration, psilocybin effects were significantly greater than placebo on 26 of 27 measures, whereas dextromethorphan was only significantly elevated on 14 measures. One month after the last session, measures suggesting preference for repeating the drug condition included psychological insight, personal meaning, and spiritual significance, which were significantly greater for 20 and 30 mg/kg psilocybin than for dextromethorphan.
These results suggested that the additional effects captured by any one or more of the states of consciousness-related scales may have provided relevant insight as to the motivations for and likelihood of nonmedical use than liking alone.
Griffiths’ influence in the continuing evolution of abuse liability assessment methods is evident with many of his mentees building on his earlier work to more reliably characterize novel new potential medicines, including psychedelics. An example includes the article by 16 diverse experts (including several former Griffiths collaborators) in this same special issue (Henningfield et al., 2025). Recommendations include more comprehensive approaches to capture the diverse and novel effects of potential new medicines, including the incorporation of behavioral economics methods and the use of psychometric scales to more comprehensively characterize changes in state of consciousness as employed by Griffiths and colleagues (Henningfield et al., 2025).
Drug-specific domains of research: Alcohol, cigarette smoking, and nicotine, caffeine, psychedelics
The remainder of this review examines substance-specific domains of Griffiths’ focus, beginning with alcohol in the 1970s until his last two decades of increasing emphasison psychedelics and consciousness.
The behavioral pharmacology of alcohol and alcoholism
When Bigelow and Griffiths and colleagues were initiating their studies to better understand the behavioral pharmacology of alcohol consumption in heavy drinkers in the early 1970s, alcohol use disorder was widely considered a unique human phenomena, with uncontrollable consumption determined strongly by personality disorders, weak morality, and social factors (American Medical Association, 1968; Garber & MacKillop, 2025; Keller, 1976; Keller and Banks, 1970; National Institute of Mental Health, 1969).
In contrast, several animal behavioral pharmacology trained researchers were exploring the behavioral pharmacology of alcohol and publishing articles that suggested the behavioral pharmacology shared certain characteristics in common with other self-administered drugs including serving as a positive reinforcer when made available for several hours per day by the intravenous route of administration (Deneau et al., 1969; Woods et al., 1971; Winger and Woods, 1973). Yanagita and Takahashi (1973) extended these findings to intragastric self-administration in rhesus monkeys.
Other such studies were ongoing in the same Thompson-led University of Minnesota laboratory in which Bigelow and Griffiths were doing their predoctoral animal research. In the late 1960s, Meisch and Thompson had begun exploring the behavioral pharmacology of oral alcohol self-administration in rats, and then rhesus monkeys beginning in 1971. 7 They found that rats induced to consume ethanol by various techniques would result in alcohol serving as a reinforcer in its own right and that, once established as a reinforcer, rats would intermittently drink to intoxication with orderly changes in their behavior, such as reduced number of drinks and total volume consumed as alcohol concentration and work requirement (lever presses per drink) increased (Meisch, 1969; Meisch and Thompson, 1971, 1973). Similar findings were later obtained in rhesus monkeys (Henningfield and Meisch, 1976; Meisch et al., 1975).
This research contributed to the understanding of alcohol use disorder as a multifaceted neurobiological process and not dependent on human moral failure and personality disorders. This work also contributed to advances in the management of drinking and treatment, later explored by Bigelow and Griffiths, that incorporated behavioral interventions, such as contingency management to sustain participants in treatment and reduced, if not eliminated, drinking. Table 3 lists the titles and year of publication of some of Griffiths’ human alcohol studies.
Alcohol studies.

Thus, Bigelow and Griffiths were familiar with the foregoing research from the published articles and in their own discussions with the researchers in the Minnesota Psychopharmacology program, and at scientific meetings. Their work with psychiatric patients, which was part of their Psychopharmacology Training program experience (e.g., Bigelow and Griffiths, 1974), had also taught them that people with psychiatric disorders could experience beneficial changes in behavior and skill acquisition by behavior modification techniques that had parallels with animal behavioral pharmacology studies. For example, increased work requirement per drug dose decreased intake in an orderly fashion, and once drugs, including alcohol, were established as reinforcers, self-administration was not random but rather was related to environmental conditions and contingencies.
To enable exploration of alcohol self-administration with more ecological validity, they modified one section of the clinical research unit to generally resemble a tavern with the addition of a counter-like bar and barstools. They recruited people with extensive heavy drinking histories, who were diagnosed as chronic alcoholics, to serve as participants in studies on the residential research unit. A stationary bicycle provided a means of manipulating the work requirement per drink; in some studies, one token was provided per two minutes of exercise, and the tokens could then be used to “pay” for alcoholic beverages. They were also able to measure caffeinated beverage consumption patterns and cigarette smoking, as both cigarettes and instant coffee packets needed to be individually obtained from the research staff.
The studies commenced in the early 1970s and over the next few years their findings began to have an impact in changing thinking about alcohol used disorder from that of a personality deficit disorder in which drinking was not controllable by people with alcohol use disorder to an orderly behavioral pharmacological process involving a substance that could serve as a robust biological reinforcer in a variety of species including humans, and with orderly, environmentally influenceable patterns of self-administration. Along the way, they also observed seemingly orderly consumption patterns of coffee and cigarettes, though drinking coffee and smoking cigarettes were not the primary focus of their studies.
One of the first published studies evaluated the effects of 10–15 minutes of social isolation (“time-out”) under conditions in which the volunteers “were given daily access to substantial quantities of alcohol” dispensed as single “drinks” of 1 ounce of 95 proof alcohol mixed with orange juice upon request of the participant (Bigelow et al., 1974: 1). Immediately following the dispensing and consumption of alcohol, the volunteer was required to sit for 15 minutes in a booth approximately 1 m2 as the social isolation or time-out contingency. This resulted in substantially decreased alcohol intake in 9 of 10 participants to an average of approximately 50% of the baseline levels without such a social time-out contingency. When the time-out procedure was discontinued, alcohol consumption returned to its baseline higher levels. The robustness of the effect in 90% of the participants was at odds with theories about the lack of control over drinking.
This study was extended in three follow-up studies (Griffiths et al., 1974a, 1974b; Griffiths et al.,1975a). The latter study compared different methods for experimental conduct and different drink-contingent time-out protocols. For example, in the residential ward condition, 14 participants had daily access to up to 17 alcoholic drinks with a minimum interval of 40 minutes between drinks. Two different time-out conditions were compared: social and activity. Fifteen minutes of social time-out after a drink was requested and consumed resulted in a reduction of drinks per day to 71% of what occurred during the baseline drinking condition with no time-out contingency. Time-out from activity time led to drinking levels of 36% of baseline, and the combination of time-out from activity plus social time-out led to drinking levels of only 24% of baseline levels. The findings were similar whether the participants were continuously exposed to each experimental condition across several consecutive days or if the conditions occurred in mixed order across days.
In addition to contributing to the understanding of alcohol use disorder as a multifaceted neurobiological process that has little to do with “low morality” and which could and does develop in many people without personality disorders, these studies also piqued Griffiths’ curiosity about the apparent association between alcoholic beverage consumption and cigarette smoking. He later explained that he wondered if the relationship was “like cookies and milk, which just go well together, or if there was a pharmacological component” (personal communication to JEH). He recognized these were not mutually exclusive potential contributors. This led Griffiths, Bigelow, and Liebson to design an elaborate series of test conditions within a single study to better understand the relationship between alcohol consumption and cigarette smoking (Griffiths et al., 1976b).
In the resulting article titled “Facilitation of human tobacco self-administration by ethanol: a behavioral analysis,” he assessed the generalizability of the association and interactions (Griffiths et al., 1976b). The study included seven distinct experiments, each controlling for and manipulating different variables in an effort to assess the generalizability of the association and rule out various hypotheses in daily 6-hour sessions in which 30 ml of alcohol was mixed with 60 ml of orange juice and compared to orange juice alone. During the sessions, cigarettes were available to each participant by pulling on a standard Lindsey operandum (as was used in his baboon studies) with either 5 or 10 pulls on the device necessary to dispense the cigarette. This was partly in an effort to ensure that cigarette consumption would not involve the social interaction of staff dispensing.
In the first experiment, each participant was required to consume 12 drinks, one every 30 minutes during the daily 6-hour sessions in which the drinks were alcoholic in a programed mixed sequence across days such that over 20 days, there were 10 days each of alcoholic drinks and vehicle drinks. In another experiment, the subjects were provided with either 10 consecutive days of alcoholic drinks or10 consecutive vehicle drink days. Cigarette consumption was measured by the number of cigarettes dispensed and butts returned in all experiments, but to provide additional measures of actual tobacco consumption, one experiment employed a butt collection protocol that included weighing the cigarette butt to estimate the amount of tobacco consumed. Another approach to quantitating the amount of smoking involved videotaping cigarette smoking to enable later review and counting of puffs per cigarette.
The main finding of this 7-part study was “that ethanol consumption is a potent determinant of cigarette smoking by alcoholics in a residential laboratory setting” (Griffiths et al., 1976b: 290). The effect was robust across participants, occurringin both social access and social isolation conditions. It was remarkably consistent across days whether alcohol days were in a mixed or consecutive schedule and whether participants could consume each drink ad libitum or every 30 minutes as a quick 30-second drink. The alcohol facilitation of tobacco consumption was similar when measured by the number of cigarettes smoked, puffs taken, or tobacco burned (assessed by the residual weight of the cigarette butts). Within days, there was no evidence of the effect being stronger during the beginning of the alcohol consumption session or later in the session; rather, the increase was uniform across the 6-hour sessions.
Griffiths received an award for this study from the American Psychiatric Association based on its scientific and potentially clinically relevant advancement in the understanding of alcoholism and cigarette smoking. The alcohol studies also provided a daily reminder to Griffiths and Bigelow that heavy coffee consumption and cigarette smoking were common among the heavy alcohol consumers in these studies and that smoking seemed especially associated with alcohol consumption. This coincided with increasing interest in research by NIDA on cigarette smoking, suggesting the possibility of funding for cigarette smoking abuse-related research and the development of applications for funding to extend the alcohol research program model to cigarette smoking. This Griffiths et al. (1976b) study contributed to the thinking that went into a subsequent application for funding to NIDA to study “licit and illicit drugs of abuse” that had a strong focus on better understanding the behavioral pharmacology of cigarette smoking.
The behavioral pharmacology of cigarette smoking and nicotine
In the 1970s, neither cigarette smoking nor nicotine were considered to be addictive by U.S. Federal agencies or most health care providers based on the widely accepted conclusion of the 1964 Report of the Advisory Committee to the Surgeon General that cigarette smoking was most appropriately considered a habitual behavior did not meet criteria for drug addiction (U.S. Department of Health, Education, and Welfare, 1964). However, early 1970s studies and reviews by Murray Jarvik, Michael Russell, and others suggested that cigarette smoking might be appropriately considered a form of drug dependence involving nicotine (Brecher and The Editors of Consumer Reports, 1973; Jarvik et al., 1970; Lucchesi et al., 1967; Pinney, 1979; Russell, 1971; Stolerman et al., 1973) but there had been little federal support for such research and few studies addressing nicotine’s addiction potential by the emerging standards used by the DEA, FDA, and NIDA.
NIDA was established in 1974 with a mandate to primarily focus on illicit drugs of abuse but its first director, Dr. Robert Dupont, suggested that cigarette smoking was a major devastating addiction in adults and youth (Hicks, 1975). DuPont’s statements caught the attention and concern of the cigarette industry about this potential area of emerging NIDA research and it began using its influence to minimize research related to the potential addiction potential of cigarettes and nicotine (Henningfield, 2011; Kessler, 2001; Tobacco Institute, 1980). Undeterred, the next NIDA Director, Dr. William Pollin, appointed in 1975, encouraged NIDA exploration of cigarette smoking and nicotine through conferences and monographs, which cited Griffiths’ cigarette smoking data collected during his alcohol studies with Bigelow and Liebson (Griffiths et al., 1977a; Krasnegor, 1978, 1979a, 1979b). In the preface to one of those monographs, Director Pollin stated that an increasing priority for NIDA was research addressing the possibility that cigarette smoking might be considered “a prototypic drug of addiction” and implied that NIDA was interested in supporting such research (Pollin, 1977).
To this end, Pollin encouraged interactions of NIDA staff with the CDC’s Office on Smoking and Health (OSH), which was established by Secretary of Health Joseph Califano in 1978, and the Secretary in turn directed OSH Director John Pinney to meet with Pollin to encourage NIDA to determine if smoking and nicotine met criteria for addiction. This led to increased extramural grant support and engagement of NIDA’s IRP, the Addiction Research Center, to recruit several scientists to support those efforts.
Griffiths and Bigelow and colleagues responded with a proposal to NIDA entitled “Licit and Illicit Drugs of Abuse” to study the behavioral pharmacology of cigarette smoking including interactions of smoking with a variety of drugs of abuse. In the spring of 1978, they were notified that the grant was funded. Separately, they had applied for but had not yet received funding for a post-doctoral training program, but they were able to hire a junior faculty position to focus on the research promised by the grant. They offered it to Henningfield who was completing a post-doctoral fellowship at the University of Minnesota. They promised further postdoctoral training and the opportunity to continue his primate alcohol research in the baboon laboratory along with cigarette smoking and other research in the BPRU.
For a junior faculty/postdoctoral trainee, it was a very exciting time. Like his earlier mentors Meisch and Thompson, Griffiths provided scientific readings, and frequent meetings to discuss them. He gave Henningfield the opportunity to collaborate on a highly demanding and engaging review article addressing similarities in animal and human drug-taking behavior, which was as much an exercise in the pros and cons of various scientific methods as it was about the findings themselves (Griffiths et al., 1980a). For Henningfield, it was one of his most meaningful collaborative writing and learning experiences with a long career-influencing impact that was useful in later contributions to U.S. Surgeons General and World Health Organization reports.
Specific to tobacco, Griffiths provided piles of articles to read and search through for the CDC’s OSH, including National Clearing House reports summarizing tobacco and nicotine publications worldwide, which were published several times per year.
They had regular weekly meetings but for the first year met at least briefly on a nearly daily basis developing the laboratory, experimental designs, and proposals with much of the research testing conducted by Henningfield himself with the aid of a research assistant.
Readings included those by psychologists and behavioral pharmacologists Charles Schuster, Ian Stolerman, Ellen Gritz, and Lynn Kozlowski; research psychiatrists, Murray Jarvik and Michael Russell; and pharmacologists including Edward Domino and John Rosecranz. Together, their work suggested that nicotine may be playing a modulating role in cigarette smoking, regardless of whether it met criteria as an addictive or abusable drug in its own right, but with many key questions needing study. 8 For example, the prominent behavioral pharmacologist, Charles R. Schuster, led a study demonstrating that intravenous nicotine infusion reduced spontaneous cigarette smoking in daily cigarette smokers, and two primate intravenous drug self-administration studies suggested that nicotine might serve as a positive reinforcer (Deneau and Inoki, 1967; Lucchesi et al., 1967; Yanagita and Takahashi, 1973).
Nonetheless, it was clear that this was new scientific territory in behavioral pharmacology. As Griffiths told Henningfield, “this is going to be fun because we really don’t know much about the role of nicotine in smoking or if smoking is an orderly behavioral pharmacological process.” The first thing that was needed was to build cigarette smoking test rooms equipped with monitoring systems to enable automated assessment of cigarette lighting, puffing, and extinguishing behaviors (Henningfield and Griffiths, 1979; Henningfield et al., 1980). Expired air carbon monoxide (CO) appeared to have potential as a biomarker of smoke intake, but research was needed to determine the association of CO with cigarettes smoked over time during test sessions (Henningfield et al., 1980).
These tools provided the framework to begin to manipulate basic variables such as cigarette deprivation time before smoking was allowed, various approaches to manipulated cigarette dose such as the use of smoke diluting ventilated cigarette holders, limiting the number of puffs per cigarette, manipulation of the inter cigarette smoking bout time interval, and varying the nicotine content of the cigarettes with research cigarettes provided by the National Cancer Institute (Chait and Griffiths, 1982; Griffiths et al., 1982; Henningfield and Griffiths, 1980; Nemeth-Coslett and Griffiths, 1984; Woodson and Griffiths, 1992; Zacny et al., 1987).
Within 2–3 years, it was becoming clear that cigarette smoking could be described as an orderly behavioral pharmacological process in which patterns of cigarette smoking were remarkably orderly. This included measuring puffs within a cigarette (each cigarette beginning with a burst of puffs, then more evenly spaced puffing over time) and measuring cigarette consumption over time (with increased smoke deprivation reducing the time to first cigarette and increasing puffs). A variety of approaches were used to manipulate the cigarette smoke dose, and these generally resulted in orderly changes in smoking (referred to as “compensation” and “titration”) such that the actual number of puffs and/or smoke intake were decreased by increases in smoke concentration and opportunity to smoke (Griffiths and Henningfield, 1982a, 1982b).
The Hopkins tobacco research program led by Griffiths was further expanded by an informal meeting between then-acting Director of the NIDA IRP, Donald Jasinski, who was leading the relocation of the Addiction Research Center from Lexington, Kentucky, to the very same building on the Johns Hopkins Bayview campus that housed the BPRU. Jasinski mentioned to Griffiths that he had been directed by NIDA Director Pollin to hire a clinical researcher to lead studies to determine if smoking and nicotine met the criteria for addiction potential. Griffiths suggested that Henningfield might be appropriate and willing, and, if so, it might foster mutually beneficial collaborations to accelerate progress, if the IRP would also be able to collaborate with BPRU scientists. This happened and with Griffiths and Jasinski’s support, research collaborations on tobacco and nicotine, along with opioids and other substances, accelerated with the sharing of resources and thinking as exemplified in their first joint NIDA monograph paper titled “Human dependence on tobacco and opioids: common factors” that included Griffiths and Jasinski (Henningfield et al., 1981b), a paper that would later contribute to the outline of the approach to what became the first Surgeon General’s report to focus on addiction to cigarette smoking and nicotine (U.S. DHHS, 1988).
Under Griffiths, direction and with collaborations with Stitzer, Bigelow, and other BPRU scientists, the tobacco research advanced rapidly, with a level of productivity and relevance to public health that resulted in several renewals of funding of the grant over the next decade. Research included studies of the effects of pretreatment with various drugs, which demonstrated that, with the exception of nicotine, most of the drugs with robust, pleasurable, and abuse-related effects (including reinforcement) led to increased cigarette smoking. These included alcohol, amphetamine, methadone, pentobarbital (Chait and Griffiths, 1984; Griffiths et al., 1976b; Henningfield et al., 1983a, 1984; Henningfield and Griffiths, 1981). Marijuana and caffeine produced weak or unreliable effects on smoking (Chait and Griffiths, 1983; Nemeth-Coslett et al., 1986a).
Neither the opioid antagonist naloxone nor nicotine antagonist mecamylamine reliably affected cigarette smoking (Nemeth-Coslett and Griffiths, 1986; Nemeth-Coslett et al., 1986b). The Griffiths et al. studies, along with others, also found that the abuse potential of nicotine itself varied widely depending on the delivery system and thus, the abuse potential of nicotine in the form of nicotine gum and other slower-delivering products was very low as compared to cigarettes (Nemeth-Coslett et al., 1987; Sobel et al., 2004; U.S. DHHS, 1988, 2010).
Key studies demonstrating the abuse potential of nicotine were encouraged by Griffiths and benefited from his input and methodological training. This included studies evaluating the human abuse potential of acute doses of intravenous nicotine compared to cigarettes across a range of doses of each, and studies comparing intravenous nicotine self-administration in parallel with similar protocols in squirrel monkeys (Goldberg and Henningfield, 1988; Henningfield and Goldberg, 1983a, 1983b; Henningfield et al., 1983b, 1985). Some of the important questions raised by these studies were then addressed by Griffiths and other colleagues in the classic Griffiths’ approach to independently, systematically extending the studies under different conditions, including a cocaine comparison (Duke et al., 2015; Garrett and Griffiths, 2001; Jones et al., 1999). Table 4 lists the titles and years of publication of some of Griffiths’ key studies on nicotine and cigarette smoking.
Nicotine and cigarette smoking studies.
The Griffiths et al. program of research in the Johns Hopkins BPRU was a major contributor, along with many other laboratories and investigators that came together to come to the conclusion that cigarette smoke and nicotine itself met all widely accepted criteria for substantial abuse potential. This supported the recommendation that cigarettes warranted labeling and warnings as an addictive substance, though the abuse potential of nicotine was highest in cigarettes and lower in all other tested nicotine delivery systems (Henningfield, 2011; U.S. DHHS, 1988, 2010).
Research by Griffiths and colleagues in the Johns Hopkins University tobacco research program established by Griffiths and colleagues contributed substantially to the landmark 1988 Report of the Surgeon General, which came to the following major conclusions:
Cigarettes and other forms of tobacco are addictive.
Nicotine is the drug in tobacco that causes addiction.
The pharmacological and behavioral processes that determine tobacco addiction are similar to those that determine addiction to drugs such as heroin and cocaine.
The 1988 Surgeon General’s report and those conclusions were supported by more than 60 studies by Griffiths and his colleagues, with his program being among the major contributors from laboratories addressing tobacco and nicotine across the nation and globally (Henningfield, 2011; U.S. DHHS, 1988, 2010). The 1988 Surgeon General’s report and further Hopkins studies were also widely cited and contributed to the FDA Final Rule for the FDA to regulate cigarettes and smokeless tobacco in 1996 (Henningfield, 2011; Kessler, 2001; U.S. FDA, 1996). 9
The Hopkins studies led by Griffiths were also cited and were important in the determination that the cigarette test method by the Federal Trade Commission (FTC) was misleading and often substantially underestimated tar and nicotine exposure due to cigarette smoker titration (National Cancer Institute, 2001) and the ultimate rescission of the FTC method by the FTC in 2008 following a hearing and recommendation by the U.S. Senate in by an active congress in 2007 (See FTC 2008 citation at https://www.federalregister.gov/documents/2008/12/08/E8-28969/rescission-of-ftc-guidance-concerning-the-cambridge-filter-method and 2007 U.S. Congress hearing at https://www.govinfo.gov/content/pkg/CHRG-110shrg73848/pdf/CHRG-110shrg73848.pdf).
Understanding the abuse liability of caffeine and implications for medicine and regulation
As he described it, Griffiths’ interest in caffeine was piqued by his own caffeine use that he found useful, as well as the reliable association of heavy caffeine consumption by people with alcohol use disorder that he noted in his own studies. His curriculum vitae lists more than 60 caffeine-related studies. The titles of some of these papers and chapters are presented in Table 5. They provide a sense of the extensive scope of his caffeine studies, some of which are summarized in this section. Griffiths’ work on caffeine has been highly influential in the recognition of caffeine as a potentially dependence-producing drug, albeit a relatively benign drug, and for many, if not most, consumers, a pleasurable substance contributing to occupational and social demands and well-being. His thinking on caffeine influenced and was influenced by addiction neuropharmacology pioneer Avram Goldstein, who cited Griffiths’ work in his classic Addiction: from Biology to Drug Policy (Goldstein, 1994).
Caffeine studies.
His first caffeine study was a continuation of the series of studies supported by his Licit and Illicit Drugs of Abuse grant and evaluated the effects of caffeine on cigarette smoking. Caffeine in doses from 50 to 800 mg, 25 mg of d-amphetamine, or placebo-filled capsules were administered under double-blind conditions 30 minutes before 90-minute sessions (Chait and Griffiths, 1983). Participants were in an isolated test room and were free to read and relax and smoke ad libitum using the earlier described cigarette holder that enabled measurement of the time of every puff on every cigarette. The main findings from this study were that caffeine was not associated with increased smoking, whereas d-amphetamine “which had stronger euphoriant effects” robustly increased smoking, as had been demonstrated in an earlier study that did not include caffeine (Henningfield and Griffiths, 1981).
Two subsequent studies on the residential research unit assessed the pattern of coffee drinking in heavy coffee drinkers. The studies examined acute caffeine effects in which the dose of caffeine added to decaffeinated coffee was manipulated over a wide range of doses with monitoring for potential withdrawal when the participants were only given decaffeinated coffee for at least 10 consecutive days (Griffiths et al., 1986a, 1986b).
These studies demonstrated higher liking and preference for caffeinated coffee. They showed an inverted U-shaped function of cups of coffee ingested per day as a function of caffeine dose per cup. That is, people who received less caffeine in their coffee drank more cups per day. Conversely, when participants were pretreated with caffeine in capsules before the start of the test session day they consumed fewer cups of coffee. These findings suggested that caffeine intake was being titrated, consistent with its apparent reinforcing effects.
In the 10-day condition in which no caffeine was added to the decaffeinated coffee, withdrawal symptoms had an onset on average 19 hours after the last caffeine consumption, peaked within about 24 hours, and began to subside within two days. Symptoms included headaches and sleepiness, and decreased alertness and activeness. Griffiths et al. concluded that these studies demonstrated the reinforcing effects of caffeine in humans and further characterized the caffeine withdrawal syndrome. They concluded that “caffeine has the cardinal features of a prototypic drug of abuse” (Griffiths et al., 1986a).
While this was not the first demonstration of caffeine withdrawal, as its characteristic headache had been delineated in earlier studies (e.g., Dreisbach and Pfeiffer, 1943), the work by Griffiths as well as Hughes and colleagues (e.g., Hughes et al., 1991) was important in defining the syndrome, its severity, and the time course, and showed symptoms went beyond headache.
Other studies by Griffiths showed that a withdrawal syndrome could appear after repeated dosing, even following relatively low daily intake (Griffiths et al., 1990b), and that caffeine dosing conditions could affect withdrawal (Evans and Griffiths, 1999). Two additional studies suggested that physical dependence and withdrawal could increase the reinforcing effects of caffeine (Garrett and Griffiths, 1998; Schuh and Griffiths, 1997).
Two major outpatient clinical trials with caffeine were particularly impactful. The first was a study that demonstrated that caffeine withdrawal could occur after double-blind cessation of a participant’s daily caffeine use (Silverman et al., 1992). The 62 participants in this study were told they were in a study looking at foods, to maintain the blind, and at baseline consumed low to moderate daily doses of dietary caffeine (the mean dose of caffeine was equivalent to about 2.5 cups of coffee per day). They underwent 2-day periods of caffeine abstinence (along with a variety of other benign dietary manipulations to maintain the blind), and during withdrawal had symptoms that included increases in ratings related to anxiety, depression, headache, and fatigue, and decreases in feelings of vigor. The findings were particularly impactful for two reasons: first, the withdrawal syndrome encompassed a broad range of symptoms, and second, withdrawal occurred for people consuming typical amounts of daily caffeine.
The second study was a case series evaluation of 16 people who self-reported problematic caffeine use and assessed them using the DSM-IV criteria for substance (caffeine) dependence (Strain et al., 1994). In addition, 11 of the participants underwent a double-blind caffeine withdrawal. Virtually all participants reported typical DSM dependence symptoms, and nine of the 11 had objective signs of withdrawal (with eight having functional impairment—in some cases, dramatically so). This study showed that caffeine dependence, or what would now be called caffeine use disorder, is clearly a clinical entity and that use of caffeine can be problematic for some people.
Another series of studies addressed questions relevant to potential regulatory actions, such as requiring that products with added caffeine disclose that fact along with the amount of caffeine per container. One question related to this regulatory action was the threshold dose at which behavioral effects of caffeine occurred. It was recognized that many dietary products—including the majority of carbonated soft drink brands, chocolate candies, mocha-flavored ice cream, yogurt, and other commonly consumed products contain caffeine. But the ingredient labels of these products rarely mentioned added caffeine, let alone the amount, and there appeared to be little public awareness of the pervasiveness of caffeine in manufactured food and beverage products.
For additional background, some experts (and the coffee industry) knew that products marketed as “decaffeinated coffee” did not mean coffee was completely caffeine free, but at that time, there were no FDA standards for what could be labeled as decaffeinated. Some manufacturers claimed that their standard was a reduction by at least 97%. A 2006 study of caffeine levels in some popular coffee serving shops found that a single shot of purported decaffeinated espresso might contain from 3 to 16 mg per shot and a 16-ounce serving of purported decaffeinated coffee might contain up to 12–14 mg (McCusker et al., 2006). Although, FDA’s 2024 website states that “decaf coffee typically has 2–15 mg in an 8-ounce fluid cup” (U.S. FDA, 2024), that would be a small serving in most coffee shops.
Beginning in the late 1980s, Griffiths was increasingly interested in the potential of low doses of caffeine and theobromine as might occur in commonly consumed products to alter mood and behavior. Several studies and techniques were used to determine the threshold doses for potential interoceptive and behavioral effects. One study titled “Discriminative Stimulus and Subjective Effects of Theobromine and Caffeine in Humans” employed seven volunteers with histories of methylxanthine consumption in various forms, including coffee, tea, and cocoa products (Mumford et al., 1994). They included colleagues and employees of the Johns Hopkins School of Medicine and NIDA IRP who did not receive monetary compensation for their participation but consented to participate and comply with the rigorous demands of methylxanthine abstinence, for the duration of the study, except as provided during the study. Two authors of this review were offered the opportunity to participate, and either declined participation or agreed to participate but withdrew before completion as they were unwilling or unable to fully comply with the demanding conditions. In brief, Mumford et al. found that caffeine had generally more reliable effects than theobromine, but there was wide individual variability across participants for both drugs. Reliable discrimination was acquired in all seven participants for oral encapsulated caffeine, with thresholds ranging from 1.8–178 mg, and for 5 of the 7 participants for oral encapsulated theobromine with thresholds ranging from 100 to 560 mg. Caffeine increased ratings of well-being, energy, social disposition, and alertness across the group and in the individuals, whereas theobromine did not reliably alter group ratings but did change ratings in some subjects. The authors discussed various limitations of the study, but the authors and journal reviewers agreed that the study demonstrated that for some people, commonly consumed cocoa products contain behaviorally active doses of caffeine and possibly theobromine, and that for some people, the levels of caffeine present in decaffeinated coffee may be behaviorally active.
Griffiths gave several of his caffeine research collaborators, including at least two coauthors of this review, the plaque shown in Figure 4.

Photograph of a plaque given by Griffiths to several of his BPRU colleagues who participated in the caffeine research.
Another issue that was increasingly recognized as of public health significance in the first decade of the 21st century was the growing popularity of mass-marketed high caffeine dose-containing energy drinks. As discussed by Griffiths and his colleagues, this increased the public health and regulatory importance of further studies of the dose-related effects of caffeine with respect to health, safety, and dependence potential and the need for more active regulatory oversight (Reissig et al., 2009).
Griffiths and his mentees conducted a series of studies using a variety of techniques to determine the doses at which subjective and behavioral effects of caffeine could be discriminated. They included scales sensitive to a variety of potential effects such as stimulation, pleasure, jitteriness, and nervousness. The initial studies established several key findings: (1) there is wide individual variation in the detection of caffeine effects, (2) daily caffeine users develop tolerance to caffeine and are less likely to discriminate the presence of low doses of caffeine, and (3) doses as low as a few milligrams could be discriminated by some people and reliability of this discrimination was directly related to increasing doses of caffeine (Evans and Griffiths 1991, 1992; Griffiths et al., 1990a; Mumford et al., 1994; Silverman and Griffiths, 1992).
Subsequent human laboratory studies of caffeine further characterized it as a reinforcer (Evans et al., 1994a; Griffiths and Woodson, 1988; Griffiths et al., 1989), and tolerance developed to caffeine effects (Evans and Griffiths, 1992).
Other studies compared acute caffeine effects to other stimulants. For example, one study examined differences in abuse potential-related subjective effects, such as euphoria for oral caffeine compared to oral d-amphetamine (Chait and Griffiths, 1983), and another study used intravenous caffeine administration and showed lower euphoria by caffeine as compared to nicotine (Garrett and Griffiths, 2001). However, in recreational stimulant users deprived of caffeine, intravenous caffeine produced some qualitatively similar euphoriant effects as cocaine and other stimulants, including participant identifications of the drug as cocaine (Rush et al., 1995). Other studies by Griffiths and colleagues further characterized caffeine’s abuse potential related stimulant effects (Johnson et al., 2010; Jones and Griffiths, 2003; Sigmon et al., 2009). Taken together, these studies by Griffiths and colleagues do not suggest caffeine has an abuse and addiction potential comparable to cocaine and amphetamines, but they do support the conclusion that caffeine is appropriately characterized as a stimulant with addiction potential.
This line of research with caffeine, spanning over two decades, systematically investigated the behavioral pharmacology and dependence potential (both physical and psychological) of this commonly used substance andhas had clear clinical implications. But this research can also be viewed as helping to provide a public understanding of drug dependence (Pickworth, 1995), by illustrating the features of a use disorder with a substance that is commonly used by a large proportion of the population.
At this writing, the American Psychiatric Association has not yet listed caffeine use disorder as a diagnostic category in its Diagnostic and Statistical Manual of Mental Disorders (American Psychiatric Association, 2022), though it recognizes caffeine withdrawal and intoxication disorders. It has, however, placed caffeine use disorder in its “conditions for further study.” Such research is continuing at Johns Hopkins and elsewhere as the legacy of the carefully conducted, innovative and impactful studies of caffeine by Griffiths continues.
Psychedelic research and the neuropsychopharmacology of consciousness
Griffiths’ interest in psychedelic research was, in part, related to his adoption of meditation practices in the early 1990s in his own efforts to improve his well-being and possibly a more rewarding balance of his research-focused life with other aspects of his life. This coincided with increasing animal and human psychedelic research in the 1990s (e.g., Marona-Lewicka and Nichols, 1995; Strassman, 1996), and his learning more about the history of meditation, which suggested some similarities in potential life-changing effects with those reported for a few or even single use of psychedelic substances
As he recounted to some of the authors of this review, he was personally skeptical of such claimed similarities, in part because his research on psychoactive substances was consistent with widespread demonstrations that their main effects were transient changes in emotional and subjective states (pharmacodynamic effects) that were generally related to their time course of onset and offset (pharmacokinetics). This was in contrast to his own experience in meditation and the history of meditation, which suggested that the potential benefits tended to emerge over months and years of practice, and then could be highly durable, that is, “life-changing.”
The notion that exposure to one dose of a substance could produce qualitatively similar and potentially persistent changes in mood and feeling seemed questionable to him. Some of this has been discussed in his publications (e.g., Griffiths et al., 2006), and in far greater depth in several interviews. 10
By about 2000, Griffiths was ready to attempt to initiate clinical psychedelic research at Johns Hopkins. He knew it might be a fruitless effort as the regulatory barriers were enormous (Belouin and Henningfield, 2018; Belouin et al., 2022), and there was no NIH funding for clinical research. It was not clear that major universities such as Johns Hopkins would permit such controversial research. Moreover, the accompanying stigma associated with these substances virtually eliminated efforts by pharmaceutical companies or academic research institutions from developing psychedelic research programs for the rest of the twentieth century (Belouin and Henningfield, 2018).
As discussed elsewhere, Griffiths and colleagues and Johns Hopkins University were not alone in efforts to rekindle clinical psychedelic research and potential medicinal applications, but their involvement and leadership unquestionably helped to re-legitimize and catalyze further research and investment by philanthropies but it would not be until 2021 that federal funding of clinical research by NIH and the Veterans Administration emerged (Xi et al., 2023).
Nonetheless, psychedelic research emerged as the primary focus of the last two decades of Griffiths’ life and was on a rapidly accelerating trajectory until his death in 2023, as shown by annual publication rates in Figure 5. This figure also illustrates an important aspect of Griffiths’ approach to mentoring and collaborating, which is to promote independence in thinking and research by others. This is shown by the increase in publications that did not include his authorship over time. This approach was reflected in part by his nature and the similar approach by his own mentors. It also seems likely to have been a key factor in the innovation and proliferation of research at Johns Hopkins as well as the many other institutions that collaborated with Griffiths and Johns Hopkins, as Griffiths was generous with his time and efforts to collaborate with and support the efforts of other scientists and institutions in their own development of psychedelic research programs.
Publications by Roland Griffiths and the Johns Hopkins Center for Psychedelic and Consciousness Research. The blue portion of each bar shows publications co-authored by Griffiths and the red portion shows the total listed by the Johns Hopkins Center for Psychedelic and Consciousness Research website from 2006 through 2024 (accessed May 4, 2025, at https://hopkinspsychedelic.org/publications). The percentage of the publications that were co-authored by Griffiths in each year is shown above the bars.
From the first psychedelic study and going forward, an important consideration was interest by Griffiths and his colleagues in laying the foundation for a program of research that might support the development of potential new medicines based on psychedelic substances for various psychiatric disorders. Research in the 1950s and 1960s suggested promising applications for disorders including depression, anxiety, alcohol, and other addictions before clinical psychedelic research was sharply reduced by the 1970 U.S. CSA and international 1971 Convention on Psychotropic Substances (Belouin and Henningfield, 2018; Henningfield et al., 2023). The intent of the CSA was not to prevent psychedelic research but to ensure that clinical research with Schedule I psychedelic substances would be safe and in the interests of public health; however, the complex and multilayered process requiring input and oversight by both DEA and FDA in addition to institutional review boards has substantially deterred Schedule I drug research (Belouin and Henningfield, 2018; Henningfield et al., 2023).
The 2006 Griffiths et al. psilocybin study catalyzed further such research
One of the studies that catalyzed the so-called psychedelic renaissance was published with the title, “Psilocybin can occasion mystical-type experiences having substantial and sustained personal meaning and spiritual significance” (Griffiths et al., 2006). The path from conception to initiation was challenging, with many points along the way that could have prevented the study. These included multiple protocol reviews by different oversight entities, in which a denial by any could have prevented the study for legal, ethical, safety, and other reasons. The reviewers included the Johns Hopkins Institutional Review Board, DEA, and FDA, and permission to conduct the studies was required by Johns Hopkins University itself.
The process took several years, but by 2004, Griffiths and colleagues had negotiated the labyrinth of required approvals that went hand in hand with the development of an acceptably safe and scientifically promising first such study. It was completed in 2005 and submitted for publication in January 2006.
Safety of the participants was the number one priority; however, a study must also be scientifically sound in design and the plan for its conduct to be considered ethically acceptable and with a favorable expected benefit-to-risk balance (NIH, 2016). Thus, the pivotal study was designed using key elements of human studies employed by Griffiths and colleagues in dozens of studies occurring over the three previous decades (e.g., Carter and Griffiths, 2009; Griffiths et al., 2003). Some of the practices they developed have since been adopted by other researchers and were later incorporated into two FDA guidance documents (U.S. FDA, 2017, 2023) and discussed by several FDA staff in a review article addressing psychedelic research (Calderon et al., 2023).
Griffiths’ approach to the psychedelic study included some elements from human abuse liability study designs but was not claimed to be an abuse potential study due to modifications for safety reasons, as has been discussed elsewhere (Henningfield et al., 2023). Similar to abuse liability studies, a comparator drug was tested (methylphenidate), with both drugs tested at a dose expected to produce robust effects. The study also included the classic abuse liability assessment instrument, the ARCI, but not the FDA-recommended bi-polar visual line analog Drug-Liking scale, which is typically the primary outcome measure recommended by the FDA for human abuse liability studies (U.S. FDA, 2017).
In this study, Griffiths et al. administered 30 mg per 70 kg of psilocybin. The 40 mg per 70 kg methylphenidate comparator was selected because it was a CSA Schedule II stimulant known to produce robust subjective effects, including euphoria, and had a similar onset and duration of acute psychological effects as psilocybin with peak effects within about 2 hours and substantially reduced within 6 hours. Thirty participants were each tested with both drugs (with the order of the conditions randomly assigned). Six additional participants received methylphenidate in two sessions, followed by unblinded psilocybin on a third session.
Prior to drug administration, potential participants were evaluated to ensure that they were “medically and psychiatrically healthy, without histories of hallucinogen use, and without family histories of schizophrenia or other psychotic disorders or bipolar I or II disorder” (p. 2). Participants all gave their informed consent following the approach and form approved by the Institutional Review Board of the Johns Hopkins University School of Medicine.
Each volunteer study participant had a primary same sex monitor and a secondary opposite-sex monitor. As described by Griffiths et al. (p. 3) “The primary monitor met with each participant on four occasions before his or her first session (for 8 hours total) and on four occasions (for 4 hours total) after each session. A major purpose of the participant-monitor meetings was to develop and maintain rapport and trust, which is believed to minimize the risk of adverse reactions to psilocybin.”
The individualized session monitor approach was among the features of the study intended to reduce the risk of emergence of serious adverse effects and optimize the likelihood of outcomes that participants would consider positive. It is part of the approach to establishing appropriate “set and setting” or “mindset” and environment that is expected to minimize the risks of adverse events such as paranoia and panic and increase the likelihood of outcomes considered positive by study participants (Griffiths et al., 2006; Leary et al., 1963; Metzner et al., 1965; Pahnke, 1963, 1969). Thus, the test rooms and music background were more living room-like in their appearance and feel than the test areas that are more hospital-like, common in most institutions conducting clinical pharmacology studies. Research on set and setting, including music selections and other factors to suit differing population, continue to be researched by Griffiths’ colleagues (e.g., Barrett et al., 2017b, 2018b; Strickland et al., 2020).
Griffiths and colleagues recognized that contributors to set and setting, including the approach to session monitor incorporation into the design of the study, had the potential to produce expectancy effects and provided the following rationale: Some expectancy effects are unavoidable because it would be unethical not to inform both the participants and the session monitors about the range of possible effects with hallucinogens. In addition, it is believed by many that ethical hallucinogen administration requires that sessions be monitored by individuals who are familiar with such altered states of consciousness (Masters and Houston, 1966; Pahnke, 1967; Roberts, 2001; Stolaroff, 2001).
While it was not possible to completely eliminate such expectancy effects, it was possible to significantly reduce such effects by studying participants without personal histories of hallucinogen use, by studying only a single participant at a time, and by using an experimental design and instructional sets that provided the expectation that sessions could involve not only the administration of a wide range of psilocybin doses but also a range of novel drugs, some of which could produce effects that overlap with those produced by psilocybin.
In fact, Pahnke’s “Good Friday experiment” (Pahnke, 1963, 1967, 1969) provided both some of the inspiration for the 2006 Griffiths et al. study, as well as some of the approaches to designing the set and setting, although other many other studies informed the design and there were important differences (for further discussion see Yaden and Newberg, 2022). Basic cardiovascular safety measures and evaluation of behavioral effects by a session monitor rating scale that included 20 dimensions 12 were collected at 30, 60, 90, 120, 180, 240, and 360 minutes after drug administration, with additional questionnaires to capture potential effects related to state of consciousness, hallucinogenic, mystical-type, and other experiences. These design features contributed to characterizing the experience while also documenting adverse events. These and other design features to ensure safety in this study and subsequent studies are discussed further in this section and in other publications (Barrett et al., 2016; Garcia-Romeu et al., 2021; Henningfield et al., 2023; Johnson et al., 2008; Nayak and Zahid, 2025; Van Elk and Yaden, 2022).
Perhaps the most important outcome was the demonstration that, at least under the conditions of this study, psilocybin could be safely administered and was tolerable to the participants. In this study, 11 of the 36 participants rated “experience of fear” at some point during the session to be “strong” or “extreme” on the States of Consciousness Questionnaire. These were tolerated, and the effects were reported to be managed with reassurance by the monitors during the sessions.
The time course of the acute effects of the two drugs on systolic blood pressure and overall drug effect as reported by the monitors is shown in Figure 6. Psilocybin effects were stronger on both measures, and the time course of observed behavioral and emotional effects was generally similar on various other effects by the session monitors.
Time-course of observer-rated effects since capsule administration (time 0 was the 10 minutes before administration baseline). (This figure has been simplified and revised from the original by Griffiths et al., 2006).
Both drugs produced elevations on the scales of the ARCI that have been validated and used in many studies to characterize effects produced by amphetamine—the Amphetamine scale; euphoriants such as morphine and Benzedrine—the
The drugs differentiated substantially on most of the monitor ratings, with psilocybin producing significantly strong effects on several dimensions, including overall drug effect, anxiety or fearfulness, distance from reality, tearing/crying, and joy/intense happiness. A total of 67% of the participants “rated the experience with psilocybin to be either the single most meaningful experience in his or her life or among the top five most meaningful experiences in his or her life” (p. 9).
A Persisting Effects Questionnaire administered two months post study did not show persisting positive or negative effects from methylphenidate but showed significantly stronger persisting positive effects for psilocybin on a variety of outcome measures, including positive attitudes about life and/or self, positive mood changes, and overall sense of well-being or life satisfaction. No participant in either drug condition reported decreased well-being or life satisfaction.
Griffiths and his colleagues followed the 2006 study with several studies that systematically replicated and extended the findings to better understand their reliability and generalizability, including follow-up evaluations of the participants of the 2006 study demonstrating potential positive enduring effects of psilocybin administration that persisted for at least 14 months after dosing (Griffiths et al., 2008, 2011; Reissig et al., 2013). These studies were important precursors to evaluation of psilocybin for potential therapeutic use including existential distress and depression associated with cancer and then other potential therapeutic applications (Garcia-Romeu et al., 2014; Griffiths et al., 2016; Grob et al., 2013).
As discussed elsewhere (Belouin and Henningfield, 2018; Belouin et al., 2022; Xi et al., 2023), Griffiths and colleagues and Johns Hopkins University were not alone in efforts to rekindle clinical psychedelic research and potential medicinal applications, but their involvement and leadership unquestionably helped to re-legitimize and catalyze further research and investment by philanthropies, and finally, in 2021, NIH and the Veterans Administration in the US.
Cultivating safe, ethical, and scientifically sound psychedelic research
Although the 2006 Griffiths and colleagues study was designed for safety and to be scientifically sound so as to optimize reliability, this study was considered the starting point at Hopkins and not represented as the final optimal study approach. Griffiths, his mentors, and many colleagues took the broad aspirational view of science as an ever-evolving endeavor, self-correcting, evidence-guided, and generalizability-assessing in perpetuity. Evolution of the methods, staff training, informed consent protocols, participant recruitment and medical screening, drug dosing protocols, and comparators are among the many factors that have evolved and will likely continue to evolve.
The evolution of methods to ensure safety, ethicality, and scientific reliability is guided by evidence and experience, and with input from institutional review boards, regulatory oversight, including from the FDA and DEA, as well as funding agencies and their reviewers, such as NIH. The process of peer-reviewed publishing and scientific meeting presentations has been another rich source of input from colleagues and on-going discussion guiding advances and evolution related to participant safety, ethics, and scientific methods.
Thus, for example, whereas at Johns Hopkins and some other institutions, early test rooms may have included cultural artifacts to enhance potential spiritual aspects of the experience, over time the rooms have become more culturally neutral. Other practices have been put into place to reduce anxiety and provide healing settings that feel less like the sterile hospital and scientific laboratory settings of the past, as discussed by Gukasyan and Nayak. These concepts are not unique to psychedelic research but are increasingly being evaluated and incorporated into the architecture and style of setting for birthing and psychotherapy worldwide (e.g., Hagerup et al., 2024; Nielsen and Overgaard, 2020). Thus, the room illustrated below is an example of the rooms of research and healing at the Bellevue Hospital in New York that are generally similar to those at Johns Hopkins and in several other leading psychedelic research programs (Figure 7).

An example of the increasingly adopted style of rooms for research with volunteer participants in psychedelic research. This example was in use by Michael Bogenschutz and colleagues at the New York University School of Medicine, affiliated with Bellevue Hospital in (with permission from Dr. Michael Bogenshutz).
With time and experience, methods have continued to evolve, with safety always foremost, but with efforts to address challenging issues including the most appropriate control and comparator conditions for assessing the efficacy of psychedelic medicines for potential approval by the FDA, and other medicines authorities worldwide, the durability of therapies and potential need for greater flexibility in dosing and potential booster therapies over time as needed by patients, the potential for incorporation of group therapy approaches to provide access to greater numbers of people without sacrificing safety or efficacy and more. These and more challenges and issues are under active exploration at Johns Hopkins and in other psychedelic research programs worldwide, as well as by pharmaceutical developers, as evidenced in this special issue of the Journal of Psychopharmacology and briefly summarized in its Introduction (Agrawal et al., 2024; Beaussant et al., 2024; Heal et al., 2018, 2023, 2025; Thrul et al., 2024).
Another challenge is the selection of the most appropriate comparator and control conditions for evaluating the efficacy of therapies as well as for exploration of the neuropharmacological mechanisms of action (Barrett and Griffiths, 2018; Gukasyan and Nayak, 2022; Nayak et al., 2023; and see commentary in this special issue by Nayak and Zahid, 2025). There may be no perfect placebo, and indeed many drugs used to treat psychiatric disorders have effects that are not readily mimicked but devoid of potential efficacy. Moreover, for people with life-threatening conditions, it is often difficult to justify conventional placebo “therapy” and so active comparators and cognitive behavioral therapies, and other comparators are used across studies with efficacy ultimately determined by the cohesiveness of the findings from differing approaches and studies in suggesting efficacy. This has been reviewed elsewhere and commented on in this special issue, and illustrated by the diversity of approaches employed in studies presented and reviewed in this special issue and in earlier special issues of other journals on psychedelics since 2018 (Curran et al., 2018; Heal et al., 2018, 2023; Nayak et al., 2023; Nayak and Zahid, 2025; Wen et al., 2024).
Discussion and additional perspectives
Most psychoactive substances have a history of social use that helps inform research and regulation. Psychedelics illustrate this point well as the CSA regulation of many of them was influenced by real-world reports and perceptions regarding their apparent abuse-related effects, along with some research that addressed their neuropharmacology. Griffiths was aware of the cultures of use around compounds like alcohol, caffeine, nicotine, benzodiazepines, and others that he studied. But of all of these, Griffiths found the cultural components of psychedelics were both most interesting—as well as most concerning, a topic he spoke and wrote about at length (e.g., Yaden et al., 2022; Yaden and Griffiths, 2021).
Griffiths’ interest in psychedelics started with his personal interest in meditation. As mentioned, later in his life, Griffiths became interested in the effects of meditation on states of consciousness, becoming an avid meditator in the 1990s. He experienced what he described as profound and lasting beneficial effects from the practice. As he told in lectures and interviews 11 , when Griffiths heard accounts that psychedelic substances could produce states of mind similar to meditative states, his first response was skepticism that this could be possible, followed closely by his characteristic curiosity. While he was somewhat aware of traditions of ceremonial use of psychedelics among indigenous groups (Magar et al., 2025; Urrutia et al., 2023), he said he had difficulty imagining these substances could come close to his meditation experiences with respect to personal meaning. So, as he had done across several other classes of substances, he sought to study the effects experimentally. His efforts further contributed substantially to an article on this topic addressing traditional practices by Indigenous Peoples (Urrutia et al., 2023; see also discussion by Magar et al., 2025 in this special issue).
The results of his 2006 study indicated that approximately two-thirds of the participants reported that their experience with psychedelics was among the most meaningful moments of their life. This observation—something he had never heard from participants in all his years of administering a wide variety of psychoactive compounds—sparked a fascination in the scientific study of psychedelics that would last until the end of his life. It seemed to him that psychedelics, under appropriate conditions, could produce profound states with some similarities to those he had experienced in meditation. He believed that this observation may help to explain psychedelics’ tendency to be socially positioned around ceremonial use and religious/spiritual concepts.
Griffiths was engaged in discussions about whether the acute subjective effects of psychedelics, such as “mystical-type experiences” and other cognitive and affective responses, are necessary for their full and enduring therapeutic outcomes. Together with his trainee David Yaden, Griffiths debated this issue with Olson et al. (2021). Olson argued that subjective psychedelic experiences might not be essential, pointing to promising therapeutic possibilities from non-hallucinogenic analogs. Griffiths and Yaden, emphasizing their extensive empirical findings, maintained that subjective experience remains associated with beneficial outcomes which may be important for understanding and optimizing psychedelics’ therapeutic potential, at least this is what the current state of the science suggests. Griffiths was open to being shown wrong and suggested tests that could demonstrate his view was incorrect (see more recent discussion by Garcia-Romeu, 2025).
For Griffiths, the intersection of psychedelic compounds and use in religion/spiritual contexts was a source of both deep curiosity and concern. He had found meditation practice personally positively transformational, and he wondered whether psychedelics could facilitate objectively demonstrable, reliable beneficial effects under the right conditions. However, he was aware of both the cultish practices that can take hold in ceremonial and spiritual contexts and the reactive and oppressive tendencies that organizations can take on, and how psychedelics could potentially exascerbate these problems. On the other hand, many individuals find great comfort and satisfaction in spiritual groups and religious traditions and how psychedelics interact with various worldviews seemed to him worth studying.
Anxiety about potential harms from psychedelics extended beyond the experiences themselves and how they might interact with worldviews to address society in general. In one of Griffiths’ last public appearances, he deliberately opposed Rick Doblin’s view of moving fast (“society is ready for psychedelics now”) to claiming that it may take decades for society to wisely incorporate psychedelics.
One of Griffiths’ postdoctoral trainees, Yaden, a research psychologist who did his doctoral training at the University of Pennsylvania, shared these concerns. Griffiths and Yaden wrote a pair of articles in JAMA Psychiatry discussing their concerns: “Keeping the Renaissance From Going Off the Rails” (also co-authored by Yaden’s wife, Mary (Bit) Yaden, a psychiatrist at Johns Hopkins) and “Preparing for the Bursting of the Psychedelic Hype Bubble” (also-co-authored by the chair of the Department of Psychiatry and Behavioral Sciences, James Potash; Yaden et al., 2021, 2022). The concerns about over-hasty, non-evidence-based, and otherwise unwise applications of psychedelic research findings expressed in these articles were routinely discussed and emphasized in Griffiths’ public lectures and other interviews and writings.
These concerns are also relevant to the goal of potential eventual FDA approval of some of these drugs for therapeutic use. A key premise in the review of new drug applications for therapeutic use by the FDA and other medications regulatory authorities worldwide, is that there must be conditions for approval, including the labeling, controlled substance scheduling as appropriate, and risk management programs that assure safety and a favorable benefit-to-risk ratio. As discussed earlier in this review, numerous studies appear to be well on the way to providing the evidence for potential FDA approval, whereas uncontrolled and unsupervised use of psychedelics can result in serious adverse events and sometimes injury and death (e.g., Hinkle et al., 2024).
Griffiths was also curious and concerned about how to characterize and label psychedelic experiences that were reported to be particularly profound and beneficial by participants. Following psychedelic researchers in previous decades, he used a measure of mystical-type experience. The term “mystical experience” comes from William James’ foundational work on the science of intense and meaningful altered states of consciousness (James, 1902; Yaden and Newberg, 2022). A self-report measure of mystical-type experience was designed in the 1960s by psychedelic researcher Pahnke (1963, 1967, 1969) and was refined and iterated over the years by others, including another of Griffiths’ trainees, Barrett et al. (2015; now the director of the Johns Hopkins CPCR). Barrett recounted a discussion in which Griffiths said, “In retrospect I rather regret calling it the mystical experience questionnaire” and “it is defined by the constellation of empirically measured phenomenological dimensions” by which he means self-reported “it doesn’t necessarily imply supernatural or non-rational explanation.”
One of his final publications described a need to transcend the mystical experience construct as insufficiently comprehensive and too prone to confusion, especially by those unable or unwilling to understand the difference between a colloquial meaning and a technical meaning of the same term (Yaden et al., 2024). The focus was consistent with Griffiths earlier growing interest in exploring consciousness and subjective/emotional states such as spirituality, connection, and awe as effects or dependent variables amenable to study by neuropsychopharmacology methods. A particular personal fascination as he put it was the ability “to be aware that we are aware” but in discussion with at least one of the coauthors of this review, he carefully distinguished his interest in studying various aspects of spirituality as legitimate self-report variables from the interest of some in studying “spirits,” which was too great a leap of faith for him.
Similarly, the inclusion of “consciousness” in the name of the research center he helped to establish and secular spirituality in the professorship he established reflected this thinking and belief that such research was legitimate and important. Nonetheless, some of Griffiths’ colleagues and others at Johns Hopkins were critical of including the word “consciousness” in the name of the center, acknowledging how the field of consciousness research itself is still fledgling and beset with many challenges and controversies. Indeed, there was also nothing directly empirically centered on consciousness in the studies conducted at Hopkins before and at the time of founding the center. However, Griffiths was adamant on the importance of including “consciousness,” even as an aspirational goal of this research program, and the potential for psychedelics to help address questions and problems of consciousness. He was excited by helping foster such research, in which it was not predictable where it would go or what would emerge. His training in behavioral and neuropharmacology provided the foundation for both his view and his methods, that so-called “subjective” reports of interoceptive effects and feelings were amenable to scientific study as was widely accepted in clinical studies of abuse liability, depression, pain and many other areas of exploration that rely heavily on research participant and patient self-reports.
Within a few years of the establishment of the CPCR, there was increasing appreciation of his commitment to using rigorous neuropsychopharmacology methods to explore consciousness, spirituality, and other constructs and its relevance to healing and well-being. At the ceremony installing Griffiths as the inaugural recipient of the Installation of the Oliver Lee McCabe, III, PhD Professor in the Neuropsychopharmacology of Consciousness, on January 30th, 2023, Johns Hopkins University President Ronald J. Daniels stated that he was proud to be president of a university that was exploring consciousness and spirituality as these were not only important frontiers for research, but vital dimensions in humanity, healing, and the practice of medicine.
In general, Griffiths was more interested in using psychedelics as a research tool to study experiences that some people consider religious or spiritual. His attempt to reconcile his curiosities and his concerns at this intersection of issues was reflected in his study of a wide range of populations adjacent to this question, including beginning meditators (Griffiths et al., 2016), those with an established meditation practice, and religious leaders (Griffiths et al., 2019), with a planned study on atheists at the time of his death.
This is also reflected in his efforts, initiated a few months after his stage four cancer diagnosis, to establish a professorship that would ensure the pursuit of such research in perpetuity following his death. With the support of Johns Hopkins and many contributors and philanthropies, The Roland R. Griffiths, PhD Professorship in Psychedelic Research on Secular Spirituality and Well-being was established, and its inaugural recipient, David B. Yaden installed several months before his death on June 14, 2023. “This professorship will advance the scientific understanding of the relationships between the uses of psychedelic substances, the possible resulting influences on spirituality and worldview, and the subsequent effects on psychological well-being, prosocial behavior, and health.” 13 The professorship will support research on these topics in perpetuity.
We close this review with appreciation and a commitment to continue the pursuit of such humanity-serving scientific research as exemplified by our friend and colleague, Roland R. Griffiths (Table 6).
Psychedelic foundational and medicine development studies co-authored by Griffiths.
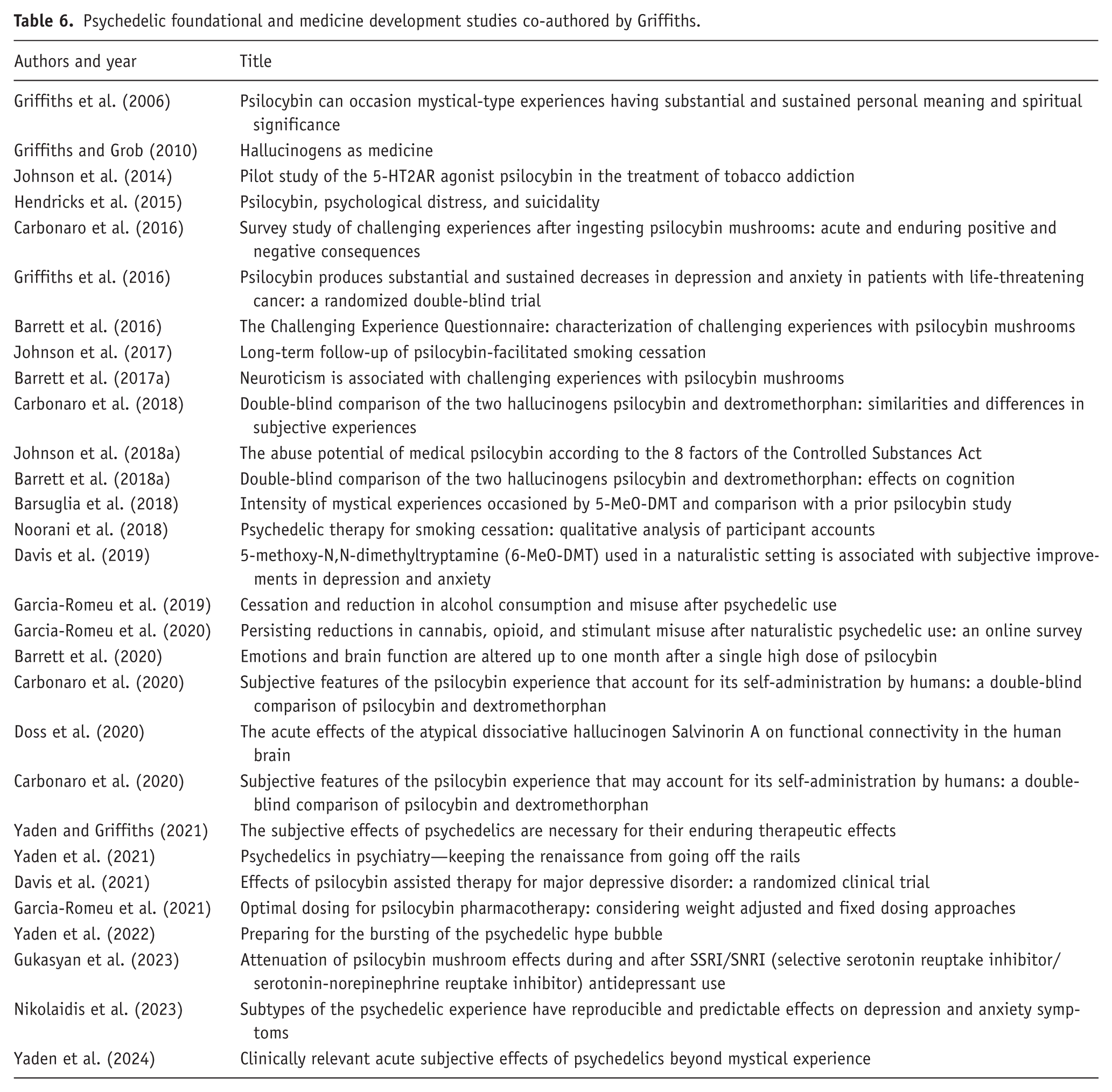
Footnotes
Acknowledgements
Acknowledgments for contributions to perspectives and history: George E. Bigelow, John Grabowski, Donald W. Kodluboy, Maxine Stitzer, Marla Weiner, and for assistance in editorial review, referencing, and development of figures and tables: Floe Foxon, Megan Harris, Christine Sweeney, and Daniel Wang.
Author note
This review was written for a special issue of the Journal of Psychopharmacology on psychedelic drug research that is dedicated to Roland R. Griffiths, PhD, a long-standing member of its International Editorial Board until his passing on October 16, 2023.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jack E. Henningfield is an employee of Pinney Associates, Inc. which provides scientific and regulatory consulting support for new drug applications (NDAs) and risk management programs for a broad range of CNS active substances and drug products including psychedelic substances, new chemical entities, and alternative formulations and routes of delivery, as well as dietary ingredient notifications, cannabinoid assessment, and noncombustible tobacco/nicotine products for FDA regulation. He received no external financial support for writing this article and no external commercial interests had any input. FSB is a scientific advisory board member for MindState Design Labs, LLC, and Lilly USA. AGR is a paid scientific advisor to Otsuka Pharmaceutical Development and Commercialization Inc., and Chair of the Scientific Advisory Board for Psyence BioMed.
