Abstract
Using data form a 14-day double-blind trial with 48 smokers randomized to either N-acetylcysteine (2400 mg) or placebo, we tested the effect of N-acetylcysteine on glutamate and gamma-aminobutyric acid concentrations in the dorsal anterior cingulate cortex and on smoking cessation. Smoking related behaviors and neurotransmitter concentrations in the dorsal anterior cingulate cortex were assessed before and after treatment. Forty-seven non-smoking males served as baseline controls. Smokers showed higher baseline glutamate but similar gamma-aminobutyric acid concentrations than non-smokers. There were no treatment effects on dorsal anterior cingulate cortex neurotransmitter concentrations, smoking cessation, craving, or withdrawal symptoms. These results confirm glutamate disbalance in smokers, but not efficacy of N-acetylcysteine.
Keywords
Substance dependence is associated with deviating glutamate and gamma-aminobutyric acid (GABA) concentrations in the dorsal anterior cingulate cortex (dACC). However, the literature is inconclusive on the direction of these neurotransmitter deviations (e.g. Durazzo et al., 2016; Schmaal et al., 2012). Pilot studies with N-acetylcysteine (NAC), a glutamatergic agent, show treatment promise (review: Deepmala et al., 2015). A first study (Schmaal et al., 2012) showed a direct effect of NAC on glutamate concentration in the dACC of cocaine dependent individuals. The aim of the current study was to test the effect of NAC on glutamate and GABA concentrations in the dACC in smokers and smoking cessation. Smokers were hypothesized to differ in dACC glutamate and GABA concentrations from non-smokers, and smokers receiving NAC were hypothesized to show normalization of these neurotransmitter concentrations and higher success-rates in smoking cessation.
Forty-eight male 15–55 year-old smokers (15+ cigarettes/day and Fagerström Test for Nicotine Dependence (FTND≥3) participated in a 14-day randomized, double-blind trial with 2400 mg/d NAC or placebo. Exclusion criteria were psychoactive medication, other mental and neurological disorders and magnetic resonance imaging (MRI)-ineligibility. MRI and psychological testing was done one day before and after treatment. Forty-seven matched non-smoking males served as baseline controls and only took part in the first assessment. Glutamate and GABA concentrations in the dACC were assessed with proton magnetic resonance spectroscopy (1H-MRS; for details see Waddell et al., 2007). Due to overlap in the spectral assignment of glutamate, glutamine and glutathione (Glx) concentrations are reported as proxy for glutamate. Breath CO concentration was assessed with a smokerlyzer. For questionnaire analysis, see Table 1 and 2. Our local Ethics Committee provided institutional review board approval. An extensive method description is available on request.
Baseline comparison of demographic, sample description measures and neurotransmitter concentrations.
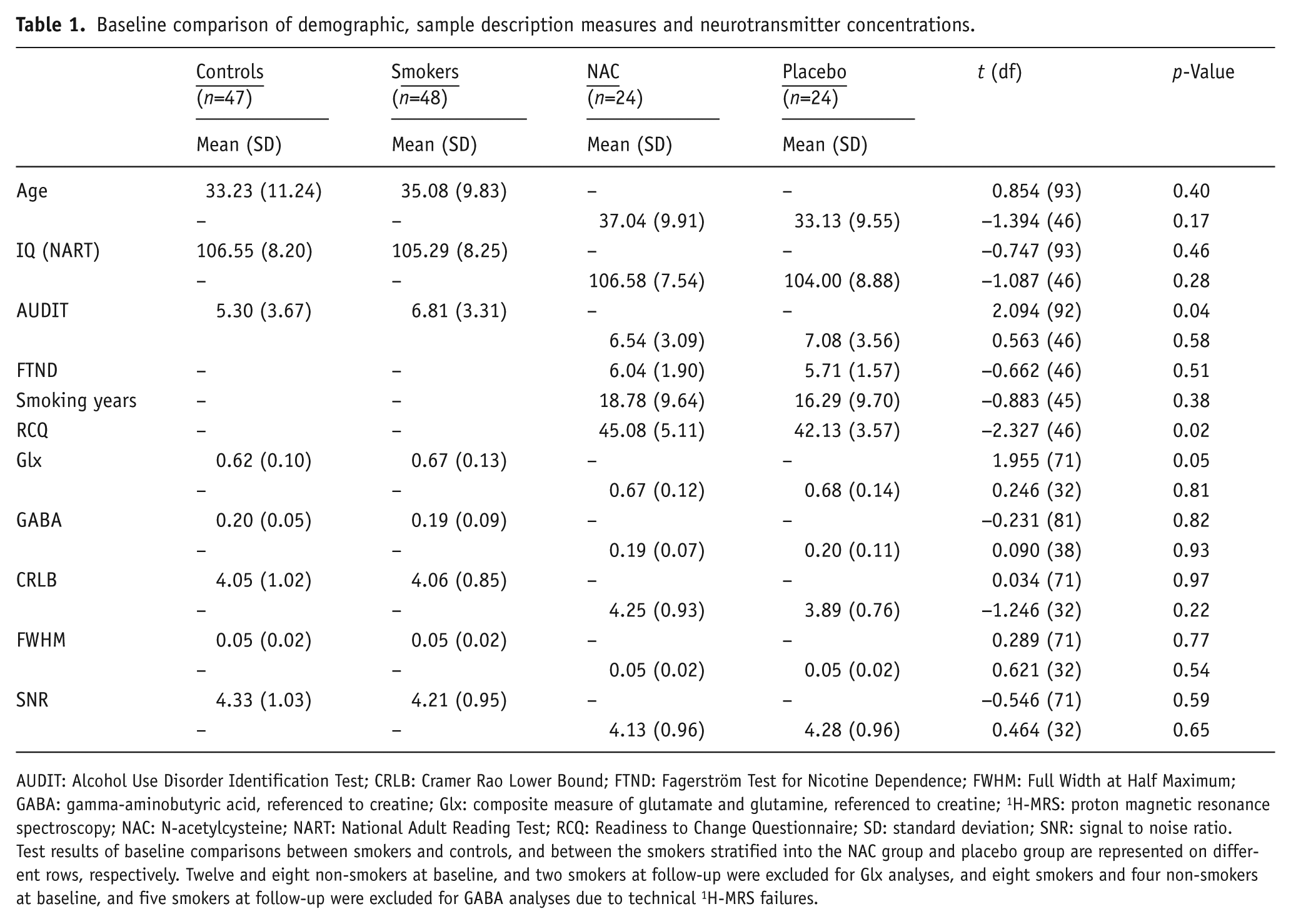
AUDIT: Alcohol Use Disorder Identification Test; CRLB: Cramer Rao Lower Bound; FTND: Fagerström Test for Nicotine Dependence; FWHM: Full Width at Half Maximum; GABA: gamma-aminobutyric acid, referenced to creatine; Glx: composite measure of glutamate and glutamine, referenced to creatine; 1H-MRS: proton magnetic resonance spectroscopy; NAC: N-acetylcysteine; NART: National Adult Reading Test; RCQ: Readiness to Change Questionnaire; SD: standard deviation; SNR: signal to noise ratio.
Test results of baseline comparisons between smokers and controls, and between the smokers stratified into the NAC group and placebo group are represented on different rows, respectively. Twelve and eight non-smokers at baseline, and two smokers at follow-up were excluded for Glx analyses, and eight smokers and four non-smokers at baseline, and five smokers at follow-up were excluded for GABA analyses due to technical 1H-MRS failures.
Effects of treatment on composite measure of glutamate and glutamine (Glx), referenced to creatine, and gamma-aminobutyric acid (GABA), referenced to creatine, concentrations, and smoking outcomes as measured by smoking behavior, craving, and withdrawal symptoms.
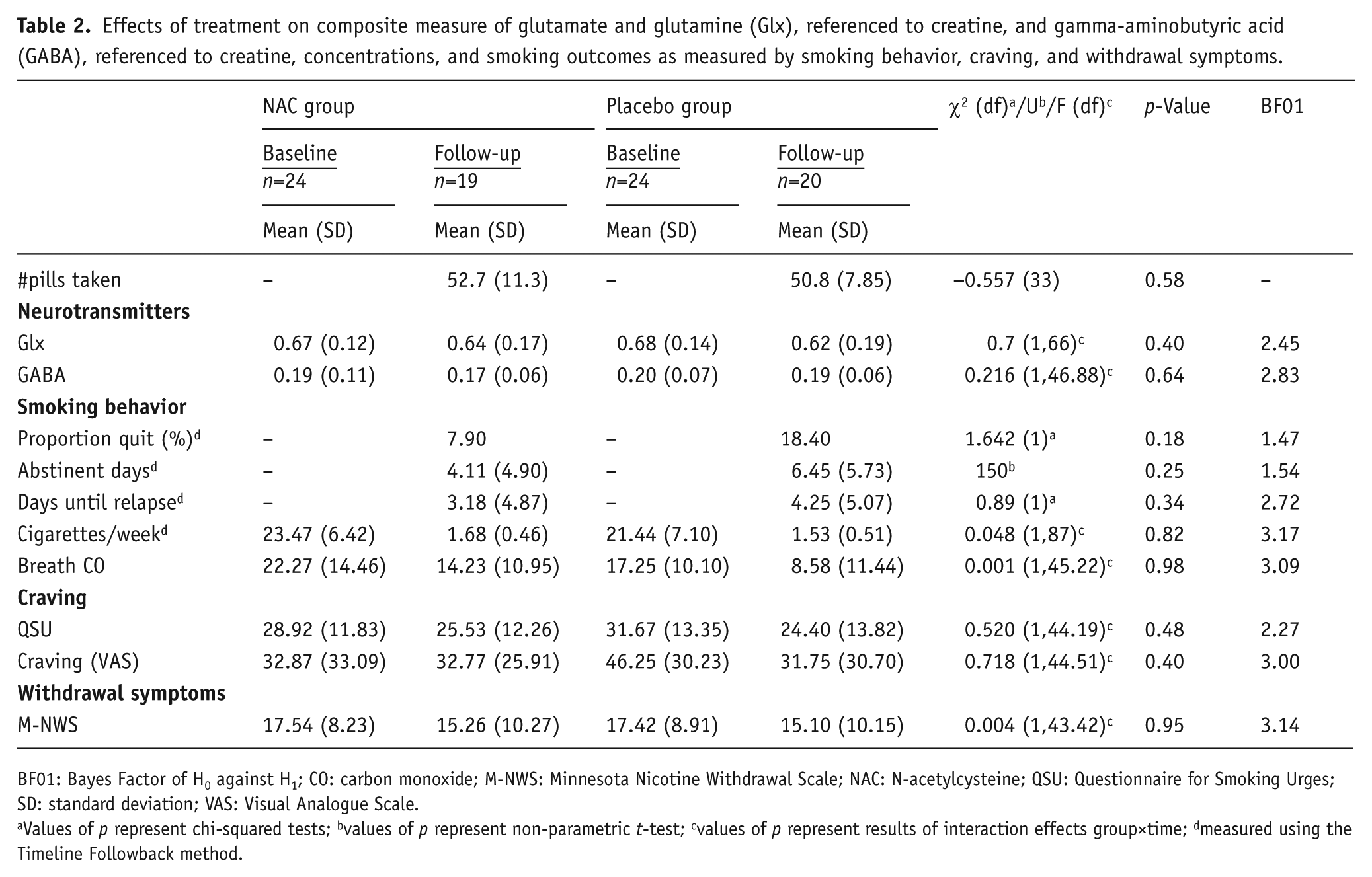
BF01: Bayes Factor of H0 against H1; CO: carbon monoxide; M-NWS: Minnesota Nicotine Withdrawal Scale; NAC: N-acetylcysteine; QSU: Questionnaire for Smoking Urges; SD: standard deviation; VAS: Visual Analogue Scale.
Values of p represent chi-squared tests; bvalues of p represent non-parametric t-test; cvalues of p represent results of interaction effects group×time; dmeasured using the Timeline Followback method.
Between-group differences were assessed with independent sample t-tests. Linear mixed model analyses were conducted to analyze treatment effects. Bayes factors quantified evidence for the null hypothesis.
There were no between-group differences in quality parameters of MRS spectra (Table 1) or medication compliance (Table 2). Smokers and non-smokers differed significantly in AUDIT-scores (Table 1), which was uncorrelated with Glx or GABA (p’s>0.50). Smokers had higher dACC Glx concentrations than non-smokers, but were not different on dACC GABA concentrations (Table 1). No baseline differences between smoking groups were detected, except for motivation to change (Table 2), which was entered as a covariate in analyses on treatment effects.
Thirty-eight smokers completed treatment. No adverse events were reported. For Glx and GABA, no main effect of time, group or time by group interaction was found (p’s>0.25). For both Glx and GABA, we had 80% power to detect a medium effect size of f=0.34 for a time by group interaction effect with α=0.05. However, the effect sizes we observed in the current study were small (Glx: f=0.28, GABA: f=0.23), and we did not have sufficient statistical power to detect such small effect sizes (Glx: 64%; GABA: 47%).
A main effect of time on weekly cigarettes smoked and breath CO concentrations (decrease, p’s<0.001) and a borderline significant main effect of group on breath CO concentrations (p=0.06) were observed, but no significant time by group interactions. For both craving measures (Visual Analogue Scale (VAS), Questionnaire for Smoking Urges (QSU)) a main effect of time was found (decrease, p’s<0.05), but no main effects for group and time by group (p’s>0.12). A trend significant main effect of time was found for withdrawal symptoms (p=0.08), but no main effect of group or time by group effect (p’s>0.80). Except for withdrawal symptoms, we had 80% power to detect medium effect sizes. The effect sizes that we actually detected were very small (f<0.10), and we did not have sufficient power to detect these effect sizes (power<17%). Bayes factors for all analyses provided anecdotal evidence for the null hypothesis (see Table 2, https://www.r-bloggers.com/what-does-a-bayes-factor-feel-like/).
Higher baseline Glx in smokers compared to non-smokers is in line with previous studies (Lee et al., 2007; Schmaal et al., 2012). However, in contrast to a previous study in cocaine dependent patients (Schmaal et al., 2012) NAC did not normalize dACC Glx. One explanation for this discrepancy is that our participants were actively smoking, whereas participants in the study of Schmaal et al. (2012) were abstinent from cocaine. NAC may still prove beneficial in a subgroup of smokers who are abstinent when starting NAC treatment (see McClure et al., 2014). However, because both treatment with NAC and placebo reduced smoking behavior and craving, we cannot draw conclusions on the specific effects of NAC on smoking outcomes. Major strengths are the presence of a non-smoking control group, and the simultaneous assessment of a range of neurobiological and clinical outcomes. The lack of assessment of additional medication to stop smoking is a limitation to this study. Finally, it should be mentioned that the applied 1H-MRS sequence was not optimized to differentiate between glutamate, glutamine and glutathione. In conclusion, even though higher baseline Glx concentrations were in line with previous results, the current study failed to replicate the normalization of dACC Glx concentrations or treatment effects of NAC.
Footnotes
Acknowledgements
The authors would like to thank Paul van Spiegel and Roos Blom from the Slotervaart Hospital in Amsterdam for their assistance in recruiting participants for this study.
Declaration of conflicting interests
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
