Abstract
Human facial emotion recognition (FER) is an evolutionarily preserved process that influences affiliative behaviours, approach/avoidance and fight-or-flight responses in the face of detecting threat cues, thus enhancing adaptation and survival in social groups. Here, we provide a narrative literature review on how human FER is modulated by neurotransmitters and pharmacological agents, classifying the documented effects by central neurotransmitter systems. Synthesising the findings from studies involving functional neuroimaging and FER tasks, we highlight several emerging themes; for example, noradrenaline promotes an overall positive bias in FER, while serotonin, dopamine and gamma-aminobutyric acid modulate emotions relating to self-preservation. Finally, other neurotransmitters including the cholinergic and glutamatergic systems are responsible for rather non-specific pro-cognitive effects in FER. With the ongoing accumulation of evidence further characterising the individual contributions of each neurotransmitter system, we argue that a sensible next step would be the integration of experimental neuropharmacology with computational models to infer further insights into the temporal dynamics of different neurotransmitter systems modulating FER.
Introduction
Facial emotion recognition (FER), the ability to identify and categorise emotional states through static or dynamic facial expressions (Calvo and Nummenmaa, 2016), is a conserved process enabling individuals to extract information about others’ affective states, threatening or affiliative behaviours and motivation (Hoffman et al., 2007; Langford et al., 2010; Tuttle et al., 2018). Near-optimal FER can promote healthy functioning in social interactions (Murphy et al., 2022), whereas frequent misattribution or non-detection errors may impair non-verbal communication and social functioning. These impairments may be manifested in socially maladaptive behaviours such as persecutory delusions in schizophrenia (Haut and MacDonald, 2010) and emotional dysregulation in attention deficit hyperactivity disorder (Staff et al., 2021). For example, angry facial expressions could be predictive of aggressive behaviour towards us, while a fearful face may signal that an individual is afraid of a nearby stimulus perceived as hostile (e.g. cueing dominance over others in conflict situations) or communicating an imminent threat. Although social behaviours can be described along dimensions of affiliation and dominance, as outlined in interpersonal models like the circumplex model (Johnson et al., 2012), recognising emotions such as anger or fear can influence behaviours differently across these axes, potentially increasing dominant responses or eliciting affiliative behaviours depending on the context. This complex interplay between emotional cues and context means that rapid and accurate interpretation of such signals is critical in allowing us to interpret and pre-empt threat and trigger fight-or-flight responses prior to escalation of the threat. As a result, FER is an important process in social cognition with established evolutionary survival utility.
In line with its survival utility, FER processing is very rapid, lasting around 100 ms (Pizzagalli et al., 1999). In this short period of time FER encompasses a sequence of discrete processes; an implicit facial emotion perception process followed by an explicit process involving facial emotion labelling (Sauter et al., 2011). Disentangling the perception from the labelling can be challenging, as neither functional imaging nor cognitive FER labelling tasks can pinpoint the source of error in emotional mislabelling and identify whether it is due to erroneous implicit perception or erroneous interpretation.
Implicit unconscious facial emotion processes often involve the amygdala and other subcortical structures associated with belief inference including the superior temporal sulcus (STS) and the insular cortex, which discriminate salient emotional cues such as fear or anger in milliseconds, even without conscious awareness (Adolphs, 2002; LeDoux, 2000; Yoshida et al., 2010). For example, the STS has been implicated in responses elicited by basic emotions such as disgust that cues certain items as unsafe or unpleasant to eat (White and Burton, 2022). Such fast recognition enables individuals to swiftly respond to potential threats or social cues in their environment, facilitating adaptive behaviour.
Explicit FER, on the other hand, involves deliberate, conscious labelling or identification of emotions displayed on a face. This process engages higher-order cognitive systems, including the prefrontal cortex, which supports working memory, attention and linguistic labelling (Ochsner et al., 2004; Phillips et al., 2003). Explicit FER is often assessed through tasks that require individuals to categorise emotions, such as selecting a label (e.g. ‘happy’, ‘sad’) for a given facial expression, although for the FER labelling process to take place the implicit perception process must have previously occurred.
Although the cognitive neurobiology of brain areas associated with FER and the sequence thereof have been anatomically well-studied (Haxby et al., 2000; White and Burton, 2022), the exact contributions of and the interactions between the neurotransmitter pathways underpinning the different components of FER have not yet been synthesised into a meaningful conceptual framework, which will represent an important step towards building unifying cognitive models of FER. In this Review article, we will take a cognitive neuroscience approach to synthesise insights into the neurotransmitter pathways involved in human FER. The evidence that we review comes from diverse study designs, which can be broadly classified into two categories: (i) psychopharmacology studies targeting a specific neurotransmitter pathway and assessing its effect on FER using performance-based tasks and (ii) functional neuroimaging studies. To ensure better classification of the findings, we structured this article in terms of the different neurotransmitter systems implicated in FER including serotonergic, noradrenergic, serotonergic, dopaminergic, cholinergic, opiate, glutamatergic and GABAergic systems and tabulated the studies referenced in these sections in the Supplemental Appendices (1–6). Before delving into these neurotransmitter systems, we briefly summarise and categorise the different FER tasks/techniques used in the studies that we included in this review. It is important to note that the methodology used to measure FER can influence the interpretation of neurotransmitter effects.
A brief overview of FER study characteristics in the literature
A wide range of experimental tasks have been used to assess human FER. The diversity of experimental designs sometimes makes the comparison of their outcomes challenging, in part because there is a lack of detailed breakdown about the ways in which they differ and how these differences affect the contextualisation of study findings. Figure 1 summarises a list of criteria that we used to classify different FER studies included in this Review.
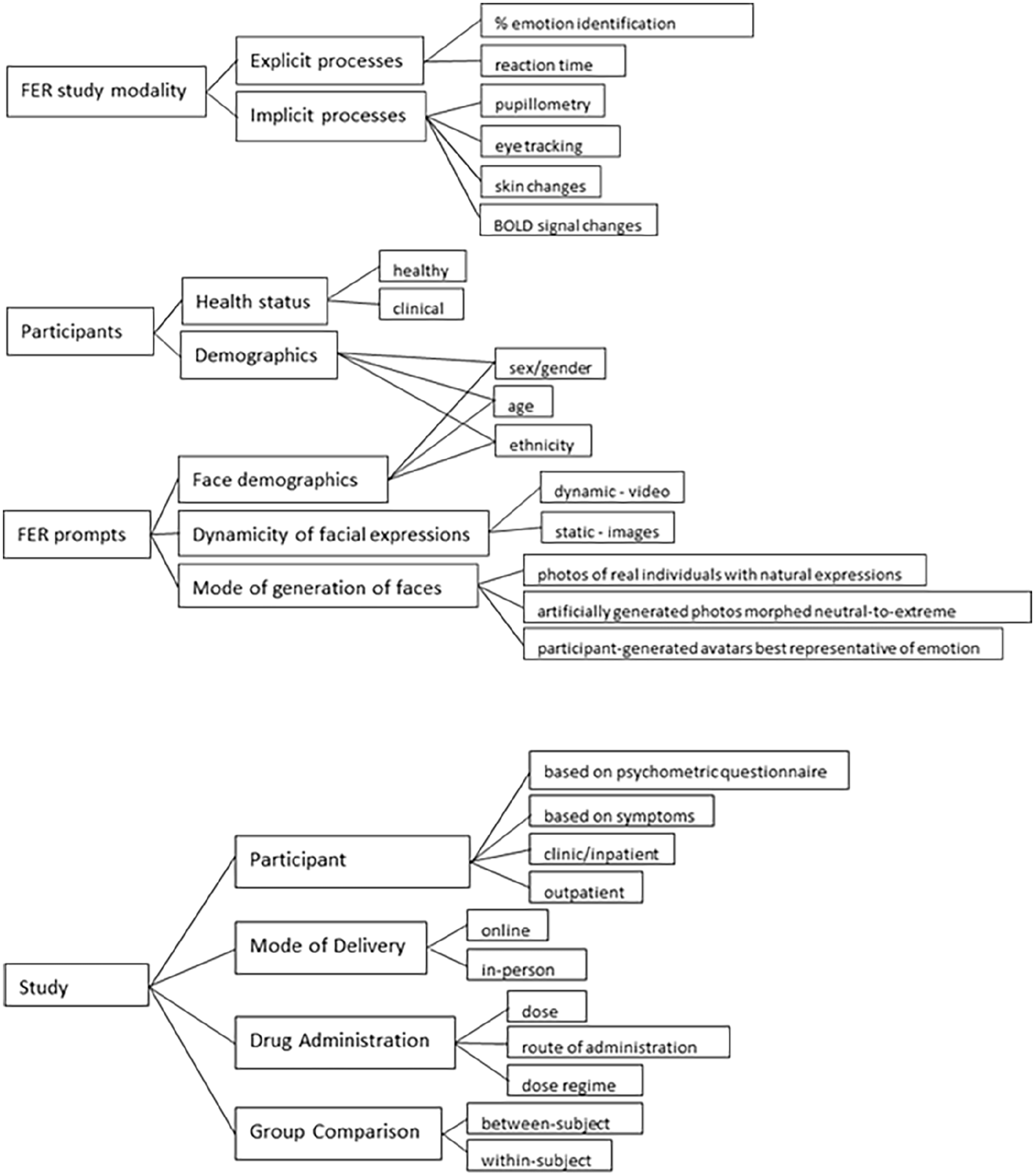
Key criteria for human facial emotion recognition (FER) study design.
This figure outlines the critical elements considered in FER study designs, including study modality, participant characteristics, FER prompt attributes, and study design considerations to highlight the highly matrixed nature of these experiments. FER study modality categorises outputs used to measure explicit processes (e.g. reaction time, emotion identification accuracy) and implicit processes (e.g. eye-tracking, pupillometry, physiological measures). Participants are stratified by health status (healthy or clinical populations) and demographic characteristics (e.g. sex, age, ethnicity). FER prompts are defined by their dynamic properties (static vs dynamic) and mode of generation (e.g. real faces, artificially generated morphs). Study design elements include participant recruitment (e.g. inpatient/outpatient, patients with low mood or recruitment based on psychometric scores) which may be associated with selection bias as for example, individuals volunteering for MRI studies may be less prone to anxiety (Charpentier et al., 2021), mode of delivery (e.g. online or in-person), drug administration details (dose, route of administration, schedule), and comparison groups (between-subject or within-subject designs). This framework highlights the various methodological choices that influence FER study outcomes and interpretations, emphasising the heterogeneity in study designs across the literature.
Serotonin
The serotonergic system, involving serotonin (5-HT) and its seven receptor families (5-HT1 to 5-HT7), plays a key role in regulating various physiological and behavioural processes (Berger et al., 2009; Barnes and Sharp, 1999; Nichols and Nichols, 2008). These include mood, appetite, sleep, cognition and social behaviours. Consequently, the involvement of the serotonergic system in FER, an important aspect of social cognition, has been extensively investigated using pharmacological agents.
Selective serotonin reuptake inhibitors (SSRIs) are a class of medications licensed in depression, anxiety disorders and other psychiatric conditions. They work by blocking the reuptake of serotonin, thereby increasing its availability in the synaptic cleft, and enhancing serotonin signalling. Studies investigating the effects of serotonin on FER have used SSRIs such as citalopram, sertraline and fluoxetine to enhance serotonergic transmission, or depletion of its amino acid precursor, tryptophan, to reduce serotonin transmission. The effects of serotonin modulation on FER have been investigated in both healthy participants and participants with major depressive disorder (MDD).
Overall, increased serotonin transmission has been found to exert a positive biasing effect on FER in healthy volunteers. Short-term administration of citalopram, a widely prescribed SSRI, reduced the accuracy of identification of anger and fear (Harmer et al., 2004) and increased positive ratings of happy infant faces (Stein et al., 2012). Recognition of anger and fear can act as important cues to trigger fight-or-flight responses, and citalopram administration has also been shown to attenuate startle responses associated with these emotions (Harmer et al., 2004), providing further evidence that serotonin modulates responses to aversive experience. These findings are echoed in a later neuroimaging study, in which citalopram was found to reduce activity in human amygdala in response to subconscious threat cues (Harmer et al., 2006). Another neuroimaging study suggested that short-term administration of escitalopram, an active isomer of citalopram, attenuates heightened amygdala activity in response to observing fearful faces in patients with MDD (Godlewska et al., 2012), mirroring the findings in non-MDD participants who received citalopram (Harmer et al., 2006).
In another study in patients with MDD, longer-term SSRI treatment improved recognition accuracy of happiness, surprise and disgust relative to baseline assessment levels prior to treatment (Tranter et al., 2009). Administration of fluoxetine, another widely prescribed antidepressant, to participants with MDD over 8 weeks showed reduced left amygdala activation to fearful faces (Sheline et al., 2001), and increased neural responses in limbic, extra-striate and subcortical regions in response to happy faces (Fu et al., 2007). These effects seem to be somewhat consistent and irrespective of the demographic characteristics of the study populations, producing similar patterns also in adolescent populations. For example, administration of fluoxetine to adolescents with MDD reversed the hypersensitivity in amygdala in response to angry faces, along with neural effects in other regions such as orbitofrontal cortex and subgenual anterior cingulate cortex (Capitão, 2014; Tao et al., 2012).
Taken together, these studies indicate that the serotonergic system modulates social cues relevant to threat detection (Osinsky et al., 2008) and the associated fight-or-flight response (Deakin and Graeff, 1991). A limited number of other studies also reported that enhancement of serotonergic transmission improves FER performance for affiliative emotions such as detecting sadness in others with some gender-specific effects (Alves-Neto et al., 2010), and less consistently for recognition of happy faces (Shiroma et al., 2014).
Besides SSRIs, other compounds have been used to manipulate serotonin transmission in experimental settings with findings globally agreeing with those from SSRI-based studies. MDMA (3,4-methylenedioxymethamphetamine or ecstasy), known for its role as a recreational mood elevating, ‘empathogen’, is a release and reuptake blocker of serotonin, noradrenaline and dopamine (Mustafa et al., 2020). Studies using MDMA have consistently shown an effect in reducing the accuracy at which healthy participants can identify negative emotions including fear, anger and sadness (Bedi et al., 2010; Hysek et al., 2014; Kirkpatrick et al., 2014; Schmid et al., 2014; Wardle et al., 2014), but the non-specific activity of MDMA on serotonergic transmission and others makes it hard to attribute its effects to one system. Furthermore, numerous studies have investigated the effect of psilocybin, a hallucinogen and mixed serotonin 5-HT1A/2A receptor agonist with promising antidepressant potential (Carhart-Harris et al., 2012, 2016; Goodwin et al., 2022) on FER. In a study by Kometer et al. (2012), psilocybin decreased the accuracy of recognition of negative facial expressions in healthy subjects, an effect which was blocked by the 5-HT2A receptor antagonist, ketanserin. These findings were mirrored in a later single-dose regime study, in which psilocybin administration in a small group of healthy volunteers also led to reduced identification of fearful facial expressions (Schmidt et al., 2013). A functional magnetic resonance imaging (fMRI) study in 2014 in which passive visualisation of emotional faces following the administration of a single dose of oral psilocybin in healthy adults triggered a reduced response to fearful and neutral faces in the amygdala and para-hippocampal gyrus, and to happy faces in the limbic and temporo-occipital area (Bernasconi et al., 2014) further reinforced the idea that psilocybin regulated response to fear.
A later fMRI study in a group of participants with moderate to severe MDD found that oral psilocybin led to increased post-treatment blood oxygenation level-dependent (BOLD) signals in the right amygdala when participants were exposed to fearful and happy faces, though no FER accuracy or reaction time were measured in this study (Roseman et al., 2018). In contrast, a subsequent fMRI study in healthy adults showed psilocybin acts via reducing the connectivity between the right amygdala and striatum, and between the right amygdala and the right fronto-median cortex during passive visualisation of angry and happy faces respectively, but no effect was shown during visualisation of fearful faces (Grimm et al., 2018).
Finally, the effects of psilocybin on reaction times in FER are inconsistent. In Grimm et al. (2018), reaction times for fearful, happy and angry faces were increased about 3.5 h post-administration of a single dose of psilocybin, with no effects on the accuracy of emotion identification. In contrast, in a contemporaneous study by Stroud et al. (2018), administration of two doses of oral psilocybin 1 week apart led to a significantly reduced reaction time in FER across all emotions in a group of MDD patients, but not in healthy volunteers.
However, it appears that the serotonin receptor specificity and concomitant activity on other receptors play a role in the observed response. A study using agomelatine, a selective 5HT2C serotonin-receptor antagonist and melatonin receptor agonist licensed for the treatment of major depression, found that following a treatment regime of 25 mg for 7 days agomelatine reduced recognition of sad facial expressions and modulated the emotion-potentiated startle response in healthy volunteers (Harmer et al., 2010).
Lysergic acid diethylamide (LSD), another known recreational drug and strong partial agonist of the 5-HT2 family of serotonergic receptors (De Gregorio et al., 2016), also appears to be implicated in fear recognition. A two-dose oral regime of LSD acutely impaired the identification of fearful expressions in healthy participants (Dolder et al., 2016). This finding aligns with a later study, which reported a significantly reduced activation of the amygdala and medial prefrontal cortex in response to fearful faces after healthy participants received an oral dose of LSD (Mueller et al., 2017).
In humans, the amino acid tryptophan is a precursor of serotonin. An early study on healthy volunteers using daily supplementation with L-tryptophan for 2 weeks found that it increased recognition of happy facial expressions and decreased recognition of disgusted faces in female but not male participants (Murphy et al., 2006). Studies investigating the effect of serotonin on FER have also used acute tryptophan depletion (ATD) regimes to reduce serotonin transmission in the brain through depletion of its precursor, tryptophan. In one study, serotonin depletion reduced recognition of fearful expressions in female but not male participants (Harmer et al., 2003b), whereas a later study found that ATD modulated happy facial expressions irrespective of participant gender, but these effects depended on vulnerability to depression (Hayward et al., 2005). Findings from fMRI studies showed that, even in the absence of any behavioural effects, the BOLD response in the right amygdala and hippocampus was enhanced when participants under the ATD regime were shown fearful expressions (Cools et al., 2005), while findings from Daly et al. (2010) also implicate the cingulate gyrus in the ATD-mediated modulation of fear processing (Daly et al., 2010). Equally, another functional neuroimaging study using ATD showed differential modulation of brain activation when processing happy versus sad faces. Specifically, ATD decreased activation in the posterior cingulate and lingual gyri during happy face processing, while increasing activation in the left inferior frontal and middle temporal gyri; conversely, during sad face processing, ATD reduced activation in the right superior/medial frontal gyrus and bilateral parietal and occipital cortices, but enhanced activation in the cerebellum and left frontal regions encompassing the inferior frontal and precentral gyri (Fusar-Poli et al., 2007).
In terms of disgust, earlier behavioural studies reported no significant impact of ATD on recognising disgust (Harmer et al., 2003; Hayward et al., 2005; Marsh et al., 2006), but Daly et al (2010) found notable interaction effects between ATD and expression intensity, observed in bilateral cingulate gyri, left postcentral gyrus and right cerebellum.
Other studies have investigated the effects of ATD on depressed cognition. Using ATD in patients with a history of depression (i.e. remitted MDD (RMDD)) has often been argued to be an experimental model for studying depressive cognition in laboratory settings (Hayward et al., 2005). Contrary to the effects observed in people without any history of depression, patients with RMDD displayed reduced accuracy for happy faces following a low-dose ATD protocol (Hayward et al., 2005), while in a separate study, high-dose ATD decreased recognition of fear in remitted depressed patients (Merens et al., 2008), which is also an effect reported in ATD studies in healthy participants. A contemporaneous study (Van der Veen et al., 2007) attempted to explain the differences across some of these findings by suggesting that ADT may have variable effects on the central concentration of serotonin in different participant groups, and that, ultimately, it is the central serotonin levels that affect FER. In their study, Van der Veen and colleagues compared FER performance following ATD between a group of healthy female volunteers with a positive family history of unipolar depression (FH+) and a group of female volunteers with no history of unipolar depression (FH−). ATD was found to affect FER, albeit differentially between the two groups and in a mood-concordant fashion; ATD led to reduced identification of negative expressions and a stronger amygdala response to intense fearful faces in the FH+ group, alongside negative effects on the mood. In contrast, ATD had no effect on the subjective mood ratings of the FH− group, nor did it affect FER (Van der Veen et al., 2007). These results suggest a complex interaction between serotonin levels and depression vulnerability both affecting FER.
In summary, the serotonergic axis is the most extensively studied in terms of its role in modulating FER in humans. Results from studies using functional imaging and cognitive tasks across healthy, individuals with different types of vulnerability to depression and depressed patients consistently show that serotonin has a strong inhibitory effect on recognising fearful faces and in some studies, but with a more variable presentation, a positive effect in improving identification of happy facial expressions.
Noradrenaline
The noradrenergic system, primarily involving the neurotransmitter noradrenaline (norepinephrine), is critical for modulating attention, arousal, memory and stress responses, as well as higher-order cognitive processes such as decision-making and problem-solving. It is centrally regulated by the locus coeruleus, a brainstem nucleus that plays a pivotal role in adapting to environmental demands and stressors (Berridge and Waterhouse, 2003; Sara, 2009; Aston-Jones and Cohen, 2005).
One common way of safely manipulating the noradrenergic system in experiments with human participants relies on the use of licensed antidepressants from the serotonin/norepinephrine reuptake inhibitors (SNRIs) family or atypical antidepressants with dual dopamine/noradrenaline activity such as bupropion. Unfortunately, pharmacological agents with truly selective activity on the noradrenergic axis are limited in number. Of those, the antidepressant reboxetine was one of the first whose mode of action was found to be highly selective for the noradrenaline reuptake site (Wong et al., 2000), while other more commonly clinically used SNRIs such as venlafaxine (Harvey et al., 2000) and duloxetine (Rodrigues-Amorim et al., 2020) have a significantly lower ability to facilitate noradrenergic activity compared to their effects on serotonin.
Earlier single-dose studies with reboxetine in healthy volunteers indicated that the noradrenergic system modulates the recognition of happy faces; a single dose of reboxetine increased the accuracy of identification of happy faces in healthy volunteers (Harmer et al., 2003a). Furthermore, longer-term administration of reboxetine over a 7-day period was shown to promote a more global positive bias in FER, such that in addition to augmenting positive FER, recognition of fearful and angry faces was diminished (Harmer et al., 2004). A subsequent study suggested that this positive reboxetine-induced bias may generalise to other related domains, including a more positive rating of infant faces in the reboxetine group compared to the control following a week-long administration (Stein et al., 2012), thus indicating a role for noradrenaline in mediating evolutionarily preserved social affiliations potentially relevant for mother-infant bonding. Finally, a study including participants with MDD suggested that reboxetine improves the recognition of positive facial expressions irrespective of the duration of the drug administration protocol (2 vs 6 weeks) in multi-sessional studies, suggesting a somewhat temporally stable role of noradrenaline on FER (Tranter et al., 2009), though the study did not use a placebo control. A subsequent, placebo-controlled study showed that a single dose of reboxetine to participants diagnosed with MDD reversed their characteristic reduction in recognition of happy facial expressions (Harmer et al., 2009).
Although most studies support a role of reboxetine in inducing a positive bias in FER, the findings in this domain are not always consistent. For example, an early fMRI study that used a single dose administration of reboxetine in healthy individuals showed an augmented neural response to fearful facial expressions and an attenuated response to neutral faces in the amygdala, though fear-versus-neutral were the only FER prompts tested (Onur et al., 2009). In contrast, Norbury et al. (2007) found that a single dose of reboxetine administered to healthy participants caused a reduced amygdala response to fearful faces and an increased response to happy versus neutral faces in the fusiform gyrus in a fMRI study. The reason for these contrasting findings is not clear but perhaps also relates to differences in sample characteristics and face paradigm.
The effects of bupropion, a dual inhibitor of noradrenaline and dopamine uptake and clinically used as an antidepressant (Stahl et al., 2004), have also been studied with regards to FER. In a placebo-controlled study, acute bupropion administration to healthy adults led to an improved recognition accuracy of happy faces, reduced response bias to sad faces and reduced vigilance towards fearful faces, likely reflecting the effects of the noradrenaline and dopamine pathways respectively (Walsh et al., 2018a, 2018b).
The other family of noradrenergic pharmacological agents frequently used in experimental psychopharmacology are the beta-adrenoceptor antagonists, a class of medications commonly used to treat anxiety in primary care settings (Boyce et al., 2021). Certain beta-blockers such as propranolol, timolol and pindolol are highly lipophilic and cross the blood-brain barrier readily, interacting not only with peripheral but also central beta-receptors (Cojocariu et al., 2021) and thus, exerting neuropsychological effects. One study suggested that healthy volunteers were slower to identify sad faces following administration of a single dose of propranolol (a mixed β1 and β2-receptor antagonist) without any compromise in the accuracy of emotion identification (Harmer et al., 2001). Similarly, findings from an fMRI study suggested that the same propranolol regime attenuated activation of the amygdala irrespective of the valence of facial emotions viewed (Hurlemann et al., 2010), positing a facilitatory role of noradrenaline in emotional processing of facial expressions. However, findings from another study in healthy volunteers who received metoprolol, a selective β1-adrenoceptor antagonist suggested that modulating the noradrenergic system with metoprolol did not lead to downstream cognitive effects in FER (Zangara et al., 2002).
Overall, taking into consideration both positive and negative noradrenergic drug modulators, the effects of noradrenaline on FER are largely consistent; increasing noradrenergic transmission induces a positive bias in FER.
Acetylcholine
The cholinergic system, which primarily relies on the neurotransmitter acetylcholine, plays a central role in memory, emotional regulation and cognitive functions. It is particularly active in brain regions such as the hippocampus and amygdala, and dysfunction in this system has been implicated in emotional disorders like anxiety and depression. The actions of acetylcholine are mediated through nicotinic and muscarinic receptors, which regulate various neurophysiological processes associated with emotional responses (Hasselmo and Sarter, 2011; Picciotto et al., 2012; Sarter et al., 2009).
The evidence supporting the involvement of the cholinergic system in FER comes from experimental pharmacology studies. One compound that has been used to manipulate cholinergic transmission is scopolamine, a non-selective antagonist of muscarinic cholinergic receptor subtypes (Drevets et al., 2013). Findings related to experimental administration of scopolamine have been inconsistent with some studies suggesting that it reduces identification specifically of anger and disgust, but not other emotions (Kamboj and Curran, 2006), while other studies suggesting no significant effect on any of the basic emotions (Bukala et al., 2019). The discrepancy between study findings could be explained by the experimental design specification; for example, Kamboj and Curran (2006) used a single 0.6 mg dose of scopolamine administered subcutaneously, whereas Bukala et al. (2019) used a 1.5 mg transdermal patch applied over 17 h. These differences can affect the clearance rate and bioavailability of the drug leading to different plasma concentrations. Currently, the literature does not include studies using other cholinergic compounds such as rivastigmine, often used in the management of Alzheimer’s disease (Farlow et al., 2013).
Considering the role of acetylcholine in modulating physiological arousal responses such as pupil dilation (Faber, 2017), more research in this area could reveal important insights into its role in FER.
Dopamine
Dopamine is a neurotransmitter involved in reinforcement learning, belief updating and motivation for goal-directed behaviour through its action on a range of dopamine receptor subtypes (Ashok et al., 2017; Schultz, 2016; Wise, 2004). Experimental manipulations of the dopamine system in healthy individuals using dopaminergic agonists and antagonists indicate a link between dopamine transmission and processing of negative emotions (Grace, 2016; Howes et al., 2017).
For example, pramipexole, a non-ergot dopamine agonist with high specificity for D2/D3 receptors, decreased amygdala activity bilaterally in response to fearful facial expressions in the absence of any behavioural differences in identifying these emotions, relative to placebo administration (Martens et al., 2021). Similarly, other dopamine agonists such as levodopa were also shown to attenuate amygdala activity in response to emotional faces, again, in the absence of any differences in overt behaviour (Delaveau et al., 2007). In contrast, blockade of the D2 receptor family using sulpiride, a licensed antipsychotic and a dopamine receptor antagonist, selectively impaired the identification of angry faces (Lawrence et al., 2002), consistent with the role of DA in processing threat cues. Furthermore, in Homan et al (2014) reduction in circulating dopamine (and noradrenaline) was achieved using catecholamine depletion with oral α -methyl- para -tyrosine (AMPT) in healthy and RMDD patient groups before exposing them to fearful and neutral faces. In RMDD, AMPT treatment increased blood flow in the left posterior cingulate cortex and decreased flow in the left dorsolateral prefrontal cortex when presented fearful faces, whereas healthy controls exhibit opposite patterns under similar conditions (Homan et al., 2014).
It is also possible to gain insight into how the dopamine system modulates FER by deriving evidence from studies in patients diagnosed with disorders with an established involvement of the dopaminergic axis, such as Parkinson’s disease (Lotharius and Brundin, 2002) which involves degeneration of dopaminergic neurons. Although dopamine abnormalities have also been implicated in a number of neuropsychiatric conditions such as schizophrenia (Brisch et al., 2014) (i.e. mainly attributed to hyperactive dopaminergic transmission) and bipolar disorder (Ashok et al., 2017). It is important to note that the strength of evidence about the magnitude of the effect of dopamine in these conditions varies. As a result, interpreting findings from these patient groups should be done with caution.
Earlier studies in PD patients also reported lower accuracy in identifying facial expressions of anger and disgust to a greater extent (Sprengelmeyer et al., 2003). Deficits in identifying these emotions were also reported by a subsequent study that further associated impairments in identifying disgust and angry faces to grey matter atrophies in the ventral striatum (vSTR), subgenual and anterior cingulate cortices (Baggio et al., 2012). Moreover, studies in patients with focal lesions to the vSTR also highlighted that striatal dopaminergic neurons are implicated in the recognition of angry faces (Calder et al., 2004).
In a cohort study of patients with bipolar disorder, participants on dopaminergic antagonists were less accurate at recognising angry faces (Bilderbeck et al., 2017). Consistent with more recent studies (Muros et al., 2021), a meta-analysis of 112 studies showed a large effect for impaired emotion processing in medicated schizophrenic patients (Hedges’ g = 0.88), with significant impairments for recognising disgust, fear and surprise, and medium effects for sadness and happiness (Barkl et al., 2014). Furthermore, a second meta-analysis investigating the effects of antipsychotic treatment on FER found an overall positive effect leading to improved target identification accuracy, particularly in the class of atypical antipsychotics (e.g. risperidone, quetiapine and olanzapine, all of which are mixed dopamine and serotonin antagonists; Hedge’s g = 0.11); such an effect was not reliably observed in patients on typical antipsychotics (Gabay et al., 2015) (e.g. haloperidol, perphenazine and perazine), which are selective D2-receptor antagonists. Comparably, in antipsychotic-naive patients with schizophrenia, FER impairments in accuracy were shown across the board for all primary emotions (Behere et al., 2009). Though there is a degree of inter-subject variability, variability in mode of action between typical and atypical antipsychotics as well as in disease phenotype, it is possible to use data from these studies to detangle the effects of neurotransmitter dysregulation due to pathophysiology of the disease and neurotransmitter effects due to mode of drug action.
Taken together, dopamine transmission seems to selectively modulate the recognition of negative emotions. If we interpret null behavioural findings in experimental pharmacology studies in healthy volunteers as indicating ‘no cognitive side effects’ in FER, the selective role of DA transmission in attenuating amygdala hypersensitivity could be meaningful for identifying treatment targets. Although earlier studies proposed an inverted-U shaped relationship between DA transmission amygdala activity in response to negative emotional faces (Delaveau et al., 2007), global evidence does not seem to affirm this assertion. As stated previously, detection of fearful, angry and disgusted faces has survival value, although across different timeframes; angry and fearful faces prompting rapid fight-or-flight responses, whereas disgust in others prompting observational/Pavlovian learning of reward values associated with aversive stimuli (e.g. unpleasant odour or taste). From this perspective, we propose that the DA system modulates evolutionarily hard-wired reactions in response to processing affective information that has survival value. The studies we review so far may suggest that the dopaminergic and serotonergic systems may represent parallel information processing streams working in an integrated fashion to guide self-preservative behaviour in the short- and medium-term through immediate reaction (fight-or-flight serotonergic inputs and anger inputs) and planned strategies to longer-term effects (cued by disgust inputs processed by the dopaminergic system).
Glutamate
Glutamate is the primary excitatory neurotransmitter in the brain, essential for synaptic plasticity, learning and memory formation (Millan et al., 2012). It plays a pivotal role in cortico-limbic circuits that regulate emotional processing and mood regulation, with dysregulation of glutamate transmission implicated in cognitive deficits and mood disorders (Krystal et al., 2019; Moghaddam and Javitt, 2012).
Studies investigating the modulatory role of the glutaminergic pathway in FER have predominantly used ketamine administration protocols. Ketamine is a non-competitive methyl-D-aspartate (NMDA) receptor antagonist binding to the phencyclidine site within the ion channel of the NMDA receptor complex, which is one of the better-understood ionotropic glutamatergic receptors (Duman et al., 2019). Earlier studies using sub-anaesthetic doses suggest that ketamine administration attenuates activity in the limbic regions and more specifically in the amygdala in response to fearful faces (Abel et al., 2003). This pattern of neural activation seems to generalise to other experimental designs inducing emotional stimulation; for example, administering ketamine to participants and presenting them with affective pictures not limited to faces (Scheidegger et al., 2016) 15 min later revealed pro-cognitive effects such as quicker valence judgements in rating affective images. However, a more recent study indicated a global deterioration of FER following intravenous ketamine infusion over a period of 5 min with more significant reductions observed in the domain of affiliative emotions such as detecting sad faces (Ebert et al., 2012).
Besides ketamine, the relationship between the glutamatergic system and FER has been studied using other ligands such as D-alanine and D-serine, which both act as co-agonists of NMDA receptors. One recent study reported pro-cognitive effects of D-alanine such as more rapid identification of facial expressions of fear, surprise and anger (Capitão et al., 2020) but did not report significant effects for D-serine. Partial agonists of NMDA receptors such as d-cycloserine (Chen et al., 2021) and memantine (Pringle et al., 2012) did not influence performance in FER categorisation, though a single dose administration of memantine led to reduced bias for negative stimuli. The studies investigating the effect of glutamate modulators in healthy volunteers are limited, hence developing a clearer understanding about how the glutamate system modulates FER is difficult. At the time of writing, we are collecting FER data across four assessment points (baseline, pre-infusion, 24 h post-infusion, +1 week) from 70 healthy volunteers who are randomised to take ketamine or placebo (ClinicalTrials.gov Identifier: NCT04850911), to be able to better characterise the role of this pathway in neuromodulation of FER.
In the past 2 decades, several studies clearly demonstrated the rapid antidepressant effects of ketamine (Berman et al., 2000; Coyle and Laws, 2015; Lally et al., 2014; Mkrtchian et al., 2020), which put NMDA receptors in the spotlight with regards to drug discovery in psychiatry. Recently, we suggested that ketamine may actualise this rapid antidepressant effect via its action on learning, affective memory recall and reward processing. These changes may involve neural networks distributed across fronto-meso-limbic networks and with the habenula playing a critical role in orchestrating this system (Pulcu et al., 2021; Yang et al., 2018). Treatment-resistant depression (TRD) patients who received serial ketamine infusions were able to correctly identify happy and faces from ambiguously congruent prompts (25% intensity) with the highest accuracy, while the same was not true for fear (Shiroma et al., 2015). In another study (Reif-Leonhard et al., 2025) in TRD patients receiving repeated infusions of esketamine, an isomer of ketamine with more selective antagonistic activity on NMDA (Moore et al., 2022), participants showed improvements in emotional misclassifications and in the accuracy of identification for all emotions except for sadness. Out of these trends, the improvement in the accuracy for recognising fear and the reduction in misclassifying other expressions as angry were statistically significant. Unfortunately, the difference in the FER tasks and emotions studied in the two studies make the comparison of their findings challenging, as Shiroma et al. (2015) only used 25% and 50% morphs showing one of fear, sadness and happiness, while in Reif-Leonhard et al. (2025), all six emotions were studied, with face morphs ranging from 10% to 100% across the six basic emotions, and including misclassification.
Our research group is also investigating how ketamine modulates FER in a group of treatment-resistant MDD patients in an independent double-blind randomised clinical trial (NCT05809609). Overall, more work needs to be done in this area to establish the cognitive profiles of novel antidepressants acting on the glutamate system, and to obtain a better understanding of the role of the glutamatergic pathway in FER beyond its pro-cognitive effects.
Gamma-aminobutyric acid
Gamma-aminobutyric acid (GABA) is the brain’s primary inhibitory neurotransmitter, essential for modulating neural excitability, anxiety and mood regulation. Dysfunction in the GABAergic system has been implicated in various psychiatric conditions, including anxiety disorders, depression and epilepsy. Pharmacological agents targeting GABA-A receptors, such as benzodiazepines, are among the most prescribed treatments for anxiety and insomnia (Chiu et al., 2018; Möhler, 2012; Rudolph and Möhler, 2014) and are known potentiators of the GABA-ergic system (Griffin et al., 2013).
Earlier studies suggested that administration of a single dose of diazepam, a commonly used benzodiazepine (GABA agonist), reduced the accuracy of correctly identifying fearful, and angry faces (Blair and Curran, 1999; Zangara et al., 2002), while preserving the recognition of other primary emotions. These effects likely underpin the anxiolytic effect of benzodiazepines, making individuals less likely to respond to threat cues from others. However, Coupland et al. (2003) found that a single dose of 15 mg diazepam impaired recognition of all emotional faces. A subsequent meta-analysis by Garcez et al. (2020), which assesses the impact of benzodiazepines on facial recognition in eight studies, showed a selective ability of benzodiazepines to inhibit the recognition of anger. From this, it seems reasonable to conclude that potentiation of GABA-ergic neurotransmission by benzodiazepines reduces the facial processing of threat.
Overall, like the dopaminergic pathway, evidence from the literature suggests that the GABAergic pathway is also implicated in guiding emotional processing that supports self-preservative behaviours. To the best of our knowledge, currently, there is no study directly comparing the dissociable effects of dopamine and GABA on FER. This could be an important direction for future studies to tease apart specific contributions of these neurotransmitter pathways to FER.
Opioids
The opioid system is a complex neural pathway in the human body that plays a crucial role in regulating pain perception, mood and the reward system. Opioid receptors are proteins found on the surface of nerve cells in the brain and throughout the body and can be classified into mu, delta, kappa and ORL-1 (Nummenmaa and Tuominen, 2018). The mu receptors are primarily implicated in nociception but also regulate the central dopamine reward pathways modulating euphoria (Al-Hasani and Bruchas, 2011). Endogenous opioids such as endorphins that help regulate pain are naturally produced by the body and regulate emotional responses, while exogenous opioids are synthetic or naturally derived compounds, like morphine or oxycodone that can activate opioid receptors and produce pain relief and euphoria (Nummenmaa and Tuominen, 2018).
The role of the opioid system in FER is thought to be mediated via the modulation of reward and fear perception. Studies administering buprenorphine, a mixed partial receptor agonist with stronger effects on the mu and weaker effects on the kappa, delta and ORL-1 opioid receptors (Lutfy and Cowan, 2004) found that following administration of buprenorphine, participants showed reduced sensitivity to recognising fearful faces. In particular, Ipser et al. (2013) used a dynamic FER task gradually morphing faces from neutral to emotional and found that administration of a single 0.2 mg dose of buprenorphine orally reduced the identification accuracy of fearful face. A later study followed the same dose regimen sublingually and reported reduced attention to fearful faces in an attention task (Bershad et al., 2016). A further placebo-controlled study (Schmidt et al., 2014) found that administration of heroin, a mu agonist, to heroin-dependent participants reduced the amygdala response to fearful faces compared to a saline injection. Interestingly, naltrexone, a mu-opioid antagonist with weaker antagonist effects on kappa and delta receptors, administered to healthy participants was also shown to impair the identification of not only fearful faces but also sadness (Wardle et al., 2016). On the other hand, a later study recruiting only female participants (N = 82) who were given a single 10 mg dose of oral morphine sulphate, a mu-opioid agonist, suggested no difference in the response times or accuracy of target emotion identification in FER (Massaccesi et al., 2022). Massaccesi et al. (2022) argued that the discrepancy between the effects of buprenorphine, naltrexone and morphine sulphate on FER performance may be attributable to their differential affinity for the kappa receptor family of opioid receptors (Løseth et al., 2018; Wardle et al., 2016). Globally, the opioid system seems to modulate perception of negative emotions, potentially with complex three-way interactions between this system and participants’ gender and psychosocial stress levels.
Synthesising the effects of pharmacological and functional imaging studies on FER
Overall, the purpose of this review paper has been to distil the effects of central (and to a lesser extent peripheral) pathways on human FER. After reviewing the extensive literature including pharmacological and functional imaging studies in different participant groups, we attempt to integrate the emerging themes with existing emotional classification models (Figure 2). Here, we integrate the basic six-emotion model with Russell’s circumplex model (Zheng et al., 2018) placing Ekman’s six basic emotions (Ekman, 1999) within the two-dimensional paradigm characterised by valence (x-axis) and arousal (y-axis) parameters (guided by Russell, 1980) to capture some of the multi-emotional effects observed when isolating the effect of different neurotransmitters. Valence refers to the degree that a stimulus is pleasurable (positive) or aversive (negative), while arousal describes the level of physiological activity and can be high or low. In Figure 2, we plot the six basic emotions in the two-dimensional axes system, with anger and fear representing emotions in the high-arousal negative-valence quadrant, surprise and happiness representing high-arousal, positive-valence emotions, and disgust and sadness plotted in the low-arousal negative-valence quadrant. This is a useful summative pictorial representation of the vast body of literature on the effect of well-studied central neurotransmitter pathways of FER and can be a useful reference point for experimental design in future studies.
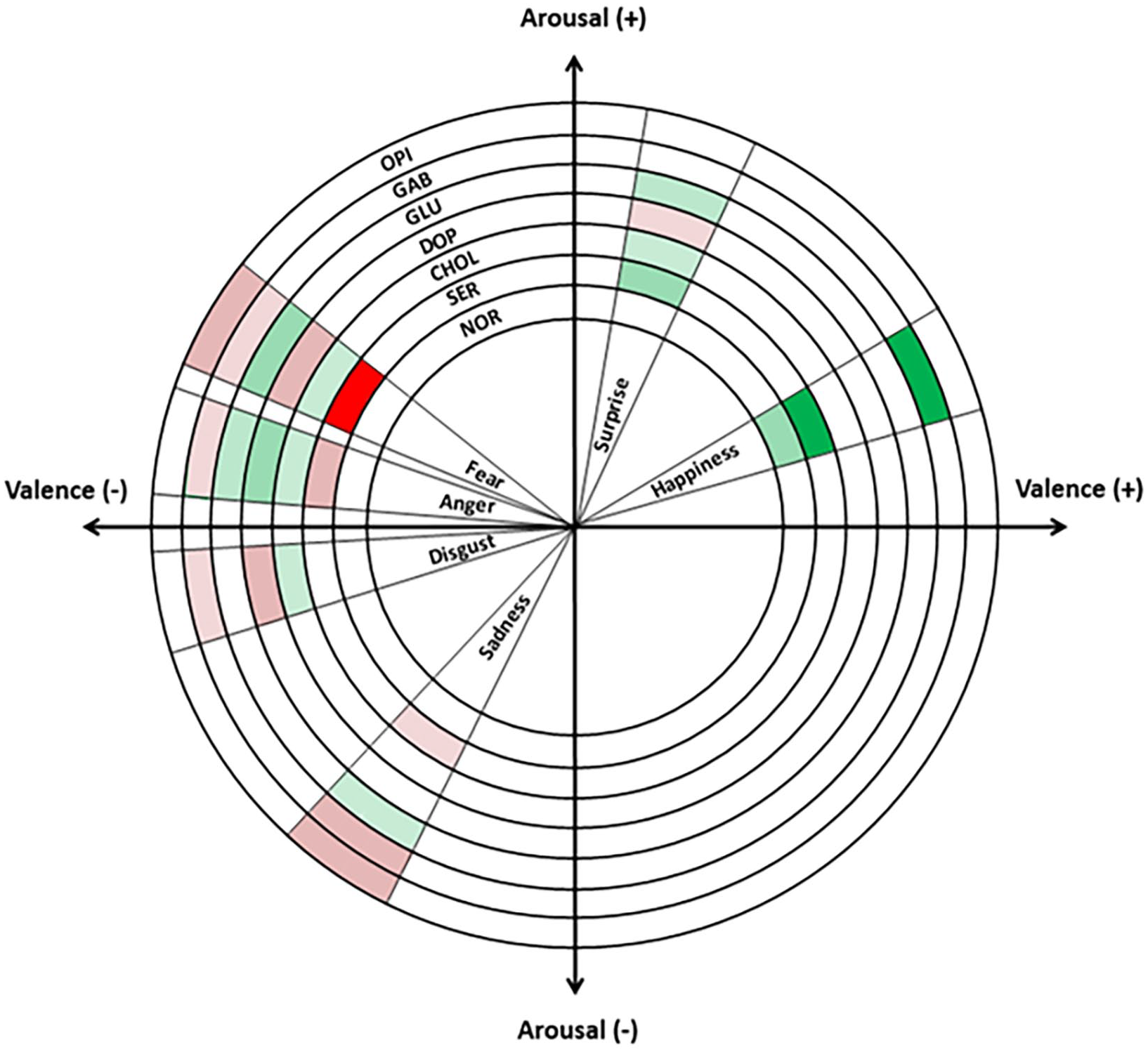
A pictorial representation of the effects of central neurotransmitter systems on facial emotion recognition.
Green colours indicate that a neurotransmitter improves a metric related to discriminating an emotion of interest or higher neural activation in functional imaging studies, while red indicates effects in the opposite direction. The shades of green and red correspond to the number of studies supporting the finding. Dark red and green correspond to >5 studies, intermediate shades correspond to 2–4 studies, and lighter shades indicate evidence from 1 study. Classification of primary emotions by their valence and arousal components is adapted from Zheng et al. (2018).
Interestingly, it appears that responses to fear and anger, which are both high-arousal and negative-valence emotions, are jointly regulated by most neurotransmitter axes, pointing to their high evolutionary value as information sources that influence critical, rapid, survival-related decision-making. In addition, they are both under positive and negative regulation by more than one pathway pointing to a degree of redundancy, overlap and competition between different neurotransmitter axes. This overlap further highlights the importance of maintaining a narrow window of perceptual and processing accuracy for danger-signalling cues. In contrast, high-arousal and positive-valence emotions such as happiness and surprise are mostly under positive regulation; although a few studies report findings that the dopaminergic pathway is a negative regulator for surprise. Currently, unpublished work from our research group suggests that fearful faces are most frequently mislabelled as surprise. Considering the similar arousal properties of fear and surprise, misattribution errors between these emotions seem to happen along the valence axis. Generally, any isolated findings for these two emotions should therefore be interpreted with caution.
We believe that our synthesis can guide clinical research into potentiation of pharmacological regimes for mental health disorders. For example, it is clinically well-established that patients with refractory depression are often given combination therapies with antidepressants acting on different neurotransmitter systems (Moret, 2005). One clinically used combination is a joint prescription of SSRIs or SNRIs with bupropion, a noradrenaline and dopamine reuptake blocker (Zisook et al., 2006). Our synthesis of the literature as shown in Figure 2 suggest that medications in this treatment strategy not only act complementarily but in fact potentiate each other’s mode of action by both increasing responsiveness to positive cues and dampening down responsiveness to negative social cues.
However, it is important to recognise the various limitations in the ambitious goal of synthesising heterogeneous literature and data in psychopharmacology and FER. First, FER tasks vary widely in terms of complexity, stimuli used (e.g. static vs dynamic facial expressions), and the required cognitive processes (implicit perception vs explicit labelling). Differences in task design can influence performance outcomes, making it challenging to compare findings across studies. It is important to acknowledge that discrepancies in FER performance can stem from a range of factors, including perceptual, attentional, emotional or strategic approaches. While these processes are often interlinked, future studies should aim to disentangle these factors to better understand the specific contributions of each to FER outcomes.
Second, the quality and availability of evidence across neurotransmitter systems are uneven, with certain neurotransmitters, such as serotonin and dopamine, being more extensively studied than others, partly due to the availability of licensed medications, which makes it easier to study a pharmacological intervention with a well-characterised mode of action. As a result, findings related to less-studied neurotransmitter pathways may be more variable (or noisier) or derived from a smaller evidence base, limiting the generalisability of conclusions. Recognising this limitation is crucial when synthesising evidence and drawing broader inferences from literature.
A third consideration one should make regards the heterogeneity of study designs and the implications they have for the interpretability of the studies; this can be seen as both a limitation and a strength because although it makes evidence synthesis less straightforward, it also allows for more holistic insights into a largely unchartered process. The inclusion of behavioural, neuroimaging and clinical studies provides a comprehensive framework for understanding the neural and cognitive processes underlying FER. Each type of study offers unique insights: neuroimaging studies help to delineate brain regions and networks involved in FER, while behavioural studies provide data on how these processes manifest in observable outcomes. However, it is important to note that these types of studies measure different constructs, and their findings may not always align due to variations in methodology, participant populations and outcome measures.
Furthermore, the granularity of information in each study differs extensively; for example, in FER cognitive tasks, some may study an emotion as a whole, whereas others may study different gradations of the emotions. A common challenge in current FER performance-based studies is that most of the available data in recognition accuracy as a function of the facial emotion morph (e.g. from 10% to 100% fear) are averaged across these gradient steps with a view to identify overall effects across different emotional categories (e.g. how SSRIs influence recognition of fearful vs happy faces). However, this approach, while providing a bird-eye view on neurotransmitter effects, fails to report the fine details that are needed to construct mechanistic computational models of FER. By incorporating diverse types of evidence, this review not only aims to synthesise the available signals into a holistic perspective but also highlights the limits of interpretation inherent to integrating findings from distinct methodologies and suggests future avenues to fill the gaps.
Despite efforts to highlight and bring to light inconsistencies across studies, several methodological variations may have influenced our findings due to the highly matrixed nature of this landscape. Differences in participant characteristics, such as age, gender and cultural background, variations in dosing specifics, including dosage levels and administration protocols can affect FER abilities, potentially leading to variability in results and their interpretation. Equally, the mere choice of the scope of a study can skew our understanding of the contributions of different neurochemical systems by virtue of differential representation in the literature. For instance, an argument often raised in the scientific community is that the frequent reporting of changes in fear recognition might be attributed to its extensive study rather than a unique sensitivity to interventions, especially when one tries to synthesise meaningful findings in a vast and methodologically diverse field such as neuropsychopharmacology of FER. However, even acknowledging this limitation can help us ideate ways to compensate by prioritising research in underlooked areas and favouring a group of standardised protocols in future research to enhance the comparability generalisability and compatibility of findings to inform a higher-order model.
Another important limitation of using findings from pharmacological, case–control and functional imaging studies to map the effects of different neurotransmitter pathways on FER is that most of the performance-based tasks rely on using the six-emotion model, providing only a few sampling points, and making inference challenging. When considered in the context of continuous emotion models (Binetti et al., 2022), like that presented in Figure 2, it is easier to summarise and interpret the general trends regarding the effect of neurotransmitters on FER in terms of resource allocation and yield. For example, threat cues such as anger and fear inform rapid decisions with high survival value and, thus, benefit from over-investment of processing resources, tight regulation, and signal integration. On the other hand, recognising positive-valence cues such as happiness is not implicated in immediate survival-critical decisions but can provide longer-term benefits such as facilitating social participation, affiliation and reproduction. As a result, it is not surprising that emotional processing on the positive end of the spectrum is not subject to tight bi-directional regulation by overlapping neurotransmitter pathways.
Conclusion and future directions
In this narrative Review, we provide a qualitative synthesis of how different neurotransmitter pathways influence human FER. Global evidence seems to indicate that the recognition of fight-or-flight-related emotional cues is the most malleable and most tightly regulated by overlapping neurotransmitter pathways. Overall, a change in the recognition of fear is the most reported finding in social cognition (Figure 2). This may be due to the evolutionary utility of recognising threat cues and the salience attributed to fearful expressions. One observation that we can qualitatively communicate is that FER studies involving functional neuroimaging overall reported lower recognition accuracy across different emotions relative to purely behavioural administration of FER tasks. This may be due to some fundamental differences in study populations that sign up to MRI versus behavioural pharmacology studies, as discussed elsewhere (Charpentier et al., 2021), or because the MRI environment is quite different from the context in which people typically perform these judgements.
Future studies might consider analysing differences in detection thresholds; for example, using drift-diffusion models that are particularly suitable for evidence accumulation (i.e. the response time that is required to judge a morphed face as fearful) could be useful in this domain. Considering the speed by which FER judgements are made, pupillometry (i.e. the iterative measurement of changes in pupil dilation), a technique with very high temporal resolution, can be an important research modality for understanding the underlying physiological correlates of FER, and prediction errors arising from misjudgements. Pupil dilation is also under the influence of a number of neurotransmitter systems (Faber, 2017), therefore experimental pharmacology studies using this modality can reveal important insights.
Overall, perhaps as expected, central neurotransmitter hubs and brainstem nuclei projecting both to limbic and cortical regions influence FER to a greater extent than other factors such as peripheral inflammatory processes. We think that future studies should adopt a combination of experimental pharmacology and behavioural modelling methods which should reveal more detailed insights into how FER responses are evoked temporally and the specific role of different neurotransmitter systems modulating this process. Designing multidimensional experiments in this area would also have clinical utility, promoting a better understanding of the cognitive effects of compounds that can treat psychiatric and neurological conditions. This is a necessary step towards personalising psychotropic treatments to individual disease correlates, hence achieving better clinical outcomes particularly in the psychosocial symptom domain.
Last but not least, one also needs to acknowledge the broader social and interpersonal context and functions of emotions beyond examining them as solely personal, chemical phenomena and reducing them to mere consequences of complex biochemical pathways. Parkinson (1996) argues that emotions are not just private but rather emerge directly through the medium of social and interpersonal interaction (Parkinson, 1996). As such, emotions take on relational meanings which can dynamically change depending on the context. In this relational framework, understanding the temporal dynamics of how different neurotransmitter networks are implicated in the process of facial emotional recognition is no longer the endgame; rather it provides a baseline that can simplify the process of understanding the plethora of complex interpersonal factors that shape the dynamic process of FER.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251338225 – Supplemental material for Neurotransmitter modulation of human facial emotion recognition
Supplemental material, sj-docx-1-jop-10.1177_02698811251338225 for Neurotransmitter modulation of human facial emotion recognition by Myrto Vlazaki, Catherine J Harmer, Philip J Cowen and Erdem Pulcu in Journal of Psychopharmacology
Footnotes
Author contributions
MV carried out the literature review. MV and EP wrote the manuscript, and CJH/PJC reviewed and contributed to the writing by critically evaluating the revisions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CJH has received consultancy fees from P1vital Ltd., Janssen Pharmaceuticals, Sage Therapeutics, Pfizer, Zogenix and Lundbeck. CJH holds grant income from UCB Pharma, Zogenix and Janssen Pharmaceuticals and holds grant income from a collaborative research project with Pfizer. She is also a company director of TnC Psychiatry and Neuroscience. EP and PJC declare no conflict of interest. MV is an employee of Hoffman-La Roche.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
