Abstract
Background:
Growing clinical interest in psychedelic-assisted therapies has led to a second wave of research involving psilocybin, lysergic acid diethylamide (LSD), 3,4-methylenedioxymethamphetamine (MDMA) and other substances. Data suggests that these compounds have the potential to treat mental health conditions that are especially prevalent in older adults such as depression, anxiety, existential distress, and posttraumatic stress disorder.
Aims:
The goal of this study was to quantify the prevalence of older adults enrolled in psychedelic clinical trials and explore safety data in this population.
Methods:
A systematic review was conducted following the 2020 PRISMA guidelines. Search criteria included all trials published in English using psychedelic substances to treat psychiatric conditions, including addiction as well as existential distress related to serious illness. Articles were identified from literature searches on PubMed, EBSCO, and EMBASE.
Results:
4376 manuscripts were identified, of which 505 qualified for further review, with 36 eventually meeting eligibility criteria. Of the 1400 patients enrolled in the 36 studies, only 19 were identified as 65 or older, representing less than 1.4% of all trial participants. For 10 of these 19 older adults, detailed safety data was obtained. No serious adverse events (AEs) occurred in any older adults and only transient mild-to-moderate AEs related to anxiety, gastrointestinal upset, and hypertension were reported during the psychedelic dosing sessions.
Conclusions:
While existing data in older adults is limited, it suggests that psychedelic-assisted psychotherapy can be safe and well tolerated in older adults. Therefore, psychedelic-assisted psychotherapy should be more rigorously investigated for the treatment of psychiatric conditions in this population.
Introduction
Research on psychedelic compounds for the treatment of psychological distress has undergone a resurgence after several decades of legal challenges. In the United States, two compounds, psilocybin and 3,4-methylenedioxymethamphetamine (MDMA), have even been given Breakthrough Therapy designation from the Food and Drug Administration for the treatment of major depressive disorder (MDD) and posttraumatic stress disorder (PTSD) respectively following promising results in phase II and III clinical trials (Reiff et al., 2020). Research and regulatory access for psychedelic-assisted therapies are evolving rapidly in many other countries as well, with increasing interest and acceptance. The increasing momentum around the application of psychedelics in clinical settings builds on more than 20 years of research conducted from the 1950s to the early 1970s, during which time approximately 40,000 individuals were treated with psychedelic compounds (Nichols, 2016).
Unfortunately, older adults have been underrepresented in psychedelic-assisted therapy clinical trials (Johnston et al., 2022; Lau et al., 2022). In fact, pioneering trials of psilocybin-assisted therapy for the treatment of depression and anxiety in patients with cancer included few older adult participants, despite a majority of new cancer diagnoses occurring in patients age 65 and older (DeSantis et al., 2019; Pilleron et al., 2019). While community prevalence rates of depression decrease from middle to old age, rates of subsyndromal distress are high, and epidemiological studies of prevalence likely underrepresent older adults due to variations in depression, anxiety and PTSD presentation (VanItallie, 2005). According to the World Health Organization (WHO), between 2015 and 2050, the proportion of the world’s population over the age of 60 will increase from 12% to 22%. Approximately 15% of all adults aged 60 and over suffer from mental health issues, most commonly anxiety and depression, which affect approximately 7% and 3.8% of the world’s older population, respectively (Mental Health of Older Adults, 2017).
Current medication treatments for psychological distress in older adults include selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), and atypical antidepressants like mirtazapine (Stahl, 2020). However, these treatments take at least 4 to 6 weeks to take effect, have numerous side effects including gastrointestinal upset, weight gain, decreased libido, numerous drug–drug interactions, and have only mild-moderate efficacy for the treatment of psychological distress in this population (Stahl, 2020). Given the promise of psychedelic-assisted therapies to address a number of mental health conditions, and the pressing need for novel psychiatric treatments in older adults, our aim was to examine the existing literature to better understand the safety and tolerability of psychedelic-assisted therapy in this population.
Methods
Eligibility criteria
Types of studies
This systematic review included clinical trials investigating psychedelics (e.g., parallel or crossover randomized controlled trials and open-label studies). Observational studies, case reports, retrospective studies, commentaries, opinions, letters, and editorials were excluded. All included studies were published in English in peer-reviewed journals from Jan 1, 1950, to Sep 14, 2023.
Types of participants
Studies that enrolled participants with primary psychiatric disorders, substance use disorder and distress related to a serious illness were included. No age criteria were applied for study inclusion. As studies of psychedelic-assisted therapy have most often excluded patients with a history of schizophrenia or other psychotic disorders, studies that used psychedelics to treat individuals with such disorders or model psychosis were excluded. In addition, studies on healthy volunteers were excluded as the primary focus of this study was on older clinical populations.
Types of psychedelics
All studies using psilocybin, dimethyltryptamine (DMT), dipropyltryptamine (DPT), lysergic acid diethylamide (LSD), MDMA, ayahuasca, mescaline, and ibogaine were included, with or without a psychotherapy component. Articles reporting the use of cannabis, ketamine or non-serotoninergic substances were excluded.
Types of outcomes
All included studies with accompanying descriptions were collected and organized in Table 1. In addition, measures used to assess primary and secondary outcomes, as well as non-ordinary states of consciousness, were listed and organized by medical conditions in Table 2. Adverse events for older adults (>65 years old) collected in included studies are presented in Table 3.
Description of included studies.
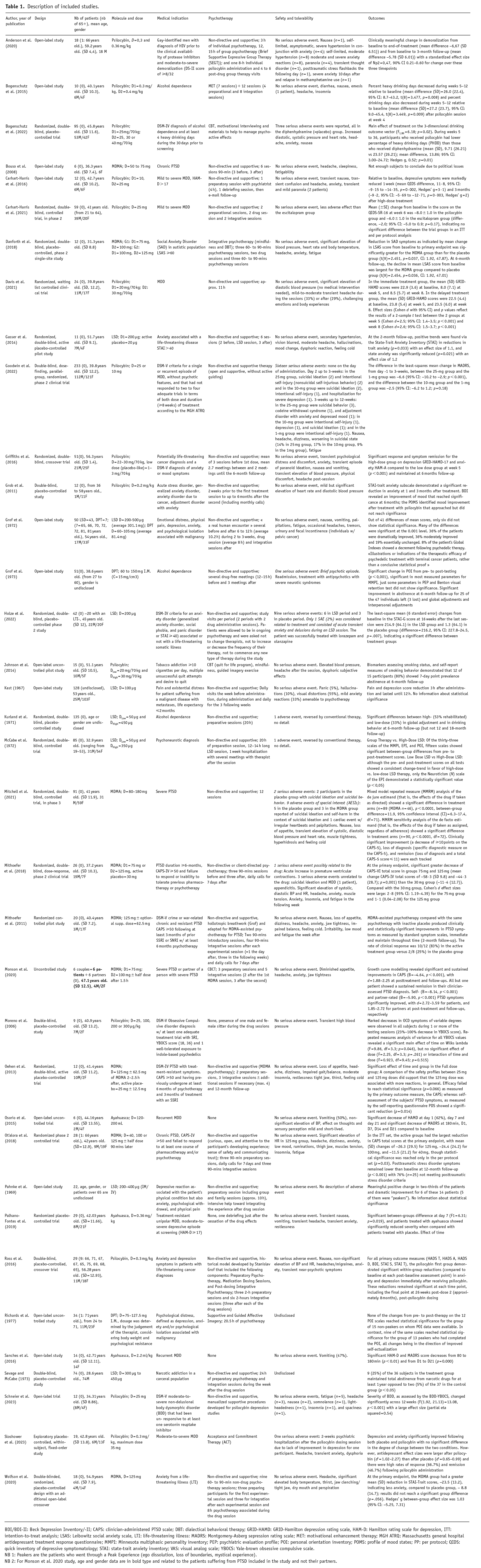
BDI/BDI-II: Beck Depression Inventory/-II; CAPS: clinician-administered PTSD scale; DBT: dialectical behavioral therapy; GRID-HAMD: GRID-Hamilton depression rating scale, HAM-D: Hamilton rating scale for depression, ITT: intention-to-treat analysis; LSAS: Leibowitz social anxiety scale, LTI: life-threatening illness: MADRS: Montgomery-Asberg sepression rating scale; MET: motivational enhancement therapy; MGH ATRQ: Massachusetts general hospital antidepressant treatment response questionnaire; MMPI: Minnesota multiphasic personality inventory; PEP: psychiatric evaluation profile; POI: personal orientation inventory; POMS: profile of mood states; PP: per protocol; QIDS: quick inventory of depressive symptomatology; STAI: state-trait anxiety inventory; VAS: visual analog scale; YBOCS: Yale-brown obsessive compulsive scale.
NB 1: Peakers are the patients who went through a Peak Experience (ego dissolution, loss of boundaries, mystical experience).
NB 2: For Monson et al. 2020 study, age and gender data are in bold type and related to the patients suffering from PTSD included in the study and not their partners.
Primary and secondary outcomes assessing efficacy in specific medical indications.
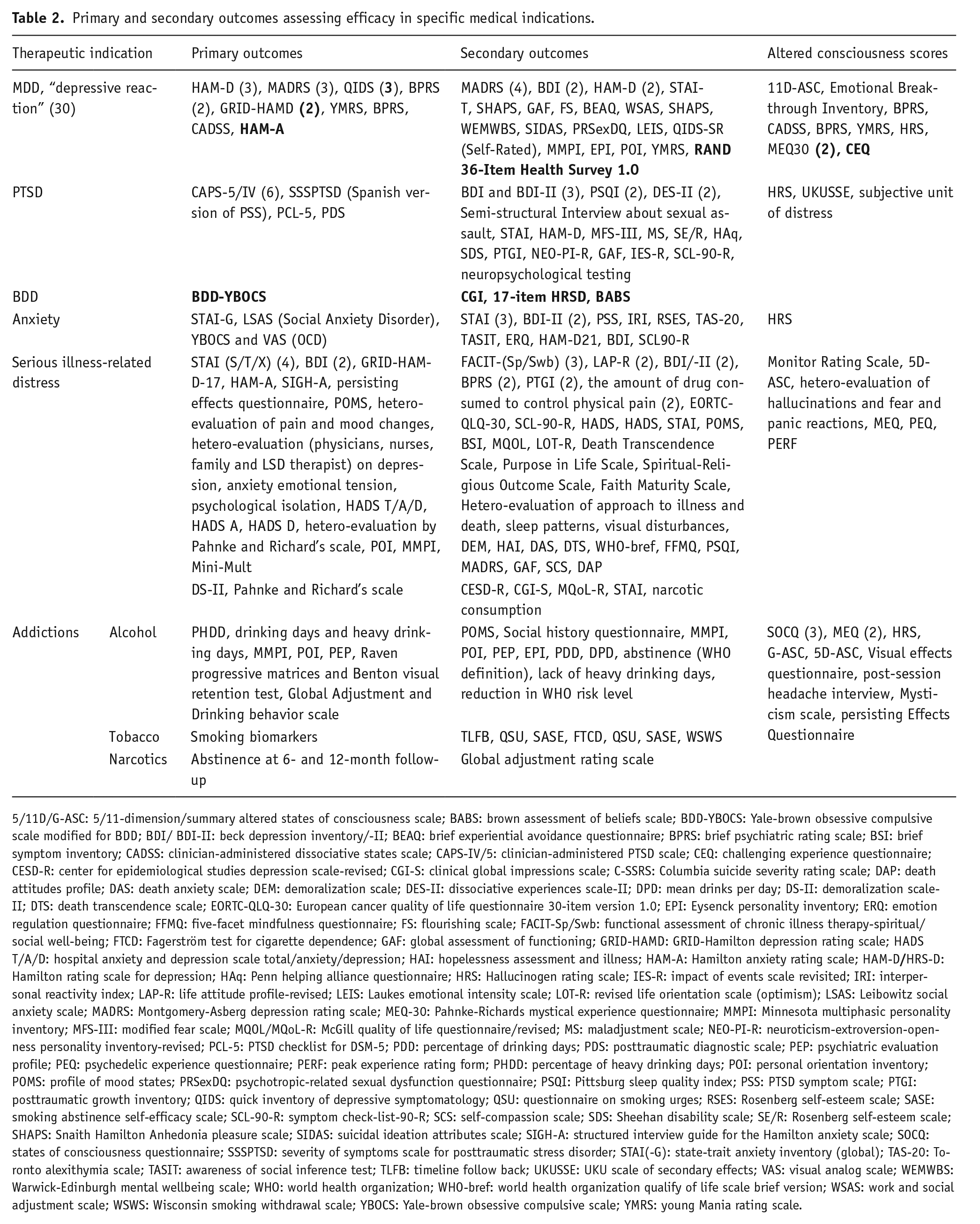
5/11D/G-ASC: 5/11-dimension/summary altered states of consciousness scale; BABS: brown assessment of beliefs scale; BDD-YBOCS: Yale-brown obsessive compulsive scale modified for BDD; BDI/ BDI-II: beck depression inventory/-II; BEAQ: brief experiential avoidance questionnaire; BPRS: brief psychiatric rating scale; BSI: brief symptom inventory; CADSS: clinician-administered dissociative states scale; CAPS-IV/5: clinician-administered PTSD scale; CEQ: challenging experience questionnaire; CESD-R: center for epidemiological studies depression scale-revised; CGI-S: clinical global impressions scale; C-SSRS: Columbia suicide severity rating scale; DAP: death attitudes profile; DAS: death anxiety scale; DEM: demoralization scale; DES-II: dissociative experiences scale-II; DPD: mean drinks per day; DS-II: demoralization scale-II; DTS: death transcendence scale; EORTC-QLQ-30: European cancer quality of life questionnaire 30-item version 1.0; EPI: Eysenck personality inventory; ERQ: emotion regulation questionnaire; FFMQ: five-facet mindfulness questionnaire; FS: flourishing scale; FACIT-Sp/Swb: functional assessment of chronic illness therapy-spiritual/social well-being; FTCD: Fagerström test for cigarette dependence; GAF: global assessment of functioning; GRID-HAMD: GRID-Hamilton depression rating scale; HADS T/A/D: hospital anxiety and depression scale total/anxiety/depression; HAI: hopelessness assessment and illness; HAM-A: Hamilton anxiety rating scale; HAM-D
Adverse events for older patients included in clinical trials.
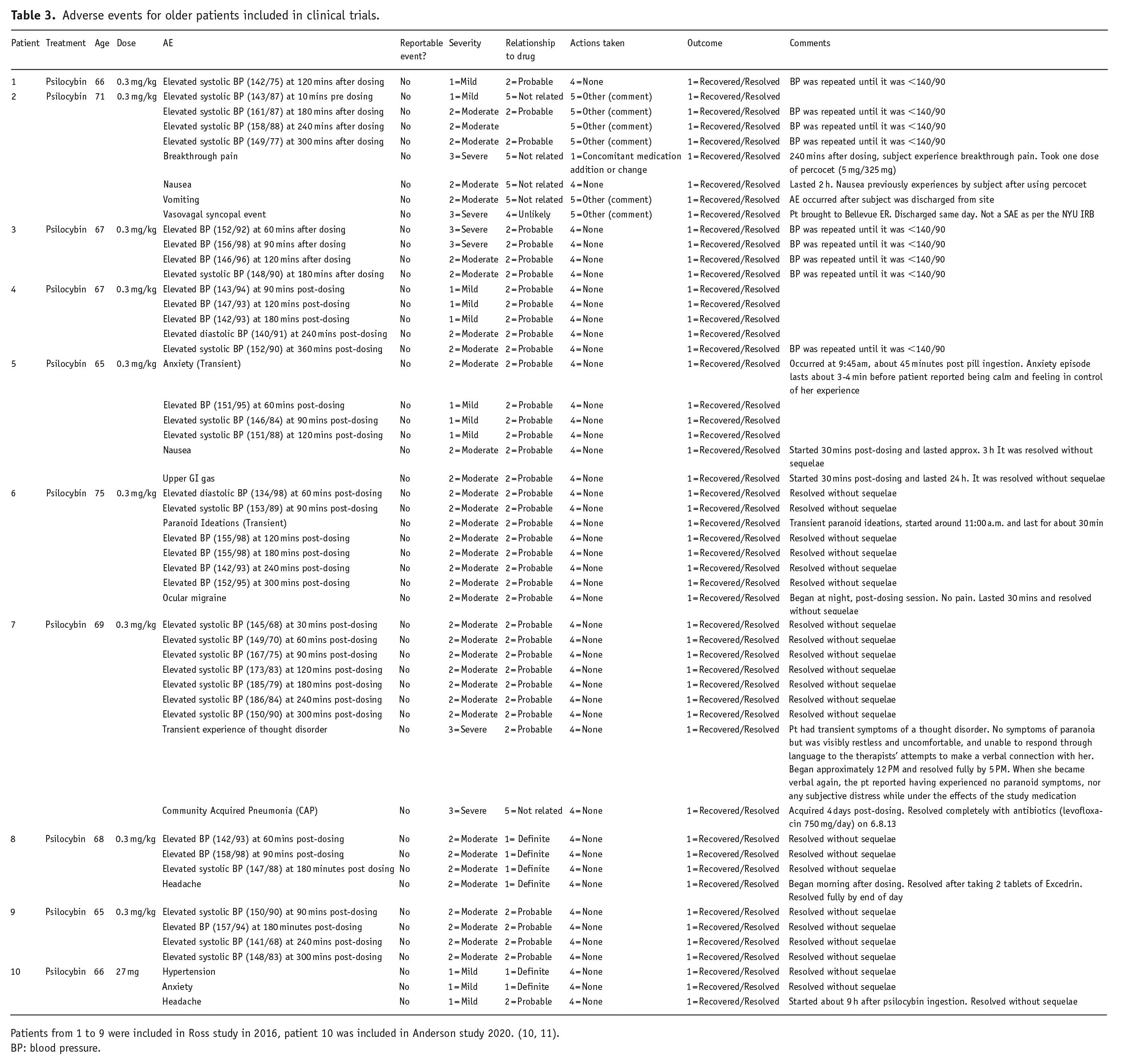
Patients from 1 to 9 were included in Ross study in 2016, patient 10 was included in Anderson study 2020. (10, 11).
BP: blood pressure.
Search strategy
The Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols of 2020 (PRISMA-P) protocol was followed (Page et al., 2021: 2020). A summary of the search strategy is presented in Figure 1 and was applied to each database used: PubMed, Embase and EBSCO. Data extraction was finalized on Sep 14, 2023. The search algorithm is detailed in Supplemental Materials S1.
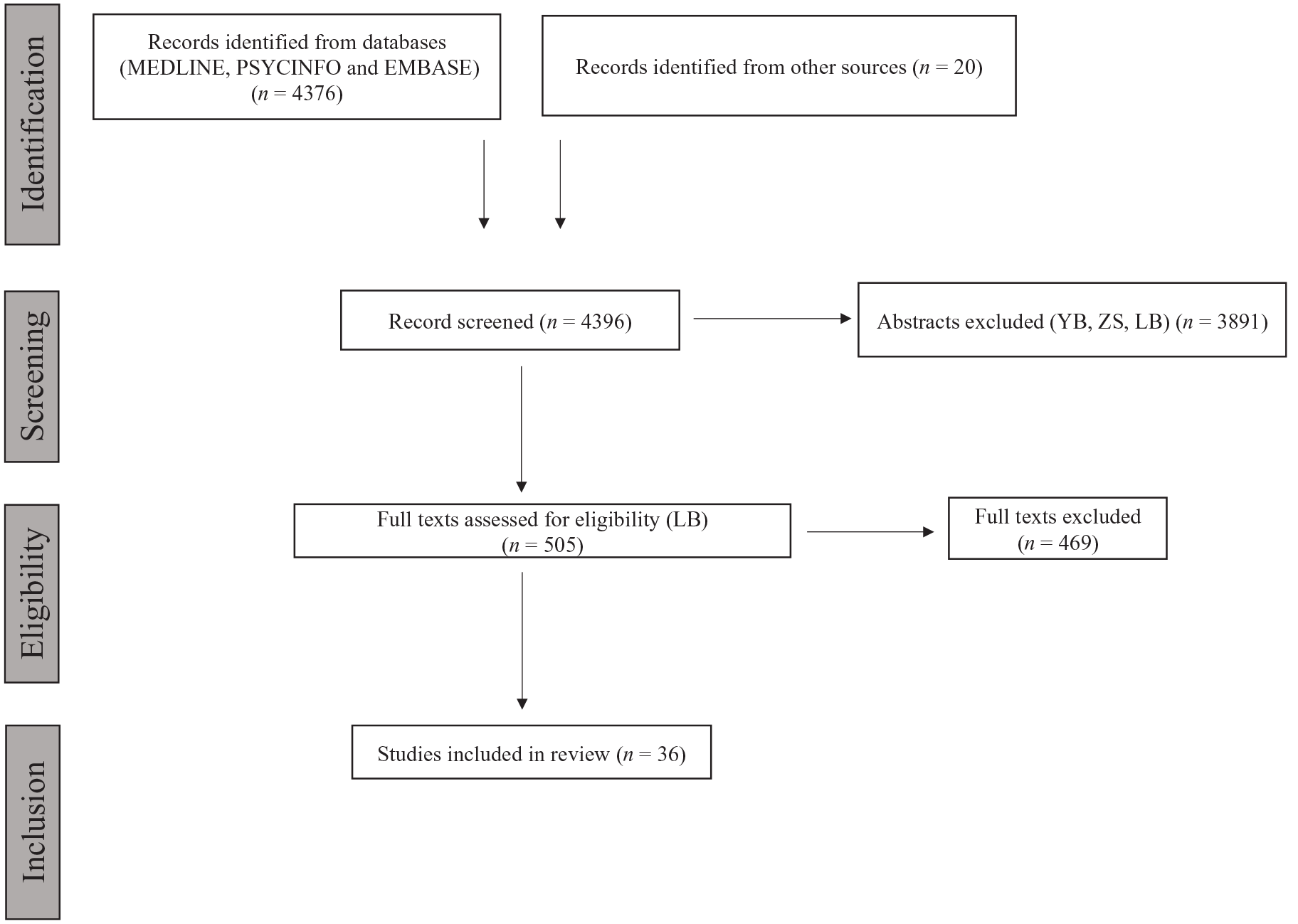
PRISMA flow diagram.
Study records
Selection process
Studies were initially selected based on their titles and abstracts according to the aforementioned eligibility criteria. Articles in which a determination of eligibility could not be made based on title and abstract alone were read in full. All included articles were read in their entirety. The study identification process was carried out independently by two teams of reviewers (YB and ZS, or YB and LB) using Rayyan software. If a team of two disagreed on the initial eligibility of an article, it was further evaluated as a group of three (YB, ZS, LB). The reviewers were not blinded to the publishing journals, authors, or institutions during any stage of the selection process. LB reviewed the bibliography of the selected articles to identify any additional sources.
Data collection and synthesis
The data extraction was carried out manually by LB, reviewed by YB, ZS and AY. The data collection included study design, population (age, gender), psychedelic compounds (dosage, route), therapeutic indications, non-drug therapy and outcome variables. For studies that enrolled older adults, their respective study teams were contacted to gather more information regarding AEs (Anderson et al., 2020; Grof et al., 1972; Ot’alora et al., 2018; Richards et al., 1977; Ross et al., 2016).
Results
Study selection
A total of 4376 articles were identified through the initial query. Through the review of their references, 20 additional articles were identified. After excluding ineligible articles and secondary analysis, 36 articles were identified for inclusion in this systematic review (Anderson et al., 2020; Bogenschutz et al., 2015, 2022; Bouso et al., 2008; Carhart-Harris et al., 2016, 2021; Danforth et al., 2018; Davis et al., 2021; Gasser et al., 2014; Goodwin et al., 2022; Griffiths et al., 2016; Grob et al., 2011; Grof et al., 1973, 1972; Holze et al., 2022; Johnson et al., 2014; Kast, 1967; Kurland et al., 1971; Mccabe et al., 1972; Mitchell et al., 2021; Mithoefer et al., 2011, 2018; Monson et al., 2020; Moreno et al., 2006; Oehen et al., 2013; Osorio et al., 2015; Ot’alora et al., 2018; Pahnke et al., 1969; Palhano-Fontes et al., 2019; Richards et al., 1977; Ross et al., 2016; Sanches et al., 2016; Savage and McCabe, 1973; Schneier et al., 2023; Sloshower et al., 2023; Wolfson et al., 2020). A PRISMA flow diagram of our systematic review is shown in Figure 1. Articles corresponding to secondary analysis of already included articles are presented in Supplemental Materials S2.
Study characteristics
Included articles were published between 1967 and 2023. Studies published prior to 1967 did not meet the inclusion criteria. Of those included, 22 were randomized controlled trials, 14 were open-label studies. The randomized controlled studies were primarily from the last decade.
Psychedelics
Fourteen of the identified studies used psilocybin (Anderson et al., 2020; Bogenschutz et al., 2015, 2022; Carhart-Harris et al., 2016, 2021; Davis et al., 2021; Goodwin et al., 2022; Griffiths et al., 2016; Grob et al., 2011; Johnson et al., 2014; Moreno et al., 2006; Ross et al., 2016; Schneier et al., 2023; Sloshower et al., 2023), nine used MDMA (Bouso et al., 2008; Danforth et al., 2018; Mitchell et al., 2021; Mithoefer et al., 2011, 2018; Monson et al., 2020; Oehen et al., 2013; Ot’alora et al., 2018; Wolfson et al., 2020), eight used LSD (Gasser et al., 2014; Grof et al., 1972; Holze et al., 2022; Kast, 1967; Kurland et al., 1971; Mccabe et al., 1972; Pahnke et al., 1969; Savage and McCabe, 1973), three used ayahuasca (Osorio et al., 2015; Palhano-Fontes et al., 2019; Sanches et al., 2016) and three used DPT (Grof et al., 1972, 1973; Richards et al., 1977). No article using DMT, mescaline or ibogaine met our inclusion criteria.
Indications
Ten of the 36 selected studies investigated treatments for psycho-existential distress related to a serious medical illness. Of those 10 studies, three used LSD (Gasser et al., 2014; Kast, 1967; Pahnke et al., 1969), four used psilocybin (Anderson et al., 2020; Griffiths et al., 2016; Grob et al., 2011; Ross et al., 2016), one used DPT (Richards et al., 1977), one used MDMA (Wolfson et al., 2020) and one studied both LSD and DPT (Grof et al., 1972). Seven studies utilized MDMA to treat PTSD (Bouso et al., 2008; Mitchell et al., 2021; Mithoefer et al., 2011, 2018; Monson et al., 2020; Oehen et al., 2013; Ot’alora et al., 2018). Eight studies investigated treatment-resistant or recurrent MDD, five using psilocybin (Carhart-Harris et al., 2016, 2021; Davis et al., 2021; Goodwin et al., 2022; Sloshower et al., 2023) and three using ayahuasca (Osorio et al., 2015; Palhano-Fontes et al., 2019; Sanches et al., 2016). One study investigated the use of LSD in treating anxiety disorders (Holze et al., 2022) and one study investigated MDMA for social anxiety disorder (SAD) in patients with autism (Danforth et al., 2018). One study investigated the use of LSD for the treatment of personality disorders (characterized as “psychoneurotic”) (Mccabe et al., 1972), one study investigated the use of psilocybin for the treatment of obsessive compulsive disorder (Moreno et al., 2006) and one for body dysmorphic disorder (Schneier et al., 2023).
Finally, regarding indications for addiction, four studies targeted alcohol dependence using psilocybin (Bogenschutz et al., 2015, 2022), DPT (Grof et al., 1973) and LSD (Kurland et al., 1971), respectively. One study investigated the use of psilocybin to treat tobacco addiction (Johnson et al., 2014) and another investigated the use of LSD to treat “narcotic addiction” (Savage and McCabe, 1973). All primary and secondary outcome measures used in the 36 included studies are detailed in Table 2.
Psychotherapy
Among the 36 studies included, only four did not combine psychotherapy with psychedelic administration (Moreno et al., 2006; Osorio et al., 2015; Palhano-Fontes et al., 2019; Sanches et al., 2016). Most studies (about 70%) used supportive, non-directive psychotherapeutic techniques to accompany the administration of psychedelics. Manualized psychotherapy interventions used in psychedelic trials included Cognitive Behavioral Therapy for alcohol and tobacco addiction (CBT) (Bogenschutz et al., 2022; Johnson et al., 2014) and Cognitive Behavioral Conjoint Therapy (CBCT) for PTSD (Monson et al., 2020), Motivational Enhancement Therapy (MET) for alcohol dependance (Bogenschutz et al., 2015), Mindfulness-based Therapy for social anxiety in an autistic population (Danforth et al., 2018), Guided Affective Imagery for psycho-existential distress (Richards et al., 1977) and Acceptance and Commitment Therapy (ACT) for MDD.
Older adults’ prevalence and characteristics
Of the 1400 participants enrolled in the 36 included studies, only 19 were of age 65 or above, yielding a prevalence of less than 1.4%. Of these 19 patients, the average age was 69.6 with a standard deviation of 4.9, with the oldest included individual being 81 years old. Regarding study interventions for those 19 older adults, 10 participants received psilocybin (Anderson et al., 2020; Ross et al., 2016), seven received LSD or DPT (Grof et al., 1972), one MDMA (Ot’alora et al., 2018) and one DPT (Richards et al., 1977).
Safety of psychedelic-assisted therapies in older patients
Psychiatric adverse events
AE data on 10 of the 19 older adults enrolled in psychedelic-assisted psychotherapy trials were received from two study teams (Anderson et al., 2020; Ross et al., 2016). All 10 participants received treatment with psilocybin-assisted therapy in the context of serious illness. Only transient mild-to-moderate psychiatric AEs were reported in four individuals and pertained to anxiety (n = 2), paranoid ideation (n = 1), and thought disorder (n = 1) during the psychedelic experience. All psychiatric AEs were successfully managed by study therapists without the need for pharmacologic interventions or psychiatric hospitalization. No participant developed persistent psychosis or hallucinogen-persisting perceptual disorder.
Medical adverse events
All 10 participants had transient episodes of hypertension (>140/90) likely related to psychedelic administration that resolved without any additional intervention or long-term sequalae. The maximum recorded systolic pressure was 186 mmHg. Among the same 10 participants, two reported headaches that began after discharge from the study site and many hours after the acute effects of the drug subsided. Both were considered to be drug-related but were time-limited (one spontaneously resolved after 30 min, the other with use of over-the-counter non-steroidal anti-inflammatories). Two participants reported GI symptoms, one of which occurred during the dosing session and self-resolved, the other after discharge from the study site. None of the 10 participants had any serious adverse events (SAEs) reported.
Table 3 summarizes the descriptions of adverse event information for these participants.
Discussion
To our knowledge, this study is the first systematic review of older adult participants in psychedelic-assisted therapy research. Despite 1400 patients participating in clinical trials of psychedelic use with methodologies that met criteria for this study, only 19 participants could be identified as aged 65 and older, yielding an estimated prevalence of less than 1.4%. Among these nineteen older adults, eighteen had received psychedelic-assisted therapy (using psilocybin, LSD or DPT) for distress related to cancer or other life-threatening illnesses. Among trials of MDMA-assisted therapy for PTSD, only one older adult was included, contrasting with the elevated prevalence of older adults experiencing trauma across the lifespan and elevated lifetime rates of PTSD in older Veterans (Cook and Simiola, 2017; Kessler et al., 2005; Schnurr et al., 2002).
Psychedelic-assisted therapy outcomes and mechanisms
Psychedelic-assisted therapies have emerged as promising interventions in various psychiatric indications, encompassing depression, anxiety, posttraumatic stress disorder (PTSD), substance use disorders, and the alleviation of distress related to serious illnesses. The positive outcomes observed in these trials underscore their potential as effective treatments for these conditions. Among the 36 studies included in our review, 29 demonstrated statistically significant clinical improvements.
However, it is important to note that five studies did not reach statistical significance (Moreno et al., 2006; Oehen et al., 2013; Richards et al., 1977; Sloshower et al., 2023; Wolfson et al., 2020), and two earlier studies from the 1970s did not report statistical significance (Kast, 1967; Pahnke et al., 1969). These discrepancies highlight the need for further investigation to determine the factors influencing treatment effectiveness, such as variations in study design, patient populations, or therapeutic protocols.
The precise mechanisms underlying psychedelic-assisted therapies remain incompletely understood. Classic psychedelics like psilocybin and LSD act as serotonin 2A receptor (5-HT2A) agonists or partial agonists (Nichols, 2016), while MDMA primarily functions as a potent releaser and/or reuptake inhibitor of presynaptic serotonin, dopamine, and norepinephrine (Vizeli and Liechti, 2017). Psychedelic-assisted therapy combines both psychotherapeutic and pharmacological components, making it a complex intervention to dissect in terms of its mechanisms of action.
These mechanisms can be broadly categorized into three domains: (1) neurocognitive mechanisms—these include the activation of the default mode network and alterations in brain connectivity patterns, which have been implicated in self-referential thought processes and ego dissolution during psychedelic experiences (Gattuso et al., 2022); (2) neurochemical mechanisms—psychedelics induce the release of serotonin and possibly other neurotransmitters, which may contribute to their therapeutic effects. Further research is needed to elucidate the precise role of these neurochemical changes in the therapeutic process (Ko et al., 2022); and (3) psychotherapeutic mechanisms—the psychotherapeutic aspects of psychedelic-assisted therapy are multifaceted and include the development of a trusting therapeutic relationship, a sense of care and safety, and the facilitation of deep emotional processing (Belser et al., 2017). These elements play a vital role in the overall therapeutic experience and need to be specifically studied in older adult populations.
General safety and tolerability
As shown in Table 1, among all adult participants (ages 18–81), adverse events were predominantly of GI, cardiovascular or psychiatric origin, a substantial majority of which were time-limited and resolved with appropriate treatment when indicated. Sixteen psychedelic-related SAEs were reported, including thirteen in the most recent studies. Although this safety profile is not specific to older adults and no SAE was reported in older adults, this provides guidance to potential complications that might occur in administering psychedelic-assisted therapy to older adults.
In the older adult population, transient episodes of hypertension and GI upset were the most significant medical AEs that may contribute to potential medical complications such as stroke, myocardial infarction and dehydration. Thus, caution must be taken with older patients predisposed to cardiovascular complications and more research is needed to better understand the cardiac risk in older adult study populations. Patients with other organ dysfunction affecting blood pressure regulation also fall into this category, specifically those with renal dysfunction. Additionally, it is possible that acute GI upset could lead to low oral intake, predisposing those with inadequate blood pressure regulatory ability to episodes of syncope. This risk could possibly be mitigated by closely monitoring orthostatic blood pressures prior to discharge and ensuring adequate fluid intake before and after the dosing session. Future studies may be warranted investigating the role of antiemetics in mitigating the GI upset caused by serotonergic agonists. In addition, it should be noted that the amphetamine-like pharmacology of MDMA may pose a higher cardiovascular risk than psilocybin, and thus may require more precautions for use in older patients whose physiology may have difficulty handling the excess sympathomimetic stimulation evoked by the release of high quantities of serotonin, norepinephrine and dopamine (Vizeli and Liechti, 2017). In the studies included, cardiac adverse events described were elevation in blood pressure and heart rate and one SAE with acute increase in premature ventricular contractions in a patient who exhibited a premature ventricular contraction at baseline (Mithoefer et al., 2018). The acute risk with MDMA may include myocardial infarction, acute heart failure and stroke. However, the overall incidence appears to be rare as described in observational literature (Ecstasy Induced Acute Systolic Heart Failure and Non-Ischemic Cardiomyopathy in a Young Female: A Rare Case Report and Literature Review—PMC, n.d.). Dose adjustment might be required in older adults with cardiac comorbidity.
Rationale and guidelines for studying PAT in older adults
As individuals age, many face the onset of new medical conditions that negatively affect organ function and contribute to development of frailty. With this decline in organ function comes changes in pharmacokinetics and pharmacodynamics (Poulose and Raju, 2014). Additionally, with increased medical burden comes new medications, which in themselves affect metabolism and may have unknown interactions with novel pharmaceuticals (Wastesson et al., 2018). As such, older adults are appropriately excluded from initial clinical trials investigating the safety, tolerability and preliminary efficacy of novel drugs, as these must be first demonstrated in healthy populations before extending their use to those with comorbidities. The average upper age limit for PAT studies is 64.3 years old (SD 8.52) just under the commonly accepted lower limit of age, 65, for older adults. The current state of psychedelic research appears to indicate that psychedelics may be tolerated in older adults, warranting studies that specifically investigate PAT in older adults where clinical needs are particularly high (Johnston et al., 2022).
There are a few studies currently underway that are paving this path forward. For example, Johns Hopkins University is currently testing the efficacy of PAT for depression in patients with mild cognitive impairment or early-onset Alzheimer’s disease (NCT04123314). Dana Farber Cancer Institute is investigating the role of PAT for demoralization in patients receiving hospice care, with no upper age limit on the inclusion criteria (NCT04950608). The Canadian government has recently allowed citizens with terminal diagnoses to file for a medical exemption to allow the use of psilocybin in a therapeutic setting to assist with the process of death and dying (Rosenblat et al., 2022). As new data emerges utilizing PAT in medically complex patients, many of whom are older adults, the field will soon have more evidence to guide the appropriate usage of PAT in geriatric patients.
As such, there will likely be utility in continued systematic reviews pooling this data to increase the power and significance of conclusions drawn. It may also be useful to determine if the degree of side-effect burden (increase in blood pressure, frequency of side effects reported, etc.) in older patients is significant relative to those below the age of 65, which again, will be possible as more data emerges.
As we come to better understand the major risks of using different psychedelic agents in older adults, there may be an opportunity to investigate the utility of combining these agents with proven pharmacological interventions to ameliorate the risks and improve tolerability (i.e., MDMA and beta-blockers, psilocybin and antiemetics). Lastly, there may be a role of PAT for well-being in the older population, including assisting with the existential transition to old age and the acceptance of loss of functionality that inevitably comes with it (Beaussant and Nigam, 2022; Johnston et al., 2022).
Limitations
Several limitations deserve attention to interpret our findings. First, the very low prevalence of patients at or above the age of 65 included in trials assessing PAT limits the ability to draw significant conclusions regarding the safety of psychedelic use in this population. However, this data can be used to guide attention to potential cardiovascular and GI complications in future trials assessing the tolerability in older adults. Second, the safety data presented in Table 3 was received upon request to the primary research teams having conducted studies with older adult participants, only two of which responded. However, while accessing safety data from studies conducted in the 1960s and 1970s might have strengthened our analysis, it is unlikely that this would have significantly changed its conclusions as reports from these studies have underlined the overall good tolerance of psychedelic-assisted therapy interventions in their respective populations. Third, the medical comorbidities of the older adults included in this study are unknown as they were often not listed in the available dataset.
Conclusion
In this systematic review, we demonstrate that patients at or over the age of 65 account for less than 1.4 % of those included in clinical studies to this day. Within the 36 studies that met inclusion criteria for this study, only 19 of the 1400 participants were age 65 or older. All medical adverse events reported in this subset were cardiovascular or GI in origin and resolved in a time-limited fashion (within 48 h of drug exposure) and without long-term sequalae. No psychedelic-related SAEs were reported in older adults, no patients required medical or psychiatric hospitalization, no one developed HPPD or persistent psychosis, and all AEs were effectively managed by study therapists. Although existing data in older adults is limited, it does provide preliminary evidence for the safety and tolerability of PAT in older patients, and as such, should be more rigorously studied in future clinical trials.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231215420 – Supplemental material for Older adults in psychedelic-assisted therapy trials: A systematic review
Supplemental material, sj-docx-1-jop-10.1177_02698811231215420 for Older adults in psychedelic-assisted therapy trials: A systematic review by Lisa Bouchet, Zachary Sager, Antoine Yrondi, Kabir B Nigam, Brian T Anderson, Stephen Ross, Petros D Petridis and Yvan Beaussant in Journal of Psychopharmacology
Footnotes
Acknowledgements
The authors thank Jason Smith for his help in the search strategy development and article extraction.
Authors contributions
YB and ZS designed the study, wrote the protocol. The study identification process was carried out independently by two reviewers (YB and ZS, or YB and LB). In case of inconsistency, articles were evaluated for inclusion as a team (YB, ZS, LB). LB assessed data extraction and wrote the initial manuscript draft. The data extraction was reviewed by YB, ZS and AY. BA, SR and PP contributed with safety data from their respective study. All the authors contributed data analysis and manuscript writing. Underlying research materials can be accessed by contacting the authors by email.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the past three years, BA has received consulting fees from Journey Colab. Dr. Ross reported receiving grant support for clinical research from the National Institute on Drug Abuse (NIDA), National Cancer Institute (NCI), Heffter Research Institute, Usona Institute, Council on Spiritual Practices (CSP), Multidisciplinary Association of Psychedelic Studies (MAPS), and Reset Pharmaceuticals. Dr. Ross is Director of the NYU Langone Center for Psychedelic Medicine Research and Training Program, funded by MindMed. Dr. Ross is listed as a co-inventor in two provisional patent applications (N420838US and N419987US) related to the use of psilocybin to treat psychiatric and existential distress in cancer, filed by New York University Grossman School of Medicine and licensed by Reset Pharmaceuticals. Dr. Ross has waived all rights and has no prospect of financial gain in relation to these patent applications. LB, ZS, AY, KN, PP and YB.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
