Abstract
Background:
There is high inter-individual variability in clozapine metabolism due to genetic and non-genetic differences. Patient-specific factors such as smoking, inflammation indicated by elevated C-reactive protein (CRP), and certain concurrent medications have a significant influence on clozapine metabolism.
Aim:
To assess which patient-specific factors best explain variability in clozapine metabolism estimated by clozapine concentration to dose (C/D) ratios.
Methods:
A retrospective cohort analysis using electronic medical data was conducted on 172 inpatients at the BC Psychosis Program. Patients with normal renal and liver function were included if they were on clozapine and had at least one steady-state plasma concentration. The degree of influence of each factor on the variability of clozapine metabolism in the entire cohort and subgroups stratified by fluvoxamine use was evaluated using multiple linear regression analysis of C/D ratios.
Results:
Model fit testing showed that the entire cohort model accounts for 52.7% of C/D ratio variability, while the no fluvoxamine and fluvoxamine models accounted for 40.8% and 43.8%. In the entire cohort (n = 172), fluvoxamine use explained the highest variance, and C/D ratios were higher by 30.6% on average. The second strongest predictor was elevated CRP > 10 mg/L, and C/D ratios were higher by 22.9% on average. Subsequently, obesity, nonsmoker status, and female sex explained a significant but modest proportion of variance. Among participants on fluvoxamine (n = 58), only fluvoxamine dose was associated with an increase, and for every 25 mg increase in dose, C/D ratios increased by 5% on average.
Conclusion:
In a clinical population, this study replicated the relationship between reduced rate of clozapine metabolism and the use of fluvoxamine, elevated CRP, obesity, nonsmoking status, and female sex; and the magnitude of the effects were large enough to be clinically relevant.
Keywords
Introduction
Clozapine has unique superior efficacy in treatment resistance, persistent hostility or violent behavior, and suicidality (Forte et al., 2021) in individuals with schizophrenia (Wagner et al., 2021). Moreover, it has been associated with lower all-cause and specific-cause mortality, fewer hospitalizations, and all-cause discontinuation (Correll et al., 2022). Yet, these findings have not translated into greater clozapine use, and its initiation is markedly delayed (John et al., 2018; Yoshimura et al., 2017). This is in part due to common and sometimes severe adverse effects, as well as knowledge gaps in preventing and managing these effects (Farooq et al., 2019).
Clozapine is a strong candidate for therapeutic drug monitoring (TDM) as it has a suggested plasma concentration–response relationship and carries an increased risk of nonresponse or adverse effects if plasma concentrations occur outside the therapeutic range (Northwood et al., 2023; Siskind et al., 2021). Available evidence suggests concentrations above 350 ng/mL (1070 nmol/L) are associated with higher rates of therapeutic response and concentrations above 600 ng/mL (1850 nmol/L) are associated with higher rates of dose-dependent adverse drug effects (Northwood et al., 2023; Siskind et al., 2021), such as sedation, seizures, myoclonus, obsessive-compulsive symptoms, tachycardia, and orthostatic hypotension (Gurrera et al., 2022). During the dose-escalation phases of treatment, other clozapine side effects, like inflammation, can become more noticeable and could be connected to the rate of titration and dose accumulation (de Leon et al., 2022). Therefore, to individualize the titration plan and target dose, it would be imperative to identify patient-specific factors that impact clozapine metabolism.
Clozapine clearance is mediated, in part, by extensive hepatic first-pass metabolism via the cytochrome P450 (CYP450) system with a modest excretion of unchanged drug in the urine of less than 3% (Schaber et al., 1998). CYP1A2 is the principal enzyme that metabolizes clozapine in vivo and results in the formation of N-desmethylclozapine (norclozapine) (Thorn et al., 2018). Although CYP3A4, CYP3A5, CYP2D6, and CYP2C19 are also involved in the metabolism of clozapine, their contribution is minor based on the current evidence (Brandl et al., 2015; Dahl et al., 1994; Jaquenoud-Sirot et al., 2009; Toth at al., 2017).
To date, the proportion of the variance in clozapine metabolism explained by genetic differences has been modest (Pardiñas et al., 2023). By contrast, other patient-specific factors such as smoking, high caffeine intake, inflammation indicated by elevated C-reactive protein (CRP), and certain concurrent medications have a significant influence because they can either induce or inhibit CYP1A2 activity (de Leon et al., 2022).
At a steady state, the rate of metabolism and clearance can be estimated by dividing the clozapine plasma concentration (ng/mL) by the total daily dose (mg/d) to derive the concentration-to-dose (C/D) ratio (de Leon et al., 2020). In this work, we evaluated the direction and magnitude of patient-specific factors’ effects on clozapine C/D ratios using TDM and other clinical data from a cohort of inpatients receiving clozapine treatment.
Materials and methods
Study setting and population
This was a retrospective study using routine care clinical data from the British Columbia Psychosis Program (BCPP), a 25-bed tertiary inpatient resource for treatment-resistant psychosis in the Canadian province of British Columbia. Ethics approval was obtained from the University of British Columbia Clinical Research Ethics Board. The study cohort consisted of inpatients with severe and treatment-resistant psychosis who were admitted to the program between February 21, 2012, and November 7, 2021, after being referred by psychiatrists working at secondary inpatient facilities or within community mental health or assertive community treatment teams. Schizophrenia or schizoaffective disorder are the most common diagnoses for patients who are admitted to the program. Most patients entering the BCPP have used clozapine in the past or are currently taking clozapine. The majority are admitted as involuntary patients under the provincial Mental Health Act.
For patients with resistant psychotic illnesses, the BCPP employs a thorough treatment strategy. This entails a formal review of the diagnosis by DSM 5 criteria, challenges, and re-challenges of the clozapine medication as necessary, trials of alternative antipsychotics and augmentation drugs, evidence-based nonpharmacological approaches, such as cognitive behavioral therapy for psychosis and social skill development, and electroconvulsive treatment trials. Unless severe tolerability or safety issues occur after admission, patients who were receiving clozapine at the time of admission continue to receive this medication.
Clozapine administration at BCPP is supervised by psychiatric nurses and if there are reasons to suspect non-adherence, nurses ask to see the inside of patients’ mouths after dose intake. In some cases, clozapine tablets may be crushed in jam or apple sauce to improve adherence. If there are concerns regarding emesis of the ingested dose, patients are observed for 30 min post-medication administration. During this period, access to their room is restricted. Moreover, patients at BCPP have a limited number of independent passes each day for the duration of their stay because it is a secured unit; consequently, the frequency of cigarette smoking in this cohort is likely lower than in studies conducted in outpatient settings. Similarly, caffeine intake is supervised and lower than studies conducted in outpatient settings.
Eligibility criteria
Patients with normal liver and renal function were eligible if they were on clozapine treatment during the study period with at least one clozapine plasma concentration at “steady-state conditions.” Steady state was qualified as samples that were taken 12 h (±2 h) after the last clozapine dose and at least 5 days after any dose changes to clozapine. Furthermore, these levels were not contaminated by new inhibitors, inducers, or infection/inflammation.
Data collection
For each patient meeting eligibility criteria, a review of the electronic medical record was conducted by five authors (R.R., A.S., A.R., N.B., and K.K.) utilizing a custom data extraction template. Extracted data included patient demographics (sex, age, self-reported ancestry), medical and psychiatric diagnoses, smoking status (yes/no), obesity (body mass index (BMI) ⩾ 30 kg/m2), prescribed medications, medication administration records, and laboratory results including clozapine plasma concentrations at steady-state conditions, complete blood counts, and CRP levels throughout their stay at the facility. The principal investigator (R.R.) maintained the consistency of retrospective data extraction and re-evaluated each concentration to ensure steady-state conditions were met.
Measurement of the rate of clozapine metabolism and clearance
Clozapine plasma concentrations were measured using liquid chromatography-tandem spectrometry at the British Columbia Provincial Toxicology Center accredited laboratory. Given the inpatient setting, many patients had repeated clozapine TDM.
Consistent with previously published literature (Ruan et al., 2019), we averaged the plasma concentrations for patients who had multiple clozapine TDM at steady-state conditions throughout their stay, provided they were not contaminated by new inhibitors, inducers, or infection/inflammation.
Measurement and inclusion of CRP levels
Non-fasting high-sensitivity CRP was measured using turbidimetric/immunoturbidimetric methodology in Vancouver Coastal Health (VCH) accredited laboratory services at the time of admission and then routinely as per clinical need. Unless there were changes to CRP re-measurements, complete blood counts, or indications of acute illness from clinicians’ records, it was assumed that all the included individuals with normal CRP range on admission would have normal CRP range throughout their stay. All the included patients with elevated CRP >10 mg/L on admission had repeated CRP levels during their hospital stay. For patients initiated on clozapine at BCPP, CRP was measured prior to initiation of clozapine and then weekly for at least 4–6 weeks as per VCH’s protocol for myocarditis monitoring. Our cutoff for CRP elevation was ⩾10 mg/L which may suggest the presence of an underlying inflammatory condition (Fathian et al., 2022; Windgassen et al., 2011).
Statistical analysis
Jamovi version 2.5 (The jamovi project, 2024) was used for all analyses. Descriptive statistics are presented as means and standard deviations (SD). The effects of age, sex, ancestry, smoking status, obesity, valproate, fluvoxamine, CYP2D6 inhibitors (e.g., paroxetine, fluoxetine, and bupropion), and CRP on C/D ratios were evaluated using a multiple linear regression model. Prior to fitting the regression model, C/D ratios were log10 transformed to correct the positive skewness of the residuals. Moreover, an evaluation of multicollinearity was performed among the independent variables and the dependent variable underwent outlier (Grubb’s) analysis. The Shapiro–Wilk test was used to test the normality of residuals. An alpha threshold of 0.05 was used for all tests.
Results
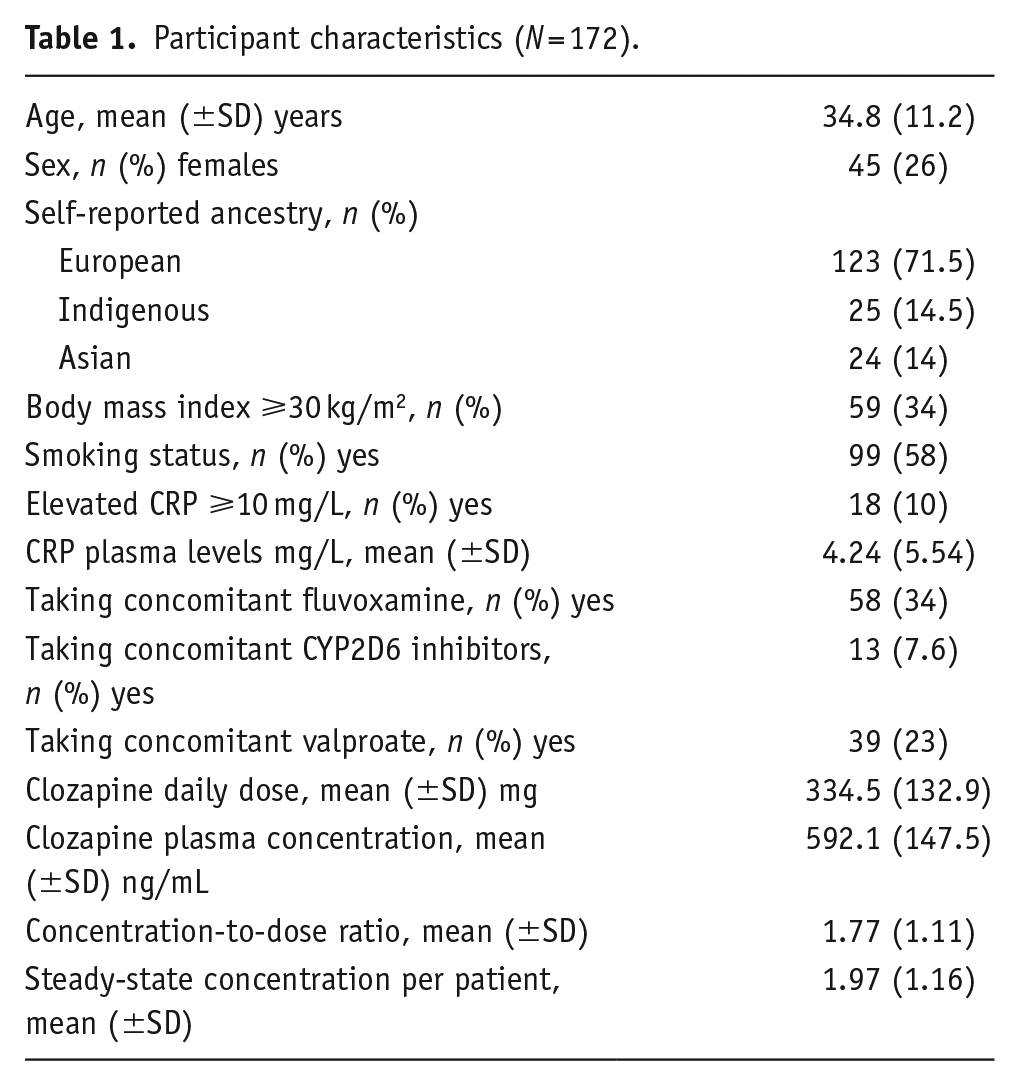
Table 1 shows demographic and clinical characteristics of the cohort. A total of 343 steady-state plasma concentrations were available for analysis from 172 individuals. These individuals were primarily of European background (71.5%). There was a relatively small representation of other ancestries with approximately 14% self-identifying as Asian (ranging from Pakistan to Japan) and 14.5% as Indigenous. Clozapine in a significant portion of this cohort was augmented with fluvoxamine (n = 58). Fluvoxamine dose ranged from 12.5 to 200 mg. Strong CYP2D6 inhibitors including bupropion, fluoxetine, and paroxetine were prescribed to 13 patients, three of whom also received fluvoxamine. For all 343 plasma concentrations, information on age, sex, ancestry, smoking status, concomitant medications and their dose, and BMI were available. There were missing CRP levels on admission and during the hospital stay for 18 patients. Consequently, the multiple imputation method was used to fill these missing values.
Participant characteristics (N = 172).
Model fitting was performed on the entire cohort and subgroups stratified by fluvoxamine use. Model fit testing showed that the entire cohort model accounts for 52.7% of C/D ratio variability, while the no fluvoxamine and fluvoxamine models accounted for 40.8% and 43.8%, respectively (Table 2). In the entire cohort (n = 172), the mean C/D ratio was 1.77 (minimum = 0.430, maximum = 6.58). Fluvoxamine use explained the highest variance in C/D ratios (R2 = 34.4%, p < 0.001), and the C/D ratios of individuals on fluvoxamine were higher by an average of 30.6% compared with those not using fluvoxamine (Table 2). The second strongest predictor of increase in C/D ratios was the elevation of CRP more than 10 mg/L (R2 = 8.7%, p < 0.001). The C/D ratios of individuals with elevated CRP were higher by an average of 22.9% relative to those with normal CRP levels (Table 2). Obesity (R2 = 2.2%, p = 0.007), nonsmoker status (R2 = 1.7%, p = 0.018), and female sex (R2 = 1.2%, p = 0.043) were associated with increased C/D ratios but explained a relatively modest proportion of the variance in C/D ratios. Notably, indigenous ancestry was associated with decreased C/D ratios in comparison with Asian ancestry (p = 0.016).
Multiple linear regression models for clozapine concentration-to-dose ratios* in full cohort and by fluvoxamine use.
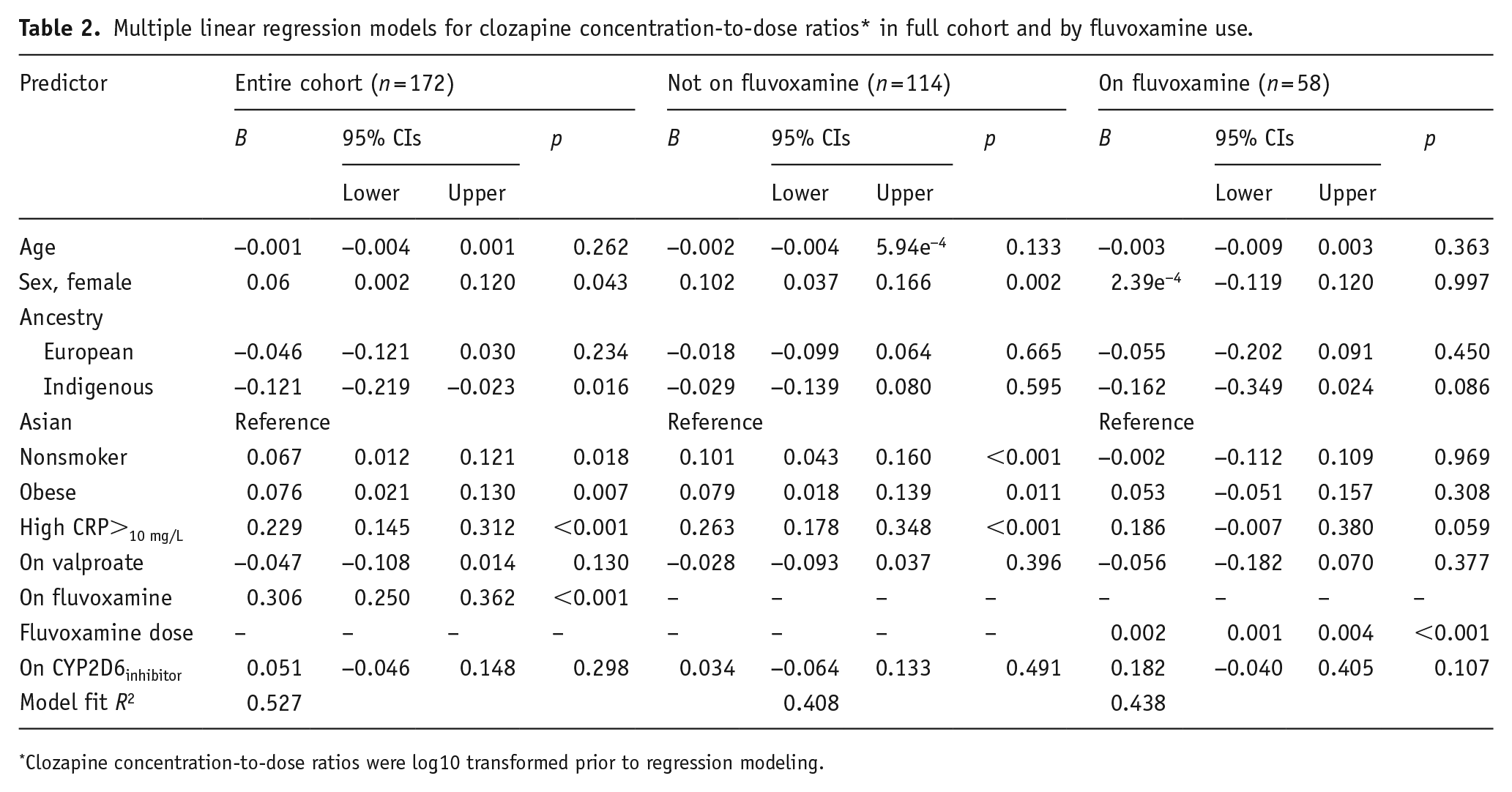
Clozapine concentration-to-dose ratios were log10 transformed prior to regression modeling.
In individuals not on fluvoxamine (n = 114), the mean C/D ratio was 1.34 (minimum = 0.430, maximum = 3.60). Model fit testing in this subgroup showed that elevated CRP was the strongest predictor of variability in C/D ratios (R2 = 21.7%, p < 0.001), with C/D ratios 26.3% higher among individuals with elevated CRP relative to those with normal CRP levels (Table 2). Like the entire cohort model, nonsmoker status (R2 = 6.7%, p < 0.001), female sex (R2 = 5.6%, p = 0.002), and obesity (R2 = 3.8%, p = 0.011) were associated with increased C/D ratios but explained a relatively modest proportion of the variance in C/D ratios.
Among participants using fluvoxamine (n = 58), the mean C/D ratio was 2.62 (minimum = 1.06, maximum = 6.58). Model fit testing in this subgroup revealed that only fluvoxamine dose was associated with an increase in C/D ratios (R2 = 20.1%, p < 0.001). For every 25 mg increase in fluvoxamine dose, the C/D ratios increased by an average of 5% (Table 2). Furthermore, examination of dose–response relationships showed positive correlations between C/D ratios (log10) and fluvoxamine dose (R = 0.515, p < 0.001) and CRP plasma levels (R = 0.339, p < 0.001) (Figure 1).
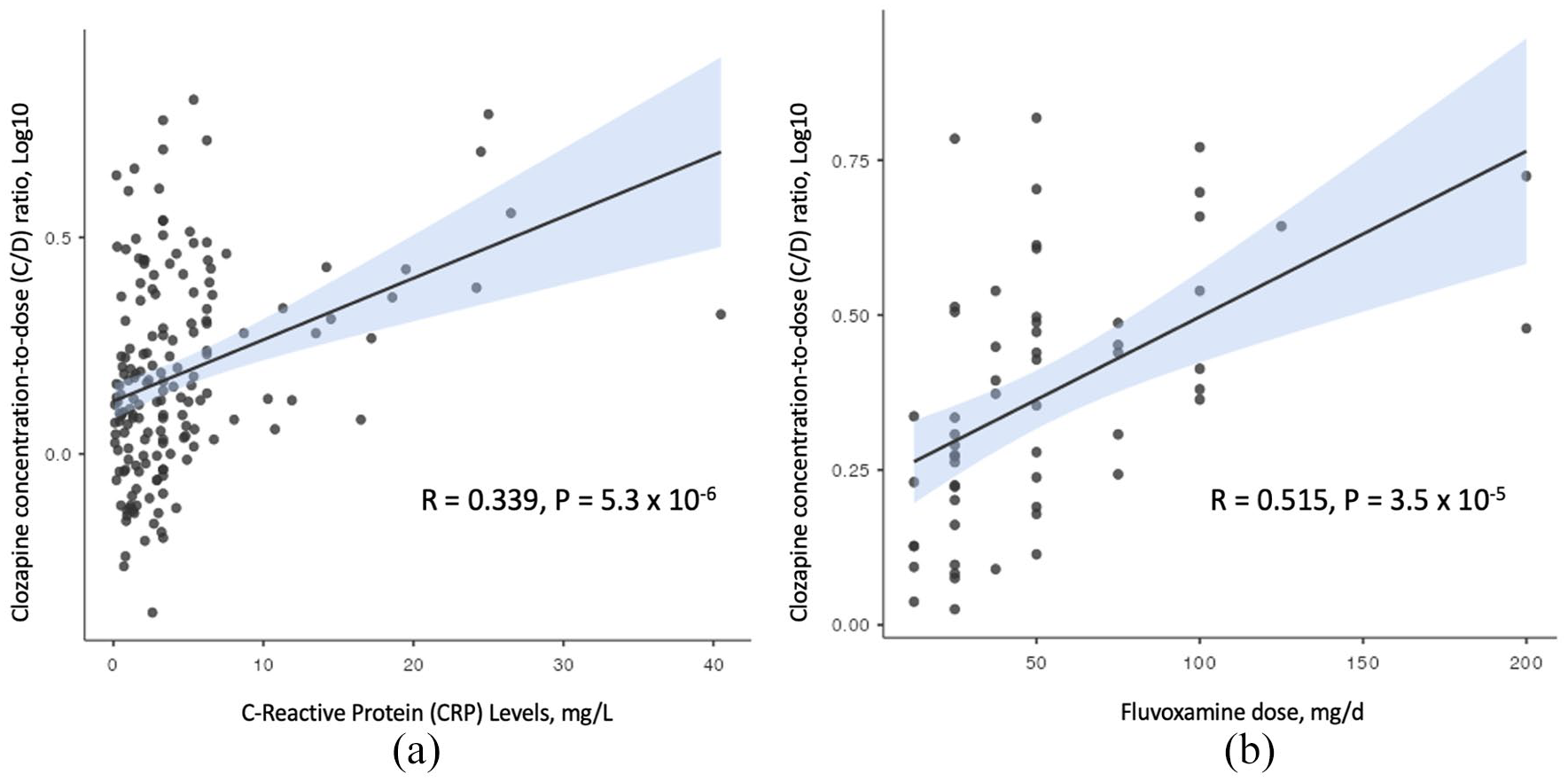
CRP n = 172 (a) and fluvoxamine n = 58 (b) dose–response relationship with clozapine plasma concentration-to-dose ratios.
Discussion
The direction and magnitude of patient-specific factors’ impact on clozapine’s rate of metabolism (measured by change in C/D ratios) was evaluated in a cohort of hospitalized patients with treatment-resistant psychotic illnesses. Given the inpatient and specialized setting of the study, there was high accuracy in the timing of clozapine dosing and blood samples for plasma concentration quantification. Furthermore, supervision by trained nurses increased the likelihood of high adherence rates.
Our results demonstrate that fluvoxamine has a strong, positive dose–response relationship with C/D ratios. Fluvoxamine use independently explained the most variance in clozapine C/D ratios and reduced the effect size of other patient-specific factors in the subgroup of participants prescribed fluvoxamine. Unlike previously published literature that suggested a possible balancing out effect between CYP1A2 inducing effects of smoking and CYP1A2 inhibiting effects of fluvoxamine augmentation (Augustin et al., 2019), our findings suggest that in a dose-related fashion, fluvoxamine will ultimately override the inducing effects of smoking. Consequently, concomitant use of fluvoxamine with clozapine, specifically at higher doses, requires rigorous TDM and should generally be avoided at the time of initiation of clozapine to decrease the risk of rapid dose accumulation and clozapine-associated inflammation (de Leon et al., 2022).
In the subgroup of patients not on fluvoxamine, CRP elevation was the strongest patient-specific factor in predicting an increase in clozapine levels followed by nonsmoking status, female sex, and obesity. Initiation of clozapine treatment is associated with inflammation, including but not limited to myocarditis, which requires close monitoring. CRP measures have been recommended at baseline prior to initiation of clozapine and weekly throughout the initial few weeks following drug commencement (Leung et al., 2023). Notably, compared to the general population, a diagnosis of schizophrenia and common comorbidities like metabolic syndrome are linked to greater levels of pro- and anti-inflammatory cytokines in the bloodstream as well as persistently elevated CRP (Goldsmith et al., 2016; Ridker et al., 2003). Using the cutoff of >10 mg/L, we were able to include individuals with persistent CRP elevation, and our results confirm its strong association with a decreased rate of clozapine metabolism not just in early treatment, but throughout. Therefore, CRP plasma levels have therapeutic significance when used in conjunction with TDM for dose adjustments and evaluation of dose-related adverse effects. Like fluvoxamine, the dose–response relationship between CRP levels and C/D ratios was strong and positive. This association has the clinical implication that acute infections or inflammations can precipitate clozapine toxicity.
In this study, Asian ancestry trended toward an association with a lower rate of metabolism without meeting statistical significance. A larger cohort free from the dominating effects of potent CYP1A2 inhibitors would likely be necessary to demonstrate statistical significance. This study could not confirm the findings of a prior small study that revealed Indigenous ancestry (González-Esquivel et al., 2021), similar to Asian ancestry (Pardiñas et al., 2023), may be linked with lower rates of clozapine metabolism. The present study is inconclusive given the small number of participants self-identifying as Indigenous Canadians, and the global designation “Indigenous people of Americas” may underestimate the genetic diversity and mixed ancestry in the vast continent and may overstate its impact on variability in plasma concentration of clozapine.
There are several limitations of this study. First, given the retrospective real-world nature of the clinical data, there may be some inaccuracies, such as the time of administration, patient adherence, and dietary habits. In addition, it was not possible to quantify the amount of coffee consumed or number of cigarettes smoked daily. However, these issues were mitigated as much as possible, as all patients were institutionalized and under direct supervision for dosing, blood sample collection, and day-to-day function. Second, in this study, ancestry was determined by self-identification and not by genetic testing. Nonetheless, this study is representative of real-world practice; genetic ancestry testing is not done in clinical settings. Third, we relied on a single measure of the serum clozapine level in patients with only one available clozapine TDM at steady-state conditions (n = 78), which cannot account for any intra-individual variation in clozapine kinetics. Fourth, we were not able to determine whether participants were assessed to be clozapine rapid metabolizers prior to the initiation of fluvoxamine by clinicians. Fifth, we acknowledge that we have combined two distinct diagnoses, schizophrenia and schizoaffective disorder which may have distinct overall underlying demographic and clinical factors.
Conclusion
In a clinical population, this study replicated the relationship between reduced rate of clozapine metabolism and the use of fluvoxamine, elevated CRP, obesity, nonsmoking status, and female sex, and the magnitude of the effects were large enough to be clinically relevant. Consequently, evaluation of patient-specific factors prior to and throughout clozapine treatment would be useful for individualization of speed of titration and dose optimization in conjunction with TDM. Because of the possibility of precipitating clozapine toxicity, the strong and positive dose–response correlations between fluvoxamine and CRP levels in reducing clozapine metabolism call for considerable caution.
Footnotes
Acknowledgements
We would like to recognize that our work takes place on historical and contemporary Indigenous lands. We also acknowledge the many Indigenous communities forged in Canada’s urban centers.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RR, AS, AR, NB, and KK have none to declare, AMB reports receiving research funding from Boehringer Ingelheim, RFW has received income from Canadian Agency for Drugs and Technologies in Health and Advisory Board Activities for HLS Therapeutics, CGS is a consultant to Clearmind Medicine, and CAB is founder and CEO of Sequence2Script Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
