Abstract
Background and aim:
Severe eating disorders, such as obesity, bulimia, and anorexia, keep increasing to epidemic proportions worldwide. Understanding of neuropeptides’ role in complex hunger/satiety mechanisms may allow new prospects for treatment and prevention. Pyroglutamylated arginine-phenylalanine-amide peptides (QRFPs) are thought to enhance feeding following the central administration.
Methods:
In our study, QRFP-26 was delivered into the lateral hypothalamic area of male Wistar rats by direct microinjections, as QRFP-26 expressing neurons and binding sights are densely present in this neural structure. The consumption of liquid food was measured over 60-min.
Results:
Both doses (100 and 200 ng) significantly decreased food intake compared to the control treatment. Neuropeptide Y Y1R/NPFF (neuropeptide FF) antagonist BIBP3226 eliminated the anorexigenic effect caused by QRFP-26 administration. QRFP-26 affects neither general locomotion, behavioral patterns examined in the Open Field Test, nor anxiety.
Conclusion:
This study is the first to report the anorexigenic action of QRFP-26 following direct administration into the hypothalamus, emphasizing steady locomotion and anxiety levels. We have shown that the effect of QRFP can be linked to the neuropeptide Y (NPY) Y1 or NPFF receptors.
Introduction
Severe eating disorders, such as overweight and obesity, have increased to epidemic proportions, with over 4 million people dying each year according to the global burden of disease. These conditions are progressively spreading in adults and children not only in high-income countries, as was considered before, but all over the world (World Health Organization, 2021). On the other side of the spectrum, anorexia nervosa characterized by decreased body mass index represents an interface between metabolic and mental disorders. There is no other group of mental diseases with a mortality rate higher than that of eating disorders, the victims of which are up to 18 times more likely to attempt suicide compared to the average population (Smith et al., 2018). Therefore, the problem of eating disorders escalates from a purely medical question into a multiplex social issue.
Understanding complex hunger/satiety regulating mechanisms has pivotal importance in the management of such disturbances. A close relation of mental and metabolic regulation suggests a crucial role of neuropeptides in these processes. The RFamide peptides have been implicated in the control of multiple physiological functions including regulation of metabolism and feeding behavior. This peptide family includes several biologically active neuropeptides sharing RF motifs at their C-terminal end. Animal experiments have revealed that most of the RFamide peptides, such as neuropeptide FF (NPFF) (Bechtold and Luckman, 2006; Lawrence et al., 2002), prolactin-releasing peptide (PrRP) (Bechtold and Luckman, 2006; Lawrence et al., 2002), RFamide-related peptide-1 (RFRP-1) (Newmyer and Cline, 2009), and kisspeptin (Smith et al., 2006; Stengel et al., 2011), induce anorexigenic effects, while RFRP-3 is rather considered to enhance feeding (Johnson et al., 2007). Pyroglutamylated arginine-phenylalanine-amide peptides (QRFPs, QRFP-26, and QRFP-43) have been demonstrated to regulate feeding behavior as well. Acute intracerebroventricular (i.c.v.) treatment of QRF peptides dose-dependently increased appetite in mice (Chartrel et al., 2003; do Rego et al., 2006; Moriya et al., 2006; Takayasu et al., 2006), rats (Lectez et al., 2009), and birds (Ukena et al., 2010). Consistently, our earlier results also suggest that administration of QRFP into the medial hypothalamic parenchyma leads to an orexigenic effect (Zagoracz et al., 2015). Specific QRFP receptors have been detected in the diencephalon in high density (Bruzzone et al., 2007; Fukusumi et al., 2006; Kampe et al., 2006). Interestingly, QRFP-binding sites in the rat’s and human’s central nervous system (CNS) have a much wider distribution than QRFP receptors’ mRNA expression (Bruzzone et al., 2007) suggesting the involvement of nonspecific pathways.
Hypothalamic regulation of hunger/satiety mechanisms involves two distinct, functionally antagonistic groups of neurons in the arcuate nucleus (Arc): the orexigenic neuropeptide Y (NPY) and agouti-related peptide (AgRP)-expressing neurons on one side, and the anorexigenic pro-opiomelanocortin (POMC)-expressing neurons on the other. In response to food consumption, POMC cells activate melanocortin receptors (MCRs) on second-order neurons in the paraventricular (PVN), dorsomedial, ventromedial hypothalamic nuclei, and lateral hypothalamic area (LH/LHA) thus signaling satiety (Kleinridders et al., 2009; Waterson and Horvath, 2015). On the other hand, fasting or relevant mediators induce activation of the NPY/AgRP neurons that have similar projections but exert opposite effects: NPY directly stimulates food intake via NPY Y1 and Y5 receptors (Y1R and Y5R) and reduces energy expenditure, while AgRP acts as an inverse agonist of MCRs. The LH plays the role of inhibitor toward the PVN. One of the major appetite/satiety regulatory mediators from the periphery is leptin which performs its function via NPY (Mercer et al., 2011). It has been shown that leptin can also modify the expression of preproQRFP mRNA in the hypothalamus (Takayasu et al., 2006). Furthermore, a subpopulation of the NPY neurons expressing QRFP receptors was revealed in the Arc (Lectez et al., 2009). Also, several studies confirmed the appetite-reducing effects of Y1R antagonists against QRFPs (Lectez et al., 2009; Primeaux et al., 2013; Takayasu et al., 2006). These findings suggest a close interaction between QRFP and NPY systems, as well as the involvement of the LHA in the food-intake regulating network (for review, see Timper and Bruning, 2017). Nevertheless, the exact mechanisms and the relation of the proposed interactions to the classical theory of the hypothalamic regulation of feeding remain poorly understood.
Thus, taking into account that both hypothalamic regions play a crucial role in metabolic regulation in a counteracting manner; both regions are rich in QRFP-synthesizing cells and receptors; and our previous findings demonstrating orexigenic activity of QRFP in the medial hypothalamus, we hypothesized that administration of neuropeptide QRFP into the LHA would inhibit feeding behavior. The hypothesis was tested using intraparenchymal administration of two doses of the neuropeptide and the corresponding dose of non-peptide Y1/FF1 receptors antagonist BIBP3226. Despite the lack of information regarding the expression of NPY or NPFF receptors on QRFP neurons, the efficacy of BIBP3226 administration was described previously (Takayasu et al., 2006). To ensure that the observed changes were due to the modification of feeding behavior itself, the effects of substances on anxiety, general locomotion, and explorative activity have been investigated as well.
Materials and methods
The study conforms to the ARRIVE Guidelines. The experimental protocols were approved by the institutional (Animal Welfare Committee of Pécs University, Medical School) and national (Scientific Ethics Council for Animal Experiments) licensing committees. All the experiments were performed according to the protocols. The subjects, materials, and protocols of the present research were similar to our previous study (Zagoracz et al., 2015). A brief description of the applied materials and methods is given below.
Stereotaxic surgeries
In the present study, 83 male Wistar rats with 250–280 g body weight (bw; LATI, Gödöllő, Hungary) were housed and cared for in accordance with institutional (Animal Welfare Committee of Pécs University, Medical School; licenses No. BA02/2000-8/2012, BA02/2000-9/2018), national (Law XXVIII, 1998, Government Decree, 40/2013. (II.14) Hungary), and international standards (European Community Council Directive 86/609/EEC; 1986, 2006; European Directive 2010/63/EU of the European Parliament and the Council; National Institutes of Health Guidelines for Laboratory Animals, 1997). Rats were anesthetized by ketamine supplemented with diazepam (Calypsol, 80 mg/kg bw and Seduxen, 20 mg/kg bw; Richter, Hungary). Stainless steel guide tubes were implanted into the LHA (coordinates referring to the bregma: anteroposterior (AP): −2.8 mm, mediolateral (ML): 1.3 mm, and dorsoventral (DV): 7.5–8.3 mm ventral from the surface of the dura mater; Paxinos and Watson, 1986) and fixed to the skull with acrylic cement (Duracryl). Following the surgery, animals had a minimum of 5 days for postoperative recovery before the experimental procedures.
Drug injections
In all the experiments, rats were randomly allocated to control or treatment groups using random-number tables. Only the principal investigator was aware of the group allocation. Within one paradigm, all the groups received their treatment on the same day, during the light phase. For the intrahypothalamic microinjections, QRFP-26 (Rat; 048-72; Phoenix Pharmaceuticals Inc., Bulingame, CA, USA) was dissolved in 0.15 M sterile saline. On the days of testing, rats received injections of peptide in the appropriate dose (100 ng, 35 pmol or 200 ng, 70 pmol) or vehicle injection in the case of control measurement (0.15 M sterile saline).
Studies with NPY/FF receptor antagonist BIBP3226 (B174; Sigma-Aldrich Kft., Budapest, Hungary) on food intake were performed with double injection volume (0.4 + 0.4 µl) to each animal. Antagonist treatment involved the administration of an equimolar dose of BIBP3226 (18 ng, 38 pmol) 15 min prior to 100 ng QRFP-26 injection (Ant + QRFP-26), or BIBP3226 administration followed by vehicle injection (Ant + Vehicle).
In the case of behavioral experiments, only one group of animals received Ant injections equivalent to a 100 ng dose of peptide (the full cycle of antagonistic experiments was omitted). Since the peptide did not modify behavior, we considered this strategy acceptable for the sake of decreasing the number of involved animals. The same animals participated in the Open Field Test (OFT) and in the Elevated Plus Maze (EPM, Figure 1(c)).
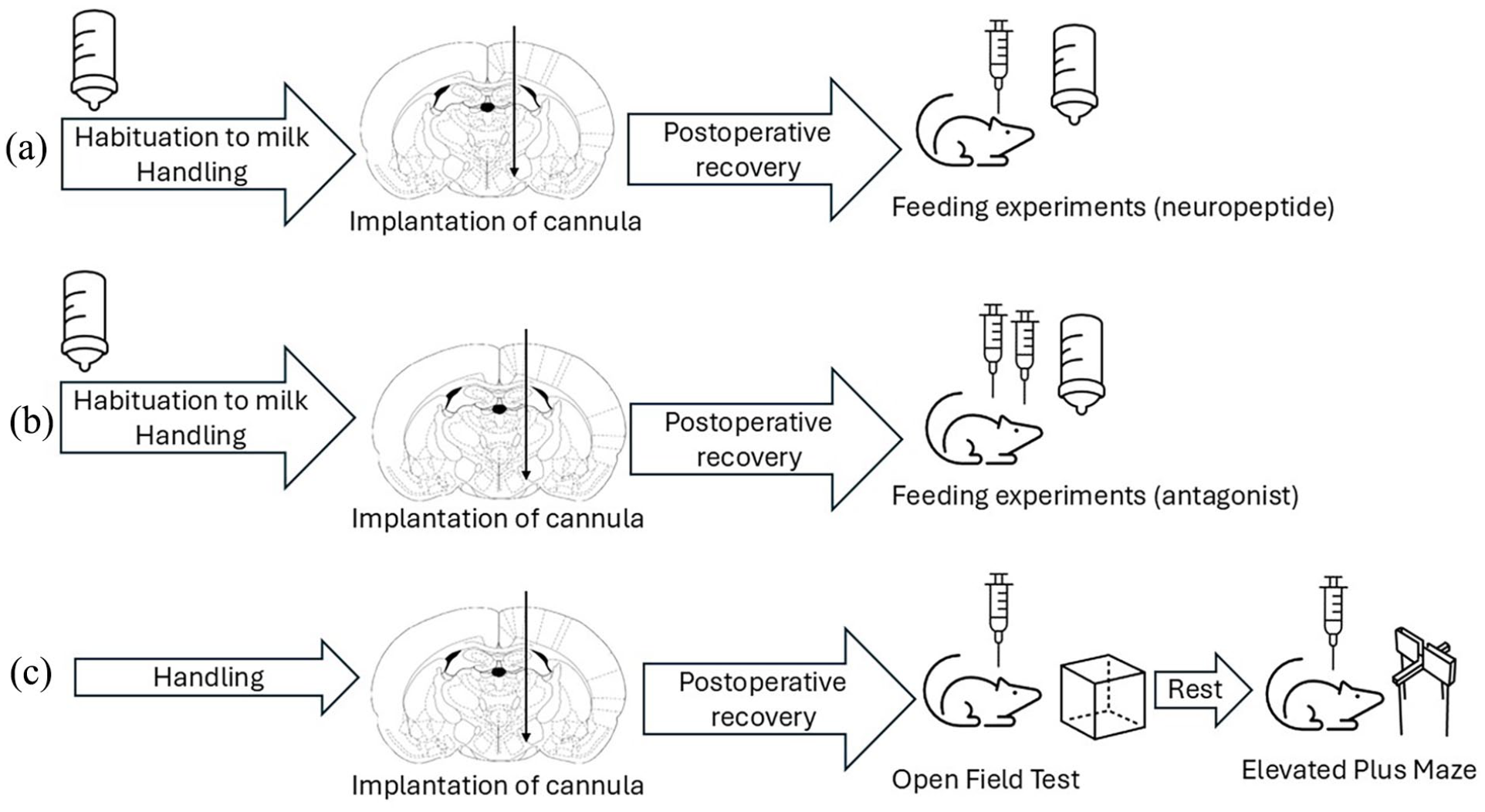
Schematic illustration of the experimental setup. (a) Feeding-related experiments with two doses of neuropeptide QRFP. (b) Feeding-related experiments with Y1/FF receptor antagonist BIBP3226 imply double injection volume. (c) Behavioral experiments.
All substances were injected through stainless steel conduits (injection cannulas, 27 gauge) extending 0.5 mm below the tips of the implanted guiding cannulas. The injection cannula was attached to a Hamilton microsyringe (10 µl; Bonaduz, Switzerland) via polyethylene tubing (PE-10). The drugs were applied by automated syringe pumps (Cole Parmer Instrument Company, LLC, Vernon Hills, IL, USA) for 1 min. The injection cannula was left in place for an additional 1 min to allow diffusion into surrounding tissues.
Food intake measurements
Numerous studies revealed that the liquid food paradigm is advantageous in many terms compared to standard chow measurements (Fekete et al., 2002, 2007; Toth et al., 2008; Vigh et al., 1999a, 1999b). Tap water and standard laboratory food chow (Charles River Laboratories, Budapest, Hungary) were available ad libitum before and between the experiments. To accustom rats to the experimental nutriment, 1 week prior to operation animals were habituated to consume the liquid food (milk, Isosource Standard Natur, Nestle). Graduated drinking cylinders were fixed to the home cages, and milk was available for the animals for 3 h a day. This feeding schedule was maintained until the end of the experiments. After the microinjections, liquid food intake was measured at milliliters accuracy every 5 min for the first half-an-hour and every 10 min for the following half-an-hour, so the 60-min measurement data were analyzed (Figure 1(a) and (b)).
Open field test
OFT was employed for measuring spontaneous motor activity and exploration behavior (Figure 1(c)). The experimental arena presented as a 50 × 50 × 50 cm gray-painted box with a floor virtually divided into 16 identical squares, thus marking central and peripheral zones of the field. The apparatus was provided with homogeneous illumination. Naive rats were placed in the center of the arena and allowed to explore the environment for 5 min (Habituation); afterward, they were returned to their home cages. In the following 2 days, the procedure was repeated for the sake of recording the level of basal activity (average of two sessions). On the last day (Test), animals received microinjection of QRFP, Ant, or vehicle, and the experimental procedure was repeated. The arena was cleaned and deodorized with acetic acid after each animal. All the environmental and animal-related pre-test conditions were identical for each rat. The distance moved in the arena was analyzed by Noldus EthoVison System (Noldus Information Technology, PA Wageningen, The Netherlands). Behavioral patterns, such as grooming activity and rearing, were analyzed on video recording. Time spent by the animals around the walls of the apparatus was recorded as an indirect indicator of anxiety.
Elevated plus maze
The anxiety level was evaluated using the EPM test (Figure 1(c)). The arena consisted of two opposite open arms (50 × 10 cm) and two opposite closed arms (50 × 10 × 40 cm) with walls but with no roof. The maze was elevated to a height of 100 cm from the floor. Following the drug administration, animals were placed into the center of the maze (central platform), facing one of the closed arms. Each rat was tested only once. The apparatus was provided with homogeneous deem light. The arena was cleaned and deodorized with acetic acid after each animal. All the environmental and animal-related pre-test conditions were identical for each rat trial lasted for 5 min, and during this period the time spent on the opened and closed arms and at the ends of the opened arms was recorded by the Noldus EthoVision system. It was considered an arm entry if all four paws crossed the border.
Histology
To confirm the positioning of cannulas, at the end of experiments, animals were anesthetized with urethane and perfused with 0.15 M saline followed by 10% formalin solution. Brains were extracted, sliced with a freezing microtome in 40 µm sections, and stained with cresyl violet. Injection sights were established according to the stereotaxic atlas (Paxinos and Watson, 1986). Only data from the rats with correctly placed cannulas were analyzed.
Statistical analysis
All results were expressed as a mean ± standard error of the mean (SEM). In feeding-related experiments, the cumulative food intake per 100 g bw was analyzed by repeated measures analysis of variance (ANOVA; SPSS for Windows 11.0, IBM TechXchange). The experimental design implicated that each animal served its control (i.e., within the subject design). Assumption criteria for the feeding experiments were tested by Q-q plot and run test.
Data from the behavioral experiments were analyzed by one-way (EPM) or two-way (OFT) ANOVA followed by the Tukey post hoc test (Kovács et al., 2012) due to the between-subjects design in these experiments. The statistical rejection criterion for all the experiments was established at p < 0.05 level.
The hypotheses were specified before the data were collected. The analytic plan was pre-specified and data-driven analyses are clearly identified and discussed.
Results
Histology
The stereotaxic operations were performed based on Paxinos and Watson’s atlas of the rat brain. Altogether 8 of 83 operated animals were excluded from data analysis. A schematic illustration of cannula placements is shown in Figure 2.
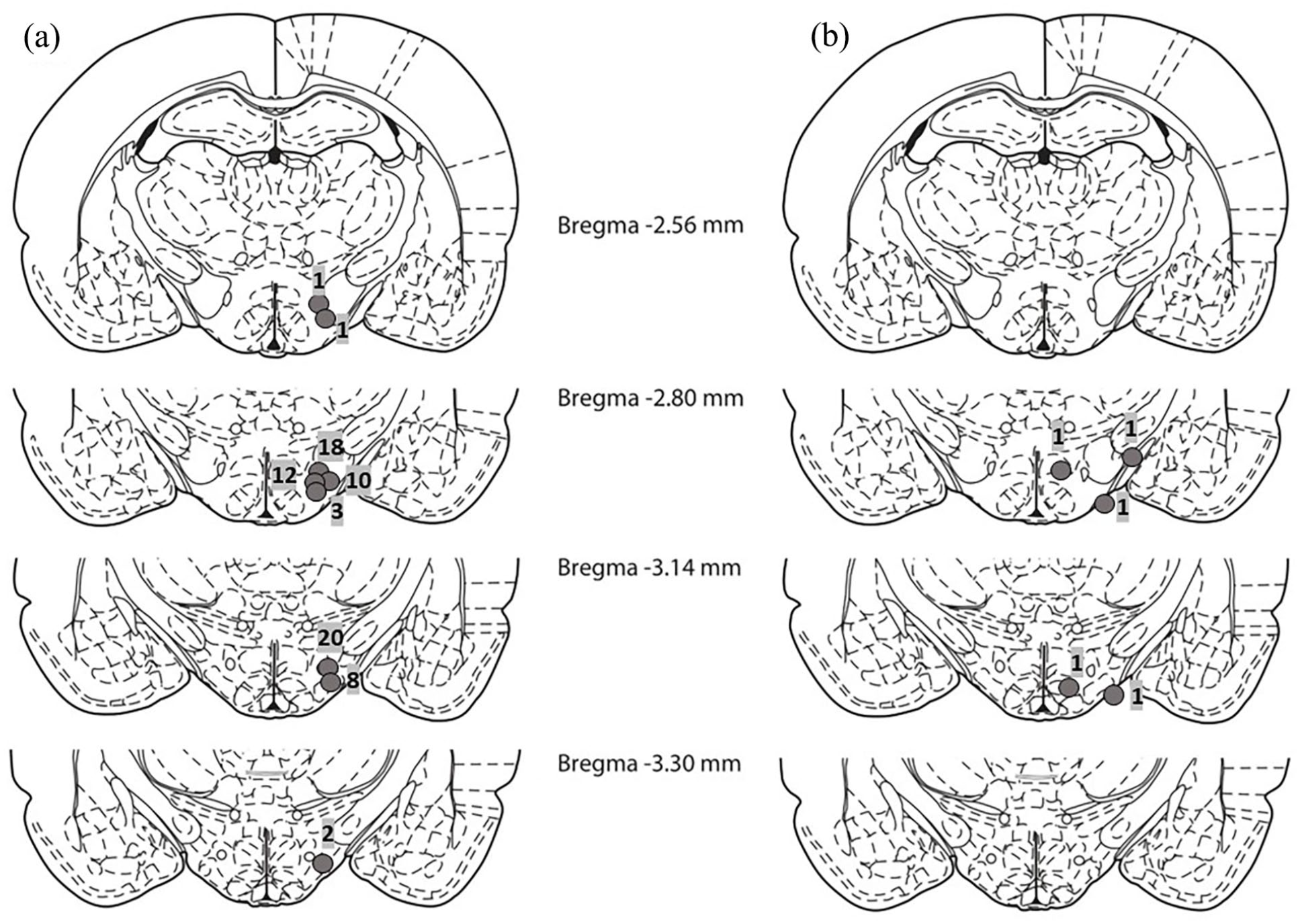
Schematic illustration of reconstructed injection sites. (a) Correct injection placements in the LHA (n = 75) and (b) incorrect injection placements (n = 5). Brain structure diagrams of coronal sections are adapted from the stereotaxic atlas of Paxinos and Watson. The numbers in the middle refer to the antero posterior distance from bregma in mm. The numbers above circle symbols on (a) and (b) indicate the numbers of animals.
In five animals, cannulas were mispositioned in the target area (Figure 2(b)). Among them, in three cases, cannulas were led to the medial hypothalamic nuclei or to the medial globus pallidus, in two other cannulas passed out of the brain.
Another three animals have been excluded from the experimental analysis due to their special characteristics: one rat was not able to drink milk from the sipper spout, and two rats’ crowns were damaged, making microinjecting impossible.
Feeding experiments
The neuropeptide QRFP injected into the LHA in 100 ng dose led to a significant decrease in food consumption compared to the control administration (Figure 3(a)). ANOVA analysis revealed a significant effect of time (F(8, 80) = 79.326, p < 0.01) and treatment (F(1, 10) = 11.271, p < 0.01), but not time × treatment interaction. Application of QRFP in 200 ng dose (Figure 3(b)) resulted in anorexigenic effects as well. ANOVA showed significant effect of time (F(8, 64) = 51.469, p < 0.01) and treatment (F(1, 8) = 8.113, p < 0.03), but again not their interaction.
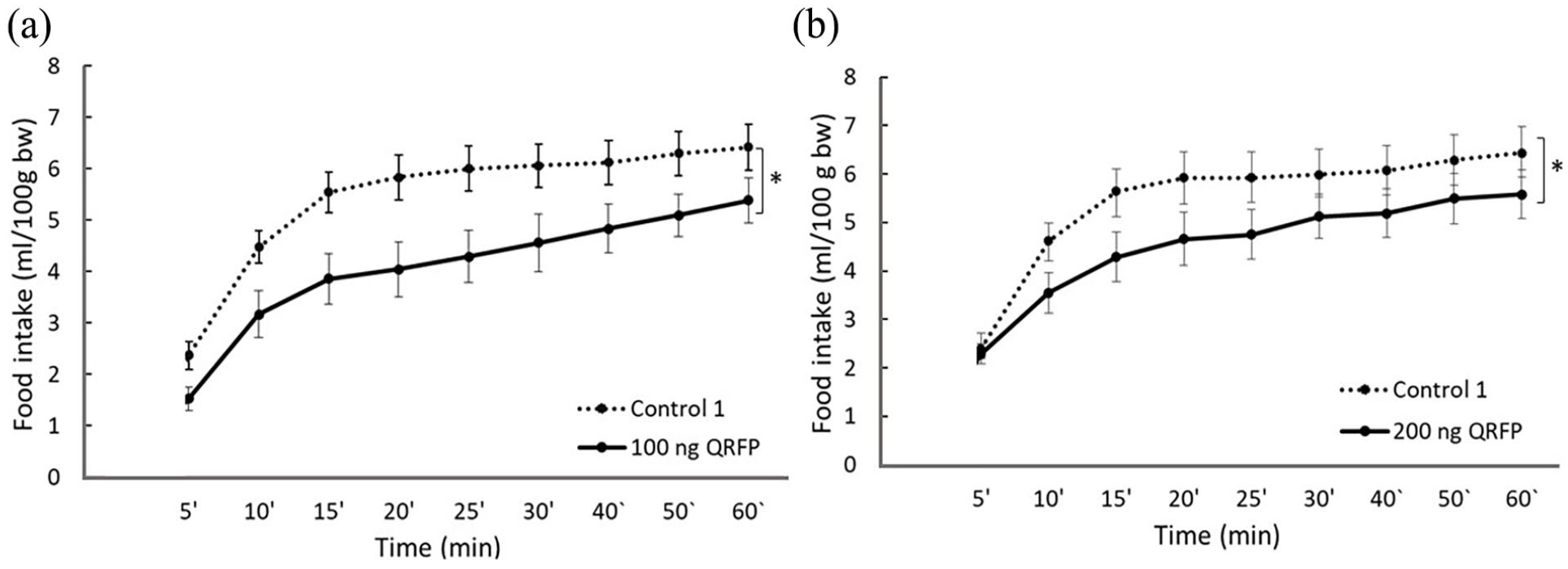
Feeding-related effects of QRFP microinjections into the LHA. Lines with symbols represent cumulative mean food intake in ml/100 g body weight (±SEM) after application of the peptide in different doses or vehicle microinjections (0.4 µl). (a) 100 ng QRFP versus Control 1 (n = 11), (b) 200 ng QRFP versus Control 1 (n = 9).
Effects of BIBP3226 in the LHA are presented in Figure 4. QRFP applied in double volume (Figure 4(a)) confirmed the previous data related to decreased food consumption. ANOVA yielded a significant effect of time (F(8, 72) = 71.800, p < 0.01), treatment (F(1, 9) = 20.882, p = 0.01), but not time × treatment interaction. Combined antagonist and neuropeptide QRFP treatment (Ant + QRFP, Figure 4(b)) led to the prevention of the anorexigenic effects induced by QRFP. Treatment with the antagonist itself in double volume (Ant + veh, Figure 4(c)) did not modify food intake. The volumes of consumed milk in the control and antagonist-treated groups were identical (p > 0.05).
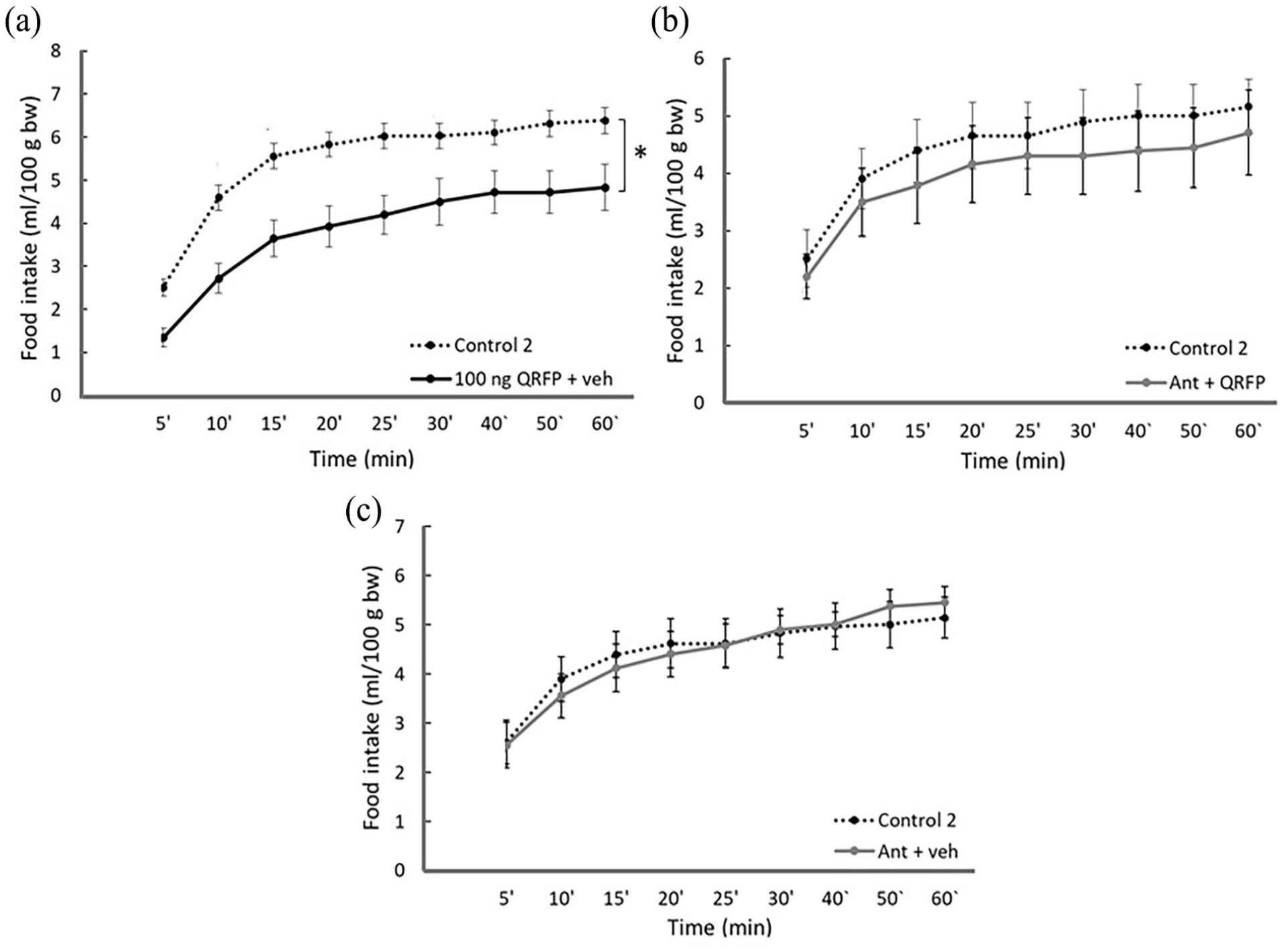
Feeding-related effects of BIBP3226 microinjections into the LHA. Lines with symbols represent cumulative mean food intake in ml/100 g body weight (±SEM) following the microinjections (0.4 + 0.4 µl). (a) Control 2 (vehicle + vehicle) versus 100 ng QRFP + vehicle (n = 10), (b) Control 2 versus Ant pretreatment followed by QRFP microinjection (Ant + QRFP, n = 7), and (c) Control 2 versus Ant + vehicle (n = 8).
Open field test
According to ANOVA, the distance moved in the OFT apparatus (Figure 5(a)) and the number of arena line crossings (Figure 5(b)) had no significant difference between or within the groups. At the same time, one-way ANOVA yielded a significant difference between the trials when analyzing the number of rearing episodes (F(1, 12) = 5.497, p < 0.01, Figure 5(c)) and between the groups in the case of grooming activity (F(3, 22) = 3.991, p < 0.03, Figure 5(d)). According to the post hoc test, Ant-treated animals had higher rearing activity compared to their basal activity (p < 0.01). They also spent significantly more time with grooming compared to controls and QRFP-treated rats during the test trial (p = 0.05, p < 0.04, p < 0.04, respectively).
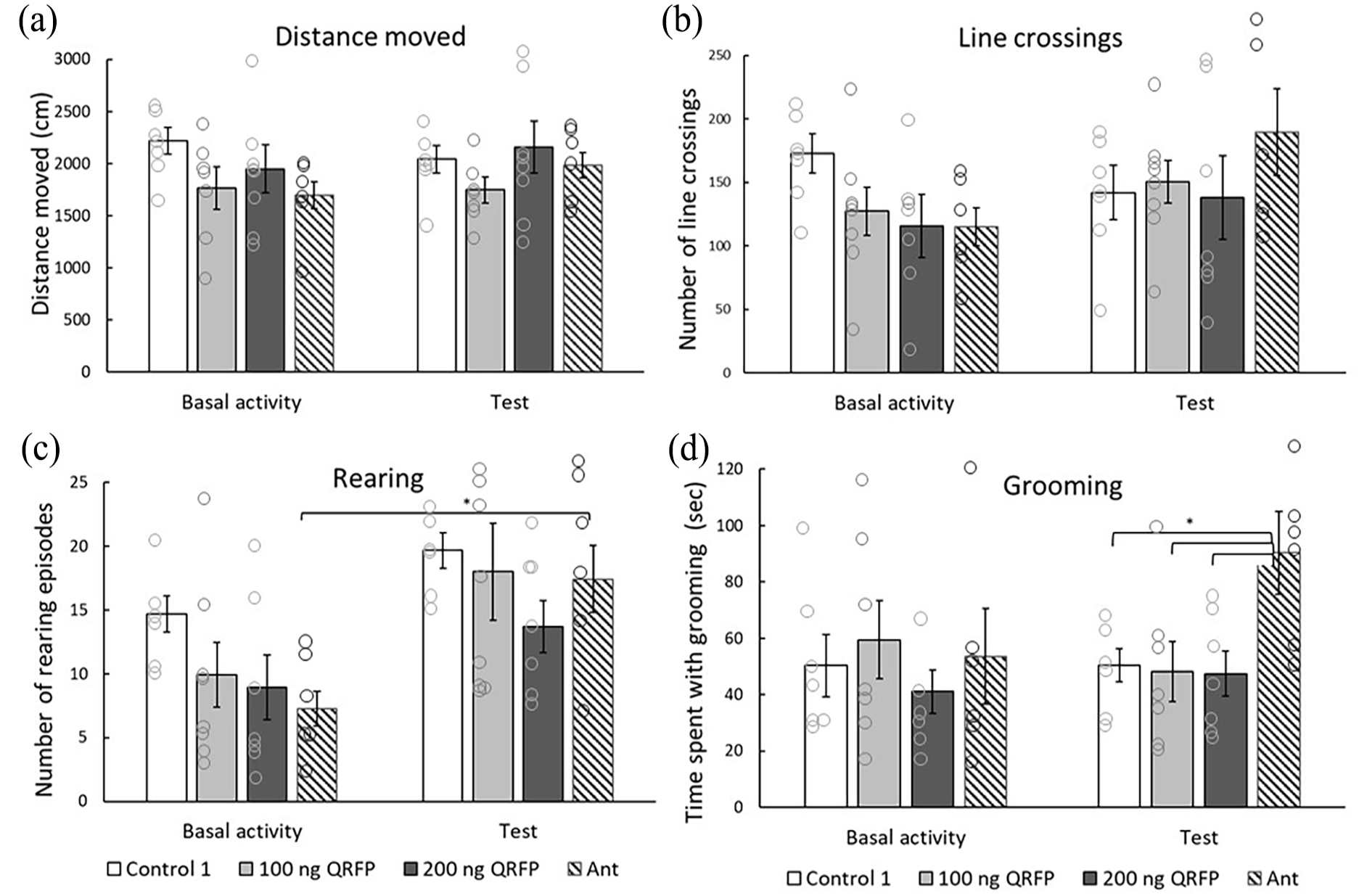
Effects of QRFP and BIBP3226 microinjections into the LHA on general locomotion in the open field test. Effects on general locomotion produced by Vehicle solution (Control 1, n = 7), 100 ng QRFP (n = 8), 200 ng QRFP (n = 7), Ant (n = 6). Columns represent (a) distance moved (cm) (±SEM), (b) number of virtual line crossings (±SEM), (c) number of rearing episodes (±SEM), and (d) time spent with grooming (s) (±SEM).
Elevated plus maze
According to one-way ANOVA, control animals as well as low- and high-dose QRFP-treated rats spent similar time in the closed and opened arms of the maze (Figure 6).
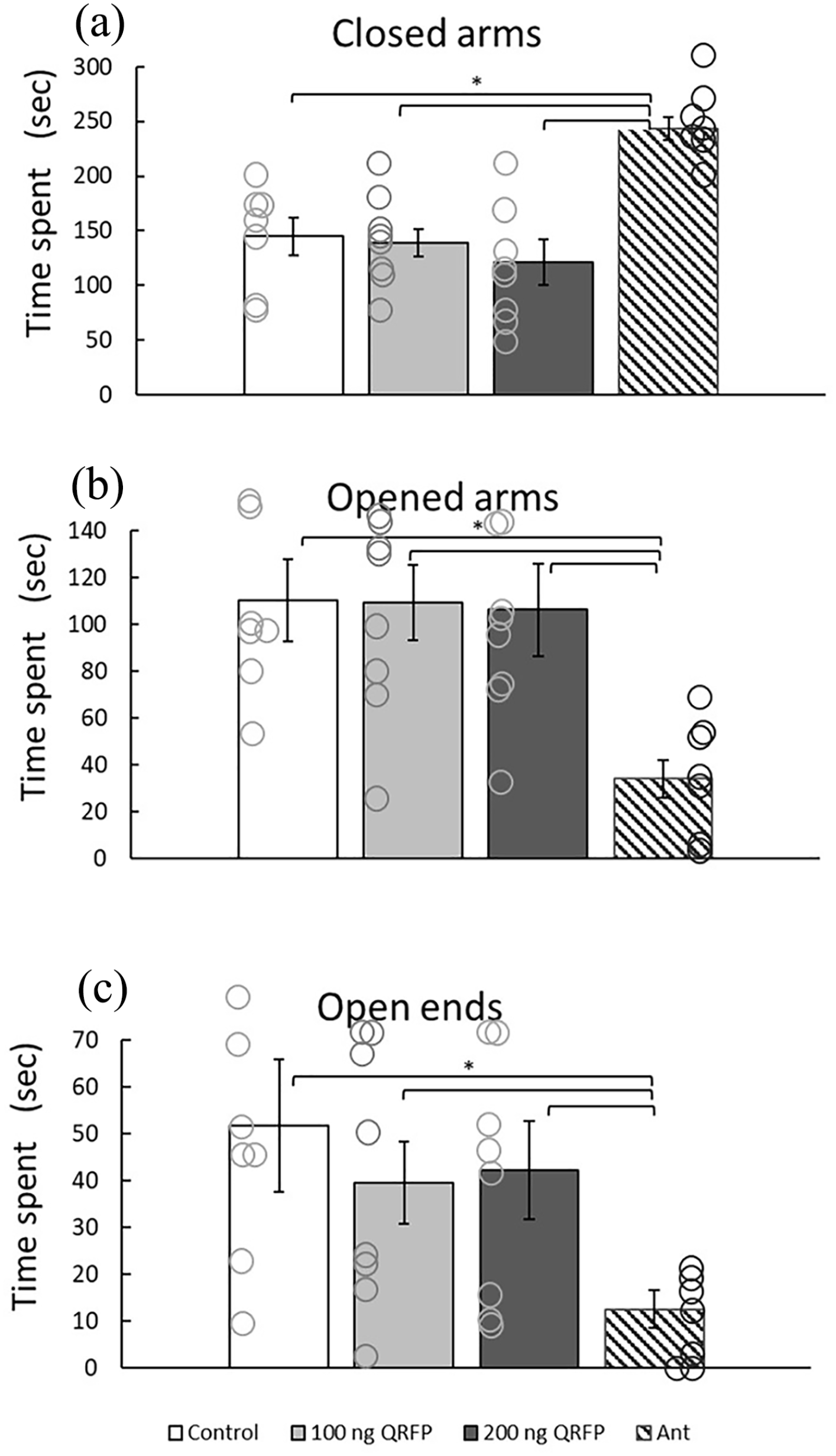
Effects of QRFP and BIBP3226 microinjections into the LHA on the anxiety level in the Elevated plus maze. Vehicle-treated rats (Control 1, n = 7), 100 ng QRFP (n = 8), 200 ng QRFP (n = 8), Ant (n = 7). Columns represent (a) the mean time spent in the closed arms (±SEM), (b) the mean time spent in the opened arms (±SEM), and (c) the mean time spent at the ends of the opened arms of the EPM apparatus (±SEM).
One-way ANOVA analysis of the experiment with Ant revealed a significant difference between the groups (F(3, 24) = 12.051, p < 0.01 for closed arms, F(3, 24) = 10.249, p = 0.01 for opened arms, and F(3, 24) = 6.330, p < 0.05 for the ends of the opened arms). In other words, BIBP3226 Ant treatment in the LHA led to longer time spent in closed arms, and shorter time spent in the opened space which suggests elevated anxiety.
Discussion
The QRFP neuropeptides have been intensively studied during the past 20 years mostly in regard to their regulation of metabolic functions (Chartrel et al., 2016). QRFP’s orexigenic effect has been observed in genetically modified mice, as well as in wild-type animals following i.c.v. (Chartrel et al., 2003; Takayasu et al., 2006) or direct microinjections into the parenchyma of the medial hypothalamic nuclei (Zagoracz et al., 2015). Nevertheless, the present findings suggest that the administration of QRFP-26 into the LHA leads to a decline in food consumption. Effective doses of QRFP delivered into the brain parenchyma have been established based on our previous experiments (Zagoracz et al., 2015). Both doses (100 and 200 ng) rapidly decreased animals’ food intake (Figure 3), and the neuropeptide acted within the first 5–10 min. However, the data represent the cumulative value of food intake; hence, the difference between the control and treated groups remained significant for a longer period. It is important to emphasize that our data were collected on healthy, young, male rodents. On one side, this way we minimized potential confounding factors. On the other hand, these restrictions mean limitations for the interpretation of the results. Further studies on female animals would uncover a possible role of estrogens in the QRFP-feeding regulation circuit, while experiments on elder rodents would give a better understanding of QRFP’s role in feeding disorders associated with aging.
According to the theory of hypothalamic “feeding and satiety centers,” stimulation of the LH should induce hunger and lead to hyperphagia (Anand and Brobeck, 1951; Shimizu et al., 1987). Our present findings verified the hypothesis and demonstrated that QRFP administration leads to opposite effects, suggesting that QRFP inhibits those elements/neurons of the LH that induce hyperphagia. The question of how it could be possible may arise.
To explain the anorexigenic effect observed in the LHA, an inhibiting element must have been included in the circuit regulating food consumption—either stimulation of inhibitory neurons or inhibition of appetite-promoting factors. Assumingly, the diffusion of injected into the brain parenchyma peptide is minimal, otherwise, we would detect orexigenic changes due to effects in the MH nuclei, as demonstrated earlier. Thus, at least first-order effector neurons should be located within the LHA. One of the options is that QRFP suppresses local NPY neurons in the LHA hunger center (Figure 7(a)) (Gray and Morley, 1986) {Gumbs, 2020 #7828}, rich in leptin and insulin receptors (El Mehdi et al., 2022), as well as in GABA neuronal cells and receptors {Jiang, 2022 #7852}. Y1R is also densely present within the LHA (Fetissov et al., 2004), and a group of Y1R-representing cells has been found to contain nitric oxide synthase (NOS) (Fetissov et al., 2003), a signaling molecule stimulating feeding in many species (Han et al., 2013). Thus, decreased NPY stimulus would lower the appetite. The phenotype of other Y1R-immunopositive neurons remains unknown (Campbell et al., 2003; Fetissov et al., 2003).
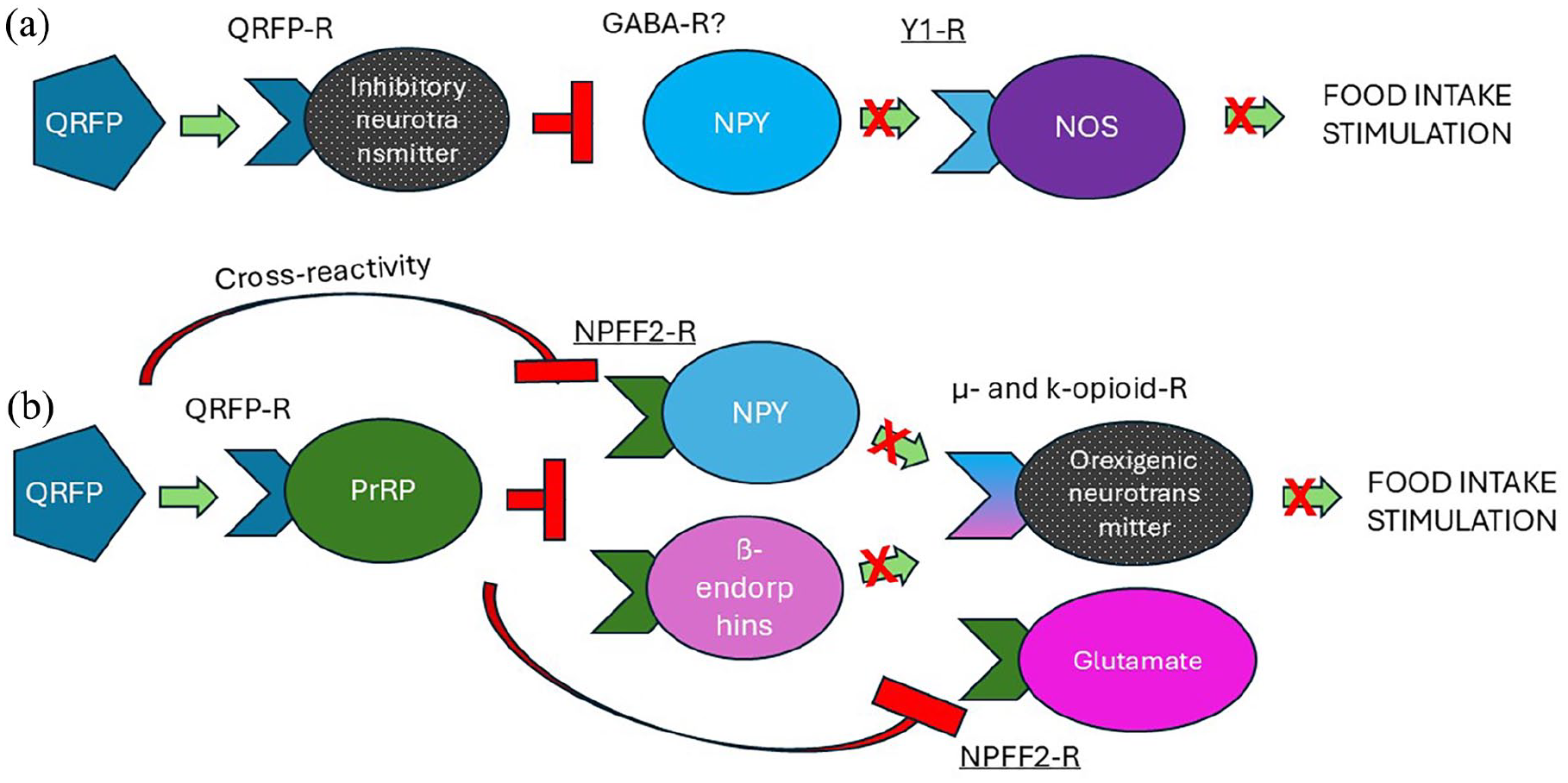
Possible neural pathways involved in anorexigenic effects of QRFP in the LHA. (a) Pathway involving NPY Y1 receptors: QRFP binds specific receptors present on the neurons with assumed inhibitory neurotransmitters. It ceases the release of NPY which would bind Y1 receptors expressed on the NOS, thus decreasing appetite. (b) The pathway involving NPFF FF2 receptors: QRFP binds specific receptors presented on PrRP neurons, which is a potent ligand for NPFF F2 receptors. Alternatively, there is a possibility of direct activation of F2 receptors by QRFP due to structural similarities. PrRP exerts an inhibitory action on F2 receptors-representing neurons, which could be NPY or beta-endorphin, potent opioid receptor ligands stimulating appetite. Inhibitory signaling might propagate toward glutamate neurons in the parabrachial nucleus, thus also blocking appetite. Green arrows represent facilitating stimuli, and red T-shaped lines are inhibitory stimuli. Black dotted circles represent hypothetical neurons with unknown neurotransmitters.
Specific QRFP receptors have been shown to share nearly 50% of the amino acidic identity with other G protein-coupled receptor subtypes: NPFF1, FF2, Orexin R1, and R2, the binding sites of the neuropeptides strongly involved in feeding regulation (Bonini et al., 2000; Jiang et al., 2003; Lee et al., 2001). Also, antagonist BIBP3226 has been shown to display antagonist properties toward both NPFF1 and FF2 receptors (Fang et al., 2006; Mollereau et al., 2002). Thus, cross-reactivity or a subsequent involvement of the NPFF receptors should be considered (Figure 7(b)). FF1 receptors subtype exhibits a higher affinity toward RFRPs (also referred to as neuropeptide SF and neuropeptide VF), while FF2 receptors are considered the endogenous receptors for NPFF itself along with the cousin neuropeptides NPAF and PrRP (Liu et al., 2001; Yoshida et al., 2003). In addition, the distribution of FF receptors is uneven: the LHA is abundant in FF2 receptors mRNA and lacks FF1, whereas medial hypothalamic nuclei demonstrate opposite characteristics (Gouarderes et al., 2004). Hence, one of the possible mechanisms of anorexigenic effects of QRFP implies direct (by cross-reactivity) or indirect (by activation of potent ligand) stimulation of NPFF2 receptors in the LHA. Out of the three potent ligands mentioned above, PrRP-expressing cell bodies and fibers have been detected in the LHA (Dodd and Luckman, 2013). Whether these neurons express QRFP-Rs or not is a topic of further research, but presumably QRFP could stimulate PrRP expression and its binding to NPFF2 receptors. The general anorexigenic effect of the FF system has been shown to involve central μ and ƙ opioid receptors (Cline and Mathews, 2008). However, as NPFF and PrRP cannot bind to opioid receptors, reduction in food intake is mediated by innate opioid ligands, for example, NPY or β-endorphins. These data are supported by the findings that concentrations of both ƙ and μ receptors were strongly modified in obese mice (Khawaja et al., 1989). FF2 receptors have also been detected in the parabrachial nucleus (PBN) (Gouarderes et al., 2004), so NPFF has been shown to depress excitatory glutamatergic transmission in the PBN neurons (Chen et al., 2000). Even though it is unlikely that QRFP injection-induced signaling would reach that far within such a short time, we would like to mention this theoretical pathway.
Generally, a decrease in food intake can be also caused by nonspecific effects of the applied drugs, for example, reduced locomotion or developed anxiety and corresponding freezing reaction. Indeed, QRFP knockout mice exhibited anxiety-like behavior (Okamoto et al., 2016). In agreement with these data, another study established the anxiolytic effect of GPR103 (QRFP-R) agonists in mice (Palotai and Telegdy, 2016). Interestingly, a recent human study on aggressive individuals revealed that the plasma levels of QRFP positively correlated with the anxiety scores in all the studied subjects and controls (Vaeroy et al., 2023). Nevertheless, in our study, QRFP applied to the LHA did not modify general locomotion, nor the anxiety level of tested animals (Figures 5 and 6). Assumingly, the applied doses were too small to reach the anxiety threshold. On the other hand, the present research design critically differs from previously mentioned studies as QRFP was applied locally so that microinjections did not affect the whole CNS. To sum up, the observed decrease in food intake seems to be linked to the appetitive mechanisms themselves and is not affected by changes in locomotion or anxiety.
In addition, administration of Y1/FF antagonist BIBP3226 led to enhanced rearing and grooming, and avoidance of the open space (opened arms in EPM test) in rats, referring to the anxiogenic effect (Figures 5 and 6). There are only a few data available regarding the anxiogenic role of NPFF2 {Lin, 2017 #7866}. However, the important role of the NPY system in the regulation of anxiety and depression has been known for a long time (Kask et al., 2002; Widerlöv et al., 1988). In recent studies, a significant decrease in NPY mRNA and upregulation of Y1R and Y2R mRNA, along with a significant decrease in expression of the NPY protein in the brains of suicidal subjects, have been reported (Carboni et al., 2022; Sharma et al., 2022). Various conditioning paradigms showed that NPY is a strong suppressor of fear, and the role of Y1R is crucial (for review, see Comeras et al., 2019). Experiments on mice also revealed that NPY neurons of the nucleus accumbens involved in the regulation of anxiety behavior have a direct and exclusive connection with the LH (Yamada et al., 2021). Therefore, our findings support the concept of NPY Y1 receptor subtype playing a role in stress and anxiety responses (Morales-Medina et al., 2010).
To summarize, the administration of neuropeptide QRFP-26 into the LHA leads to a decrease in food consumption. The anorexigenic phenomenon is most likely to be linked to QRFP-NPY(Y1) or QRFP-NPFF (FF2) regulating pathways, as Y1/FF receptor antagonist BIBP3226 prevented the appetite-modulating effect of QRFP-26. The possible involvement of GABA, opioid, and other neurotransmitter systems may be a subject for further research. QRFP-26 did not modify general locomotion or anxiety levels in the applied doses. In addition, our observations suggest that Y1/FF Ant treatment itself induces anxiogenic behavior following the intralaterohypothalamic microinjections, thus supporting the hypothesis regarding the involvement of NPY in stress and anxiety regulation.
Footnotes
Acknowledgements
The authors express their gratitude to Anna Schulteisz, Erika Szábó, Erzsébet Korona, Tiborné Takács, Ferenc Wilhelm, Ákos Klonga, András Belvárácz, and Dr. Anita Hegedűs for their technical contribution to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the University of Pécs Medical School, Pécs, Hungary (PTE ÁOK/KA-2022-23) and by the Hungarian Academy of Sciences.
