Abstract
Background:
Neuropeptide Y1 receptor (NPY1R) heteroreceptor complexes with galanin receptor 2 (GALR2) and Tropomyosin receptor kinase B (TrkB) contribute to neuroplasticity and mood regulation.
Aims:
To determine whether transient intracerebroventricular (ICV) knockdown of NPY1R is sufficient to alter hippocampal neurogenesis and depressive-like behaviour.
Methods:
Adult Sprague-Dawley rats received a single ICV injection of Accell Smart-Pool small interfering RNA (siRNA) targeting NPY1R or a scrambled control. NPY1R protein levels, NPY1R-GALR2 and NPY1R-TrkB complexes (in situ proximity ligation), proliferating cell nuclear antigen (PCNA) counts, brain-derived neurotrophic factor (BDNF) expression and forced-swim behaviour were assessed 6, 8 and 10 days post-injection in the ventral hippocampus.
Results:
ICV siRNA significantly reduced NPY1R immunoreactivity (peak at day 8; p<0.05) and lowered NPY1R-GALR2 and NPY1R-TrkB heteroreceptor complexes (p<0.01 and p<0.001, respectively). PCNA-positive cell numbers and BDNF optical density were unchanged at all time points. Forced-swim immobility, climbing and swimming times likewise remained unaltered.
Conclusions:
Transient ICV knockdown effectively disrupts NPY1R heteroreceptor complexes but fails to impact neurogenesis or depressive-like behaviour, indicating compensatory mechanisms that preserve hippocampal plasticity. Within the time window and conditions tested, transient NPY1R knockdown disrupted GALR2/TrkB heteroreceptor complexes without altering neurogenesis or depressive-like behaviour, delineating receptor-complex-level target engagement and motivating studies using sustained and circuit-specific manipulations to assess therapeutic potential.
Keywords
Introduction
Adult hippocampal neurogenesis is a form of neuroplasticity crucial for learning, memory and mood regulation, and its disruption is implicated in affective disorders (Baptista and Andrade, 2018; Boldrini et al., 2013, 2018; Kang et al., 2016; Terreros-Roncal et al., 2023). Beyond monoaminergic systems, neuropeptides have emerged as potent modulators of hippocampal plasticity. Neuropeptide Y (NPY) and galanin (GAL) are abundantly expressed in limbic brain regions—including the dentate gyrus (DG) of the hippocampus—where they influence synaptic function and behaviour (Eliwa et al., 2017; Sahay and Hen, 2007; Santarelli et al., 2003; Thorsell and Mathe, 2017). NPY, a 36–amino acid peptide, has well-documented effects on stress resilience, feeding and cognition, and can bidirectionally modulate emotional and stress responses depending on receptor subtype and context (Kormos and Gaszner, 2013; Reichmann and Holzer, 2016; Zaben and Gray, 2013). Likewise, GAL, a 29/30–amino acid neuropeptide, broadly impacts central nervous system physiology; notably, activation of its receptors can promote trophic effects on hippocampal progenitor cells in vitro, suggesting a role in neurogenesis (Lu et al., 2005; Luo et al., 2019; Yun et al., 2019). These observations raise the possibility that NPY and GAL signalling pathways converge on hippocampal plasticity mechanisms relevant to mood disorders (Juhasz et al., 2014; Sharma et al., 2022).
NPY has been identified as an endogenous antidepressant and neuroprotective factor in the hippocampus. Levels of NPY are reduced in chronic stress and depression models, and postmortem studies show decreased NPY in the hippocampus of patients with major depression (Cohen et al., 2012; Corvino et al., 2014; Sharma et al., 2022). Conversely, virtually all effective antidepressant treatments elevate brain NPY expression, and rodents engineered to overexpress NPY in the hippocampus exhibit reduced depression-like behaviour (Bjornebekk et al., 2006; Husum et al., 2000; Thorsell et al., 2000). Importantly, NPY exerts pro-neurogenic effects in the adult hippocampal niche: NPY stimulates the proliferation of neural progenitors both in vitro and in vivo and can increase hippocampal brain-derived neurotrophic factor (BDNF) levels associated with neuronal survival. Many of these actions are mediated by the Neuropeptide Y1 receptor (NPY1R), the predominant NPY receptor in the brain and a critical regulator of mood-related circuitry (Cohen et al., 2018; Corvino et al., 2014; Rana et al., 2022). Genetic ablation of NPY1R reduces baseline hippocampal neurogenesis, diminishing dentate-gyrus progenitor proliferation and immature neuron numbers, indicating a key role for NPY1R signalling in maintaining the neurogenic niche (Decressac et al., 2011; Howell et al., 2003). Pharmacological activation of NPY1R produces antidepressant-like effects in adult rodents, including reduced immobility in the forced-swim test (FST) and anxiolytic-like behaviour, linking NPY1R activity to mood regulation (Goyal et al., 2009; Redrobe et al., 2002).
The galanin system plays a similarly critical, albeit more complex, role in emotional behaviour and hippocampal plasticity. GAL signals through three G-protein-coupled receptors (GPCR; galanin receptors (GALR1–3)), all expressed in the hippocampus and implicated in mood modulation (Branchek et al., 2000; Lu et al., 2005; O’Donnell et al., 1999). Depression-related phenotypes vary with receptor subtype: activation of GALR1 (or GALR3) tends to promote depressive-like behaviour, whereas stimulation of GALR2 produces antidepressant-like effects. Accordingly, central administration of galanin can induce despair-like behaviour via GALR1/3 pathways, but selective GALR2 agonism has the opposite effect, enhancing hippocampal neurogenesis markers and reducing depression-like behaviours in rats (Barr et al., 2006; Kuteeva et al., 2007, 2008). Increased GALR2 expression in the ventral hippocampus has been associated with antidepressant treatment efficacy, and GALR2 knockout mice exhibit heightened depressive phenotypes, underlining GALR2’s pivotal role in mood regulation (Lu et al., 2008; Luo et al., 2019). Emerging therapeutics targeting GALR2, such as intranasal delivery of GALR2-specific peptide agonists, further highlight its promise as a mediator of neurogenic and antidepressant outcomes (Yun et al., 2019).
Intriguingly, the NPY and GAL systems do not act independently in the hippocampus but converge through heteroreceptor interactions. Recent studies demonstrate that NPY1R and GALR2 can form physical heteroreceptor complexes in ventral dentate gyrus neurons (Alvarez-Contino et al., 2023; Borroto-Escuela et al., 2021, 2024; Beltran-Casanueva et al., 2024). These NPY1R-GALR2 heterocomplexes enable direct cross-talk between the two peptidergic signalling pathways, yielding synergistic effects on neuroplasticity. For example, the combined intranasal administration of a selective NPY1R agonist with a GALR2 agonist dramatically increased the proliferation of granule cell precursors in the adult rat dentate gyrus, effects accompanied by a sustained reduction in depression-like behaviour. Such enhancements were linked to increased proximity between NPY1R and GALR2, as detected by in situ proximity ligation assays (PLA). Blocking either receptor blocked the pro-neurogenic and behavioural benefits of co-treatment, confirming that the interaction between NPY1R and GALR2 is essential for the full synergistic effect (Alvarez-Contino et al., 2023; Beltran-Casanueva et al., 2024).
Neurotrophic signalling via BDNF/TrkB is a fundamental pathway underlying both neurogenesis and antidepressant responses, intersecting with NPY/GAL networks. BDNF is highly expressed in the hippocampus and is critical for the maturation and survival of newborn neurons; reduced BDNF levels are consistently observed in depressed patients and in animal models of stress (Eliwa et al., 2017; Castren and Kojima, 2017; Mahar et al., 2014; Thorsell and Mathe, 2017). Antidepressant treatments elevate BDNF levels and activate its receptor TrkB—a mechanism necessary for their behavioural efficacy. Recent evidence suggests that NPY1R can physically interact with TrkB receptors, forming NPY1R-TrkB heteroreceptor complexes that potentiate BDNF-driven signalling, thereby promoting robust neuronal growth and synaptic plasticity (Arrabal-Gomez et al., 2024; Casarotto et al., 2021; Castren and Monteggia, 2021). The pro-neurogenic and antidepressant-like effects of combined NPY1R and ketamine stimulation are abolished by pharmacological NPY1R and TrkB antagonism, indicating that BDNF/TrkB signalling is indispensable for these synergistic effects (Arrabal-Gomez et al., 2024).
In light of these findings, the current study seeks to elucidate the specific role of NPY1R in modulating hippocampal neurogenesis and mood-related behaviours through its interactions with GALR2 and TrkB. To this end, we developed a small interfering RNA (siRNA)-mediated NPY1R knockdown model in Sprague-Dawley rats. This approach enables us to directly assess how a transient reduction in NPY1R expression affects the formation of NPY1R-GALR2 and NPY1R-TrkB heteroreceptor complexes, as well as downstream markers of neurogenesis in the ventral hippocampus (e.g. proliferating cell nuclear antigen (PCNA) and BDNF) and depressive-like behaviour measured via the FST. We hypothesize that targeted knockdown of NPY1R will disrupt these heterocomplexes, leading to altered neurogenic processes and behavioural response. The results of this study are expected to provide critical mechanistic insights into the interplay between neuropeptidergic and neurotrophic signalling in mood regulation, thereby supporting the development of novel, targeted therapies for major depressive disorder.
Materials and methods
Animal housing, care and experimental design
Male Sprague-Dawley rats (6–8 weeks old, 200–250 g) were procured from CRIFFA (Barcelona) and housed in large group cages, with a maximum of six animals per cage, to foster social interaction and naturalistic behaviours. We used a single-sex cohort to limit variance for receptor-level endpoints in line with institutional animal care and use committee (IACUC) 3Rs guidance. Each cage was enriched with paper shavings and cardboard tubes to stimulate exploratory and burrowing activities, thereby enhancing overall well-being and reducing stress-induced variability in experimental outcomes (Baptista and Andrade, 2018; Boldrini et al., 2013). The housing environment was strictly controlled with a 12-hour light/dark cycle, maintaining ambient temperature at 22°C ± 2°C and relative humidity between 55% and 60%. Behavioural experiments were uniformly conducted between 09:00 and 14:00 hours to minimize diurnal fluctuations. All procedures complied with the EU Directive 2010/63/EU and Spanish legislation (Real Decreto 53/2013) and were approved by the University of Málaga’s Local Committee for the Ethics of Animal Experimentation (CEUMA 45-2022-A).
Animals were assigned randomly to experimental groups using a drawing lot method to ensure unbiased distribution of inherent biological variability, thereby enhancing the reliability and validity of our findings. Moreover, data collection and analysis were conducted under blinded conditions; experimenters were not involved in treatment administration, and group identities were coded to eliminate observer bias. Animals were allocated to post-injection time-points (day 6, day 8 and day 10; n = 5 per group per time-point). A between-subjects design was used across time-points; no rat contributed data to more than one time-point.
Intracerebral cannulation procedure
For precise intracerebral delivery, rats were anaesthetized using 5% isoflurane (ISOFLO®, Zoetis Abbott liquid gaseous anaesthetic, Madrid, Spain) in an induction chamber, with anaesthesia maintained via a 1.5%–2.5% isoflurane mask. Under aseptic conditions, a chronic 22-gauge stainless-steel guide cannula (Plastics One Inc., Roanoke, VA, 24018, US) was stereotaxically implanted into the right lateral cerebral ventricle using the following coordinates: +1.4 mm lateral, −1.0 mm posterior to bregma, and 3.6 mm ventral from the skull surface, as delineated by Paxinos and Watson (2006). Post-surgery, animals were housed individually to facilitate recovery and reduce post-operative stress. This standardized protocol for anaesthesia, cannulation and post-surgical management has been rigorously validated in our previous work (Borroto-Escuela et al., 2021; Mirchandani-Duque et al., 2022; Narváez et al., 2015, 2016, 2018).
Knockdown NPY1R siRNA rats
NPY1R knockdown was performed following the protocol previously established for GALR2 siRNA administration (Narváez et al., 2018). Briefly, after stereotaxic implantation of the guide cannula (see “Intracerebral Cannulation Procedure”), each rat received a single intracerebroventricular (ICV) injection of Accell Smart-Pool siRNA targeting NPY1R(Dharmacon) (5 µg; 0.35 nmol) in 5 µL diluted in Accell siRNA Delivery Media, infused at 1 µL/minutes; the injector remained in place for 2 minutes to prevent reflux. Animals were randomly assigned to experimental groups and allowed to recover for 6, 8 or 10 days post-injection to achieve progressive reduction in NPY1R expression. Control animals received 5 µL of Accell Non-targeting Pool siRNA diluted in the same delivery media. Immediately after behavioural testing at each time-point, the same animals were deeply anaesthetized and perfused; brains were processed for PLA, NPY1R-IR, PCNA and BDNF as described. Rats were deeply anaesthetized with pentobarbital (Mebumal, 100 mg/kg, i.p.) and transcardially perfused with 4% paraformaldehyde (Sigma Aldrich, St. Louis, MO, USA). Brains were then extracted and coronally sectioned at 30 μm using a Cryostat (HM550, Microm International, Walldorf, Germany) across the ventral hippocampus (Bregma −5.20, −6.72 mm, according to Paxinos and Watson, 2006).
Evaluation of NPY1R expression in the hippocampus
To quantify NPY1R protein levels, free-floating sections from the dentate gyrus were processed for antigen retrieval by incubation in saline sodium citrate buffer (pH 6; 10 nM sodium citrate) at 65°C for 90 minutes, followed by a 30-minute incubation in 0.6% H₂O₂ to quench endogenous peroxidase activity. Sections were then incubated overnight at 4°C with a primary antibody against NPY1R (goat anti-NPY1R, 1:200, sc-21992, Santa Cruz Biotechnology, CA; and a mouse anti-NPY1R, 1:200, Santa Cruz Biotechnology, CA) in 2.5% donkey serum. Concordant labelling patterns were obtained with both primaries. Negative controls included omission of primary antibody and species-matched isotype controls. After washing in phosphate-buffered saline (PBS), sections were incubated for 90 minutes with a biotinylated anti-mouse IgG (1:200, Sigma, St. Louis, MO, USA) followed by a 1-hour incubation in ExtrAvidin peroxidase (1:100, Sigma, St. Louis, MO, USA) in darkness. The immunoreactivity was visualized using a diaminobenzidine (DAB) solution (0.05% diaminobenzidine and 0.03% H₂O₂ in PBS), and sections were mounted on gelatin-coated slides and coverslipped with DePeX mounting medium (Merck Life Science SLU, Darmstadt, Germany). NPYY1R-positive cells were quantified using an optical fractionator method under unbiased stereological microscopy (Olympus BX51, Olympus, Denmark) as detailed in our Supplemental Material (Borroto-Escuela et al., 2022; Mirchandani-Duque et al., 2022; Narváez et al., 2018).
Analysis of heteroreceptor complexes through in situ proximity ligation assay
To assess the NPY1R with GALR2 and TrkB heteroreceptor complexes formation in the ventral dentate gyrus, we employed the in situ PLA method using NaveniFlex Tissue MR Atto 647N (Navinci, Sweden). Free-floating brain sections were first incubated in blocking buffer at 37°C for 1 hour in a pre-warmed humidity chamber, then incubated overnight at 4°C with primary antibody pairs: mouse anti-NPY1R (1:200, Santa Cruz Biotechnology, CA) with rabbit anti-TrkB (1:200, Sigma Aldrich, ZRB1281) for NPY1R-TrkB detection, and mouse anti-NPY1R (1:200, Santa Cruz Biotechnology, CA) with rabbit anti-GALR2 (1:100, Alomone Lab) for NPY1R-GALR2 detection. Following thorough washing, a mixture of Navenibody goat and rabbit was applied for 1 hour at 37°C. Unbound probes were removed, and Enzymes A and B were sequentially applied for 60 and 30 minutes, respectively, in a humidity chamber at 37°C. Excess connector oligonucleotides were washed away, and the sections were then incubated with the rolling circle detection mixture (Enzyme C, Tex615) for 90 minutes at 37°C. Finally, sections were mounted with a fluorescent mounting medium containing 4′,6-diamidino-2-phenylindole (DAPI) (Abcam, ab104139, Cambridge, UK), stored at −20°C, and later imaged using confocal microscopy as described previously (Borroto-Escuela et al., 2021, 2022; Diaz-Sanchez et al., 2023; Narváez et al., 2020, 2021).
Hippocampal cell proliferation assessment
Brain sections designated for cell proliferation analysis underwent antigen retrieval by incubating at 65°C for 90 minutes in saline sodium citrate buffer (pH 6; 10 nM sodium citrate). To eliminate endogenous peroxidase activity, sections were subsequently incubated in 0.6% H₂O₂ for 30 minutes. They were then incubated overnight at 4°C with a primary antibody against PCNA (1:1500, P8825, Sigma, St. Louis, MO, USA). The following day, sections were incubated with a secondary antibody (1:200, B8895, Sigma, St. Louis, MO, USA) for 90 minutes. Amplification was achieved using ExtrAvidin peroxidase (1:100, Sigma, St. Louis, MO, USA) for 1 hour at room temperature in darkness, and detection was performed with a solution of 0.05% DAB and 0.03% H₂O₂ in PBS. After multiple washes, sections were mounted on gelatin-coated slides, dehydrated through graded alcohols, and coverslipped. PCNA-labelled cells were quantified using the optical fractionator method under unbiased stereological microscopy (Olympus BX51, Olympus, Denmark), as previously described (Alvarez-Contino et al., 2023; Mirchandani-Duque et al., 2022; Narváez et al., 2018).
Evaluation of BDNF induction in the ventral dentate gyrus
For evaluation of BDNF induction in the ventral dentate gyrus, a separate set of sections underwent similar antigen retrieval and peroxidase neutralization procedures. These sections were then incubated at room temperature with a primary BDNF antibody (Chemicon, AB1534SP, 1:500 in 2.5% donkey serum, Merck KGaA, Darmstadt, Germany). After thorough PBS washes, sections were incubated with a biotinylated anti-rabbit IgG secondary antibody (1:200, Sigma, B8895) followed by ExtrAvidin peroxidase (1:100, Sigma) in darkness. Visualization of BDNF-expressing cells was achieved using a DAB and H₂O₂ mixture, and processed sections were mounted, coverslipped, and analysed using the optical fractionator method with unbiased stereology on an Olympus BX51 microscope.
Behavioural assessment
Depression-like behaviour was evaluated using the FST, a widely accepted model for assessing despair-like behaviour in rodents (Planchez et al., 2019; Porsolt et al., 1977; Yankelevitch-Yahav et al., 2015). Behavioural testing was conducted between 09:00 and 14:00 hours at three post-injection time-points (days 6, 8 and 10) in alignment with the molecular endpoints. Prior to testing, rats were acclimatized to handling and the experimental environment and then randomly allocated into treatment groups. To ensure that any alterations in FST performance were not confounded by locomotor impairments, an Open-Field Test (OFT) was conducted, with total distance moved analysed using video tracking software (Smart2.5, Panlab, SL; Kalynchuk et al., 2004; Morales-Medina et al., 2012, 2013; Ribeiro et al., 2020). Within each time-point, the same rats performed the OFT (24 hours before) and the FST; behavioural testing was completed prior to perfusion and tissue collection for PLA, NPY1R immunohistochemistry, PCNA and BDNF analyses.
For the FST, each rat was placed individually in a cylinder containing water maintained at 25 ± 0.2°C for a total of 10 minutes. Immobility (defined as floating with minimal movement to keep the head above water) and active swimming were recorded during the final 5 minutes using Raton Time 1.0 software (Fixma S.L., Valencia, Spain). After the test, rats were carefully dried in heated cages before being returned to their home cages. All behavioural assessments were performed by observers blinded to the treatment conditions (Beltran-Casanueva et al., 2024; Borroto-Escuela et al., 2024; Cryan et al., 2005; Porsolt et al., 1977; Redrobe et al., 2002).
Statistical analysis
Data were treated such that each rat was considered an independent experimental unit. Results are expressed as mean ± standard error of the mean, with sample sizes specified in the figure legends. Data were analysed using GraphPad PRISM 8.0 (GraphPad Software, La Jolla, CA, USA). Comparisons between groups were performed using one-way analysis of variance (ANOVA) followed by Newman–Keuls post hoc tests or Student’s unpaired t-test where appropriate. Significance was defined at *p < 0.05, **p < 0.01, and ***p < 0.001.
Results
Evaluation of NPY1R expression in the ventral hippocampus
Immunohistochemical analysis was performed to quantify NPY1R protein expression in the dentate gyrus of the ventral hippocampus over a time course of 6, 8 and 10 days following ICV injection of NPY1R siRNA. Quantitative analysis revealed a statistically significant, time-dependent reduction in NPY1R immunoreactivity, with maximal reduction observed at 8 days post-injection (one-way ANOVA, F(3,16) = 4.96, p < 0.05; Figure 1(a)–(d)). Specifically, the 8-day group displayed markedly lower NPY1R protein levels compared with the control, 6-day and 10-day groups, indicating that the 8-day time-point is critical for achieving optimal knockdown.
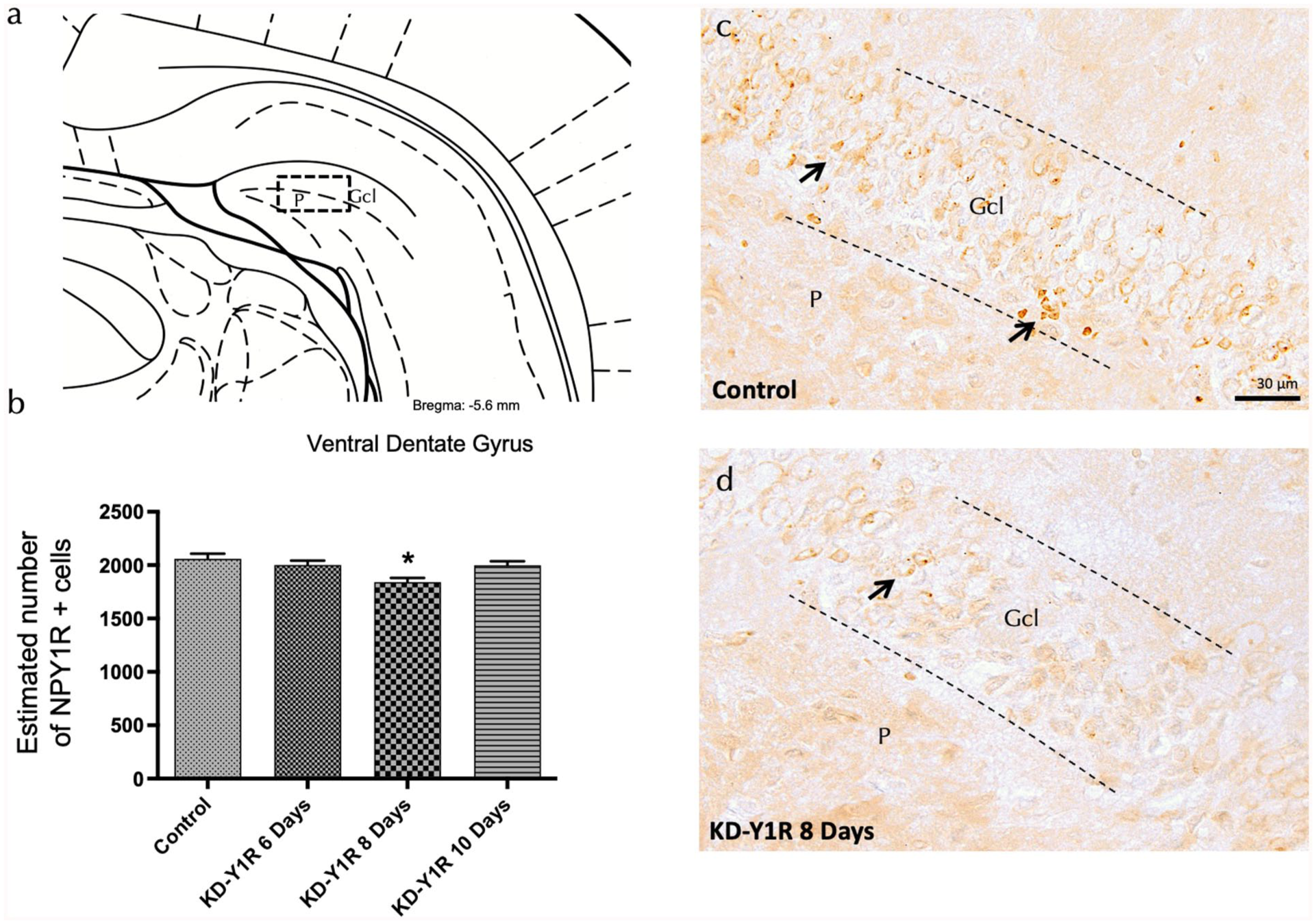
NPY1R expression in the ventral dentate gyrus on NPY1R-knockdown rats.
Knockdown of NPY1R siRNA decreases NPY1R-GALR2 and NPY1R-TrkB heteroreceptor complexes
To assess the impact of NPY1R knockdown on receptor complex formation, we employed in situ PLA to detect NPY1R-GALR2 and NPY1R-TrkB heteroreceptor complexes in the ventral dentate gyrus. Analysis revealed that positive red PLA signals, corresponding to these heterocomplexes, were predominantly localized within the subgranular zone and dispersed in the polymorphic layer. A one-way ANOVA confirmed a significant reduction in the density of NPY1R-GALR2 complexes 8 days after siRNA administration (F(3,16) = 4.89, p < 0.01; Figure 2(a)–(d)). Newman–Keuls post hoc tests revealed that the 8-day group exhibited a significantly lower density compared to the control group (p < 0.01) and to both the 6-day and 10-day groups (p < 0.05 for each comparison).
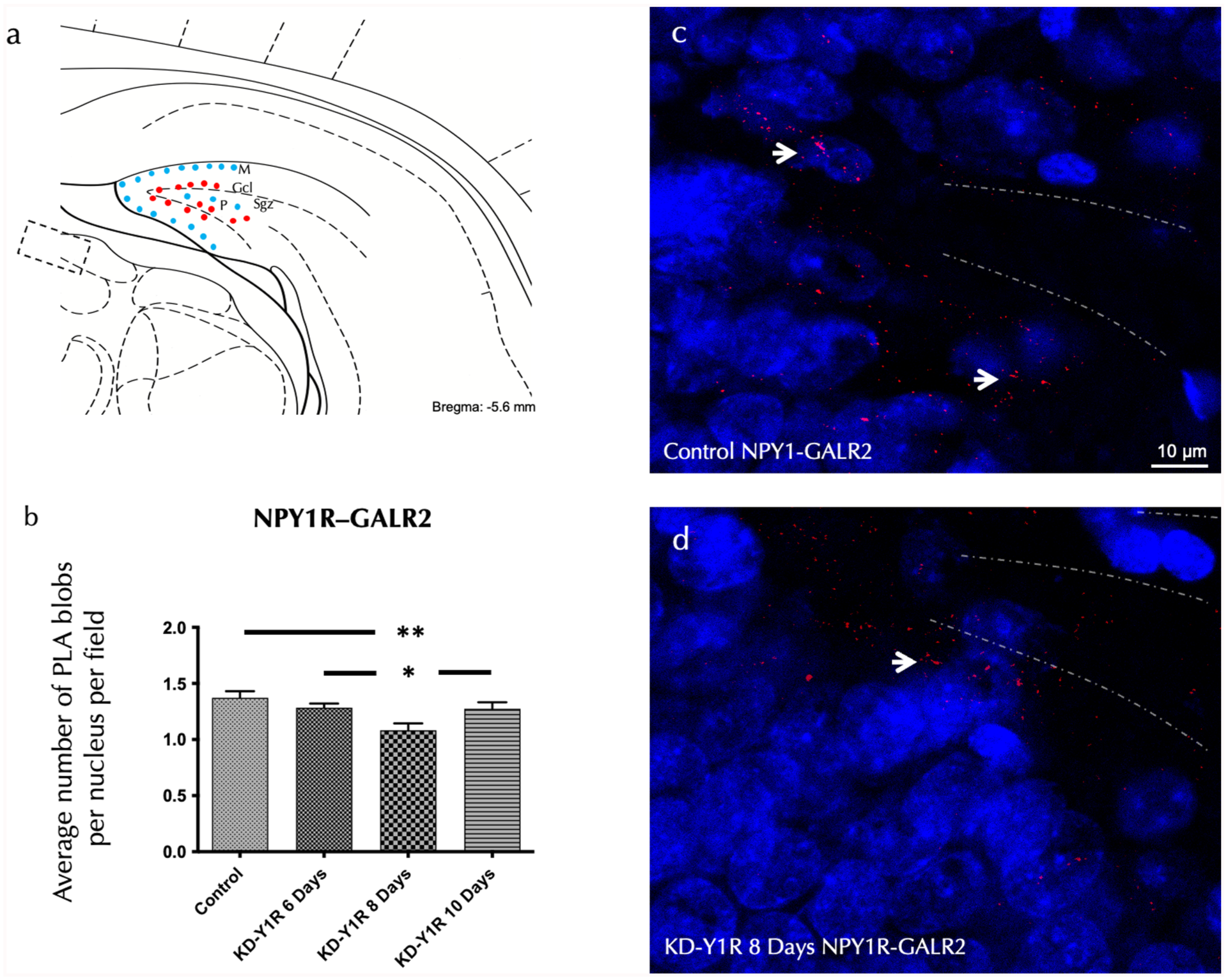
In situ detection of NPYY1R/GALR2 heteroreceptor complexes within the ventral hippocampal DG via PLA.
Similarly, analysis of NPY1R-TrkB heteroreceptor complexes revealed a significant reduction in their density 8 days post-siRNA administration. A one-way ANOVA demonstrated a significant effect (F(3,16) = 9.61, p < 0.001; Figure 3(a)–(d)). Newman–Keuls post hoc tests showed that the 8-day group differed significantly from the control group (p < 0.001), from the 6-day group (p < 0.01) and from the 10-day group (p < 0.05). These data confirm that the NPY1R knockdown not only reduces overall receptor levels but also disrupts its ability to form functional heteroreceptor assemblies with TrkB.
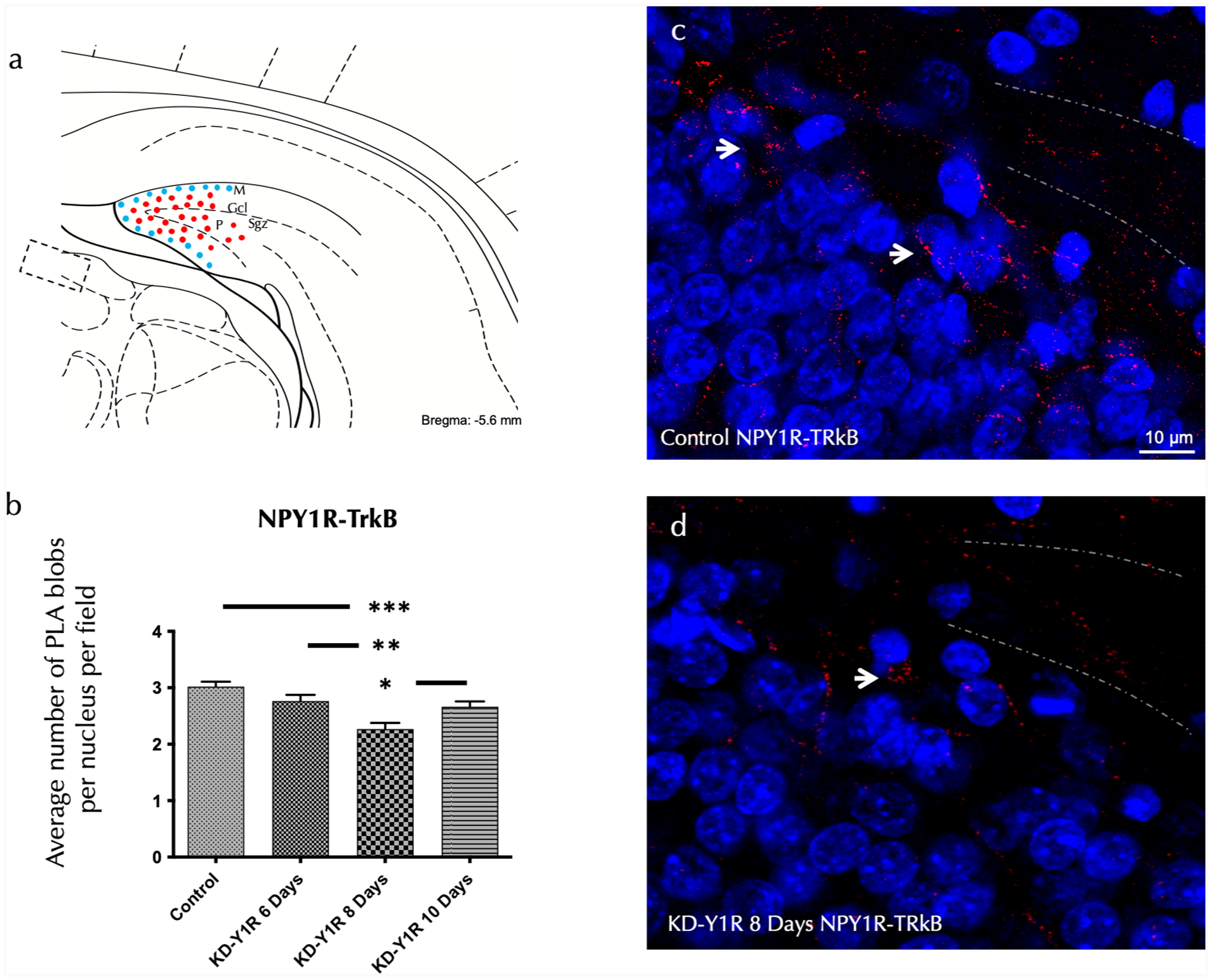
In situ detection of NPY1R/TrkB heteroreceptor complexes within the ventral hippocampal DG via PLA.
Cell proliferation in the ventral hippocampus is preserved in NPY1R knockdown siRNA rats
Cell proliferation in the ventral hippocampal dentate gyrus was evaluated by quantifying PCNA-positive cells following NPY1R siRNA administration at 6, 8 and 10 days. Notably, one-way ANOVA analysis revealed no significant differences in the number of PCNA-positive cells among the groups (F(3,16) = 0.57, p > 0.05; Figure 4(a)–(d)). This suggests that the transient knockdown of NPY1R does not adversely affect the overall proliferation rate of hippocampal progenitor cells within the time frame examined.
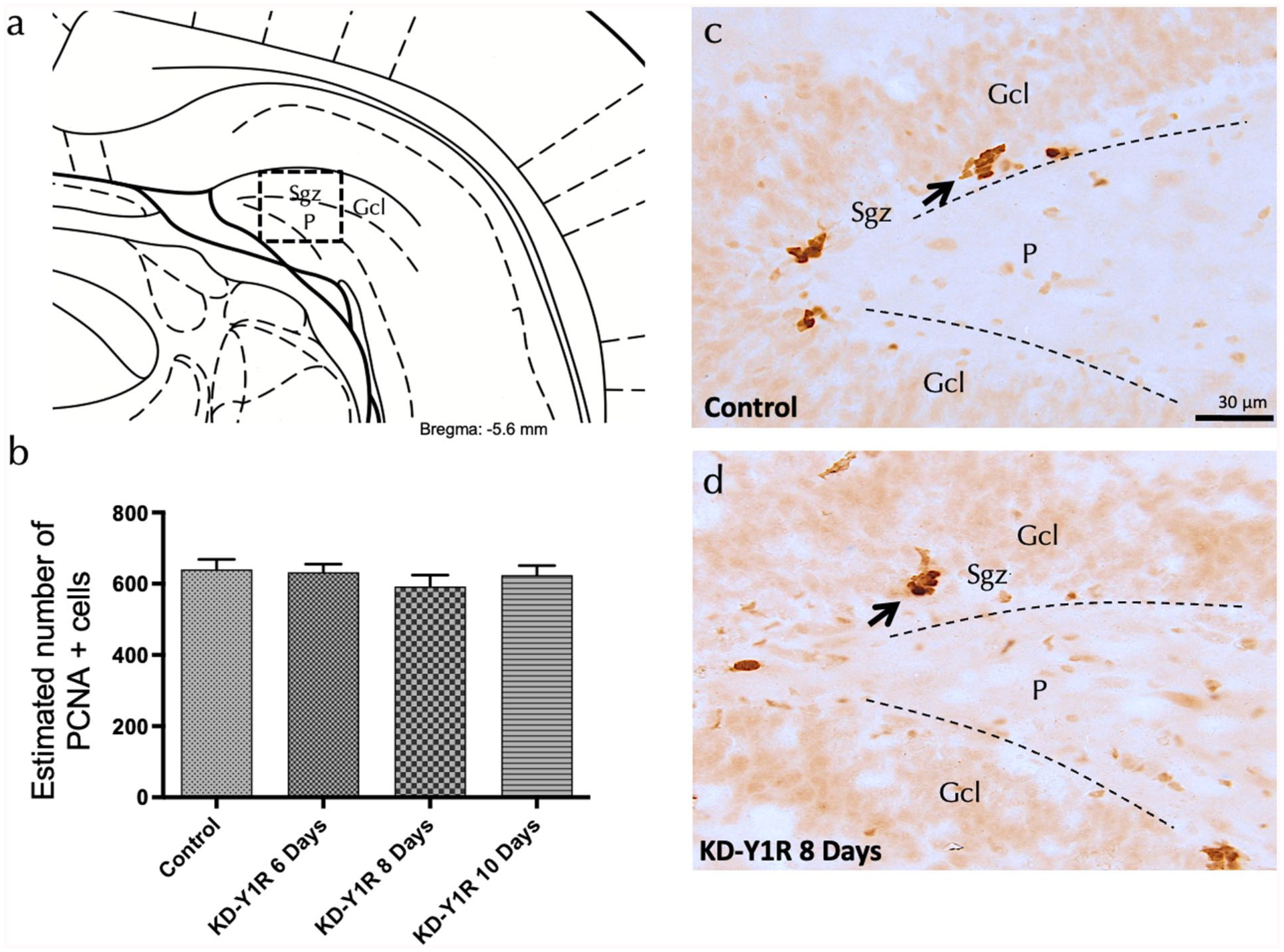
Cell proliferation assessment in the ventral dentate gyrus on NPY1R-knockdown rats.
Preserved proliferation is associated with maintained BDNF expression
To elucidate potential compensatory mechanisms supporting preserved neurogenesis, we examined BDNF expression in the ventral dentate gyrus following NPY1R siRNA administration. Brain sections were processed for BDNF immunohistochemistry, and stereological quantification revealed that BDNF-positive cell counts in the granular layer remained comparable across control and siRNA-treated groups (one-way ANOVA, F(3,16) = 0.15, p > 0.05; Figure 5(a)–(d)). BDNF-positive cells were primarily localized in the granular region, with only sporadic expression in the polymorphic layer, suggesting that despite the reduction in NPY1R and associated heteroreceptor complexes, neurotrophic support via BDNF is maintained.
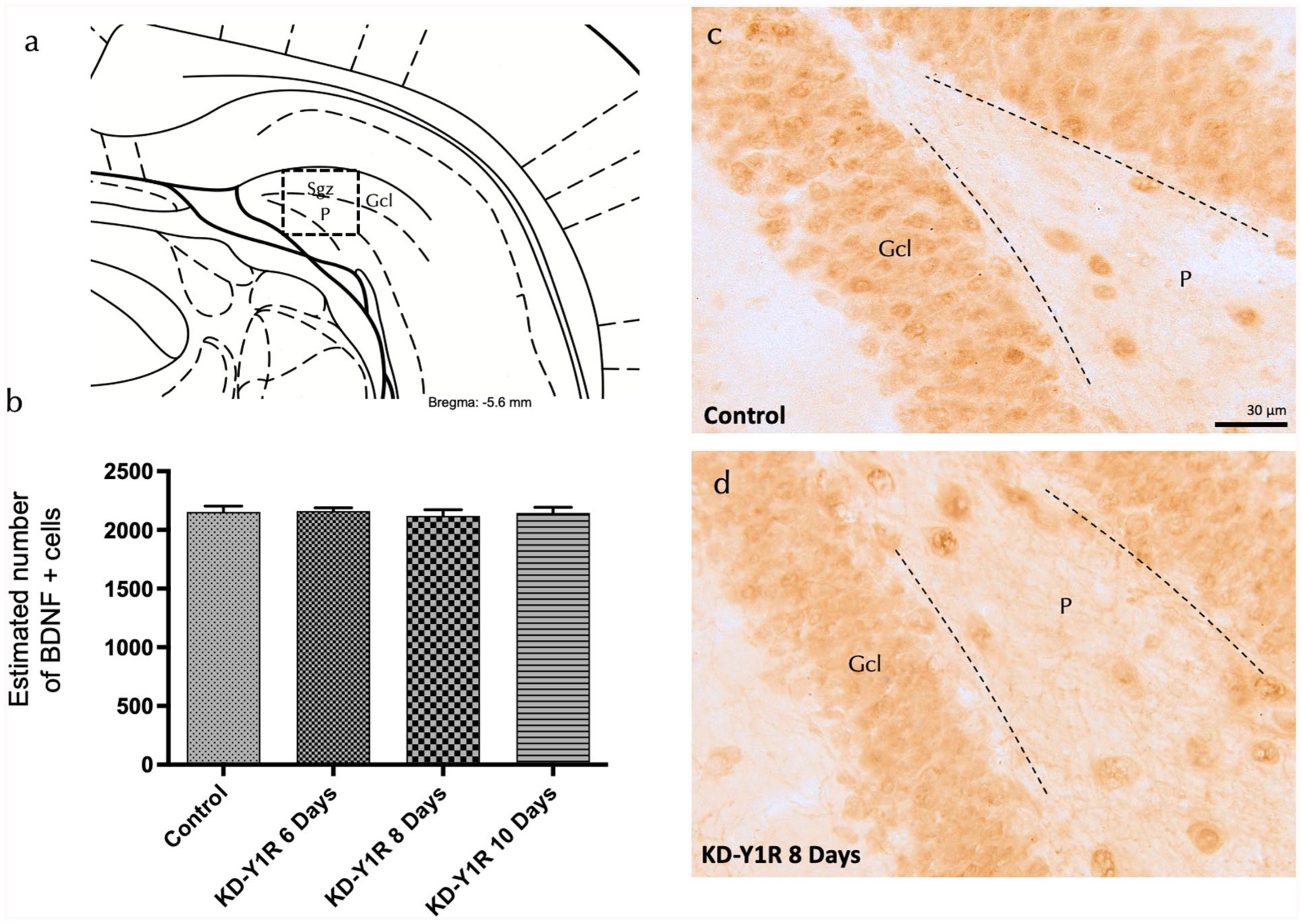
NPY1R-siRNA effects on hippocampal BDNF-IR cells of the ventral DG hippocampal region.
Knockdown of NPY1R siRNA induction does not affect depressive-like behaviour in the FST
Behavioural assessments using the FST were conducted 24 hours after siRNA administration at 6, 8 and 10 days. Analysis of immobility and swimming times revealed no significant differences between the NPY1R siRNA-treated groups and the vehicle control (one-way ANOVA, F(3,16) = 1.17 for immobility and F(3,16) = 0.07 for swimming; Figure 6(a)–(b)). Furthermore, no changes in locomotor activity were observed in the OFT (data not shown). These findings indicate that the transient knockdown of NPY1R does not elicit measurable alterations in depressive-like behaviour, suggesting that compensatory mechanisms (e.g. maintained BDNF signalling) may counterbalance the reduction in NPY1R and heteroreceptor complex density.
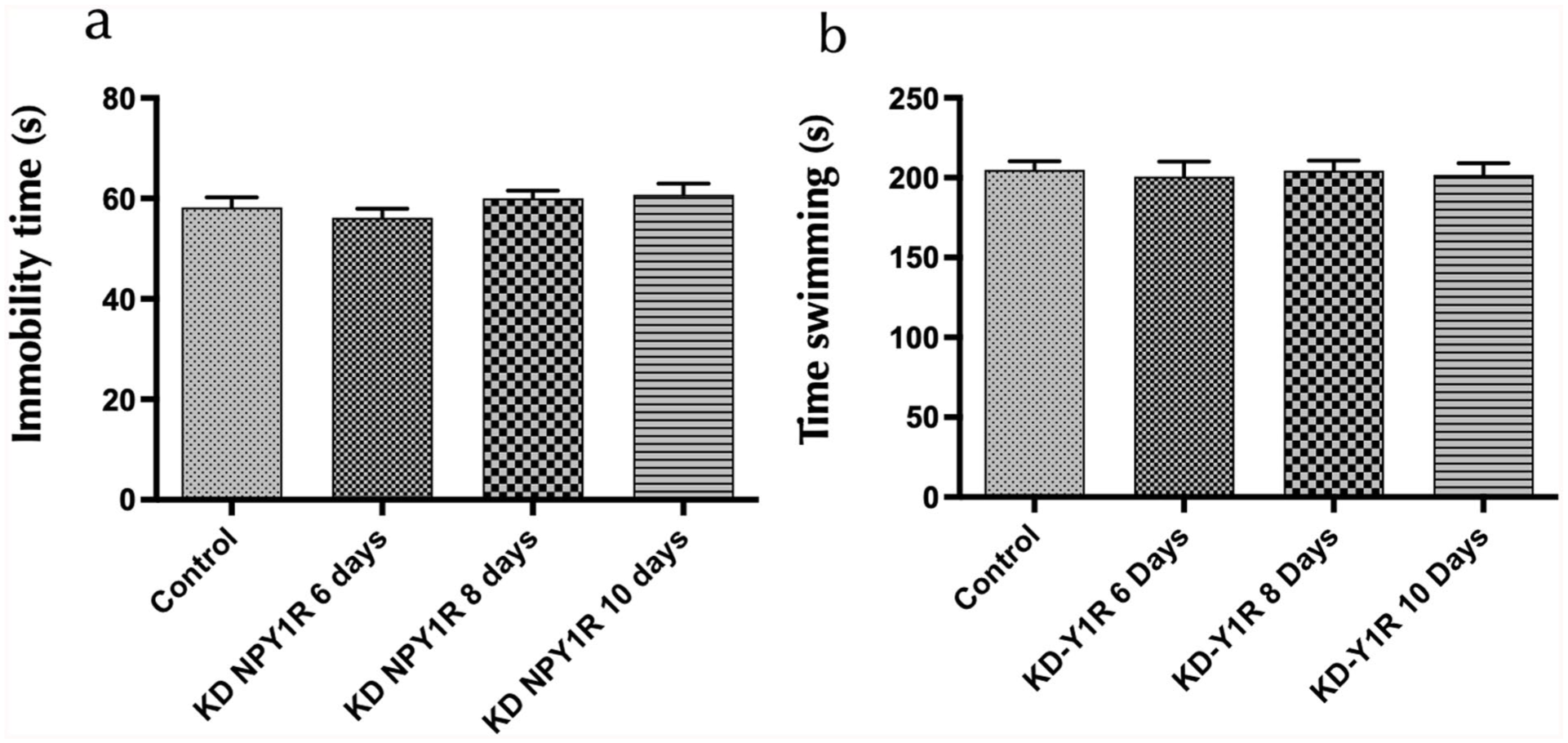
Behavioural responses to NPY1R-knockdown rats in the FST.
Discussion
Reduction in NPY1R expression and heteroreceptor complex disruption
We observed that ICV administration of NPY1R-targeted siRNA produced a time-limited, partial decrease in NPY1R expression that peaked at day 8 in both the ventral and dorsal dentate gyrus. This aligns with previous work showing that GALR2 knockdown also leads to a marked reduction in heterocomplex formation at similar time-points (Narváez et al., 2018), suggesting a conserved temporal window optimal for receptor depletion and functional assessment. The reduction in NPY1R was paralleled by a significant decline in the density of NPY1R-GALR2 and NPY1R-TrkB heteroreceptor complexes, indicating a direct dependence on NPY1R availability for the stable formation of these assemblies. Beyond confirming their presence and specificity in vivo, these measures serve as pharmacodynamic markers of target engagement that report on neuropeptide–neurotrophin receptor interactions in the hippocampus (Borroto-Escuela et al., 2021; Narváez et al., 2016, 2018). The formation of NPY1R-GALR2/TrkB complexes correlates with downstream signalling efficiency, reinforcing their modulatory role in integrating neuropeptidergic and neurotrophic inputs within the hippocampus (Arrabal-Gomez et al., 2024; Castren and Monteggia, 2021).
Although the maximal effect on day 8 is temporally circumscribed, the magnitude of target engagement at this time-point was substantial, with clear reductions in NPY1R immunoreactivity and in the density of NPY1R-GALR2/TrkB heteroreceptor complexes. The absence of changes in PCNA, BDNF and forced-swim measures under these conditions therefore constrains causal models linking these heterocomplexes to hippocampal neurogenesis and stress-coping behaviour in naïve rats. In the broader context of receptor–receptor interactions, this finding complements prior work establishing GPCR heteroreceptor complexes as modulators of mood-related circuitry, including systems engaging galanin and neurotrophin signalling; here we provide in vivo evidence of robust heterocomplex disruption without downstream functional change within the tested window (Borroto-Escuela et al., 2017a; Narváez et al., 2021). This dissociation is noteworthy given prior reports of antidepressant-like effects with hippocampal Y1R engagement (Ishida et al., 2007).
Consistent with single-dose Accell-siRNA delivery to the adult rodent brain, the peak reduction in NPY1R we observed in the ventral DG occurred 8 days after ICV injection and then waned as protein re-synthesis progressed. This time-limited profile matches prior demonstrations of neuron-selective, in vivo knockdown following a single ICV Accell dose (Nakajima et al., 2012). Importantly, the partial magnitude of receptor loss helps explain the preserved downstream readouts: for GPCR systems with receptor reserve, non-linear occupancy–response relationships allow near-normal signalling until a depletion threshold is exceeded, such that moderate decreases in receptor abundance may be insufficient to alter network or behavioural outputs within short observation windows (Kenakin, 2013; Sum et al., 2004). In the NPY literature, antidepressant-like actions are robust when NPY1R is actively engaged (and with NPY1R overexpression), whereas compensatory adaptations, including adjustments in other NPY receptor populations, can blunt phenotypes under partial Y1R loss (Olesen et al., 2012; Redrobe et al., 2002; Wittmann et al., 2005). Taken together, our data indicate clear target engagement at the receptor/heterocomplex level but suggest that greater or more sustained depletion, or circuit-targeted interference, may be required to drive changes in neurogenesis or stress-coping behaviour.
The day-8 peak and the return towards baseline by day 10 are consistent with the expected pharmacodynamic lag between siRNA-induced mRNA suppression and protein/complex turnover, followed by re-synthesis and re-assembly. In vivo studies using Accell-type siRNAs have documented neuron-selective uptake with knockdown spanning roughly 1 week, and proteome-wide analyses in neurons show protein half-lives ranging from hours to >10 days, supporting a scenario in which complex abundance can recover rapidly once synthesis resumes. These temporal considerations suggest that longer-lasting engagement and/or circuit-restricted interference may be required to drive downstream plasticity or mood-related changes (Dorrbaum et al., 2018, 2020; Nakajima et al., 2012; Taniguchi et al., 2020).
While the decreases in NPY1R-GALR2 and NPY1R-TrkB density were significant, they were partial, which likely contributed to the preserved downstream outcomes. GPCR heteroreceptor complexes are dynamic, undergoing activity-dependent assembly/disassembly, trafficking and compartmentalized signalling; as a result, moderate reductions in complex abundance may not surpass the functional threshold required to impact circuit-level behaviour within short observation windows. Moreover, receptor reserve and system-level amplification at the heteromer level, together with compensatory crosstalk among GPCRs and Receptor tyrosine kinases (RTKs) (e.g. mechanisms that sustain TrkB/BDNF signalling), can preserve output despite partial complex loss. These considerations align with current views of heteromer plasticity and with reports that GPCR-RTK interactions can maintain neurotrophic drive even when specific GPCR complexes are reduced (Borroto-Escuela et al., 2017a, 2017b; Lao-Peregrin et al., 2024; Sutkeviciute and Vilardaga, 2020).
Preservation of hippocampal cell proliferation and BDNF expression
Interestingly, despite a robust decrease in NPY1R expression and its heteroreceptor complexes, our results did not reveal a significant reduction in cell proliferation as measured by PCNA-positive cell count in the dentate gyrus. This contrasts with previous findings in constitutive knockout mouse models, where complete genetic ablation of NPY1R consistently reduced hippocampal progenitor proliferation, highlighting differences between transient knockdown and permanent genetic deletion (Decressac et al., 2011; Howell et al., 2003). This notion is supported by earlier reports in genetic knockout studies where lifelong absence of NPY1R resulted in significant neurogenic deficits (Howell et al., 2003). It is possible that a transient reduction of receptor expression, as induced by siRNA, may not sufficiently disrupt proliferative processes, perhaps due to compensatory mechanisms involving other neuropeptide receptors or signalling pathways. Therefore, future studies might consider using genetically engineered rat models with permanent knockout or selective conditional knockouts to conclusively elucidate the role of NPY1R in neurogenesis.
In agreement with the preserved proliferative capacity, BDNF expression remained unaffected by NPY1R knockdown. Given that BDNF signalling via TrkB receptor activation is a critical mediator of neurogenesis and antidepressant responses, the unaltered BDNF expression observed here is consistent with the absence of proliferative and behavioural deficits (Arrabal-Gomez et al., 2024; Beltran-Casanueva et al., 2024). Previous studies in transgenic animals indicate that a congenital absence of specific receptors or prolonged suppression of BDNF signalling is required to significantly impact BDNF-mediated neurogenesis and associated behaviours (Castren and Kojima, 2017; Mahar et al., 2014). These findings suggest resilience mechanisms in transient receptor knockdown models that preserve neurogenic and trophic processes in the hippocampus, highlighting the need for genetically stable knockout models to clearly delineate receptor-specific contributions (Howell et al., 2005). Moreover, compensatory upregulation of neurotrophic factors has been observed in studies where partial receptor loss did not translate into reduced neurogenesis (Cohen et al., 2018), which is consistent with the literature indicating that BDNF is a robust modulator of hippocampal plasticity and can buffer against receptor-level perturbations (Castren and Kojima, 2017; Miranda et al., 2019). This robustness may reflect an inherent capacity of the hippocampal microenvironment to safeguard neurogenesis, even when heteroreceptor complex formation is compromised.
Behavioural outcomes in the FST
Behavioural analyses further supported these neurobiological findings. The lack of observed changes in depressive-like behaviour, assessed using the FST, aligns with the maintained proliferation and BDNF levels following transient NPY1R knockdown. This absence of a detectable behavioural effect, despite the molecular alterations, may be interpreted in several ways. First, the transient knockdown of NPY1R might be insufficient to elicit changes in behaviour if compensatory signalling (such as maintained BDNF levels) balances receptor loss. Second, previous studies using chronic genetic ablation of NPY1R have reported marked behavioural alterations, suggesting that long-term loss rather than transient reduction is necessary to impact mood-related behaviours (Howell et al., 2003). Third, it is possible that additional aspects of hippocampal circuitry, such as synaptic integration and connectivity, require more prolonged receptor deficiency to manifest behavioural changes. These interpretations are consistent with literature indicating that antidepressant-like effects often correlate with sustained neuroplastic changes rather than acute receptor-level modifications (Goyal et al., 2009; Redrobe et al., 2002).
Limitations and translational implications
Our study utilized an siRNA-mediated knockdown approach to transiently reduce NPY1R expression in the ventral hippocampus, which enabled us to capture early molecular changes, specifically, the reduction of NPY1R expression and the concomitant decline in NPY1R-containing heteroreceptor complexes. However, this transient method has several limitations. First, the temporary nature of siRNA knockdown may not fully recapitulate the chronic or congenital loss of NPY1R function, suggesting that sustained receptor deficiency might be required to uncover the full impact on neurogenesis and mood regulation. We evaluated a single siRNA dose across three time-points and observed a transient maximal effect at day 8; longer-lasting or repeated interventions (e.g. adeno-associated virus (AAV)-mediated short hairpin RNA (shRNA)/clustered regularly interspaced short palindromic repeats interference (CRISPRi)) will be needed to test duration-dependence. Moreover, the ICV route does not restrict knockdown to specific microcircuits; future region- or cell-type-targeted approaches (e.g. DG-focused AAV-shRNA with Cre-dependent constructs) may reveal context-dependent effects (Kim et al., 2022; Xie et al., 2021).
Second, our findings indicate that, despite significant receptor knockdown and a reduction in heteroreceptor complex density, key neurogenic markers such as PCNA-positive cell counts and BDNF expression remained unaltered. This preservation could be due to compensatory mechanisms that maintain neurotrophic support in the short term. It is possible that prolonged or permanent NPY1R ablation may eventually lead to deficits that were not detected within the 6- to 10-day window of our study.
This study used male rats only to limit variance for mechanistic receptor-level outcomes and to adhere to reduction within the 3Rs. However, given reports of sex-dependent features in NPY and BDNF/TrkB systems, the generalizability to females remains to be established. Future work will include female cohorts with oestrous-cycle monitoring and adequate power to test Sex × Treatment interactions across molecular and behavioural endpoints (Justice, 2024; Miller et al., 2017).
Clinically, within the present model and time window, receptor-heterocomplex measures are best interpreted as pharmacodynamic markers of target engagement. The observed reduction in NPY1R-containing heteroreceptor complexes indicates engagement of the intended receptor systems but does not by itself establish changes in neurogenesis or behaviour. Whether such measures can serve as clinical biomarkers of neurotrophic signalling will require studies with sustained and region-restricted manipulations and human validation. Second, our findings motivate exploration of multi-target strategies that co-activate NPY1R with GALR2 or facilitate NPY1R-TrkB coupling, as these mechanisms are linked to neurotrophic support. Prospective approaches—such as heterobivalent ligands—merit evaluation for their capacity to enhance plasticity, but their antidepressant efficacy remains to be determined. Moreover, whether heteroreceptor-complex-related readouts (e.g. in cerebrospinal fluid or peripheral cells) can be developed as feasible pharmacodynamic or predictive biomarkers should be examined in future proof-of-concept studies. Lastly, clinical trials evaluating intranasal delivery of neuropeptide-based therapeutics, particularly those designed to enhance NPY1R-GALR2-TrkB signalling, may pave the way for non-invasive, rapid-acting treatments for major depressive disorder and other neuroplasticity-related disorders.
In summary, while our transient NPY1R knockdown approach confirms the importance of receptor integrity for heterocomplex formation, it also reveals a robust compensatory capacity of the hippocampal neurogenic machinery. These findings lay the groundwork for future research aimed at chronic receptor modulation and the development of multi-target therapeutic strategies that could significantly improve clinical outcomes in mood disorders.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251389528 – Supplemental material for Intracerebroventricular knockdown of NPY1R disrupts NPY1R-GALR2/TrkB heteroreceptor complexes without affecting neuroplasticity or depressive-like behaviour
Supplemental material, sj-docx-1-jop-10.1177_02698811251389528 for Intracerebroventricular knockdown of NPY1R disrupts NPY1R-GALR2/TrkB heteroreceptor complexes without affecting neuroplasticity or depressive-like behaviour by Isabel Moreno-Madrid, Carlos Arrabal-Gómez, Jesús Romero-Imbroda, Amelia Díaz-Casares, Kjell Fuxe, Dasiel Borroto-Escuela, Pedro Serrano-Castro and Manuel Narváez in Journal of Psychopharmacology
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by the PI24/01816 from Instituto de Salud Carlos III,ProyExcel_00613 Junta de Andalucía, Spain, and PPRO-B4-2024-011 from Universidad de Málaga, Spain, to MN and PS-C. Funding for open access charge: Instituto de Investigación Biomédica de Málaga y Plataforma en Nanomedicina–IBIMA Plataforma Bionand. . Additional funding support came from Cátedra Imbrain: Neurociencia Integrada y Bienestar to MN. This work also received support from Stiftelsen Olle Engkvist Byggmästare in 2018 and 2021, as well as from the Swedish Medical Research Council (Grant No. 62X-00715-50-3) awarded to KF and DB-E. Additionally, funding was provided by Hjärnfonden (Grants F02018-0286 and F02019-0296), Karolinska Institutet Forskningsstiftelser 2022, EMERGIA 2020-39318 (Plan Andaluz de Investigación, Desarrollo e Innovación 2020), and CONSOLIDACION INVESTIGADORA (CNS2022-136008, Programa Estatal para Desarrollar, Atraer y Retener Talento, del Plan Estatal de Investigación Científica, Técnica y de Innovación 2021–2023) awarded to DB-E. DB-E is affiliated with the Academia de Biólogos Cubanos and the Observatorio Cubano de Neurociencias (Yaguajay, Cuba).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
The data that support the findings of this study are openly available in the Institutional repository of the University of Málaga (RIUMA) and from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
