Abstract
The effects of propranolol in the treatment of anxiety disorders have not been systematically evaluated previously. The aim was to conduct a systematic review and meta-analysis of randomised controlled trials, addressing the efficacy of oral propranolol versus placebo or other medication as a treatment for alleviating either state or trait anxiety in patients suffering from anxiety disorders. Eight studies met the inclusion criteria. These studies concerned panic disorder with or without agoraphobia (four studies, total
Introduction
Propranolol, a ß1,2-adrenoreceptor antagonist, competes at receptor level with catecholamines, thereby blocking their orthosympathetic effects (Black et al., 1964). Clinically, propranolol is used widely to target peripheral sites of the noradrenergic system to treat hypertension, coronary artery disease and tachyarrhythmias (Freemantle et al., 1999; Fuster et al., 2006; Webb et al., 2010). Furthermore, propranolol can be deployed to block ß-adrenoreceptors in the central nervous system, as the lipophilic compound readily enters the blood–brain barrier.
Soon after the discovery of propranolol in the early 1960s, Turner and Granville-Grossman fortuitously noted its anxiolytic effects in an attempt to reduce tachycardia caused by hyperthyroidism (Turner and Granville-Grossman, 1965). Ever since, propranolol has gained increasing interest in psychiatry. Several trials studying off-label use of propranolol would follow, such as its use in the treatment of high trait anxiety (Becker, 1976; Kathol et al., 1980; Meibach et al., 1987; Wheatley, 1969), substance disorder and withdrawal symptoms (Grosz, 1972), schizophrenia (Yorkston et al., 1974), autism (Ratey et al., 1987), and aggression (Fleminger et al., 2006). In addition, propranolol has been shown to mitigate milder distressing states such as exam nerves (Brewer, 1972; Drew et al., 1985; Stone et al., 1973), stage fright (Brantigan et al., 1982), performance anxiety in musicians (Clark and Agras, 1991), performance anxiety in surgeons (Elman et al., 1998), and fear of undergoing surgery (Dyck and Chung, 1991; Jakobsson et al., 1995; Mealy et al., 1996).
Propranolol’s generic status, and the fact that selective serotonin reuptake inhibitors (SSRIs) have become first-line pharmacological treatment across the range of anxiety disorders (Baldwin et al., 2014), have probably contributed to a gradually declining attention for the agent as a potential treatment of anxiety-related conditions. More recently however, with advanced insights into the way the brain processes emotional experiences and their pivotal role in the development and persistence of several mental disorders (McGaugh, 2000), the psychopharmacological properties of propranolol have regained research attention (Johansen et al., 2011; Kindt et al., 2009; Soeter and Kindt, 2010).
Whereas propranolol was first studied as a general anxiolytic in the treatment of anxiety disorders, today it is mainly the amnesic effect on retrieved fear memory that is the subject of interest. To this end, there is evidence to suggest that after a fear memory is recollected, and followed specifically by a prediction error (a discrepancy between actual and expected negative events), propranolol selectively blocks protein synthesis, thereby prohibiting the ‘reconsolidation’ of the fear memory while sparing declarative memory (Debiec and Ledoux, 2004; Finnie and Nader, 2012; Kindt et al., 2009; Merlo et al., 2014, 2015; Nader et al., 2000; Sevenster et al., 2013). A recent meta-analysis of eight experiments with healthy human volunteers (total
Clinical evidence for the effects of propranolol in the treatment of anxiety disorders has never before been systematically reviewed. Accordingly, in an effort to determine the current place of propranolol within the therapeutic armamentarium of treatments for anxiety disorders, the aim of this study was to review both published and unpublished reports of randomised controlled trials (RCTs) that evaluated the effects of oral propranolol versus placebo or other medication as a treatment for alleviating state and/or trait anxiety in patients suffering from anxiety disorders. In addition, meta-analyses of pooled summary statistics were undertaken where possible.
Method
Systematic review
A systematic review was performed, which is reported in accordance with the PRISMA Statement (Moher et al., 2009).
Eligibility criteria
Only placebo-controlled, comparative parallel group and crossover RCTs were eligible when they included human subjects with any of the anxiety disorders as included in the current version (American Psychiatric Association, 2013) or previous versions of the Diagnostic and Statistical Manual (DSM) for an evaluation of the therapeutic effects of propranolol. Unpublished abstracts and reports were also considered. The comparator was either a placebo or other medication. The search excluded experimental fear conditioning trials and secondary prevention trials. There was no restriction on the basis of sample size, duration of follow-up, primary or secondary outcomes, duration or severity of symptoms, presence of comorbid disorders, or demographic variables of subjects. The search was not restricted to any language.
Information sources and search
An electronic systematic literature search, updated until 18 March 2014, was performed in the online databases: PubMED (all indexed years), Ovid Embase (Embase Classic and Embase 1947 to present), PsycINFO (1806 to present), Web of Science SCI-EXPANDED 1975–present [v.5.13.1], and the World Health Organization International Clinical Trials Registry Platform (WHO ICTRP) that includes unpublished reports, using the following strategy (see Table 1). When necessary, authors of included articles were contacted in order to retrieve summary continuous data that were not provided in their trial report.
Search terms.

Trial selection
During the primary screening process, two raters (SAS and RvW) independently assessed the information in the title and abstract of retrieved articles on their eligibility in a standardised but non-blinded manner. Thereafter, full text articles were retrieved and screened for eligibility. RCTs were included when both raters regarded all inclusion criteria (i.e. RCT, comparator agent, anxiety disorders) as fulfilled. Unpublished reports were also considered in order to rule out publication bias. There were no disagreements in assessment and inclusion. Figure 1 shows a flow diagram of the inclusion process.
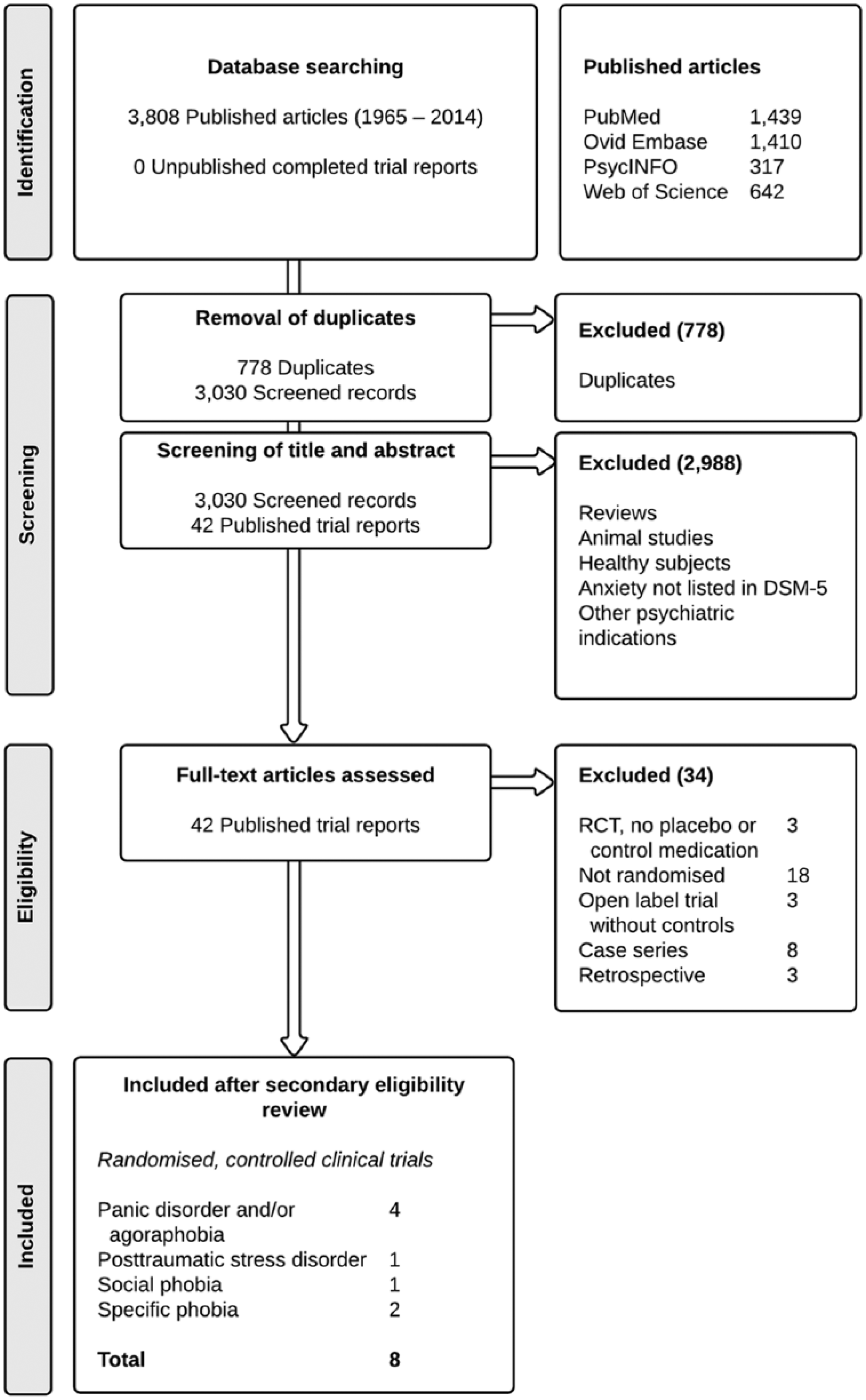
Flow of information through the different phases of the systematic search.
Data extraction and collection
One reviewer (SAS) extracted the data, and a second reviewer (RvW) checked the extracted items. The following information was obtained from each trial: (a) description of the trials, including the primary researcher, the year of publication, and the source of funding; (b) characteristics of the interventions, including the number of participants randomised to treatment and control groups, the number of total dropouts per group as well as the number that dropped out due to adverse effects, the dose of the medication and the period over which it was administered; (c) characteristics of trial methodology, including the diagnostic criteria employed; (d) characteristics of participants, including gender distribution and mean and range of ages; and (e) outcome measures employed (both primary and secondary), and summary statistics of reported continuous (means and standard deviations; SD) and dichotomous outcome measures (categorical treatment outcome). Summary outcome data were entered into Review Manager (RevMan software, version 5.2; Cochrane Collaboration, 2012). Eight authors were contacted for further information. Seven responded but none provided requested information or data.
Risk of bias
To ascertain the validity of eligible RCTs, two of the reviewers (SAS and AJvW) independently assessed the quality of the trials by means of the Cochrane Collaboration’s tool for assessing risk of bias (Higgins et al., 2011a). Disagreement in assessment was solved by discussion with a third person (AdJ).
Meta-analysis
Meta-analyses of pooled summary statistics were undertaken only if trials were combinable; i.e. if these included participants with the same anxiety disorder and if treatment effects in these trials were expressed in the same variable.
Data analysis and synthesis
For dichotomous treatment outcomes of interest, relative risks with 95% confidence intervals (95%-CIs) were used as the summary statistic. Results from continuous data were expressed as weighted mean differences (WMDs) with 95%-CIs. These data were pooled over studies using invariance weighting. Results were combined using the random effects model, in order to prevent substantially overstated precision of final estimates of effects even when statistical heterogeneity was low (
Sensitivity analysis
To explore the degree to which the findings of the meta-analysis could be affected by bias, sensitivity analyses were performed, when considered appropriate.
Results
Study selection
The initial search yielded a total of 3030 citations after adjusting for duplicates (see Figure 1). After a primary screening process, 42 published trial reports (all in English language) were read full text for detailed examination. It appeared that 34 trials needed to be excluded after secondary review due to their study design (i.e. the full text article revealed that these trials were not randomised or that there was no comparator medication). Our eligibility criteria were met by a total of eight published trials of propranolol for the treatment of panic disorder with or without agoraphobia (four studies,
Study characteristics
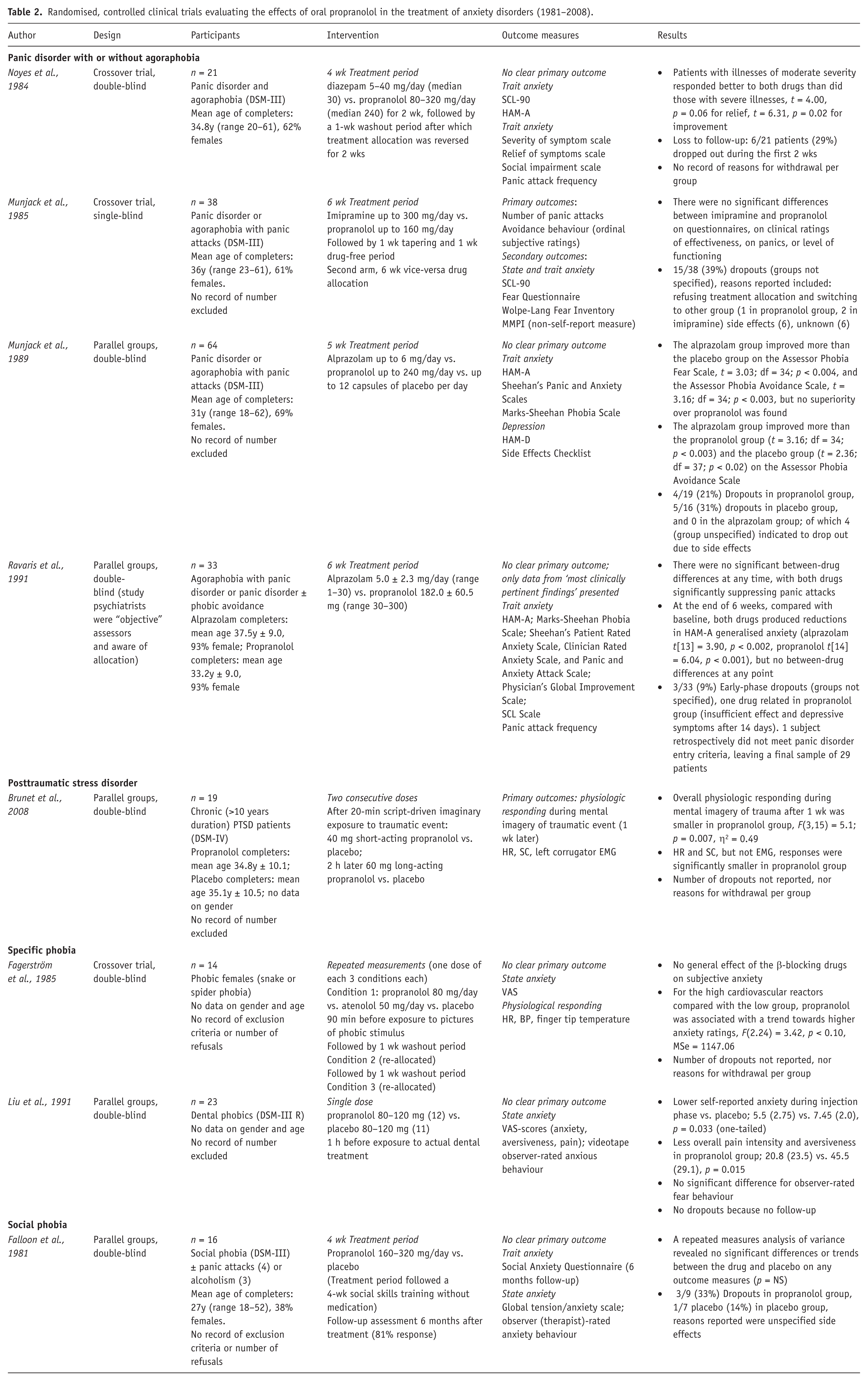
The characteristics and results of included trials are summarised in Table 2.
Randomised, controlled clinical trials evaluating the effects of oral propranolol in the treatment of anxiety disorders (1981–2008).
Panic disorder with or without agoraphobia
Four trials found mixed effects of propranolol versus other medication in the treatment of panic disorder with or without agoraphobia (Munjack et al., 1985, 1989; Noyes et al., 1984; Ravaris et al., 1991). Three of these four studies (Munjack et al., 1989; Noyes et al., 1984; Ravaris et al., 1991) qualified for formal meta-analyses.
Posttraumatic stress disorder
Only one trial with chronic PTSD patients showed that 40 mg of short-acting oral propranolol given prior to imaginary exposure, followed by 60 mg of long-acting oral propranolol, statistically reduced physiological responding (reduced heart rate and skin conductance) during imaginary exposure 1 week later (
Specific phobia
One trial with specific (dental) phobia subjects was found (Liu et al., 1991). It showed that, as compared with placebo, 80–120 mg of oral propranolol significantly diminished self-reported state anxiety during injection of local dental anaesthesia (self-reported anxiety at injection: 5.5 (2.75) versus 7.45 (2.0),
Social phobia
One trial with social phobia subjects was found. It reported absence of statistically significant effects of propranolol on state and trait anxiety 6 months follow-up after a social skills training (Falloon et al., 1981).
Risk of bias
Study quality
Risk of bias judgements are summarised in Figure 2. The majority of trials (9/12; 75%) failed to provide complete outcome data. On average, 19 percent (37 out of 191) of the participants in the six trials that provided dropout data did not reach study endpoint; hence the risk of attrition bias is considerable. The authors of one trial reported that “data from only the most clinically pertinent findings” were presented in the paper, although they specified all outcome measures that were initially recorded (Ravaris et al., 1991). Therefore, this trial was judged to have a high risk of selective outcome reporting.
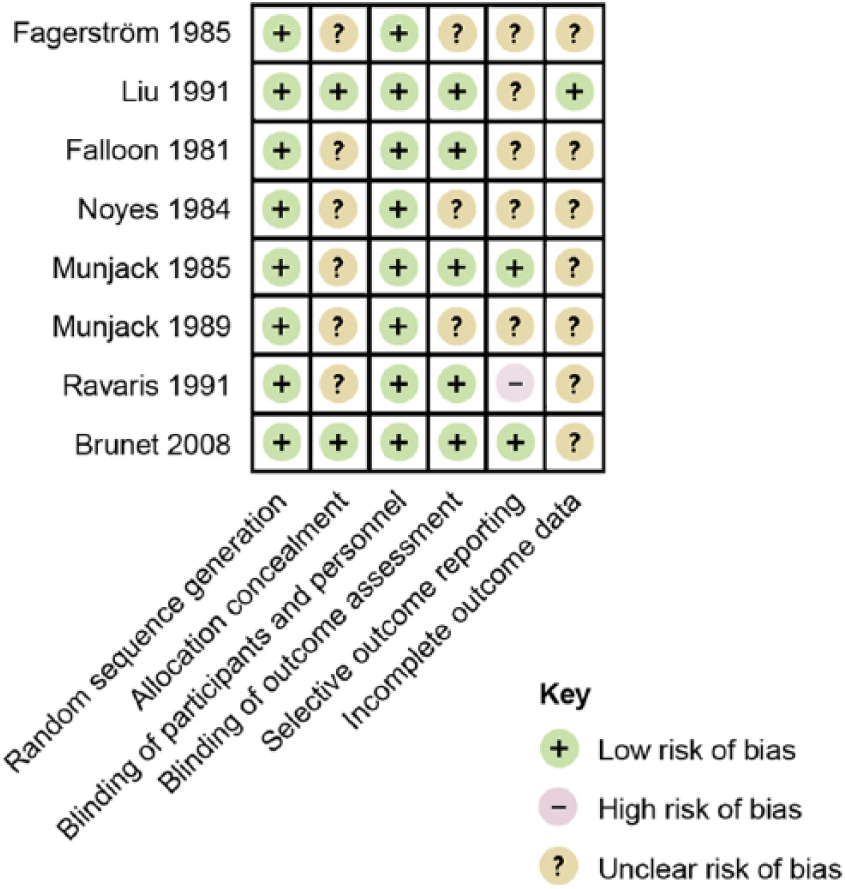
Risk of bias summary figure.
Publication bias
No unpublished reports of completed RCTs were obtained. Three RCTs were found (ClinicalTrials.gov Identifiers: NCT00645450, NCT00648375 and NCT01239173) that had been terminated because of inadequate subject recruitment. The authors of these three discontinued trials were contacted. All responded, but none provided data.
Meta-analyses
Propranolol versus benzodiazepines in panic disorder with or without agoraphobia
Meta-analysis I. Outcome: number of panic attacks after 2 weeks of treatment
Two trials report the effects of propranolol (30–320 mg/day) versus benzodiazepines (1–30 mg/day alprazolam or 5–40 mg/day diazepam) prescribed to adults with panic disorder with or without agoraphobia for a 2-week period, expressed in mean number of panic attacks after 2 weeks of treatment (Noyes et al., 1984; Ravaris et al., 1991). Effect size for each study was expressed as a WMD. There was substantial statistical heterogeneity of studies (two trials, 50 participants in total; χ2 = 3.04, df = 1,
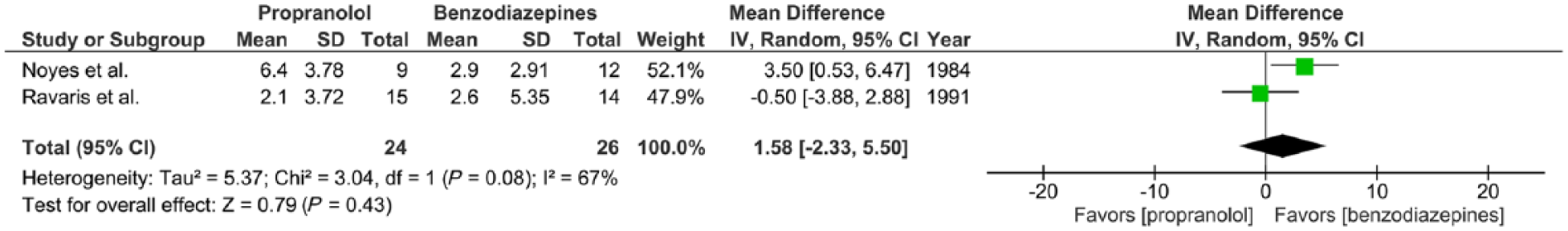
Meta-analysis I. Propranolol versus benzodiazepines in panic disorder with or without agoraphobia (outcome: number of panic attacks after 2 weeks of treatment).
Meta-analysis II. Outcome: HAM-A after 2 weeks treatment
Two trials report the effects of propranolol (30–320 mg/day) versus benzodiazepines (1–30 mg/day alprazolam or 5–40 mg/day diazepam) given to adults with panic disorder with or without agoraphobia for a 2-week period, expressed in mean HAM-A after 2 weeks of treatment (Noyes et al., 1984; Ravaris et al., 1991). WMDs were calculated for continuous summary data obtained from the Hamilton Anxiety Rating Scale (HAM-A), a widely used scale to assess the severity of symptoms of anxiety (Hamilton, 1959). There was substantial statistical heterogeneity of studies (two trials, 50 participants in total; χ2 = 7.11, df = 1,
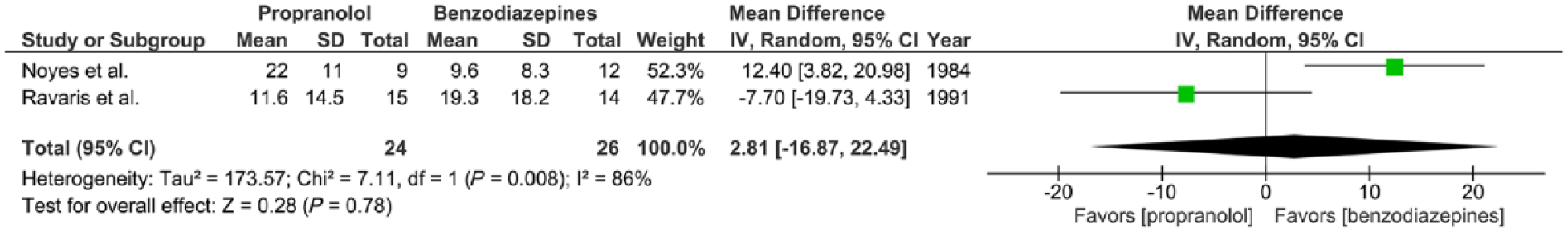
Meta-analysis II. Propranolol versus benzodiazepines in panic disorder with or without agoraphobia (outcome: HAM-A after 2 weeks treatment).
Meta-analysis III. Outcome: HAM-A after 2–6 weeks treatment
Three trials report the effects of propranolol (20–320 mg/day) versus benzodiazepines (0.5–30 mg/day alprazolam or 5–40 mg/day diazepam) given to adults with panic disorder with or without agoraphobia for a 2–6-week period, expressed in mean HAM-A scores at completion of treatment (Munjack et al., 1989; Noyes et al., 1984; Ravaris et al., 1991). There was substantial statistical heterogeneity of studies (three trials, 92 participants in total; χ2 = 7.74, df = 2,
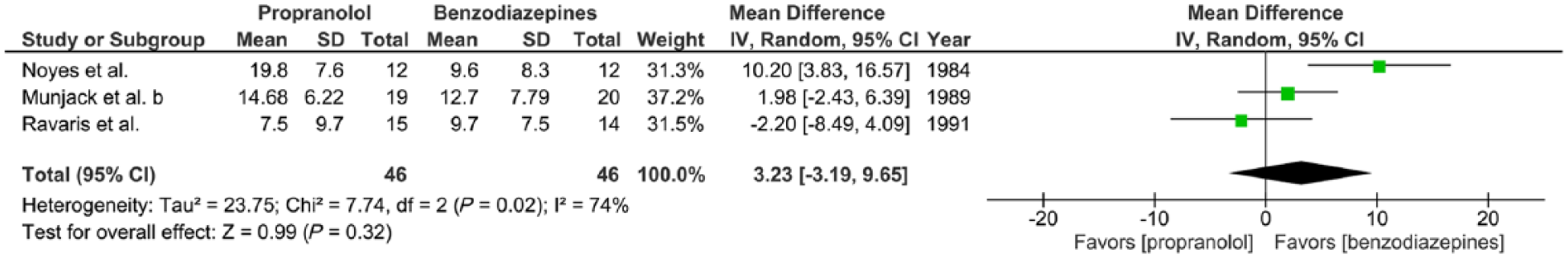
Meta-analysis III. Propranolol versus benzodiazepines in panic disorder with or without agoraphobia (outcome: HAM-A after 2–6 weeks treatment).
Meta-analysis IV. Outcome: HAM-A after 6 weeks treatment
Two trials examined the effects of propranolol (30–300 mg/day) versus alprazolam (1–30 mg/day) given to adults with panic disorder with or without agoraphobia for a 2-week period, expressed in mean HAM-A after 6 weeks of treatment (Munjack et al., 1989; Ravaris et al., 1991). There was little statistical heterogeneity of studies (two trials, 68 participants in total; χ2 = 1.14, df = 1,
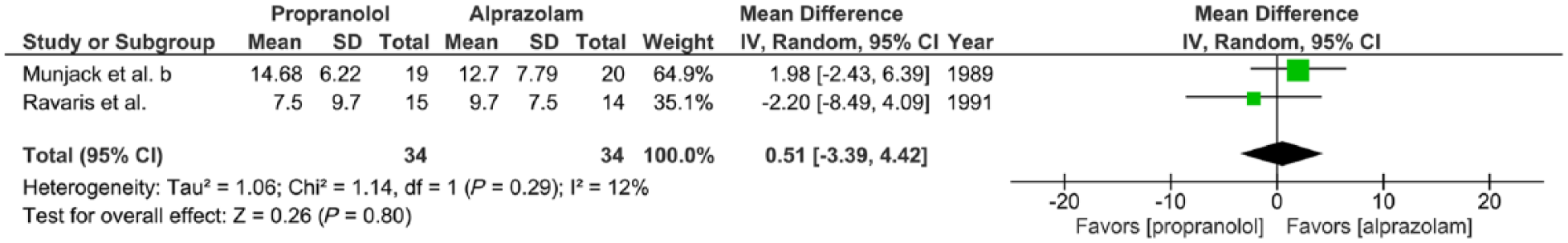
Meta-analysis IV. Propranolol versus alprazolam in panic disorder with or without agoraphobia (outcome: HAM-A after 6 weeks treatment).
Sensitivity analysis
For one study, included in all four meta-analyses, a high risk of selective outcome reporting was found (Ravaris et al., 1991). The impact of this bias on the findings of our meta-analyses was assessed. After excluding this study only one study remained for meta-analyses I,
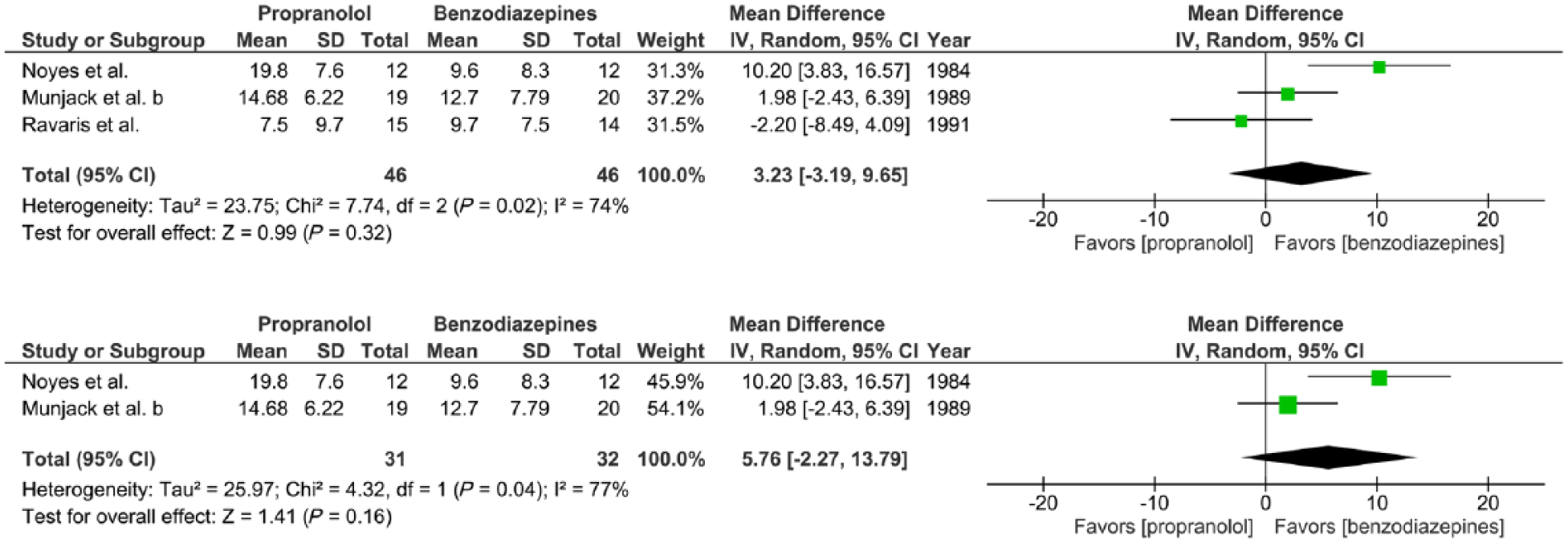
Sensitivity analysis. Meta-analysis III with one study with high risk of selective outcome reporting included and excluded (Ravaris et al., 1991).
Discussion
This systematic review and meta-analysis shows a lack of well-designed clinical studies. This limits the scientific evidence and allows neither firm conclusions in favour or against the use of propranolol in the treatment of anxiety disorders, nor recommendations for informed decision-making in clinical practice. More specifically, our meta-analyses found no statistical difference between the effects of propranolol and benzodiazepines on anxiety and panic attack frequency (Munjack et al., 1989; Noyes et al., 1984; Ravaris et al., 1991). In addition, four moderate risk of bias trials failed to show solid evidence on the therapeutic effect of propranolol in patients with dental phobia (Liu et al., 1991), animal-type specific phobia (Fagerström et al., 1985), and social phobia (Falloon et al., 1981). No RCTs were available on the effects of propranolol in the treatment of any of other anxiety disorders (e.g. generalised anxiety disorder, obsessive–compulsive disorder (OCD), separation anxiety disorder, or selective mutism; note that PTSD and OCD have been relocated to separate chapters in the DSM-5 (American Psychiatric Association, 2013). Moreover, despite widespread suggestions (Gardner, 2010; Giles, 2005; Lehrer, 2012), no evidence was found for the effects of propranolol on PTSD symptom severity, through inhibition of memory reconsolidation (Brunet et al., 2008) or any other mechanism.
To date, statistical equivalence of the efficacy of propranolol versus benzodiazepines regarding the treatment of individuals with panic disorder with or panic disorder without agoraphobia has not been shown. Because the evidence converges to suggest that propranolol and benzodiazepines prescribed in clinical settings have similar effectiveness in the short-term treatment of these conditions (Munjack et al., 1989; Noyes et al., 1984; Ravaris et al., 1991), other factors also need to be considered. First, it takes time for an effect to become prominent upon administration. SSRIs generally require a period of 2–4 weeks, while in some patients with panic disorder the onset of action may take up to 12 weeks (Michelson et al., 2001; Oehrberg et al., 1995). Therefore, early adjuvant therapy with propranolol may be taken into consideration. Second, the side effects profile should be taken into account. Whereas the clinical effects of benzodiazepines are considered equivalent to the first-line pharmacotherapy of panic disorder (SSRIs; Mitte, 2005; Roy-Byrne et al., 2013; Wilkinson et al., 1991), they carry a high risk of unwanted sedative effects, cognitive impairment and dependence, and tolerance will develop over time (Baldwin et al., 2014). Conversely as compared with benzodiazepines, the side effects of SSRIs are temporary, reversible and relatively benign, albeit still considerably frequent (among others, >10% gastrointestinal complaints, fatigue, insomnia, and headache; Sandoz, 2012). Although side effects of propranolol occur less frequently (among others, 1–10% sleeping disturbances, nightmares, transient fatigue, and cold extremities; Roy-Byrne et al., 2013; Sandoz, 2012), the extent of the supporting evidence base is currently broader and more consistent for SSRIs than for propranolol (Otto et al., 2001). Therefore, it would seem most reasonable not to divert from current treatment guidelines recommending SSRIs as the first-line medication for panic disorder (Baldwin et al., 2014) until robust data regarding the comparative efficacy and tolerability of propranolol versus SSRIs become available.
With regard to the therapeutic effects of propranolol, it has been proposed that propranolol’s anxiolytic properties may result from its peripheral (autonomic) rather than its central activity (Balon et al., 1990; Clark, 1986; Roy-Byrne et al., 2006). This may explain the lack of evidence for propranolol’s efficacy in the long-term treatment of anxiety disorders other than panic disorder. To this end, it seems most likely that propranolol aids in breaking a vicious cycle of anxiety in which catastrophic misappraisal of bodily sensations of orthosympathetic origin, such as palpitations or increased ventilation, fuel the occurrence of panic attacks. This explanation is supported by research that found that subjects suffering from high levels of general trait anxiety improved little on propranolol (Becker, 1976; Kathol et al., 1980; Meibach et al., 1987; Wheatley, 1969), whereas more favourable effects were found in the treatment of performance anxieties, in which enhanced sensitivity for adrenergic hyperactivation may similarly initiate the fear response (Brantigan et al., 1982; Brewer, 1972; Clark and Agras, 1991; Drew et al., 1985; Elman et al., 1998; Stone et al., 1973).
The present systematic review was limited by the moderate number of small studies examining the effects of propranolol on anxiety disorders, and by the risk of bias these trials presented. Notably, the average loss to follow-up was nearly one-fifth of all participants. As withdrawal reasons were seldom reported, the possibility of selective loss to follow-up in some studies could not be ruled out.
In conclusion, the quality of evidence for the efficacy of propranolol at present is insufficient to support the routine use of propranolol in the treatment of any of the anxiety disorders.
Footnotes
Acknowledgements
The authors would like to thanks Joost G Daams, MA, clinical librarian at the Academic Medical Centre of the University of Amsterdam, for helping constitute adequate search terms for the electronic database search.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
