Abstract
Introduction and objectives:
The ability to adapt to new task demands flexibly and to stabilise performance in the presence of distractors is termed cognitive control and is mediated by dopaminergic and cholinergic neurotransmission. We aimed to test the hypothesis that the effect of the cholinergic agonist nicotine on cognitive control depends on baseline dopamine levels.
Methods:
Thirty-eight healthy non-smokers (16 males; Mage=24.05 years) performed a cognitive control task including distractor and switch trials twice. Subjects were split into two parallel groups. One group received 2 g of L-tyrosine two hours prior to testing to manipulate dopamine availability experimentally, while the other group received placebo on both days. One hour later, both groups received in a within-subject design: on one day, a 7 mg nicotine patch; on the other day, a matched placebo. Response time costs for distractor and switch trials served as measures of cognitive stability and flexibility.
Results:
Nicotinic modulation reduced response time costs in switch trials and increased costs in distractor trials (nicotine×condition, p=0.027) with a trend-wise interaction between nicotine, L-tyrosine and trial type (nicotine×L-tyrosine×condition, p=0.068), which was due to stronger nicotine effects under L-tyrosine.
Conclusions:
Our data provide preliminary evidence that nicotine has opponent effects on cognitive stability and flexibility. Subjects who received the dopamine precursor L-tyrosine were more prone to nicotine effects on behaviours, which are improvements in cognitive flexibility at the cost of decreased cognitive stability.
Introduction
Cognitive control describes the ability to adapt behaviours successfully in response to relevant changes in the environment or current tasks and is often impaired in neurological and psychiatric diseases. It comprises the functions of cognitive stability and flexibility, which are commonly tested by assessing distractor suppression and task switching, respectively. Cognitive control has been associated with the dopaminergic neurotransmitter system and relies on a balance between prefrontal and striatal dopamine signalling, with striatal dopamine being important for flexible updating of behaviour and prefrontal dopamine for cognitive stabilisation (Cools and D’Esposito, 2011; Klanker et al., 2013). There is some evidence for antagonistic changes of flexibility and stability with dopaminergic stimulation (Cools et al., 2001, 2003; Dreisbach and Goschke, 2004). Recent evidence in animals also indicates a possible role of nicotinic acetylcholine receptors (nAChR) in cognitive flexibility. Allison and Shoaib (Allison and Shoaib, 2013) studied the effects of nicotine on cognitive flexibility in rats and demonstrated improved attentional set shifting between visual stimuli of the same dimension (i.e. intra-dimensional) and different dimensions (i.e. extra-dimensional). They suggested that nicotine may mediate these processes via alterations in prefrontal circuitry. In a follow-up study, nicotine and specific α7 and β2* nicotinic agonists improved extra-dimensional set shifting and reversal of the shift dose dependently (Wood et al., 2016). Additionally, the drugs improved ketamine-induced deficits in extra-dimensional set shifting. Similar findings were obtained by Terry et al. (2016) in monkeys, where varenicline, a partial α4β2 and full α7 agonist, reversed ketamine-induced deficits in reversal learning. Human evidence on nicotinic modulation of cognitive flexibility is scarce. In contrast to the results from animal research, one study found a decrease in intra-dimensional set shifting in highly dependent smokers under nicotine (Nesic et al., 2011). A recent study in a larger sample of non-smokers found no evidence for an improvement of response inhibition or interference control after nicotine administration (Ettinger et al., 2017). Inconsistent findings were also obtained with respect to nicotinic modulation of electrophysiological markers of attention- and memory-related cognitive control processes in non-smokers (Evans et al., 2014; Knott et al., 2014).
Baseline performance levels have frequently been found to modulate the effects of nicotine and may account for inconsistent effects in human nicotine studies (Newhouse et al., 2004). Low baseline performance predicted nicotine-induced improvements in attentional reorienting and cognitive control tasks in non-smokers (Behler et al., 2014; Breckel et al., 2015; Petrovsky et al., 2012; Wachter and Gilbert, 2013) and smokers (Hammersley et al., 2016), while high performers were found to deteriorate in a measure of cognitive control under nicotine (Ettinger et al., 2017). Baseline performance in cognitive control tasks may depend on individual dopamine levels (for a review, see Cools and D’Esposito, 2011) For example, individuals with lower prefrontal dopamine levels (Val/VAl carriers of the Val158Met polymorphism of the COMT gene; Chen et al., 2004) perform better in task switching and instrumental reversal learning, thus showing increased cognitive flexibility (Cameron et al., 2018; Krugel et al., 2009). Comparable results were found by Stelzel et al. (2010), who reported that subjects with higher D2 receptor densities and reduced striatal dopamine synthesis (homozygous A2 allele carriers of DRD2/ANKK1-TaqIa polymorphism; Laakso et al., 2005; Ritchie and Noble, 2003) show increased task-switching costs and switching-related neural activity in the prefrontal cortex.
Given the abundance of NAChRs on midbrain dopamine neurons that project to the striatum, nucleus accumbens and prefrontal cortex (Livingstone and Wonnacott, 2009; Wallace and Bertrand, 2013 for review), it is reasonable to assume that dopaminergic and cholinergic mechanisms may interact to adapt behaviours flexibly and that differences in baseline dopamine levels may modulate the effects of cholinergic drugs. Several human pharmacogenetic studies show that polymorphisms in dopamine system genes impact behavioural effects of nicotine in smokers, non-smokers or mixed samples (Ahrens et al., 2015; Gilbert et al., 2005; Jacobsen et al., 2006) and neural effects of nicotine in non-smokers (Bowers et al., 2015). For example, we were previously able to show that nicotine stabilised behaviour by reducing distractor interference only in non-smokers who were C/C carriers of the DRD2 SNP rs6277, a polymorphism which has been suggested to be related to increased striatal dopaminergic tone (Hirvonen et al., 2009; Smith et al., 2017).
To test the hypothesis that increased dopaminergic activity contributes to baseline-dependent nicotine effects in cognitive control, we experimentally manipulated dopamine levels with the dopamine precursor L-tyrosine. L-tyrosine is converted intracellularly by the enzyme tyrosine hydroxylase into the direct dopamine precursor L-DOPA. Tyrosine hydroxylase is the rate-limiting step in dopamine syntheses, as it is regulated by end-product inhibition (Daubner et al., 2012). However, under basal conditions, tyrosine hydroxylase is saturated to only 75% (Carlsson and Lindqvist, 1978). Microdialysis measures of DOPA or dopamine concentration in the rat striatum and medial prefrontal cortex are increased after L-tyrosine application (Brodnik et al., 2012, 2017; During, et al., 1989), indicating that dopamine availability can be increased by additional L-tyrosine. Effective systemic doses of L-tyrosine supplementation in humans vary between 500 and 12 g per day (for a review, see Jongkees et al., 2015). Recently, several studies by Colzato et al. have repeatedly shown cognitive effects on working memory and other tasks of executive function with 2 g of L-tyrosine supplementation (Colzato et al., 2013, 2016; Jongkees et al., 2017; Steenbergen et al., 2015). We therefore used 2 g of L-tyrosine or a respective placebo to manipulate dopamine levels experimentally prior to the application of nicotine or its respective placebo. Subsequently, participants performed a cognitive control task that gauged both flexible shifting between task rules and stable performance in the presence of distractors. Additionally, we included an n-back task following the cognitive control task in order to validate the effectiveness of our L-tyrosine administration, as shown previously (Colzato et al., 2013, 2016; Jongkees et al., 2017). Based on the assumption that the effects of nicotine depend on basal dopamine levels, as suggested by prior pharmacogenetic studies, we hypothesised that we would find stronger nicotinic modulation in the cognitive control task in those subjects receiving L-tyrosine. Whether this modulation is similar or different for distractor processing and task switching is not entirely clear based on the literature and was therefore tested with a three-way interaction of nicotine, L-tyrosine and task condition.
Methods
Participants
Forty-five volunteers were recruited via announcements on the university’s electronic bulletin board. All met the following criteria: healthy non-smokers, normal or corrected vision, between 18 and 35 years old, not on medication (except for contraceptives), no history of neurological or psychiatric disease, body mass index (BMI) between 18 and 26 kg/m2, right-handed, no thyroid gland malfunction, not suffering from phenylketonuria and not pregnant. Female volunteers using hormonal contraception were tested when taking the contraceptives (not on pause), and those not taking contraceptives were tested within the first 10 days of their menstrual cycle (follicular phase). One subject accidentally received nicotine on both sessions which led to exclusion of the data. Three subjects did not complete the nicotine session due to side effects such as nausea or dizziness. Further, subjects’ data were excluded from analysis if they showed poor performance in the respective test. Poor performance was defined as an accuracy not being significantly higher than chance level (permutation testing, p=0.05). This led to 38 data sets (15 male; MBMI=22.99 kg/m2; Mage=23.86 years) for the cognitive control task and 39 data sets (16 male; MBMI=23.06 kg/m2; Mage=24.05 years) for the n-back task, which were used for statistical analysis.
Drug administration and experimental design
Drug administration was double blinded and placebo controlled. Subjects were randomly assigned to the L-tyrosine group or the placebo group in a between-subject design (see Figure 1 for study design). On both experimental sessions they either received 2 g of L-tyrosine (Bulk Powders, Colchester, UK) or microcrystalline cellulose (placebo; Sigma–Aldrich, St. Louis, MO) dissolved in 330 mL of orange juice. Participants were encouraged to drink the whole glass at a good pace within a few minutes. L-tyrosine was administered two hours prior to the task performance which corresponded to the time point of maximal plasma levels after oral doses of 100 mg/kg or 150 mg/kg L-tyrosine (Glaeser et al., 1979). Nicotine was administered transdermally in a within-subject design using a 7 mg nicotine patch (NiQuitin Clear 7 mg; Omega Pharma, Nazareth, Belgium). A pharmacologically inactive patch served as placebo (DracoHydro ultra; Dr Ausbuettel & Co. GmbH, Dortmund, Germany). The patch was administered one hour prior to the task. With this administration schedule, plasma levels during the task were approximately half of the maximal levels present in smokers after patch application (Rasmussen, et al., 2018). The schedule, however, minimises side effects in non-smokers and has previously yielded behavioural effects (Ahrens et al., 2015; Behler et al., 2014; Breckel et al., 2015; Potter and Newhouse, 2008). The order of patch administration was randomised and balanced. The patch was placed on the subject’s back, directly above the belt line and covered with band-aid to avoid subjects being able to see the patch type during the administration period. Furthermore, patch application and preparation of the drinks were performed in absence of the experimenter by a third person. The combination of a between- and within-subject design was chosen to reduce practice effects due to frequent testing.
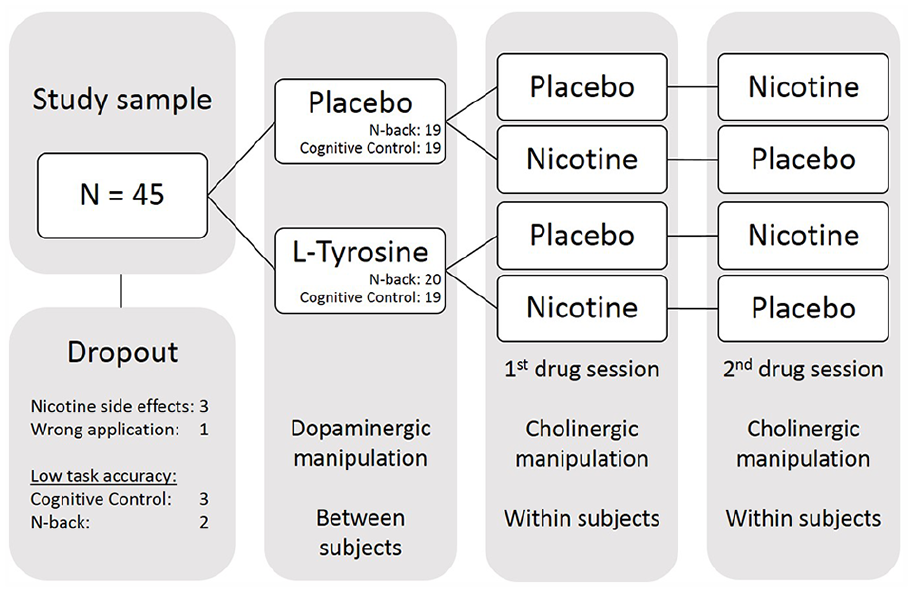
Study design. In total, 45 subjects participated in the study. Due to drop-outs and low performance in task accuracy, the final sample compromised 38 sets for cognitive control task data and 39 sets for n-back task data. While dopaminergic manipulation with L-tyrosine occurred between subjects, cholinergic manipulation was carried out within subjects. Drug application took place double blinded and pseudo randomised.
Volunteers attended three sessions. During the first session, subjects received information about study goals and schedule, and they signed a statement of informed consent. Furthermore, they performed the following neuropsychological paper and pencil or computer-based tests: CANTAB (Cambridge Cognition, 2019) tests: intra-extra dimensional set shift and rapid visual information processing; multiple-choice word test (German verbal intelligence estimate; Schmidt and Metzler, 1992) and impulsive behaviour scale (Kovaleva et al., 2012). Subsequently, they were introduced to the cognitive control task using a short demonstration with a longer inter-stimulus interval. Afterwards, they performed a complete run (25 minutes) of the whole cognitive control task at normal speed to ensure adequate performance levels prior to drug administration. The second session took place one day after the first session. Volunteers were instructed to fast overnight, to refrain from alcohol the evening before and not to drink coffee for at least two hours prior to the appointments. The second session always started at 8:30am or 9:30am. Depending on their group, subjects received either L-tyrosine or placebo. Half an hour later, they were allowed to eat one piece of apple or banana. Another 30 minutes later, either a 7 mg nicotine patch or an inactive placebo was administered in a randomised order. Additionally, subjects were allowed to eat a piece of bread (approximately 50 g of pretzel products) with vegetarian spread. The patch was removed after 50 minutes, right before the subjects started to perform the cognitive control task for approximately 25 minutes. Afterwards, subjects were given a short break for several minutes before performing the n-back task, which was always performed as the second task. The third session took place approximately one week after the second session, and both sessions were identical except for the administered patch (nicotine/placebo). At the end of each session, subjects gave feedback about whether they thought they had received treatment or placebo. Subjects’ well-being was monitored by the experimenter and assessed by means of Bond and Lader (1974) rating scales and cardiovascular measures (heart rate and blood pressure) four times throughout each session. The status was measured before L-tyrosine/placebo administration, before nicotine/placebo patch administration, before nicotine/placebo patch removal and after performing the cognitive control task. The study was conducted in accordance with the Declaration of Helsinki, with ethics approval obtained from the Ethics Committee of the German Psychological Association. All subjects gave their written informed consent and were reimbursed (10€/hour) for their time.
Cognitive control task
We used a sustained attention task with switch and distractor trials to gauge both cognitive flexibility and stability (adapted from Armbruster et al., 2012; see Figure 2). Subjects were presented with digits on the upper part of a computer screen for 900 ms every two seconds and had to indicate with their right hand whether it was odd or even (80% of all trials, ongoing trial). After every three to six trials, this ongoing task was interrupted by one of three conditions (each 6.6%), indicated by the appearance of a second digit in the lower part of the screen which differed in brightness from the upper digit. If the upper digit was brighter (distractor trial), the task rule was unchanged; if the lower digit was brighter (switch trial), subjects had to indicate, using the left hand, whether this number was smaller or larger than five. Assignment of task rules to the hands was counterbalanced across subjects. In the third condition (ambiguous trial), it was impossible to perceive brightness differences visually, although numbers differed by up to 8% in brightness. As the ambiguous condition is not the focus of this article, it will not be analysed further. In total, 720 trials were presented, with a short pause after half of the trials.
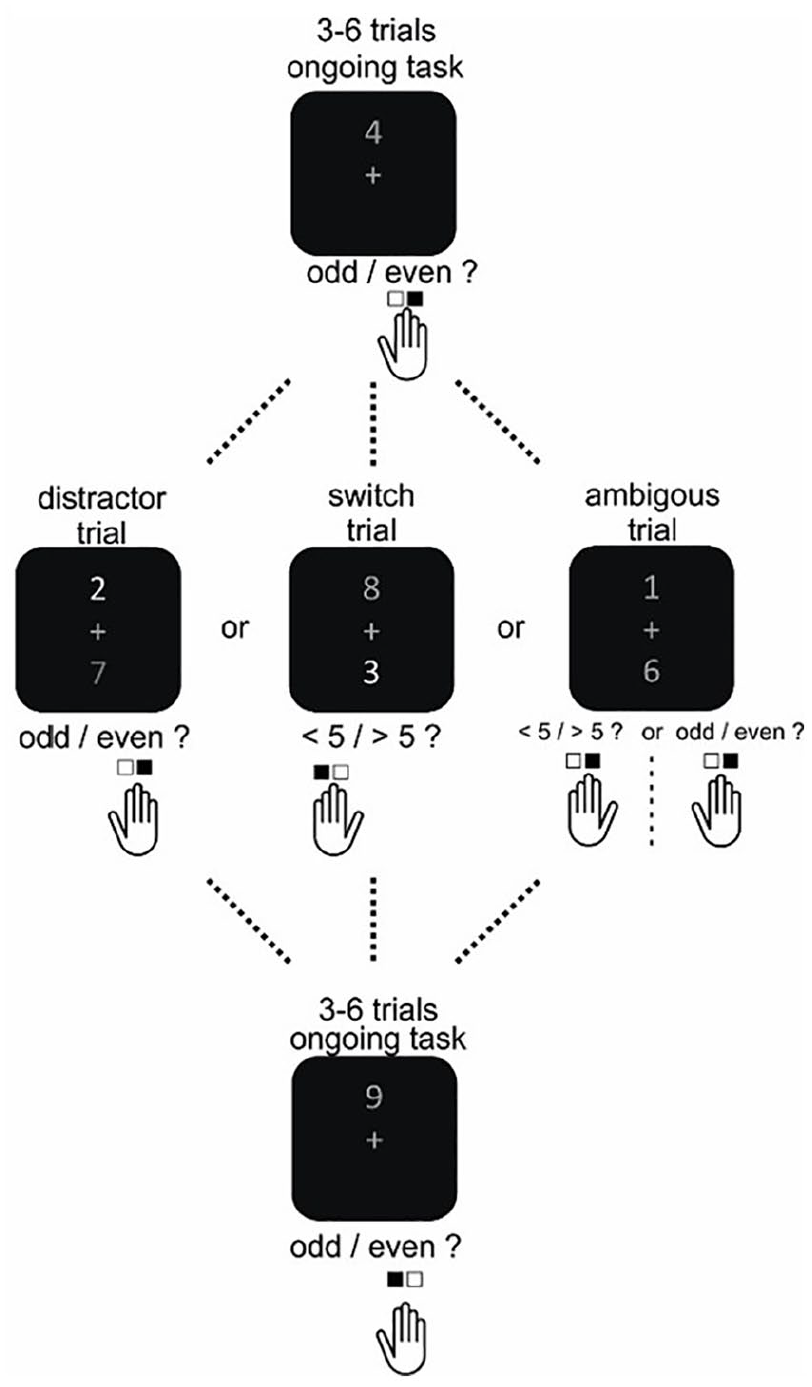
Schematic representation of the cognitive control task. After every three to six trials, the ongoing task condition was interrupted by one of three conditions. In case of a distractor trial, task rules remained equal to the ongoing condition, and the second digit was not of relevance. In case of a switch trial, attention had to be shifted towards the second digit, and another response rule was applied. See text for further details. Note that the ambiguous condition is not of relevance for this paper and is not analysed further here.
N-back task
Subjects performed an n-back task with a two-back and a three-back condition. Three blocks of each condition were presented alternatingly. A stream of single digits (0–9) was displayed on screen, with each stimulus being present for 500 ms, and an inter-stimulus interval jittered between 2400 ms and 3100 ms. Participants responded to targets (33% of the trials) and to non-targets by pressing the index or middle finger of the right hand, respectively. A total of 180 stimuli were presented, and responses could be given within 2000 ms after stimulus onset. The n-back task was primarily included to validate the effectiveness of L-tyrosine in a task, which is well known to rely on prefrontal dopamine (Aalto et al., 2005; Wang, et al., 2004). If L-tyrosine increased dopamine levels, working memory performance should be increased under L-tyrosine. Both paradigms were programmed with Presentation® software (Neurobehavioural Systems, Berkeley, CA). To ensure that the viewing distance was 50 cm, a chin rest was used. Responses were given via response boxes and recorded with Presentation®.
Data analysis
Data from the cognitive control task were analysed with respect to response time (JASP v0.9; JASP Team, Amsterdam, The Netherlands). We calculated the mean response time for each condition and response time costs for distractor and switch trials (differences in means between the distractor or switch and the ongoing condition). Statistical analysis focussed on response time costs. Further, neuropsychological test and cognitive control task data from the first session (baseline measurement without drugs; only second half taken due to learning effects in first half) were used to test for differences between groups. Except for switch costs in the cognitive control task, none of the tested variables showed significant differences between groups (see also Supplemental Table S1). Hence, to correct for group differences, reaction time costs were baseline corrected by the use of change scores (response time costs under drug–response time costs at baseline). Corrected response time costs were then entered into one repeated-measures analysis of variance (rmANOVA) with the between-subject factor L-tyrosine (placebo/L-tyrosine) and within-subject factors nicotine (placebo/nicotine) and condition (distractor/switch costs). The rmANOVA with three factors was chosen to enable testing, among other things, whether the dopaminergic modulation of nicotine effects is similar or different for distractor and switch trials. For the n-back task, accuracy values (sum of hits and correct rejections) were calculated separately for the two-back and three-back condition. Accuracy rather than reaction time was analysed, since prior L-tyrosine studies found larger effects on accuracy-based measures (Colzato et al., 2013; Jongkees et al., 2017). Results were analysed usingone rmANOVA with the between-subject factor L-tyrosine (placebo/L-tyrosine) and within-subject factors nicotine(placebo/nicotine) and condition (two-back/three-back). Cardiovascular effects and mood ratings were analysed by calculating the difference between time point 3 (before nicotine/placebo patch take-off) and time point 1 (baseline, before L-tyrosine/placebo administration). These values were then entered into rmANOVAs with the within-subject factor nicotine (placebo/nicotine) and the between-subject factor L-tyrosine (placebo/L-tyrosine).
Results
Cognitive control task
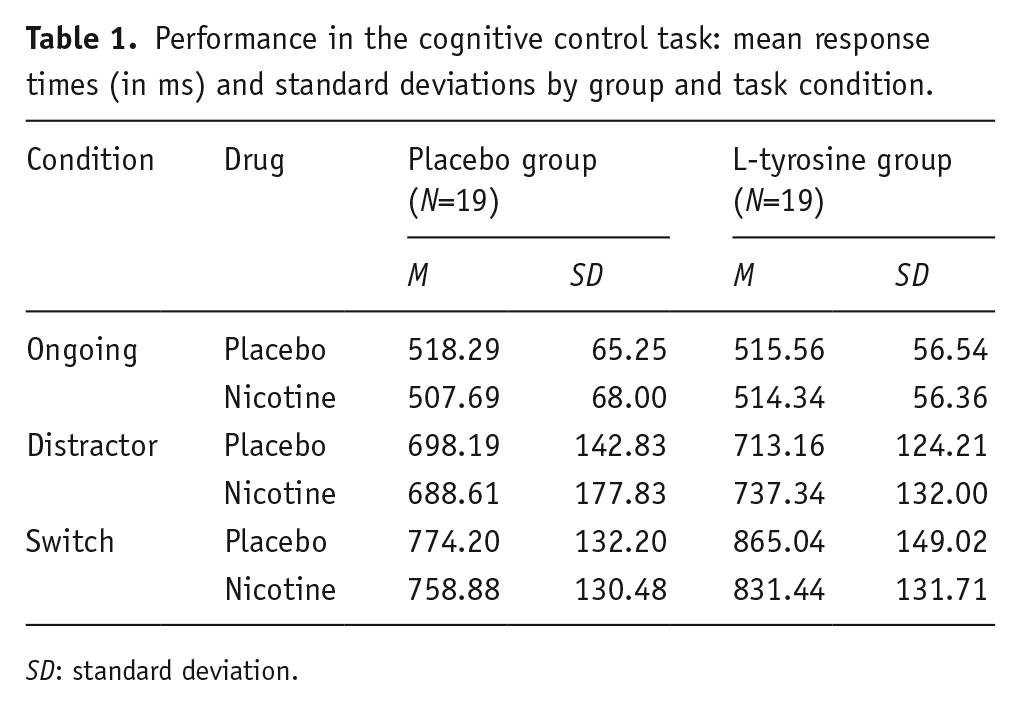
Subjects showed high accuracy with mean values >90%. Table 1 displays response times for ongoing, distractor and switch trials under placebo and nicotine for the placebo and L-tyrosine group. Statistical analyses focussed on switch costs and distractor costs as measures of cognitive flexibility and stability, respectively. There was no main effect of nicotine on response time costs (F(1, 36)=0.117, p=0.735), but there was an interaction between nicotine and condition (F(1, 36)=5.286, p=0.027, η2p=0.128) driven by reduced switch costs and increased distractor costs under nicotine. Interestingly, we found a trend for a three-way interaction between both drugs and condition (F(1, 36)=3.547, p=0.068, η2p=0.09). Neither the main effects of condition nor L-tyrosine nor other interactions were significant (p>0.1). To characterise the three-way interaction better, we conducted two separate ANOVAs for the L-tyrosine and placebo group, respectively, with the within-subject factors nicotine and condition. Figure 3 illustrates the effects of nicotine in the L-tyrosine and placebo group, and shows that the effects of nicotine were stronger when subjects received L-tyrosine before nicotine application. In this group, the nicotine application had opposing effects on distractor and switch costs, with increased distractor costs and decreased switch costs (interaction nicotine×condition, F(1, 18)=7.407, p=0.014, η2p=0.292). In the placebo group, this interaction was not significant (F(1, 18)=0.106, p=0.749). Further, in both groups, the main effect of nicotine remained insignificant (p>0.7). Hence, the effects of nicotine on cognitive control processes are increased under L-tyrosine with opposing effects on cognitive flexibility and stability.
Performance in the cognitive control task: mean response times (in ms) and standard deviations by group and task condition.
SD: standard deviation.
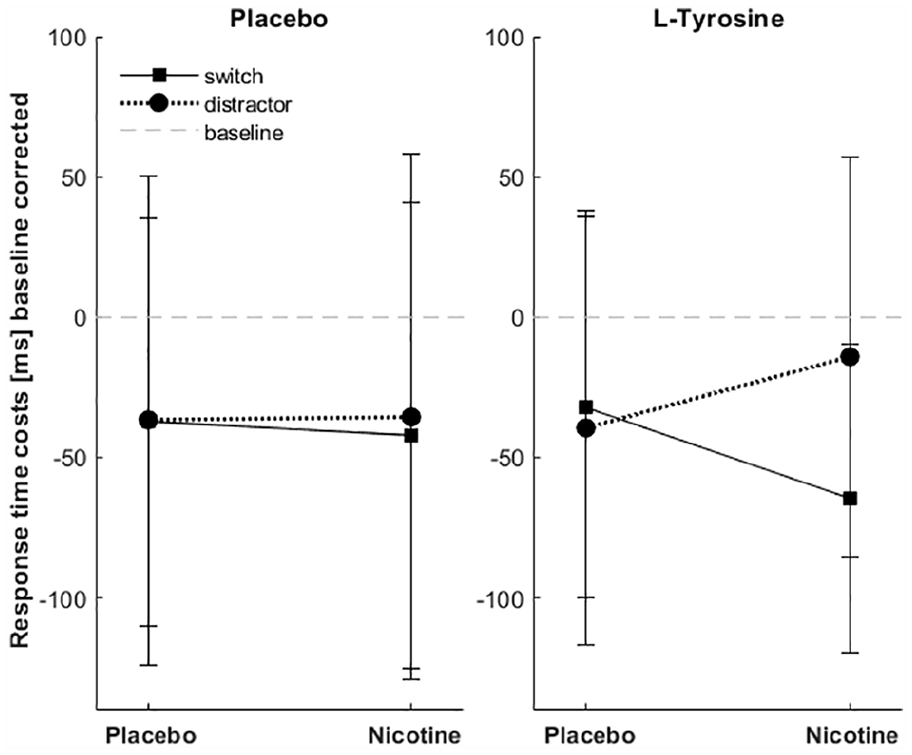
Nicotine-induced modulation of switch and distractor costs in the placebo and L-tyrosine groups. Response time costs were calculated as the difference between response times in distractor or switch trials and response times in the ongoing task. Data were further corrected for baseline differences (drug sessions–baseline session). Therefore, decreased (negative) values represent smaller costs under the drug. Nicotine effects were stronger when dopamine levels were increased with L-tyrosine (right). Note the opposing effects of nicotine on distractor and switch costs. Error bars represent standard deviation.
Working memory task
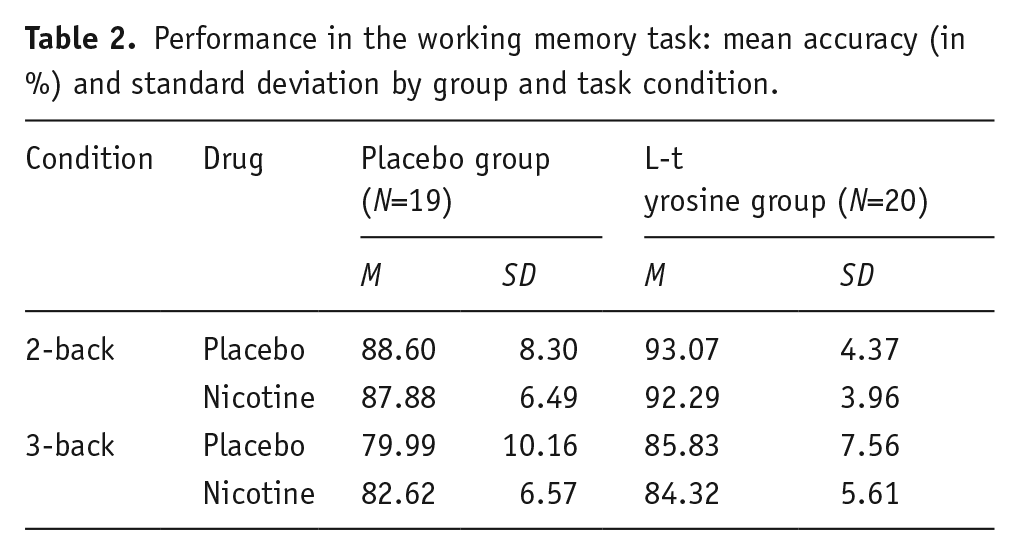
Accuracy in the n-back task was significantly higher in the two-back condition compared to the three-back condition (main effect condition, F(1, 37)=75.79, p<0.001, η2p=0.672). The L-tyrosine group showed significantly higher accuracy compared to the placebo group, independent of condition (main effect of L-tyrosine, F(1, 37)=7.13, p=0.011, η2p=0.162). All other effects, including a nicotine main effect or interactions (also L-tyrosine×nicotine), were not significant (p>0.1). Figure 4 illustrates the main effect of condition and of L-tyrosine; data as a function of all individual conditions are listed in Table 2.
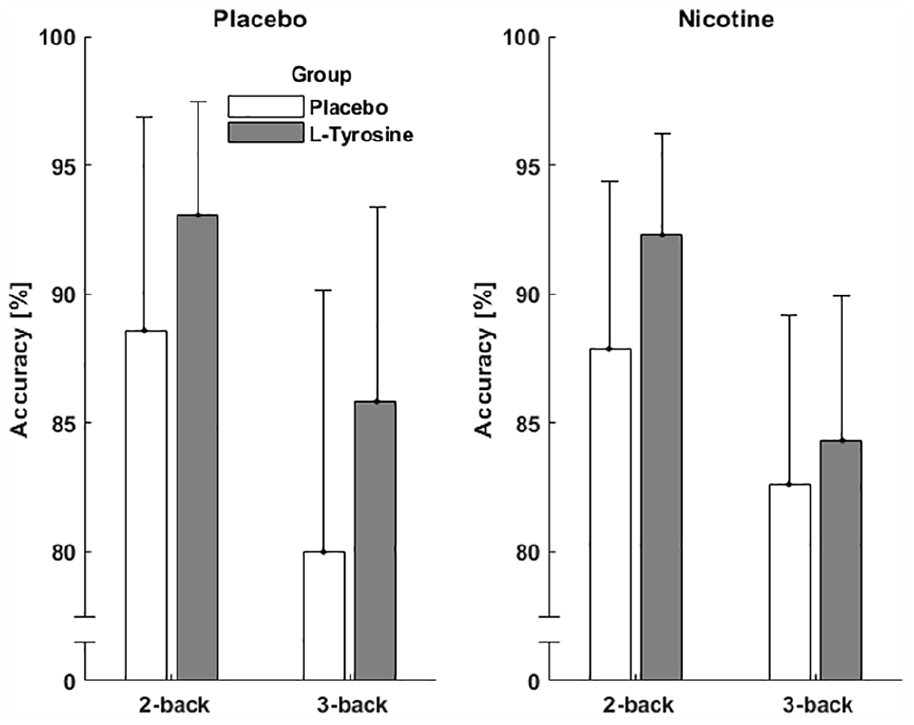
Improvement of working memory with L-tyrosine. L-tyrosine significantly increased accuracy compared to placebo. Error bars represent standard deviation.
Performance in the working memory task: mean accuracy (in %) and standard deviation by group and task condition.
Physiological measures and mood ratings
Analysis of heart rate showed a main effect of nicotine (F(1, 36)=9.174, p=0.005, η2p=0.203), with a larger increase under nicotine (5.7 beats per minute) compared to placebo (0.4 beats per minute) after drug application. L-tyrosine did not significantly affect heart rate nor did it interact with nicotine (p>0.1). The mean arterial pressure (MAP), which is a combined metric of systolic and diastolic blood pressure (BP) measures (MAP=BPdiast+1/3×(BPsyst−BPdiast) was affected by L-tyrosine (F(1, 36)=4.214, p=0.047, η2p=0.105) but not by nicotine. There was no interaction between both drugs (p>0.1). Under L-tyrosine, MAP decreased on average by 2.77 mmHG from baseline until the end of patch administration compared to an increase of 5.26 mmHG under placebo (pooled over placebo and nicotine sessions). Subjective ratings of alertness were significantly affected by nicotine (F(1, 36)=6.471, p=0.015, η2p=0.152). While subjects became more alert from baseline to the end of patch administration under placebo (end of administration minus baseline = −6.377), ratings were unchanged under nicotine (−0.001). Further, nicotine significantly affected contentedness (F(1, 36)=6.882, p=0.013, η2p=0.16). Contentedness of subjects increased under placebo (−3.281) but decreased with nicotine (1.476). L-tyrosine did not have significant effects nor did it show interactions with nicotine on alertness and contentedness (p>0.1). Nicotine had a significant effect on calmness (F(1, 36)=98.234, p<0.001, η2p=0.732), with decreased under nicotine (35.29) compared to placebo (−0.409). There was no main effect of L-tyrosine on calmness (p>0.1), but a trend for an interaction with nicotine (F(1, 36)=3.056, p=0.089, η2p=0.078). An overview of physiological measures and mood ratings is shown in Supplemental Table S2. Additionally, subjects gave feedback at the end of each treatment session on whether they thought they had received placebo or treatment. In the placebo patch sessions, 20/41 subjects correctly identified the placebo patch; in the nicotine session, 26/41 identified that they had received nicotine. A 2×2 chi-square test showed that subjects’ guesses did not significantly differ from an equal distribution (χ2=1.246, p=0.264). Regarding the L-tyrosine treatment, in the first treatment session, 3/19 subjects in the L-tyrosine group compared to 10/19 in the placebo group thought they had received L-tyrosine (χ2=5.729, p=0.017). In the second treatment session, 6/19 subjects in the L-tyrosine group compared to 11/19 in the placebo group thought they had received L-tyrosine (χ2=2.245, p=0.134). Hence, subjects were not able to identify the received substances correctly.
Discussion
We aimed to investigate whether the effects of nicotine on cognitive control are mediated by baseline dopamine levels. We found stronger effects of nicotine in subjects who received L-tyrosine additionally, with a nicotine-induced increase in cognitive flexibility but a decrease in cognitive stability.
Effectiveness of L-tyrosine in increasing baseline dopamine levels
Administration of L-tyrosine, a precursor of the catecholamines dopamine and noradrenaline, increases L-Dopa levels and catecholamine metabolism in the striatum and medial prefrontal cortex in rats (Brodnik et al., 2012, 2017). We used L-tyrosine to manipulate dopamine levels experimentally in human subjects. Even though we cannot exclude that the effects of L-tyrosine were mediated to some extent by an elevation of noradrenaline levels, we would like to underline that only 5% of dopamine is hydroxylated to noradrenaline (Nutt and Fellmann, 1984). Further, L-tyrosine depletion shows much stronger effects on dopamine compared to noradrenaline release (Le Masurier et al., 2014), making a dopaminergic contribution more likely than a noradrenergic contribution.
The effectiveness of L-tyrosine was validated behaviourally with a working memory task, which is well known to rely on levels of prefrontal dopamine measured for example in humans by means of positron emission tomography (PET; Aalto et al., 2005) or in monkeys by means of in vivo single-cell recordings (Wang et al., 2004) or microdialysis (Watanabe et al., 1997). Consistent with our expectations and several prior studies on the effects of acute L-tyrosine on n-back performance (Colzato et al., 2013; Jongkees et al., 2017), we found increased accuracy in the n-back task under L-tyrosine which may be due to increased prefrontal dopamine. In a computational model, Durstewitz and Seamans (2008) showed that an increase in dopamine concentration in the prefrontal cortex leads to a D1-dominated state of prefrontal cortex activation. This is associated with an energy landscape showing deeper attractor states, supporting highly robust representations and stable performance but impairing flexible switching to an alternative behaviour. If we assume that L-tyrosine primarily increased dopamine levels in the prefrontal cortex leading to a D1-dominated state, this would also be consistent with the improved working memory performance in the n-back task. Note that performance on the working memory task after the challenging cognitive control task may have increased the effects of L-tyrosine, which is more effective in cases of dopamine depletion occurring after stress or high cognitive demands (Fernstrom and Fernstrom, 2007; Lehnert et al., 1984; Mahoney et al., 2007). In contrast to the cognitive control task, manipulations of dopamine levels with L-tyrosine did not modulate the effects of nicotine on working memory task performance in our sample of non-smokers. An absence of nicotinic modulation of working memory in non-smokers is consistent with most prior studies (Fisher et al., 2012; Kleykamp et al., 2005; MacQueen and Drobes, 2017; but see Kumari et al., 2003), and a genetic modulation of nicotine effects on working memory was only found in a mixed sample of smokers and non-smokers (Jacobsen et al., 2006). A meta-analysis by Heishman et al. (2010), which reviewed nine different studies, including smoker and non-smoker samples with different application routes, also yielded non-significant effects of nicotine on working memory accuracy. Even though n-back performance also relies on aspects of stability, such as maintenance of representations in working memory and flexibility such as updating, these processes differ from those needed in the cognitive control task. For example, updating demands are much higher in the working memory task, where working memory contents have to be updated in every trial. In contrast, updating in the cognitive control task relates to updating of stimulus–response associations and is only required in a minority of trials – the switch trials. In line with these commonalities and differences of cognitive operations involved, neuroimaging studies also point to common and distinct prefrontal brain areas involved in task switching, stroop and n-back performance (Derrfuss et al., 2004).
Effects of increased baseline dopamine levels on nicotinic modulation of cognitive stability and flexibility
Cognitive stability and flexibility were studied within one task focussing on distractor and switch costs, respectively. Administering L-tyrosine compared to placebo did not affect switch or distractor costs significantly. Nicotine administration reduced switch costs and increased distractor cost compared to placebo. This effect was largely driven by subjects who additionally received L-tyrosine, suggesting an interaction between increased dopamine availability and nicotinic stimulation.
The literature about L-tyrosine effects on flexibility and stability is diverse. A previous study which used the same dose of acute L-tyrosine in female participants reported increased task switching under L-tyrosine (Steenbergen et al., 2015). In contrast to our study, task switches were predictable, and effects of the drug were only present in case of long response–stimulus intervals. The authors suggest that L-tyrosine supports conflict-resolving rather than preparatory components of switching and may act primarily in striatal brain regions. This would be in line with a recent PET/functional magnetic resonance imaging study showing that greater striatal dopamine synthesis capacity is associated with better task switching (Berry et al., 2016). In contrast, studies that compared subjects with polymorphisms that impact on dopamine signalling showed that genotypes associated with presumably lower dopamine levels are better in task switching (Cameron et al., 2018; Colzato et al., 2010; Krugel et al., 2009; Stelzel et al., 2013).
The major aim of this study was to investigate whether experimentally increased dopamine levels may explain interindividual differences in the effects of nicotine on cognitive control. Using PET imaging, Samanez-Larkin et al. (2013) investigated whether the effects of dopaminergic drugs depend on individual differences in dopamine signalling. Since dopamine receptor availability correlated positively with a reduction in switch costs after amphetamine administration, the authors conclude that thalamic and cortical dopamine receptor availability may contribute to such baseline-dependent drug effects on cognitive control. Further, prior pharmacogenetic studies suggest that effects of cholinergic drugs, such as nicotine, could also depend on putative differences in dopamine signalling. For example, subjects who are homozygous for the C allele of the DRD2 SNP rs6277 polymorphism, which presumably increases striatal dopamine signalling (Hirvonen et al., 2009; Smith et al., 2017), showed improved distractor resistance after nicotine (Ahrens et al., 2015). Reduced distractor interference after nicotine administration was also observed in carriers of the A1 allele of the DRD2/ANKK1-TaqIa polymorphism, which leads to lower D2 receptor densities but increased striatal dopamine synthesis (Gilbert et al., 2005; Laakso et al., 2005; Ritchie and Noble, 2003). Nicotine was also shown to modulate resting-state alpha frequency depending on COMT genotype (Bowers et al., 2015). Hence, nicotine seems to increase cognitive stability in subjects with genetically increased striatal dopamine signalling, and it may be speculated that it rebalances fronto-striatal dopamine systems from a flexible to a more stable state. However, increasing dopaminergic neurotransmission with L-tyrosine experimentally yielded the opposite pattern, that is, nicotine induced increases in cognitive flexibility and decreases in stability. Such a discrepancy has recently been reported by Jongkees et al. (2017, 2019) who found L-tyrosine administration but not the COMT polymorphism to impact on the effects of transcranial direct current stimulation on working memory. The magnitude of changes in neurotransmitter levels might be one reason for such a difference between state (i.e. experimental manipulation) and trait (i.e. genotype-dependent baseline) effects on behaviour. Another reason could be the specific modulation of D1 or D2 receptors by polymorphisms of dopaminergic neurotransmission: They are either more related to D2 receptors and striatal functions (DRD2 SNP rs6277 or DRD2/ANKK1-TaqIa polymorphism) or to dopaminergic signalling in prefrontal cortex dominated by D1 receptors (COMT polymorphism). In contrast, systemic L-tyrosine presumably modulates both receptor types and brain areas. Future human neuroimaging studies will need to identify whether L-tyrosine impacts stronger or to the same extent on brain networks implicated in cognitive stability and flexibility. Dissociations of brain networks implicated in dopaminergic modulation of cognitive versus motor flexibility have already been described (Stelzel et al., 2013).
Limitations
Given our sample size of 45, the data can only be seen as a preliminary evidence for the reported cholinergic–dopaminergic interaction. Future studies with larger samples are needed to reproduce and verify our findings. Further, the sample size did not allow us to consider individual differences in genotype which may already lead to differences in dopaminergic or cholinergic tone and signalling. Second, our systemic increase of dopamine availability is rather unspecific in terms of regional or receptor subtype specificity. Additionally, we chose a rather low dose of both drugs and did not adjust for body weight. Finally, the between-subject design may be prone to confounding interindividual differences in cognitive performance.
Conclusions
We here provide first preliminary evidence that experimentally increased dopaminergic signalling increases the effects of nicotine in a cognitive control task. Such combined stimulation of dopaminergic and nicotinic cholinergic receptors had opposing effects on cognitive stability and flexibility. Under increased dopaminergic signalling, nicotine seems only beneficial for flexible adaptation to new task demands. These findings may also be of relevance for nicotinic action in psychiatric disorders with changes in dopamine signalling such as schizophrenia or attention-deficit/hyperactivity disorder. For example, both patient populations show a higher rate of (heavy) smokers compared to healthy controls, and it has been suggested that smoking is used to self-medicate psychiatric symptoms and cognitive deficits (Dalack, et al., 1998; Kumari and Postma, 2005; Van Amsterdam et al., 2018). For both disorders, nicotinic receptors are discussed as potential targets for drug development (Potter et al., 2006, 2014; Tregellas and Wylie, 2019). Our data, however, suggest that improvements in cognitive flexibility with nicotine may come at the expense of increased distraction.
Supplemental Material
SupplementTables – Supplemental material for Increased dopamine availability magnifies nicotine effects on cognitive control: A pilot study
Supplemental material, SupplementTables for Increased dopamine availability magnifies nicotine effects on cognitive control: A pilot study by Stefan Ahrens, Joana Laux, Christina Müller and Christiane M Thiel in Journal of Psychopharmacology
Footnotes
Acknowledgements
The authors would wish to thank C Fiebach for advice on the paradigm.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by a grant from the German Research Foundation DFG TH766/8.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
