Abstract
Introduction:
Eating disorders are characterized by maladaptive eating behaviors and preoccupations around body shape, weight, and eating. The serotonin system has been among the most widely studied neurobiological factors in relation to eating disorders. Recent research also highlighted the role of oxytocin.
Aims and methods:
This article aims to review animal and human studies on the involvement of central serotonin and oxytocin, and their interplay in eating disorders in particular. We synthesize results from studies using animal models of eating disorders and from research conducted in healthy individuals and clinical populations.
Results/Outcomes:
Altered serotonin neurotransmission and oxytocin levels in the brain—particularly in the hypothalamus, brainstem, and limbic regions—were associated with disturbances in eating behaviors and related maladaptive cognitions and emotions. These brain regions were found to constitute a typical neural network through which both central serotonin and oxytocin might operate in a bidirectional manner.
Conclusions/Interpretation:
Based on the preceding findings, we describe a developmental biopsychosocial model relevant to eating disorders, including the role of serotonin–oxytocin interactions in the brain. While it is clear that eating disorders are multifactorial in which many biopsychosocial pathways are involved, the current review highlights the importance of well-designed translational research when studying mechanisms of serotonin–oxytocin interactions in the brain. Such research would help to better understand the effects of joint central oxytocin and serotonin administration as a possible preventive or therapeutic intervention for eating disorders.
Introduction
Eating disorders (EDs), including Anorexia Nervosa (AN), Bulimia Nervosa (BN), and Binge Eating Disorder (BED)—the worldwide lifetime prevalence of which has been estimated to be approximately 4%–5%—have been defined as psychiatric disorders characterized by disturbances in eating behaviors, and related preoccupations around body shape, weight, and eating (Treasure et al., 2020). The widely known approach to the pathogenesis of EDs is the interplay of environmental factors with genetic predisposition (Booij and Steiger, 2020; Brewerton, 2022; Bulik et al., 2022; Le et al., 2017; Wade et al., 2000). Although the exact mechanism underpinning this interplay is still unclear, a widely accepted assumption of environmental effects on neurobiological processes is the epigenetic modification of key regulatory genes (Booij and Steiger, 2020). Dynamic epigenetic processes help fine-tune the gene expression repertoire in response to environmental stimuli (Jaenisch and Bird, 2003). For example, higher hippocampal serotonin (5-hydroxytryptamine; (5-HT)) 1A receptor expression was reported in individuals with posttraumatic stress disorder relative to controls (reviewed by Lewis et al., 2020), and among unmedicated participants with a current depressive episode, who were exposed to childhood adversity (Bartlett et al., 2023). In addition, recent life stress was associated with elevated 5-HT1A receptor levels in the amygdala, insula, and orbital cortex of depressed individuals in another cohort (Galfalvy et al., 2023). Similarly, heightened 5-HT1A receptor density in the fronto-temporo-parietal brain regions was observed in individuals with EDs (not only in actively ill but also in weight-restored individuals) relative to healthy controls (reviewed by Bailer and Kaye, 2011; Gianni et al., 2020). In other words, environmental stressors might disrupt developmental trajectories and outcomes by interfering with the neurobiological framework (Nelson, 2013), changing receptors’ expression, which, in turn, can increase individual vulnerability to psychiatric disorders, including EDs.
Over the years, many neurobiological systems have been linked to EDs (e.g., Bulik et al., 2022; von Hausswolff-Juhlin et al., 2015), with 5-HT possibly as the most commonly investigated system (e.g., Bailer and Kaye, 2011; Frank, 2015). Alterations in central 5-HT system functioning are thought to contribute to dysregulation in eating behaviors as well as to related cognitive and emotional correlates of EDs, including perfectionism, impulsivity, and emotional reactivity (Steiger et al., 2010). Such alterations may occur at various levels, including (but not limited to) the functioning of multiple receptors (e.g., 5-HT1A, 5-HT2A), the serotonin transporter (5-HTT/SLC6A4), enzyme involved in amine neurotransmitters’ metabolism (monoamine oxidase A; (MAOA)), and rate-limiting enzymes for brain 5-HT synthesis (tryptophan hydroxylase 1 and 2; (TPH1, TPH2)) (Steiger et al., 2023). It is important to highlight the multiple layers of serotonergic neurotransmission control, with 5-HT1A autoreceptors localized on the cell body attenuating the firing rate of the serotonergic neurons, and the 5-HT1B autoreceptors at the axon terminals blocking the synaptic 5-HT release. These receptors can also act as heteroreceptors, controlling other neurotransmitters’ release in different brain regions (Garcia-Garcia et al., 2014; Sari, 2004). Therefore, their multiple roles should be taken into account when interpreting the consequences of their pharmacological stimulation/inhibition via systemic administration of agonist or antagonist drugs. Moreover, the functions of 5-HT1A receptors can change throughout developmental phases. Therefore, we should consider both their spatial and temporal roles when interpreting experimental results (Albert and Vahid-Ansari, 2019).
In addition to 5-HT, research has supported the role of oxytocin (OT) in the pathophysiology of EDs (e.g., Giel et al., 2018; Romano et al., 2020; Spetter and Hallschmid, 2017; Russell and Hunt, 2023). Like 5-HT, OT has been shown to play a significant role in regulating affect, cognitive control, metabolism, stress, and social behaviors (e.g. Neumann, 2008; Spetter et al., 2018), all important correlates of risk or maintenance factors for disordered eating behaviors. Also, OT has been found to stimulate the release of 5-HT and modulate its availability in the primates’ central nervous system (Lefevre et al., 2017). Hence, the therapeutic effects of central oxytocinergic and serotonergic stimulation might shed light on the neurobiological mechanisms in eating patterns, namely the regulation of caloric intake, metabolism, and weight (for review, see Giel et al., 2018; Lam et al., 2010; Neumann and Slattery, 2016).
The current paper aims to review specifically the role of 5-HT and OT, particularly their interactions, in the (risk for) disordered eating. It is clear that EDs have multifactorial causes and involve various neurobiological (e.g., metabolic, immunological, neurodevelopmental) substrates (Bulik et al., 2022), which is why alterations in 5-HT and OT would not necessarily fully explain the variance in ED risk for a given individual. That being said, synthesizing results of research conducted in animals and humans on 5-HT and OT and their interactions is relevant, considering that these two biological processes are among the most widely studied neurobiological factors in relation to eating behaviors and EDs. In spite of extensive animal work on maladaptive eating behaviors, translational studies in humans remain scarce. While this limits the applicability of existing research evidence in the context of EDs, animal models including (but not limited to) an activity-based anorexia model have generated certain key symptoms in individuals with AN (Schalla and Stengel, 2019), hence ensuring their relevance in the current review. Furthermore, in light of the view that EDs have neurodevelopmental origins (Connan et al., 2003; Vannucci et al., 2015), we discuss the role of the central 5-HT and OT in EDs from a neurodevelopmental perspective. Next, we review studies assessing the impact of serotonergic intervention on brain OT and vice versa, those examining the effect of OT on brain 5-HT. We conclude by synthesizing findings from animal models and human studies and describe a developmental biopsychosocial pathway relevant to EDs that includes the role of 5-HT-OT interactions in the brain. Avenues for future research are also discussed.
Brain serotonin and disordered eating
Development of the 5-HT system
Serotonin is a molecule derived from an amino acid called tryptophan, synthesized mainly in the neuroendocrine cells of the stomach and the intestines by TPH1 and decarboxylase enzymes. Behaving as a paracrine messenger in the periphery, 5-HT is also released into the blood, where platelets take it up and release it upon their activation (see review by Jones et al., 2020). It acts as a neurotransmitter in the enteric nervous system, and in the central nervous system where TPH2 isoform is predominant (reviewed by Pratelli and Pasqualetti, 2020). For the current review, we will mainly focus on brain 5-HT. The central serotonergic neurons are located in the raphe nuclei, and project to almost all brain regions, most importantly to the prefrontal and cingulate cortical regions, the limbic system, and the hypothalamus (Booij et al., 2015). Serotonergic pathways’ development in the brain, including synthesis, innervation, and fiber density, has been reviewed extensively (e.g., Booij et al., 2015; Brummelte et al., 2017). Research findings indicate that the 5-HT neurons are one of the earliest to appear in the developing living organism, emerging as early as the first month of the gestational period. Moreover, each component of the central 5-HT system (e.g., receptors, enzymes) has a unique role in brain development, including neurogenesis, neuronal differentiation, axon myelination, synaptic remodeling, pruning, and maintenance (Booij et al., 2015; Brummelte et al., 2017).
Alterations in the central 5-HT system, especially early in life, may lead to long-lasting changes in neurotransmission and brain structure. Indeed, findings of pharmacological studies and genetic (“knock-out” and “knock-in”) animal models that experimentally induced 5-HT depletion during prenatal and early postnatal periods showed associations between persistent reductions in 5-HT concentration and the volume of adult rats’ brain regions, including frontal cortex, hippocampus, thalamus, and raphe nuclei (e.g., Ellegood et al., 2018; Ghaheri et al., 2022; Migliarini et al., 2013).
Early environmental adverse factors (such as chronic stress during pregnancy or in infancy) could also alter brain serotonergic activity. For instance, adolescent and adult offspring of rodent and primate mothers subjected to stress during pregnancy—in the form of restraint stress, exposure to nicotine, or altered diet (ethanol-based, high-fat, or protein-deprived)—displayed an increase in TPH2, 5-HTT, and 5-HT2A receptors’ availability in the raphe, hippocampal, and frontal cortical regions (e.g., Miyagawa et al., 2011; Mokler et al., 2003; Muneoka et al., 2001; Ngai et al., 2015; Slotkin et al., 2007; Sullivan et al., 2010; Van den Hove et al., 2014). A decrease in 5-HT1A receptor availability in the cerebral cortex and brainstem has also been reported in adult male rats after perinatal nicotine exposure (Slotkin et al., 2007). In addition, exposure to early-life social stress in the form of parental deprivation (peer-reared condition) in rhesus monkeys has been associated with a decrease in 5-HT1A receptor density and reduced 5-HTT availability in the hypothalamus, hippocampus, and raphe (Ichise et al., 2006; Spinelli et al., 2010). However, inconsistent results have been reported regarding the 5-HT concentration in the prefrontal cortex in adult male offspring exposed to postnatal maternal separation, with some findings showing an increase (Xue et al., 2013) and others indicating a decrease (Récamier-Carballo et al., 2017).
Possible detrimental effects of prenatal adversity on central 5-HT functioning have been observed in human studies. However, studies are mostly correlational. For instance, 5- to 12-year-old children with fetal alcohol syndrome exhibited lowered 5-HTT availability in the prefrontal cortex relative to control children (Riikonen et al., 2005). In addition, exposure to perinatal adversity, including in utero exposure to nicotine or experiencing asphyxia at birth, has also been linked to lowered 5-HT synthesis capacity in the hippocampus and the orbitofrontal cortex in healthy adults (Booij et al., 2012). Overall, the evidence highlights the importance of 5-HT for the developing brain and the susceptibility of the 5-HT system to early adverse environmental experiences.
The developmental role of central 5-HT: Relevance for EDs
Various research groups examined rodent eating behaviors, measuring different serotonergic components in the brain and using paradigms that include food restriction (acute restraint or starvation) and activity-based models (for review, see Scharner and Stengel, 2021). For instance, a decrease in 5-HTT availability in the frontal cortex has been observed in young adult rats experiencing up to 2 weeks of food restriction, relative to freely fed controls (Huether et al., 1997). Research using the food restriction models showed that pharmacological stimulation of the 5-HT1A receptor in the nucleus accumbens (NAcc) was associated with a dose-dependent reduction in food intake in food-restricted rats (Clissold et al., 2013). Later on, Pratt et al. (2016) reported only transiently inhibited feeding in food-deprived mice after stimulation of 5-HT1A and 5-HT1B receptors, when the drugs were injected in the ventral tegmental area (VTA). Stimulation of 5-HT1B and 5-HT2C receptors in the raphe and hypothalamus was associated with hypophagia in food-deprived mice (reviewed by Compan et al., 2015). Similarly, 5-HT4 receptor stimulation in the NAcc has been shown to induce hypophagia in food-restricted rats (Jean et al., 2012, 2017). The (in)activity of 5-HT3,5,6 receptors has not been consistently linked to changes in food intake and body weight (reviewed by Compan, 2013, 2020). The preceding findings suggest that brain 5-HT effects on eating behaviors depend on the brain area stimulated, the dose administered, and the type of receptors targeted (Pratt et al., 2016). Nonetheless, while increased central 5-HT level/activity may contribute to hypophagia, decreased central 5-HT levels/activity may contribute to hyperphagia (Compan, 2020).
Opposite effects of serotonergic drugs have been shown in activity-based anorexia models in which rodents have restricted food access along with access to a running wheel (Schalla and Stengel, 2019). Activity-based symptoms of anorexia were intensified following a primarily peripheral increase of 5-HT (e.g., via subcutaneous or intraperitoneal administration of 5-HT releasing fenfluramine) while the same effect would be observed following a central 5-HT reduction (e.g., via an irreversible TPH enzyme inhibitor parachlorophenylalanine administration; reviewed by Schalla and Stengel, 2019).
Furthermore, the modulatory role of substance use such as nicotine in both brain 5-HT functioning and food intake has been studied. A 3-day-long food deprivation (compared to ad libitum feeding regime) in rats was associated with a decrease in serotonergic neurons in the raphe, which, in turn, was linked to an increased desire for food (Lee et al., 2002). Co-administration of intraperitoneal nicotine to these food-deprived rats counteracted this effect by increasing the number of serotonergic cells in the raphe, which, in turn, was linked to a suppressed food desire (Lee et al., 2002). Such findings are relevant for AN, an ED characterized by food restriction and, at least in a subgroup of individuals, a heightened rate of smoking and nicotine dependence (Anzengruber et al., 2006). Indeed, alterations in the brain 5-HT levels might influence our susceptibility to such reinforcers as tobacco and food (Erritzoe et al., 2009), increasing vulnerability to various disorders, including EDs.
In living humans, 5-HT effects on mood, cognition, and food intake can be studied reliably in the laboratory with acute tryptophan depletion—a method used to experimentally lower 5-HT levels by administering an amino acid mixture that includes all essential amino acids except tryptophan, thereby temporarily reducing 5-HT synthesis (Young, 2013). Research has shown that acute tryptophan depletion leads to increases in depressive symptoms, anxiety, irritability, desire to binge, food intake, and body image-related fears in individuals currently diagnosed with or recovered from BN, compared to healthy controls (e.g., Bruce et al., 2009; Kaye et al., 2000; Smith et al., 1999; Weltzin et al., 1995). In individuals diagnosed with AN, one study found that, relative to controls, acute tryptophan depletion led to decreased anxiety among currently symptomatic and recovered women (Kaye et al., 2003), while another study did not find any significant change in either mood or anxiety among individuals recovered from AN (see Weinert et al., 2023). Further research is necessary to clarify the link between 5-HT neurotransmission and psychological functioning in individuals recovered from AN.
No acute tryptophan depletion studies so far have been conducted in individuals with BED. In the general population, an ad libitum consumption of high-carbohydrate (vs high-protein or mixed) meals has been associated with an increased tryptophan to other large neutral amino acid ratio (Gendall and Joyce, 2000; Wurtman et al., 2003). Blood tryptophan level has been negatively associated with the desire to binge, regardless of the meal type (Gendall and Joyce, 2000). It could be argued that, unlike healthy individuals in whom tryptophan availability and food intake work together to regulate appetite and eating patterns, individuals with (or susceptible to) BN present a disturbed relation between tryptophan, 5-HT, and food intake. On one hand, lowered tryptophan levels (induced by dietary restriction) can trigger binge-eating episodes, thereby perpetuating a cycle of bingeing and purging. On the other hand, food intake in individuals with AN may contribute to alterations (most likely, increase) in 5-HT levels and to dysphoric mood. This, in turn, increases the likelihood of these individuals re-engaging in fasting to reduce distress (Kaye et al., 2009). The preceding would perpetuate a reciprocal relation between 5-HT disturbances, impaired psychological functioning, and maladaptive (restrictive) eating behaviors despite self-starvation.
Human research on neurobiological correlates of EDs has used brain imaging techniques of single-photon emission computed tomography (SPECT) and positron emission tomography (PET) combined with various radioligands to characterize the functioning of the brain serotonergic components in vivo. Although PET studies are usually not feasible in research on a large-scale basis and are primarily correlational, PET is, to date, the most direct method to study neurotransmitters in the living human brain (Ceccarini et al., 2020). Considering the clinical presentation of EDs involving a complex interplay between motivational processes, cognitions, and affect, PET studies provide insight into neurochemical correlates of ED symptoms that animal models cannot address. Indeed, findings of PET neuroimaging research comparing the functioning of elements in the brain serotonergic system of healthy adults and those with AN and BN showed heightened 5-HT1A receptor activity (represented by an increase in [11C]WAY100635 or [18F]MPPF ligand’s binding potential) in the clinical groups (for review, see e.g., Frank et al., 2019). In addition, lowered central 5-HTT activity (indicated by reduced [11C]DASB, [11C]MADAM, or [123I]nor-beta-CIT ligands’ binding potential) in the hypothalamus, NAcc, and raphe was observed in individuals diagnosed with AN, BED, and in those diagnosed with or recovered from BN (vs healthy controls) (e.g., Majuri et al., 2017; Mele et al., 2020; Pichika et al., 2012; Tauscher et al., 2001; Yokokura et al., 2019).
Overall, animal and human research supported the role of the 5-HT system in disordered eating. Alterations in the functioning of central 5-HT components, including a decrease in 5-HTT in the raphe and an increase in 5-HT1A receptors’ activity in the frontal brain regions and the hypothalamus, have been observed in hypophagic food-deprived animals as well as in individuals with EDs.
Brain OT and disordered eating
Development of the OT system
OT is a neuropeptide mainly produced by hypothalamic neurons projecting to the pituitary gland and other parts of the central nervous system (Grinevich et al., 2016; Quintana and Guastella, 2020) to influence various behaviors including eating and social behaviors (Caldwell et al., 2017; Giel et al., 2018; MacDonald and MacDonald, 2010). The major part of OT production occurs in the paraventricular and supraoptic nuclei shortly after birth (Altstein and Gainer, 1988; Choy and Watkins, 1979; Grinevich et al., 2016), with OT receptor (OXTR) emerging as early as the first day after birth in rats (Shapiro and Insel, 1989; Yoshimura et al., 1996) and potentially within the first postnatal weeks in primates (Vaidyanathan and Hammock, 2017).
OT operates through its receptor, expressed in the amygdala, hippocampus, NAcc, and hypothalamus (Geerling et al., 2010; Gould and Zingg, 2003; Vaidyanathan and Hammock, 2017). OT signaling is involved in hypothalamic development by governing the formation of new blood vessels (Gutnick et al., 2011). Animal research indicated that in addition to modulating brain activity (Febo and Ferris, 2014; Liu et al., 2015), OT affects the maturation of various brain regions (e.g., amygdala, hippocampus, hypothalamus, neocortex) across the lifespan, with higher contribution for subcortical regional development within two postnatal weeks and for neocortical development during the third postnatal week (Hammock and Levitt, 2013; Insel et al., 1993). Hence, alterations occurring in the brain OT system, particularly early in life, might be followed by persistent modifications in the central OT pathway and brain development. For instance, findings from knockout animal studies showed that OT depletion was associated with persistent reductions of neuronal activity in the adult rodents’ brain regions, including hypothalamus and amygdala (Ferguson et al., 2001; Smith et al., 2019; Zimmermann-Peruzatto et al., 2017).
Early environmental adverse factors could also alter OT levels in the brain. Research evidence stems primarily from animal studies in which rodents have been exposed to prenatal stress in the form of exposure to ethanol (via a liquid diet) or exposure to nicotine (via an osmotic minipump) and to early postnatal stress in the form of maternal separation or deprivation. For instance, relative to non-stressed controls, adult rats born to dams subjected to stress during pregnancy presented impaired social behaviors and diminished OT levels in the brain, particularly in the hypothalamus and VTA (e.g., Lee et al., 2007; McMurray et al., 2008). The social impairments were reversed via central OT administration into the amygdala (Lee et al., 2007). Consistent with the preceding finding, mice exposed to high (relative to low) levels of maternal and peer interactions from birth to weaning displayed greater OXTR availability in the amygdala (Branchi et al., 2013; Francis et al., 2000) and more affiliative (less aggressive) behaviors (Branchi et al., 2013).
Overall, early-life environmental conditions influence brain OT and social behaviors later in life. Essentially, OT is involved in various prosocial behaviors such as sharing resources, including food, and bonding with others, particularly mates, family, and peers (e.g., Kendrick, 2000; MacDonald and MacDonald, 2010; Soumier et al., 2022). OT effects on these behaviors depend on whether the environment increases or hinders closeness to others (Bartz et al., 2011; Bartz, 2016). Impairments in social bonding—mainly characterized by lowered parental sensitivity to the offspring and by offspring’s insecure attachment early in life and their subsequent relationship difficulties with mates—are central in many interpersonal models of EDs (Arcelus et al., 2013). More precisely, inadequate childhood attachment plays a key role in developing maladaptive interpersonal skills, including fear of being perceived negatively by others and avoidance of expressing emotions in front of others, resulting in poor social support. In turn, poor social support could perpetuate disordered eating (Arcelus et al., 2013).
Social attachment and disordered eating: Implication of brain OT
Enduring attachment, particularly the early-life bond between the child and the primary caregiver, is crucial for the quality of interpersonal relationships and general well-being. Impaired social attachment has been discussed as a risk factor for developing EDs (Jewell et al., 2016; Tasca and Balfour, 2014). Social attachment has been argued to enhance the functioning of an internal working model—a framework featuring various mental representations allowing the understanding of the self, others, and the world—contributing to social cognition, emotion processing, and behavior regulation (Amianto et al., 2017; Tops et al., 2014). In line with the preceding, individuals with a personal or parental history of EDs were more likely to have experienced insecure attachment, poor parent child interaction, in particular during feeding, as well as to have difficulties recognizing other people’s emotions and regulating one’s emotions and behaviors, relative to healthy individuals without any history of EDs (Cimino et al., 2016; Jewell et al., 2016).
OT is perhaps the most commonly investigated neuroendocrine modulator of social attachment and eating behaviors. Released primarily during breastfeeding, OT is essential for mother–infant attachment as it increases parental sensitivity toward the child and positive parent–child interactions (Szymanska et al., 2017). The impairment of mother–infant bonding may affect attachment style, with possible consequences for the future development of relationships (Nagasawa et al., 2012). Furthermore, attachment anxiety has been associated with a greater risk of impaired self-identity (Salande et al., 2017), with a possible tendency to employ body weight as a basis of self-definition (Monteleone et al., 2017). In addition, insecure early-life attachment has been associated with psychological inflexibility in adulthood (Salande et al., 2017), the central components of which have been posited to be such maladaptive coping strategies as cognitive fusion, defined as a tendency to get deeply entangled in one’s thoughts, and experiential avoidance, characterized by unwillingness to acknowledge unwanted inner experiences (Bardeen and Fergus, 2016). In turn, impaired self-identity, more ruminative thoughts about eating, shape, and weight, and attempted avoidance of related negative thoughts and emotions have been associated with ED symptoms (Cowdrey and Park, 2012; Monteleone et al., 2017). Accordingly, nurturing familial context and secure parent–infant attachment may play a protective role by promoting body image satisfaction and positive relationships across the lifespan, potentially employing the internalization of stable working models from early in life (Amianto et al., 2017).
OT has been involved in various aspects of social attachment and affiliation, including the formation of social bonds and prosocial behaviors (Szymanska et al., 2017; MacDonald and MacDonald, 2010). The effect of endogenous peripheral OT concentration in social attachment remains however unclear, with some studies reporting a positive association between (blood or plasma) OT and social attachment anxiety and avoidance (e.g., Ebner et al., 2019; Marazziti et al., 2006), and other studies reporting no association between plasma OT levels and attachment dimensions (e.g., Aydin et al., 2019; Plasencia et al., 2019). A recent meta-analysis on the relationship between OT and attachment showed that endogenous OT levels, measured peripherally in blood or saliva, were not associated with attachment dimensions (Zhang et al., 2021). However, exogenous OT administered intranasally was shown to reduce insecure attachment-related behaviors in adults, especially those related to attachment avoidance (Zhang et al., 2021).
In addition to its involvement in social attachment, OT has been linked to eating behaviors. Its primary effect is to reduce food intake (Maejima et al., 2018). Heightened OT levels have been argued to represent a state variable in the context of dietary restriction and socioemotional impairments (Giel et al., 2018). In line with the preceding, a small-scale study showed heightened postprandial serum OT levels in individuals with AN (relative to healthy controls) that were positively associated with symptoms of AN and heightened anxiety (Lawson et al., 2013). However, overnight serum OT levels are the lowest in individuals with AN, higher in normal-weight healthy individuals, and the highest in individuals who are overweight or obese (Schorr et al., 2017). One hypothesis attempting to explain the possibly contradictory OT pattern in AN suggests that low fasting OT levels represent a trait variable or a response to chronic undernutrition, gradually contributing to the pathogenesis of AN (Giel et al., 2018). This idea is supported by animal research showing that acute fasting was associated with a transient decrease in hypothalamic OT-containing neuronal activity, which was reversible by subsequent food consumption, while chronic fasting-related change remained persistent (Suyama et al., 2016). In addition, lowered fasting OT levels (in the brain and blood) were observed in individuals with current AN (Demitrack et al., 1990; Lawson et al., 2013; Monteleone et al., 2016).
Altered OT system functioning may further exacerbate problems in (socio)emotional regulation (Ebner et al., 2019), contributing to the development of maladaptive eating behaviors. For example, individuals with dysfunctional eating behaviors may tend to eat alone and avoid social meals. Maladaptive eating behaviors might constitute an attempt to regulate emotions and cope with stress, a link not devoid of oxytocinergic underpinnings. While low (plasma) OT level might reflect a downregulated hormone level due to chronic fasting/undernutrition in the context of AN (Monteleone et al., 2016), heightened levels of OT were observed in individuals with AN following food intake (Lawson et al., 2012).
Early-life environmental influences on OT levels in the rodents’ brain—as seen in the previous section—appear to convene in such brain areas as hypothalamus, amygdala, and hippocampus (Branchi et al., 2013; Lee et al., 2007; McMurray et al., 2008; Smith et al., 2019; Zimmermann-Peruzatto et al., 2017), which might constitute a neural basis through which OT operates to regulate eating behaviors in humans as well.
The developmental role of central OT: Relevance for EDs
Cumulative evidence of animal studies, involving a change in the brain OT levels via pharmacological manipulations, highlights hypophagic properties of OT (for review, see Leslie et al., 2018; Olszewski et al., 2016). For instance, a single OT administration along the brainstem or in the hypothalamus in food-deprived adult male rats has been associated with a decrease in hunger, time spent eating, weight, and food consumption, including sucrose intake (e.g., Arletti et al., 1989; Ho et al., 2014; Maejima et al., 2014; Noble et al., 2014; Ong et al., 2015). In addition, OT administration into the NAcc and VTA has been associated with reduced sucrose intake (Mullis et al., 2013), while the administration of OXTR antagonists (e.g., L-368,899) into the VTA has been associated with an increase in sugar consumption in rodents (Herisson et al., 2016; Mullis et al., 2013).
Most experimental studies in humans used a double-blind placebo-controlled crossover design involving an intranasal OT administration to individuals with a current diagnosis of an ED and their healthy counterparts. The intranasal administration of OT—via potentially increased central OXTR activity—has been suggested to improve stress reactivity, socioemotional functioning, and (eventually) maladaptive eating behavior in individuals with EDs (Quintana et al., 2018). However, results of human studies of intranasal OT effect on eating behaviors and eating-related cognitions are mixed. In one study, women with AN (relative to healthy controls) showed a decrease in attentional bias to food- and body-related images following 10 consecutive intranasal OT treatments, while there was no change in caloric consumption in either group (Kim et al., 2014). In another study, women with BN and BED (relative to healthy controls) displayed increased attentional bias to food images and (sad) emotion recognition (Kim et al., 2015; Leslie et al., 2020). However, there was no OT-related change in binge-eating behavior after a double-dose intranasal OT treatment (Leslie et al., 2019). Furthermore, the results of a meta-analysis showed that a single dose of intranasal OT had not been linked to a change in emotion recognition and attentional bias in individuals with AN (Hasselbalch et al., 2020). However, exogenous OT has been associated with lowered caloric consumption in healthy individuals but not in individuals with EDs (Chen et al., 2021). The preceding findings indicate the irreproducibility of the intranasally administered OT-related outcomes in humans as well as the importance of taking the OT dose and specific ED diagnosis into account while studying experimentally the potential effect of OT in individuals with EDs. That being said, peripheral administration of OT has been associated with decreased food intake in rats and humans who underwent fasting (Arletti et al., 1989, 1990; Lawson et al., 2015; Ott et al., 2013). The hypophagic property has also been reported in rats following a central administration of OT (Ho et al., 2014; Noble et al., 2014; Ong et al., 2015), which prompts the need to investigate brain OT levels in vivo in humans. While researchers have developed and tested various PET radioligands to map OXTR in vivo (Tan et al., 2022; Vidal et al., 2017), a reliable OT ligand has yet to be established.
Functional magnetic resonance imaging (fMRI) studies combined with intranasal OT administration have shown some insight into the OT-related effects on regional brain function in individuals with EDs. Although methodologies of studies are highly heterogeneous, reviews and meta-analyses of fMRI studies generally conclude OT-related decreased activation in response to food images in various brain areas, including amygdala, VTA, insula, and prefrontal, orbitofrontal, and anterior cingulate cortices among individuals diagnosed with or recovering from AN, relative to healthy controls (Simon et al., 2019). In addition, reduced medial prefrontal and posterior cingulate cortical activity in response to social stimuli has been found in individuals with a history of AN carrying a common OXTR gene variant (rs2254298 A allele) (Sala et al., 2018), which was previously associated with more severe ED symptoms (e.g., eating obsessions and appearance concerns) among individuals with AN by the same research group (Acevedo et al., 2015). Such altered corticolimbic functioning might underlie some of the maladaptive cognitions and affect regulation processes typically occurring in EDs, including body concerns, fears of being judged by others, as well as lowered motivation and pleasure experienced from food (Simon et al., 2019). These maladaptive emotional-cognitive processes may overturn hypothalamic signaling of hunger (Simon et al., 2019; Steward et al., 2018) and/or promote restrictive eating and purging behaviors.
The preceding findings highlight the role of brain regions such as the amygdala, insula, and frontal cortical regions as potential neural bases through which OT operates to regulate eating behaviors and associated emotions and cognitions. Considering that 5-HT is released within corticolimbic brain regions (Booij et al., 2015), both OT and 5-HT might operate within a common neural network to manage similar behaviors, emotions, and cognitions. In the next section, the effect of 5-HT alterations in the central nervous system on subsequent OT functioning, and vice versa, is discussed.
Bidirectional relation between serotonin and OT
Effect of 5-HT intervention on central OT
One of the earliest types of evidence about the interplay of the 5-HT and OT systems in the brain came from electrophysiological experiments. An increase in the firing rate of hypothalamic OT-containing neurons was observed following electrical stimulation of serotonergic dorsal raphe neurons in adult male albinos (Saphier, 1991). Using pharmacological treatments, Saydoff et al. (1993) found that 5,7-dihydroxytryptamine-induced central 5-HT depletion was associated with reduced OT concentration in the pituitary gland, located below the hypothalamus. Intraperitoneal administration of the 5-HT releaser (and 5-HT2 receptor agonist) d-fenfluramine stimulated OT-expressing cells in the rat’s hypothalamus, which was blocked by pre-treatment of selective reuptake inhibitor fluoxetine (Javed et al., 1999). The same research group also found that subcutaneous administration of the 5-HT2 receptor agonist 2,5-dimethoxy-4-iodoamphetamine stimulated OT-expressing cells in the rat’s hypothalamus, which was blocked by pre-treatment of MDL100,907, a 5-HT2A receptor antagonist (Van de Kar et al., 2001).
In addition to the impact of 5-HT on the central OT system, peripheral effects on plasma OT concentration have also been shown. For instance, intraperitoneal administration of the 5-HT releaser d-fenfluramine has been associated with an increase in plasma OT levels (Saydoff et al., 1991). In addition, intraperitoneal administration of 6-chloro-2-[1-piperazinyl]-pyrazine (CPP) and m-chlorophenylpiperazine (mCPP) has been associated with increased plasma OT levels in rats (Saydoff et al., 1991). While CPP is mostly known as an agonist to 5-HT2A/C receptors (Hemrick-Luecke and Fuller, 1996; Vickers et al., 2001), the mCPP has an affinity for α2-adrenoceptors and most 5-HT receptors (Baumann et al., 2001; Hamik and Peroutka, 1989), with its strongest action reported as agonists to 5-HT2C receptors (Hayashi et al., 2005; Hewitt et al., 2002; Kennett et al., 1994). The effects of CPP and mCPP were attenuated by pre-treatment of the 5-HT2A/C antagonist ritanserin (Saydoff et al., 1991). Later on, the 5-HT1A agonist alnespirone was also used to experimentally elevate plasma OT levels in conscious male rats; this effect was blocked by pre-treatment of the 5-HT1A and ß1,2-adrenoceptor antagonist pindolol (Van de Kar et al., 1998). Worth noting is that the heteroreceptor effects of 5-HT1A are possibly more important in this pharmacological action of pindolol in the human brain (Raurich et al., 1999), as this drug has similar efficacy at both the pre- and postsynaptic 5-HT1A receptors (Newman-Tancredi et al., 2001). These findings indicate the influence of serotonergic elements on peripheral OT levels, with agonists of the 5-HT1A and 5-HT2 receptor subtypes increasing plasma OT concentration. Results may also point to the possible involvement of other monoamines, such as noradrenaline (see Vacher et al., 2002).
The effects of altered 5-HT levels on OT functioning have also been examined via gene deletions of metabolizing enzymes. For instance, MAOA knockout mice had heightened 5-HT levels in the central nervous system and presented increased hypothalamic OT concentration, compared to intact wild-type controls (Vacher et al., 2002). Accordingly, another animal study showed that adult TPH2 knockout mice with experimentally reduced 5-HT levels in the central nervous system presented lowered hypothalamic OT levels (Xi et al., 2019). Overall, results of the studies focusing on the action of 5-HT on OT levels in the brain and in the plasma showed that 5-HT releasers, as well as 5-HT2A/C receptors’ agonists (in contrast with antagonists), tend to stimulate OT-expressing cell activity in the hypothalamus and promote peripheral OT secretion (e.g., Javed et al., 1999; Saydoff et al., 1991; Van de Kar et al., 2001).
Effect of OT intervention on central 5-HT
Akin to the animal research findings described in the previous subsection, alterations in OT levels have been associated with changes in the brain 5-HT functioning in animals and humans. For instance, OT (vs saline) administration along the brainstem was associated with a decreased 5-HT concentration in the hypothalamus and the hippocampus of adult male rats (Schwarzberg et al., 1981, 1984). Furthermore, results of a PET study conducted in adult macaques showed that OT (vs placebo) injected along the brainstem was associated with decreased 5-HTT and increased 5-HT1A receptor availability in the amygdala and insula (Gianni et al., 2020). Similar results were found in a PET study conducted in human adults, where intranasal OT (vs placebo) administration was associated with increased in vivo measures of 5-HT1A receptor availability in the amygdala/(para)hippocampus complex and the raphe (Mottolese et al., 2014). Overall, animal and human research evidence suggests that OT exerts an action on the brain 5-HT system, indicated by decreased regional 5-HTT availability and increased availability of 5-HT1A receptors in the amygdala and hippocampus. Also, prairie voles that have been injected intraperitoneally with OT (vs saline solution) during their first postnatal days displayed higher 5-HT axon length in the amygdala and hypothalamus in adolescence (Eaton et al., 2012). Considering that axon length is argued to contribute to the velocity of propagation of the action potentials (Seidl, 2014), further research is necessary to assess the impact of OT on 5-HT neurotransmission.
The abovementioned findings indicate that OT and 5-HT might operate within a typical neural network, including the hypothalamus, amygdala, and brainstem, contributing to maladaptive eating behaviors and related emotions. Adult rats exposed to early postnatal maternal deprivation displayed decreased 5-HT concentration in the hypothalamus, heightened anxiety, as well as decreased standard chow food intake compared to the intact control group (de Lima et al., 2020). Underlying such cooperation may be simultaneous alterations in the hypothalamic concentration of 5-HT and OT, especially considering that the hypothalamus is the neural root of the neuropeptides, including (but not limited to) OT (Blevins et al., 2016). Following this line of thought, findings of another recent study showed preliminary evidence of physical interaction between brain 5-HT2C and OXTR receptors in vitro (Chruścicka et al., 2021). However, the interaction effect between OT and 5-HT in EDs has not been studied systematically.
Discussion
Integrative summary
In the present review, we summarized the existing evidence on the involvement of both central 5-HT and OT in EDs. Animal studies have shown that pharmacological stimulation of 5-HT1B and 5-HT2C receptors in the hypothalamus and raphe nucleus of the brainstem, and 5-HT4 receptor stimulation in the NAcc was associated with lowered food intake in food-restricted and food-deprived rats (reviewed by Compan, 2020). In humans, PET studies have shown that the presence of AN or BN diagnoses was associated with increased 5-HT1A post-synaptic receptor density in the fronto-temporo-parietal regions (Gianni et al., 2020), and with decreased 5-HTT availability in such brain regions as the hypothalamus, raphe, and NAcc, as assessed in vivo (Mele et al., 2020; Pichika et al., 2012; Yokokura et al., 2019). In addition, increased OT levels in the hypothalamus and brainstem were associated with reduced food desire and intake in food-deprived rats (Ho et al., 2014; Maejima et al., 2014; Noble et al., 2014). To date, human studies involving individuals with EDs and healthy controls used a double-blind, placebo-controlled crossover design at intranasal OT treatment, suggesting to act through central OXTR activity (Quintana et al., 2018). The published findings indicate the irreproducibility of intranasal OT-related outcomes (Chen et al., 2021; Hasselbalch et al., 2020). That being said, decreased food intake has been reported in humans and rats who underwent fasting following a peripheral administration of OT (Arletti et al., 1990; Lawson et al., 2015) as well as in rats following a central administration of OT (Noble et al., 2014; Ong et al., 2015), prompting the need to investigate brain OT levels in humans. However, a reliable PET radioligand to map OT in vivo remains to be established.
Central serotoninergic and oxytocinergic systems were found to exert a reciprocal effect via a common neural network. Specifically, 5-HT2A/C receptors’ activity in the brain was positively associated with OT-expressing cell activity in the hypothalamus (Javed et al., 1999; Van de Kar et al., 2001), while OT increased 5-HT1A receptor availability in the amygdala, hippocampus, and raphe (Mottolese et al., 2014) and decreased 5-HTT availability in the amygdala, insula, and hippocampus (Lefevre et al., 2017).
Considering that both 5-HT and OT are involved in brain development (Booij et al., 2015; Hammock and Levitt, 2013), they could both be susceptible to environmental stressors, especially those occurring early in life. For instance, the quality of caregiving and interaction with peers during the early postnatal period was associated with alterations in serotoninergic and oxytocinergic systems in the brain (Branchi et al., 2013; Francis et al., 2000; Spinelli et al., 2010).
Because impaired early-life social bonding has been central in interpersonal models of overeating (Arcelus et al., 2013), we discussed the effects of a negative social environment and disrupted attachment on the brain 5-HT and OT systems. Biological embedding theory, one of the predominant theories attempting to explain the underpinning links between adverse early-life conditions and mental health, posits that stressful experiences can disrupt human developmental course by “getting under the skin” through a specific neurobiological framework, ultimately affecting individual mental health (Nelson, 2013). These effects on the brain 5-HT and OT (dys)regulation might render some people susceptible to (mal)adaptive eating behaviors and related phenotypes (e.g., anxiety, impulsivity). In addition, elements of the brain 5-HT system that have been consistently linked to brain OT functioning (e.g., Lefevre et al., 2017; Mottolese et al., 2014)—particularly, 5-HT1A and 5-HT2A/C receptors and 5-HTT—have also been found to play an important role in active stress coping (Carhart-Harris and Nutt, 2017; Puglisi-Allegra and Andolina, 2015), reflecting an organism’s ability to adapt to early-life stressors. Therefore, it could be of relevance for future treatment of EDs to concurrently target the OXTR as well as 5-HT1A and/or 5-HT2A/C receptors and 5-HTT. The preceding, however, remains to be further tested.
Relevance of 5-HT and OT interactions for a developmental biopsychosocial model of EDs
Overall, the research findings reviewed in the present review align with a developmental model of EDs. While EDs are driven by biopsychosocial pathways in which numerous hormones and neurotransmitters (and the interactions between them) are involved, concerning the role of 5-HT and OT, we conclude that vulnerability-inducing 5-HT and OT levels are shaped by early-life environmental adversity (see Figure 1). Epigenetic processes underlying environmental effects may convey alterations in the central 5-HT and OT, through their joint (emotion and reward) neural network, including the prefrontal cortex, hypothalamus, amygdala, hippocampus, NAcc, VTA, and the brainstem. Along with other risk factors, bidirectional interactions between dysregulated 5-HT and OT in the brain, especially early on during development, may contribute to risk for EDs. In addition, combined with an early-life adverse environment and various other neurobiological risk factors, alterations in the central 5-HT and OT interplay might contribute to the formation of insecure attachment. In turn, inadequate social bonding would translate to impairments in cognitive and socioemotional functioning, including lowered emotion recognition and control and heightened anxiety, which, in turn, might impede the individual’s accessibility to and use of social support. Restrictive eating, malnourishment, and food deprivation-induced neurobiological alterations are expected to further trigger the onset of the ED. The ongoing presence of ED symptoms, in turn, may further exacerbate the existing alterations in central 5-HT and OT functioning, progressively aggravating cognitive and socioemotional impairments.
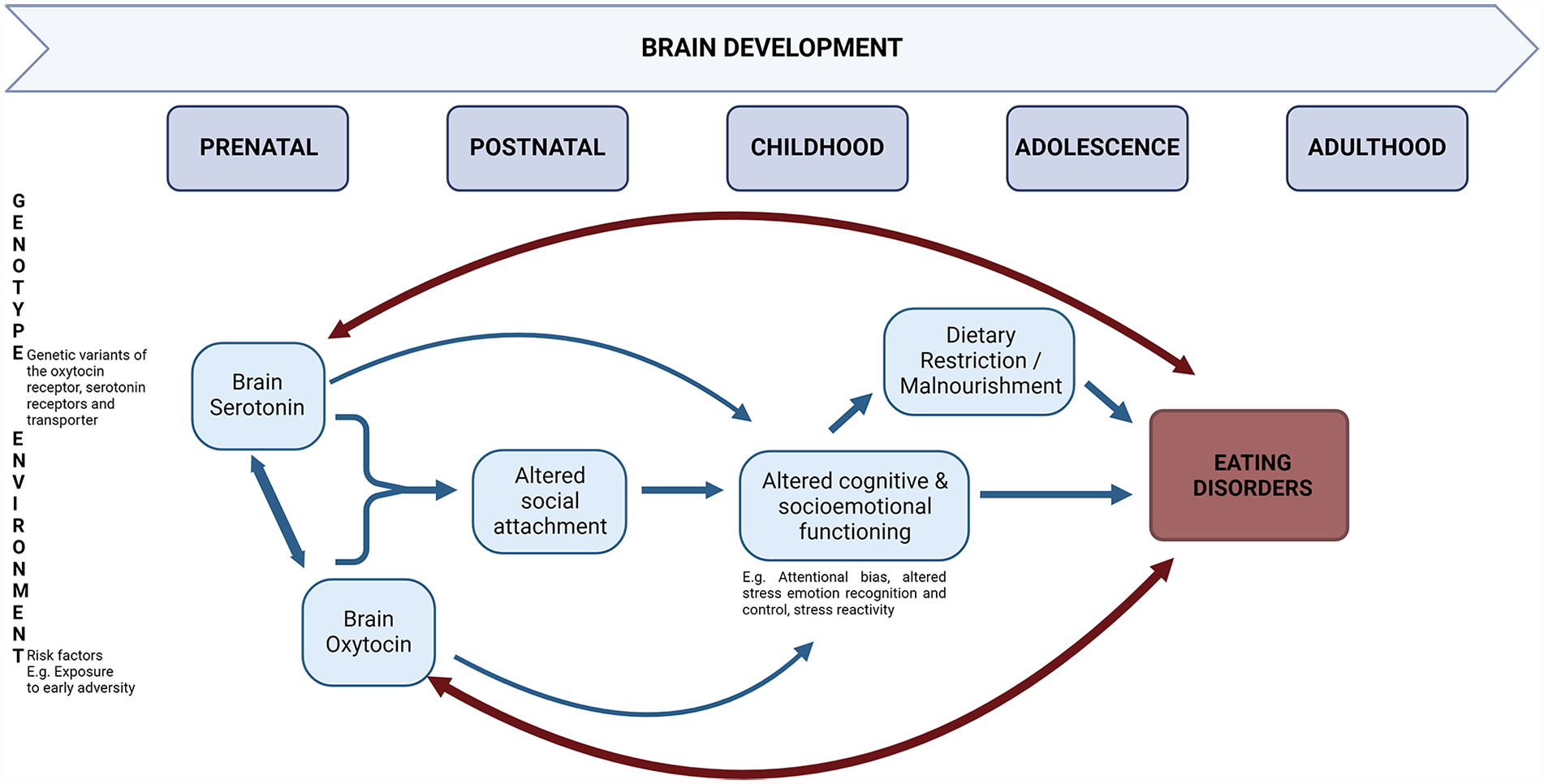
Schematic presentation of the role of the interaction between serotonin and oxytocin in eating disorder development from a developmental biopsychosocial perspective.
While the preceding explanation of how 5-HT and OT may interact and contribute to ED development is plausible, the proposed model requires further study, and the exact physiological mechanisms remain unclear. In addition to early-life environmental stressors, adverse life events later in life have also been associated with altered 5-HT and OT levels as well as with maladaptive eating behaviors and related cognition and affect. For instance, adverse life events at the age of 15 years have been positively associated with the drive for thinness and binge-purge behaviors at the age of 18 years (Akkermann et al., 2012). Moreover, this effect of current life stress was found to be moderated by the short allele of the 5-HTT-linked promoter region (5-HTTLPR), coding for reduced serotonin transporter expression (Akkermann et al., 2012). Various research findings suggest that this 5-HTTLPR interacts with stressful life events, with the short allele (as opposed to the long variant) linked to greater sensitivity to negative environmental factors (e.g., Landoni et al., 2022). As for the OT system, the presence of the rs2254298 and the rs53576 minor (A) alleles of intronic polymorphisms in the OXTR gene has been associated with increased vulnerability to stress and negative mental health outcomes, including symptoms of AN and BN (e.g., Acevedo et al., 2015; Saphire-Bernstein et al., 2011). By contrast, the presence of the rs53576 major (G) allele was found to enhance socioemotional functioning by promoting self-confidence, security, and a general sense of well-being (e.g., Krueger et al., 2012; Monin et al., 2019; Saphire-Bernstein et el., 2011). Such psychosocial resources would likely decrease the risk of developing an ED. Overall, research evidence supports that life stressors may heighten susceptibility to 5-HT and OT dysregulation following stress and result in disturbed eating behaviors in at-risk individuals (having “risk” gene variants, for instance). Future research is necessary to elucidate the cumulative role of early- and current-life stressors in shaping the joint vulnerability of 5-HT and OT dysfunctioning to the development of EDs. Furthermore, it would be important to further examine how a 5-HT/OT vulnerability interacts with other neurobiological risk factors, including (but not limited to) metabolic and immunological risk factors (e.g., see Bulik et al., 2022).
It could be speculated that 5-HT and OT-based interventions have the joint potential to contribute to treating EDs or prevention in at-risk individuals by optimizing the functioning of brain regions involved in social rewards and emotion regulation, such as the prefrontal cortex, amygdala, and brainstem. However, refinement of methodologies and a thorough investigation of the intranasal pharmacodynamics are necessary.
Conclusion and avenues for future research
It is clear that EDs are highly multifactorial, involving numerous neurobiological substrates. As such, alterations in 5-HT, OT, and their interaction likely explain only a small fraction of the variance in ED risk for a given individual. Overall, this review highlights the importance of acknowledging the reciprocal interaction between well-studied neurobiological substrates of EDs such as 5-HT and OT. Furthermore, synthesizing relevant animal and human studies’ findings, the present review illustrated the importance of future translational research in EDs that integrates basic animal experiments with applied research in clinical populations. As such, developing more refined animal models of EDs and new methodologies to study peripheral and central 5-HT or OT in humans would be an important step forward to better understand the clinical relevance of joint administration of serotonergic and oxytocinergic drugs as a possible therapeutic or preventive intervention for EDs. Considering the clinical heterogeneity of EDs, future work also needs to consider the role of demographic and clinical factors such as sex, diagnostic status, illness duration, and psychiatric comorbidities to understand how 5-HT and OT affect (or reduce) the ultimate expression of ED-related cognitions, affect, and behaviors.
Footnotes
Author contributions
EI designed the review, did the literature search, and drafted the manuscript. ZN revised and edited the manuscript. LB designed the review and revised and edited the manuscript. All authors approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was facilitated by a grant from the Natural Sciences and Engineering Research Council of Canada (NSERC) RGPIN/5025-2018 awarded to Dr. Linda Booij and by the Hungarian National Research Development and Innovation Office for bilateral science and technology cooperation project #2019-2.1.11-TÉT-2020-00242 awarded to Dr. Zsofia Nemoda.
