Abstract
Background:
The treatment of major depressive disorder (MDD) with available antidepressant drugs is characterized by considerable ineffectiveness. Classical psychedelics such as psilocybin and N,N-dimethyltryptamine (DMT), which act primarily as 5-hydroxytryptamine 2A (5-HT2A) receptor agonists, have shown preliminary efficacy for inducing long-term remission in MDD after one or two doses. GM-2505 is a novel, 5-HT2A receptor agonist, developed for treating MDD.
Methods:
In this single-ascending dose, randomized, placebo-controlled, double-blind study, we characterized GM-2505’s safety, tolerability, pharmacokinetics (PK), and pharmacodynamic (PD) profile in 48 healthy participants.
Results:
Single intravenous (IV) doses up to 20 mg demonstrated an acceptable safety profile of mild transient adverse events, short-term, non-clinically significant increases in blood pressure and pulse, and no significant changes in electrocardiographs, consistent with other 5-HT2A receptor agonists. In general, GM-2505 Cmax and AUClast increased dose proportionally, with t1/2 of 40–50 minutes. Generally, dose-dependent effects were observed for neuroendocrine hormones, several neuropsychological and neurophysiological measures, and subjective drug effects. Dose-related effects were also observed in resting-state electroencephalography (rsEEG), with decreased power in the low frequency rsEEG bands (theta and alpha), and increased in the high frequency bands (slow and fast gamma).
Conclusions:
These PD findings were similar in nature and magnitude to other 5-HT2A receptor agonists that have been studied clinically. In line with the GM-2505 PK profile, the duration of cardiovascular and subjective effects was shorter than psilocybin but longer than DMT, demonstrating a potentially more practical temporal profile for use in a supervised clinical setting compared to longer-acting 5-HT2A receptor agonists, with an optimal dose range of 10–15 mg IV. Clinical trial (ISRCTN64428072) registration: https://www.isrctn.com/ISRCTN64428072.
Keywords
Introduction
Major depressive disorder (MDD) in the 21st century poses a significant disease burden with considerable associated mortality (Ferrari et al., 2013). The pharmacological treatment of MDD with currently available antidepressant drugs is characterized by considerable ineffectiveness (Cipriani et al., 2018). Real-world data from the STAR*D trial, and data across clinical trials suggest that only ~30–40% of patients achieve remission (Fava et al., 2003; Gaynes et al., 2009; Rush et al., 2006). However, remission rates may be even lower, as indicated by Pigott (2015). Moreover, while the benefits of antidepressant treatment may be detectable after 1 week of treatment (Taylor et al., 2006), clinically relevant improvements often take longer to occur (Kojic et al., 2022). Patients also often experience burdensome adverse effects and/or residual depressive symptoms. Drug developers and clinicians are currently endeavouring to develop novel, rapid-acting antidepressant medications that are superior to conventional ones in terms of efficacy, temporal trajectory, and side effect profile.
With the resurgence in psychedelic research over the last two decades, compounds such as psilocybin, N,N-dimethyltryptamine (DMT) have shown an ability to rapidly induce long-term remission in MDD after a limited number of administrations in preliminary clinical trials (Goodwin et al., 2022; Raison et al., 2023; von Rotz et al., 2023). The 5-hydroxytryptamine 2A (5-HT2A) receptor plays a central role in mediating the psychedelic effects of these agents in humans, as demonstrated by the correlation of 5-HT2A receptor occupancy with the subjective effects of psilocybin and the ability of the selective 5-HT2A receptor antagonist ketanserin to block and reverse these subjective effects (Holze et al., 2024; Quednow et al., 2012). Psychedelics also appear to show long-lasting therapeutic effects in mood disorders that endure even after the compound has been cleared, potentially mediated in part by increased plasticity in key circuits relevant to these indications (Inserra et al., 2021; Nichols, 2016; van Elk and Yaden, 2022). Since psychedelics share activation of 5-HT2A receptors as a common mechanism-of-action (MoA), they differentiate mechanistically from conventional antidepressants (van Elk and Yaden, 2022), presenting a novel MoA in the rapid treatment of MDD.
GM-2505 (N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methy-lethanamine; bretisilocin) is a novel, 5-HT2A receptor agonist/5-HT releaser under development for the treatment of MDD. Based on its preclinical profile, GM-2505 is expected to produce psychedelic effects, similar to other 5-HT2A agonists, including changes in perception, emotion, and cognition with a duration of pharmacodynamic (PD) effect shorter than psilocybin and lysergic acid diethylamide (LSD), but longer than DMT. The in vitro and in vivo pharmacology of GM-2505 has been extensively profiled (Supplemental Section 1) and support its primary activity as a 5-HT2A receptor agonist while it also causes 5-HT release and has agonist activity at 5-HT2C receptors and is an antagonist at 5-HT2B receptors with similar potency. The in vivo plasma t1/2 was ~45 minutes following intravenous (IV) administration, consistent with a duration of psychedelic-like effects of ~90 minutes.
Taken together, GM-2505 is a novel, 5-HT2A receptor agonist expected to induce consciousness-altering effects similar to DMT, psilocybin, or LSD. The expected shortened half-life is anticipated to enhance the convenience and scalability of supervised, in-clinic sessions compared to longer-acting agents such as psilocybin and LSD. This shortened half-life maximizes the degree of target engagement during a session of reasonable duration, which may improve both the response and durability of the treatment compared to shorter-acting agents such as DMT. The current study evaluated for the first time, the safety, pharmacokinetics (PK), and PD of single-ascending IV doses of GM-2505 in healthy volunteers.
Methods
This was a single-ascending dose (SAD), randomized, placebo-controlled, double-blind, safety, tolerability, PK, and PD study. This trial (ISRCTN64428072; European Union Drug Regulating Authorities Clinical Trials Number, 2022-003014-37) was conducted at the Centre for Human Drug Research (Leiden, the Netherlands). The study was approved by the Medical Ethics Committee of Stichting Beoordeling Ethiek Biomedisch Onderzoek (Assen, the Netherlands) and was conducted according to the Dutch Act on Medical Research Involving Human Subjects (WMO) and in compliance with all International Conference on Harmonisation-Good Clinical Practice guidelines and the Declaration of Helsinki.
Study participants
Six cohorts of eight healthy male and female participants (aged 18–55 years), with a self-report of at least one prior psychedelic drug experience in the last 5 years were enrolled in this study. Participants with current or previous hypertension, cardiovascular disease, risk factors for seizures, suicidality, substance abuse, DSM-5 psychiatric disorders (including a family history of psychotic disorder in first- or second-degree relatives), or persistent negative psychological effects following past use of psychedelics were excluded. Each participant provided written informed consent before any screening procedures were performed.
Randomization and study procedures
Eligible participants were randomly assigned to GM-2505 or placebo (randomization ratio of 3:1 in each cohort). All participants underwent medical screening and training for the PD assessments 21–22 days before dosing. Subsequently, eligible participants were invited for an ~1 hour preparation session with their psychedelic guide and were given a tour of the dosing room. Dosing rooms were modified to accommodate the participants’ perceptual changes and increase feelings of safety and comfort, in line with safety guidelines for psychedelics (Johnson et al., 2008). Participants were at the clinical unit between days 1 and 2, with dosing on the morning of day 1. The majority of safety, PK, and PD measurements were carried out on day 1. One psychedelic guide, one nurse, and one research assistant were present during study drug administration. Prior to discharge, participants were examined by the study physician, ensuring physical and psychological safety, and were debriefed by the psychedelic guide. After discharge, participants were followed up for safety assessments for up to 28 days.
Study drug and dose rationale
All study treatments were prepared at the GMP Clinical Trial Pharmacy of Leiden University Medical Center (Leiden, The Netherlands). Placebo syringes were identical in appearance to those containing GM-2505 and were filled with saline.
Single IV infusions of GM-2505 were administered over 10 minutes, followed by a 5 minutes saline flush of the infusion line. A starting dose of GM-2505 0.34 mg was anticipated to be 33- and 100-fold below the half-maximal effect dose ED50 in the rat head twitch response assay (HTR) and plasma exposure at the no observed adverse effect level (NOAEL) dose in the sensitive species (NOAEL(beagle dog plasma) = 432.5 ng/mL), respectively. The maximal dose was set to not exceed a peak plasma exposure (Cmax) of 87 ng/mL (i.e. one-fifth of the NOAEL Cmax plasma exposure). Dose escalation decisions after each cohort were based on review of all available blinded safety, PD, and PK data.
GM-2505 pharmacokinetics
Blood samples were taken 15 minutes pre-dose, and at 10, 20, and 40minutes, and 1, 1.5, 2, 3, 4, 6, 12, and 24 hours post-dose for plasma PK analysis. Urine was collected in 0–6, 6–8, 8–12, and 12–24 hours intervals. The overview of PK endpoints can be found in Supplemental Section 2.
Safety evaluations
Safety assessments consisted of adverse events (AEs), clinical laboratory tests, vital signs, 12-lead electrocardiography (ECG), the Brief Psychiatric Rating Scale (BPRS), Columbia Suicide Severity Rating Scale (C-SSRS), Hunter’s Serotonin Toxicity criteria, and continuous electroencephalography (EEG). The safety evaluations are described in detail in Supplemental Section 3. In summary, AEs were reported from providing informed consent until the follow-up visit. Clinical laboratory values were assessed at screening, upon admission to the clinical unit, prior to discharge, and during the follow-up visit. Vital signs, including systolic and diastolic blood pressure (SBP and DBP), pulse rate, and temperature, were assessed, and ECG was performed upon admission, 5 minutes pre-dose, and 6 and 40 minutes, 1, 2, 4, 8, and 24 hours post-dose. The BPRS and C-SSRS were used to assess emergence of psychotic symptoms and suicidality, respectively, and were assessed at screening, upon admission to the clinical unit, prior to discharge, and at the follow-up visit. Continuous recordings of EEG from 4 minutes pre-dose to 40 minutes post-dose were used to assess potential epileptiform abnormalities. Study stopping criteria for dose escalation included a serious AE and prolonged SBP and DBP increases >160 and >120 mmHg, respectively.
PD assessments
PD assessments included various subjective and objective measures. Visual analogue scales (VAS), the NeuroCart test battery (Groeneveld et al., 2016), and cognitive and reward processing tasks are described in Supplemental Section 4. The remaining PD assessments are described below.
Real-Time Intensity
The Real-Time Intensity (RTI) scale is a rater-based scale assessing the subjective intensity of psychedelic experiences (Timmermann et al., 2019). Participants were asked to verbally rate the current intensity of their experience on a scale from 0–10 (0 = not at all; 10 = extremely) on visual, bodily, and emotional dimensions. The scores on all three dimensions were also averaged to obtain a total RTI score. RTI measurements were obtained on Day 1, 15 minutes pre-dose, and 10, 20, and 40 minutes, 1, 1.5, 2, 3, 4, 6, 12, and 24 hours post-dose.
Altered States of Consciousness Rating Scale
The Altered States of Consciousness Rating Scale (ASC) is a 94-item retrospective VAS to assess altered states of consciousness induced by various psychedelic drugs including alterations in mood, perception, experience of self, and thought disorder (Hasler et al., 2004; Kraehenmann et al., 2017a, 2017b; Liechti, 2017; Mathai et al., 2020; Pokorny et al., 2016; Quednow et al., 2012; Schmid and Liechti, 2018). The ASC was administered
Mystical Experiences Questionnaire 30
The Mystical Experiences Questionnaire 30 (MEQ30) is a 30-item scale to assess a range of mystical or spiritual-like effects and was administered
Neuroendocrine hormones
Serum adrenocorticotropic hormone (ACTH), cortisol, prolactin, and oxytocin levels were measured to assess neuroendocrine effects by collecting blood samples at 15 minutes pre-dose, and 10 and 40 minutes, 1.5, 4, 6, and 24 hours post-dose.
Resting-state EEG
The resting-state electroencephalography (rsEEG) was continuously recorded pre-dose, and at 30 minutes, 1, 1.5, 3, 4.5, 6, and 24 hours post-dose using a 40-channel recording system (Refa-40; TMSi B.V., Oldenzaal, the Netherlands). The acquisition and analysis methods are described in Supplemental Section 4. Power values were averaged across canonical rsEEG bands including delta (1–4 Hz), theta (4–8 Hz), alpha (8–13 Hz), beta (13–25 Hz), slow gamma (30–50 Hz), and fast gamma (65–95 Hz) to obtain band-specific spectral averages. Finally, rsEEG power values were averaged across all electrodes to obtain single values of rsEEG band-specific power per participant, timepoint, band, and eye state (open or closed).
Statistical analysis
Safety and PK parameters were summarized by intervention and dose groups. Categorical variables (e.g. AEs) were summarized by counts and percentages, while continuous variables (e.g. ECG, BP, and PK parameters) were summarized by descriptive statistics, including means, change from baseline (CFB, where baseline was considered the last measurement before dosing for safety and PK, and the mean of the two pre-dose assessments for PD, where applicable), standard deviation (SD), and coefficients of variation. PK endpoints were derived by non-compartmental analysis of the plasma concentration-time data, using the PKNCA (Denney et al., 2015) package in R V4.0.3 for Windows (R Foundation for Statistical Computing, Vienna, Austria, 2010; R Core Team, 2023).
Repeatedly measured PD data were evaluated with mixed model analyses of covariance with treatment, time, and treatment by time as fixed effects, with participant as random effect, and with the (mean) baseline value as covariate. Least square means estimates over time by treatment were presented with 95% complex imagery (CI). Treatment effects were analysed as the contrasts between placebo and the different dose levels of GM-2505 for each PD endpoint, and these were reported along with 95% CI. This exploratory trial was not prospectively designed for formal hypothesis testing with statistical power; therefore, presented P-values (α = 0.05) for each comparison should be considered nominal. No corrections for multiple comparisons were made. Variables were log-transformed before analysis as needed. All PD calculations except those for EEG were performed using SAS for Windows V9.4 (SAS Institute, Inc., Cary, NC, USA; SAS Institute Inc., 2014). EEG data were evaluated in a post-hoc analysis using a linear mixed-effects model with repeated measures. The analysis was conducted using MATLAB’s “fitlme” function. The model included fixed effects for dose, time, and their interaction, as well as random intercepts for subjects to account for intra-subject variability. Each dose’s effect was tested against placebo at each time point using specific contrasts.
Results
Summary of GM-2505 preclinical characterization
The in vitro and preclinical in vivo pharmacology of GM-2505, with supporting methodology, are described in Supplemental Section 1. In brief, GM-2505 was a potent 5-HT2A and 5-hydroxytryptamine 2C (5-HT2C) receptor agonist in Ca2+ flux assays, with functional selectivity versus 5-hydroxytryptamine 2B (5-HT2B; i.e. no agonist activity detected at 10 mM) and 5-hydroxytryptamine 1A (5-HT1A) receptors (EC50 17 μM (microMolar rather than milliMolar). Obtained EC50 values and the % activation at max concentration were: 5-HT2A (17 nM, 82%); 5-HT2C (9.5 nM, 85%); 5-HT2B (>10,000; 0.8%); 5-HT1A (16,918 nM, 83%). GM-2505 increased the release of 5-HT from rat synaptosomes with an EC50 value of 12.9 nM, in a similar fashion to DMT (EC50 84.7 nM) in the same assay. In C57BL/6 mice, GM-2505 produced dose-dependent induction of the HTR with an ED50 of 0.3 mg/kg, subcutaneous (95%, CI: 0.21–0.41).
Participant characteristics and disposition
Forty-eight participants met eligibility criteria and were randomized to six cohorts of eight individuals. GM-2505 was administered to participants as SADs of 0.34 mg (n = 4), 1 mg (n = 6), 3.3 mg (n = 6), 10 mg (n = 6), 15 mg (n = 6), or 20 mg (n = 6) and a total of 14 participants received placebo (in total, 34 GM-2505 and 14 placebo). Two participants in the placebo group were excluded from PD analyses as they did not complete all measurements. Participant demographics and baseline characteristics are found in Table 1. A CONSORT diagram is presented in Supplemental Section 4.6.
Participant characteristics and baseline physiological measures.
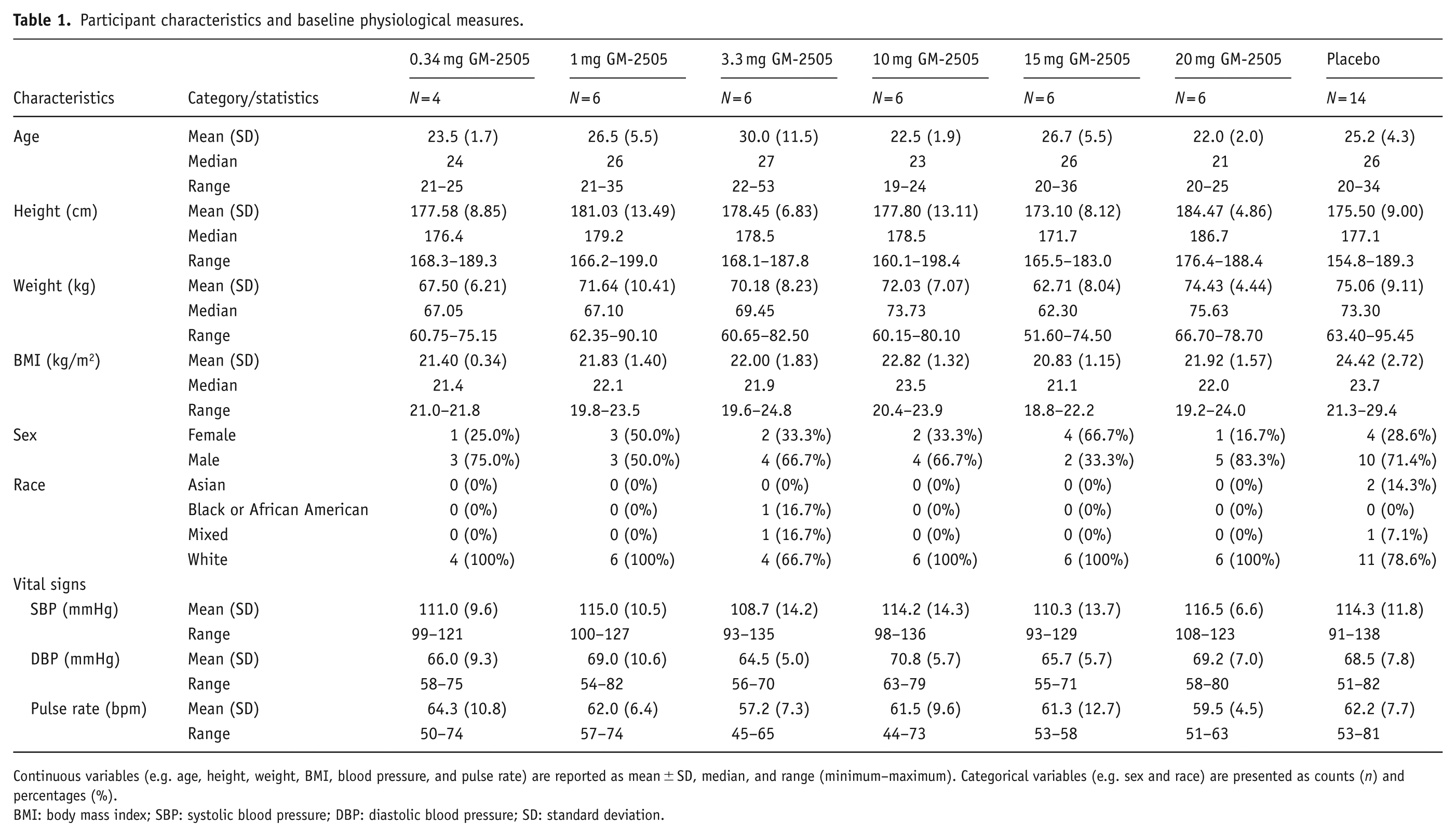
Continuous variables (e.g. age, height, weight, BMI, blood pressure, and pulse rate) are reported as mean ± SD, median, and range (minimum–maximum). Categorical variables (e.g. sex and race) are presented as counts (n) and percentages (%).
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; SD: standard deviation.
Pharmacokinetics
GM-2505 Cmax and AUClast increased dose proportionally, although the increase seemed to be more than dose proportional for the dose levels of 10–20 mg as compared to 1 and 3.3 mg (see Figure 1). Median time to maximum plasma concentration (Tmax) was 10–20 minutes (Figure 1), consistent with the duration of the infusion and subsequent line wash. Mean terminal half-life (t1/2) was 40–50 minutes (Figure 1). An overview of all PK parameters is provided in Supplemental Section 2.
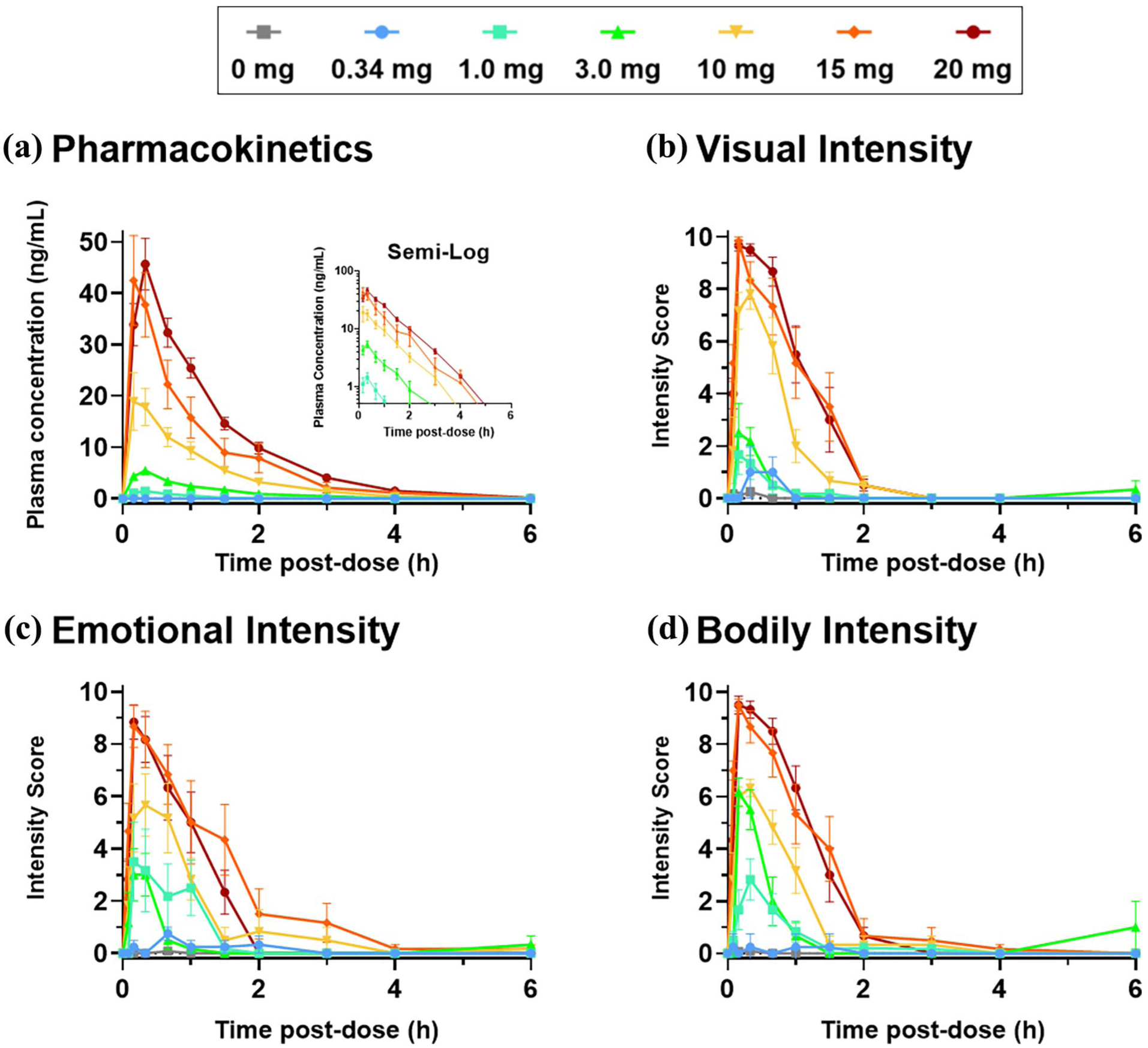
Plasma concentration-time profiles and subjective effects reported in real time following IV administration of GM-2505. (a) Mean (± SEM) plasma GM-2505 linear concentration-time profile following a single 10 minutes infusion of GM-2505 at doses of 0.34, 1.0, 3.3, 10, 15, or 20 mg of GM-2505 in healthy volunteers. Each data point represents the mean (+ SEM) value from 6 participants with the exception of four participants receiving 0.34 mg dose, which also was limited by most time points for most subjects being below the lower limit of detection. Inset shows the same GM-2505 concentration-time data on a semi-log plot. (b) Subjective effects of GM-2505 single infusions of 0.34, 1.0, 3.3, 10, 15, and 20 mg over time reported by healthy volunteers following dosing at time -0- on Visual Intensity items (mean + SEM) in RTI scale. (c, d) Effects on Emotional Intensity and Bodily Intensity items of the RTI scale, respectively, reported by subjects in the same format as B. RTI plots represent mean (+ SEM) for placebo (n = 12) and 6 participants in each dose group with the exception of the 0.34 mg dose (n = 4).
Safety assessments
Adverse events
All AEs were self-limiting, and the majority were mild (93.1%) to moderate (5.8%) in intensity. No serious AEs occurred, and no AEs led to study discontinuation or required psychiatric intervention. AEs generally emerged several minutes after starting the infusion, with psychedelic-related AEs resolving by ~2 hours post-dose (though lasting up to 4–6 hours in several subjects). A limited number of non-psychedelic-related AEs (e.g. fatigue and headache) lasted up to 24 hours post-dose. Across GM-2505 doses, the most frequently reported AEs were sensory processing disorder, altered state of consciousness, feeling of body temperature change, and feeling of relaxation (see Table 2). One participant experienced irregular menstruation following treatment with 15 mg GM-2505, considered possibly related to GM-2505, and required a subsequent referral to a gynaecologist for a diagnostic workup. A summary of AEs (with incidence rate >5%) can be found in Table 1, and all AEs are listed in Supplemental Section 7.
Summary of treatment-emergent adverse effects.
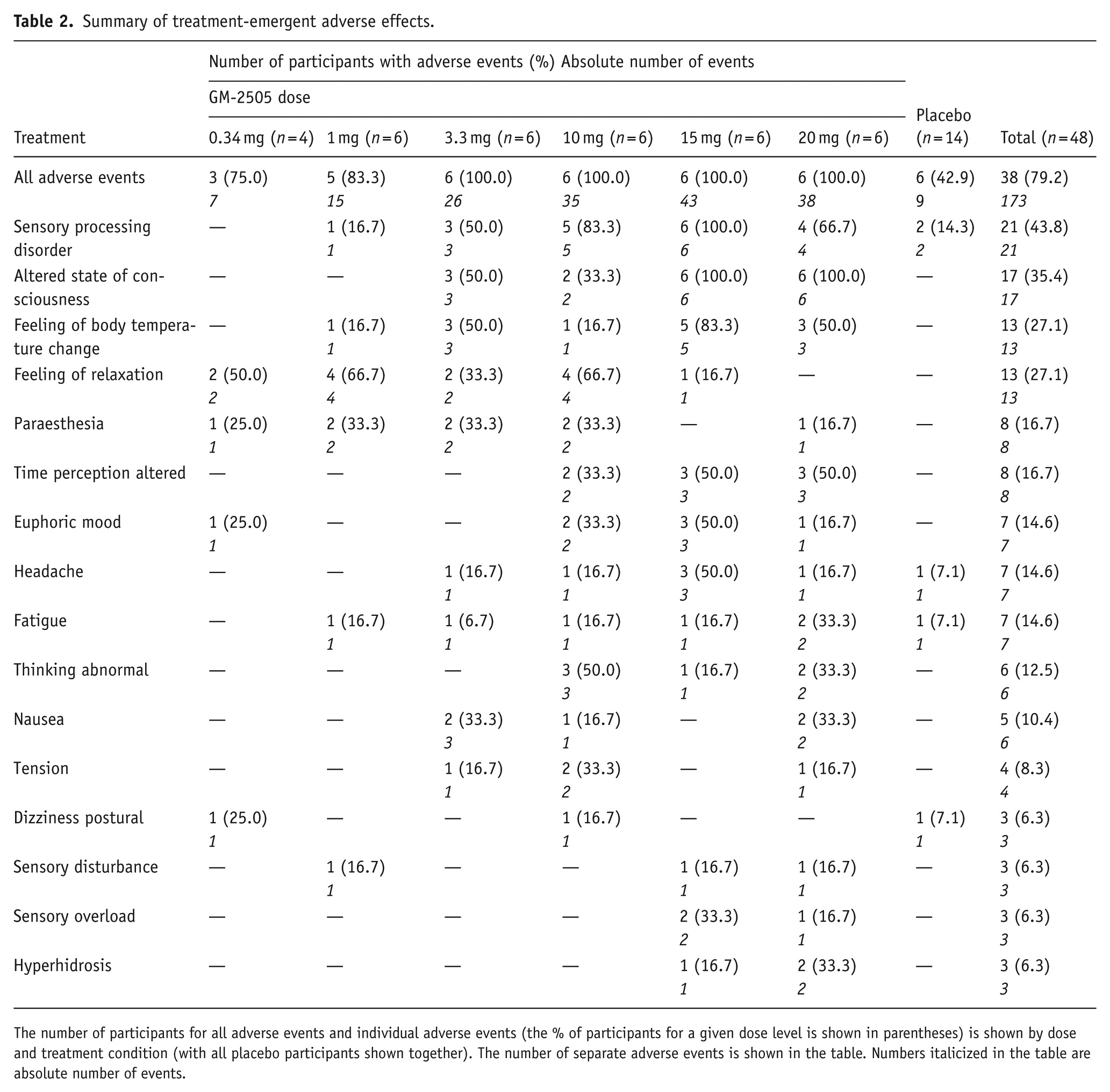
The number of participants for all adverse events and individual adverse events (the % of participants for a given dose level is shown in parentheses) is shown by dose and treatment condition (with all placebo participants shown together). The number of separate adverse events is shown in the table. Numbers italicized in the table are absolute number of events.
Vital signs
Compared with placebo, GM-2505 increased mean SBP, DBP, and pulse rate at doses ⩾1 mg. Vital sign measurements, including the largest absolute values and changes from baseline for SBP, DBP, and pulse rate, are summarized in Table 3. The largest increases were observed at 6 minutes post-dose for all dose levels. All increases were self-limiting and were generally returned to baseline within 2 hours post-dose. GM-2505 10 mg was associated with the largest, albeit still moderate increase of 27.8 mmHg, 19 mmHg, and 35.3 bpm for SBP, DBP and pulse at 6 minutes post-dose, respectively. These increases corresponded to absolute mean SBP, DBP and pulse of 142 mmHg, 89.8 mmHg, and 96.8 bpm, respectively. An overview over the 24 hours period of vital sign assessments can be found in Supplemental Section 6.
Vital sign values and changes from baseline across GM-2505 dose levels.
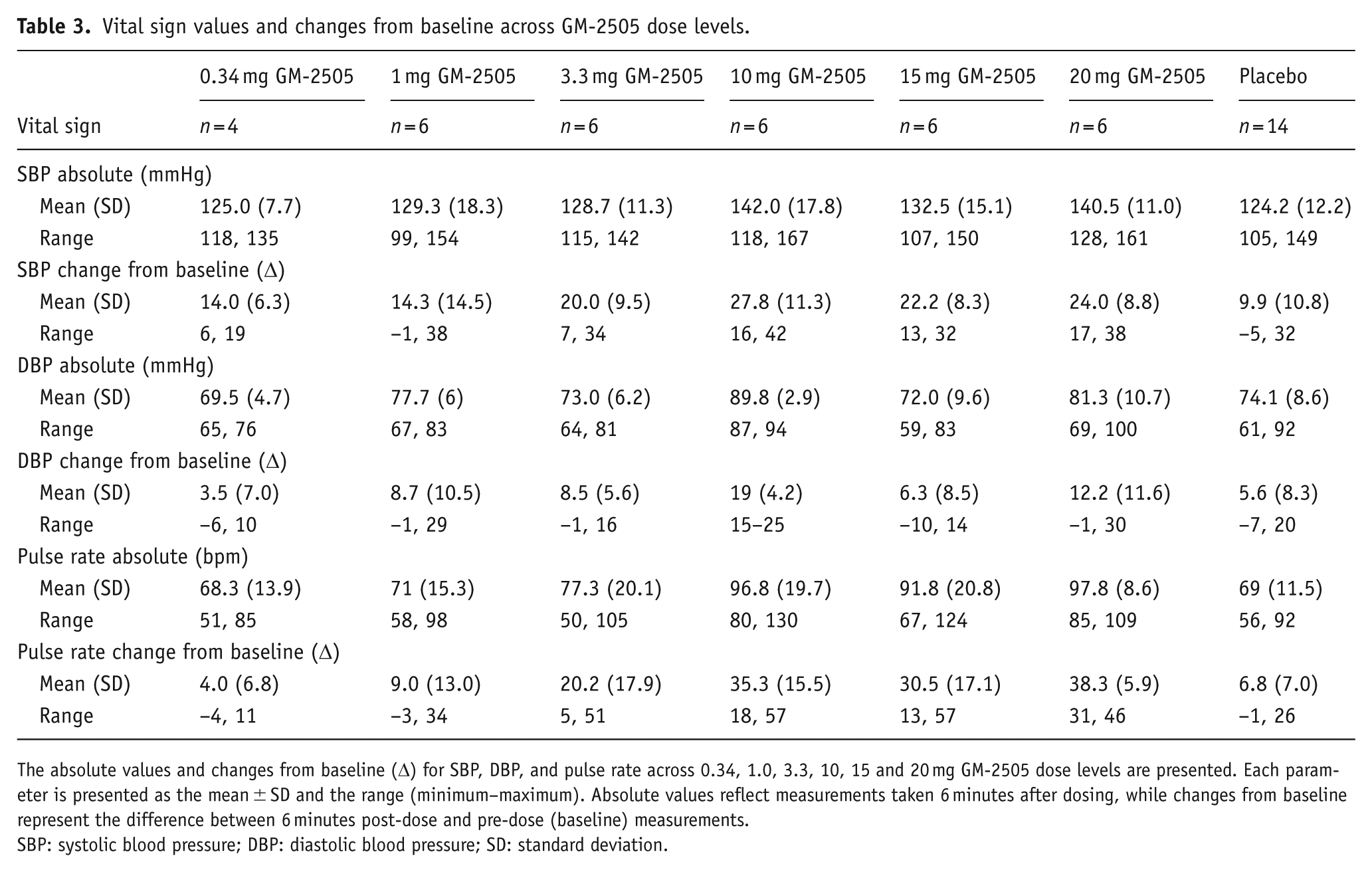
The absolute values and changes from baseline (Δ) for SBP, DBP, and pulse rate across 0.34, 1.0, 3.3, 10, 15 and 20 mg GM-2505 dose levels are presented. Each parameter is presented as the mean ± SD and the range (minimum–maximum). Absolute values reflect measurements taken 6 minutes after dosing, while changes from baseline represent the difference between 6 minutes post-dose and pre-dose (baseline) measurements.
SBP: systolic blood pressure; DBP: diastolic blood pressure; SD: standard deviation.
Electrocardiography
No clinically relevant ECG findings were observed. Limited, transient QTcF increases were reported for GM-2505 and placebo, which were generally largest 30 minutes post-dose and resolved between 1 and 4 hours post-dose for GM-2505 ⩽15 mg, and by 8 hours for GM-2505 20 mg. The highest mean QTcF was 421.2 mseconds, 30 minutes post-dose, for GM-2505 20 mg. There was no individual QTcF CFB >60 mseconds, and no absolute QTcF values were >500 mseconds for any participant.
Other safety assessments
No clinically relevant findings were noted in laboratory values, safety-EEG, C-SSRS, or BPRS.
PD effects
Real-Time Intensity
RTI increased with higher doses up to 15 mg, after which it reached a plateau at or near the scale maximum in most subjects (see Figure 1). No substantial differences were observed in the relative increases of the total, visual, bodily, and emotional dimensions of the RTI between different dose levels. All RTI scores for all dose levels returned to baseline ~2–3 hours post-dose.
Altered States of Consciousness Rating Scale
Mean total ASC scores tended to increase with rising doses up to 15 mg, after which these reached a plateau (see Figure 2). The 5-Dimensional Altered States of Consciousness Scale (5D-ASC), dread of ego dissolution dimension score showed the largest increase at the 20 mg dose, the other 5D-ASC sub-scores appeared to reach maximal scores between 10 and 15 mg (Figure 2). Most 11D-ASC sub-scores appeared highest for GM-2505 15 mg (Figure 2), except impaired control of cognition and anxiety, which appeared highest for GM-2505 20 mg. Overall, the data suggest a possible inflection point between 3.3 and 10 mg doses, which may represent the threshold for noticeable psychedelic effects.
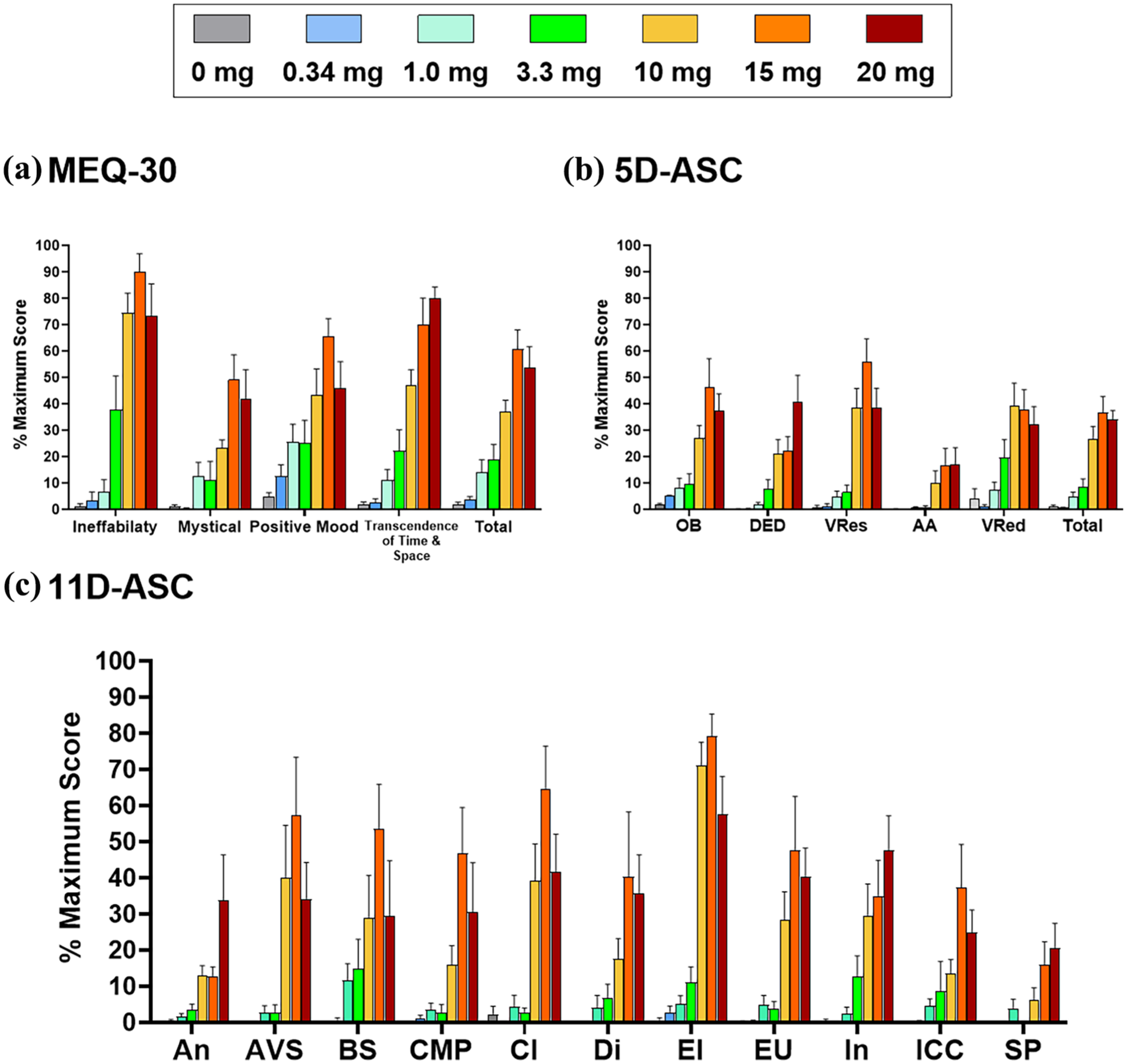
Characteristics of psychedelic experience indicated by the 5D-ASC and MEQ reported retrospectively by participants following IV dosing of escalating doses of GM-2505 (mean
Mystical Experience Questionnaire 30
All MEQ scores appeared larger than scores in the placebo group at all dose levels ⩾1 mg (Figure 2). MEQ mean score and sub-scores tended to increase dose dependently and reached maximal values between 10 and 20 mg.
VAS “feel drug.”
Compared to placebo, GM-2505 significantly increased mean VAS “feel drug” by 0.42, 0.33, 0.98, 0.72, and 0.91 log (mm) at 1, 3.3, 10, 15, and 20 mg, respectively (p < 0.01).
Neuroendocrine hormones
Compared to placebo, GM-2505 significantly increased mean ACTH (Figure 3) at doses ⩾3.3 and ⩾10 mg, respectively (cortisol data not shown). ACTH and cortisol peak effects were observed at 10 and 40 minutes post-dose, respectively (see Figure 3). Prolactin increased at doses ⩾10 mg (Figure 3), with peak effects occurring at ~40 minutes post-dose, while no effects were observed on oxytocin (data not shown).
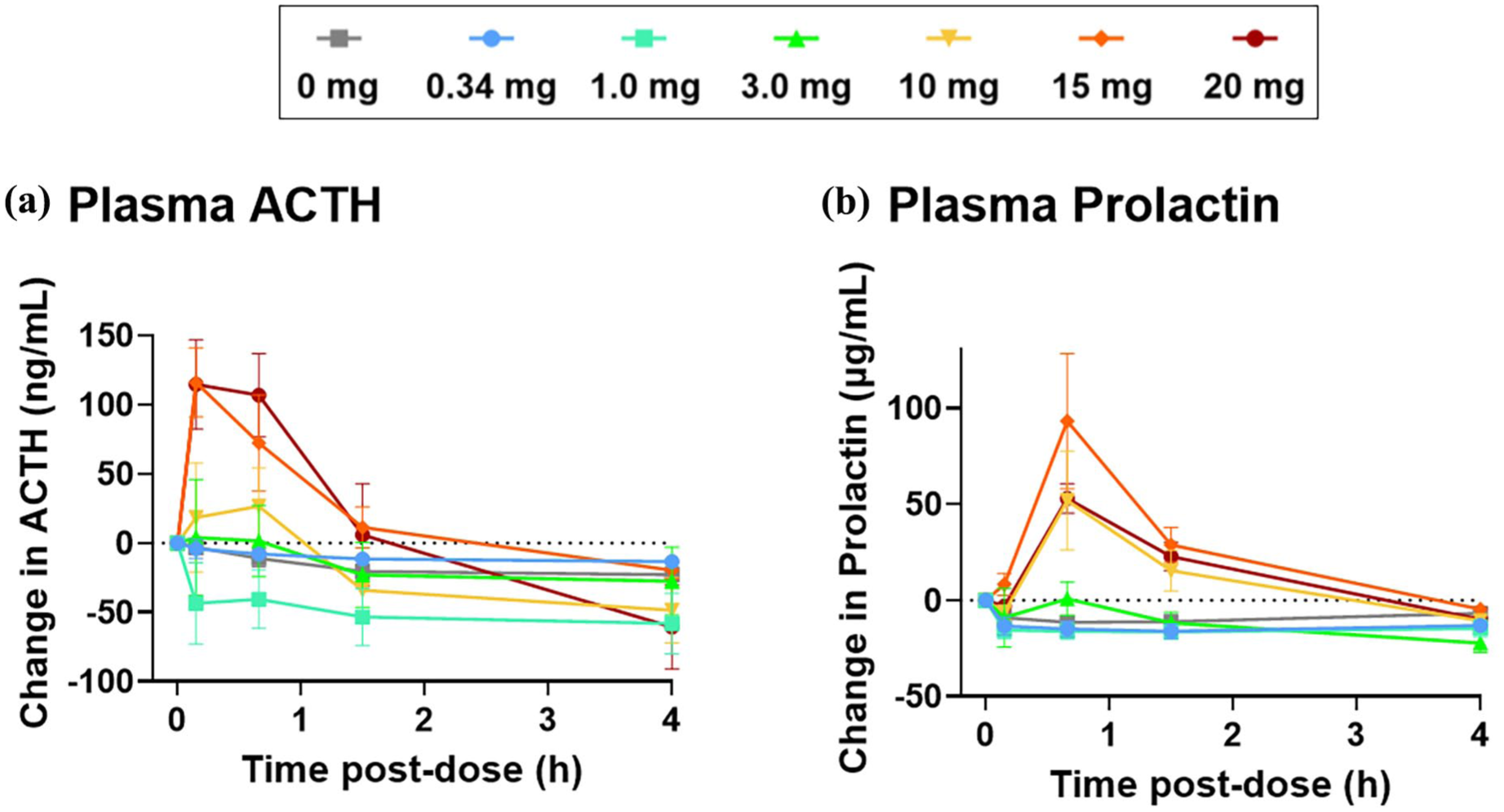
Neuroendocrine effects following IV administration of GM-2505. (a) The acute effects on ACTH levels for participants administered an IV infusion of either placebo (n = 12) or GM-2505 at doses of 0.34, 1.0, 3.3, 10, 15, and 20 mg with six participants in each dose group with the exception of the 0.34 mg dose (n = 4). Compared to placebo, GM-2505 significantly increased mean ACTH at GM-2505 10 mg (p = 0.007), 15 mg (p < 0.001), and 20 mg (p < 0.001) with peak effects observed at 10 minutes post-dose and lasting up to ~4 hours post-dose (mean
Resting-state EEG
Dose-dependent reductions in theta and alpha power and increases in slow and fast gamma power were observed 30 minutes post-dose (see Figure 4). Compared to placebo, significant theta power reductions occurred at all doses ⩾1 mg, with eyes closed, starting at 30 minutes and lasted up to 3 hours post-dose. Eyes closed alpha power reductions were limited to doses ⩾10 mg and lasted up to 1 hour post-dose. Gamma power, especially in the fast gamma band, was increased compared to placebo starting at 30 minutes with doses ⩾3.3 mg and lasting up to 90 minutes post-dose. Power changes during the eyes open condition followed comparable patterns, although the magnitude of these effects was somewhat reduced compared to the eyes closed condition (data not shown). No changes were seen in the other frequency bands.
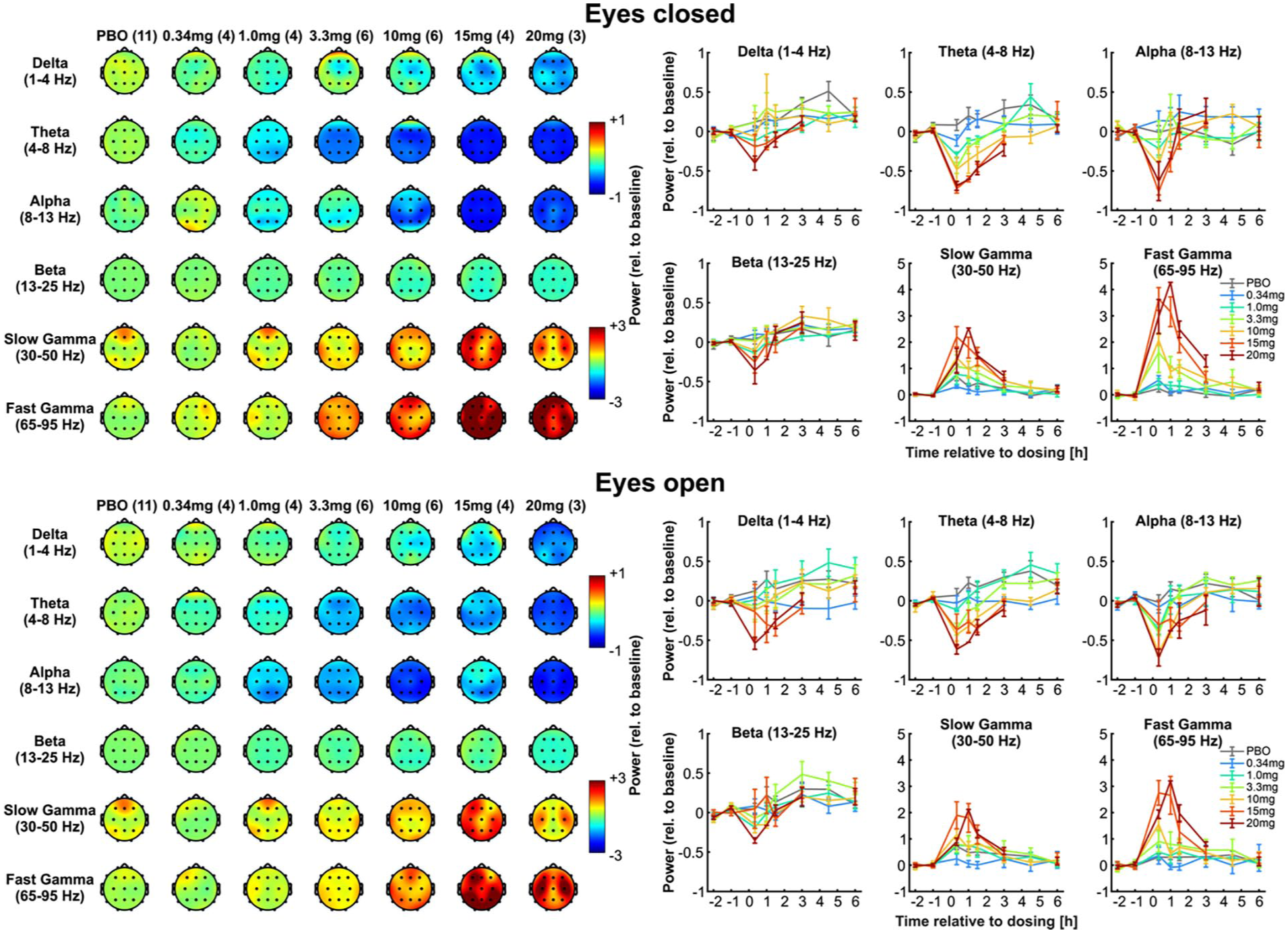
Resting-state EEG findings following IV administration of GM-2505. The left panels show tomographs with placebo (n = 14) and the GM-2505 IV infusion dose level followed in parentheses for the number of subjects in the EEG analysis. Rows of the tomograph displays are the EEG frequency bands (delta, theta, alpha, beta, slow gamma, fast gamma). Columns show escalating GM-2505 dose levels. The upper left panel shows these effects for the eyes closed condition, and the bottom left panel shows the data for the eyes open condition. The right panels show the EEG changes normalized to baseline (scale on left) at each timepoint following dosing of GM-2505 for delta, theta and alpha bands (columns) for Eyes closed (upper right panel) and Eyes open (lower right panel) conditions (mean
NeuroCart
Compared to placebo, GM-2505 produced significant dose-dependent increases in saccadic peak velocity (SPV) at doses ⩾3.3 mg lasting until ~26 hours post-dose (Figure S2). Respective left and right pupil/iris ratio significantly increased at GM-2505 ⩾10 mg, with maximal effects occurring at ~50 minutes post dose. No significant effects were observed on saccadic reaction time and postural stability. The results of the remaining PD endpoints are included in Supplemental Section 9.
Missing data
Since the RTI and ASC predominantly demonstrated a score of 0 for placebo, only descriptive statistics are presented in summary tables and figures. Moreover, due to the intense altered state of consciousness at the higher doses, several PD measures could not be collected at 30–60 minutes post-dose (see Supplemental Section 8), including rsEEG assessments, where 33% and 50% of observations were missing, 30 minutes post-dose, for 15 and 20 mg, respectively.
Discussion
The present first-in-human, SAD study characterized the safety, PK, and PD profile of the novel, highly potent 5-HT2A receptor agonist, GM-2505, in healthy participants. In general, GM-2505 exposures increased dose-dependently, as did PD endpoints, including rsEEG effects, changes in neuroendocrine hormones, and SBP increases, as well as real-time (i.e. RTI scale) and retrospective (i.e. 11D-ASC and MEQ-30) measures of subjective effects. Overall, the clear psychedelic effects induced by GM-2505, particularly for doses of 10–20 mg, were consistent with findings observed for other 5-HT2A receptor agonists, as described below.
GM-2505 administered IV up to 20 mg demonstrated an acceptable safety profile consisting of mild transient AEs, short-term increases in blood pressure and pulse rate that were self-limiting and not clinically relevant, and no clinically significant changes on ECG. The most common AEs included sensory processing disorder, altered state of consciousness, abnormal thinking, euphoric mood, and fatigue, representing a profile consistent with 5-HT2A receptor agonists (Hinkle et al., 2024). Further, the magnitude of cardiovascular effects was consistent with previously reported findings with IV DMT (Vogt et al., 2023). However, in line with its PK profile, the duration of GM-2505’s cardiovascular effects was shorter than that of psilocybin but longer than that of DMT (Goodwin et al., 2022; Holze et al., 2022; Vogt et al., 2023), demonstrating a more practical temporal profile compared to relatively longer-acting 5-HT2A receptor agonists such as psilocybin and LSD.
Subjective PD effects emerged at doses of 1 mg and/or 3.3 mg and increased substantially at 10 mg. Specifically, the RTI visual score demonstrated dose-dependent psychedelic effects peaking at 15 mg. These RTI effects largely dissipated within 2 hours even for the highest doses, consistent with GM-2505’s half-life of ~45 minutes. Increases on the 11D-ASC and the 5D-ASC were dose-dependent, with maximal effects occurring at 15 or 20 mg GM-2505. Although the magnitude of these psychedelic effects at 15–20 mg was comparable to those reported for 30 mg psilocybin or 100–200 µg LSD in healthy volunteers (Holze et al., 2022), participants reported the 20 mg dose to be particularly intense. Consistent with these reports, 5D-ASC dimensions related to pleasant subjective psychedelic experiences, such as “oceanic boundlessness” and “visionary restructuralization,” were typically maximal at 15 mg, while 20 mg seemed to be associated with increased challenging psychedelic effects, such as “anxiety” and “impaired control of cognition” on the 11D-ASC and “dread of ego dissolution” on the 5D-ASC scale. Together, therefore, participant narratives, subjective drug effects as reflected by the VAS scales and ASC scores, in combination with the AE profiles over the 10–20 mg dose range, seem to suggest superior tolerability of the 10–15 mg dose range compared with 20 mg (Vogt et al., 2023). Lastly, mean GM-2505 exposures correlated, analysed retrospectively, with mean total ASC score (r = 0.87) across dose groups, further suggesting that subjective alterations in consciousness were exposure dependent. Dose-related psychedelic effects were also observed for the MEQ-30 total score and the individual factors, with transcendence of time and space showing the most graded dose–response. Similar to the other subjective measures, the magnitude of effects on the 5/11D-ASC, MEQ-30, for 15–20 mg GM-2505 appeared similar to or greater than that reported for 30 mg psilocybin or 100–200 µg of LSD in healthy volunteers (Holze et al., 2022). In contrast, effects for 10 mg of GM-2505 were lower than those reported for the same doses of psilocybin or LSD (Holze et al., 2022). Taken together, these findings confirm that GM-2505 elicits psychedelic effects similar to existing 5-HT2A receptor agonists, with a dose between 10 and 15 mg appearing to represent an optimal balance between a substantial psychedelic effect and limited potential unpleasant subjective effects. Taken together, while similarities appear to exist between higher doses of GM-2505 (10–20 mg) and those of psilocybin, LSD, and DMT at comparable levels of psychedelic effect, further research in both healthy volunteers and patient populations is needed to confirm these apparent parallels.
Generally, dose-related effects were also observed for neuroendocrine hormones, several neurophysiological and neuropsychological measures (SPV and pupillary dilation), and rsEEG. Interestingly, since SPV is considered a central nervous system (CNS) arousal-related biomarker that has been shown to be sensitive to effects of CNS stimulants such as an alpha-amino-3-hydroxy-5-methyl-4-isoxazole (AMPA) receptor positive allosteric modulator (Dijkstra et al., 2022), and dexamphetamine (Recourt et al., 2020), increases in SPV and increased ACTH are indicative of hypothalamus-pituitary-adrenal (HPA) axis activation, and seem to indicate stimulatory CNS effects. HPA axis activation has been associated with 5-HT2A receptor agonists, like LSD (Schmid et al., 2015), psilocybin (Hasler et al., 2004), and IV DMT (Strassman and Qualls, 1994). However, GM-2505 was not associated, on average, with altered levels of mood and calmness (as illustrated by VAS “calmness” and “mood”), in contrast with the primarily CNS stimulant, dexamphetamine (Recourt et al., 2020). However, further dose work in both healthy volunteers and patient populations is needed to understand the reliability of these effects.
Apparent dose-dependent effects were observed on rsEEG, with rsEEG power being attenuated in the low frequency bands including theta and alpha, while conversely being increased in the high frequency bands (i.e. slow and fast gamma). Spectral effects in the delta and beta rsEEG bands were limited. These results appear in line with previous human, rsEEG findings with other 5-HT2A agonists including psilocybin (Muthukumaraswamy et al., 2013) and DMT (Timmermann et al., 2023). Importantly, this study demonstrated the robustness of this biomarker as apparent dose-dependence was observed over the dose range evaluated, from doses that were non-psychedelic to those that produced a maximal psychedelic experience. Furthermore, the sensitivity is also apparent in that low doses (1 and 3.3 mg GM-2505), which had minimal subjective effects, produced detectable changes in theta rsEEG power. This approach provides a framework for thorough evaluation of the neurodynamic effects of psychedelic agents. These rsEEG effects are the first instance to our knowledge where the rsEEG dose–response has been explored over a nearly 60-fold dose range. Overall, these results provide robust, evidence of 5-HT2A target engagement by GM-2505.
The major strength of the current study lies in the systematic assessment of PD effects and safety measures in a placebo-controlled, ascending dose design. In that context, PD biomarkers were selected which previously demonstrated sensitivity to CNS penetrant compounds relevant to 5-HT2A agonists (Cohen et al., 2015; Groeneveld et al., 2016). These measures were repeatedly performed following drug administration, based on the observed PK profile, which allowed for a thorough understanding of PD effects in relation to drug exposure. This approach enabled demonstration of CNS penetrance, and, moreover, provided convincing indirect evidence of target engagement, characterized by the full dose/exposure effect relationship. As a result, the current study identified the optimal pharmacologically active exposure range for GM-2505 in humans.
The main limitation of the study is the relatively low sample size and large number of missing PD measurements at GM-2505 doses ⩾10 mg as a result of altered state of consciousness. For instance, 30 minutes post dose, two and three out of six participants completed the rsEEG in the 15 and 20 mg groups, respectively, at the 30 minutes time point so these results should be interpreted with caution. Furthermore, the NeuroCart assessments and VAS scales were characterized by comparable and larger amount of missing data, limiting reliability and establishment of clear dose dependence. A noteworthy exception is the RTI scale, which could be performed by all participants, highlighting the importance of including simple, rater-based, PD measures in studies with consciousness-altering compounds. On the other hand, as opposed to assessments such as SPV and VAS scales that rely on instruction, repeated inquiry into intensity of psychedelic-related effects might arguably lead to unintended underreporting by participants (Roseman et al., 2024). Nonetheless, these findings highlight the importance of integrating existing and novel methodologies that generate potential biomarkers that are not only dependent on instruction, in an attempt to guarantee reliable PD data during both the acute and post-acute phases 5-HT2A mediated psychedelic effects.
Taken together, in the present first-in-human study, single doses of GM-2505 administered IV up to 20 mg demonstrated an acceptable safety profile, robust 5-HT2A receptor-mediated psychedelic effects in an optimal dose range between 10 and 15 mg IV, and PK characteristics considered more practical for in-clinic use than classical psychedelics like psilocybin or LSD.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251378512 – Supplemental material for A novel psychedelic 5-HT2A receptor agonist GM-2505: The pharmacokinetic, safety, and pharmacodynamic profile from a randomized trial healthy volunteer
Supplemental material, sj-docx-1-jop-10.1177_02698811251378512 for A novel psychedelic 5-HT2A receptor agonist GM-2505: The pharmacokinetic, safety, and pharmacodynamic profile from a randomized trial healthy volunteer by Gerard J. Marek, Soma Makai-Bölöni, Daniel Umbricht, Edward P. Christian, Jason Winters, Dino Dvorak, Shane Raines, Zoë A. Hughes, Eric W. Austin, Adam K. Klein, William Leong, Fas J. Krol, Anne J. van der Graaf, Maria J. Juachon, Marije E. Otto, Laura G. J. M. Borghans, Gabriël Jacobs, Andrew C. Kruegel and Jonathan Sporn in Journal of Psychopharmacology
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
