Abstract
Background:
A dearth of evidence exists on how to include children and young people in palliative care research.
Aim:
We aimed to identify successful practices in involvement, recruitment and data collection with children and young people with life-limiting illness in research.
Design:
We synthesised methods from five primary studies from three geographical regions in which children with life-limiting conditions were recruited and interviewed. Using Expert Elicitation Methodology we identified successful practices in the three areas of involvement, recruitment and data collection. We established consensus on methodological challenges and solutions, and developed 10 recommendations for inclusion in research protocols.
Setting:
Primary cross-national research in three regions; Middle East (one study), sub-Saharan Africa (one study), Europe (three studies), reporting on studies that recruited N = 244 children aged 5–18 years.
Results:
Recommendations are: (1) research team supported by advisory group of children for entire research process; (2) appropriate distress protocol tailored to population; (3) opt not to use term ‘palliative care’ in study materials if significant distress is a risk; (4) be deliberate in purposive sampling to ensure diagnoses heterogeneity where appropriate; (5) age-appropriate information materials pre-tested by children; (6) clinical teams receive training in recruitment; (7) time to build rapport before starting data collection; (8) consider potential biases and advantages of having parent/carer present during interview; (9) use age-appropriate toys/games during interviews; (10) selfcare for researchers to manage distress.
Conclusions:
These recommendations can guide design and conduct of research, enabling children with life-limiting illness to meaningfully participate and express their views.
Keywords
Over 21 million children worldwide could benefit from palliative care each year, yet many do not receive it. The majority of research on children with life-limiting and life-threatening conditions is conducted with parents and other proxies, rather than with children themselves.
Children and young people have the right to be involved in research that is about them.
This paper provides successful practices for involving, recruiting, and collecting data with children and young people in paediatric palliative care research.
It identifies common challenges in conducting data collection with children with life-limiting and life-threatening conditions and offers applied solutions.
The operationalisation of solutions to methodological challenges in five international studies through discussion between authors, informs the development of generation of 10 useful practices for involving children and young people in the planning, design and conduct of palliative care research.
Through cross-national innovation in conducting robust research with children living with life-limiting and threatening illness, evidence will better reflect the concerns of children and young people and policy can be better aligned to their self-report priorities.
Introduction
It is estimated that annually 21 million babies, children and young people (hereafter ‘children’) would benefit from palliative care. 1 A systematic review of the evidence on what matters to children and young people with life-limiting and life-threatening illness (hereafter ‘life-limiting’) 2 found that most studies relied on proxy not self-report. 3 While the vast majority of need is in low- and middle-income countries, the majority of evidence originates from high-income settings. 4 The lack of robust child-centred research is a barrier to evidence-based service development in paediatric palliative care. 2
The United Nations Convention of the Rights of the Child 11 states a child’s rights to be involved in decisions that affects their lives and have their views listened to. Design and delivery of care that aligns with the needs of children requires their meaningful involvement in the research which shapes and informs their care. Challenges to participation of children with life-limiting illness include unpredictably of these health conditions, limited access to relatively small populations with common developmental delay, concern regarding participant burden, concerns among ethical review committees, gatekeeping from health professionals and families and perception of potential burden for children and their families of research participation.5–10 Although improvement in treatment and care requires the child’s voice in research, 11 the strategies for recruitment, data collection and involvement are rarely reported in the literature. 5 The aim of this paper is to order to identify successful practices that have been proven effective in enabling involvement, meeting recruitment targets and collecting data with children.
Methods
We present specific successful solutions and overarching recommendations for involving children and young people with serious illnesses in research, drawing on strategies used across primary five studies in three WHO regions which have successful recruited and collected data with children with life-limiting illness. We drew on adapted Expert Elicitation Methodology developed from ‘IDEA’ methods, 12 which involved an author of this paper to develop strategies to improve the design and conduct of research with family carers in palliative care, 13 and has also been applied in the field of palliative sedation.12,14–16
Data collection and procedure
We utilised the ‘IDEA’ (Identify, Discuss, Estimate and Aggregate) expert elicitation framework, which relies on the expertise and opinions of people with significant relevant experience and knowledge of a specified topic. 12 This approach is pertinent as the ‘D’ of IDEA (i.e. discussion) seeks to promote critical thinking and to share evidence and insights. As we did not seek to estimate probability, we did not conduct 12 the quantitative (aggregate) step of IDEA. To ensure that the contribution of experts was based in evidence, the exercise was rooted in peer-reviewed scientific literature that had successfully recruited and engaged children. 14
The identification phase was developed by EN and RH, who developed a proforma to elicit salient methodological innovation. The five primary studies were chosen because they had successfully involved, recruited and collected data with children and young people with life-limiting illness into primary studies studies. This proforama was refined by the wider expert team members. The pro-forma data extraction headings sought to answer the study aims, that is, involvement, recruitment and data collection. The expert panel also provided key study data for each of the primary research papers assessed: country, number of studies, reference, aim, study design, number of children recruited, age range, method of data collection, refusal rate, involvement practices, interviewers’ gender composition, age range versions of information sheets. Data analysis involved three rounds. First, responses to the three themes were collated by EN and RH and core thematic categories were identified under each theme. Then, the draft analysis was circulated to all experts, and themes (and adequate relevant detail within each to enable replication by others who wish to incorporate the learning in future research) were refined based on two rounds of feedback. The project was conducted 09/2023–03/2024.2,17–22
Results
Data sources
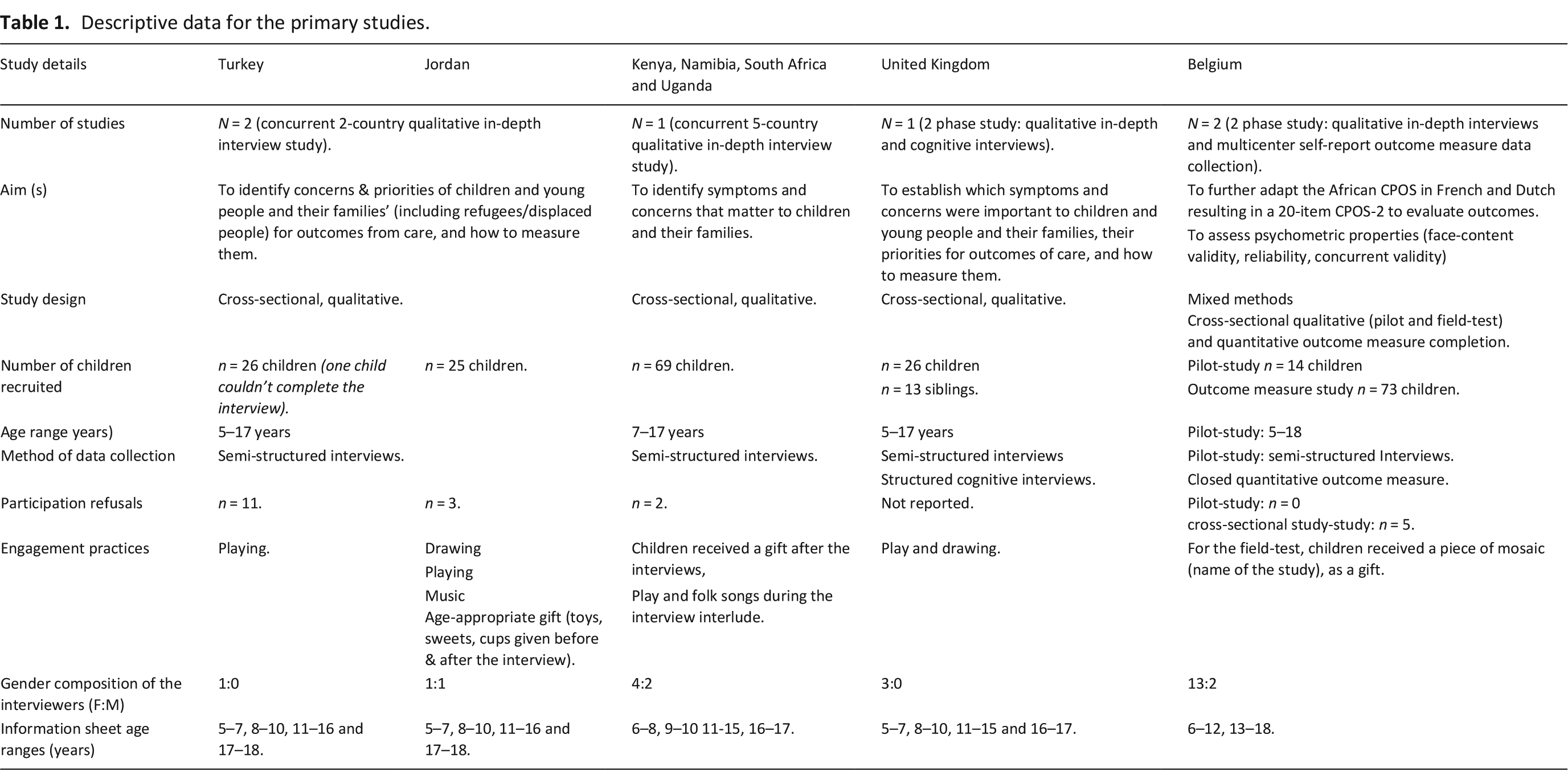
The studies were conducted in three geographical regions, Middle East (one cross national study across Jordan and Turkey),17,18 Africa (one cross national study across Kenya, Namibia, South Africa and Uganda),19,20 and Europe (two studies in Belgium and one across the United Kingdom).2,21 The studies each recruited n = 23–73 children and young people with life-limiting illness between the ages of 5–18 years (N = 244). Each study addressed the multidimensional symptoms and concerns of living with their diagnosis. Four of the studies used qualitative methods (semi-structured interviews) and one used a mixed method approach (see Table 1).
Descriptive data for the primary studies.
Ethics for the primary studies
The primary studies received local ethical approval: Kenya (KEMRI/RES/7/3/1), Namibia (17/3/3/EN), South Africa (HPCA 03/10), Uganda (UNCST SS2366), Jordan (18 KHCC 162), Turkey (16969557-25 or GO 19/40), Belgium (B403201837760), UK (HRA:19/LO/0033). Data were collected from 23 April 2019 to 29 July 2020 (Jordan). No unpublished primary study data was shared for this analysis.
The identified challenges and solutions experienced are detailed in Table 2 in the themes of (1) Involvement in planning and design of the research (i.e. child and public participation in the research), (2) recruitment and (3) data collection.
Challenges and solutions to involvement, recruitment and data collection and with children and young people.
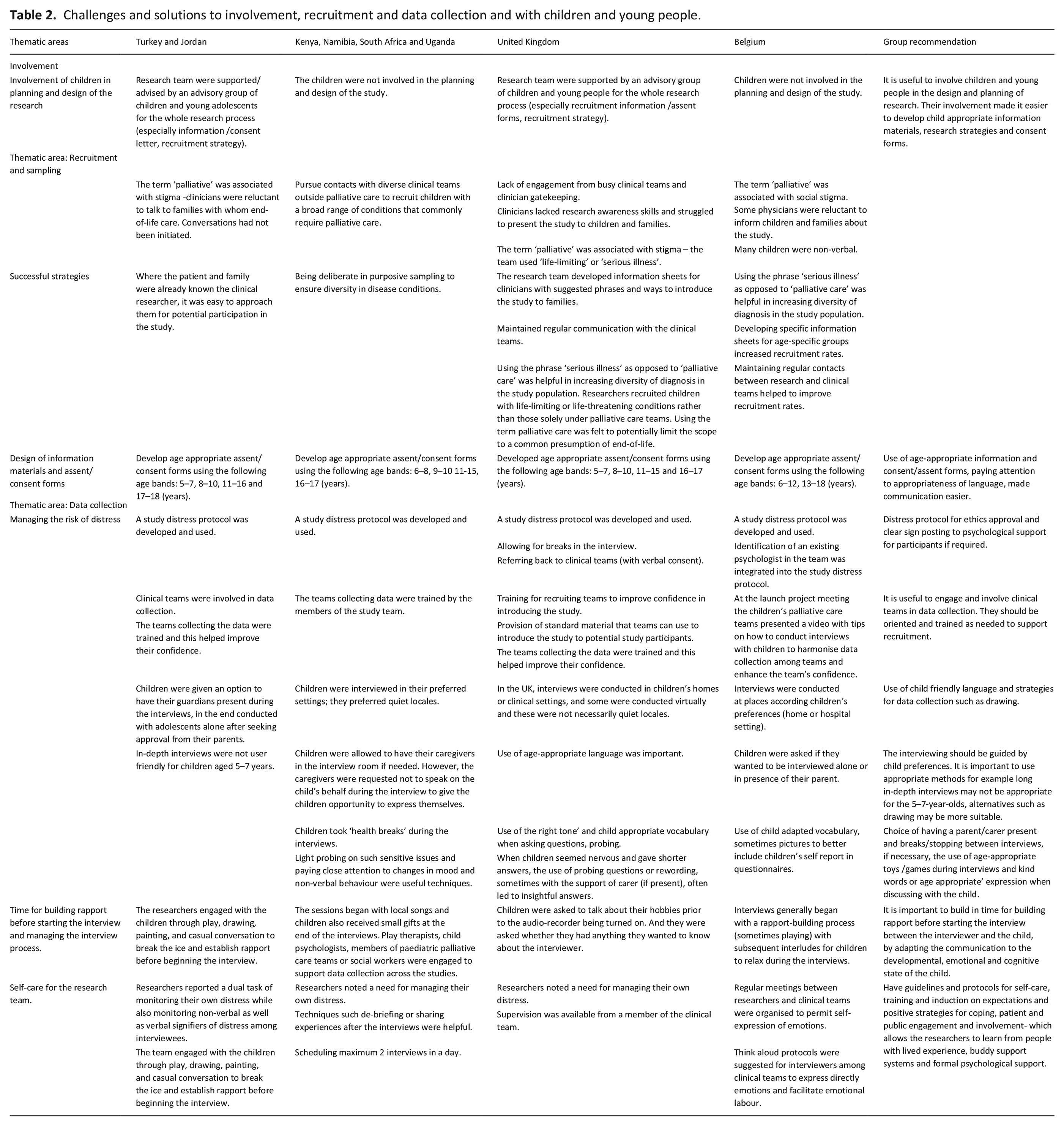
In summary, involving children, families and clinical teams in study design and conduct was described as essential. 23 Their input included the wording on study materials, child-led selection of images and naming of research outputs, recruitment processes and interpretation. However, it was also noted that clinician involvement led to successful recruitment as they were trusted by potential participants.
With respect to recruitment, in the UK and Belgium, not adopting the term palliative care and using the term ‘serious illness’ in inclusion criteria was helpful in increasing diversity of diagnosis in the study population. For example, in the UK researchers recruited children with a life-limiting conditions rather than those solely under palliative care teams, which allowed for involvement of diverse medical specialities and a focus away from solely end-of-life. Lack of prior experience among paediatric palliative care clinical teams in research recruitment meant that they were not confident in presenting the study to families. This was addressed by developing information sheets for clinicians with suggested phrases and ways to introduce the study to families. Regular communication between the clinical and research teams ensured that these concerns could be overcome. In Africa, 24 networking with a wide range of paediatric specialities for potential participants who may benefit from palliative care and therefore were appropriate for the study was very time demanding. Altruism was a driver for participation in Jordan, the authors noted that recognition that their data will be used for the benefit of those other patients like themselves increase children’s their propensity to participate in the study. It was also felt to be important to demonstrate to families a close working relationship between clinical and research teams to reduce suspicion that the data might be used for other research purposes, thus reducing reluctance to participate. If clinicians are relied on to approach families, teams found that asking the local/site investigator to nominate a specific clinician (or two) to be responsible for recruitment was useful. This gave the research team a dedicated contact in the clinical team. All studies found that families were very busy (often with additional sibling caring duties) and required sensitive and non-intrusive follow-up requests, with text messages being popular as they could be answered when an appropriate moment arose.25,26 In all instances, working with child development specialists, child and educational psychologists, play therapists and patient and public involvement on this issue was useful.
Lastly, data collection was largely conducted with an option to have their guardians present during the interviews. The role of caregivers accompanying the child during the interview varied. In some instances, caregivers responded to child-focussed interview questions themselves instead of playing a supportive role to help children respond. Conversely, parent carers also helped the child to remain focussed, and sometimes corrected information given by the child especially on the chronology of events. In some instances, researchers felt that caregivers influenced the child’s responses. For younger children, the use of play, art and drawing tailored to their areas of interest were commonly used to re-focus their attention and to keep them engaged in interviews. 27 Building rapport, trust and engagement was particularly challenging during the COVID-19 pandemic where some interviews were conducted online and children and their families were already experiencing social isolation. 28 This carries implications for future web-based data collection. 29 Interestingly, clinical staff identified benefits associated with having collected research data, for example, it rebalanced activities in that they received as well as gave information, and enabled learning from children and families.
Based on findings in Table 2, the researchers developed the following recommendations for involving, recruiting and collecting data with children with life-limiting illness (Table 3).

Recommendations for research recruitment, data collection and involvement with children and young people who have life-limiting and life-threatening illnesses.
Discussion
The recommendations are generated from successfully conducted and reported studies which have enabled children and young people to meaningfully participate in self-report research. This is important given that evidence for the development and use of health outcome measures with children suggests that children can meaningfully self-report alone from the age of 8. 30 Their participation is also in line with core ethical principles for conducting human research. 26
Collaboration with a child advisory group in research is essential for guidance on how to best meet their varying needs for involvement. Children’s input on the wording on study materials, child-led selection of images, recruitment processes and interpretation, made the planning and implementation of the research child-centred and therefore more inclusive and acceptable. The use of child-centered data collection methods such as playing, drawing, photography, observation and talking mats made research process more inclusive feasible and inclusive especially for younger children. 31 Given the heterogeneity of the population of children with life-limiting conditions and high prevalence of developmental delay it is important that data collection processes allow for more inclusive participation, by focussing on children’s capabilities, considering duration of interviews and the need to allow participants time to relax and play between interviews.31–33
The study further sheds light on the motive of ‘protection from harm’ that may drive gatekeeping (enacted by ethics committees, clinicians and caregivers). Key protocol components include ensuring core competencies among data collectors, ensuring capacity among participants with attention to verbal and non-verbal behaviours, distinguishing consent and assent, using developmentally appropriate language and materials and pragmatic protocols for managing adverse events such as distress. Further we note the importance of protecting researchers from harm, and note the evidence on researcher support in potentially distressing situations.34,35
Researchers delivering these studies highlighted concern associated with the use of the term ‘palliative care’, which may be misinterpreted by the patients and caregivers as ‘end-of-life’ care. Avoiding the use of the term palliative care where necessary may improve research recruitment, in the diverse clinical settings in which children receive their medical care. 36 Providing tailored information to the clinic teams to aid introductory conversations with potential participants is recommended. Recruiting children outside palliative care services increases the opportunity for children living with life-limiting illnesses to provide their views, especially given the many barriers to paediatric palliative care referral. 37 Clinical teams should also know the general content of the data collection instruments that children may be asked to complete.
Our study has several limitations. Firstly, the studies that inform our recommendations were conducted in eight countries from three geographical regions. This reflects diverse cultures and settings, but other regions and cultures may have additional successful practices to consider. Second, the analysis draws on protocols and expert views and as such may not have captured some methods and innovations that prospective research may address. Given that 98% of children with life-limiting illness live in low- and middle-income countries, 38 it is a strength that we included data from these regions but acknowledge that further countries have great need for palliative care research with children.
We note that, given the criticism that methods are not well reported in children’s palliative care research, 5 the solutions applied in the studies we report here were often not described in the original manuscripts. Going forward we urge all studies to fully report their methods of involvement, recruitment and data collection. 5
We propose essential protocol features and researcher practices strategies for meaningfully engaging and involving children and families in palliative care. These can stimulate the development of research and evidence that is more inclusive of children’s voices.
Footnotes
Acknowledgements
The authors thank the clinical teams who supported the recruitment, the children and young people and their families for participating in these studies.
Authors’ note
The data can be obtained by contacting the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The UK study C-POS study was supported by the European Research Council’s Horizon 2020 programme [Grant ID: 772635]; this article reflects only the author’s views and the European Research Council is not liable for any use that may be made of the information contained therein. The C-POS study wass supported by the National Institute for Health and Care Research (NIHR) Applied Research Collaboration South London (NIHR ARC South London) at King’s College Hospital NHS Foundation Trust. The views expressed are those of the author[s] and not necessarily those of the NIHR or the Department of Health and Social Care. EN and RH are supported by the NIHR (GHRUG NIHR134440) using UK aid from the UK Government to support global health research. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR or the UK government. The Turkey and Jordan study was funded by the UK Research and Innovation GCRF Research for Health in Conflict in the Middle East and North Africa (R4HC-MENA) project; ES/P010962/1. The authors have no financial relationships relevant to this article to disclose.
