Abstract
Background:
Psilocybin has shown rapid and sustained antidepressant effects in patients with major depressive disorder, yet the neurobiological mechanisms underlying these outcomes remain unclear.
Aims:
This study aimed to bridge clinical and preclinical findings by investigating the relationships between 5-HT2A receptor occupancy (RO) achieved after administration of psilocybin and its effects on behavior and markers of neuroplasticity in mice.
Methods:
In vivo 5-HT2A RO was determined via displacement of [3H]MDL-100,907 in the prefrontal cortex (PFC). To relate RO with behavioral outcomes of psilocybin, we assessed the head twitch response (HTR) acutely and investigated behavior in the elevated zero maze (EZM) and forced swim test (FST) 20–24 hours post-drug. Neuroplastic changes were assessed by measuring α-tubulin post-translational modifications (PTMs) and expression of key synaptic proteins in both the PFC and amygdala.
Results:
Psilocybin produced dose-dependent 5-HT2A RO (RO₅₀ = 0.88 mg/kg) and an inverted-U dose–response in HTR, with peak effects occurring between ~44% and 62% RO. Behaviorally, a 1.5 mg/kg dose increased the open areas ratio in the EZM, while 3 mg/kg reduced immobility in the FST, 20 and 24 hours after dosing, respectively. Both dose levels shifted α-tubulin PTMs toward a more dynamic microtubule state and selectively increased synaptic marker expression in the PFC, not in the amygdala.
Conclusions:
These findings suggest that the therapeutic effects of psilocybin could be mediated by dose- and region-specific enhancement of neuronal plasticity, with distinct signatures associated with anxiolytic-like and antidepressant-like properties.
Keywords
Introduction
The seminal works by Charles Grob and Roland Griffiths demonstrating the efficacy of psilocybin in treating anxiety and or depression in patients with terminal illness were instrumental in 5-HT2A agonists being considered as potential therapeutics for the treatment of mood and anxiety disorders (Griffiths et al., 2016; Grob et al., 2011). Recent clinical trials of the effects of psilocybin in the treatment of major depressive disorder (MDD) and treatment-resistant depression have shown extremely impressive results after only 1–2 doses, suggesting that psilocybin could be approved as a rapid-acting antidepressant in the near future (Goodwin et al., 2022; Raison et al., 2023). It is apparent that psilocybin produces antidepressant efficacy in humans that is both rapid acting (i.e., immediate symptom relief within hours) and durable (efficacy persists for weeks to months after treatment), and this is likely true for other classical psychedelic 5-HT2A agonists. However, our understanding of the physiological and/or pharmacological mechanisms underlying the durability of psilocybin’s antidepressant efficacy has lagged efforts to develop psilocybin into a clinical treatment.
In humans, the psychedelic effects of psilocybin are mediated by agonist activity at 5-HT2A receptors (Vollenweider et al., 1998). In rodents, the head twitch response (HTR) assay (originally described by Corne et al., 1963) is commonly used as an in vivo proxy for 5-HT2A activation and to study the effects of psychedelic compounds, as potency in this assay strongly correlates with the potency in humans (Halberstadt et al., 2020). It is well established that the HTR is driven primarily through 5-HT2A receptor activation, and the effects of various psychedelic 5-HT2A receptor agonists can be blocked by the selective 5-HT2A receptor antagonist, M100,907 (Halberstadt, 2015). While the HTR is directly linked to activation of 5-HT2A receptors, other 5-HT (and non-5-HT) receptor subtypes can modulate the HTR; notably, agonists at 5-HT1A receptors reduce the expression of the HTR caused by 5-HT2A agonists (Shahar et al., 2022). Typically, the total number of HTRs is counted over a period of 20–30 minutes after dosing and forms a characteristic ‘inverted-U’ dose–response curve (De La Fuente Revenga et al., 2022; Fantegrossi et al., 2008; Halberstadt and Geyer, 2013, 2011, 2016). However, how this behavior relates to occupancy of central 5-HT2A receptors has yet to be demonstrated.
While there have been no clinical studies investigating whether the therapeutic effects of psilocybin are also mediated by activation of 5-HT2A receptors, this is more than likely given that multiple 5-HT2A agonists (including dimethyltryptamine (DMT), 5-methoxy-dimethyltryptamine, and lysergic acid diethylamide (LSD)) have been shown to have therapeutic effects in depression or anxiety disorders (Jacobsen et al., 2024; Reckweg et al., 2023). The preclinical literature has mixed results demonstrating the role of this receptor in the antidepressant-like and anxiolytic effects of psilocybin (Cameron et al., 2023; Hesselgrave et al., 2021; Sekssaoui et al., 2024; Takaba et al., 2023). There have also been mixed reports on the durability of the antidepressant-like effects of psilocybin in rodents (Hibicke et al., 2020; Jefsen et al., 2019; Takaba et al., 2023; Wang et al., 2025). A recent multi-institutional effort to characterize the behavioral effects of psilocybin (2 mg/kg, intraperitoneal) in mice found several robust and replicable acute effects on increased anxiety and avoidance behaviors, as well as reduced fear expression. However, they did not find any reliable effects 24 hours post-psilocybin on reducing anxiety or depression-like behaviors, or on facilitating fear extinction (Lu et al., 2025). These discrepancies in the literature may be due to the dose of psilocybin/psilocin, the timing between drug administration and testing, the specific assay evaluated, or whether the experiment was conducted in untreated animals or those that have undergone perturbations intended to manifest aspects of depression, such as stress or inflammation. Any one of these variables could affect the results and could help explain the variability seen across preclinical studies of psilocybin.
While the mechanism through which 5-HT2A agonists achieve antidepressant efficacy has not been conclusively established, given that the clinical efficacy of psilocybin markedly outlasts (multiple weeks) the pharmacokinetic effects (<12 hours; Brown et al., 2017; Holze et al., 2023; Otto et al., 2025), increased neuroplasticity is strongly implicated in the durability of the efficacy. Preclinically, psilocybin has been shown to increase dendritic spine density in the prefrontal cortex (PFC) in vivo (Shao et al., 2021) as well as increase the expression of synaptic plasticity biomarkers, such as brain-derived neurotrophic factor (BDNF) and synaptic vesicle glycoprotein 2A (SV2A) (Raval et al., 2021; Shahar et al., 2024). Other 5-HT2A agonists such as LSD, DMT, and 2,5-dimethoxy-4-iodoamphetamine (DOI) have also been shown to induce neuritogenesis both in vitro and in vivo through increased dendritic arbor complexity, dendritic spine growth, and increased synapse formation, hypothesized to occur through a combination of TrkB, mTOR, and 5-HT2A signaling (Ly et al., 2018; Ohtani et al., 2014). These lasting structural and functional changes in neurons are likely preceded by cytoskeletal re-organization to facilitate both the structural changes associated with neurite outgrowth and spine enlargement, and delivery of key molecular components to drive functional synaptic connectivity (Dakic et al., 2017; Ohtani et al., 2014; Schätzle et al., 2018). Indeed, pharmacological stabilization of microtubules using paclitaxel, or inhibition of microtubule polymerization using nocodazole, inhibits long-term potentiation-induced synaptic plasticity and memory formation (Barnes et al., 2010; Jaworski et al., 2009; Shumyatsky et al., 2005).
Microtubule structures, composed of α- and β-tubulin heterodimers, are critical for the formation and maintenance of the complex neuronal cytoskeleton that provides structural neuronal stability during basal periods as well as periods of neuronal projection growth and shortening. Post-translational modifications (PTMs) of α-tubulin, such as N-terminal acetylation at position Lys-40 and the C-terminal enzymatic cycle of tyrosination/detyrosination, dynamically regulate microtubule stability and function, influencing processes ranging from axonal transport to dendritic and synaptic remodeling (Moutin et al., 2021). Increases in both N-terminal acetylation and C-terminal detyrosination of α-tubulin associate with decreased microtubule dynamics by reducing microtubule elongation and shortening, which correlates with a reduction in their ability to remodel in response to cellular stimuli (Carmona et al., 2023; Peris et al., 2009; Piperno et al., 1987). Conversely, deacetylation of α-tubulin at Lys-40 as well as the re-addition of tyrosine residues to the C-terminus of detyrosinated α-tubulin is associated with an increased dynamic state of microtubules, whereby elongation and shortening of microtubule structures can occur during processes such as neurite outgrowth and synaptic remodeling.
Dysregulation of these α-tubulin PTMs has been implicated in the pathogenesis and treatment of neurological disorders characterized by synaptic pathology, including MDD. Previous studies indicate that behavioral abnormalities in animal models of depression are paralleled by decreased brain microtubule dynamics as observed in restraint stress (Bianchi et al., 2003), social isolation (Bianchi et al., 2006), chronic unpredictable mild stress (Yang et al., 2009), and psychosocial stress models (Parésys et al., 2016). These changes in microtubule dynamics can be rescued, together with depression-like behaviors, by chronic treatment with typical antidepressants such as fluoxetine, or acute treatment with rapid-acting drugs such as pregnenolone-methyl-ether (PME) (Barbiero et al., 2022; Bianchi and Baulieu, 2012; Bianchi et al., 2009a; Yang et al., 2009). Importantly, PME is a neuronal microtubule-targeting compound with no known affinity for central nervous system (CNS) receptors; therefore, its behavioral and molecular efficacy in animal models of depression supports the hypothesis of a potential link between antidepressant efficacy, cytoskeletal remodeling, and neuronal plasticity (Bianchi and Baulieu, 2012; Parésys et al., 2016). More recently, it was shown that α-tubulin PTMs are detectable in human plasma and that a decrease in acetylated α-tubulin was observed in the plasma of healthy volunteers treated acutely with the antidepressant dose of ketamine (0.5 mg/kg, intravenous (IV)) and that those changes statistically correlated with the altered metabolism of glutamate in the pregenual anterior cingulate cortex (pgACC) (Colic et al., 2019). Taken together, these findings are in line with preclinical and clinical evidence indicating a role of neuronal plasticity in the pathogenesis and treatment of depression (Liu et al., 2017; Wang et al., 2023).
The goal of the work described here is twofold: first, we aim to understand the relationship between HTR and behavioral effects in simple pharmacodynamic assays associated with anxiolytic-like and antidepressant-like activity and 5-HT2A receptor occupancy in mice, and assess how this translates to what is known clinically about the degree of target occupancy associated with subjective and therapeutic effects of psilocybin. Next, given the durability of the clinical antidepressant efficacy of psilocybin, to link these acute effects in mice to biochemical measures of neuroplasticity, such as biomarkers of microtubule dynamics (α-tubulin acetylation and tyrosination/detyrosination) and synaptic marker expression (PSD-95, SV2A, and synaptophysin) in the PFC and amygdala.
Methods
Drugs
Psilocybin and ketanserin (Cayman Chemical Company, Ann Arbor, MI, USA) were prepared fresh in 0.9% saline <1 hour prior to injection. Drug doses were calculated as mg/kg free base. Unless otherwise stated, all dosing was subcutaneous (SC) using 0.9% saline as the vehicle and a dose volume of 10 mL/kg.
In vivo receptor occupancy
Receptor occupancy experiments were performed at Kesla Translational Models (Kingston, RI, USA). Studies were conducted in 8- to 10-week-old male C57BL6/J mice (n = 6/group; The Jackson Laboratory, Bar Harbor, ME, USA). Mice were housed in a controlled (temperature: 20°C–24°C; standard 12/12 hours light/dark cycle; lights on at 7 am) vivarium at the University of Rhode Island (URI), an AAALAC-approved animal facility that meets all federal and state requirements for care and treatment of laboratory animals. Mice were housed up to five per cage in a climate-controlled room on a standard light cycle and were provided with ad libitum access to food and water. Mice were randomly assigned to be dosed with vehicle (saline), psilocybin (0.1, 0.32, 1, 3.2, 10 mg/kg; SC), or ketanserin (10 mg/kg, SC) followed 5 minutes later by retroorbital administration (3 mL/kg) of the tracer [3H]MDL-100907 (American Radiolabeled Chemical, Inc., St. Louis, MO, USA; 50 μCi/kg). Mice were euthanized by live decapitation 30 minutes after dosing, and brains were removed (30 minutes was chosen to optimize the ratio between specific and non-specific binding). Brain regions (PFC and cerebellum) were rapidly dissected and homogenized for ~3 seconds each using a polytron at its highest setting (5–6) in 10 volumes (100 mg/mL) of ice-cold assay buffer (50 mM Tris, 2 mM MgCl2, pH 7.4). Aliquots (400 μL each) of the resulting homogenates were filtered using a vacuum manifold apparatus with 25 mm Whatman GF/B filters presoaked in 0.3% polyethylenimine. The filters were soaked overnight in 20 mL of Biosafe scintillation fluid and analyzed the following day using a scintillation counter. Nonspecific binding was determined using the cerebellum of mice treated with vehicle (Knauer et al., 2008). Total binding was measured as counts per minute (cpm) in the frontal cortex of vehicle-treated mice. Specific binding was calculated by subtracting nonspecific binding from the total cpm for each sample. Psilocybin receptor occupancy was calculated as:
HTR and locomotor activity
Automated HTR studies were conducted at Kesla Translational Models (Kingston, RI). Mice were housed under the same conditions as for in vivo RO described above. Testing was conducted between 1000 and 1800 hours. All animal experiments were carried out in accordance with NIH guidelines and were approved by the URI Institutional Animal Care and Use Committee.
Studies were conducted in 8- to 10-week-old male C57BL6/J mice (n = 12 mice/group; Jackson Labs, Bar Harbor, ME). Mice were randomly assigned to be dosed with vehicle or psilocybin (0.1, 0.32, 1, 3.2, and 10 mg/kg, SC; Cayman Chemical Company, Ann Arbor, MI, USA) immediately prior to testing. Mice were placed in behavioral chambers (12″ × 12″ × 12″) and behavior was recorded for 20 minutes using high-speed cameras (240 fps, 2.7k resolution). The data were processed using a custom algorithm in DeepLabCut to detect and classify the HTR while tracking the locomotor activity of the animals. A similar method for the automated detection of HTR was published by Glatfelter et al. (2022) at the National Institute on Drug Abuse (NIDA).
HTR data were analyzed in 2-minute bins. The total number of head twitches in the 20-minute period was measured. In addition, the peak response rate was determined by taking the 2-minute bin for each animal with the highest HTR count and dividing by two to determine the peak rate of HTR/minute.
Elevated zero maze and forced swim test
Male C57BL/6J mice (8–10 weeks old, n = 6–10/group), sourced from an internal breeding colony at Trinity College Dublin, Ireland, were used in these experiments. The animals were group-housed in a controlled environment (temperature: 20°C–24°C; 12/12 hours light/dark cycle; lights on at 7 am), with water and food available ad libitum. Animals were acclimated to the facility environment and handled by experimenters prior to starting experiments. Animal care and procedures were conducted in accordance with European Communities Council Directive (86/609/EEC), and the experimental protocols were approved by the animal ethical committee of Trinity College Dublin and by the Health Products Regulatory Authority of Ireland.
All animals were randomly assigned to treatment groups, with a target group size of n = 8. An additional two animals were included in the vehicle control group. Mice were tested in the Elevated Zero Maze (EZM) 20 hours post-administration of psilocybin (1.5 or 3 mg/kg, SC) or vehicle. The assay was performed as described in Shepherd et al. (1994). Briefly, the apparatus (UgoBasile, Gemonio, Italy, #40163) consisted of an annular track (5 cm in width) with an inner diameter of 60 cm, divided into two opposite open quadrants and two opposite closed quadrants (with side walls 15 cm in height). The entire maze was elevated 60 cm above the floor. At the start of each test session, mice were placed at the end of an open area, facing the closed area, designated as the starting point. The mice were allowed to explore the EZM for 6 minutes. Both open areas had the same level of brightness (150 ± 5 lux), as did both closed areas (90 ± 10 lux). The behavior of the mice was recorded with a top-view camera (Canon) using a tripod with horizontal extension. After the 6-minute EZM session, each mouse was returned to their home cage and the EZM was cleaned to remove odor sources (1% Virkon Rely + On, Lanxess) before testing the next mouse.
Anxiety-related variables were scored by an experimenter unaware of the treatment after the experiment. These variables include the time spent in the open and closed areas. An entry into an open or closed area was only considered valid when all four paws of the mouse were fully inside the area. Behavior in the EZM was represented as a ratio of the percent time exploring the open areas (time in open areas/(time in open areas + time in closed areas) × 100).
Forced swim test (FST) sessions were conducted 24 hours post-administration by placing the same mice in individual glass cylinders (18.5 cm tall, 12 cm diameter) filled with water (25°C ± 1°C) to a depth of 12 cm for 6 minutes. The water was changed between each animal. A digital camera (Canon) positioned directly above the cylinders recorded the swim sessions. After the FST, mice were dried under a heat lamp for 5 minutes and transferred to a CO2 chamber for euthanasia and tissue collection. The last 4 minutes of the FST test videos were manually scored for start and stop times of immobility by observers unaware of the treatment group. Immobility was defined as the absence of movement except to stay afloat, including passive floating where the animal drifts on the water surface without initiating swimming (Crowley et al., 2004).
Molecular biomarker analyses
Animals were euthanized 24 hours post-administration of psilocybin and 5 minutes post-FST. Brains were removed and regions dissected immediately after euthanasia, and were subsequently flash-frozen and stored at −80°C until analysis. PFC and amygdala tissues were homogenized on ice in 300 µL ice-cold lysis buffer (5 mM Tris-HCl, 2 mM EGTA, 2% (w/v) Protease/Phosphatase Inhibitor Cocktail (Sigma #P8340-5M)) using a probe sonicator (QSonica Q55) in 2 × 15 seconds pulses at 15 kHz. Brain homogenates were centrifuged at 10,000 × g for 30 seconds at 4°C to pellet debris, and the supernatant was taken for downstream molecular analyses.
Protein concentration of brain homogenates was analyzed by Bradford Assay and were equalized to uniform concentrations of 0.10 µg/µL for analysis of α-tubulin PTMs, or 3 µg/µL for analysis of synaptic markers, in 2× Laemmli Sample Buffer (Sigma #S3401-10VL). Samples were denatured at 95°C for 5 minutes prior to loading 10 µL per well (1 µg/well for analysis of α-tubulin PTMs, 30 µg/well for analysis of synaptic markers) of NuPage Novex Bis-Tris 26-well Gels, followed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Proteins were transferred to polyvinylidene fluoride (PVDF) membranes using the Invitrogen™ iBlot™ 2 system at 20 V for 10 minutes. Membranes were blocked for 1 hour at RT using Odyssey Intercept™ Tris-buffered saline (TBS) Blocking Buffer (LI-COR Biosciences, Lincoln, NE, USA) and incubated in primary antibodies overnight at 4°C on a circular rocker (acetylated α-tubulin (T6793, 1:6,000), total α-tubulin (EP1332Y, 1:2,000), tyrosinated α-tubulin (T9028, 1:500), de-tyrosinated α-tubulin (AB3201, 1:500), PSD95 (MA1-045, 1:2,000), SV2A (AB32942, 1:2,000), synaptophysin (AB32127, 1:1,500), and glyceraldehyde 3-phosphate dehydrogenase (AB8245, 1:5,000)). Membranes were washed for 3× 10 minutes in TBS (0.1% (v/v) Tween-20) before incubation in secondary antibodies for 1 hour at RT. Secondary antibodies were diluted in Odyssey Intercept™ TBS Blocking Buffer (LI-COR Biosciences, Lincoln, NE, USA) with 0.01% (w/v) SDS (IRDye® Goat anti-Rabbit 680RD (926–68,071, 1:10,000) and IRDye® Goat anti-Mouse 800CW (926–32,210, 1:10,000)). After incubation with secondary antibodies, membranes were washed for 3× 10 minutes in TBS (0.1% (v/v) Tween-20) before imaging with infrared LI-COR Odyssey CLx and signal intensity analyzed with LI-COR Image Studio software (LI-COR Biosciences, Lincoln, NE, USA).
Statistics
Total HTR and total distance data were analyzed using one-way ANOVA with Dunnett’s post hoc multiple comparisons test. The time course data were analyzed using two-way ANOVA with repeated measures (Group × Time) and Geisser–Greenhouse corrections for sphericity violations were applied (ε < 0.75). Median effective doses (ED50/RO50 values) and 95% confidence intervals (95%CI) were calculated with nonlinear regression.
FST/EZM data were analyzed using one-way ANOVA, followed by Fisher’s least significant difference (LSD) post hoc multiple comparison test. One animal (1.5 mg/kg psilocybin group) was excluded from analysis as it failed to engage with the task (biological non-responder). No statistical outliers were identified or excluded.
Prior to statistical analyses of molecular Western Blot data, Western Blot data were normalized and presented as the fold of the vehicle mean for different brain regions and for each analysis performed. Samples were blocked and analyzed by Western Blot across multiple gels, and raw densiometric ratios were first normalized to the mean of the control group within each gel before pooling and analysis of full sample cohorts from multiple gels to control for innate gel-to-gel variability in raw densiometric signals produced. Molecular data were subsequently analyzed by one- or two-way ANOVA with Fisher’s LSD post-hoc multiple comparison test. All statistical analyses were conducted using GraphPad Prism 10.4 (GraphPad, Boston, MA, USA).
Results
Receptor occupancy
Psilocybin (0.1–10 mg/kg, SC) produced dose-dependent displacement of [3H]MDL-100,907 in vivo binding to mouse frontal cortex (Figure 1). The calculated dose achieving 50% displacement (RO50) was 0.88 mg/kg (95%CI: 0.143–6.67). Ketanserin (10 mg/kg, SC) achieved 90% occupancy.
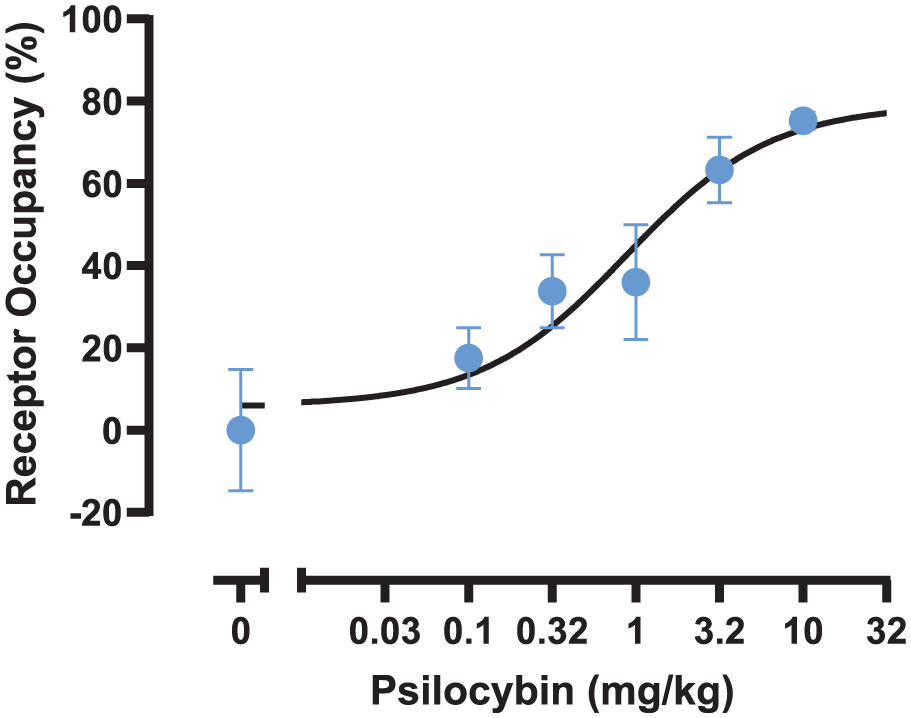
5-HT2A receptor occupancy produced by administration of psilocybin.
HTR LMA mouse
Psilocybin (0.1–10 mg/kg, SC) caused a dose-dependent increase in the total number of HTRs with the typical ‘inverted-U’-shaped dose–response curve with a maximum response at ~1 mg/kg (Figure 2(a)). The higher doses of psilocybin (3.2–10 mg/kg, SC) also caused a significant decrease in the total locomotor activity over the duration of the session (Figure 2(b)).
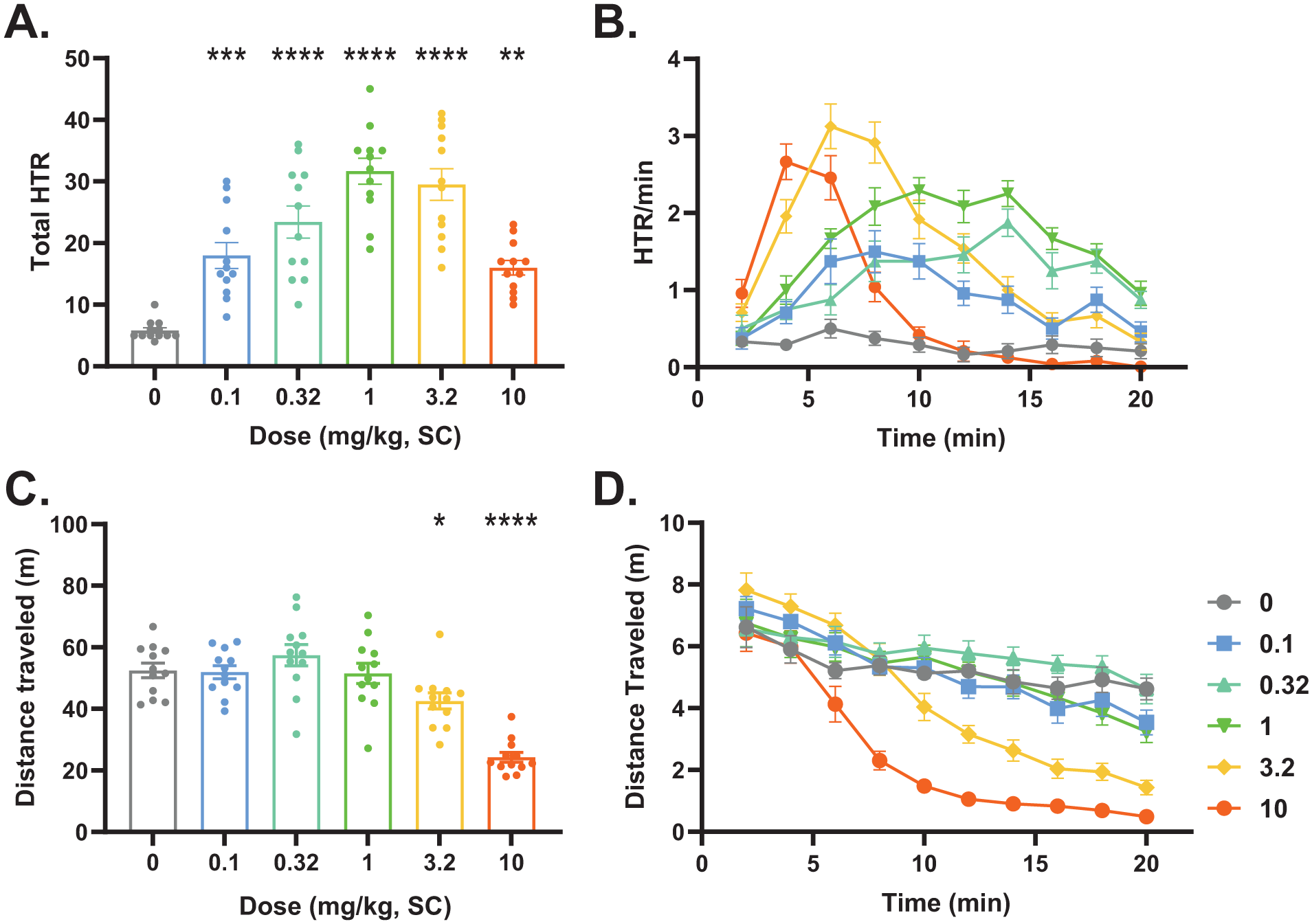
Effects of psilocybin on HTR and locomotor activity.
When the results were analyzed over time (Figure 2(c)), there was a clear difference in the pattern of the HTR response between lower (0.1–1 mg/kg) and higher doses (3.2–10 mg/kg), with the lower doses causing the HTR to increase slowly and plateau, while the higher doses produced a rapid increase followed by a steep decline. At these higher doses, the suppression of locomotor activity coincides with the drop-off in HTR activity (Figure 2(d)).
Fitting the HTR data to a Gaussian distribution, the peak response (Emax = 31.66 HTRs; 95%CI: 28.47–34.97) was calculated to occur at a dose of 1 mg/kg, with an ED50 = 0.145 mg/kg (95%CI: 0.077–0.248) (Figure 3(a)). However, when looking at the rate of HTR over time (Figure 2(c)), the 3.2 mg/kg dose produced a higher peak rate of HTR than the 1 mg/kg dose, despite 1 mg/kg producing more HTRs overall.
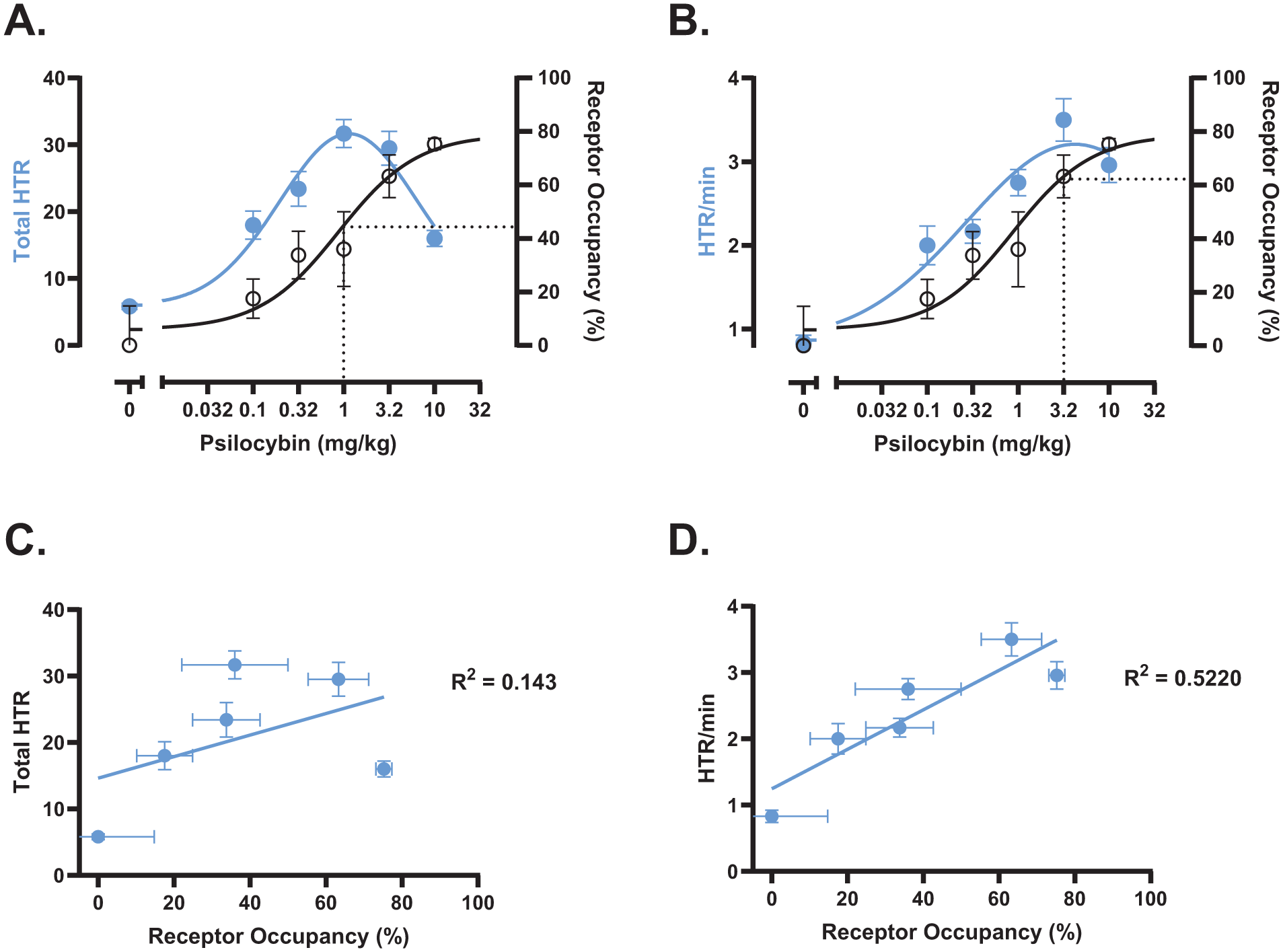
The relationship between 5-HT2A RO and the HTR for psilocybin.
Next, we calculated the average peak rate (over 2-minute bins) and fit a curve to these data using nonlinear regression (Figure 3(b)). The calculated Emax = 3.21 HTR/minute (95%CI: 2.94–3.48) occurred at 3.2 mg/kg, and the ED50 was 0.169 mg/kg (95%CI: 0.053–0.375). Figure 3(c) shows the total HTR plotted as a function of occupancy, while Figure 3(d) shows the peak HTR rate as a function of occupancy. Although both the total count and the peak rate were correlated with 5-HT2A occupancy, the relationship was stronger for the peak rate (R2 = 0.5220, p < 0.0001) than for the total number of HTRs (R2 = 0.1434, p = 0.001), suggesting that the peak HTR rate may be a better in vivo proxy of 5-HT2A activation than the total count.
Elevated zero maze and forced swim test
For behavioral analyses, mice were dosed with psilocybin (1.5 or 3 mg/kg, SC) or vehicle and were tested in the EZM (20 hours post-administration) and FST (24 hours post-administration) (Figure 4).
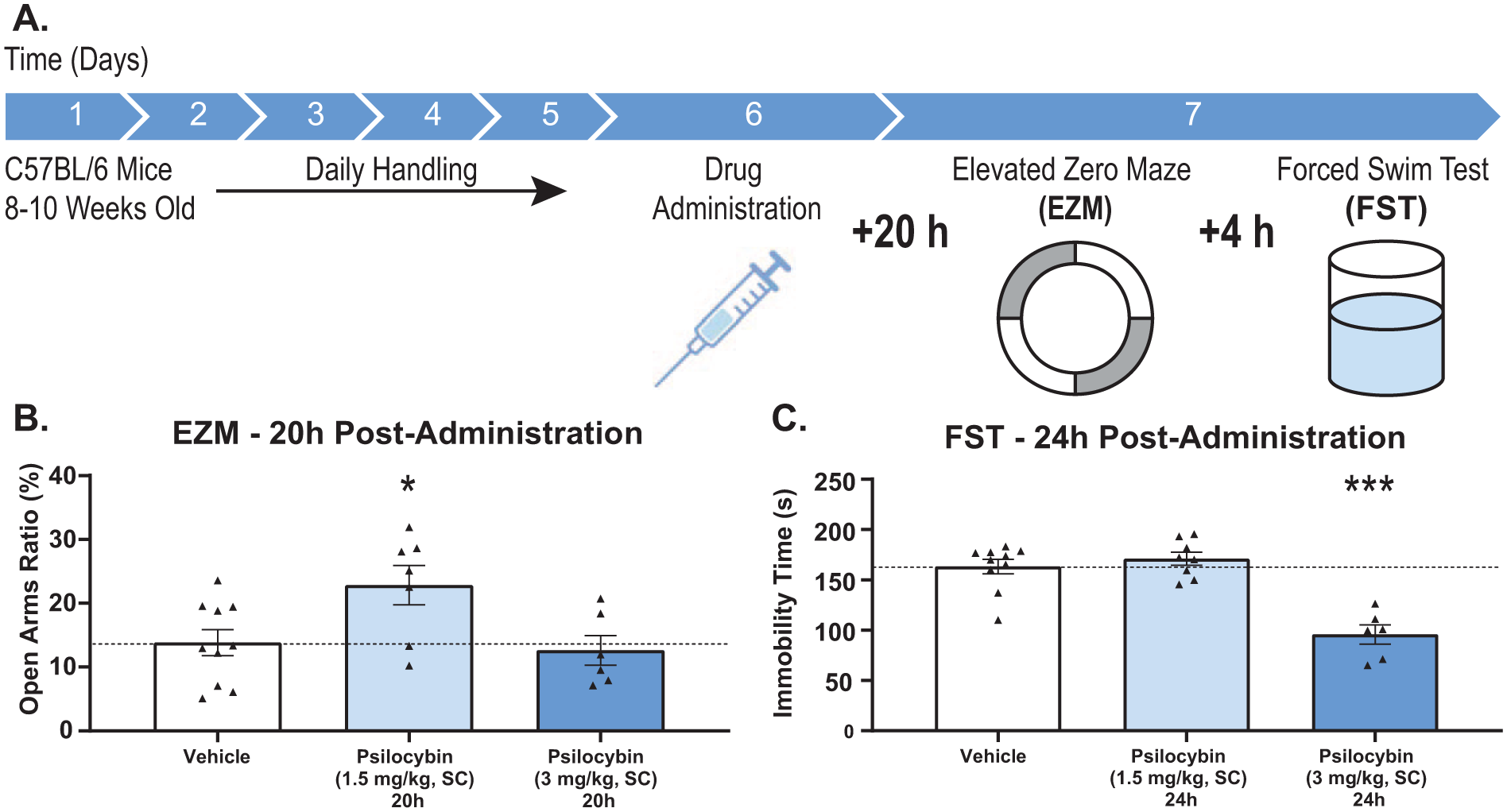
Psilocybin produces dose-dependent effects on mouse behavior in the EZM and FST.
In the EZM, psilocybin-treated animals displayed a significantly increased open arms time ratio at the dose of 1.5 mg/kg, SC (22.83 ± 3.08%, n = 7, p < 0.05) but not at 3 mg/kg, SC (12.61 ± 2.31%, n = 6, p = 0.73), compared to vehicle-treated controls (13.82 ± 2.03%, n = 10).
In the FST, we observed an inverted picture showing that the immobility time was significantly reduced at 3 mg/kg, SC psilocybin (95.68 ± 9.60 seconds, n = 6, p < 0.001), but not at 1.5 mg/kg, SC psilocybin (171.0 ± 6.6 seconds, n = 8, p = 0.26), compared to vehicle controls (158.2 ± 8.23 seconds, n = 11).
Psilocybin increases microtubule dynamics
Psilocybin treatment at 1.5 and 3 mg/kg (SC) produced a dose-dependent decrease in the levels of acetylated α-tubulin in both the PFC and amygdala (Figure 5(a)). 1.5 mg/kg treatment elicited a 30.5% reduction in the PFC (p = 0.0020 vs. Vehicle) and 18.6% reduction in the amygdala (p = 0.0036 vs. Vehicle), while 3 mg/kg treatment elicited a 45% reduction in the PFC (p = 0.0001 vs. Vehicle) and 30% reduction in the amygdala (p = 0.0001 vs. Vehicle). Psilocybin treatment showed a statistically greater effect in reducing the levels of acetylated α-tubulin in the PFC when compared to the amygdala for both 1.5 and 3 mg/kg doses analyzed by two-way ANOVA (Row Factor: F(1,42) = 4.131, p = 0.0485) (Figure 5(a)).
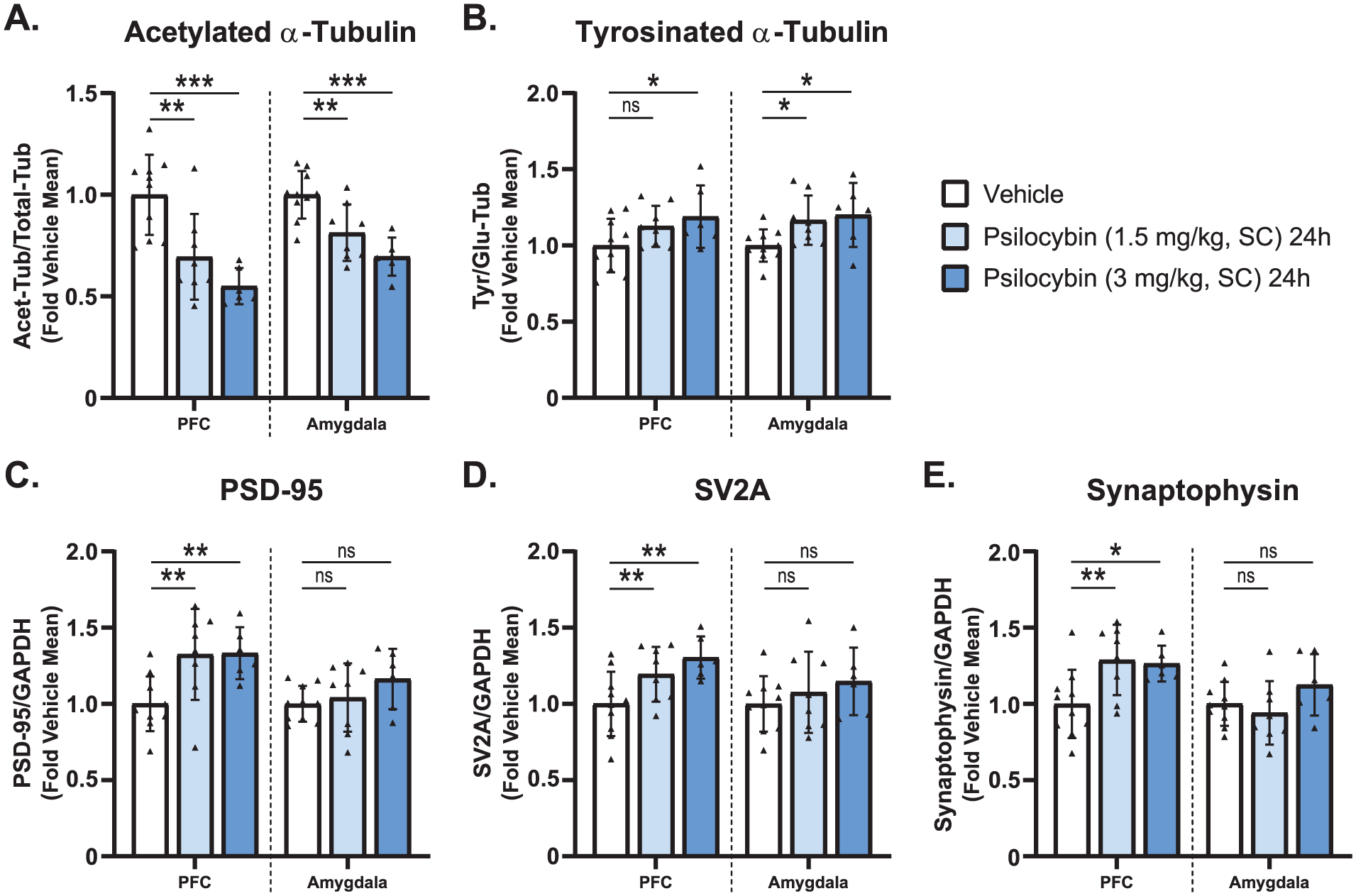
Psilocybin increases the expression of cytoskeletal and synaptic plasticity markers in the PFC and amygdala.
Psilocybin treatment had a minor effect on increasing the levels of tyrosinated α-tubulin in both the PFC and amygdala (Figure 5(b)), 3 mg/kg psilocybin treatment induced an 18.9% increase in the PFC (p = 0.0444 vs. Vehicle), and a 20% increase in the amygdala (p = 0.0206 vs. Vehicle), while the 1.5 mg/kg dose induced a 16.5% increase in the amygdala (p = 0.0353 vs. Vehicle). No statistically significant differences were observed for the effects of psilocybin treatment on the levels of tyrosinated α-tubulin between the PFC and amygdala (F(1,42) = 0.1220, p = 0.7286) (Figure 5(b)).
These data indicate an overall shift to a state of increased microtubule dynamics in both the PFC and amygdala upon treatment with both doses of psilocybin when compared to vehicle-treated controls.
Psilocybin increases synaptic marker expression in the PFC
PSD-95 expression was significantly increased in the PFC upon treatment with both 1.5 and 3 mg/kg (SC) psilocybin (Figure 5(c), Left), with a 32.4% and 33.3% increase, respectively, compared to vehicle-treated controls (1.5 mg/kg; p = 0.006, 3 mg/kg; p = 0.009). In the amygdala, no significant changes were observed for PSD-95 expression upon treatment with 1.5 or 3 mg/kg psilocybin compared to vehicle-treated controls. Psilocybin treatment showed a statistically greater effect in increasing the expression of PSD-95 in the PFC when compared to the amygdala for both 1.5 and 3 mg/kg doses (Row Factor: F(1,42) = 6.361, p = 0.0155) (Figure 5(c)).
SV2A expression increased in a dose-dependent manner after administration of 1.5 or 3 mg/kg (SC) psilocybin in the PFC (Figure 5(d), Left), with SV2A expression increased by 19.4% and 30.4%, respectively, compared to vehicle-treated controls (1.5 mg/kg; p = 0.0391, 3 mg/kg; p = 0.0046). In the amygdala, no significant changes were observed for SV2A expression upon treatment with 1.5 or 3 mg/kg psilocybin compared to vehicle-treated controls.
Synaptophysin expression was significantly increased in the PFC after treatment with either 1.5 or 3 mg/kg (SC) psilocybin (Figure 5(e), Left), with 28.8% and 26.4% increases, respectively, compared to vehicle-treated controls (1.5 mg/kg; p = 0.0077, 3 mg/kg; p = 0.0215). In the amygdala, no significant changes were observed for synaptophysin expression upon treatment with 1.5 or 3 mg/kg psilocybin compared to vehicle-treated controls. Psilocybin treatment showed a statistically greater effect in increasing the expression of synaptophysin in the PFC when compared to the amygdala for both 1.5 and 3 mg/kg doses (Row Factor: F(1,42) = 8.030, p = 0.0070) (Figure 5(e)).
These data indicate an overall shift to a state of increased synaptic plasticity upon treatment with both doses of psilocybin when compared to vehicle-treated controls.
Discussion
Measurement of in vivo receptor occupancy in mice showed that administration of psilocybin produced dose-dependent occupancy of 5-HT2A receptors (presumably by its active metabolite psilocin) in the mouse frontal cortex with an ED50 of 0.88 mg/kg. Psilocybin produced the expected ‘inverted-U’-shaped dose–response curve in HTR, with an ED50 of 0.15 mg/kg and the peak response occurring at 1 mg/kg. This is nearly identical to the published ED50 value of 0.17 mg/kg for psilocin (Klein et al., 2021) and consistent with other studies of psilocybin/psilocin in the HTR (Erkizia-Santamaría et al., 2022; Halberstadt et al., 2011; Lu et al., 2025; Sherwood et al., 2020). One key finding is that doses on either side of the HTR peak produce a similar total number of head twitches over 20 minutes but are clearly differentiated in terms of peak rate. These results show the importance of running the HTR assay across a range of doses and considering the temporal dynamics of the HTR in addition to the total count.
The peak of the total HTR dose–response curve occurred at 1 mg/kg, a dose which corresponds to 44% RO, while the peak rate occurred at a dose of 3.2 mg/kg, which corresponds to 62% occupancy. When each of these measures of the HTR was correlated with in vivo 5-HT2A receptor occupancy, the peak response rate showed a stronger correlation than the total count (R2 = 0.5220 and 0.143, respectively). Madsen et al. (2019) performed a human PET study showing that doses of psilocybin which resulted in >60% RO produced maximal subjective intensity. This clinical finding is consistent with the peak HTR in mice occurring at a dose that achieved ~60% RO in the PFC. Interestingly, psilocybin did not fully displace the radiotracer in the current study or the human PET study, suggesting a small proportion of non-displaceable 5-HT2A binding. Despite not demonstrating 100% receptor occupancy in either human or rodent studies, the data are sufficient to determine an ED50, particularly given the knowledge that zero drug exposure results in 0% displacement of radioligand, and it is impossible to achieve >100% displacement of radioligand binding. Overall, these data suggest that, compared to the total HTR count, the peak HTR rate may be a better proxy for in vivo 5-HT2A activation and more representative of the intensity of psychedelic effects in humans.
It was also apparent that doses >1 mg/kg of psilocybin produced suppression of locomotor activity, which notably corresponded in time with the decline in the rate of the HTR. This suggests that there may be a common mechanism responsible for both the descending arm of the HTR dose–response curve and the locomotor suppression produced by high doses of psilocybin or other psychedelics (Halberstadt et al., 2011; Halberstadt and Geyer, 2016, 2011). However, additional research is needed to understand the relationship between suppression of locomotor activity and suppression of the HTR seen at doses above those which produce peak responding. It is important to note that other studies have demonstrated that the suppression of head twitch is not secondary to the suppression of locomotor activity, as GABAA PAMs like diazepam will reduce locomotor activity without inhibiting the HTR to DOI (Miyata et al., 2006; Wettstein et al., 1999). This behavioral suppression may confound interpretation of the data, particularly at the higher end of the dose range. Thus, we decided to present both the total count of HTRs over 20 minutes, as well as the peak rate of responding.
As described earlier, doses of psilocybin that achieve >50% RO produce profound psychedelic effects in healthy subjects (Madsen et al., 2019). Robust and sustained antidepressant efficacy of an oral dose of 25 mg psilocybin (Goodwin et al., 2022; Raison et al., 2023) corresponds to a dose that achieves ~69% RO in humans (Madsen et al., 2019). Interestingly, a 10 mg dose of psilocybin, which achieves <50% RO, was not associated with such robust or durable efficacy (Goodwin et al., 2022). Similarly, doses of MM120 (a form of LSD), which produce robust subjective effects in healthy volunteers, have also been shown to be efficacious in generalized anxiety disorder (Jacobsen et al., 2024). As such, there is now a better understanding of the relationship between 5-HT2A RO and both subjective and therapeutic effects. We have attempted to build a parallel understanding using mouse models to explore the relationship between RO and HTR, and behavior in the EZM/FST. Overall, the relationship demonstrated in mice was consistent with that established in humans.
Both ketamine and psilocybin have been shown to have rapid and durable efficacy in various naturalistic and pharmacological assays of antidepressant-like activity in rodents, for instance, ketamine has shown efficacy in chronic mild stress (Klein et al., 2024), chronic social defeat stress (Zhang et al., 2014), and the interferon-alpha assay of depressive-like behavior (Clark et al., 2025). Similarly, psilocybin has been shown to selectively attenuate a negative affective bias in the affective bias test and reverse chronic stress-induced deficits in motivation in mice 24 hours after a single dose (Hesselgrave et al., 2021; Hinchcliffe et al., 2024).
Hibicke et al. (2020) showed that both LSD and psilocybin produced persistent effects of reducing immobility time in the mouse FST lasting for at least 35 days, while the beneficial effect of ketamine only lasted ~2 weeks. In a study by Wang et al. (2025), two doses of 1 mg/kg psilocybin were efficacious at blocking the effect of stress when tested 2 weeks after the second dose. By contrast, Jefsen et al. (2019) did not find any effect of either psilocybin or psilocin 3 hours after drug administration; however, this could be due to the short interval between dosing and testing, or the relatively high doses of psilocybin/psilocin. In a study by Takaba et al. (2023), psilocin showed an inverted-U shaped dose–response curve in the FST 24 hours after dosing, suggesting there is an optimal dose to reduce immobility time in the FST. These studies highlight the difficulty of modeling both the rapid onset of pharmacodynamic effects associated with antidepressant-like efficacy of psilocybin (or ketamine), as well as the durability of those effects. While the EZM and FST employed in this study are often referred to as models of anxiolytic- and antidepressant-like effects, we recognize the limitations of these assays and consider them as simple pharmacodynamic readouts which are sensitive to the effects of psilocybin at a time when both the drug and its active metabolite psilocin have been cleared. Debating their relevance to human depression or anxiety is well beyond the scope of this manuscript.
Further to the analysis of the relationship between RO and HTR, our findings here also demonstrate that subcutaneous administration of psilocybin induces a significant shift in α-tubulin PTMs toward a more dynamic microtubule state, particularly in the PFC. This is characterized by a dose-dependent reduction in acetylated α-tubulin levels and increases in tyrosinated α-tubulin. The observed decrease in N-terminal acetylated α-tubulin, mediated by deacetylases such as Histone Deacetylase 6 and Sirtuin 2 (Hubbert et al., 2002; North et al., 2003), as well as increase in C-terminal tyrosination of α-tubulin, mediated by Tubulin Tyrosine ligase (Sanyal et al., 2023), suggest enhanced microtubule plasticity, consistent with prior studies showing that deacetylation and re-tyrosination of α-tubulin promote microtubule structural dynamicity necessary for neurite extension and synaptic remodeling (Carmona et al., 2023; Peris et al., 2009; Schätzle et al., 2018). Decreased levels of acetylated α-tubulin and/or increased levels of tyrosinated α-tubulin have been previously observed in the hippocampus and medial PFC of animals chronically treated with clinically effective antidepressant drugs such as fluoxetine or agomelatine. These changes were noted both under baseline (naïve) conditions and following exposure to experimental models of depression, where α-tubulin PTMs were shifted toward a less dynamic cytoskeletal state (Bianchi et al., 2006, 2009a, 2009b; Ladurelle et al., 2012; Yang et al., 2009, 2024). Neurosteroids such as PME, which specifically target neuronal microtubule-associated proteins (MAPs) and exhibit no affinity for known CNS receptors, have been shown to reverse these tubulin alterations. PME restores increased acetylated α-tubulin and reduces tyrosinated α-tubulin levels, while also rescuing depression-like behaviors in animal models such as social isolation in rats and psychosocial stress in tree shrews (Bianchi and Baulieu, 2012; Parésys et al., 2016). Moreover, we have recently provided the first clinical evidence that circulating plasma levels of acetylated α-tubulin are altered in healthy volunteers acutely treated with an antidepressant dose of ketamine (0.5 mg/kg, IV), with these changes correlating with altered glutamate metabolism in the pgACC (Colic et al., 2019). Taken together, these findings support the hypothesis of a potential role of microtubule dynamics in the promotion of neuronal plasticity, potentially associated with antidepressant efficacy (Bianchi et al., 2005). Thus, several lines of preclinical and clinical evidence underline a potential role of neuronal plasticity in the pathogenesis and treatment of depression (Liu et al., 2017; Wang et al., 2023).
The exact mechanism by which psilocybin induces these cytoskeletal modifications is not clear; however, the observed shifts in α-tubulin PTMs likely reflect upstream regulation via 5-HT2A-activated intracellular signaling cascades. Psilocybin is known to activate multiple pathways downstream of 5-HT2A receptor engagement, including the mTOR and PLC/IP3/PKC pathways, both of which are critical modulators of cytoskeletal and synaptic plasticity, as well as phosphorylation status of MAPs (Costa-Mattioli and Monteggia, 2013; González-Maeso et al., 2007).
GSK3β, a downstream effector of mTOR, regulates the phosphorylation and stability of MAPs through phospho-regulation and modulates MAP association and dissociation (Goold et al., 1999; Liz et al., 2014; Zumbrunn et al., 2001). This, in turn, contributes to microtubule stability and accessibility, with MAP dissociation leading to increased remodeling by generating more free tubulin substrate for enzymatic action (Beurel et al., 2015).
In addition, a direct role of 5-HT2A receptor in promoting microtubule dynamics has been experimentally demonstrated in vitro. In cultured rat cortical neurons, Ohtani et al. (2014) showed that activation of 5-HT2A receptors with the agonist DOI significantly increased tyrosinated α-tubulin and decreased acetylated α-tubulin specifically in dendritic growth cones. This shift toward a more dynamic microtubule state was accompanied by increased morphological plasticity of dendritic growth cones and was reversed by a 5-HT2A receptor antagonist, indicating receptor-specific effects. These findings provide compelling evidence that serotonergic modulation via 5-HT2A directly regulates the cytoskeletal machinery at the level of α-tubulin PTMs.
These observations suggest that psilocybin may exert its structural plasticity-promoting effects via a combination of upstream signaling cascades and direct modulation of microtubule dynamics through 5-HT2A receptor engagement. There is also a potential, yet to be fully elucidated, for direct interaction of psychedelic compounds with microtubule structures themselves, potentially influencing the polymerization state independently of or in synergy with receptor-mediated signaling (De Abreu et al., 2023).
Shifts in α-tubulin PTMs occurred in both the PFC and amygdala, with a statistically greater magnitude observed for acetylated α-tubulin in the PFC compared to the amygdala (F(1,42) = 4.131, p = 0.0485). This suggestion of region-specific increase in plasticity aligns with clinical and preclinical models, suggesting that increased neuroplasticity in the PFC is associated with antidepressant efficacy, while suppression of plasticity in the amygdala may be required for anxiolytic effects (Fogaça and Duman, 2019; Otte et al., 2016).
Psilocybin administration at both doses investigated also significantly elevated the expression of key pre- and post-synaptic markers (PSD-95, SV2A, and synaptophysin) in the PFC. These findings support previous work showing that serotonergic psychedelics such as LSD, DMT, and psilocybin increase dendritic spine density and synaptogenesis through 5-HT2A receptor-dependent activation of intracellular signaling cascades (Ly et al., 2018; Raval et al., 2021; Shao et al., 2021). Notably, no significant increases in expression of synaptic markers were detected in the amygdala despite the observed increase in microtubule dynamics, suggesting that dynamic remodeling in this region may remain below the threshold for structural synaptic changes, potentially serving as an intrinsic safety mechanism and preventing anxiogenic synaptic remodeling in the amygdala. These region-specific effects of psilocybin on α-tubulin PTMs and synaptic protein expression may have important implications for its psychotropic and therapeutic actions. We have planned future studies aimed to further explore the effects of psilocybin on α-tubulin PTMs and synaptic markers in animal models of depression and anxiety.
The regional pattern of plasticity observed may provide a molecular context when interpreting the behavioral results obtained. Psilocybin at 1.5 mg/kg significantly increased exploration in the EZM, which can be interpreted as an anxiolytic-like behavior. However, the 3 mg/kg dose, which induced stronger microtubular and synaptic plasticity in the PFC, did not increase exploration in the EZM but instead produced a robust reduction in immobility time in the FST. This suggests that moderate microtubule remodeling in the amygdala, without inducing synaptic restructuring, may be sufficient for anxiolytic-like effects, while the more profound plasticity in the PFC seen at a higher dose supports effects in the FST in mice. Importantly, the absence of synaptic upregulation in the amygdala, even at 3 mg/kg, might be a protective mechanism in preventing anxiogenic side effects, consistent with clinical reports of low anxiety induction following psilocybin treatment (Carhart-Harris et al., 2018).
These findings highlight that the durability of the therapeutic efficacy of psilocybin may arise from a dose- and region-specific modulation of microtubule dynamics and neuroplasticity in the PFC. At 1.5 mg/kg psilocybin enhanced microtubule dynamics in the amygdala without increasing synaptic protein expression, consistent with the anxiolytic-like effects seen in the EZM, while a 3 mg/kg dose promoted synaptic and cytoskeletal remodeling in the PFC, aligned with the sustained reduction of immobility observed in the FST, as well as antidepressant outcomes observed in clinical studies (Goodwin et al., 2022; Raison et al., 2023). Given the role of the amygdala in fear processing and memory of negative emotional states, suppression of plasticity in this region likely contributes to the anxiolytic-like effects observed (Janak and Tye, 2015). Alternatively, the regional expression pattern of 5-HT2A receptors may also contribute to the differences seen in the amygdala versus the PFC, whereby the robust effects in microtubule and synaptic protein plasticity observed in the PFC are underpinned by a higher expression of 5-HT2A receptors in the PFC (Andrade and Weber, 2010; Saulin et al., 2012). This differential molecular profile may define an optimal therapeutic window for achieving distinct clinical benefits.
One of the limitations of the current dataset is that we did not assess the effects of doses of psilocybin achieving <50% RO on behavioral (EZM and FST) or molecular endpoints to understand the minimum level of 5-HT2A RO required to see these effects. Additional studies are being conducted to examine the effects of psychedelics on behavior and biochemistry in models incorporating a perturbation (e.g., stress or immunological challenges) in an attempt to increase construct/face validity and understand whether any of the induced phenotypes can be reversed by 5-HT2A receptor agonists. An additional limitation of the current work is that while it was possible to determine an ED50 for in vivo binding, such a binding assay is unable to determine the level of receptor reserve in vivo. The literature indicates that the level of HTR produced by agonists of 5-HT2A receptors varies according to their degree of intrinsic efficacy in vitro (Wallach et al., 2023), suggesting that the level of receptor reserve in the circuit involved in the HTR is low. Further experiments are required to demonstrate this definitively.
Finally, in this study, we restricted psilocybin testing to male mice to minimize biological variability and focus on core molecular and behavioral mechanisms. Although preclinical data suggest that hormonal differences can influence serotonergic signaling and 5-HT2A receptor function (Shadani et al., 2024) and that psilocin may elicit sex-specific neural responses in limbic circuits such as the central amygdala (Effinger et al., 2023), these differences have to date not translated into measurable sex effects in human clinical studies. Major randomized controlled trials of psilocybin for treatment-resistant or MDD (Goodwin et al., 2022; Davis et al., 2020; Carhart-Harris et al., 2021; Raison et al., 2023) and subsequent meta-analyses (Swieczkowski et al., 2025) report robust antidepressant efficacy without evidence of sex-related differences in treatment response. As additional clinical data become available, it will be important to carefully review whether there are any differences in the effects of psilocybin in males and females.
In conclusion, we have explored the relationship between 5-HT2A RO, HTR, and behavior in the EZM and FST in mice and shown that this was consistent with that established in humans. While EZM and FST are often referred to as models of anxiolytic- and antidepressant-like effects, we recognize the limitations of these assays and consider them as pharmacodynamic assays that are sensitive to the effects of psilocybin at a time when the drug has been cleared. Our findings demonstrate that doses of psilocybin that achieve similar 5-HT2A RO as clinical doses are associated with robust and durable antidepressant efficacy, promote a state of increased neuronal plasticity by enhancing microtubule dynamics and selectively increasing synaptic protein expression in the PFC of mice. These changes may provide a mechanistic basis for the durability of the therapeutic effects of psilocybin.
Footnotes
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All experiments were funded by Ulysses Neuroscience or Gilgamesh Pharmaceuticals.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available on request.
Animal Welfare Declarations
All animals were cared for in strict accordance with the Guide for the Care and Use of Laboratory Animals (NIH Publication No. 85-23, revised 1996), and the protocols for animal experiments were approved by the URI IACAUC (Approval # AN2324-001) on September 14, 2023. Animal care and procedures were conducted in accordance with European Communities Council Directive (86/609/EEC), and the experimental protocols were approved by the animal ethical committee of Trinity College, Dublin.
Behavioral Experimentation Justification
In this study, the EZM and FST were conducted sequentially in the same animals, with a 4-hour recovery period between tests to reduce cumulative stress. This design was chosen to minimize animal use while allowing investigation of behaviors associated with putative anxiolytic and antidepressant-like effects. Both assays are well-validated and widely used to assess core affective domains relevant to human psychiatric conditions. While the use of EZM and FST as assays to predict human anxiolytic or antidepressant efficacy for novel mechanisms has not always been successful (e.g., mGluR5 NAMs, CRF1 antagonists) (Barnes et al., 2018; Koob and Zorrilla, 2012; Valdez, 2006) here these assays are used to demonstrate pharmacodynamic effects shared by known antidepressant and anxiolytic drugs at a time when psilocybin is no longer present. The procedures involved are mild (EZM) or moderate (FST), and animals only briefly experienced stress, which was essential to meet the experimental objectives.

