Abstract
Background:
Schizophrenia is considered to have a lifetime prevalence of around 1%. Up to 30% of patients diagnosed with schizophrenia are subsequently categorised as treatment resistant. Current guidelines advise against the use of antipsychotic polypharmacy (APP) or high-dose antipsychotic therapy (HDAT) in the treatment of schizophrenia; however, these treatment approaches continue to be used in up to 25% of cases.
Aims:
This review was to evaluate the evidence for the efficacy and tolerability of APP and HDAT as an alternative to antipsychotic monotherapy at standard doses in the treatment of schizophrenia.
Methods:
This is a systematic review. We searched PubMed, EMBASE and PsycINFO, for eligible trials published prior to 24 March 2023. The protocol was registered on PROSPERO (CRD42023408785). Quality assessment was conducted using the Revised Cochrane risk-of-bias tool for randomised trials.
Results:
A total of 14 studies were included in this review. Two studies demonstrated clinically significant improvement with APP compared to standard treatment. There was no clear evidence that APP or HDAT is definitively less tolerable than antipsychotic monotherapy at a standard dose.
Conclusions:
This review found limited evidence for the efficacy of APP and HDAT in the treatment of schizophrenia over the use of antipsychotic monotherapy at a standard dose. The relative tolerability was unclear. Management of treatment-resistant schizophrenia remains a prominent clinical issue and further research, including high-quality large-scale Randomised Controlled Trials (RCTs) of APP and HDAT in patients who have been unresponsive to clozapine, would be of significant benefit to the field of psychiatry.
Keywords
Introduction
Schizophrenia is a severe, lifelong mental disorder, characterised by symptoms such as delusions, hallucinations and disorder of thought form. It can have significant impacts on cognitive, behavioural and emotional function, and the mean lifetime prevalence is considered to be just under 1% (Kahn et al., 2015). Up to 30% of patients diagnosed with schizophrenia are subsequently categorised as being treatment resistant (Correll and Howes, 2021; Siskind et al., 2022). Treatment-resistant schizophrenia is broadly defined as patients who have not responded to at least two sequential trials of a single dopamine receptor antagonist or dopamine receptor partial agonist, at an effective dose, duration and degree of concordance (Correll and Howes, 2021). Current practice is that patients who are deemed treatment resistant should undergo a trial of treatment with clozapine; however, between 10% and 25% of patients are treated instead with antipsychotic polypharmacy (APP), defined as a combination of more than one dopamine receptor antagonist or dopamine receptor partial agonist, or high-dose antipsychotic therapy (HDAT), defined as one or more dopamine receptor antagonist or dopamine receptor partial agonist prescribed at a dose which exceeds 100% of the British National Formulary (BNF) maximum dose (Burness et al., 2021; Rajan and Clarke, 2013; RCPsych, 2023; Paton et al., 2008; Takahashi et al., 2020).
Current national and local guidance advise against the use of APP and HDAT in the treatment of either schizophrenia or treatment-resistant schizophrenia (NICE, 2014; RCPsych, 2023). Previous reviews have suggested, at the time of their publication, that there was insufficient evidence to support these treatment approaches (CADTH, 2011; Galling et al., 2017; Tracy et al., 2013). Contributing to concerns regarding lack of efficacy were concerns regarding decreased tolerability including, but not limited to, the increased prevalence of extra-pyramidal side effects (EPSEs), increased metabolic side effects and sexual dysfunction (Carnahan et al., 2006; Hashimoto et al., 2012; Suzuki et al., 2008). Since the publication of previous reviews, the development and increasing use of dopamine receptor partial agonists, such as brexpiprazole and cariprazine, may be reflected in changes in practice and present new combinations of drugs for psychosis that would not have been included in previous reviews (Frankel and Schwartz, 2017). In 2019, a nationwide cohort study conducted in Finland suggested that the combination of clozapine and aripiprazole may reduce rehospitalisation rates (Tiihonen et al., 2019). Given the continued prevalence of APP and HDAT within clinical practice, it is important to re-examine the current evidence.
This review aims to evaluate whether progress has been made and if there is sufficient evidence for the efficacy and tolerability of APP and HDAT to suggest either approach as a viable alternative to antipsychotic monotherapy at a standard dose in the treatment of schizophrenia.
Methods
The systematic review of the literature was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standard (Page et al., 2021). The study protocol was registered on PROSPERO (CRD42023408785) and its details are available at crd.york.ac.uk/PROSPERO/display_record.php?RecordID=408785
Search strategy
We searched PubMed, EMBASE and PsycINFO, from the date of database inception to 24 March 2023, with no language restrictions; however, non-English language studies were subsequently excluded. Study authors were contacted to obtain missing data. We used the search terms ((schizophreni* or schizoaffect* or schizo affect* or hebephreni* or schizophreniform or dementia praecox or dementia precox or shared paranoid disorder* or (delusional adj2 disorder*) or (brief psychotic adj2 disorder*) or first psychotic episode* or first episode psychos*)), combined with a list of drugs for psychosis (including trial drug numerical identifiers) AND ((augmentation or add-on or adjunctive or adjunct or adjuvant or added or polypharmac* or polytherap* or combination* or combined or combining or co-therap* or cotherap* or co-administration or coadministration or (dual adj2 therap*) or concomitant or concurrent or monotherap* or monotreatment or mono-therap* or mono-treatment* or mono-administration))) AND ((dose or doses or dosage* or dosing))) AND ((Randomized Controlled Trial or Controlled Clinical Trial))) AND ((random* or sham or placebo*)).
Inclusion and exclusion criteria
We included randomised controlled trials that enrolled adults with a primary diagnosis of schizophrenia or other primary psychotic disorder as defined by ICD-10, ICD-11, DSM-IV or DSM-V. We included studies that compared treatment with HDAT or APP to treatment with a single drug for psychosis at less than 100% BNF maximum (standard dose). We excluded studies if they did not report a quantifiable measure of symptoms of psychosis alongside a quantifiable measure of tolerability. Searches were completed by author CL and verified by reviewer CR, and disagreements were resolved by consensus.
Quality assessment
An assessment of the risk of bias was completed using the Revised Cochrane risk-of-bias tool for randomized trials (RoB 2; Sterne et al., 2019). RoB 2 assesses for bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in the measurement of the outcome and bias in the selection of the reported result.
Data extraction
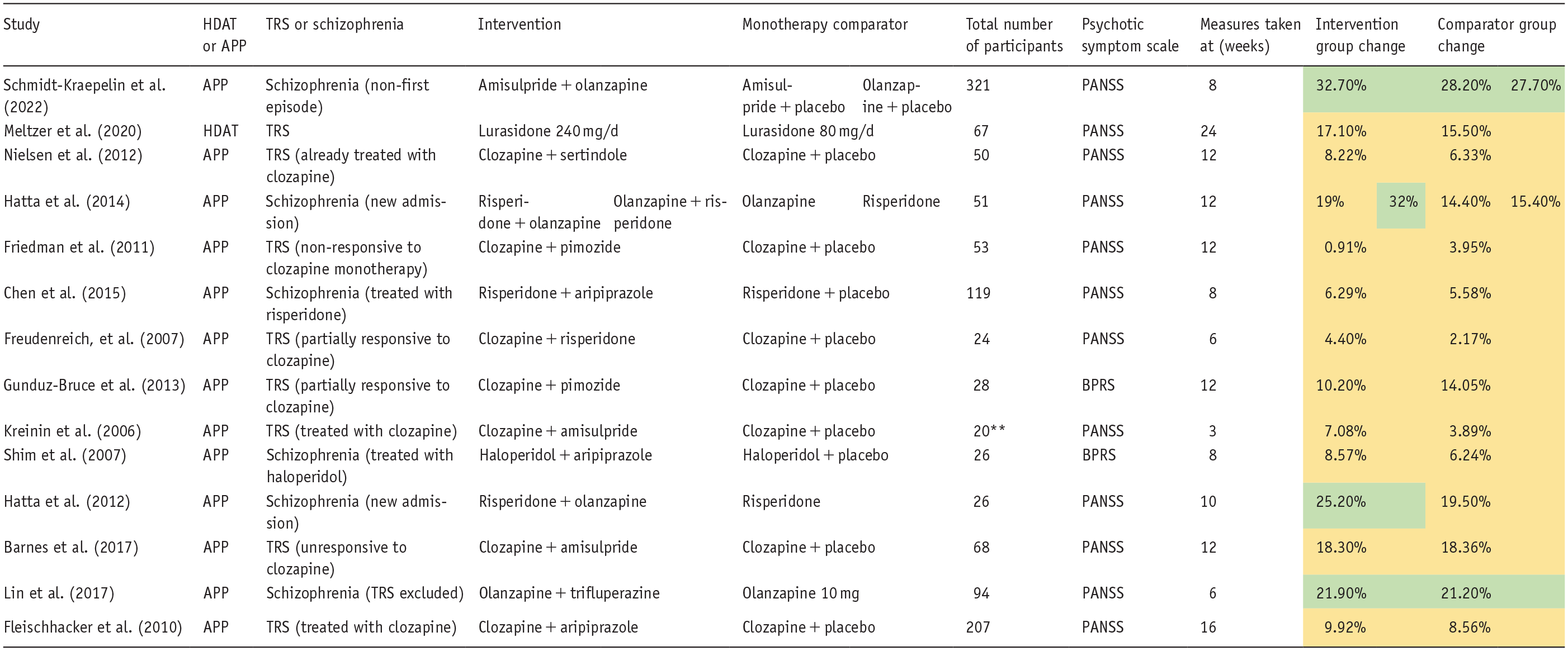
Data extraction was completed by authors CL, CR and CG using a predesigned data-extraction form. Data extracted for each treatment and comparator group included: the number of participants, positive and negative syndrome scale (PANSS) (or other specified psychotic symptom scale) score before and after intervention, Glasgow Antipsychotic Side-Effect Scale (or other specified, quantifiable, adverse effect scale) score before and after intervention, and any reported changes to weight, BMI or HbA1c (See details in Table 1).
Study characteristics.
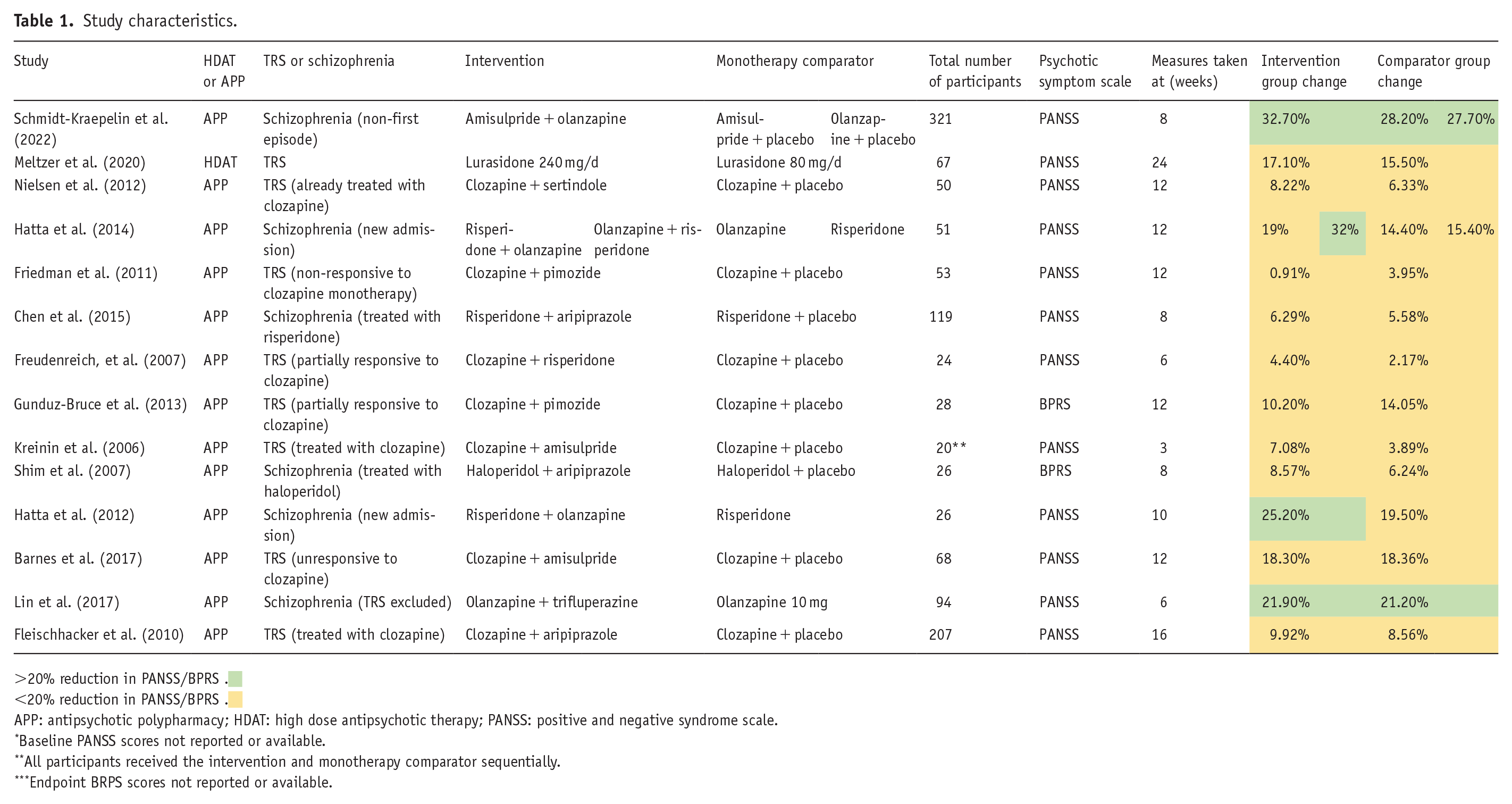
>20% reduction in PANSS/BPRS .
<20% reduction in PANSS/BPRS .
APP: antipsychotic polypharmacy; HDAT: high dose antipsychotic therapy; PANSS: positive and negative syndrome scale.
Baseline PANSS scores not reported or available.
All participants received the intervention and monotherapy comparator sequentially.
Endpoint BRPS scores not reported or available.
Data synthesis
The intention at the point of study design was to complete a meta-analysis of the primary outcomes, efficacy and tolerability of HDAT and APP compared to treatment with antipsychotic monotherapy at a standard dose. However, due to the degree of heterogeneity between studies, this was not feasible. Therefore, the findings have been summarised descriptively.
Results
Search results and study characteristics
A total of 1004 records were identified from the combined searches of the PubMed, PsycInfo and EMBASE databases. After duplicates were removed, 446 titles, then 223 abstracts were screened, and records were excluded if they did not appear relevant or to meet the inclusion criteria based upon title or abstract, respectively. In all, 42 full reports were retrieved and a further 28 were excluded following full-text screening. Fourteen studies were included in the final review (Figure 1).
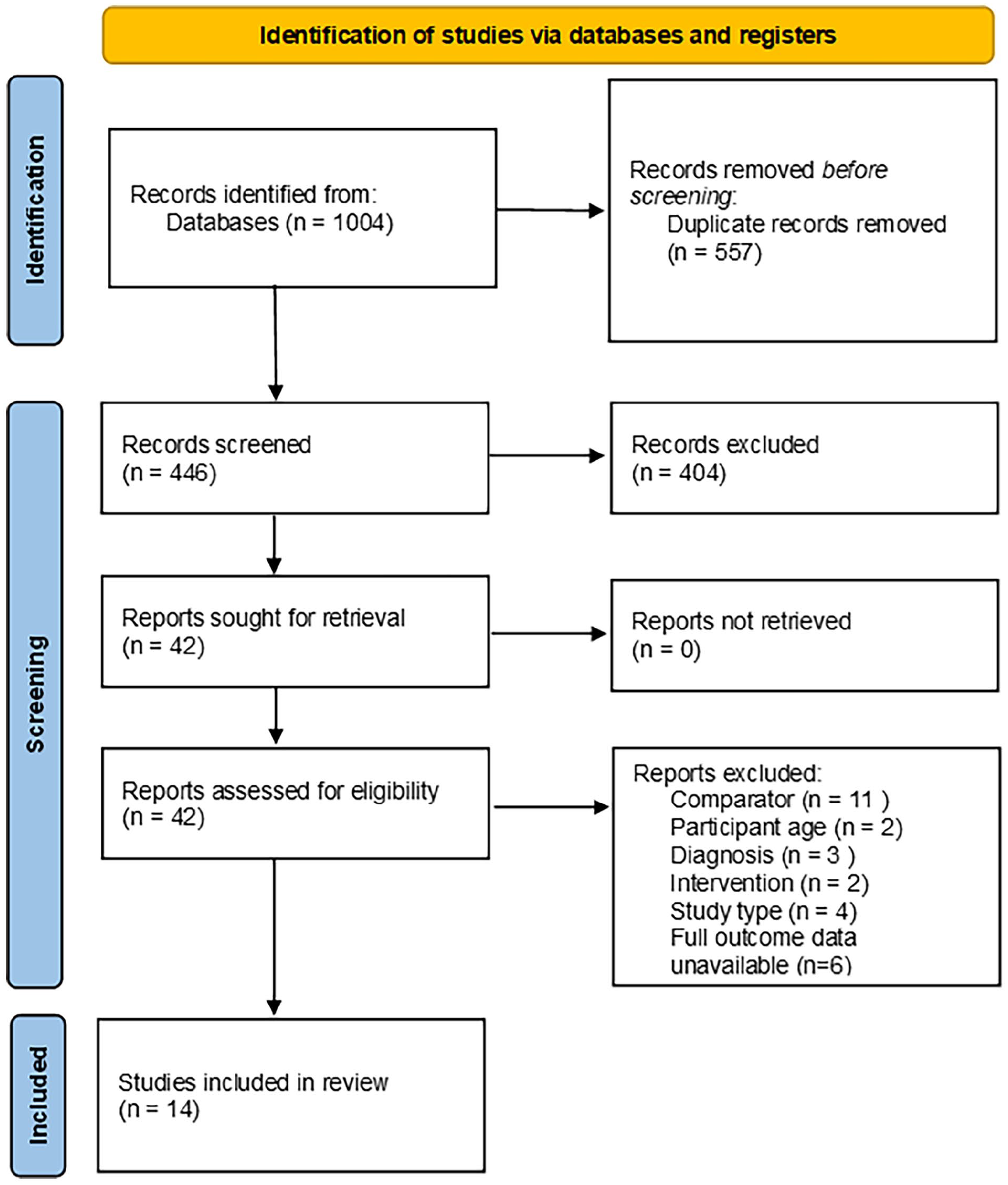
PRISMA flow chart of study selection.
Thirteen of the studies were assessed as low risk of bias. Three of the studies showed ‘some concerns’ regarding bias due to (1) no information on whether an appropriate analysis was used to estimate the effect of assignment to intervention, (2) whether raters were blinded to the intervention or (3) if measurement of the outcome could have differed between intervention groups (Chen et al., 2015; Fleischhacker et al., 2010; Meltzer et al., 2020).
A meta-analysis was not performed as the differences in reporting outcomes across the studies, principally variability in the range of time points used for measurement of psychotic symptoms, precluded a statistical synthesis of the results of the included studies.
All 14 included studies were reports of randomised control trials comparing either APP or HDAT against at least one monotherapy comparator group (treatment with a single drug for psychosis at a standard dose).
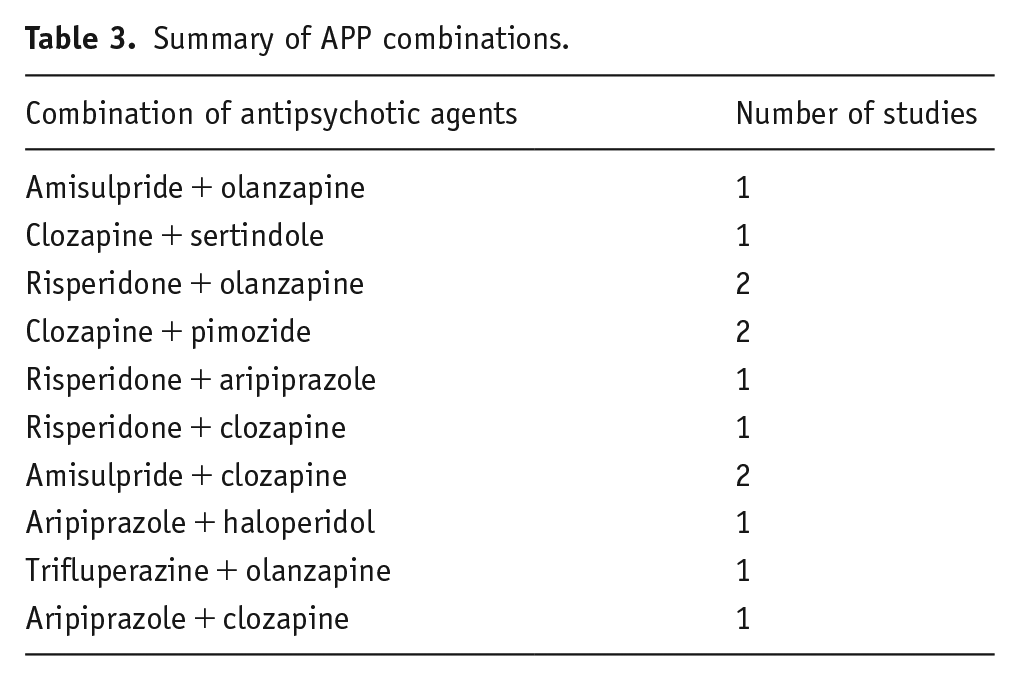
Thirteen studies compared APP to a monotherapy comparator. Combinations were diverse, with no combination being used more than twice. The different combinations are shown in Table 2. The most commonly combined agents were clozapine, risperidone and olanzapine, used seven, four and four times, respectively. HDAT was utilised in one study (Meltzer et al., 2020) (Table 3).
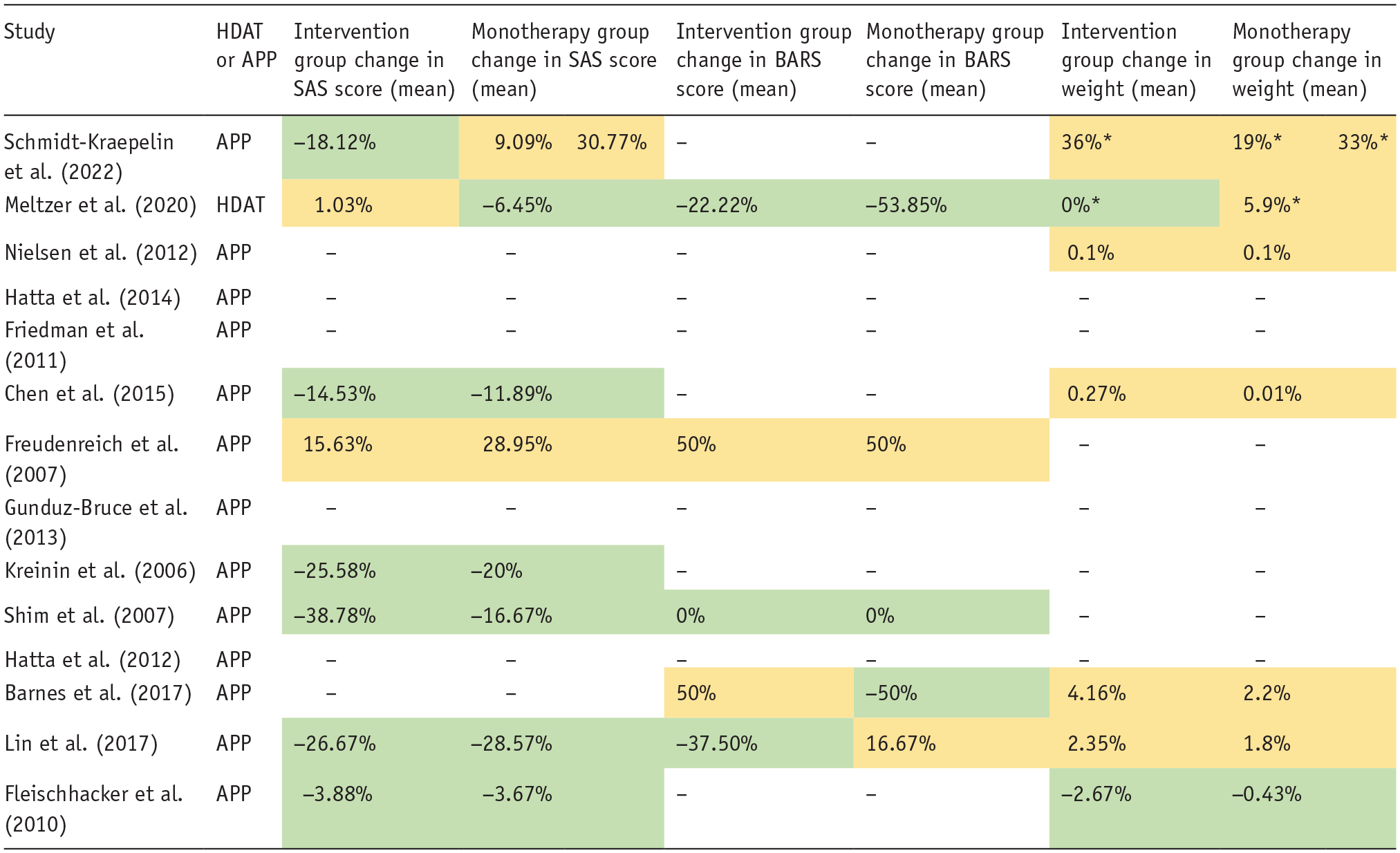
Summary of tolerability scores.
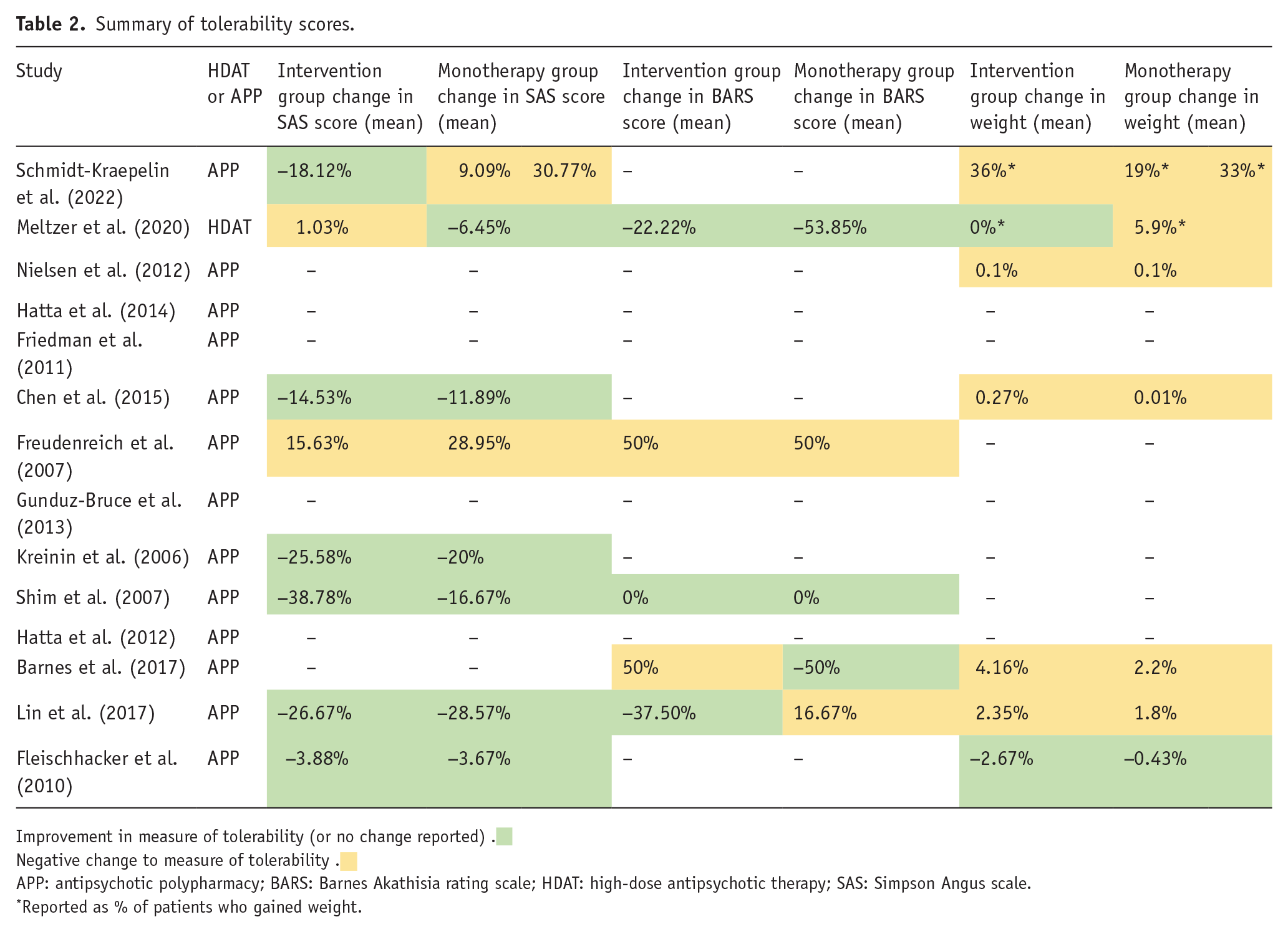
Improvement in measure of tolerability (or no change reported) .
Negative change to measure of tolerability .
APP: antipsychotic polypharmacy; BARS: Barnes Akathisia rating scale; HDAT: high-dose antipsychotic therapy; SAS: Simpson Angus scale.
Reported as % of patients who gained weight.
Summary of APP combinations.
Comparison of APP to monotherapy
Eleven of the included studies compared APP to antipsychotic monotherapy at a standard dose and reported PANSS scores at baseline and endpoint. Four studies demonstrated an improvement in total PANSS score of more than 20% in at least one APP group (Hatta et al., 2012, 2014; Lin et al., 2017; Schmidt-Kraepelin et al., 2022). This has been used by previous studies as the minimum change for clinical significance (Paton et al., 2007). However, in two of those four studies, the monotherapy groups also demonstrated improvement in total PANSS scores of more than 20% (Lin et al., 2017; Schmidt-Kraepelin et al., 2022). Hatta et al (2014) demonstrated an improvement in total PANSS score of 32% in the APP arm where olanzapine was augmented with risperidone, compared to an improvement of 19% in the APP arm where risperidone was augmented with olanzapine (Hatta et al., 2014). Four studies demonstrated clinically significant changes in PANSS positive scores, these changes were also seen in the monotherapy comparator groups (Lin et al., 2017; Hatta et al., 2012, 2014; Schmidt-Kraepelin et al., 2022). Only two studies demonstrated clinically significant changes in PANSS negative scores in the APP groups, in both of those studies there was also a similar clinically significant change within the monotherapy comparator groups (Hatta et al., 2014; Schmidt-Kraepelin et al., 2022) (Figure 2).
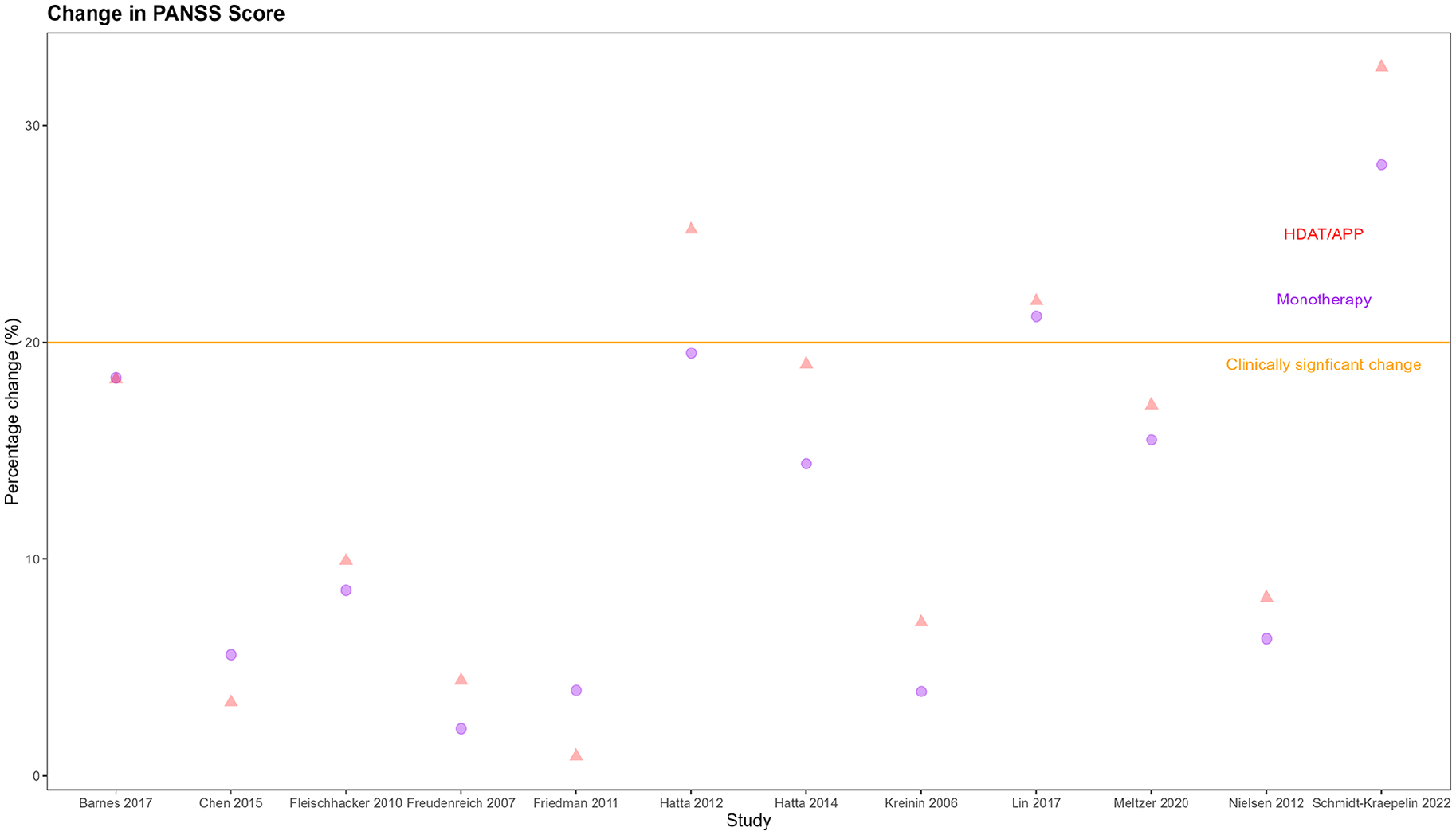
Change in total PANSS score HDAT/APP compared to monotherapy.
Two of these studies reported BPRS scores at baseline and endpoints (Gunduz-Bruce et al., 2013; Shim et al., 2007). Neither study demonstrated a clinically significant improvement in BPRS scores in either the APP or monotherapy groups. One study demonstrated an improvement of 8.57% compared to 6.24% at 8 weeks with the addition of aripiprazole to haloperidol compared to treatment with haloperidol alone (Shim et al., 2007). One study demonstrated less improvement in BPRS scores with pimozide augmentation of clozapine at 12 weeks compared with placebo (Gunduz-Bruce et al., 2013).
Seven of these studies reported CGI-S scores at baseline and endpoint. Five studies demonstrated greater reductions in CGI-S in the APP groups compared to monotherapy groups (Chen et al., 2015; Kreinin et al., 2006; Lin et al., 2017; Schmidt-Kraepelin et al., 2022; Shim et al., 2007). One study showed no difference (Nielsen et al., 2012). One study showed a greater improvement in the monotherapy group compared to the APP group (Gunduz-Bruce et al., 2013).
Seven of these studies measured EPSEs using the Simpson Angus Scale (SAS) and reported scores at baseline and endpoint. Six studies demonstrated lower SAS scores in the APP groups compared to the monotherapy groups (Chen et al., 2015; Fleischhacker et al., 2010; Freudenreich et al., 2007; Kreinin et al., 2006; Lin et al., 2017; Shim et al., 2007). Only two studies demonstrated an increase in EPSEs scored via the SAS between baseline and endpoint in the APP groups, and in both of those studies, there was a higher increase in the monotherapy groups (Chen et al., 2015; Lin et al., 2017).
Four of these studies measured symptoms of akathisia using the Barnes Akathisia Rating Scale (BARS) and reported scores at baseline and endpoint (Barnes et al., 2017; Freudenreich et al., 2007; Lin et al., 2017; Shim et al., 2007). One of those studies demonstrated a worse increase in akathisia symptoms in the monotherapy group compared to the APP group between baseline and endpoint (Lin et al., 2017).
Six of the included studies reported the average weight (kg) at baseline and endpoint, or the average weight gain, within the APP and monotherapy groups. Individuals within the intervention group gained weight between baseline and endpoint in five studies (Barnes et al., 2017; Chen et al., 2015; Lin et al., 2010, 2017; Nielsen et al., 2012; Schmidt-Kraepelin et al., 2022). Individuals within the monotherapy groups gained weight in five studies. In Fleischhacker et al. (2010), individuals lost less weight in the monotherapy group than in the APP group, which augmented clozapine with aripiprazole (Fleischhacker et al., 2010).
Neither Prolactin nor HbA1c results at baseline and endpoint were reported consistently in more than one study and therefore these have been excluded from further analysis within this review.
Comparison of HDAT to monotherapy at standard dose
Meltzer et al. (2020) was the only study that met the inclusion and exclusion criteria for this review and included an HDAT group (Meltzer et al., 2020). This study reported a PANSS score at baseline and endpoint and demonstrated an improvement of 17.10% in the HDAT group (lurasidone 240 mg/d, 163% BNF maximum) compared to 15.50% in the comparator (lurasidone at a standard dose) (Meltzer et al., 2020). This would not be considered a clinically significant improvement.
Similar improvements were seen in the CGI-S scores, an improvement of 15.22% in the intervention group compared to 11.11% in the comparator group (Meltzer et al., 2020).
With regards to tolerability, the HDAT group showed worse outcomes for both EPSEs and akathisia, as demonstrated by comparative changes in the SAS and BARS scores, as seen in Table 2. However, more patients gained weight in the comparator group (5.8%) than in the HDAT group (0%) (Meltzer et al., 2020).
Discussion
This review presents the systematically acquired evidence for the use of HDAT and APP in the treatment of schizophrenia from 14 RCTs involving 1174 participants. The findings reveal no significant improvement in the efficacy and tolerability of either HDAT or APP over the use of antipsychotic monotherapy at a standard dose, in the available literature.
The findings of this review are largely in keeping with those of previous reviews of both APP and HDAT. Despite the continued use of APP and HDAT by clinicians internationally, there remains limited evidence to support either of these treatment approaches.
Thirteen of the studies included in this review compared APP to antipsychotic monotherapy at a standard dose. Ten of those studies did not demonstrate a clinically significant change in quantifiable measures of psychotic symptoms within the intervention groups. One study, which compared the augmentation of olanzapine with trifluperazine to treatment with olanzapine alone, demonstrated a clinically significant change that was replicated within the monotherapy comparator group (Lin et al., 2017). Two studies did demonstrate clinically significant changes in quantifiable measures of psychotic symptoms within intervention groups that were not replicated in monotherapy comparator groups (Hatta et al., 2012, 2014). Both of those studies combined risperidone with olanzapine; however, the number of total participants was relatively small and there was not significantly more improvement in the APP treatment groups compared to the monotherapy comparators. In Hatta et al. (2012), the 26 patients included in the study had all already failed to respond to initial treatment with risperidone; therefore, this may have reduced the likelihood of a subsequent positive response to further dose increases in risperidone in the monotherapy comparator group compared to the addition of olanzapine within the intervention group (Hatta et al., 2012). In both Hatta et al. (2012) and Hatta et al. (2014), all patients included were new emergency admissions who met the criteria for a diagnosis of a primary psychotic illness and therefore this excluded patients who had experienced long admissions or multiple trials of alternative antipsychotics and this difference in the patient group may explain the higher level of improvement when compared to those studies which specifically included patients who met the criteria for treatment-resistant schizophrenia (Hatta et al., 2012, 2014). This review did not find evidence that any specific combination of drugs for psychosis was more effective than antipsychotic monotherapy at a standard dose, which is in line with previous reviews that have focused specifically on APP (Galling et al., 2017).
The inclusion and exclusion criteria used in this review were designed to enable a comparison of HDAT and APP to antipsychotic monotherapy at a standard dose; however, this review found only one RCT that both met the inclusion criteria and included an HDAT treatment group (Meltzer et al., 2020). Meltzer et al. (2020) compared lurasidone 80 mg/d (55% BNF maximum) to lurasidone 240 mg/d (163% BNF maximum) in treatment-resistant schizophrenia. While both groups improved in PANSS scores from baseline, neither group demonstrated a clinically significant improvement and while the intervention group performed slightly better, the difference was not significant and the authors themselves acknowledge that the results were comparable. One recent review has specifically examined the efficacy of high-dose olanzapine in treatment-resistant schizophrenia (Gannon et al., 2023). However, none of the five included RCTs would have met the inclusion and exclusion criteria for this review, due to the lack of an appropriate monotherapy comparator group, the lack of a quantifiable adverse effect scale or that the intervention groups included patients treated with a range of doses of olanzapine which included both standard doses and HDAT (Conley et al., 1998, 2003; Ermilov et al., 2013; Meltzer et al., 2008; Tollefson et al., 2001; Volavka et al., 2002). A limitation of this review is that the inclusion and exclusion criteria may have led to the exclusion of studies, such as those included in the Gannon et al. (2023) review, examining the efficacy of other drugs for psychosis used within HDAT dose ranges (Gannon et al., 2023). Furthermore, non-English language studies were excluded due to financial and capacity restraints within the review team, and this may have led to the exclusion of further potentially relevant studies. However, there is a clear lack of high-quality RCTs examining the efficacy and tolerability of HDAT in the management of treatment-resistant schizophrenia within the literature.
There remains scope for future research to adequately explore the efficacy of HDAT but the current lack of clear evidence for efficacy over monotherapy within standard BNF dose limits should prompt clinicians to exercise caution with regard to this treatment option.
This review did not demonstrate clear evidence that HDAT or APP is definitively less tolerable than antipsychotic monotherapy at standard doses. Monotherapy comparator groups performed worse than intervention groups in measures of akathisia, extra-pyramidal side effects and weight gain in several studies included in this review. As noted within the results section, the lack of consistency in reporting markers of metabolic syndrome meant that it would not be appropriate to draw definitive conclusions from the data available. It may be that future research looking specifically at these markers may be beneficial. It is possible that the adverse effects associated with drugs for psychosis are seen at standard doses and are not exponentially worse as doses increase or agents are combined. However, the observed length of treatment within the studies included did not exceed 24 weeks and 14 studies did not record measures beyond 12 weeks. While it may be difficult to maintain a high-quality RCT for a longer period, it may represent an inherent limitation in observing the long-term tolerability and possible adverse effects associated with the use of HDAT and APP. Non-RCT studies likely represent a more appropriate method to achieve this aim, and previous studies including self-rating questionnaire studies, retrospective reviews of treatment data and open-label studies have demonstrated evidence of decreased tolerability of both HDAT and APP (Carnahan et al., 2006; Hashimoto et al., 2012; Ray et al., 2009; Suzuki et al., 2008). There are also some situations where some dopamine receptor antagonists, such as clozapine or olanzapine, have been augmented with agents such as aripiprazole (as in Fleischhacker et al. (2010)), based on evidence that this can improve metabolic outcomes (Fleischhacker et al., 2010). This is supported by the British Association for Psychopharmacology (BAP) guidance (Cooper et al., 2016).
Nine of the included studies specifically included patients who met the criteria for treatment-resistant schizophrenia, while two of the remaining seven studies explicitly excluded patients with treatment resistance. It stands to reason that those studies that include a new cohort of patients who have not previously been treated with antipsychotic therapy are more likely to include patients who will respond well to initial treatment compared to a cohort made up solely of those individuals who have already met the criteria for treatment-resistant schizophrenia. The combinations of those cohorts within this review may therefore be seen as a limitation and future reviews may wish to examine one of these cohorts independently.
In conclusion, this review has found little further evidence to support the use of HDAT and APP in the treatment of schizophrenia, not yet ascertained to be treatment resistant, over the use of antipsychotic monotherapy at a standard dose. This is in line with the results of previous reviews and current national guidelines. Treatment-resistant schizophrenia remains a prominent clinical issue within psychiatry. Previous research and clinical guidelines have outlined approaches for treating these individuals (NICE, 2014; RCPsych, 2023; Taylor et al., 2018; Tracy et al., 2013). Several of the included studies have attempted to explore alternative treatment options for patients who have not been fully responsive to, or were intolerant of, treatment with clozapine; however, there remains a clear lack of high-quality evidence to support the use of HDAT and APP within this cohort and it is clear that further research in this regard, which would ideally include high-quality large-scale RCTs of HDAT or APP, would be of significant benefit to the field of psychiatry.
Footnotes
Acknowledgements
The authors would like to thank Dr Elizabeth Graves and Sana Sajid for their review of the protocol during its development and Dr Abigail Hood for her review of this paper during the draft stage.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: CL has previously acted as a Sub-Investigator for trials run by Boehringer Ingelheim and Janssen through his role at Hampshire and Isle of Wight Healthcare NHS Foundation Trust but did not receive any direct funding or payment for this. SRC’s research work was previously funded by Wellcome. He receives honoraria for editorial work at Elsevier Journals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.