Abstract
Background:
Previous research has suggested that controlled administration of psychedelic substances can modulate emotional reactivity, enhancing positive and diminishing negative emotions. However, it is unclear whether similar effects are associated with using psychedelics in less-controlled naturalistic environments.
Aims:
This cross-sectional study investigated the neural markers associated with the perception of emotional stimuli in individuals with extensive experience of naturalistic psychedelic use (15 or more lifetime experiences), comparing them to non-users.
Methods:
Electroencephalography (EEG) signals were recorded from two groups: experienced psychedelics users (N = 56) and non-users (N = 55). Participants were presented with facial images depicting neutral or emotional expressions (anger, sadness, and happiness). Event-related potential (ERP) components were analyzed as indices of emotional reactivity.
Results:
Psychedelic users were characterized by significantly lower amplitudes of the N200 component in response to fearful faces, in comparison to non-users. In addition, interaction effects between Group and Emotional expression were observed on N170 and N200 amplitudes, indicating group differences in the processing of fearful faces. However, no significant between-group differences emerged in the analysis of later ERP components associated with attention and cognitive processes (P200 and P300).
Conclusions:
The results suggest that naturalistic use of psychedelics may be linked to reduced reactivity to emotionally negative stimuli at the early and automatic processing stages. Our study contributes to a better understanding of the effects related to using psychedelics in naturalistic contexts.
Introduction
Psychedelics, such as psilocybin, lysergic acid diethylamide (LSD), mescaline, and N,N-dimethyltryptamine (DMT), are a class of psychoactive substances that can robustly alter perception and the state of consciousness. In the last decade, a growing number of studies have investigated the acute subjective effects of psychedelics and their neurophysiological mechanisms of action (Preller and Vollenweider, 2018; Stoliker et al., 2022; Vollenweider and Preller, 2020). A related line of research has focused on their potential to cause long-term changes in personality, mood, and well-being (Aday et al. 2020; Hendricks et al., 2015; Studerus et al., 2011), in both healthy and clinical samples.
Psychedelics induce profound changes in emotional experience (Preller and Vollenweider, 2018; Vollenweider and Preller, 2020), often leading to intense positive emotions and mystical-like states (Griffiths et al., 2006; Liechti et al., 2017). Subjective reports regarding psychedelic experiences are in line with the results of experimental studies revealing that psychedelics acutely affect behavioral and neural responses to emotional stimuli. Specifically, psilocybin was shown to impair recognition of sad and fearful faces (Dolder et al., 2016; Schmidt et al., 2013), and also to reduce both the amplitude of the face-selective N170 event-related potential (ERP) component in response to fearful faces (Schmidt et al., 2013) and the amplitude of the P300 ERP component in response to words inducing negative emotions (Kometer et al., 2012). Relatedly, low doses of LSD reduced the amplitudes of the N170 and P300 ERP components in an emotional-faces oddball task in a dose-dependent manner (Murray et al., 2021). Psychedelics have also been shown to modulate the activity and connectivity of the amygdala, which is a brain region involved in the processing of emotions, especially fear (LeDoux, 2003). Specifically, psilocybin reduces amygdala reactivity to negative stimuli (Kraehenmann et al., 2015) and disrupts its functional connectivity with other emotion-related brain regions, including the striatum, frontal pole, and ventromedial prefrontal cortex during perception of negative facial expressions (Grimm et al., 2018; Mertens et al., 2020). In line, LSD also lowered the reactivity of the amygdala and the medial prefrontal cortex in response to the presentation of fearful faces (Mueller et al., 2017). Regarding reactivity to positive stimuli, psychedelic substances have been shown to acutely promote goal-directed behavior toward positive cues (Kometer et al., 2012) and increase subjective vividness and neuronal activity during the recollection of positive autobiographical memories (Carhart-Harris et al., 2012). Thus, a growing number of studies suggest that, in research settings, psychedelics acutely alleviate the processing of negative and enhance the processing of positive emotional stimuli (review: Rocha et al., 2019).
Importantly, there is initial evidence that psychedelics might also cause more prolonged changes in emotional reactivity (Vollenweider and Preller, 2020). Barrett and Colleagues (2020) found that a single psilocybin session led to an increase in positive affect and a decrease in negative affect when measured 1 week after the psychedelic experience. Self-reported changes in affect intensity were accompanied by reduced amygdala activity during the processing of negative emotional stimuli and increased prefrontal cortex activity when processing emotionally conflicting stimuli. A month after the psilocybin session, negative affect and amygdala reactivity returned to the baseline level but the participants’ positive emotional state remained elevated, as suggested by a decrease in trait anxiety. A similar effect was reported in a study by Stroud et al. (2018), who measured long-term alterations in emotional processing through a behavioral assessment conducted 1 month after two psilocybin sessions separated by 1 week. Stroud et al. (2018) found that psilocybin improved the processing of emotional faces (i.e., reduced reaction times in an emotional recognition task) in patients diagnosed with treatment-resistant depression, and this improvement was correlated with reduced anhedonia. Finally, several previous cross-sectional and experimental studies have reported long-term improvements in well-being in both healthy and clinical populations after a single intake of psychedelics (Aday et al., 2020; Nichols et al., 2017; Vollenweider and Preller, 2020). While perception or processing of emotions was not measured directly in those studies, it is plausible that changes in emotional reactivity might be the mechanisms behind the observed changes in well-being (Andersen et al., 2021).
However, it is not known to what extent the acute and long-term effects of psychedelic sessions conducted in well-controlled laboratory or clinical settings apply to using psychedelic substances in less-controlled naturalistic environments. This is an important research question as the majority of psychedelics users use them in naturalistic contexts (Winstock et al., 2021) and the effects of naturalistic use may substantially differ from those observed in therapeutic settings (Carhart-Harris et al., 2018; Glynos et al., 2022). We therefore investigated the relation between naturalistic use of psychedelics and emotional reactivity in our recent cross-sectional study (Orłowski et al., 2022). Analyzing questionnaire data collected from a large sample of participants, we revealed that psychedelic users exhibit higher positive and lower negative emotional reactivity compared to non-users. Our results suggest that the naturalistic use of psychedelics might be associated with persistent trait-level “positivity bias” and are thus generally in line with experimental work on the acute or long-term effects of laboratory-based use (Barrett et al., 2020; Schmidt et al., 2013).
The aim of the present study was to further investigate how naturalistic use of psychedelics is related to emotional reactivity by using measurements that are independent of participants’ reports and are thus less prone to a range of subjective biases. To this end, we employed similar experimental procedures as used in studies on the acute effects of psychedelics (Kometer et al., 2012; Schmidt et al., 2013) to compare neural reactivity to emotional stimuli between groups of experienced psychedelic users and non-users. Specifically, participants were presented with images of faces with neutral or emotional expressions while electroencephalography (EEG) signals were recorded to perform an analysis of ERPs. Based on previously reported results (Kometeret et al., 2012; Schmidt et al., 2013), we preregistered our analysis of the N170 and P300 ERP components, which are well-established indexes of emotional reactivity (Olofsson et al., 2008). The N170 component is associated with early face processing and encoding and typically exhibits a higher amplitude in response to faces expressing negative emotions, such as anger or sadness, compared to faces expressing positive emotions, such as happiness or joy (Eimer, 2000; Pourtois et al., 2004). The P300 component, in turn, reflects higher-level cognitive processes, such as attention allocation, working-memory encoding, and evaluation of the emotional significance of stimuli (Polich, 2007; Olofsson et al., 2008); its amplitude reflects the emotional valence and level of arousal associated with stimuli (Hajcak et al., 2010; Foti et al., 2009). We expected the psychedelic users to be less reactive to negative emotional expressions (fear and anger) than the non-users; thus, we hypothesized that we would observe a lower amplitude of both the N170 and P300 ERP components in response to fear- and anger-expressing faces in the psychedelics users group but no between-group differences in response to neutral and happy expressions (i.e., an interaction effect).
Furthermore, we conducted exploratory (i.e., not pre-registered) analyses of the N200 and P200 ERP components, which are considered to reflect early attentional and perceptual processes involved in the evaluation of emotional stimuli (Schupp et al., 2006). The N200 component has been reported to be sensitive to the detection of emotional stimuli, and its amplitude is modulated by the level of arousal and valence of emotional stimuli (Olofsson et al., 2008), with several studies showing more negative N200 amplitude in response to fearful faces in comparison to neutral faces (Eimer and Holmes, 2007; Schupp et al., 2003). The P200 component is thought to reflect the initial perceptual processing of emotional stimuli, with larger amplitudes for emotional stimuli compared to neutral stimuli (Eimer and Holmes, 2007). By including these two components, the conducted analysis captured the whole range of processes involved in the perception of facial expressions: from the early perceptual and face-selective N170 component, through the mid-level attention-related N200 and P200 components, to the late P300 component, which is associated with cognitive processing. This allowed an exhaustive characterization of the potential differences between psychedelic users and non-users in terms of their reactivity to emotional stimuli.
Materials and methods
Participant sample
The present study’s methods and hypotheses were pre-registered on the Open Science Framework https://osf.io/z6tky). The study was conducted in accordance with the Declaration of Helsinki. The experimental procedure was approved by the Human Ethics Committee of SWPS University of Social Sciences and Humanities (Warsaw, Poland; 13/2020). All participants signed an informed consent document and received monetary compensation of 100 PLN (approximately 20 EUR). All participants were native Polish speakers and had normal or corrected-to-normal vision.
The sample size was not precalculated as we aimed to maximize the sample size in line with the available resources. The experimental sessions were conducted at two laboratories: at the Department of Psychology, SWPS University of Social Sciences and Humanities (Warsaw, Poland), where data were collected from 36 psychedelics users and 36 non-users; and at the Consciousness Lab, Psychology Institute, Jagiellonian University (Cracow, Poland), where data were collected from 20 psychedelics users and 19 non-users. In the conducted analyses, data from both laboratories were pooled, as described in subsequent sections. Thus, the analyzed sample consisted of 56 psychedelics users and 55 non-users (see Table 1 for characteristics of both groups), which is close to the preregistered sample size of 58 participants per group. Data from three additional participants (one user and two non-users) were excluded from the analysis due to technical problems with data recording.
The groups of users and non-users were compared in terms of demographic variables, substance use, and behavioral results of the experiment.
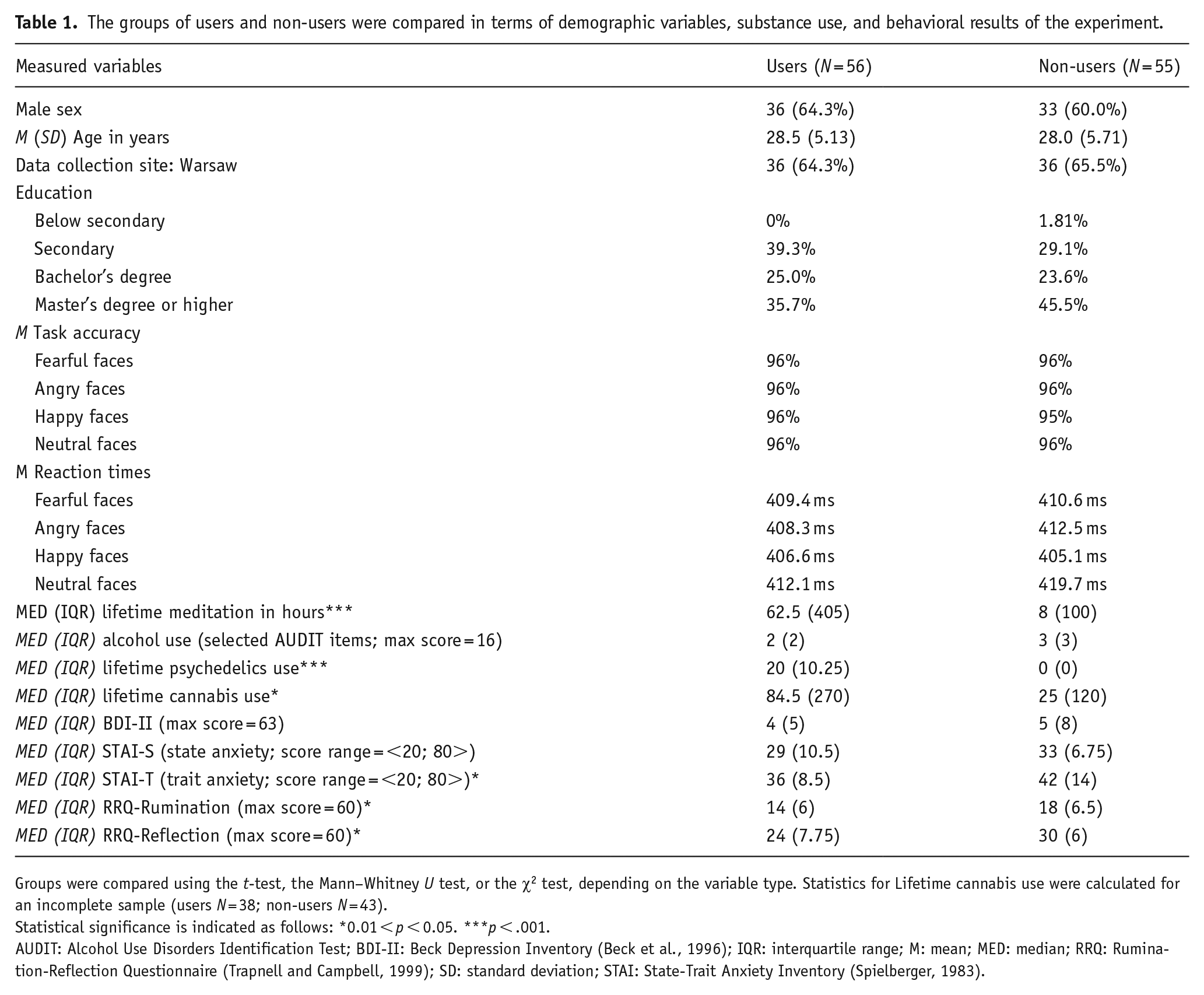
Groups were compared using the t-test, the Mann–Whitney U test, or the χ2 test, depending on the variable type. Statistics for Lifetime cannabis use were calculated for an incomplete sample (users N = 38; non-users N = 43).
Statistical significance is indicated as follows: *0.01 < p < 0.05. ***p < .001.
AUDIT: Alcohol Use Disorders Identification Test; BDI-II: Beck Depression Inventory (Beck et al., 1996); IQR: interquartile range; M: mean; MED: median; RRQ: Rumination-Reflection Questionnaire (Trapnell and Campbell, 1999); SD: standard deviation; STAI: State-Trait Anxiety Inventory (Spielberger, 1983).
Recruitment of participants
Participants were recruited via an online survey created with the open-source LimeSurvey software (Limesurvey GmbH, Hamburg, Germany). The survey was distributed online via the social media profiles of several Polish organizations involved in harm reduction and drug policy (politykanarkotykowa.pl; sin.org.pl; psychodeliki.org).
Participants first read and electronically accepted the informed consent form and the General Data Protection Regulation (GDPR) clause. Next, they provided detailed information regarding their history of psychoactive substance use (lifetime and the last year) and provided basic socio-demographic data, information about meditation practice, history of medication use, and psychological, psychiatric, and neurological disorders; they also completed a shortened version of the Alcohol Use Disorders Identification Test (items 1–4; Bush et al., 1998). Due to a programming error, data on cannabinoid use were collected only from a subsample of participants (n = 81, 38 users). Finally, participants were asked for consent to be contacted regarding the EEG study and about their preferred location for the EEG experimental session (Warsaw or Cracow). The survey was distributed in two versions: one version also contained a set of psychological questionnaires and took about 30 min to complete (the data collected in this version were published in Orłowski et al., 2022); the other version was used solely to recruit additional participants for the EEG study and took about 10 min.
Data from circa 5000 respondents were collected. The following exclusion criteria were used when screening the respondents for the EEG study: no consent to be contacted regarding the EEG study; frequent use of psychoactive substances such as empathogens, stimulants, or dissociatives (more than 15 lifetime or more than 5 in the past year); occasional use of substances from the class of benzodiazepines, synthetic cannabinoids, or opioids (more than three lifetime uses); obtaining 4 points for any question of the shortened version of the Alcohol Use Disorders Identification Test (AUDIT) questionnaire (0–4 point scale); diagnosis of a psychiatric or a neurological disorder; medical diagnosis of an addiction; and current use of prescribed drugs with psychoactive effects.
Respondents not meeting any of the described exclusion criteria were divided into two subgroups based on the number of lifetime experiences with psychedelics: users who reported 15 or more lifetime experiences with classical psychedelic substances, that is, LSD, psilocybin mushrooms, crystal DMT, changa, ayahuasca, mescaline, synthetic psilocybin or derivatives of any of these, that is, 5-MeO-DMT, 4-AcO-DMT; and non-users who had never used psychedelic substances but indicated a willingness to use them in the future. The subgroups were further subdivided according to the respondents’ preferred location for the experimental session (Warsaw or Kraków). Next, participants were chosen such that the subgroups (users and non-users) to be tested in each location were matched in terms of gender, age, education, and alcohol use. We aimed to also match both groups in terms of lifetime cannabinoid use (the estimated number of days in a lifetime on which cannabinoids were used) and meditation practice (the estimated number of lifetime hours spent on meditation) but we were not able to create a sufficiently large experimental group when considering these variables. Participants were invited to the laboratory in advance and asked to not use any psychedelic substances for at least 30 days before the EEG session (as the study aimed to investigate the long-term effects associated with the use of psychedelics).
Apparatus
The experimental procedure was written in Presentation® software (Version 22.1, Neurobehavioral Systems Inc., Berkeley, CA, USA). In Warsaw, a 24-inch DELL XL2411 (refresh rate: 144 Hz; resolution: 1920 × 1080; Dell Inc., Round Rock, Texas, USA) monitor was used for stimuli presentation; in Cracow, a 23.8-inch HP EliteDisplay E243i monitor (refresh rate: 60 Hz; resolution 1920 × 1080; Hewlett-Packard Company, Palo Alto, CA, USA) was used. The viewing distance was approximately 60 cm. A Cedrus Button Box (RB-840; Cedrus Corporation, San Pedro, CA, USA) with eight buttons were used to collect behavioral responses. In both laboratories, data collection took place in small isolated rooms. In Warsaw, the room was not sound-attenuated, but the room in Cracow was sound-attenuated. Participants were seated in comfortable chairs for the whole experiment.
Stimuli
Face images from the Warsaw Set of Emotional Facial Expression Pictures (Olszanowski et al., 2015) were used as stimuli. We selected 13 female (Models IDs: AD, JS, KO, KP, Ks, MJ, MK1, MR1, MS, OG, PS, SO, and SS) and 13 male models (Models IDs: AG, DC, HW, KA, KM, MG, MK, MR2, PA, PB, PO, RA, and RB). From each model, four images were used in the experimental procedure: angry, fearful, happy, and neutral facial expressions. Thus, 104 images were used as stimuli in total.
Experimental procedure
Each session started with the signing of the relevant documents, verbal instructions, and EEG preparation, all of which took around 45 min. Resting-state EEG data were recorded for 10 min at the beginning of each session (not analyzed in the present study). This was followed by a short break, during which the experimenter explained the details of the procedure to the participant.
The procedure consisted of 416 trials (104 per facial expression condition). Within each condition, female faces were presented in half of the trials, and male faces were used in the other half. Thus, the face of one actor or actress was presented four times per condition. Pictures of faces were presented in a pseudo-random order, with an additional constraint that images of the same facial expression or a person of the same gender would not be presented more than twice in a row. Participants were instructed to maintain their gaze on the fixation cross and perform a gender discrimination task. Specifically, they were asked to press one response pad button after seeing the face of a male actor, and another button after seeing the face of a female actor. The assignment of left and right response pad buttons to gender was counterbalanced across participants.
Each trial started with a fixation cross (size: 84 × 84 pixels), presented centrally for 700–1200 ms (chosen randomly for each trial; Figure 1). Next, a face image (image size: 1063 × 720 pixels) was displayed for 100 ms, followed by a question mark presented centrally and the letters “K” and “M” (the first letters of the words woman and man in Polish) presented on the left and right sides of the question mark. Participants responded by pressing one of two response pad buttons with the index finger of their left or right hand. The left button of the response pad always corresponded to the letter displayed on the left side of the screen, while the right button corresponded to the letter displayed on the right side of the screen. Participants were instructed to perform the task as quickly and accurately as possible. Pressing a response button ended the current trial and started the next one. At the beginning of the procedure, participants performed a brief training session of eight trials before moving to the main part of the procedure. A break was provided after every 104 trials, the duration of which was controlled by participants (they ended a break by pressing a button). The duration of the whole procedure was circa 15 min.
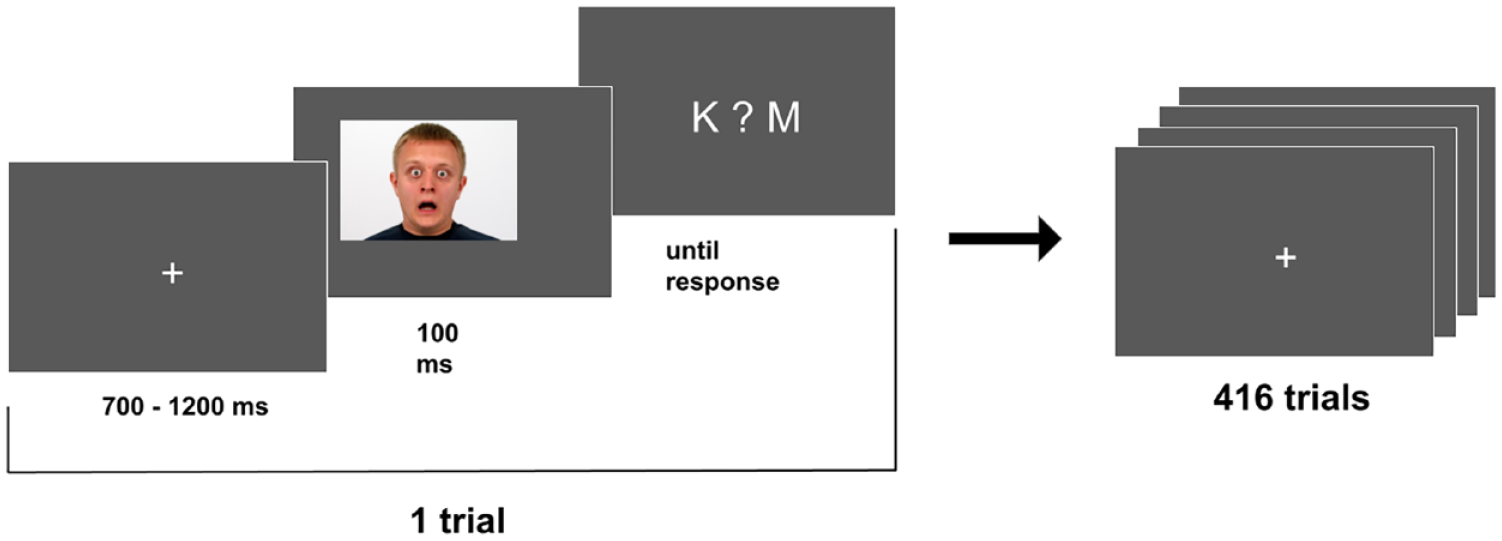
Scheme of the experimental procedure.
EEG recording
In Warsaw, EEG was recorded from 64 scalp sites (Ag/AgCl active electrodes placed according to an extended 10–20 system) using a Brain Products ActiCap system and the Brain Vision Recorder software at a sampling rate of 1000 Hz. In Cracow, the EEG signal was recorded from 64 scalp sites (Ag/AgCl active electrodes placed according to an extended 10–20 system) using a BioSemi ActiveTwo amplifier system (BioSemi, Amsterdam, Netherlands) and the BioSemi ActiView software at a sampling rate of 2048 Hz. In both laboratories, we additionally recorded signals from both earlobes. Moreover, four electrooculograms—2 vertical (VEOG) and two horizontal (HEOG)—were recorded using bipolar electrodes (VEOG placed at the supra- and infra-orbit of the left eye; HEOG placed at the external canthi of the left and right eyes).
EEG preprocessing
Custom-made Matlab scripts based on EEGlab functions (Delorme and Makeig, 2004) were used for the preprocessing of EEG and EOG data. Raw data were filtered using a 0.1 Hz high-pass FIR filter (33,792 filter order) and a 40 Hz low-pass FIR filter (220 filter order). EEG signals were then re-referenced to the mean value of all scalp electrodes and down-sampled to 250 Hz. The clean_channels() function from the clean_rawdata plugin (https://github.com/sccn/clean_rawdata) was used to automatically identify noisy channels and remove them from the data (2.95 ± 2.38 channels per participant). The removed channels were spherically interpolated based on the activity of neighboring channels. Next, the continuous signal was divided into epochs time-locked to the stimulus (face image) presentation (−400 ms to 1200 ms; 416 epochs in total; 104 per condition). Epochs containing no behavioral response, incorrect responses (17.01 ± 13.25 epochs per participant), or in which the response was faster than 100 ms after the stimulus (6.64 ± 13.53 epochs per participant) were excluded from further analysis. The FASTER plugin (Nolan et al., 2010) was then used to automatically detect epochs containing artifacts and to remove them from the data (18.89 ± 7.53 epochs per participant). Next, to remove residual artifacts from the data, independent component analysis (ICA) was used. To obtain the most optimal decomposition, ICA was performed on data filtered with a 1 Hz high-pass filter (other preprocessing steps were the same as described above). Signals were decomposed into 50 independent components and the obtained ICA decomposition was exported and used to automatically identify artifactual components in the main dataset (filtered with 0.1 Hz high-pass filter). Artifactual components were identified by the Multiple Artifact Rejection Algorithm and removed from the data (41.31 ± 4.53 components removed per participant). As the final step, signals from the scalp electrodes were re-referenced to the mean of the earlobe electrodes.
The numbers of epochs that entered the statistical analysis did not differ statistically between groups (users: 373.38 ± 18.89; non-users: 372.96 ± 18.58) or between conditions (Angry: 93.14 ± 5.46; Fearful: 93.54 ± 5.43; Happy: 93.36 ± 5.33; Neutral: 93.14 ± 5.00).
ERP analysis
For the present study, we pre-registered analyses of two ERP components: N170 and P300. The spatiotemporal windows used to analyze each component were defined as follows: First, topographic maps averaged over all participants, and conditions (Figure 2) were used to define clusters of electrodes at which each component reached its maximum amplitude. Then, the N170 component was identified in two symmetric occipitoparietal clusters (electrodes: P7, TP7, P8, and TP8), while the P300 component was identified in the centroparietal cluster (electrodes: CP1, CPz, CP2, P1, Pz, and P2). Next, for each ERP component, the time window was specified based on visual inspection of waveforms that were averaged within the chosen cluster of electrodes (Figure 2). A time window of 132–200 ms was selected for the N170 component, while for P3 it was 320–520 ms. To avoid inflation of the type I error rate, the spatiotemporal windows were defined independently of differences caused by experimental manipulations (Keil et al., 2014).
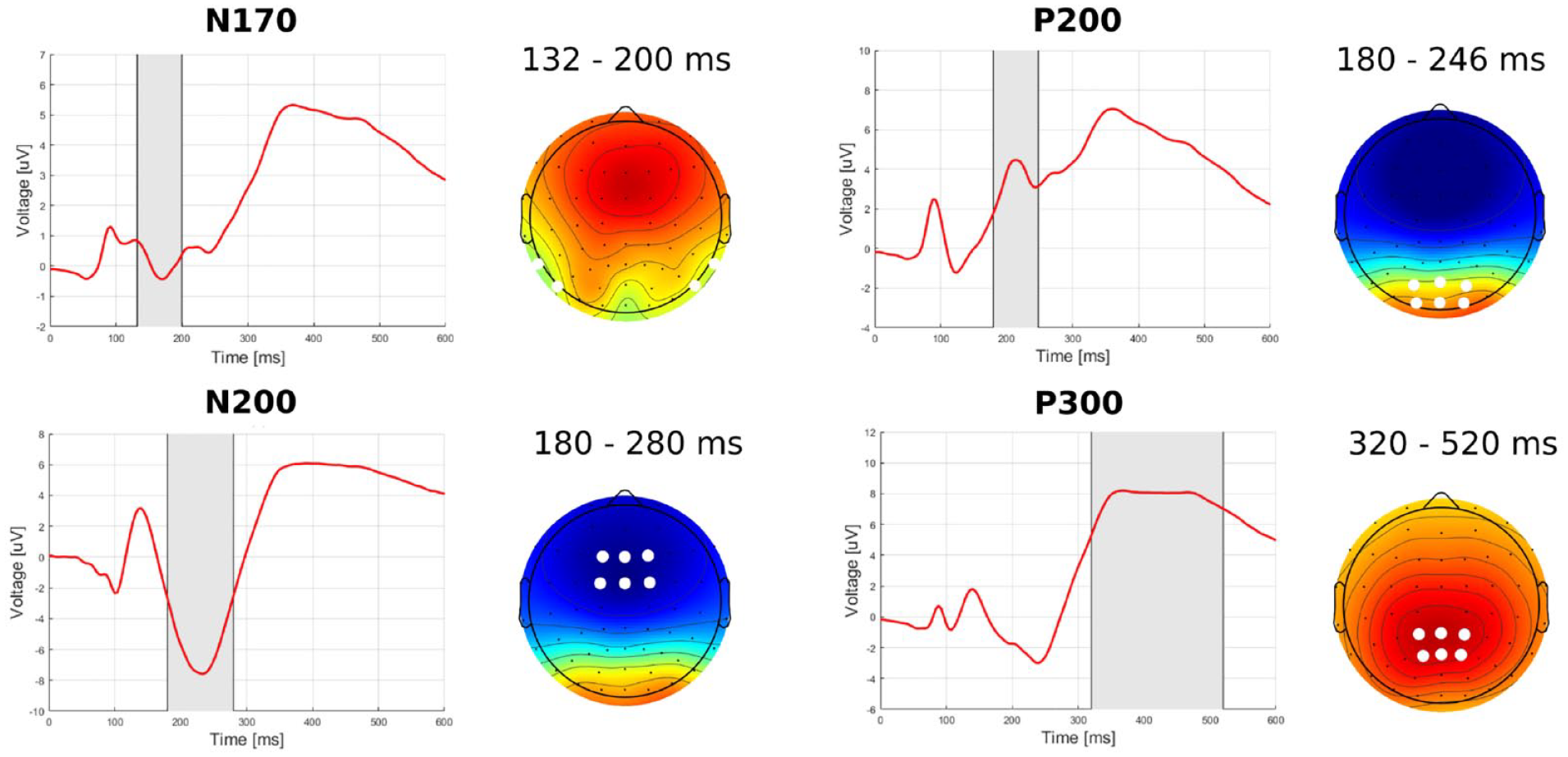
Waveforms of ERP components averaged over all conditions, participants, and electrodes selected for a given component (marked by white dots). The analyzed time windows are marked in gray. Topographic maps of EEG activity averaged over all participants and conditions and within the selected time window.
In addition to the pre-registered analysis, we conducted an exploratory analysis by examining the topographic maps and grand-average ERPs. This led to the identification of two additional components, which we analyzed in a non-pre-registered manner. The first component was the occipitoparietal P200, which typically represents various aspects of perceptual processing and attention (Rosburg et al., 2009, Singhal et al., 2002). The second component was the frontal N200, which is associated with the processing of emotional stimuli and spatial attention (Eimer and Holmes, 2007; Olofsson et al., 2008). The electrode clusters (P200: PO3, POz, PO4, O1, Oz, and O2; N200: F1, F2, Fz, FC1, FC2, and FCz) and time windows (P200: 180–248 ms; N200: 180–280 ms) of these components were identified in the same way as for N170 and P3.
For all four components, mean amplitudes computed for each trial within the defined spatiotemporal windows (i.e., average over selected electrodes and timepoints) were used in further single-trial statistical analyses.
Statistical analysis
All statistical analyses were conducted in R (R Core Team, 2022). We used linear mixed models (R lmerTest package; Kuznetsova et al., 2017) to analyze the effects of group (psychedelics users, non-users) and facial expression (angry, fearful, happy, and neutral) on the amplitude of the ERP components. A random effect of the participant was included in all models. In addition, for each ERP component, we constructed a model incorporating the fixed effect of the data collection site (Warsaw or Cracow). The recruited groups were not matched in terms of meditation practice, which can have similar effects on emotionality as the use of psychedelics (Millière et al., 2018; Orłowski et al., 2022; Simonsson and Goldberg, 2023; Tang et al., 2016), we utilized weights in our statistical models to mitigate the influence of individuals with a history of extensive meditation practice following the methodological guidelines of the statistical tool employed (Bates et al., 2015). Weights were generated as follows: (1) the number of lifetime meditation hours is transformed using the Yeo-Johnson method (Weisberg, 2001) to decrease dispersion (range 0–2000); (2) values are centered around five to obtain positive values only; (3) the inverse of the values is computed to down weight participants with higher numbers of lifetime mediation hours. In the Results section, we present the results of weighted models. In addition, we conducted control analyses using unweighted models. Details of the weighing procedure and the results of the unweighted models, which provided qualitatively similar results, have been included in the Supplemental Material.
In both analyses (the main with weighted models and the control with unweighted models), the models with and without the fixed effect of the data collection site were compared in terms of the Bayesian Information Criterion, and the model with a significantly better fit to the data was selected and reported in the Results section. In cases where there were no significant differences between models, the simpler model (i.e., without a data collection site) was chosen. Post hoc tests for all significant effects were corrected for multiple comparisons using the Holm method.
Results
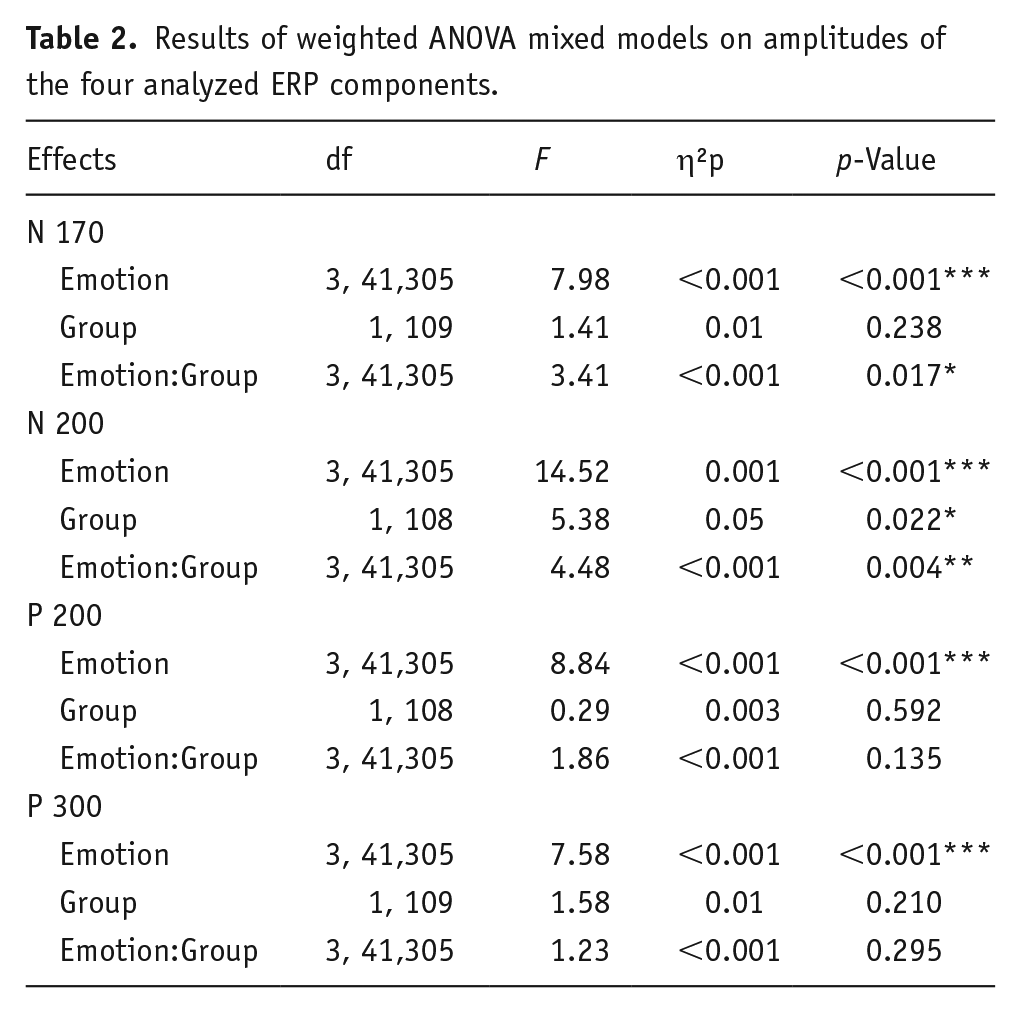
Results of the mixed-model analysis of variance (ANOVA) conducted for each ERP component are presented in Table 2.
Results of weighted ANOVA mixed models on amplitudes of the four analyzed ERP components.
N170
We found a significant effect of facial emotional expression on N170 amplitude (p < 0.001, Table 2). The post hoc tests indicate that the perception of Fearful faces was associated with lower N170 amplitude (less negative value) than the perception of Happy (z = 3.01, p = 0.011) or Neutral (z = 2.57, p = 0.030) ones. Also, the perception of angry faces resulted in lower N170 amplitude compared to happy (z = 4.14, p < 0.001) and neutral faces (z = 3.70, p = 0.001).
The main effect of the Group was not significant (p = 0.238, Table 2) but we found a significant Emotion x Group interaction (p = 0.017, Figure 3(a)). Post hoc comparisons within groups indicated that the amplitude of the N170 component in the psychedelics user group was lower during the perception of Fearful faces than during the perception of Neutral (z = 3.75, p = 0.012, Figure 3(a)) or Happy faces (z = 3.57, p = 0.023, Figure 3(a)). By contrast, we did not find any significant differences between the processing of different emotions within the non-user group. We also did not find any between-group effects within any of the Emotion conditions (Angry: z = 0.93, p = 0.87; Fearful: z = 1.85, p = 0.26; Happy: z = 1.06, p = 0.87; Neutral: z = 0.80, p = 0.87).
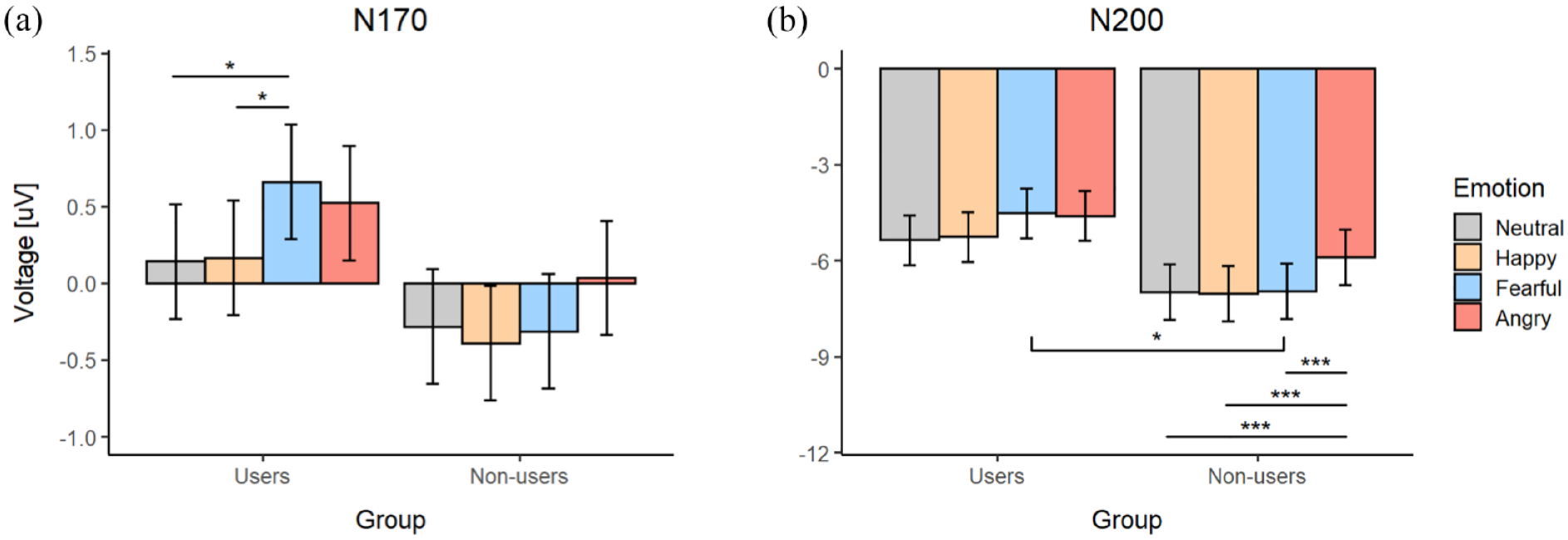
Weighted mean values of N170 (a) and N200 (b) ERP components for four emotional facial expressions (neutral, happy, fearful, and angry) in the two groups: psychedelic users and non-users. The y-axis represents values of N170 and N200 amplitudes in microvolts (μV); error bars represent standard error (SE) values. The statistical significance of post hoc comparisons is indicated as follows: *p < 0.05. ***p < 0.001.
P300
A significant effect of facial emotional expression was found on P300 amplitude (p < 0.001, Table 2). Post hoc comparisons indicated that the perception of Fearful faces was associated with greater (more positive) P300 amplitude in comparison to the perception of Neutral (z = 4.72, p < 0.001) or Happy faces (z = 2.93, p = 0.017). However, neither the Group effect nor the Emotion × Group interaction effect was statistically significant (Table 2).
N200
Once again, a significant effect of facial emotion was found (p < 0.001, Table 2). Specifically, the perception of Angry faces was associated with a lower N200 amplitude (less negative values) in comparison to all other expressions (Angry vs Fearful: z = 3.58, p = 0.001; Angry vs Happy: z = 5.52, p < 0.001; Angry vs Neutral: z = 5.88, p < 0.001). Next, we found a significant effect of Group on N200 amplitude (p = 0.022, Table 2, Figure 4). Here, the users group exhibited lower N200 amplitudes than the non-users group.
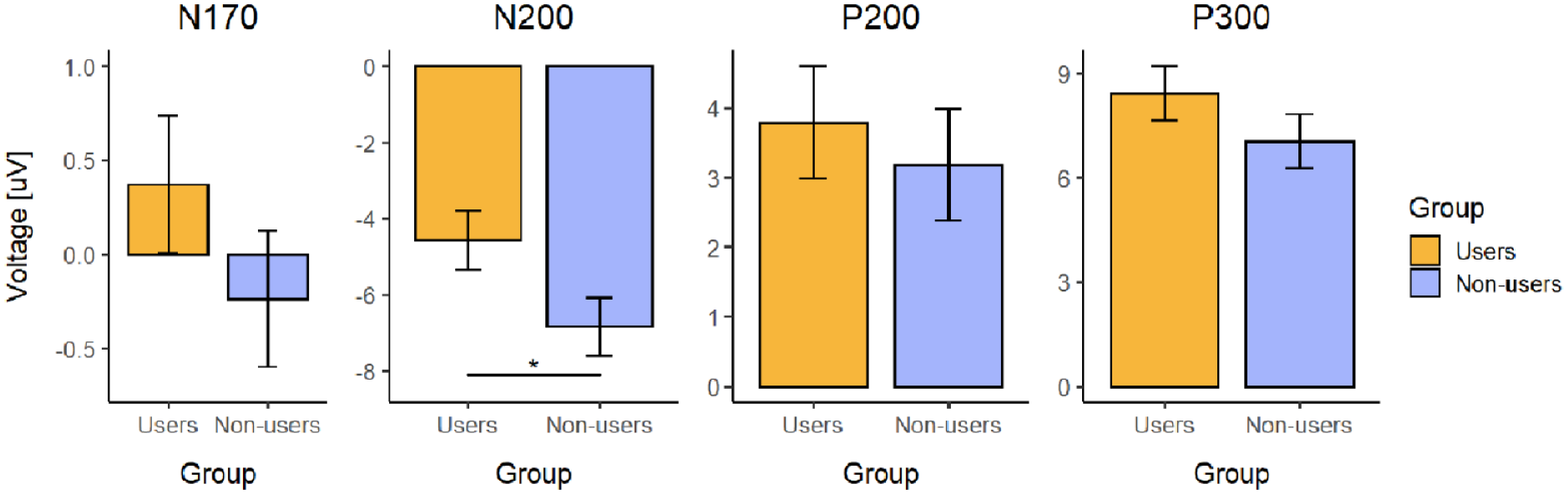
Weighted mean values of analyzed ERP component amplitudes between two groups: psychedelic users and non-users. The y-axis represents values of amplitudes of components in microvolts (μV) and error bars represent SE values. Statistical significance of post hoc comparisons: *p < 0.05.
Furthermore, we found a significant Emotion x Group interaction (p = 0.004, Figure 3(b)). Post hoc comparisons revealed that the perception of Fearful faces resulted in lower amplitude in the users than in the non-users group (z = 2.79, p = 0.021, Figure 3(b)), but the groups did not differ in terms of N200 response to other facial expressions (Angry: z = 1.75, p = 0.081; Happy: z = 2.34, p = 0.059; Neutral: z = 2.27, p = 0.059).
Next, post hoc comparisons within groups indicated that N200 amplitude in the non-user group was lower during the perception of Angry faces than during the perception of faces belonging to the other conditions (Angry vs Fearful: z = 5.28, p < 0.001; Angry vs Happy: z = 5.55, p < 0.001; Angry vs Neutral: z = 5.63, p < 0.001; Figure 3(b)). By contrast, within the psychedelics users group, we did not find any significant differences between the processing of different emotional expressions.
P200
Facial emotional expression had a significant effect on P200 amplitude (p < 0.001, Table 2). Specifically, the perception of Angry faces was associated with greater (more positive) P200 amplitude in comparison to other emotional expressions (Angry vs Fearful: z = 3.90, p < 0.001; Angry vs Happy: z = 3.81, p < 0.001; Angry vs Neutral: z = 4.68, p < 0.001). However, we found a main effect of neither Group nor the Emotion × Group interaction (Table 2).
Discussion
In recent years, we have seen growing interest in investigating the mechanisms of action and the psychological consequences of using psychedelic substances. While there is evidence that classic psychedelics may have a lasting positive impact on well-being and quality of life (reviews: Aday et al., 2020; Vollenweider and Preller, 2020), these effects have been observed after psychedelic sessions carried out in well-controlled laboratory or clinical settings under the supervision of a trained therapist. Therefore, it remains unclear whether qualitatively similar effects are associated with using psychedelics in naturalistic environments, which are the primary settings for the vast majority of psychedelics users (Winstock et al., 2021) but typically involve less precise control over substance quality, dosage, and situational factors (Golden et al., 2022).
That naturalistic use of psychedelics might be associated with better mental health, including more positive emotional reactivity, has been suggested by cross-sectional studies using surveys and questionnaires to measure the variables of interest (Jiménez-Garrido et al., 2020; Nygart et al., 2022; Orłowski et al., 2022; Raison et al., 2022). In line, a recent prospective longitudinal study by Nayak and colleagues (2023) found that after a single self-administration of psilocybin in a naturalistic context participants reported enduring reductions in anxiety, depression, and alcohol misuse; and exhibited enhanced cognitive flexibility, emotion regulation, spiritual well-being, and extraversion. Nonetheless, it is crucial to recognize that the methodology employed in these studies is limited by a range of biases associated with the self-reported nature of measurements (Karpen, 2018), including those related to memory accuracy (Levine et al., 2002) and social norms (Fay et al., 2012). Therefore, in the present pre-registered study, we investigated the association between the naturalistic use of psychedelics and emotional reactivity by measuring brain activity evoked by emotional stimuli in a large sample of experienced psychedelics users (N = 56) and a matched control group of non-users (N = 55). Specifically, we recorded EEG while participants were presented with images of faces with neutral or emotional expressions (fear, anger, and happiness), and we analyzed four ERP components, which provided markers of emotional reactivity that were independent of participants’ reports.
Main ERP findings in the context of previous studies on acute and long-term effects of psychedelics
Starting with the early N170, which is a well-established marker of face-encoding that is particularly sensitive to the emotional content of facial expressions (Eimer, 2000; Pourtois et al. 2004), our analysis revealed that psychedelic users exhibit lower N170 amplitude during perception of fearful faces compared to perception of neutral and happy faces. By contrast, no significant differences in N170 amplitude were observed between emotional expressions in the non-users group. Similarly, when analyzing the subsequent frontal N200 component, which is thought to reflect processing of the emotional significance of stimuli and to be modulated by affective arousal and valence judgments (review: Olofsson et al., 2008), we found that the psychedelic users exhibited lower N200 amplitude during perception of fearful faces compared to the non-users. Therefore, the ERP analysis suggests that using psychedelics in naturalistic settings might be associated with weaker neural responses to negative emotional stimuli in the early and automatic stages of their evaluation. In relation to previous work, our results are consistent with studies which found that administration of psychedelics acutely impairs recognition of fearful faces (Kometer et al., 2012; Schmidt et al., 2013), reduces amplitude of the face-selective N170 ERP component (Schmidt et al., 2013), and decreases reactivity of the right amygdala during processing of negative emotional pictures (Kraehenmann et al., 2015) and fearful faces (Mueller et al., 2017). However, it is important to keep in mind that while these previous studies investigated the causal link between psychedelics and the perception of emotions, our work provides only correlational evidence. Furthermore, the ERP findings reported here are also in line with the results of our recent analysis of questionnaire data from a large sample of participants (N = 2561, 1661 psychedelics users), which revealed that the user’s group was characterized by lower negative and higher positive emotional reactivity compared to non-users, and the number of lifetime psychedelic experiences was correlated to the magnitudes of the observed effects (Orłowski et al., 2022). Therefore, despite differences in the methods used to measure emotional reactivity, both the present ERP study and the previous questionnaire study provide consistent results, thus indicating psychedelic users’ lower reactivity to negative emotional stimuli.
However, we did not find any between-group differences or interactive effects in the later attention- and cognition-related ERP components (P200 and P300). P200 is considered to reflect late attentive information processing (Rosburg et al., 2009, Singhal et al., 2002), while P300 reflects higher-level cognitive processes, such as attention allocation and evaluation of the significance or relevance of stimuli (Olofsson et al., 2008; Polich, 2007). Kometer and colleagues (2012) reported that psilocybin acutely reduced P300 amplitude in response to negative emotional stimuli, whereas we found no P300 effect in our data. Thus, our results suggest that long-term psychedelic use may be related to effects at the early and automatic stages but not necessarily in the later and cognition-related stages of emotional processing. However, further electrophysiological studies are needed to support this conclusion.
We also did not find any between-group behavioral differences, neither in accuracy nor in reaction times. This indicates that emotional expressions did not interfere with the gender discrimination task, possibly due to its low difficulty, as evidenced by the ceiling effect of accuracy scores (Table 1). Finally, we did not observe differences in any of the analyzed ERP components that would indicate differences in the perception of positive emotions between users and non-users, although using psychedelics was associated with greater positive emotional reactivity in the above-mentioned questionnaire study (Orlowski et al., 2022). Thus, while at present there is evidence that using psychedelics is associated with attenuated negative emotional reactivity, whether or not greater positive emotional reactivity is also observed in psychedelics users remains to be investigated.
Methodological aspects and limitations
To facilitate between-experiment comparisons, in our study, we utilized images of facial emotional expressions, as they have also been used in previous work on emotional processing in the acute phase of the psychedelic experience (Grimm et al., 2018; Kometer et al., 2012; Kraehenmann et al., 2015; Mueller et al., 2017; Schmidt et al., 2013). However, an important difference is that we used the gender discrimination task, which did not require active evaluation of the emotional content of stimuli, while previous studies investigating the acute effects of psychedelics used emotion discrimination or recognition tasks (i.e., emotions were task-relevant; Grimm et al., 2018; Kometer et al., 2012; Kraehenmann et al., 2015; Mueller et al., 2017; Schmidt et al., 2013). Nevertheless, the fact that we found a main effect of emotional expression in each analyzed ERP component validates our procedure and confirms that it was sensitive enough to capture the effects of emotions, even when they were task irrelevant.
To ensure that the experimental group consisted of individuals who had used psychedelics for an extended period of time, we recruited users with at least 15 experiences with classic psychedelics (excluding microdosing). Furthermore, all participants reported that they had not used any psychedelic substances in the 30 days before the EEG measurement, which was essential to capture only the long-lasting effects associated with psychedelic use. This 30-day period was chosen on the basis of the results of Barrett et al. (2020), who demonstrated that brain responses to facial affect stimuli returned to the baseline levels after 30 days of a single-dose psilocybin experience.
Importantly, in our study, the control group consisted of participants who did not have any previous experiences with psychedelics but indicated interest in using these substances in the future. Furthermore, because both users and non-users were recruited through the same social media profiles dedicated to psychedelics, we assume that the control group participants exhibited interest in psychedelic substances. Although the cross-sectional design of our study prevents us from inferring causality, this approach reduces the possibility that any of the observed effects could be attributed to differences in lifestyle or personality traits rather than effects related to using psychedelics.
Subjective experiences induced by psychedelics appear to share similarities with those associated with meditation (Millière et al., 2018; Simonsson and Goldberg, 2023). Specifically, both practices are associated with improved emotional regulation (Orłowski et al., 2022; Tang et al., 2016); also, both have therapeutic potential for mood disorders (Marchand, 2012; Pascoe et al., 2021; Vollenweider and Kometer, 2010) and have similar effects on neuronal activity, such as reduction of activity in the default mode network (Gattuso et al., 2023; Jerath et al., 2012; Taylor et al., 2013). In our study, we attempted to control for confounding variables such as lifetime meditation practice, but despite collecting data from around 5000 responders we were not able to create subgroups of psychedelics users and non-users that did not differ in terms of meditation practice (Table 1). This indicates a strong relationship between both practices in the general population and highlights the need for future cross-sectional studies on psychedelics to control for meditation practice (and vice versa). To address this limitation in our study, we employed a statistical weighting approach which reduced the impact of individuals with extensive meditation experience. Interestingly, the inclusion of weights in our statistical models increased the interactive effects, which were the primary focus of this study. It is also worth noting that in our previous questionnaire study, we found that psychedelics had an effect on emotional reactivity independently of meditation (Orłowski et al., 2022). Unfortunately, in the present study, the sample size was insufficient to conduct such statistical analyses.
Furthermore, we were not able to match the users and non-user groups in terms of lifetime cannabis use. Importantly, cannabis use has been associated with alterations in emotional processing, including difficulties in accurately perceiving and distinguishing the intensity of emotions (MacKenzie and Cservenka, 2023). Considering the strong correlation between the use of these substances in the general population, isolating their individual effects becomes a significant challenge for studies examining naturalistic substance use. Regarding other limitations of our study, we did not collect data about the contextual factors and intentions underlying participants’ psychedelic use. These are important components of the “set and setting,” which could significantly influence the trajectory of the psychedelic experience and its long-term effects, including those related to emotional processing (Carhart-Harris et al., 2018). Moreover, it is crucial to acknowledge the potential impact of self-selection bias, as individuals who have had negative or challenging experiences with psychedelics may be less inclined to use them repeatedly, and may also be less likely to participate in research studies, potentially leading to an overrepresentation of participants with predominantly positive experiences (Aday et al., 2020; Muthukumaraswamy et al., 2022). Together, these limitations underscore the importance of interpreting our results with caution and suggest that future research may benefit from employing more controlled and context-specific methodologies.
Conclusion
In conclusion, the present study found differences in emotional reactivity to negative emotional stimuli between experienced users of classic psychedelics and non-users. Our data suggest the naturalistic use of psychedelics may be associated with attenuation of the initial and automatic phase of emotional processing but not necessarily of later stages involving cognitive processes. Therefore, our findings align with previous research indicating that psychedelic use may be related to a lasting reduction of negative emotional reactivity, a mechanism that potentially contributes to improved quality of life and well-being. Our study contributes to a better understanding of the effects related to using psychedelics in naturalistic contexts but, at the same time, exhibits limitations typical of cross-sectional studies, including the inability to establish causality and to exclude potential impact of confounding variables.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231216322 – Supplemental material for The relation between naturalistic use of psychedelics and perception of emotional stimuli: An event-related potential study comparing non-users and experienced users of classic psychedelics
Supplemental material, sj-docx-1-jop-10.1177_02698811231216322 for The relation between naturalistic use of psychedelics and perception of emotional stimuli: An event-related potential study comparing non-users and experienced users of classic psychedelics by Paweł Orłowski, Justyna Hobot, Anastasia Ruban, Jan Szczypiński and Michał Bola in Journal of Psychopharmacology
Footnotes
Acknowledgements
We thank Stanisław Adamczyk, Agata Chrobak, Amanda Hajska, Małgorzata Paczyńska, and Barbara Zając for assistance in data collection.
Author’s Note
Michał Bola is also affiliated to Centre for Brain Research, Jagiellonian University, Kraków, Poland.
Author contributions
PO: designed the study, programmed the experimental procedure, collected the data, analyzed the data, interpreted the data, and wrote and revised the manuscript. NR: designed the study, collected the data, interpreted the data, and revised the manuscript. JH: designed the study, collected the data, interpreted the data, and revised the manuscript. JS: analyzed the data, interpreted the data, and revised the manuscript. MB: obtained the funding, conceived the study, designed the study, interpreted the data, and revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a National Science Center Poland grant (2020/39/O/HS6/01545). Jan Szczypiński was supported by the Foundation for Polish Science START scholarship and NCN ETIUDA grant (2020/36/T/HS6/00092).
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
