Abstract
Objective:
(-)-OSU6162 is an antagonist at dopaminergic D2 receptors which – like other D2 antagonists – dampens spontaneous activity in animals exploring a novel environment. However, unlike other D2 antagonists, (-)-OSU6162 has unexpectedly been found to stimulate locomotor activity in inactive, habituated rodents. To what extent the compound may increase activity also in other situations characterised by reduced locomotion is unknown, and it has also not been clarified whether two other receptors also targeted by (-)-OSU6162 – serotonergic 5-HT2A receptors and sigma-1 receptors – are involved in its unusual behavioural profile. The objective of the present study was to investigate the possible impact of (-)-OSU6162 on the pronounced inactivity in the form of freezing displayed by rats exposed to context-conditioned fear. For comparison, the effect of amphetamine – another dopamine-augmenting and activating compound – was also explored in the same paradigm.
Methods:
The impact of (-)-OSU6162 and amphetamine on the expression of freezing was assessed in rats exposed to contextual fear conditioning using electrical foot shocks. It was also assessed whether the increase in activity observed in animals treated with (-)-OSU6162 could be countered by pretreatment with a D2/D3 antagonist (raclopride), a 5-HT2A inverse agonist (MDL100907), or a sigma-1 receptor antagonist (BD1063).
Results:
While (-)-OSU6162 markedly reduced freezing behaviour, amphetamine abolished it completely. The effect of (-)-OSU6162 was countered by raclopride but neither by MDL100907 nor by BD1063.
Conclusion:
(-)-OSU6162 reduces the expression of context-conditioned fear displayed as freezing by a mechanism involving D2 but not 5-HT2A or sigma-1 receptors.
Introduction
The so-called dopamine stabiliser (-)-OSU6162 has been shown to modulate rodent activity in an atypical manner (Rung et al., 2008). In line with being an antagonist at dopaminergic D2 receptors (Natesan et al., 2006), it causes inhibition of locomotor activity in active animals exploring a novel environment or treated with amphetamine. Unlike other D2 antagonists, however, (-)-OSU6162 does not elicit catalepsy even at high doses in rodents (Natesan et al., 2006) and does not cause extrapyramidal side effects in humans (Nilsson et al., 2020; Tolboom et al., 2015). Also, unlike other D2 antagonists, (-)-OSU6162 was unexpectedly shown to cause locomotor stimulation in animals habituated to a testing arena (Carlsson et al., 2011; Natesan et al., 2006). Striatal D2 receptor occupancy displayed by (-)-OSU6162 appears relatively high in rodents (Natesan et al., 2006) and non-human primates (Ekesbo et al., 1999) but more modest in humans (Tolboom et al., 2015).
In addition to being a D2 receptor ligand, (-)-OSU6162 also displays affinity to serotonergic 5-HT2A and 5-HT2B receptors (acting as partial agonist) (Carlsson et al., 2011), high affinity to the sigma-1 receptor (Sahlholm et al., 2013), and relatively low affinity to dopaminergic D3 receptors and serotonergic 5HT1A and 5HT1D receptors, respectively (Burstein et al., 2011). In contrast, it has little or no affinity to adrenergic, histaminergic, or other dopaminergic receptors, and no affinity to the noradrenaline, dopamine, or serotonin transporters (Burstein et al., 2011).
Given the involvement of dopamine in numerous psychiatric and neurological conditions, the possible impact on spontaneous activity of a compound displaying this highly unusual pharmacological profile deserves further attention, especially since (-)-OSU6162 is entirely devoid of the side effects marring D2 receptor antagonists used for psychosis, hyperprolactinemia being the sole exception (Nilsson et al., 2020). Likewise, it lacks the side effects of central stimulants and appears not to display abuse liabilities (Asth et al., 2021).
Rats re-introduced to a neutral context in which they have previously been exposed to an aversive stimulus in the form of foot shocks display context-conditioned fear expressed as freezing,
Material and methods
Animals
A total of 243 male Sprague Dawley rats (Janvier, Le Genest-Saint-Isle, France) were used in this study. Animals were 8–9 weeks upon arrival and housed in groups of 3 per cage. They were maintained on a 12-hour light/dark cycle, had access to food and water
Drugs
(-)-OSU6162 was gifted from Arvid Carlsson Research AB (Gothenburg, Sweden), S(-)-raclopride (+)-tartrate salt, and D-amphetamine sulfate were purchased from Merck KGaA (Darmstadt, Germany), and MDL100907 and BD1063 dihydrochloride were purchased from Tocris (Bristol, UK). All drugs were dissolved in 0.9% saline on the day of the experiment and given subcutaneously in volumes of 2 ml/kg with the exception of MDL100907, which was dissolved in a few drops of hydrochloric acid, made up to volume with 0.9% saline, and buffered to pH > 5.5 with sodium hydroxide. BD1063 was given at a dose of 15 mg/kg which was selected based on pilot experiments following a survey of the relevant literature; for example, BD1063 at a dose range of 3–30 mg/kg has been shown to reduce alcohol-seeking behaviour (Blasio et al., 2015; Sabino et al., 2009), reduce binge-like eating (Cottone et al., 2012), and to counteract MDMA-induced (but not spontaneous) motor activity (Brammer et al., 2006), while lower doses of 7.5–10 mg/kg have shown to specifically counteract sigma-1 receptor induced increase of substance intake (Hiranita et al., 2010; Sabino et al., 2011). MDL100907 was given at a dose of 1 mg/kg, which is a dose that in our laboratory has been shown to effectively antagonise 5-HT2A receptor agonist-induced reduction of contextual fear conditioning (Hagsater et al., 2021) and which, according to numerous previous studies, should be sufficient to block other 5-HT2A-mediated effects (Carbonaro et al., 2015). The dose of raclopride was chosen following a pilot experiment investigating dose-response effects on (-)-OSU6162-induced and spontaneous locomotion (see section “Discussion”).
Contextual fear conditioning
The contextual fear conditioning experiments were performed using the contextual near-infrared Video Fear Conditioning System for Rat (Med Associates, Sant Albans, VT, USA). The conditioning chamber was enclosed by a sound-attenuating cubicle in which a constant 60-dB white background noise was delivered during all experimental sessions. In the fear acqusition session, rats were allowed to habituate for five minutes prior to receiving five electric foot shocks (0.6 mA, 30 seconds inter-shock interval). Seven days later, the animals were administered (-)-OSU6162, amphetamine, or saline 40 minutes before they were re-exposed to the chamber for five minutes during which contextual fear-conditioned freezing was assessed. In other experiments, antagonists at D2, 5-HT2A, or sigma-1 receptors, or saline were administered 10 minutes prior to (-)-OSU6162 or saline. Freezing was calculated by automatic scoring of video recordings (immoblity > 1 second) and presented as the % time the animal spent immobile during the five-minute test (Hagsater et al., 2019). Normally, animals subjected to a novel cage display considerable exploratory activity – in the present study, gross observation (though without quantification) was applied to confirm that immobile animals displayed typical signs of freezing, that is, rigged tail, arched back, erected fur, and retracted ears, rather than any unusual form of inactivity not pertaining to the typical freezing characteristics. In a control experiment aimed at addressing the possible impact of (-)-OSU6162 and raclopride, respectively, on spontaneous activity in animals not displaying freezing, rats were exposed twice to the same context as above but without receiving foot shocks. Faeces trays were always emptied, and the wire mesh cage was cleaned using 70% ethanol before every new test session.
Locomotor activity
In order to assess if the notably low dose of raclopride (0.03 mg/kg) found to counter the effect of (-)-OSU6162 in the freezing paradigm could also prevent the locomotor stimulation elicited by (-)-OSU6162 in habituated animals, rats were placed in the centre of ventilated and sound-attenuated motron boxes illuminated by a dim light (40 × 40 cm, Med Associates Inc., Fairfax, VT, USA) and allowed to habituate for 30 minutes before being administered raclopride followed 10 minutes later by (-)-OSU6162 after which the motor activity of the animals was assessed for 30 minutes. Movement was registered as ambulatory counts caused by crossing a square pattern of infrared beams scattered across the floor.
Statistical analysis
Data were analysed, and graphs were created using GraphPad Prism version 10.2.3 (Boston, MA, USA) and Microsoft Excel version 16.97 (Redmond, WA, USA). Assessment of normality was undertaken using the Shapiro-Wilk test. Comparisons between groups were performed with one-way analysis of variance (ANOVA) when normally distributed, or the Kruskal-Wallis test when not, followed by the Benjamini-Hochberg procedure. The significance threshold was set to 0.05. For all ANOVAs, homogeneity of variance was estimated using Levene’s test. Normally distributed data are displayed as means + standard errors of the mean (SEM), while data not normally distributed are displayed as medians + interquartile ranges (IQR).
Results
(-)-OSU6162 at doses of 10 and 30 mg/kg – but not at doses of 1 or 3 mg/kg – reduced the expression of contextual fear-conditioned freezing (H(4) = 16.23, η² = 0.284,
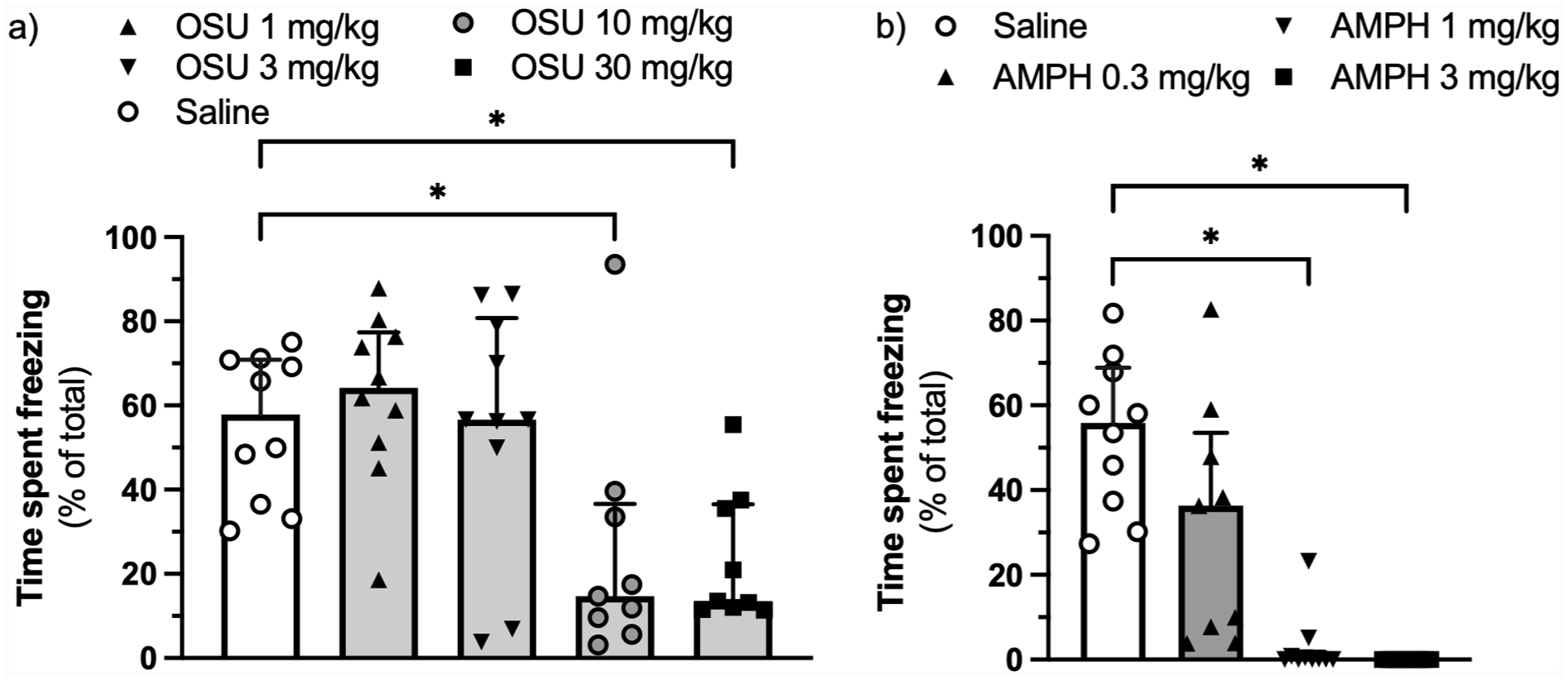
Effect of (-)-OSU6162 (a) and AMPH (b) on the expression of context-conditioned fear assessed as percentage of time spent freezing.
The D2/D3 antagonist raclopride (0.03 mg/kg) (F(2,33) = 6.793, ηp² = 0.292,
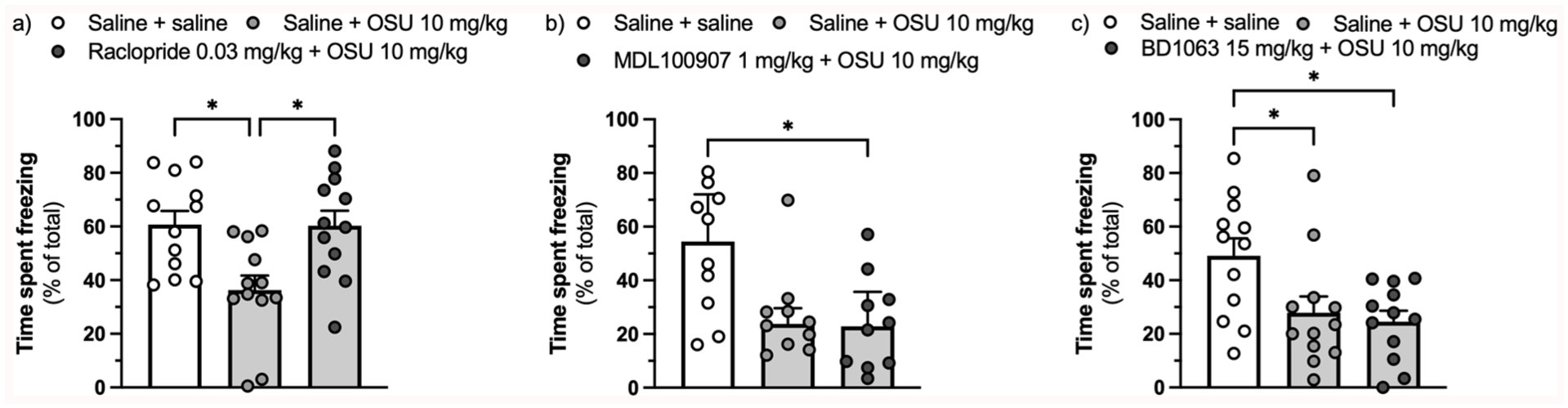
Effect of pretreatment with the D2/D3 receptor antagonist raclopride (0.03 mg/kg) (a), the 5-HT2A receptor inverse agonist MDL100907 (1 mg/kg) (b), and the sigma-1 receptor antagonist BD1063 (15 mg/kg) (c) on the reduction in the expression of context-conditioned fear assessed as percentage of time spent freezing caused by (-)-OSU6162 (10 mg/kg).
The same (low) dose of raclopride (0.03 mg/kg) that was found to antagonise the (-)-OSU6162-induced reduction in freezing also countered the stimulatory effect of (-)-OSU6162 in rats habituated to a locomotor activity arena (F(2,15) = 4.583, ηp² = 0.379,
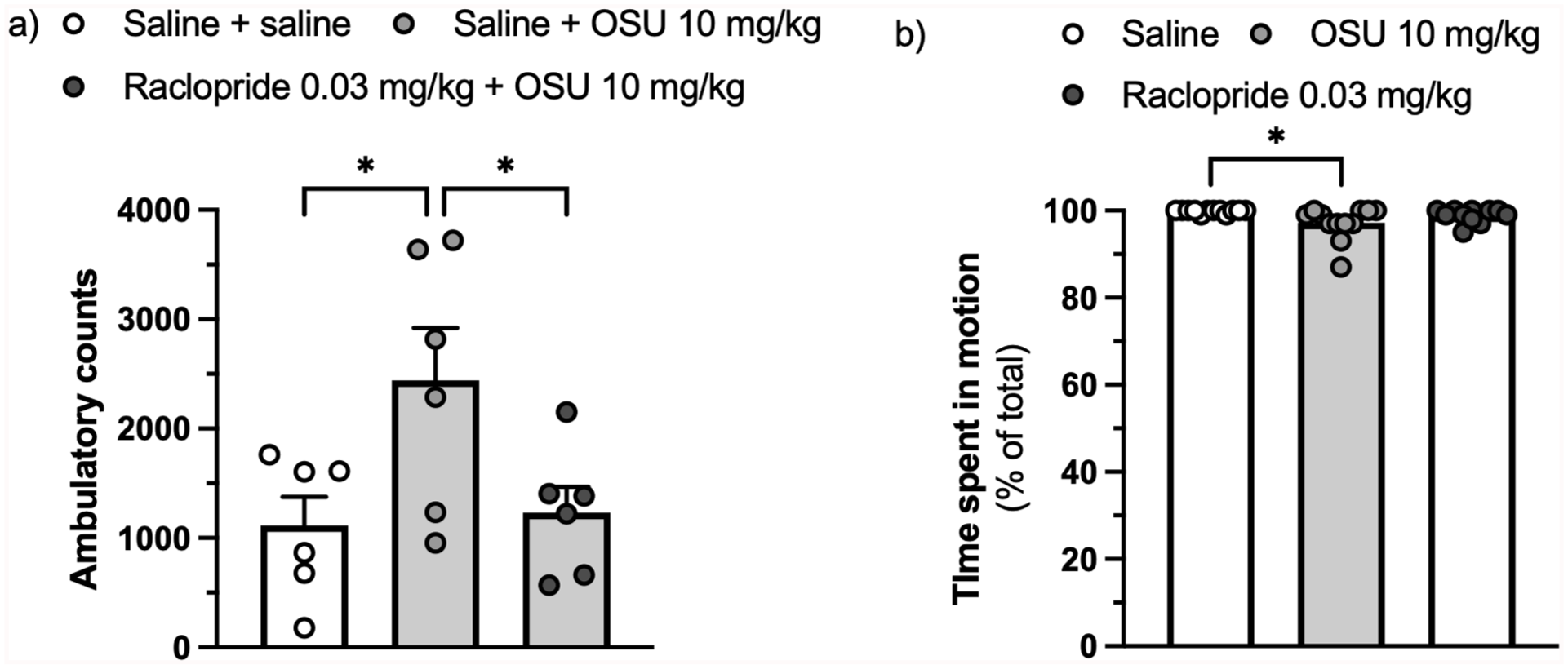
Effect of the D2/D3 receptor antagonist raclopride (0.03 mg/kg) on the increase in locomotion (ambulatory counts) caused by (-)-OSU6162 (10 mg/kg) in rats habituated to a locomotor activity arena.
Discussion
While amphetamine usually stimulates locomotion in rodents, regardless of their baseline activity, the impact of (-)-OSU6162 is more complex. Thus, while the drug dampens locomotion in active, exploring animals – as would be expected from a D2 receptor antagonist – it has been found to stimulate locomotion in habituated, non-active animals (Carlsson et al., 2011). While an activating impact of (-)-OSU6162 has previously been demonstrated in this situation only, we now report that a robust stimulatory effect of (-)-OSU6162 is at hand also in rats displaying a lack of motion when being exposed to context-conditioned fear. The activating effect of (-)-OSU6162 was blocked by a low dose of the selective D2/D3 antagonist raclopride but neither by a 5-HT2A antagonist nor by a sigma-1 receptor antagonist.
The activating effect of (-)-OSU6162 in rats displaying freezing at the second exposure to the conditioning chamber was not merely a reflection of the previously reported increase in activity in rats habituated to their chamber. Thus, animals exposed to the same conditioning chamber twice, but without having experienced foot shocks at their first visit, displayed no signs of habituation when re-introduced to the chamber, and their activity was dampened rather than stimulated by (-)-OSU6162.
While it cannot be excluded that the freezing-reducing effect of (-)-OSU6162 is caused by impaired memory retrieval, this seems unlikely since the drug has been attributed no negative impact on memory, but – on the contrary – been shown to prolong object location memory as well as to reverse memory impairments following scopolamine administration in mice (Nilsson and Carlsson, 2013). Likewise, (-)-OSU6162 has been reported not to impair cognitive function in humans (Khemiri et al., 2020).
The dose of the selective D2/D3 receptor antagonist raclopride presently shown to prevent the activating effect of (-)-OSU6162 has previously been reported too low to impact motor activity
The anti-freezing effect of (-)-OSU6162 being effectively countered by a low dose of raclopride suggests an involvement of D2/D3 receptors. A possible explanation to why the stimulatory effect of a D2/D3 antagonist, (-)-OSU6162, is antagonised by another D2/D3 antagonist, raclopride, might be that (-)-OSU6162 increases extracellular levels of dopamine by preferentially blocking autoreceptors, hence causing an indirect stimulation of postsynaptic locomotion-stimulating D2/D3 receptors not blocked by (-)-OSU6162 but by a low dose of raclopride. In line with this possibility, in vivo microdialysis experiments have revealed (-)-OSU6162 to increase extracellular levels of dopamine in the rat striatum (Steensland et al., 2012). Needless to say, this mechanism could be relevant also for the stimulatory effect of (-)-OSU6162 in habituated rodents.
It was to address this possibility we compared the effect of (-)-OSU6162 to that of an indirect D2 receptor agonist, amphetamine, using the same paradigm and the same setting. In line with the possibility of (-)-OSU6162 reducing freezing by increasing extracellular levels of dopamine, the central stimulant also reduced freezing but to an even greater extent. Thus, while the maximum reduction in the expression of freezing after (-)-OSU6162 was approximately 85%, high doses of amphetamine eliminated the freezing reaction entirely, and even rendered the animals hyperactive, as judged by gross observation. Such a discrepancy is not counterintuitive since (-)-OSU6162 and amphetamine are likely to elicit an increase in extracellular dopamine levels by different mechanisms: autoreceptor blockade and reversal of the direction of the dopamine transporter, respectively. Likewise, it is compatible with the notion of (-)-OSU6162 acting as a dopamine
While the observed antagonism between (-)-OSU6162 and raclopride may thus tentatively be explained by (-)-OSU6162 enhancing extracellular levels of dopamine by blocking D2 autoreceptors, a different possibility should also be considered. There are hence some indications that (-)-OSU6162 – instead of being merely a silent antagonist at the orthostatic site of the D2 receptor – may also act as a positive allosteric modulator at D2 receptors (and/or to exert limited intrinsic activity upon the same); until now there is, however, only
Apart from its impact on D2 receptors, (-)-OSU6162 is also a partial agonist at 5-HT2A receptors. We have previously, using the same experimental setting, found two 5-HT2A receptor agonists – psilocybin and 25CN-NBOH – to reduce the expression of conditioned fear, and shown these effects to be effectively blocked by the 5-HT2A antagonist/inverse agonist MDL100907 (Hagsater et al., 2021). Also, it has been shown that MDL100907 blocks the (-)-OSU6162-induced stimulation of locomotion elicited in mice rendered monoamine-depleted by means of pretreatment with reserpine, a response that could not be blocked by D2 antagonism (Carlsson et al., 2011). It was therefore deemed important to explore the possible impact of MDL100907 at a dose effectively blocking 5-HT2A receptors (Carlsson et al., 2011; Hagsater et al., 2021) on the freezing-reducing effect of (-)-OSU6162. The response being the same in animals pretreated with MDL100907 as in those pretreated with saline, however, argues against 5-HT2A receptor activation being involved in the studied effect of (-)-OSU6162.
Previous reports suggest that sigma-1 receptor agonists reduce the expression of freezing (Noda et al., 2000). (-)-OSU6162 displaying high affinity for this receptor (Sahlholm et al., 2013), thus prompted an attempt to block the effect of (-)-OSU6162 on freezing also with a sigma-1 receptor antagonist, BD1063, at a dose previously shown to effectively block the sigma-1 binding site (Brammer et al., 2006). (-)-OSU6162, however, reduced freezing to the same extent also after pretreatment with this sigma-1 antagonist.
While the primary aim of the present study was to shed further light on the enigmatic stimulatory impact of (-)-OSU6162 on locomotor activity in rat, it should be noted that contextual fear conditioning has often been used as a putative animal model of human anxiety (Grillon, 2002; Hagsater et al., 2019; Izumi et al., 1999). The possibility that (-)-OSU6162 reduced freezing in the present experiments by reducing fear, rather than by a non-specific stimulation of locomotor activity, should thus not be ruled out (de Souza Caetano et al., 2013). The compound has not displayed effects similar to those of drugs for anxiety in the elevated plus maze (Studer et al., 2016) but did exert such an effect in anxiety-prone Flinder rats exposed to the novelty suppressed feeding test (Melas et al., 2021).
In conclusion, the results of this study add to the growing body of data suggesting that the D2 receptor antagonist (-)-OSU6162 impacts dopamine transmission in an unusual manner. The drug displaying both similarities and differences when compared to other dopamine-modulating substances, such as D2 receptor antagonists, D2 receptor agonists, and dopamine releasers, warrants further exploration of its possible use for various dopamine-related clinical conditions.
Footnotes
Acknowledgements
We gratefully thank Jenny Steen for expert technical assistance and Fredrik Hieronymus for valuable statistical advice. Ingrid Norrlin and Clara Augustsson are also acknowledged for valuable contributions. Arvid Carlsson Research AB is acknowledged for the provision of (-)-OSU6162.
Ethical considerations
All experiments followed the legislative directive 2010/63/EU of the European Union and were approved under permit nr 2354/20 and 4122/21 by the regional animal ethics committee at the University of Gothenburg in accordance with the guidelines of the Swedish Board of Agriculture. The animals received electric foot shocks of moderate intensity (0.6 mA) every 30 seconds during the training session which lasted for five minutes. In our experience, this is the minimum intensity, frequency, and duration required to elicit a robust context-conditioned fear response. For the advancement of our insight into mechanisms underlying fear and freezing, we find this commonly used approach justified given that the stressor is both moderate and short-lasting.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swedish Research Council (grant number 2015-02515) and the Swedish Brain Foundation (grant number FO2018-0331).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. Eriksson is a board member of the non-profit Arvid Carlsson Foundation which holds an (-)-OSU6162-related patent. The other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available upon request to the corresponding author.
