Abstract
Background:
Drugs that act on the central nervous system (CNS) and have sedative effects can lead to abuse in humans. New CNS-active drugs often require evaluation of their abuse potential in dedicated animal models before marketing approval. Daridorexant is a new dual orexin receptor antagonist (DORA) with sleep-promoting properties in animals and humans. It was approved in 2022 in the United States and Europe for the treatment of insomnia disorder.
Aims:
Nonclinical evaluation of abuse potential of daridorexant using three specific rat models assessing reinforcement, interoception, and withdrawal.
Methods:
Reinforcing effects of daridorexant were assessed in an operant rat model of intravenous drug self-administration. Similarity of interoceptive effects to those of the commonly used sleep medication zolpidem was tested in an operant drug discrimination task. Withdrawal signs indicative of physical dependence were evaluated upon sudden termination of chronic daridorexant treatment. Rat experiments were conducted at a dose range resulting in daridorexant plasma concentrations equaling or exceeding those achieved at the clinically recommended dose of 50 mg in humans.
Results:
Daridorexant had no reinforcing effects, was dissimilar to zolpidem in the drug discrimination task, and did not induce any withdrawal-related signs upon treatment discontinuation that would be indicative of physical dependence.
Outcomes:
Daridorexant showed no signs of abuse or dependence potential in rats. Our data indicate that daridorexant, like other DORAs, has a low potential for abuse in humans.
Keywords
Introduction
Many central nervous system (CNS)-active drugs that are widely used in clinical practice have the potential to be abused by either patients or healthy individuals who may take them for recreational rather than medical purposes. This poses a major public health issue. History has shown that drugs with either stimulating effects (e.g., amphetamines) or sedating effects (e.g., opioids, gamma-aminobutyric acid type A (GABA(A)) receptor agonists or positive allosteric modulators, including zolpidem) often bear abuse potential. Therefore, regulatory agencies require novel drug candidates with such action to be evaluated for abuse potential as part of a new drug or marketing authorization application (EMA Guideline, 2006; FDA Guidance, 2017). Based on their profile, the drugs are categorized according to their likelihood for abuse such that certain measures of control be exerted by central agencies (e.g., the US Drug Enforcement Administration (DEA)) on drug availability and the frequency of prescriptions by physicians.
Daridorexant is a new sleep medication that was approved in the United States in 2022 (FDA Approval Daridorexant, 2022). It is the third dual orexin receptor antagonist (DORA) entering the US market, following approval of suvorexant (Merck; FDA Approval Suvorexant, 2014) and lemborexant (Eisai; FDA Approval Lemborexant, 2019). Daridorexant was designed with an optimized pharmacokinetic (PK) and pharmacodynamic profile for an expected effect duration lasting through the night (Treiber et al., 2017) and is the first DORA to be also available in Europe (EMA Approval Daridorexant, 2022). As a CNS-active and sleep-promoting drug (Dauvilliers et al., 2020) daridorexant qualified for abuse potential assessment in animal models before approval.
DORAs act on the brain by blocking the orexin receptors (OXRs) type 1 and type 2 that are located in wake promoting centers (Roch et al., 2021; Sakurai, 2007). Hereby, DORAs interfere with the driving force of arousal-enhancing orexin neuropeptides, which activate these regions. As a consequence, DORAs allow sleep-promoting areas of the brain to take over from wake-promoting areas as the intrinsic sleep pressure builds up and demands rest (for instance at the end of the day) (de Lecea, 2021). By such specific mechanism of action DORAs enable natural sleep to occur.
In addition to their location in wake-controlling regions of the brain, OXRs are found in areas of the mesolimbic dopamine system (Cole et al., 2020; Kallo et al., 2022; Kawashima et al., 2022; Schmeichel et al., 2017) where they help to orchestrate the amount of arousal necessary for successfully engaging in highly motivational and rewarding tasks (de Lecea, 2021). For example, in rats, OXR signaling mediates natural or drug-related reward seeking under large effort requirements (Schmeichel et al., 2018) and is required to make cognitive associations of the environmental context to reward reception (Harris et al., 2005). Therefore, it appears unlikely that DORAs would exert reinforcing effects. Indeed, the DORA almorexant did not have any conditioned rewarding effects in a conditioned place preference paradigm in rats (Steiner et al., 2013), and suvorexant helped patients with opioid use disorder reduce their insomnia, withdrawal, and craving symptoms during opioid withdrawal without eliciting any drug-liking (Huhn et al., 2022).
To fulfill regulatory requirements (EMA Guideline, 2006; FDA Guidance, 2017), we investigated the abuse potential of daridorexant in three well-validated animal paradigms that can generate behavioral signs indicative of drug effects in humans, namely drug self-administration, drug discrimination, and physical dependence.
Methods
General procedures
Experiments were conducted according to Good Laboratory Practices under authorization of the Aptuit Committee on Animal Research and Ethics and the Italian Ministry of Health. Protocols were approved by the FDA and the Controlled Substance Staff.
Animals
For all experiments, female, non-ovariectomized Sprague Dawley rats (Crl:CD(SD); Charles River, Calco (LC), Italy) were used. Prior toxicity studies during the development of daridorexant revealed no relevant sex differences at comparable systemic exposure, justifying the use of one sex only. This choice was also made in respect of efforts to reduce experimental animals (i.e., 3Rs). Females were preferred to males due to their lower body weight (BW) and behavioral agility. Adult rats were singly housed under standard conditions with
Selected dose ranges of daridorexant according to regulatory guidelines
Based on Idorsia-internal nonclinical pharmacology and toxicology studies, doses of daridorexant were selected such as to lead to pharmacologically active rat plasma and brain concentrations (Treiber et al., 2017), and to cover and exceed the human total plasma exposure at the recommended clinical dose of 50 mg (
Pharmacokinetics
Blood was drawn from the caudal vein after exposing rats for 10 min to warm air (39 ± 1°C). Samples were collected in K2-EDTA tubes and centrifuged for 10 min at 2000
Statistics
Drug self-administration
The goal of the drug self-administration study was to evaluate whether daridorexant, when given intravenously, would exert reinforcing effects and sustain a learned operant response (i.e., lever pressing) in rats trained to obtain cocaine. The stimulant cocaine, listed as a Schedule II drug in the United States, was used both as the training drug and positive reference.
Sixty-eight rats that were implanted with femoral vein catheters (see Supplemental Methods) entered the study. They were food-restricted to maintain their BW in the range of 250 ± 15 g throughout the study.
Self-administration sessions were performed with each rat individually placed in a regular operant chamber, equipped with two levers and operable lights (MedAssociates, Fairfax, VT, USA), and enclosed inside a sound- and light-attenuating box. Individual infusion pumps (Model PHM-100VS, Med Associates) were connected to the implanted catheters via infusion tubing and liquid swivels mounted on the top of each chamber. Infusion volumes were set at nominal 80 μL over 4.3 s. Infusion volumes and the maximum number of infusions achievable remained constant throughout the study. Catheter patency was lost and could not be regained for 27 rats; these rats were removed from the study.
Rats underwent daily experimental sessions during which they were first trained to self-administer cocaine under a fixed ratio (FR) schedule of reinforcement. Upon the start of each operant session, the house light was illuminated and both levers were exposed (one active, one inactive). If the active lever was pressed once (FR1), the infusion pump delivered an infusion of cocaine, and then the house light was switched off for 20 s (time-out (TO)). Cocaine hydrochloride (MacFarlan Smith, UK) was freshly dissolved in an aqueous 0.9% w/v NaCl solution at a concentration of 2.5 mg (free base)/mL. The nominal cocaine dose of 0.80 mg/kg/infusion was based on prior experiments at Aptuit and on a rat BW of 250 g. Any presses during TO had no programmed consequences. Active (left or right) levers were randomized across rats. Each session ended after 2 h, or when rats had achieved 25 reinforcements. Once rats achieved stable responding under an FR1 schedule, the FR was gradually increased to FR2, FR5, and finally FR10. Training was continued until stable responding under FR10 was achieved (i.e., when the percent coefficient of variation of the average number of infusions during the last three sessions was ⩽10).
Next, rats underwent an extinction phase, in which cocaine was substituted with saline. Each animal received 3–14 extinction sessions (one session/day; total number of sessions depended on performance) until baseline levels of responding were reduced to at least 50%.
Rats were then re-exposed to cocaine to ascertain that rats could again achieve stable cocaine self-administration behavior under FR10. Three rats did not achieve stable responding on time and had to be removed.
During the following substitution phase, based on their levels of responding, rats were evenly divided in five groups, in which cocaine was either continued or substituted with one of three dose levels of daridorexant or its vehicle. Daridorexant hydrochloride (Idorsia) was dissolved in its vehicle (hydroxypropyl-beta-cyclodextrin, HP-β-CD, 30% [w/v]/buffer pH 4 in purified MilliQ water) up to final concentrations of 0.313, 0.938, and 3.125 mg (free base)/mL. The selection of nominal dose levels of daridorexant, 0.1, 0.3, and 1 mg/kg/iv infusion was based on an intravenous PK study (see Supplemental Methods). Substitution was carried out during six consecutive sessions (1 session/day) in 38 rats (beforehand, 30 rats had to be removed from the study for several reasons, see above). To increase the individual group sizes to nine, seven rats from the positive cocaine treatment control group were redistributed among the other groups after the sixth cocaine substitution session, and underwent a consecutive, second substitution phase. Plasma levels of daridorexant were evaluated approximately 15 min after the end of the first (Day 1) and last (Day 6) substitution session.
Hereafter, all rats were switched again to self-administration of cocaine under an FR10 schedule for at least three additional sessions.
Statistical analysis
Inactive and active lever presses and number of infusions were recorded. Within each treatment group, each day of substitution was compared with the last day of the cocaine self-administration baseline within the same treatment group, using separate one-way analyses of variances (ANOVAs; repeated measure: Time) followed by Dunnett’s post-hoc test. Additionally, each daridorexant treatment group was compared with the vehicle and cocaine treatment groups for each substitution day using separate one-way ANOVAs followed by Dunnett’s post-hoc test.During the second period of cocaine re-exposure, the first day of stable cocaine self-administration after the substitution phase was compared with the last day of the cocaine self-administration-baseline prior to the substitution phase using independent, paired Student’s
Drug discrimination
The goal of the drug discrimination study was to investigate whether daridorexant would elicit similar interoceptive effects (i.e., bodily sensations) as zolpidem. For that purpose, rats were trained to discriminate “drug (zolpidem)” from a “no drug (vehicle)” condition in a two-lever, food-reinforced operant task. Lorazepam, a sedative/hypnotic benzodiazepine (FDA Prescribing Information for Ativan), and suvorexant (both schedule IV) served as comparators in the generalization test.
The experiment was conducted in operant chambers equipped with two retractable levers, cue lights, a house light, and a dispenser that delivered 45-mg sugar pellets (Bio-Serv, Flemington, NJ, USA). Chambers were controlled by MedPC® IV software (version 4.39, MedAssociates, Fairfax, VT, USA).
Eleven rats were successfully trained, up to the defined criterion, to discriminate zolpidem (zolpidem hemitartrate; Toronto Research Chemicals, ON, Canada) at 3 mg/kg (free base; dose based on (Born et al., 2017)) from its vehicle (0.5% methylcellulose in water), both dosed orally 15 min before each session (see Supplemental Methods for details).
During the following generalization phase, we investigated whether different doses of zolpidem (0.3, 0.56, 1, 1.8, and 3 mg (free base)/kg; tested first, as positive control) and daridorexant (15, 30, and 60 mg (free base)/kg; tested second) would cause stimulus generalization to zolpidem 3 mg/kg. The generalization test sessions were carried out with a design identical to that of the training sessions (Supplemental Methods) with the difference that both levers were reinforced to prevent bias. After daridorexant, we tested lorazepam (0.3, 1, and 3 mg/kg; Cambrex Profarmaco Milano, Italy), followed by suvorexant (10, 30, and 100 mg/kg; PharmaBlock Sciences (Nanjing), Inc., China). In between switching from one drug to the next, at least five maintenance training sessions and PK assessments (see below) were performed.
All drugs or their respective vehicles (0.5% methylcellulose in water, for zolpidem hemitartrate, daridorexant hydrochloride, and suvorexant; 40% propylene glycol, 10% polyethylene glycol 400, and 50% 0.9% saline, for lorazepam) were administered orally at 5 mL/kg in a cross-over design 15 min (zolpidem and daridorexant), 1 h (lorazepam), or 2 h (suvorexant) prior to the start of the independent generalization test sessions. Rats treated with lorazepam or suvorexant received a second oral administration of water 15 min before the beginning of the generalization sessions. Pre-treatment times were chosen based on the
Generalization sessions were performed in intervals of at least 72 h and were interspersed with at least two consecutive maintenance training sessions (i.e., either with vehicle or zolpidem 3 mg/kg, in randomized order), during which the performance criterion (Supplemental Methods) had to be met. Eleven rats were used for generalization testing with zolpidem, daridorexant, and lorazepam; nine rats for suvorexant (two rats were removed due to health or performance issues).
A single blood sample was collected from each rat (not those of the zolpidem-treated group) approx. 15 min after termination of the generalization sessions. Additional PK/time profiles were assessed after each respective drug discrimination dose–response curve.
Statistical analysis
The zolpidem-associated lever presses up to the first reinforcement (FRF), and as percentage of the total lever presses during the whole session were qualitatively assessed. A treatment was considered to produce full, partial, or no generalization when there was ⩾80%, >20 and <80%, or ⩽20% selection of the drug-associated lever, respectively (Solinas et al., 2006).
The response rate (number of responses during the entire session divided by session time minus the sum of TO) was analyzed using ANOVA for repeated measures followed by post-hoc Dunnett’s test.
Physical dependence
The objective of the physical dependence study was to assess whether daridorexant would cause physiological and/or neurobehavioral signs of withdrawal following abrupt discontinuation of chronic treatment (see Figure 3(a) for a schematic). Forty rats were evenly divided in four groups and treated daily for 28 days with either daridorexant at 20 or 200 mg (free base)/kg/day, its vehicle (0.5% methylcellulose in water), or the positive control chlordiazepoxide (CDP; dissolved in water) by oral gavage at 5 mL/kg. The CDP group received an additional CDP treatment approximately 8 h following the morning dose because of its short half-life in rats (~4 h) (Froger-Colleaux et al., 2011; Koechlin et al., 1965). CDP was up-titrated (to overcome developing tolerance with repeated dosing) from 10 to 200 mg/kg/day in daily increments of 10 mg/kg/day, reaching on Day 20 the dose of 200 mg/kg/day, which was maintained up to Day 28. This treatment scheme was based on previous studies (Goudie et al., 1993; Koechlin et al., 1965; Ryan and Boisse, 1983; Takada et al., 1989) and experience at Aptuit. The classical benzodiazepine CDP was selected as a reference because of its sedative properties and dependence potential (schedule IV).
Starting from Day 29, drug treatment was discontinued. However, to maintain the daily handling routine, all animals continued to receive daily vehicle treatments. The potential occurrence of physiological and behavioral withdrawal signs was monitored until Day 42.
From Days 1 to 42, BW and food consumption (FC; food intake over 24 h, normalized by BW) was assessed daily before the morning dosing. Body temperature (BT) was measured via implanted subcutaneous transponders (IPTT-300, BioMedic Data Systems, Inc., Waterford, WI, USA) daily at 1 h after morning dosing. General clinical observations were performed daily at 1 h after morning dosing (and for the CDP group, also after the afternoon dosing from Days 11 to 28; for welfare monitoring). Spontaneous locomotor activity (SLMA) and detailed neurobehavioral observations (functional observation battery (FOB)) were recorded at 1 h after the morning dosing (at the expected
For PK assessments, satellite animals were treated with 20 and 200 mg/kg/day of daridorexant (
Statistical analysis
Potential drug effects on BW, FC, BT, and SLMA parameters were assessed at each observation time by comparing each treatment group to vehicle or to the last day of treatment (for the withdrawal phase). Differences were evaluated using ANOVA, followed by Fisher’s Least Significance Difference (LSD) post-hoc test. Potential drug effects on neurobehavioral signs were evaluated based on the group median values for those signs that were scored on a continuous scale using a non-parametric Mann–Whitney
Results
Drug self-administration study
The responding on the active lever during the three cocaine self-administration baseline sessions prior to substitution was comparable among the five treatment groups (Figure 1(a)). Rats that continued to receive cocaine during substitution maintained stable active lever responding. All four groups in which cocaine was substituted with vehicle or daridorexant had significantly fewer active lever presses during each of the six substitution sessions than during the last baseline self-administration session (
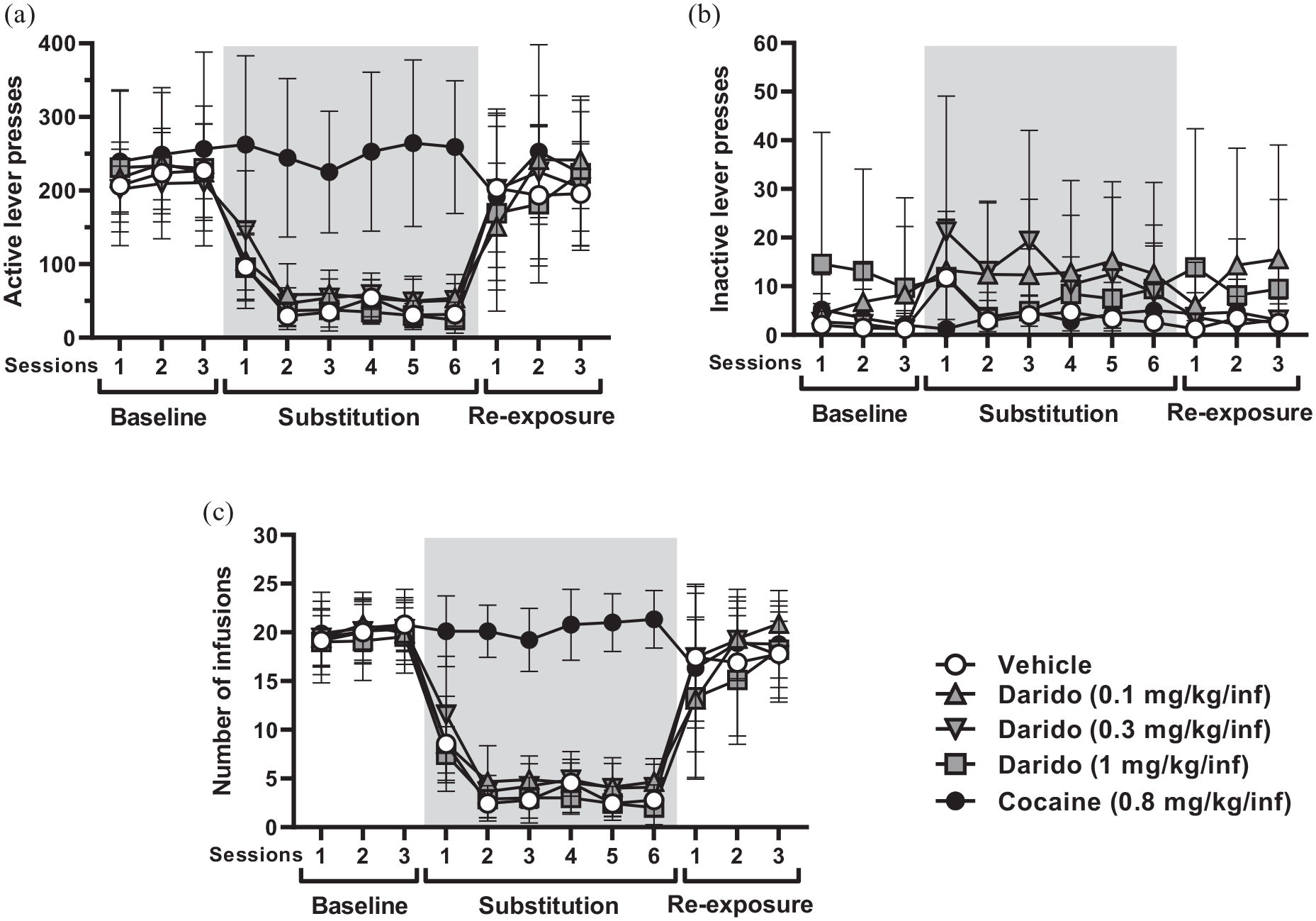
Active (a) and inactive (b) lever presses, and number of intravenous infusions (c) obtained during the baseline (sessions 1–3), substitution (sessions 1–6), and re-exposure (sessions 1–3) phase of the operant drug self-administration paradigm.
When rats were re-exposed to cocaine, active lever responding was reinstated in the daridorexant and vehicle groups to previously established cocaine-reinforced levels (Figure 1(a)).
Plasma concentrations of daridorexant at the end of the first and last substitution sessions ranged from 18 to 2660 ng/mL, depending on the self-administered dose and substitution session (Table 1). As drug infusions decreased by approximately 50% or more from the first to the sixth substitution session, daridorexant plasma concentrations also decreased.
Number of intravenous infusions and associated plasma concentrations of daridorexant in rats at the end of the first and last (i.e., sixth) substitution session of the drug self-administration paradigm.
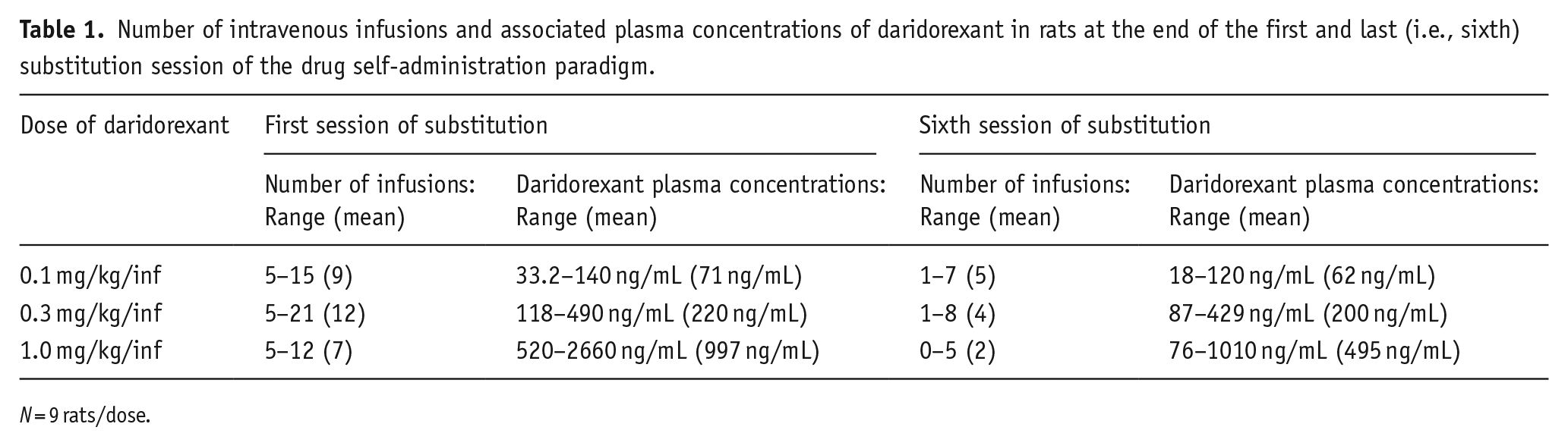
All vehicle-treated samples, bar one, showed daridorexant concentrations below the lower limit of quantification. The reason for this isolated case measured during the first substitution (41.1 ng/mL, and reconfirmed in a duplicate analysis at 46.1 ng/mL) remains unknown (e.g., cross-contamination during sample work-up for analysis), but was not expected to affect the interpretation of the overall data.
Drug discrimination study
All assessed drugs were well tolerated. Zolpidem generalization testing resulted in a sigmoidal dose–response curve (Figure 2(a)). Full generalization (group means of 99.6% and 97.8% of responding on the zolpidem-associated lever in the whole session) was achieved with zolpidem doses of 3 and 1.8 mg/kg, partial generalization (78.1 and 37.3% of responding) with 1 and 0.56 mg/kg, and no generalization (4.1% of responding) with 0.3 mg/kg. After vehicle administration, all rats responded on the vehicle-associated lever. This dose–response profile was mirrored when only the zolpidem-associated lever presses up to the FRF were considered (Supplemental Figure 1). The response rate was first slightly increased and then decreased with increasing doses of zolpidem (ANOVA:
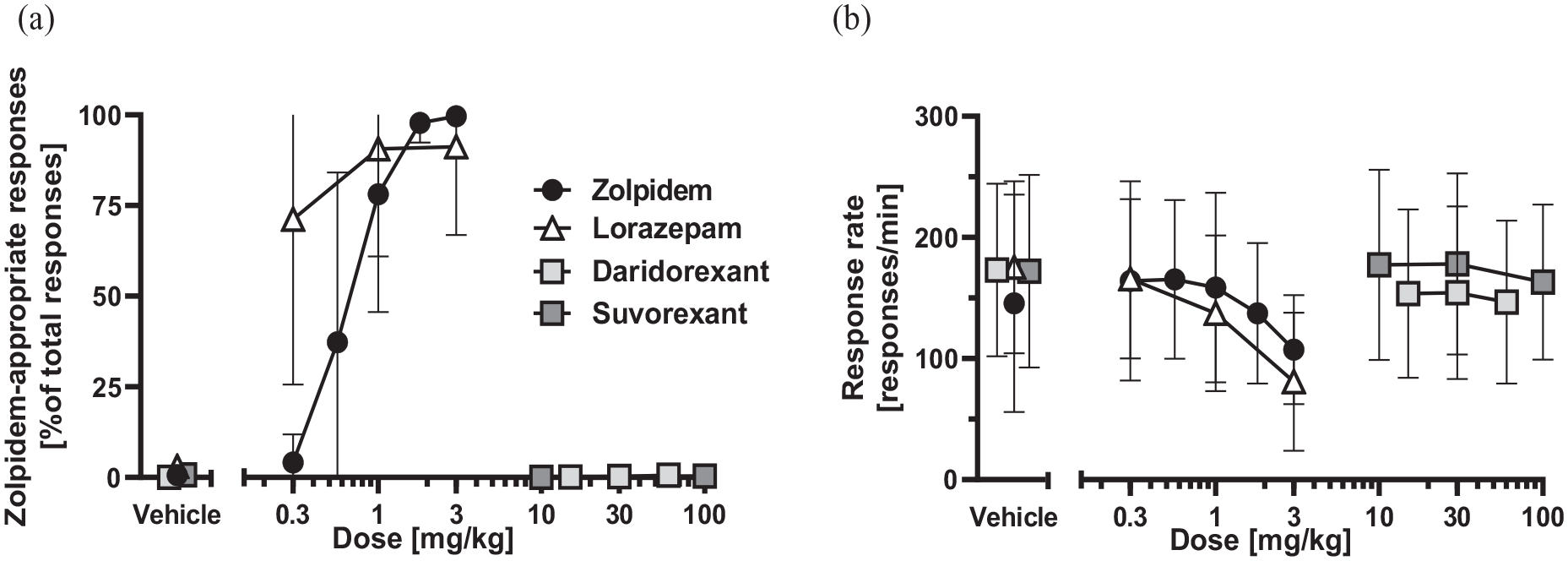
Percentage of responding on the zolpidem-appropriate lever (a) and response rate (b) during the entire test session following oral treatment with zolpidem, lorazepam, daridorexant, and suvorexant. Data points depicting the vehicle groups are slightly nudged for better visibility. Shown are group means ± SD (standard deviation).
Daridorexant did not cause any generalization to zolpidem. The group mean value for responding on the zolpidem-associated lever in the entire session was ⩽0.6% across all daridorexant dose levels (Figure 2(a)), which was similar to vehicle (0.1%). The daridorexant-treated rats also obtained all their first rewards by pressing the previously vehicle-associated lever with an FRF of 10 (
Plasma exposure to daridorexant after acute oral dosing of rats enrolled in the drug discrimination study.
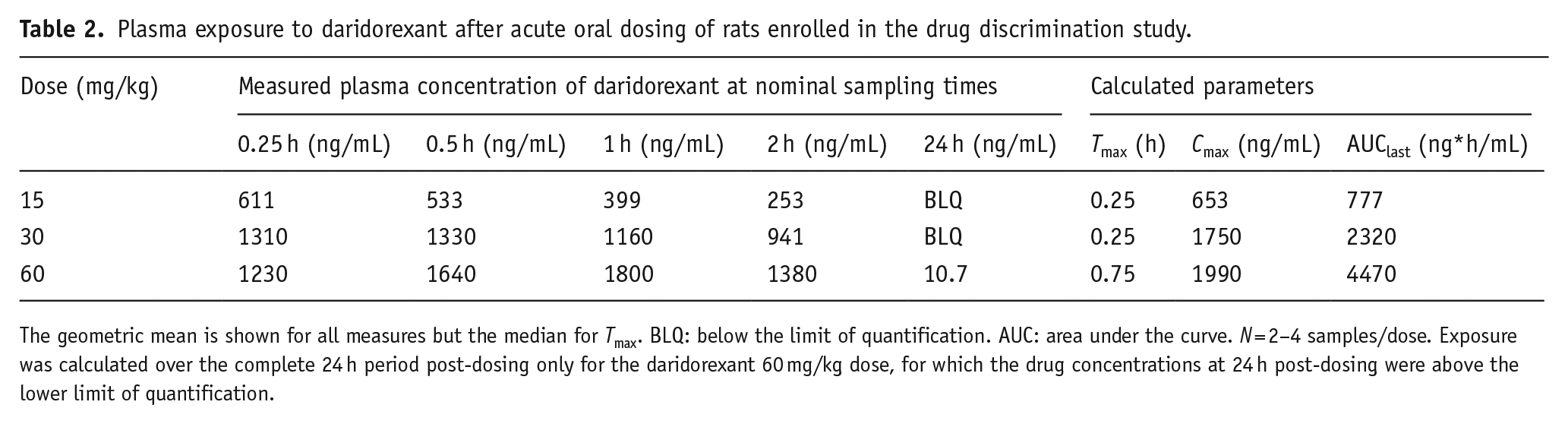
The geometric mean is shown for all measures but the median for
Daridorexant plasma concentrations during drug-discrimination. Blood samples were collected at the end of the daridorexant generalization test sessions at approximately 60 min after oral treatment.
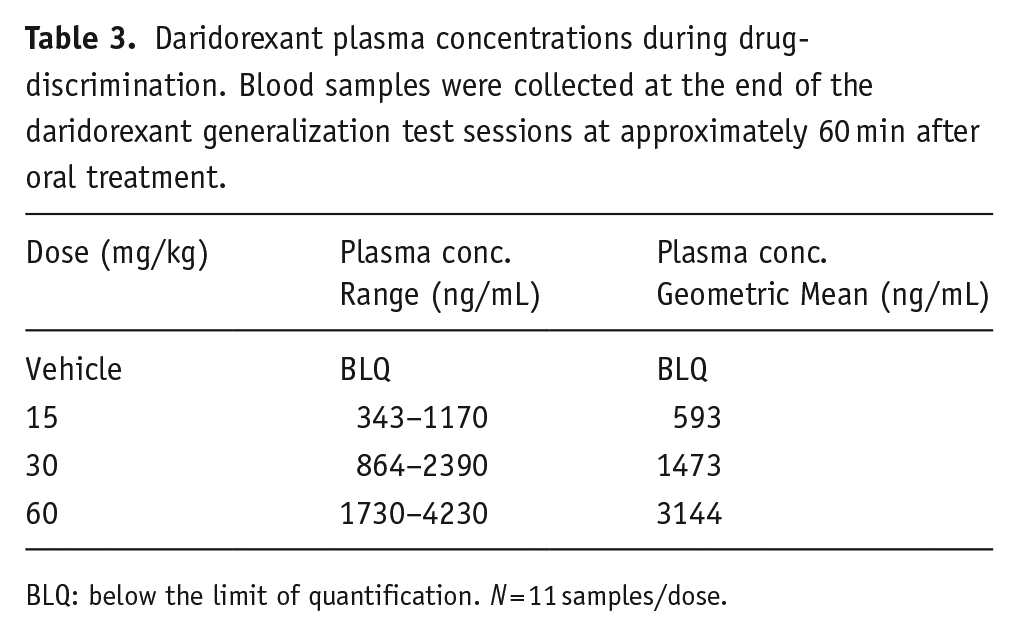
BLQ: below the limit of quantification.
Lorazepam caused partial (at 0.3 mg/kg; Figure 2(a)) or full (at 1 and 3 mg/kg) generalization to zolpidem, and the first rewards within the test session were also mostly obtained on the zolpidem-associated lever at an FRF of 10 (Supplemental Figure 1). Lorazepam dose-dependently reduced the response rate (ANOVA:
Suvorexant-treated rats responded on average ⩽0.4% on the previously zolpidem-associated lever (Figure 2(a)), which was comparable to vehicle (⩽0.7%). Thus, suvorexant did not cause any generalization to zolpidem. Consistently, suvorexant-treated rats obtained almost all their first rewards on the vehicle-associated lever with a FRF of 10 (in 30/33occasions (Supplemental Figure 1)). Suvorexant did not affect the response rate (ANOVA,
Physical dependence study
Daridorexant treatment had no effect on BW (Figure 3(b)). The slightly lower BW of the group of rats treated with 200 mg/kg/day of daridorexant was already apparent before the first dosing (Day-1) and, therefore, judged non-treatment related. During the withdrawal period, the BW of rats treated formerly with daridorexant remained overall similar to that of the vehicle group. Rats treated with daridorexant 200 mg/kg/day displayed a slightly reduced rate of BW gain due to mild, temporary BW reductions on Days 31 and 32 compared with Day 28 (
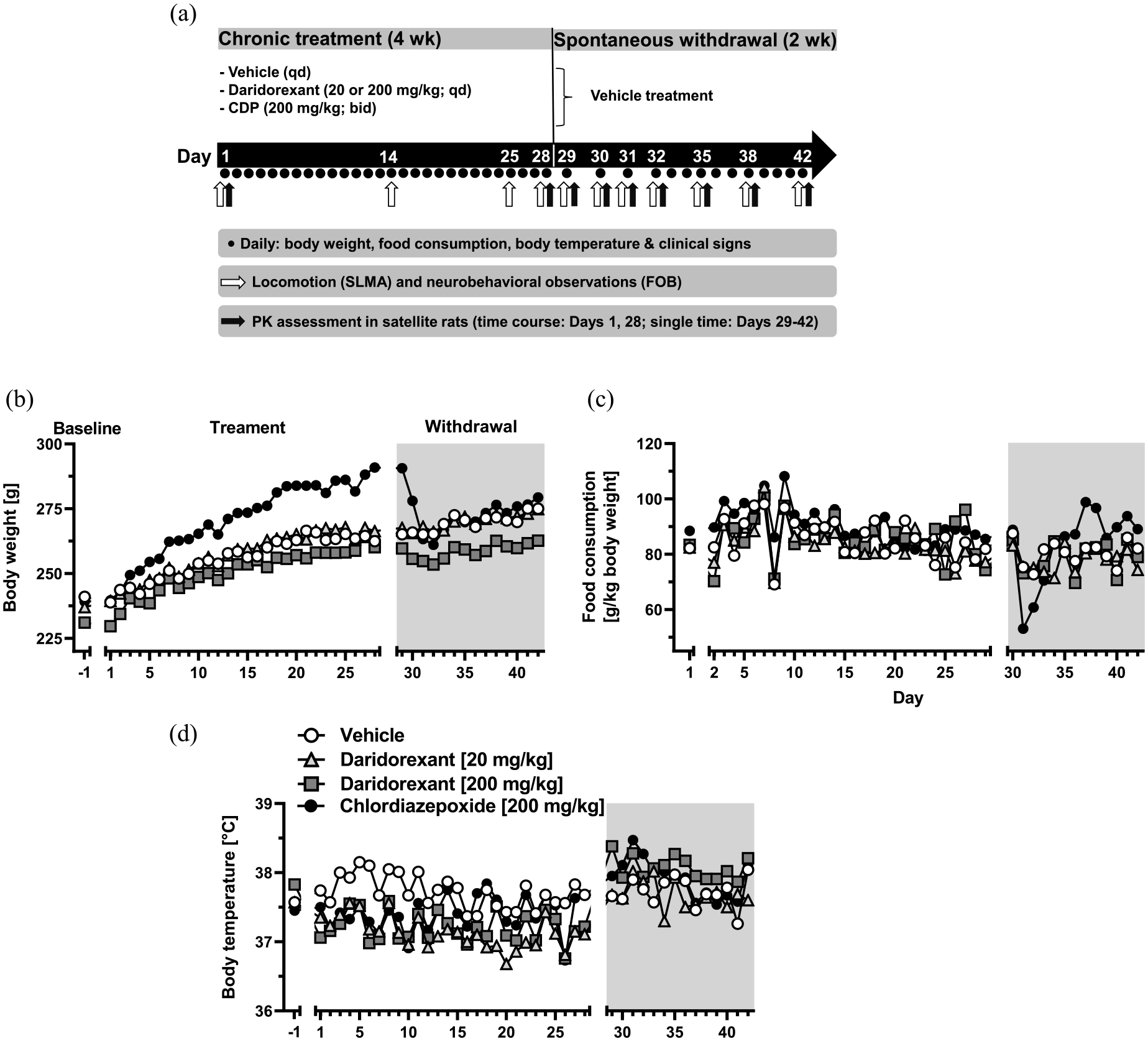
In the experiment assessing physical dependence (schematic in (a)), body weight (b), food consumption (FC) over 24 h (c), and body temperature (d) were monitored during baseline (D-1, for (b) and (d), D1 for (c); followed by a line break in the X axes), 28 days of oral vehicle or drug treatment, and 14 days after discontinuation of treatment (start of withdrawal phase indicated by a line break of the X-axis (b–d), and duration by the gray rectangle). Since FC is calculated over 24 h, the X-axis is shifted by one day for (c). The sudden and temporary drop of FC on Day 8 was likely related to a systemic error in the weighing procedure on that day. The scheme of blood sampling of satellite animals depicted in (a) only refers to daridorexant-treated rats. For better visibility, only group means are shown. For a figure with error bars depicting the standard deviation see Supplemental Figure 4.
CDP treatment increased BW (statistically significant differences vs. vehicle on Days 8 and 11, and between Days 14 and 28;
Daridorexant did not affect FC during the treatment period (Figure 3(c)). A few incidental, statistically significant differences from vehicle were considered unlikely to be drug-related, particularly, because the daily FC of vehicle-treated rats was also variable. During the withdrawal period, the level of FC in the formerly daridorexant-treated groups remained comparable to that of the vehicle group, which showed slightly reduced FC around Days 31 and 32 (statistically significant difference to Day 29 only for Day 32;
CDP led to slightly increased FC compared to vehicle, which reached statistical significance on Days 4, 8, 9, 19, and 26 (
Both daridorexant and CDP reduced BT during treatment by approximately −0.5°C (Figure 3(d)). Daridorexant reduced BT during the entire dosing period, and BT returned to normal levels upon treatment discontinuation. CDP reduced BW most prominently during the first half of the dosing period after which it remained comparable to that of the vehicle group. For a detailed description and statistical evaluation, see Supplemental Results.
Rats treated with daridorexant (either dose) moved less, had lower vertical activity, and spent more time in the margins than vehicle-treated rats on Days 1 and 14 (Supplemental Figure 2). On the following test days during the treatment and withdrawal periods, daridorexant-treated rats behaved similar to vehicle.
CDP-treated rats moved slightly more than, but did not differ in terms of vertical activity and margin time from, vehicle-treated rats during the treatment period. During withdrawal, formerly CDP-treated rats moved slightly less and spent more time in the margins, mostly during the late withdrawal period (Days 32–38). For details and statistics, see Supplemental Results.
Neurobehavioral observations, during treatment with daridorexant and following treatment discontinuation, revealed no statistically significant changes in incidence (for absence or presence of signs) or severity (for scored signs) of signs in comparison to vehicle. This was supported by the absence of any abnormal clinical signs recorded during the daily, welfare-related observations.
Treatment with CDP induced several neurobehavioral and somatic signs that were statistically different from vehicle and were recorded on Days 14, 25, and 28 (Table 4); signs included elevated body and/or tail position, piloerection, arousal and escape attempts, chewing, and a decrease in respiration. Following discontinuation of CDP treatment, several withdrawal signs such as abnormal body and/or tail position, chewing, and piloerection occurred during Days 29–32. The general welfare observations corroborated those findings: CDP-treated rats had several clinical signs including piloerection, tip-toe gait, and hunched posture, during both the late phase of dosing (starting from Day 10) and abstinence.
Neurobehavioral observations—summary of statistically significant results for rats treated with chlordiazepoxide (up-titrated to 200 mg/kg/day over 20 days; constant dose from Days 20 to 28) at defined observation time points during the treatment period, and after treatment discontinuation (Days 29–42). For the full list of all symptoms that were monitored, see Supplemental Methods.
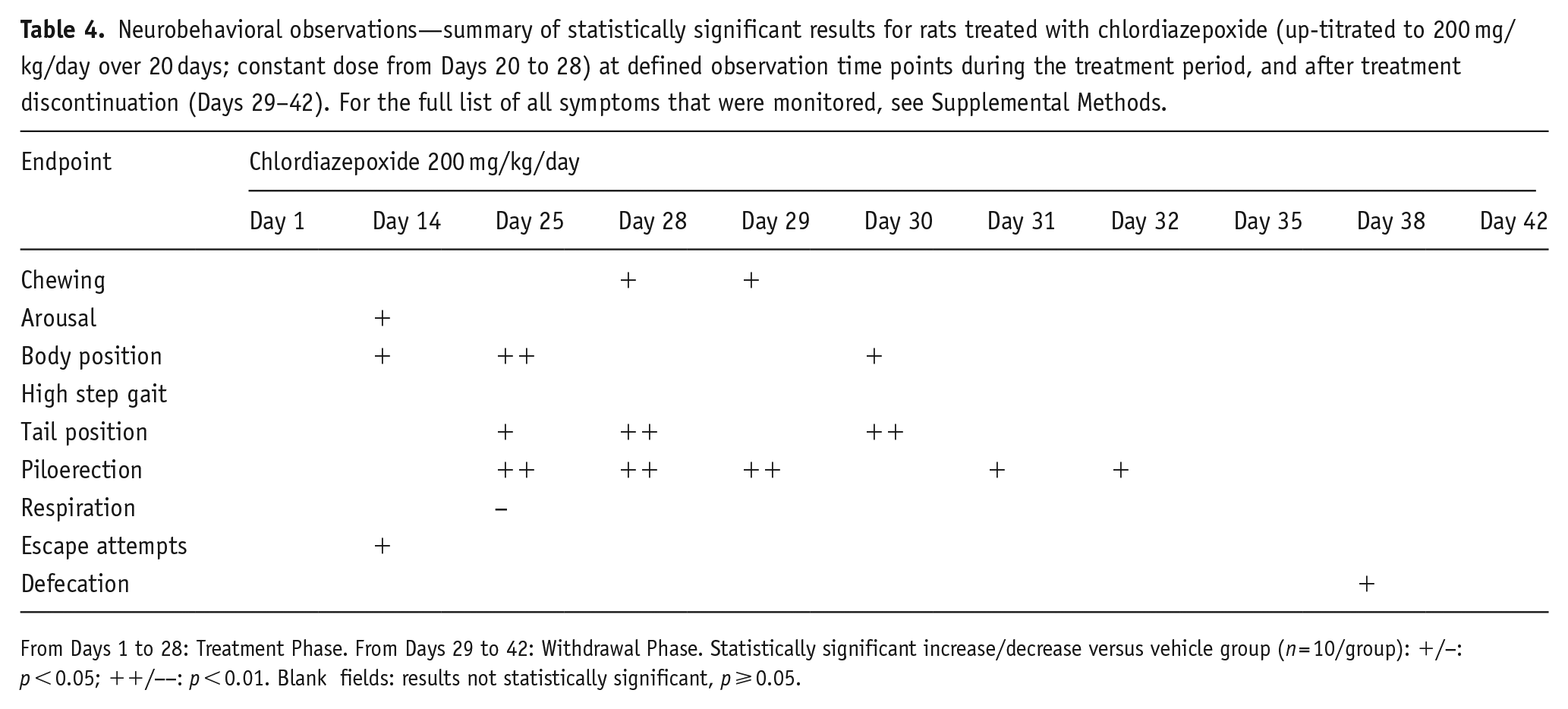
From Days 1 to 28: Treatment Phase. From Days 29 to 42: Withdrawal Phase. Statistically significant increase/decrease versus vehicle group (
The total
Plasma exposure to daridorexant after acute (Day 1) or chronic (Day 28; once daily for 4 weeks) oral administration to satellite rats (
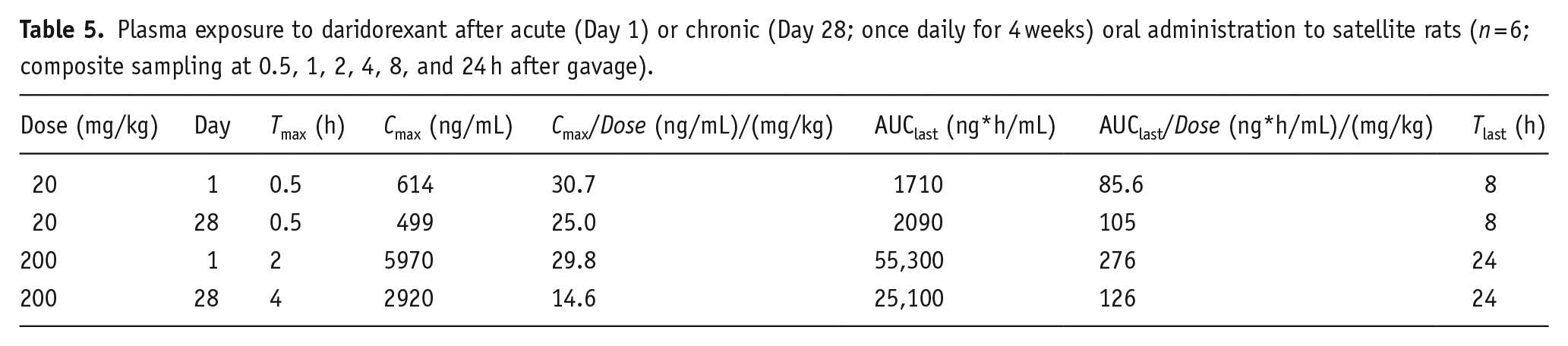
CDP was largely eliminated within 48 h after the last administration on Day 28 (Supplemental Figure 3), with very low, residual plasma levels remaining at 72 h after administration in two out of three rats.
Discussion
Daridorexant showed no signs of abuse potential in three well-recognized animal paradigms. Unlike benzodiazepines and zolpidem (Griffiths and Weerts, 1997; Licata and Rowlett, 2008), daridorexant did not cause reinforcing effects or tolerance and withdrawal symptoms. It also did not produce any interoceptive cues resembling zolpidem. This profile of daridorexant aligns with that of other DORAs (Asakura et al., 2021; Born et al., 2017) and clearly differentiates this class from conventional sleep medications based on GABA(A) receptor signaling.
The orexin system is well conserved between rat and human (Soya and Sakurai, 2020). In our experiments, rat plasma exposures to daridorexant were in a pharmacologically active range (Treiber et al., 2017) and comparable to those achieved with therapeutic or supratherapeutic human doses (Muehlan et al., 2018, 2020). This allowed for a meaningful assessment of daridorexant’s abuse potential in rats (EMA Guideline, 2006; FDA Guidance, 2017).
Drug self-administration
When cocaine was substituted with daridorexant, rats extinguished their responding similarly to rats for which cocaine was substituted with vehicle. At the same time, lever presses of daridorexant-treated rats were never lower than those of vehicle-treated rats demonstrating that daridorexant did not impair operant task performance. Re-exposure to cocaine after the substitution phase reinstated responding, indicating that neither the extinction procedure nor daridorexant disrupted cocaine-seeking behavior. This suggests that repeated, intermittent DORA administration does not interfere with the memory or perception of formerly established rewarding or reinforcing cues (Steiner et al., 2013).
The lack of reinforcing effects of daridorexant in rats trained to self-administer cocaine is in accordance with data on suvorexant and lemborexant, which were tested on monkeys trained to self-administer barbiturates. Neither suvorexant nor lemborexant caused any reinforcing effects, even at plasma concentrations several folds above the clinical
These animal results seemingly contradict the findings from human abuse potential (HAP) studies. All DORAs produce drug-liking effects similar to zolpidem in people who take sedative drugs for recreational purposes (Cruz et al., 2014; Landry et al., 2022; Schoedel et al., 2016; Ufer et al., 2022). It is unknown, however, whether the rewarding effects underlying such ratings of “drug-liking” are also sufficient to lead to reinforcement (i.e., motivated behavior involving drug taking). This requires specific testing in humans.
The DEA classified the three DORAs marketed in the United States as schedule IV substances, citing potential for psychological dependence based on the HAP study outcomes (Department of Justice, 2014, 2020, 2022). However, there is currently insufficient real-world evidence for abuse of DORAs. During the last decade, only very rare cases of non-medical use or abuse have been reported for suvorexant in official US databases such as the FDA Adverse Event Reporting System (FEARS) and the National Survey on Drug Use and Health (NSDUH) (see the Citizens Petition Daridorexant, 2023). Similarly, the phase III studies of daridorexant indicate that abuse-related adverse events were rare and not associated with positive affective states (e.g., euphoria) (Kunz et al., 2023; Mignot et al., 2022; FDA: Daridorexant Review, 2022). The FDA recently conducted a preliminary evaluation of the relationship between positive HAP data and actual post-marketing abuse for drugs and has concluded that this correlation is not always reliable, particularly not for drugs with novel mechanisms of action including DORAs (Caro et al., 2022).
Our rat self-administration study had a limitation in not using another positive reference with sedative action. The reason was that such a reference in rats trained to self-administer cocaine had not yet been established in the scientific literature. Including one could have strengthened our conclusion regarding the lack of reinforcement observed for daridorexant. Self-administration behavior is influenced by various factors, such as drug, dose, training history, and FR requirements (Solinas et al., 2006). Additionally, the duration of the substitution period can influence results. Therefore, we cannot fully exclude that daridorexant may engender self-administration in animals differently trained than in our experiment.
Drug discrimination
Lorazepam caused stimulus generalization to zolpidem as expected given the similar binding sites of both drugs at GABA(A) receptors and confirming previous findings (Sanger and Zivkovic, 1986). In contrast, daridorexant did not cause any stimulus generalization to zolpidem, indicating a lack of similarity between the interoceptive cues produced by daridorexant and 3 mg/kg zolpidem. Suvorexant up to 100 mg/kg also failed to elicit stimulus generalization to zolpidem, which is consistent with prior research (Asakura et al., 2021; Born et al., 2017). Likewise, lemborexant was devoid of stimulus generalization to zolpidem at exposures exceeding those attained with clinically approved doses (Asakura et al., 2021). Together, these behavioral outcomes with DORAs can be explained by the lack of off-target activity on GABA(A) receptors for suvorexant (Australian Public Assessment Report, 2015), lemborexant (Beuckmann et al., 2017), and daridorexant (Roch et al., 2021).
All four drugs evaluated in our drug discrimination paradigm were well tolerated. However, while the overall response rate was reduced by the two GABA(A) receptor modulators at high doses, it was not (suvorexant) or only slightly (daridorexant) affected by the DORAs. This aligns with the well-known lack of motor impairing or muscle relaxant effects of DORAs, even at supratherapeutic doses (Beuckmann et al., 2019; Roch et al., 2021; Steiner et al., 2011).
In HAP studies, people who use recreational drugs perceive the effects of DORAs as similar to those of sedatives, including benzodiazepines (Schoedel et al., 2016; Ufer et al., 2022). This is expected since participants are pre-selected based on liking sedatives. Although subjective effects are related to the discriminative-stimulus properties of drugs, they are not necessarily interchangeable (Bolin et al., 2016). Discrimination can be caused by various bodily sensations of which, for instance, only one is captured in a specific visual analog scale measuring a drug effect. Therefore, it is still unclear whether DORAs cause stimulus generalization to zolpidem in humans, which could be tested using for instance money as a reinforcer (Bolin et al., 2016).
It must be underlined that we tested daridorexant against only one dose of zolpidem used for rat training. The quality of the interoceptive effects is determined by the drug concentration, which dictates the level of engagement of the drug’s main and potential off-targets. Therefore, we cannot fully exclude that daridorexant might partially generalize to zolpidem if a different dose was used for training. Similarly, generalization to a different type of sedative with abuse potential might potentially occur. We must keep in mind that suvorexant and lemborexant were also tested against zolpidem 3 mg/kg.
Physical dependence
Chronic treatment with daridorexant was well tolerated. The reduced locomotion in a non-stimulating environment, and the increase in time spent in the margin area of the open field were consistent with the sleep-promoting effects of daridorexant (Roch et al., 2021). Likewise, the slight reduction of BT under daridorexant treatment was considered a consequence of OXR antagonism. Orexinergic neurons in rats activate brain-stem areas involved in thermoregulation (Berthoud HR, 2005) and generate heat (Monda et al., 2004). DORAs also reduce stress-induced hyperthermia (Boss et al., 2014; Martin et al., 2019). The effect on BT observed was likely due to a reduction of the gavage- and handling stress-induced hyperthermia. Benzodiazepines and CDP have similar effects (Conley and Hutson, 2007) but unlike CDP, the BT reduction observed with daridorexant was maintained throughout the entire dosing period. This is congruent with a general lack of tolerance development to DORA’s effects in animals (Roch et al., 2021).
Discontinuation of daridorexant did not lead to withdrawal symptoms or changes in physiological, neurobehavioral, or locomotor activity variables except a slight decline of BW at 200 mg/kg/day. In the absence of any other neurobehavioral observations, this decline was deemed unlikely to be a withdrawal sign. In the run-out periods of the phase III daridorexant trials, there were no group differences in the Benzodiazepine Withdrawal Symptom Questionnaire between previously placebo- or daridorexant-treated patients (FDA: Daridorexant Review, 2022; Mignot et al., 2022). Consequently, daridorexant was considered by the DEA to have no physical dependence potential (Department of Justice, 2022), which is aligned with the ruling on suvorexant and lemborexant (Department of Justice, 2014, 2020).
In contrast to daridorexant, during the discontinuation of CDP rats experienced typical withdrawal symptoms (i.e., a marked decline of BW, reduced FC and an upsurge of expression of neurobehavioral signs). In addition, the increased time spent in the marginal zone of the test arena could suggest an increase in anxiety-like behavior. These findings are consistent with the expected withdrawal signs of a benzodiazepine and with historical (Aptuit) or published data (Born et al., 2017).
Conclusion
The current results with daridorexant confirm that DORAs do not produce abuse potential signs in the most often used and officially recommended set of preclinical experiments employed for abuse potential assessment.
Supplemental Material
sj-docx-1-jop-10.1177_02698811231215415 – Supplemental material for Abuse potential assessment of the dual orexin receptor antagonist daridorexant in rats
Supplemental material, sj-docx-1-jop-10.1177_02698811231215415 for Abuse potential assessment of the dual orexin receptor antagonist daridorexant in rats by Michel A Steiner, Michael Toeroek-Schafroth, Maria Elena Giusepponi, Lisa Dacome and Michela Tessari in Journal of Psychopharmacology
Footnotes
Acknowledgements
We thank Katerina Neou-North (Idorsia Pharmaceuticals Ltd) and Jessica Beake (Beake Medicom Ltd) for careful reading of the manuscript, Melek Sevinc (Idorsia) and Vera Gros (formerly Idorsia, now F. Hoffmann-La Roche AG) for help with the coordination of the studies, Marianne Treher (Idorsia) for support in reviewing the original study reports, Susanne Globig (Idorsia), Nathalie Grignaschi (Idorsia), and Stephane Barth (Swiss BioQuant) for analysis of the drug levels in plasma samples, Alexander Treiber (Idorsia) and Kerstin Niggemann (Idorsia) for help on the design of the PK studies, Aude Weigel (Idorsia) for the supervision of the in vivo part of the PK studies, and Stephane Delahaye (Idorsia) for the supervision of its analysis. We also thank the Early Formulation and Pharmacometrics departments of Aptuit for excellent technical and analytical support.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MAS and MTS were employees and shareholders of Idorsia Pharmaceuticals Ltd at the time the study was conducted. Idorsia has developed daridorexant as a treatment for insomnia and is selling the drug under the trade name Quviviq®. MT, LD, and MEG have no conflicting interests to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Idorsia Pharmaceuticals Ltd. MAS and MTS were employees of Idorsia Pharmaceuticals Ltd. at the time the study was conducted. They received salary and financial support for the experiments. MT, LD, and MEG were employees of Aptuit, an Evotec company, at the time the study was conducted. They were commissioned by and received funding from Idorsia Pharmaceuticals for conducting the experiments that were carried out at their site.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
