Abstract
Background:
Cochrane recently published a review of esketamine and other glutamate receptor modulators in depression.
Aim:
To address the limitations of the review, analyses of esketamine data were conducted to provide additional perspective to the reviewers’ interpretation of their findings.
Methods:
Response rate, remission rate, and change from baseline in Montgomery-Åsberg Depression Rating Scale (MADRS) total score were determined using data from all esketamine phase 2/3 registration studies of treatment-resistant depression (TRD) and, separately, all esketamine phase 2/3 registration studies of major depressive disorder (MDD) and active suicidal ideation with intent. Outcomes were assessed at all timepoints (i.e., 24 h, 72 h (MDD with active suicidal intent only), and 1, 2, and 4 weeks). Enrollment criteria of the TRD studies were different than those of the studies of MDD and active suicidal ideation with intent, resulting in differences in patients’ clinical characteristics and depression severity between the cohorts. Thus, we did not compare results between these cohorts (as was done in the Cochrane review).
Results/outcomes:
In the combined TRD studies, a statistically significant between-group difference favored esketamine plus antidepressant over antidepressant plus placebo at 24 h (based on response, remission, and change in MADRS score), 1 week (change in MADRS score), 2 weeks (response and change in MADRS score), and 4 weeks (response, remission, and change in MADRS score). In the combined studies of MDD and active suicidal ideation with intent, the between-group difference was statistically different, favoring esketamine plus standard-of-care over placebo plus standard-of-care, at 24 h (response, remission, and change in MADRS score), 72 h and 1 week (change in MADRS score), 2 weeks (response), and 4 weeks (response, remission, and change in MADRS score). For both study types, the between-group difference in outcomes was not statistically significant at the other timepoints.
Conclusions/interpretation:
Esketamine improves response, remission, and depressive symptoms as early as 24 h post-first dose among patients with TRD and among patients with MDD and active suicidal ideation with intent.
Major depressive disorder (MDD) is one of the most common psychiatric disorders, affecting an estimated 350 million people worldwide (De Leo and Too, 2014; World Health Organization, 2012), and results in increased risk of suicide (Hawton and van Heeringen, 2009), substantial disability (World Health Organization, 2008), impaired quality-of-life (Rapaport et al., 2005), and substantial economic burden (Donohue and Pincus, 2007). Monoamineric antidepressant(s) have been a mainstay of MDD treatment for more than four decades; however, these traditional antidepressants take weeks to exert full effect. Approximately 30% of patients with major depression fail to achieve remission despite treatment with multiple such antidepressants (Fava, 2003). Thus, alternative medications to treat depression are needed, especially to achieve rapid symptom control. In this regard, investigation led to the discovery of the rapid onset and robust mood-elevating effects of glutamatergic N-methyl-D-aspartate receptor modulators (antagonists), including ketamine and esketamine, in treating depression (Duman et al., 2016; Newport et al., 2015). A review of the evidence on ketamine and other glutamate receptor modulators in depression was first published by Cochrane Database of Systematic Reviews in 2015 (Caddy et al., 2015), and recently updated (Dean et al., 2021). In the updated review by Dean et al. (2021), the authors stated that “reliable information about ketamine and other glutamate receptor modulators in unipolar depression (including modes of administration, comparative efficacy, duration of efficacy, and safety) is not only clinically useful but also urgently needed because such evidence can improve patients’ outcomes in the treatment of depression and provide a basis for future clinical research and treatment guidelines.”
We appreciate the substantial work that went into the review by Dean et al. (2021). This Commentary will focus on analyses within the review pertaining specifically to esketamine, with the aim of providing additional perspective to the authors’ interpretation of their findings.
By way of background, Dean et al. (2021) reported that they included 64 double- or single-blinded randomized controlled trials in their meta-analysis (31 of ketamine and 9 of esketamine). The esketamine studies included six of patients with treatment-resistant depression (TRD) (Daly et al., 2018; Fedgchin et al., 2019; Järventausta et al., 2013; Ochs-Ross et al., 2020; Popova et al., 2019; Singh et al., 2016) and three of patients with MDD and active suicidal ideation with intent (Canuso et al., 2018; Fu et al., 2020; Ionescu et al., 2021). In the intranasal esketamine registration studies, patients in the TRD trials received esketamine or placebo nasal spray, each combined with an oral antidepressant (Daly et al., 2018; Fedgchin et al., 2019; Ochs-Ross et al., 2020; Popova et al., 2019) and patients in the trials of MDD and active suicidal ideation with intent received esketamine or placebo nasal spray, each with comprehensive standard-of-care treatment (i.e., initial psychiatric hospitalization and newly initiated or optimized oral antidepressant[s] therapy) (Canuso et al., 2018; Fu et al., 2020; Ionescu et al., 2021). Efficacy outcomes included response rate (primary outcome of the Cochrane review), remission rate, and mean change scores in depression severity from baseline to various time points.
Across all esketamine studies (i.e., those of patients with TRD and of patients with MDD and active suicidal ideation with intent), Dean et al. (2021) reported that esketamine increased response (defined as reduction ⩾50% from baseline on Montgomery-Åsberg Depression Rating Scale [MADRS] total score), increased remission (MADRS <11), and reduced depressive symptoms (i.e., decrease in MADRS total score from baseline), beginning at 24 h after the first dose. Thereafter, they reported that esketamine (plus antidepressant) was more efficacious than placebo (plus antidepressant) at 72 h (change in MADRS score), at 1 week (response, change in MADRS score), at 2 weeks (response, remission, change in MADRS score), and at 4 weeks (response, remission, change in MADRS score).
Based on the consideration that patients with a depressive disorder who are being treated as outpatients have different characteristics than those requiring inpatient treatment, Dean et al. (2021) conducted sub-group analyses of efficacy outcomes by treatment setting (outpatient and inpatient). Their outpatient subgroup was comprised of patients who participated in the TRD studies. Their inpatient subgroup was comprised of patients who participated in studies of MDD and active suicidal ideation with intent, who by protocol specification were also in need of psychiatric hospitalization. However, the authors did not include data from all available esketamine registration studies of patients with MDD and active suicidal ideation with intent; in this regard, the ASPIRE II study (Ionescu et al., 2021) was not included in their inpatient subgroup analyses. Furthermore, the authors did not include data from all timepoints in their analyses. While we agree with the rationale for conducting the sub-group analyses, we consider that the authors’ interpretation of their subgroup analyses results—that is, “outpatients may have a very quick increased response to esketamine, whilst inpatients could take longer to respond”—is inappropriate and does not reflect the results of their analyses or the results from all the available data. Our critique follows.
The enrollment criteria of the TRD studies were different than those of the studies of MDD and active suicidal ideation with intent, resulting in differences in patients’ clinical characteristics and depression severity between the subgroups. Patients in the TRD studies were enrolled based on documented, treatment-resistant chronic illness, whereas patients enrolled in the studies of MDD with active suicidal ideation with intent had acute symptoms that necessitated emergency treatment. Notably, over half of patients in the trials of MDD and active suicidal ideation with intent had a prior history of suicide attempt, one-fourth of patients had a suicide attempt within 1 month of study entry, and all were at imminent risk for suicide, necessitating hospitalization for at least 5 days after randomization (Canuso et al., 2021). In contrast, suicidal ideation with intent in the past 6 months (Fedgchin et al., 2019; Popova et al., 2019) or during screening/baseline (Daly et al., 2018) was an exclusion criterion in the TRD studies. Patients in the trials of MDD and active suicidal ideation with intent also had greater depression severity at baseline (mean MADRS total score of 38.6 (Canuso et al., 2018), 41.1 (Fu et al., 2020), and 39.7 (Ionescu et al., 2021) vs. 34.1 (Daly et al., 2018), 37.6 (Fedgchin et al., 2019), and 37.1 (Popova et al., 2019) for patients with TRD).
We consider that for esketamine, the differences in efficacy results between the Dean et al. (2021) post hoc-defined outpatient and inpatient subgroups are related to a combination of factors, including patients’ clinical characteristics and phase (acute vs. chronic)/severity of illness, not to the treatment setting, per se. To clarify and expand upon the findings reported by Dean et al. (2021), we conducted post-hoc analyses using all esketamine phase 2/3 registration studies of TRD, and separately, all esketamine phase 2/3 registration studies of MDD and active suicidal ideation with intent (Figures 1–6). In the combined TRD studies, there was a statistically significant between-group difference favoring esketamine plus antidepressant over antidepressant plus placebo at 24 h (based on response, remission, and change in MADRS score), at 1 week (change in MADRS score), at 2 weeks (response and change in MADRS score), and at 4 weeks (response, remission, and change in MADRS score). In the combined studies of patients with MDD and active suicidal ideation with intent, the between-group difference was statistically different, favoring esketamine plus standard-of-care over placebo plus standard-of-care, at 24 h (based on response, remission, and change in MADRS score), at 72 h and 1 week (change in MADRS score), at 2 weeks (response), and at 4 weeks (response, remission, and change in MADRS score). In the ASPIRE studies of MDD and active suicidal ideation with intent (Fu et al., 2020; Ionescu et al., 2021), patients in both the esketamine plus standard-of-care and placebo plus standard-of-care groups experienced rapid reduction in the severity of their suicidality, as measured by Clinical Global Impression—Severity of Suicidality-Revised (CGI-SS-r) at 24 h; however, the treatment difference for all patients was not statistically significant. The between-group difference (95% confidence interval (CI)) for change in CGI-SS-r at 24 h was −0.20 (−0.43 to 0.04) for all patients and −0.31 (−0.61 to −0.01) for those with a history of suicide attempt (Canuso et al. 2021). The totality of evidence demonstrates early treatment benefit of esketamine for patients with TRD as well as for patients with MDD and active suicidal ideation with intent, the latter notable given the large nonspecific benefits afforded by the background of comprehensive standard-of-care (including initial hospitalization) which was initiated at baseline (No Author, 2003; Wasserman et al., 2012).
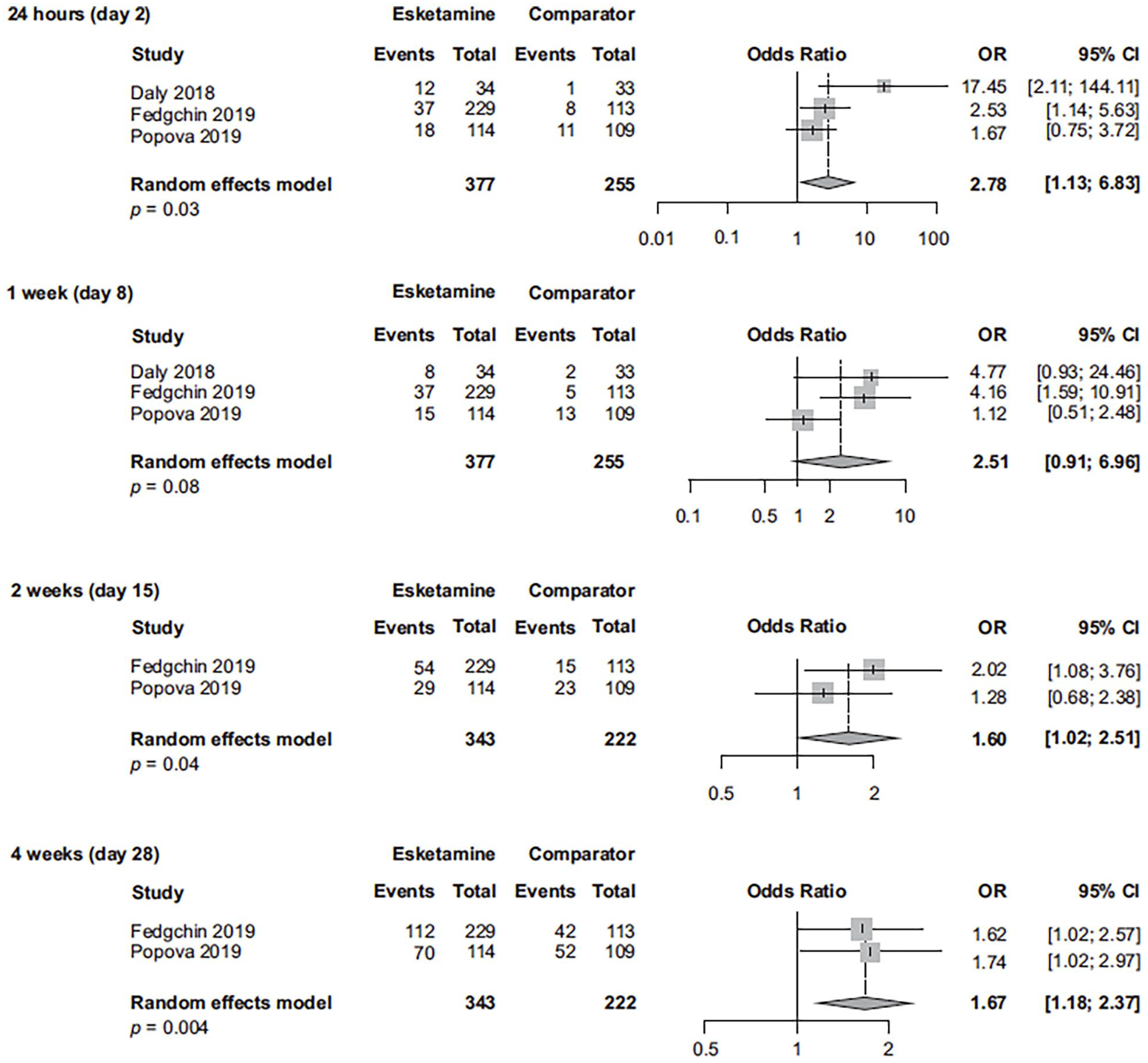
Response rate in esketamine registration studies of patients with TRD (observed cases at each visit).
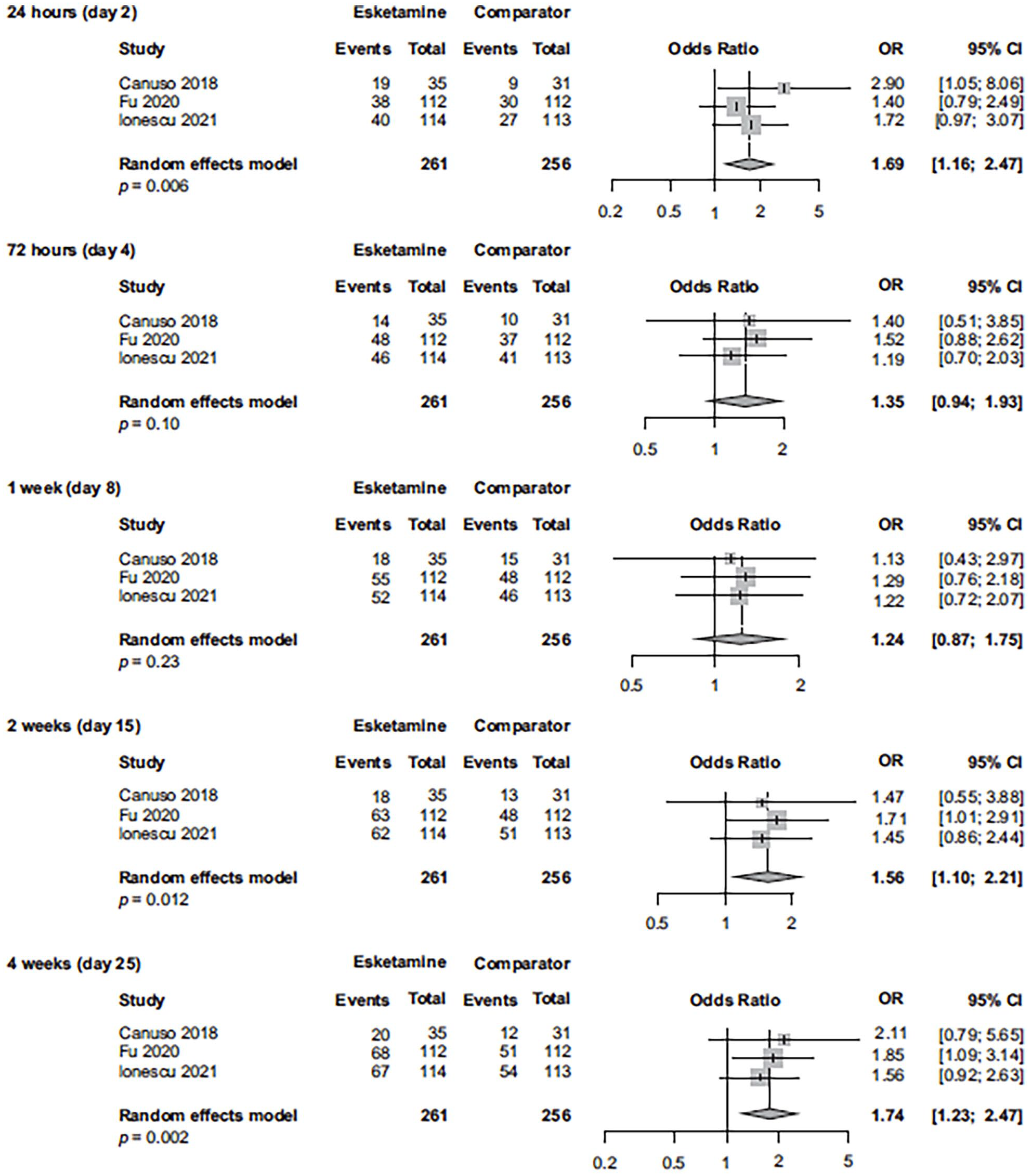
Response rate in esketamine registration studies of patients with MDD with active suicidal ideation with intent (observed cases at each visit).
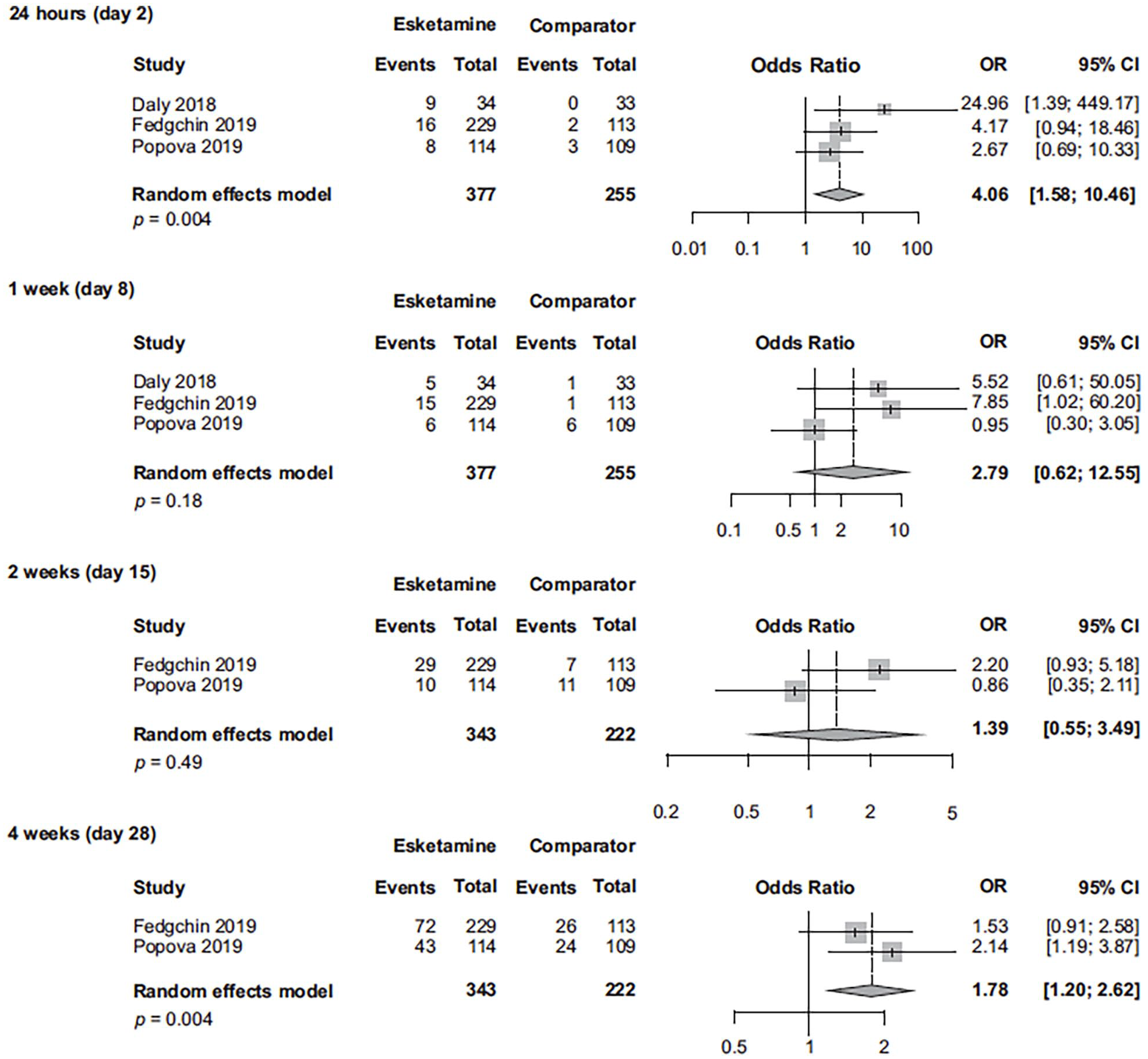
Remission rate in esketamine registration studies of patients with TRD (observed cases at each visit).
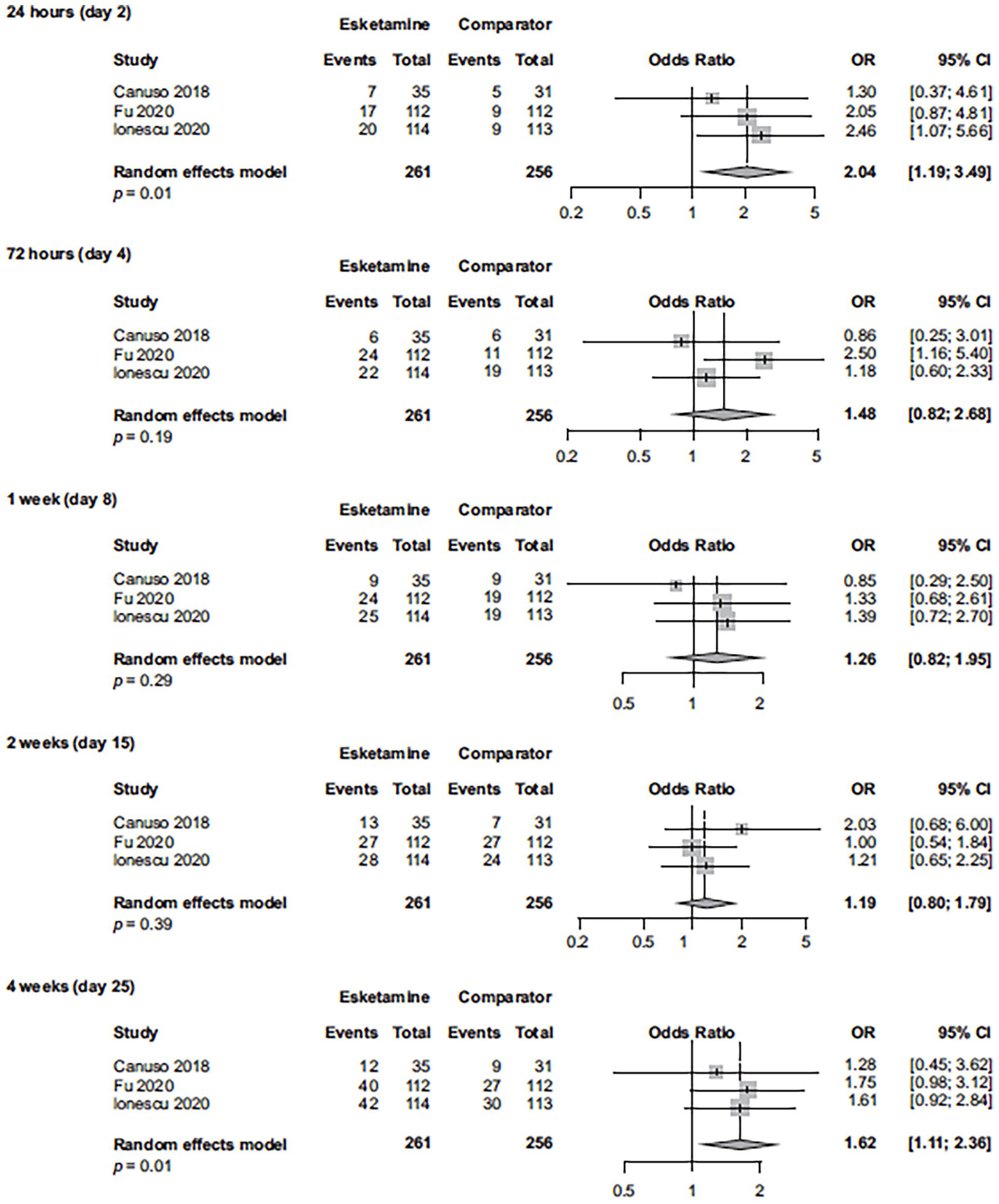
Remission rate in esketamine registration studies of patients with MDD with active suicidal ideation with intent (observed cases at each visit).
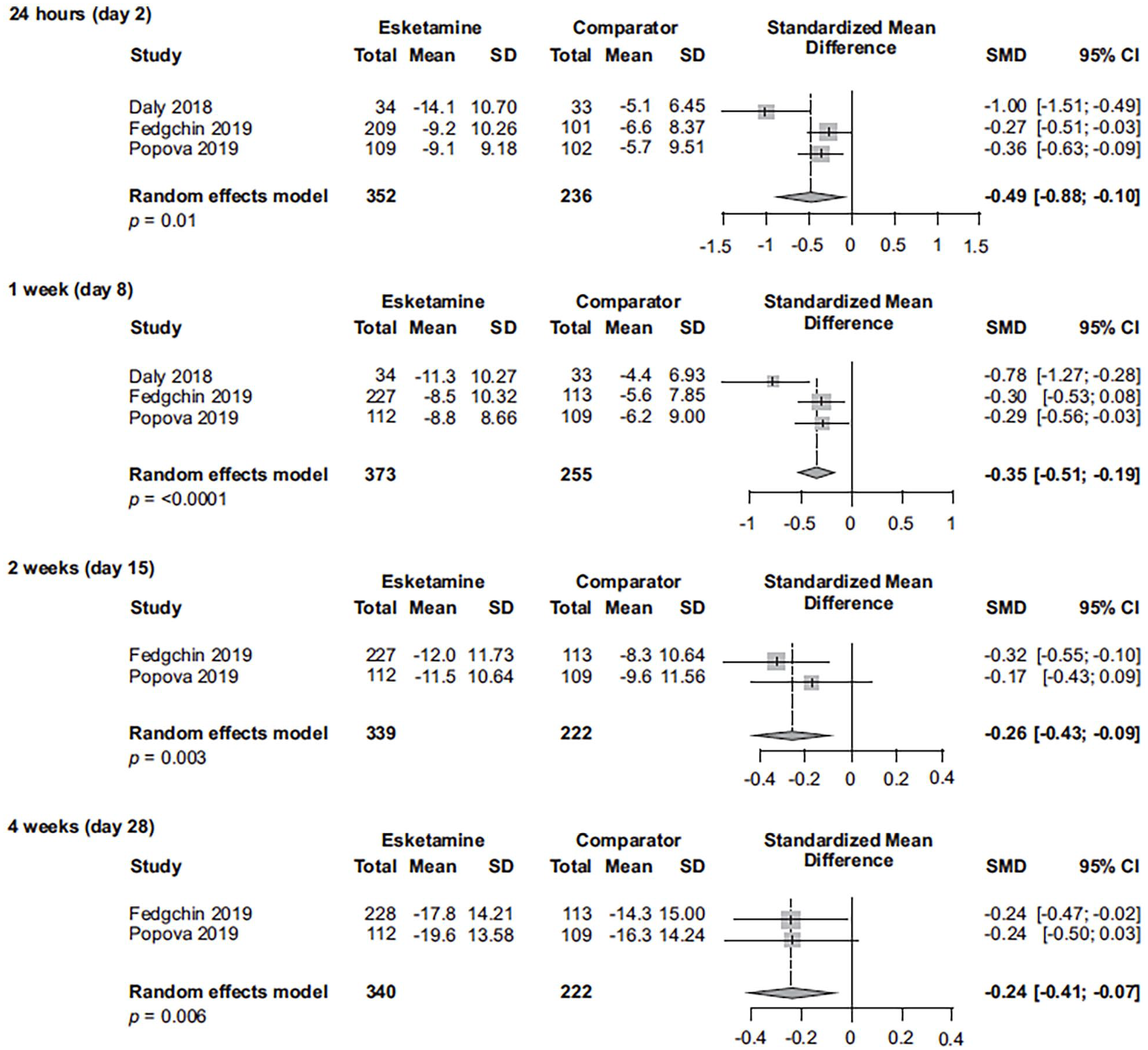
Change in MADRS from baseline in esketamine registration studies of patients with TRD (last observation carried forward).
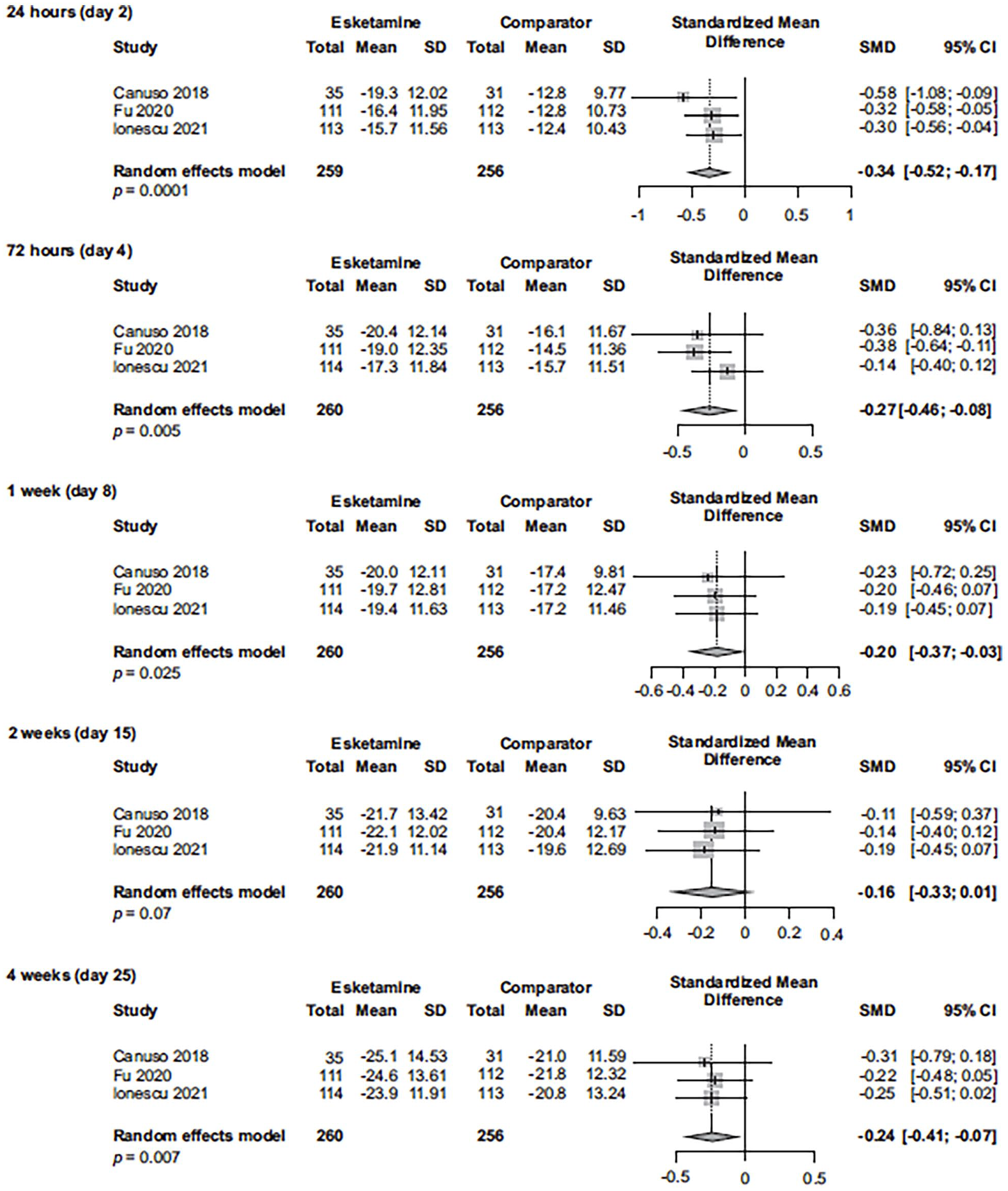
Change in MADRS from baseline in esketamine registration studies of patients with MDD with active suicidal ideation with intent (last observation carried forward).
The authors (Dean et al., 2021) stated “further studies exploring long-term outcomes are needed,” citing only 3-month esketamine data reported by Fedgchin et al. (2019). They did not acknowledge other published results of esketamine’s long-term efficacy and safety, which were reported by Daly et al. (2019) in a randomized withdrawal study after 16 weeks of treatment (esketamine plus oral antidepressant decreased relapse risk by 51% (hazard ratio [HR] = 0.49; 95% CI: 0.29–0.84) among patients who achieved stable remission and 70% (HR = 0.30, 95% CI: 0.16–0.55) among those who achieved stable response, compared with antidepressant plus placebo) and by Wajs et al. (2020) for up to 1 year of treatment (response rate of 76.5% at endpoint of optimization/maintenance phase). In line with these findings, just after publication of the Cochrane review (Dean et al., 2021), Zaki et al. (2021) presented data from an ongoing open-label study on the long-term (~4.5 years) safety and sustenance of response with esketamine nasal spray in patients with TRD. MADRS total score decreased during a 4-week induction period; the reduction persisted during an optimization/maintenance phase (mean (standard deviation) change from baseline to phase endpoint of induction, −12.8 (9.73); change from baseline to endpoint of optimization/maintenance (week 200), +1.1 (9.93)). Additional studies reporting long-term outcomes with esketamine will further inform its use in clinical practice.
In summary, the Cochrane review by Dean et al. (2021) reported rapid reduction of depressive symptoms with esketamine, but suggested that inpatients could take longer to respond. We clarified this latter point: Esketamine improves response, remission, and depressive symptoms as early as 24 h post-first dose among patients with TRD as well as among patients with MDD and active suicidal ideation with intent.
Footnotes
Acknowledgements
We acknowledge Sandra Norris, PharmD of the Norris Communications Group LLC, supported by Janssen Research & Development, LLC, for medical writing assistance and Ellen Baum, PhD (Janssen Global Services, LLC) for additional editorial support.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of Janssen Research & Development, LLC (the developer of esketamine nasal spray [Spravato®]), with the exception of Dr. Mathews who was an employee of Janssen Research & Development, LLC when this work was performed. All authors are stockholders of Johnson & Johnson.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Janssen Research & Development, LLC funded this work.
