Abstract
Background:
The number of young adults living with life-limiting conditions is increasing. This population requires palliative care responsive to their needs and preferences.
Aim:
To identify patient reported outcome measures developed, adapted and validated to assess the health outcomes of young adults (aged 18–25 years) living with life-limiting conditions. To examine their measurement properties and identify the most comprehensive, valid and reliable measures.
Design:
A systematic review and evaluation of measurement properties. PROSPERO ID (CRD42023443273).
Data sources:
MEDLINE; EMBASE; CINAHL; PsycInfo; AMED and Cochrane Library from inception to 03/07/2023. Searches were emented by hand-searching references.
Results:
Four thousand nine hundred twenty-two papers were identified. Five hundred and fifty-six full texts were assessed for eligibility. Thirty-five papers reporting 68 patient reported outcome measures were included. Most studies recruited young adults living with cancer (n = 29/35), we did not identify any studies with young adults living with complex neurodisability. Most measures (n = 61/68) were existing paediatric or adult patient reported outcome measures. Seven (n = 7/68) were young adult measures. Most were two-dimensional. The most assessed measurement properties were internal consistency, construct validity and structural validity. None of the measures were recommended for use as they did not meet sufficient criteria for content validity and internal consistency.
Conclusion:
There is a lack of multi-dimensional patient reported outcome measures for young adults living with life-limiting conditions, especially for non-cancerous conditions. Future studies may identify existing holistic measures developed for children or adults and adapt them for use with young adults. Studies should ask young adults and professionals about the relevance, comprehensiveness and comprehensibility of items.
Patient reported outcome measures can facilitate communication, allowing patients to report their needs, symptoms and concerns to healthcare professionals and providing an opportunity to address unmet needs, leading to improved care.
Young adults (aged 18–25 years) living with life-limiting conditions are a heterogeneous population, with a range of unique needs which are often unmet.
There is a lack of multi-dimensional patient reported outcome measures for young adults living with life-limiting conditions, especially those with health conditions other than cancer.
The measures identified have limited evidence of their psychometric properties.
Holistic patient reported outcome measures developed and validated for young adults with life-limiting conditions are needed.
Future studies may identify existing holistic measures developed for children or adults and adapt them for use in young adults. Collaborating with young adults, their family members, clinicians and researchers during the development of patient-reported outcome measures (PROMs) is essential to ensure the items specifically address the unique physical, psychological, social and spiritual needs of young people.
Clinicians play a vital role in ensuring patient reported outcome measures are relevant to real-world clinical settings, where they are used to support the care of young people by guiding treatment decisions and assessing well-being.
PROMs should undergo rigorous evaluation of their measurement properties following the COSMIN (COnsensus-based Standards for the selection of health Measurement INstruments) guidelines, which ensure the tools are scientifically robust, reliable and valid for use with young people.
Clear and consistent reporting of PROM development and validation studies, including transparent acknowledgment of limitations, is crucial. This allows clinicians and researchers to critically assess the quality of studies and select the most suitable and psychometrically sound measures tailored to young people’s needs.
Background
Young adults (aged 18–25 years) living with life-limiting conditions are a heterogenous population. 1 The population includes patients with childhood onset conditions who are transitioning into adult health services and may have cognitive and developmental delays 2 and young adults diagnosed with a life-limiting condition in adulthood who require care from adult services. 1
Internationally, the number of young adults living with life-limiting conditions or complex care needs is increasing, 3 but there is a lack of global epidemiological data. 4 This is further complicated by the inconsistent definition of the young adult age range between countries. 3 In England, the number of people aged 18–25 years diagnosed with a life-limiting condition increased from 18,522 in 2009/2010 to 25,766 in 2017/2018, an increase of nearly 40%. 5 Future projections estimate that by 2030 the prevalence of young people aged 14–25 years living with a life-limiting condition in England will be between 46.0 and 62.2 per 10,000, due to advances in medical technologies and improvements in the management of acute events. 5 In the UK, congenital, neurological and respiratory conditions are more prevalent in young adults aged 18–25 years than oncology diagnoses. 5 In Taiwan, the prevalence of life-limiting conditions in children and young adults (aged 0–25 years) increased from 45,311 in 2008 to 52,226 in 2017, with the highest prevalence in young adults aged 21–25 years, and oncology diagnoses being the most common. 6 International trends in the incidence of cancer among adolescents and young adults (aged 15–39 years) showed an increase in cancer cases across 23 countries. 7
The needs of young adults often differ to the needs of children and adults receiving palliative and end of life care. 8 They are living with a life-limiting condition at a time of ‘physical, emotional, social and cognitive’ development 8 meaning their needs are often broader than health, encompassing developmental needs including education, vocation, sexual relationships and living independently.8,9 Their illness may lead to ‘isolation and dependency’ on their families and healthcare services. 10 Young adults require age-specific palliative care that is responsive to their needs and preferences at the end of life. 11 Identifying unmet need is particularly important for young adults (aged 18–25 years) as they represent a ‘high-risk population who are underserved, with unique needs which are often unmet’. 12
Patient reported outcome measures are standardised questionnaires to assess patients’ symptoms, quality of life and wellbeing. 13 Patient reported outcome measures can facilitate communication between healthcare professionals, patients and family members.2,3 They allow patients to report their symptoms to health and social care professionals and provide an opportunity to address unmet needs, leading to improved care. 14 Professionals should consider the psychometric properties of measures 15 as well as factors such as diagnoses, geographical location, age, language, educational level and socioeconomic and cultural background of the target population when selecting outcome measures for use in research and clinical practice. 16
To the best of our knowledge, there is no systematic assessment of the psychometric evidence of patient reported outcome measures which assess the health outcomes of young adults living with life-limiting conditions. This systematic review aims to identify patient reported outcome measures developed, adapted and validated to assess the health outcomes of young adults (aged 18–25 years) living with life-limiting conditions, to examine their measurement properties and to identify the most comprehensive, valid and reliable measures.
Methods
This review was undertaken with a registered protocol (PROSPERO ID: CRD42023443273). This paper reports on review questions two and three in the registered protocol.
The review was carried out according to the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines. 17 It is reported in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines 18 (Supplemental Material File 1: PRISMA Checklist).
Searches
We searched MEDLINE; EMBASE; CINAHL (Cumulative Index to Nursing and Allied Health Literature); PsycInfo; AMED via Ovid and Cochrane Library from inception to 03/07/2023. Searches were supplemented by hand-searching reference lists of systematic reviews and included articles.
Search strategy
The search was developed in MEDLINE using text words and subject headings with input and support from a librarian at King’s College London with expertise in systematic searches in medical research databases. The search strategy was adapted from Knighting et al’s (2021) systematic review of respite care and short breaks for young adults aged 18–40 years with complex healthcare needs. 19 It follows the search strategy recommended by COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN). 17 No date limits were applied. The full search strategy can be accessed in Supplemental Material File 2: Search Strategy.
Eligibility criteria
The eligibility criteria were based on those used in previous systematic reviews.20,21 Original, full-text peer-reviewed research studies, reporting the development, adaptation or validation of a patient reported outcome measure for young adults (aged 18–25 years) living with a life-limiting condition were included. Detailed inclusion and exclusion criteria are shown in Table 1.
Inclusion/exclusion criteria.
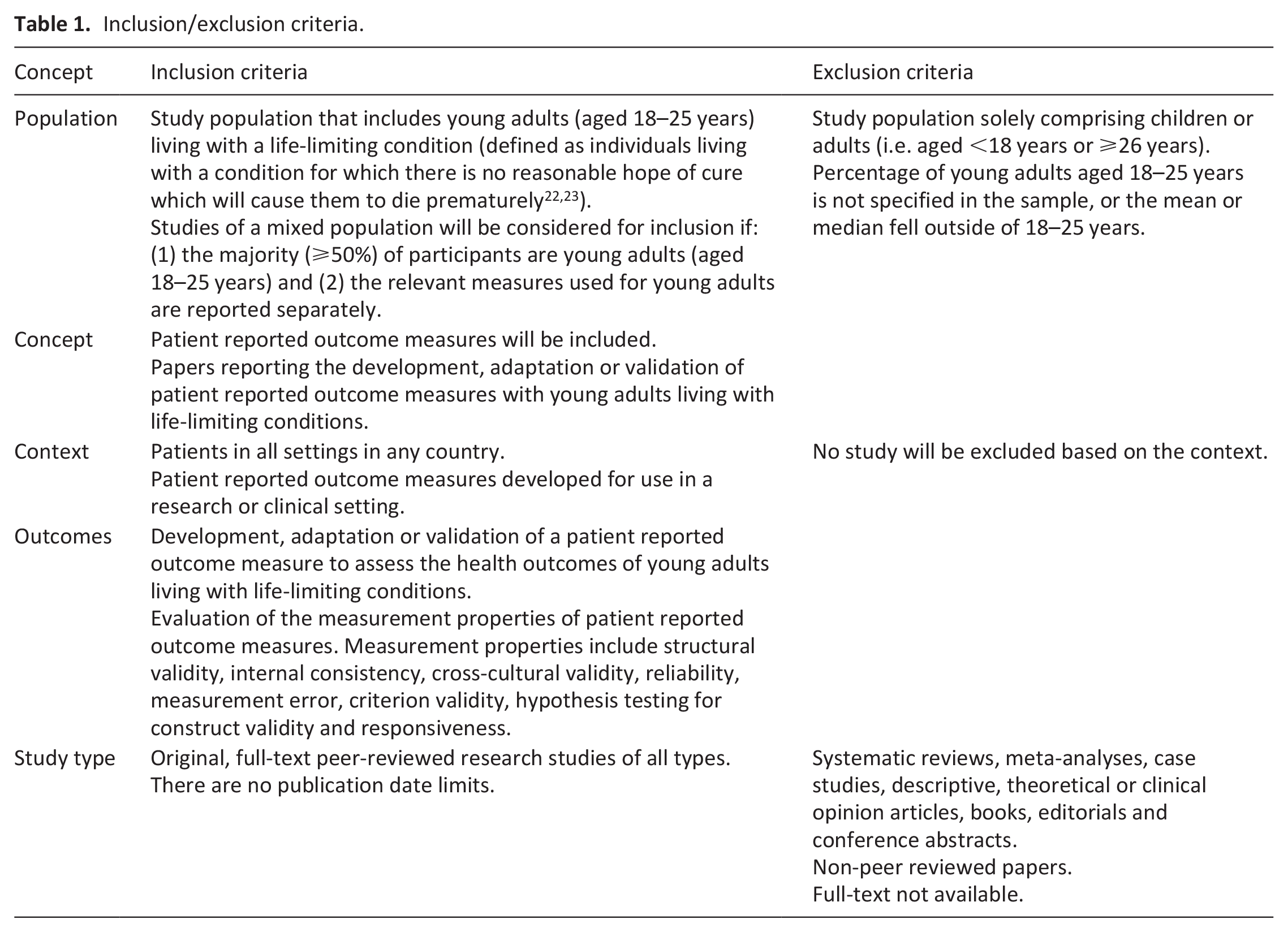
The World Health Organisation defines adolescence as those aged 10–19 years and young people as those up to the age of 25 years. 24 NHS England define young adults as those aged 18–25 years. This definition of young adults is based on biological evidence that the brain continues to develop until the mid-twenties,25,26 and people continue to develop socially and developmentally, as they transition from childhood to adulthood. 27 This definition of young adult (those aged 18–25 years) was used for our eligibility criteria.
Study selection
The search results were imported into Covidence (www.covidence.org). Duplicates were removed.
The first author (RLC) independently screened titles, abstracts and full texts against the inclusion criteria. A subset (20%) of papers were independently screened by a second reviewer (LKF). Conflicts were resolved by discussion.
Data extraction
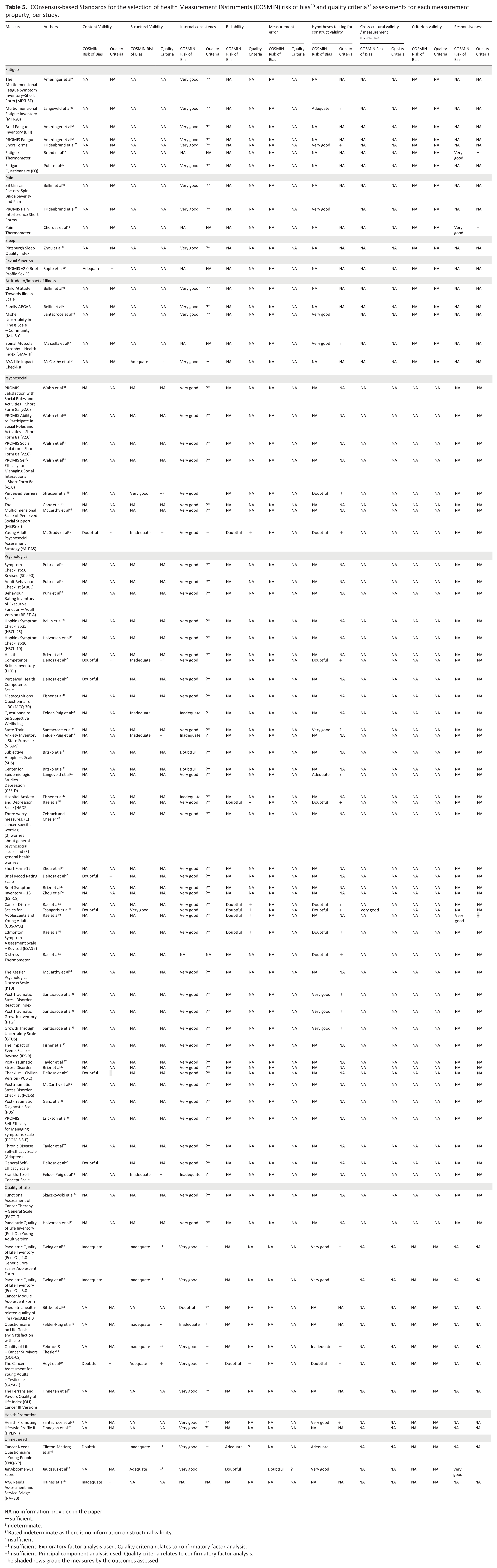
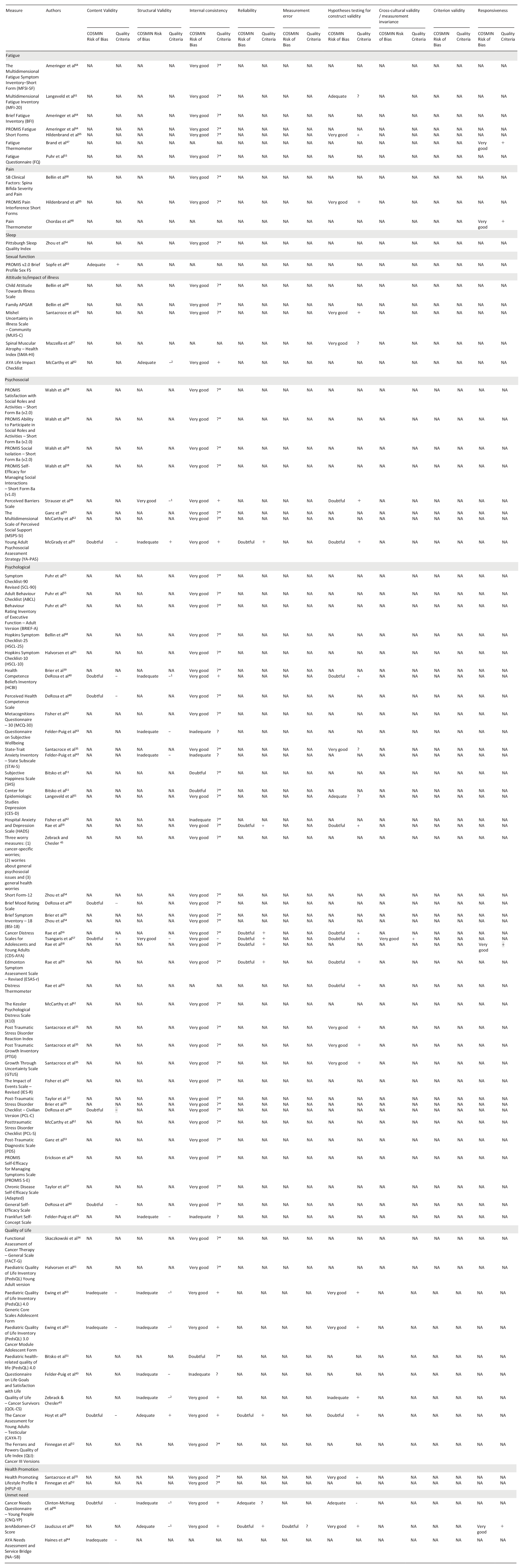
The first author (RLC) extracted data from each study into a standardised form (see Supplemental Material File 3: Data Extraction Form). Data extraction included: (1) study characteristics: author, year of publication, journal, geographic location, study design and characteristics of study sample (age, gender and disease); (2) characteristics of the patient reported outcome measure: patient reported outcome measure name, health outcome, mode of administration, recall period, length of instrument, number of items, response options, completion time, original language, available translations and validation methods used; (3) measurement properties including reliability, measurement error, criterion validity, structural validity, internal consistency, cross-cultural validity/measurement invariance, construct validity and responsiveness of the measure (see Table 2 for COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) definitions of measurement properties). 17
COnsensus-based standards for the selection of health measurement INstruments (COSMIN) definitions of measurement properties. 17

Information on interpretability of scores (distribution of scores, percentage of missing items, floor and ceiling effects and minimal important change/difference) were also extracted including information on feasibility of scores (patient/clinician comprehensibility, type and ease of administration, ease of standardisation, ease of score calculation, copyright, cost of instrument, required equipment, availability in different settings and requirements for approval). A second reviewer (MBH) independently checked the data extraction of all studies. Where it was unclear which measurement property papers were reporting on, the authors of this review made their own judgement based on the information provided. Conflicts were resolved by discussion.
Data synthesis
The domains of the patient reported outcome measures identified were mapped onto a conceptual framework developed by Namisango et al. 28 The framework consists of five categories: (1) physical, (2) psychological; (3) social; (4) spiritual and (5) other, based on the World Health Organisation’s definition of palliative care. 29
We qualitatively summarised the measurement properties of the measures used to assess the health outcomes of young adults (18–25 years) living with life-limiting conditions.
Assessment of methodological quality
We used the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) Risk of Bias checklist17,30,31 to appraise the methodological quality of studies. The checklist comprises standards for patient reported outcome measure development and measurement properties (content validity, structural validity, internal consistency, cross-cultural validity/measurement invariance, reliability, measurement error, criterion validity, hypotheses testing for construct validity and responsiveness). Each were rated as ‘very good’, ‘adequate’, ‘doubtful’, ‘inadequate’ or ‘not applicable’. If authors did not specify which aspect of validity or reliability they were evaluating, the researchers made a judgement based on the methods used. 32 The overall rating of each study was based on the ‘worst score counts’ principle. Two reviewers (RLC and MBH) independently assessed the methodological quality of included studies. Conflicts were resolved by discussion.
Next, the results of each study were rated against quality criteria developed by Terwee et al. 33 The results of each measurement property, per study, were rated as sufficient (+), insufficient (−) or indeterminate (?).
Recommending a patient reported outcome measure
After assessing the methodological quality of each of the studies we recommended patient reported outcome measures for use in accordance with COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines. 17 Measures were classified into three categories: (1) Category A: patient reported outcome measures with evidence for sufficient content validity (any level) and at least low quality evidence for sufficient internal consistency; (2) Category B: Measures categorised not in A or C and (3) Category C: Measures with high quality evidence for an insufficient measurement property. 17
Results
After de-duplication, 4386 titles and abstracts were screened, 556 full-text papers were reviewed. Citation and reference searching identified an additional 17 papers for inclusion in the review. Thirty-five papers met the inclusion criteria (see Figure 1).
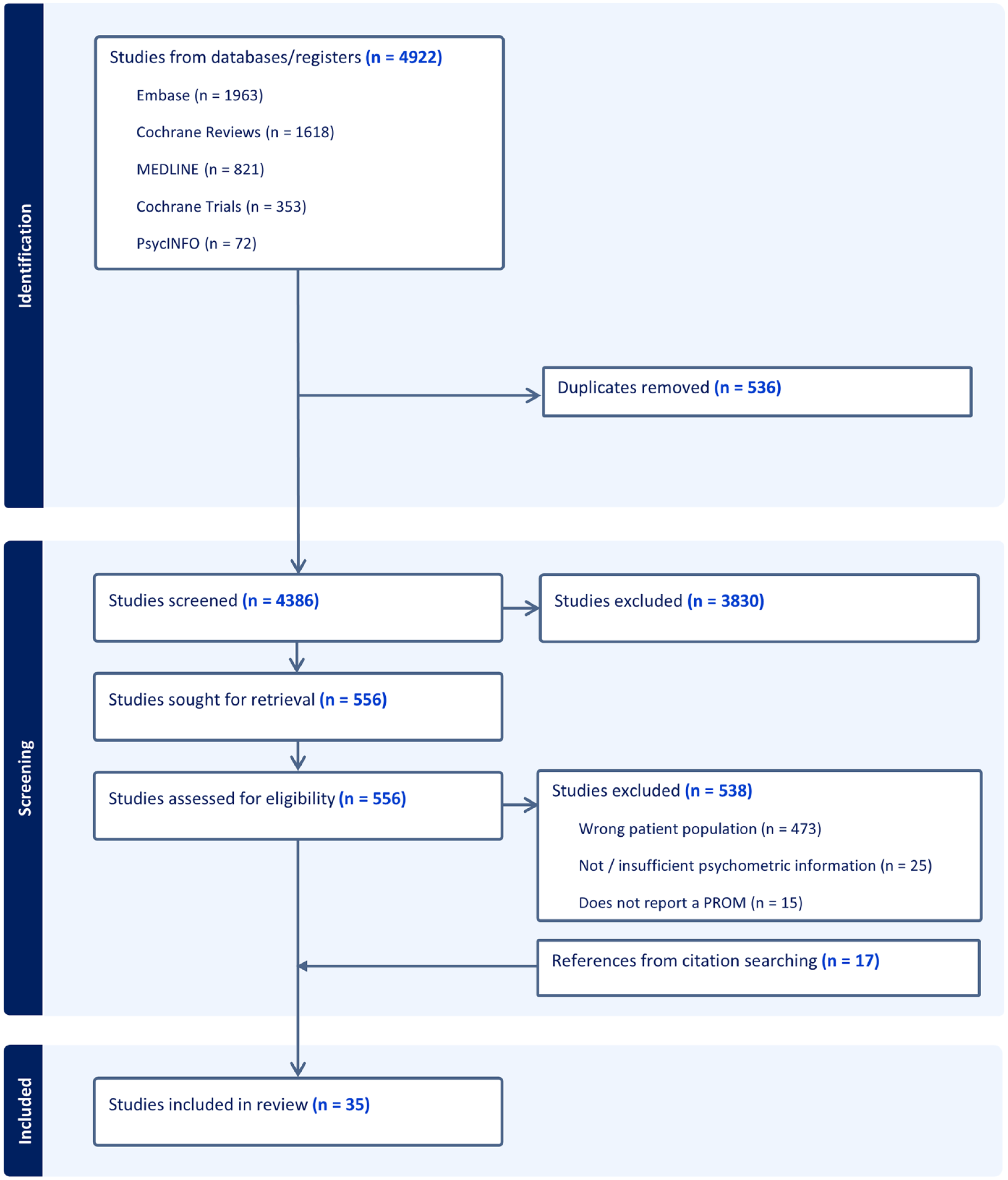
PRISMA flow chart.
The main reason for exclusion was wrong patient population, specifically studies with outside of our age range of 18–25 years. Our search identified papers that developed, adapted or validated patient reported outcome measures for ages that spanned children, adolescents and young adults living with life-limiting conditions. In these studies, the age range was often 0–25 years or 14–30 years. This encompassed the age range for inclusion in our study (18–25 years), however, they were excluded from this review.
Characteristics of included studies
Most studies included patients living with cancer (n = 29).34–62 One study included a mixed population, recruiting individuals living with cancer and blood disorders (n = 1). 63 Other populations included young adults living with sickle cell disease (n = 2),64,65 cystic fibrosis (n = 1), 66 spinal muscular atrophy (n = 1) 67 and spina bifida (n = 1; see Table 3). 68
Characteristics of included studies.
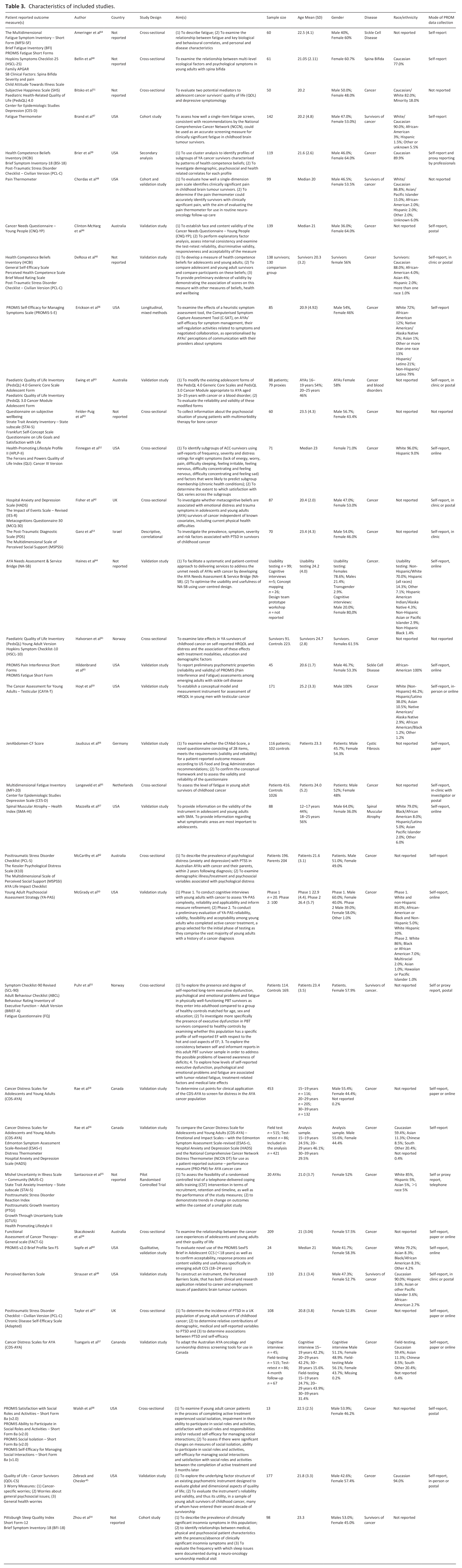
The population included in studies were predominantly from White ethnic groups. Sixteen studies did not report on race or ethnicity when describing the demographics of their sample. There was limited information on socioeconomic status including income, education and occupational status.
The 35 included studies reported the use of 68 patient reported outcome measures. Sixty one of these were existing paediatric, adolescent or adult patient reported outcome measures used and validated with young adults living with life-limiting conditions. Of these 61 measures, six were adapted or modified for use with young adults living with life-limiting conditions.35,44,56–58,62,63 Of the 68 measures identified, one measure was an existing young adult patient reported outcome measure . Of the 68 measures, 6 were new young adult patient reported outcome measures developed specifically for this population.40,46,49,50,59,66
Of the 68 patient reported outcome measures identified, 57 were generic measures and 11 were disease specific. Of these disease-specific measures most were developed for cancer populations (n = 8),34,45,46,52,56–59,63 followed by cystic fibrosis (n = 1), 66 spinal muscular atrophy (n = 1) 67 and spina bifida (n = 1). 68 Most measures were two-dimensional, some were unidimensional (see Table 4). A minority of patient reported outcome measures (n = 3)45,52,59 for young adults living with cancer were shown to assess all domains of palliative care (physical, psychological, social and spiritual domains) when mapped onto Namisango et al’s 28 conceptual framework (see Table 4).
Patient reported outcome measures, conditions and domains. 28
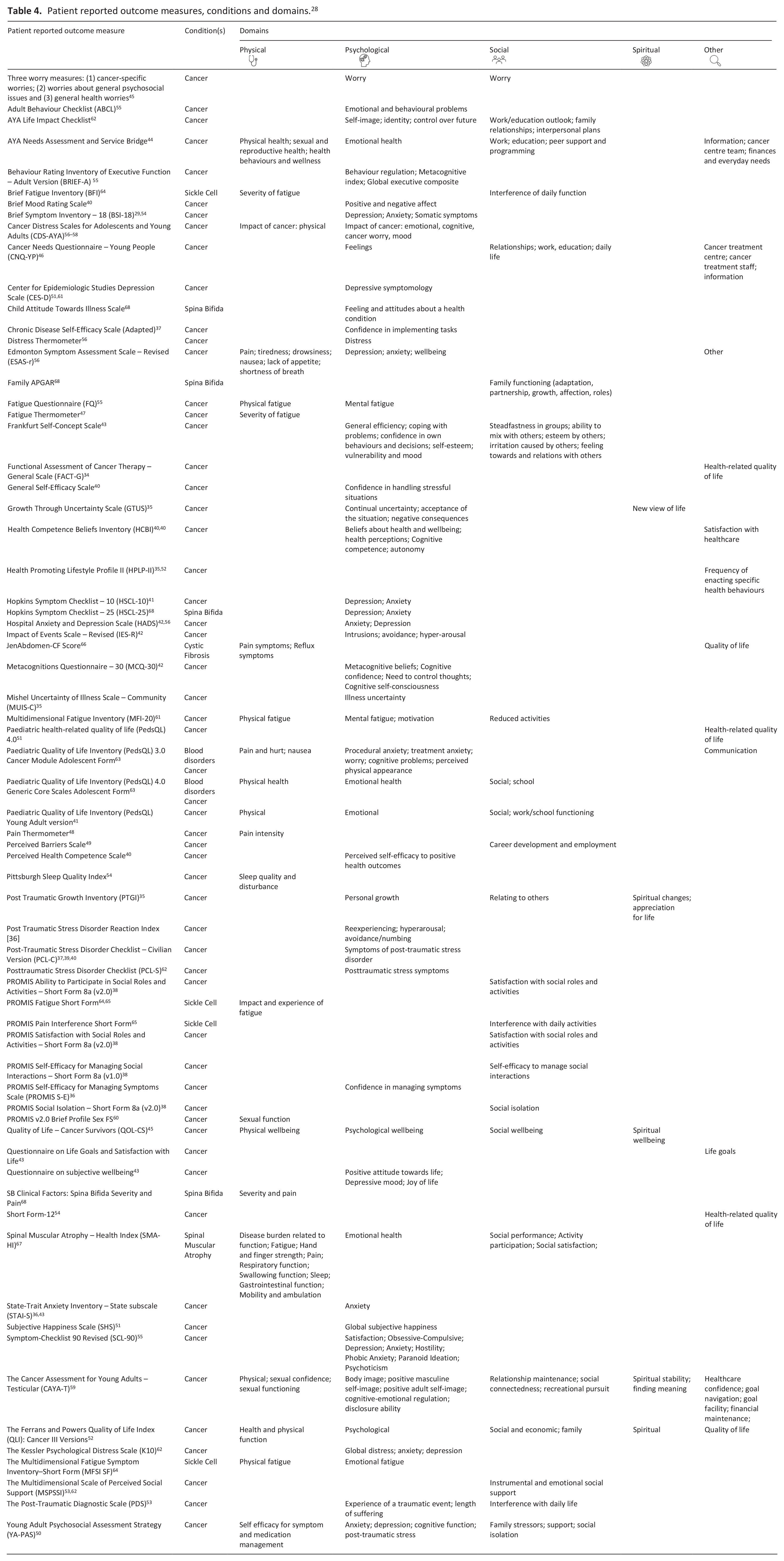
Most measures (n = 44) assessed psychological outcomes. Some measured multiple psychological domains including: mood (n = 17),35,39–43,45,50,51,54–58,61–63,68 cognition (n = 7),39,40,41,50,55–59,63 Posttraumatic Stress Disorder (n = 7),35,37,29,40,42,50,53,62 feelings towards their health condition (n = 6)35,36,39,40,56–58,68; body/self-image (n = 3)29,62,63; emotional fatigue (n = 3)55,61,64; distress (n = 2),56,62 behavioural problems (n = 2), 55 identity (n = 1), 62 control over the future (n = 1) 62 ; motivation (n = 1), 61 psychoticism (n = 1), 55 paranoid ideation (n = 1). 55
Some patient reported outcome measures (n = 23) measured physical symptoms, some measured multiple physical domains. Eleven measures (n = 11) assessed general physical health.41,44,45,50,52,56–29,63,66,67 Other measures assessed fatigue (n = 7),47,55,61,64,65,67 pain (n = 6)48,56,63,66–68 sexual/reproductive function (n = 3),44,59,60 sleep (n = 2),54,67 respiratory symptoms (n = 2),56,67 nausea (n = 2),56,63 tiredness (n = 1), 56 drowsiness (n = 1), 56 lack of appetite (n = 1), 56 and mobility (n = 1). 67
Some patient reported outcome measures measured social outcomes (n = 21). Six measures assessed general social outcomes (n = 6):35,41,52,59,63,67 interference with daily activities (n = 5) 46,53,61,64,65; social support (n = 3),44,50,53,62 satisfaction with social roles/activities (n = 3)38,67; education (n = 5),41,44,46,62,63 career/employment (n = 3),41,46,49 activity participation (n = 2),59,67 family functioning (n = 2),62,68 ability to manage social interactions (n = 3),38,43,59 social isolation (n = 1) 38 and worries about psychosocial issues (n = 1). 45
A minority of measures (n = 5) assessed the spiritual needs of young adults living with life-limiting conditions. Most of these measures asked about general spiritual needs (n = 3),45,52,59 others (n = 2) asked about the individual’s perception of life. 35 All measures that assessed spiritual needs were validated for young adults living with cancer.35,45,52,59
Measurement properties
Some measures had multiple research studies reporting on their measurement properties. They were the PROMIS Fatigue Short Form,64,65 The Multidimensional Scale of Perceived Social Support,53,62 Hopkins Symptom Checklist,41,68 Health Competence Beliefs Inventory,39,40 State-Trait Anxiety Inventory,43,64 Center for Epidemiologic Studies – Depression,51,61 Hospital Anxiety and Depression Scale,42,56–58 the Brief Symptom Inventory439,54 and the Cancer Distress Scales for Adolescents and Young Adults (CDS-AYA).56–59
It was not possible to quantitatively pool results as studies were heterogenous; studies evaluating the same tools collected data from respondents with different medical conditions or used different methodologies (e.g. randomised controlled trials vs. cross sectional designs). Therefore, the data for each study are reported separately,69,70 categorised by each patient reported outcome measure (see Supplemental Material File 4: Patient reported outcome measure characteristics).
Methodological quality
The methodological quality of the included studies is shown in Table 5. The following measurement properties were assessed in the included papers: content validity (n = 8), structural validity (n = 15), internal consistency (n = 73), test-retest reliability (n = 9), hypothesis testing for construct validity (n = 24) and responsiveness (n = 4). One study reported information on measurement error. 66 Measurement invariance was assessed by one study evaluating the Cancer Distress Scale for Adolescents and Young Adults (CDS-AYA). 57 Criterion validity was not reported in any of the studies included in this review. Most studies were limited by their small sample size which reduced the strength of the reported evidence. None of the studies reported information on all measurement domains
NA no information provided in the paper.
Sufficient.
Indeterminate.
Rated indeterminate as there is no information on structural validity.
Insufficient.
insufficient. Exploratory factor analysis used. Quality criteria relates to confirmatory factor analysis.
insufficient. Principal component analysis used. Quality criteria relates to confirmatory factor analysis.
The shaded rows group the measures by the outcomes assessed.
Content validity
Content validity is the degree to which the content of a patient reported outcome measure is an adequate reflection of the construct to be measured. 30 Eight studies reported the assessment of content validity. The methodological quality of most (n = 7) studies was considered ‘doubtful’. Of these, six studies consulted both healthcare professionals and patients on the relevance and understanding of the items included in the measure. In one study the methodological quality was considered ‘adequate’ for the components reported but they did not appear to consult healthcare professionals about the content of the patient reported outcome measure. Five studies assessed the measure for relevance, comprehensibility and comprehensiveness. Most provided little description of the qualitative methods used (e.g. whether skilled interviewers were used and their approach to data analysis).
Structural validity
Structural validity is the degree to which the scores of a patient reported outcome measure are an adequate reflection of the dimensionality of the construct to be measured. 30 Fifteen studies assessed this domain. Methodological quality was rated as ‘very good’ in two studies, ‘adequate’ in three studies and ‘inadequate’ in 10 studies. Studies of higher quality reported findings of confirmatory factor analysis. Most studies were graded as adequate/inadequate due to insufficient sample size as recommended by COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines.18,30,31
Internal consistency
Internal consistency refers to the degree of interrelatedness among items. 30 Most papers reported internal consistency. Thirty-two patient reported outcome measures were only assessed for internal consistency.34,36–42,45,51–55,62,64,68 The Fatigue Thermometer, Pain Thermometer, Distress Thermometer (single item measures) and the Spinal Muscular Atrophy – Health Index (SMA-HI) did not report the results of Cronbach’s Alpha. Most studies were assessed as having ‘very good’ methodological quality for internal consistency. Those that did not score ‘very good’ were downgraded as structural validity was not assessed or was deemed inadequate, in line with COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidance.18,30,31,33
Test-retest reliability
Test-retest reliability is the extent to which scores for the patients (who have not changed) are the same for repeated measurements under several conditions over time. 30 Nine studies reported on test-retest reliability. All were rated ‘doubtful’ as it was unclear whether patients were stable in the interim period, whether the time interval was appropriate, and whether test conditions were similar.
Measurement error
Measurement error is the systematic and random error of a patient’s score that is not attributed to true changes in the construct to be measured. 30 Only one study reported information on this domain. 66 The methodological quality was rated as ‘doubtful’ as it was unclear whether patients were stable in the interim period, whether the time interval was appropriate, and whether test conditions were similar.
Construct validity
Construct validity is the extent to which scores of an outcome measure are consistent with hypotheses. 30 In most studies, the hypotheses were clearly defined or reported in the papers. Twenty-four studies reported results for construct validity. Methodological quality was mixed. Eleven studies were rated as ‘very good’, three were rated as ‘adequate’, nine were rated as ‘doubtful’ and one was rated as ‘inadequate’.
Measurement invariance
Measurement invariance was assessed by one study. The methodological quality was rated as ‘very good’. All criteria were sufficiently described. 57
Responsiveness
Responsiveness is the ability of a patient reported outcome measure to detect change over time in a construct to be measured. 30 Three studies compared the patient reported outcome measure to a gold standard, whilst one compared the patient reported outcome measure with other outcome measurement instruments. The methodological quality of all studies was ‘very good’.
Recommended patient reported outcome measures
Most patient reported outcome measures included in this review were evaluated based on the results of a single validation study. Some measures had multiple studies reporting different measurement properties, but the populations studied were heterogeneous and did not allow us to quantitatively pool results. This limited our ability to make recommendations.
No study assessed all psychometric properties. Most patient reported outcome measures (n = 39/68) had evidence of one measurement property, the majority of which reported on internal consistency which was rated as ‘indeterminate’ as there was no evidence for structural validity. Sixteen patient reported outcome measures had evidence of two psychometric properties. The remaining measures (n = 13/68) reported three or more measurement properties.
All outcome measures included in this review were placed in Category B. They have potential to be recommended for use, but they require further research to assess their comprehensiveness, reliability and validity in the young adult population. 30 None of the measures met the criteria to be placed into Category A as they do not meet sufficient criteria for content validity and internal consistency.
Discussion
Main findings/results of the study
Our review identified 68 patient reported outcome measures to assess the health outcomes of young adults (aged 18–25 years) living with life-limiting conditions. Of these measures, most were generic (not disease-specific). Most measures were validated in cancer populations, we did not identify any measures for young adults living with neurodisabilties. The majority were two-dimensional rather than holistic, focussing on the assessment of psychological symptoms such as mood and wellbeing. Most papers measured internal consistency. Few studies reported on content validity, measurement error, measurement invariance and responsiveness. No studies assessed criterion validity.
What this study adds?
In young adults (aged 18–25 years) congenital, neurological and respiratory conditions are more prevalent than oncology diagnoses. 5 Despite this, most measures we identified were psychometrically assessed in young adults living with cancer. There was a lack of measures developed, adapted or validated for young adults living with non-cancerous conditions. Specifically, there were no studies assessing the health outcomes of young adults living with complex neurodisability despite the large number of young adults affected by these conditions.
Most of the measures identified were generic, and not specific to a particular condition. Generic measures allow for comparisons of health outcomes across populations 71 which may be beneficial for heterogenous groups such as young adults living with life-limiting conditions as the types of diagnoses and complexity varies. 1 Alternatively, disease-specific measures allow for comparisons of health outcomes within a particular condition 71 and can be sensitive to change when evaluating the effects of interventions or treatments71,72 but given the large number of different diseases within this population this makes it challenging to develop disease-specific measures. Therefore, this is not something that we think would be useful as the process of develop disease-specific measures would be complex and less efficient and the benefit to young adults living with life-limiting conditions would be limited.
A review by Contri et al 16 explains that the validity of a patient reported outcome can be affected by the context in which they are applied. This includes ‘geographical location, age, language, educational level and socioeconomic and cultural background of the target population’. 16 We found that most studies did not report educational level or socioeconomic characteristics of their sample. In line with a review of patient reported outcome measures for young adults living with cancer, 73 we found that minority groups were underrepresented in most studies. Sixteen papers did not report on ethnicity or race when describing participant demographics. This makes it unclear whether the items or constructs measured by the patient reported outcome measure are meaningful, comprehensive or valid for different groups. The need for representation of racially and ethnically minoritised groups has been called for by Rhodes et al 74 who recommend that descriptions of race and ethnicity are standardised across research, and researchers should ‘strive for diversity, equity and inclusion among research participants’. They suggest that quota sampling on race and ethnicity may facilitate this. 75 This would help to ensure that outcome measures are comprehensive of the needs of diverse groups of young adults living with life-limiting conditions when implemented in clinical practice and research studies.
Most of the included papers did not comprehensively report the methods used to assess the reliability and validity of patient reported outcome measures. In some papers it was unclear which measurement property the authors were reporting on. In these circumstances, the authors of this review made their own judgement based on the information provided. This may explain the variation in methodological quality (COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) risk of bias assessment). This is consistent with previous research which identified that ‘studies examining measurement properties are often missing key information that allows the reader to determine the methods used, what the results are and what the research means for the evidence of the quality of a particular patient reported outcome measure’. 76 This may also be explained by the publication dates of included studies which spanned the years of 1998 to 2021. During this period there have been numerous guidelines and recommendations for the reporting of validation studies. 76 To standardise and facilitate the comprehensive reporting of validation studies, COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines should be utilised. This should improve the ability of reviewers to quality appraise measurement papers 76 and will support the selection of patient reported outcome measures for use in clinical practice and research studies.
Internal consistency, construct validity and structural validity were the most assessed measurement properties. Three studies assessed responsiveness; it is therefore unclear whether the measures identified were responsive to change. 71 This should be considered when implementing these tools in clinical practice or measuring the effects of an intervention. Content validity is the extent to which the content of a patient reported outcome measure is relevant, complete and comprehensive of the construct to be measured and for the target population and is considered to be the most important measurement property by COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN). 17 To meet COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) criteria to be recommended for use, a patient reported outcome measure requires at least sufficient evidence for content validity and internal consistency. We found insufficient evidence for content validity and internal consistency so all of the measures were placed in category B and have the potential to be recommended for use. Thirteen studies reported on content validity, but the quality of evidence for 12 of these studies was rated as insufficient. One patient reported outcome measure (PROMIS v2.0 Brief Profile Sex FS) had sufficient criteria for content validity but did not have any evidence for internal consistency. 60 COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidance outlines that in circumstance where all patient reported outcome measures are placed in category B, the measure with best evidence for content validity can be provisionally recommended for use until additional evidence is provided. 17 In this circumstance the patient reported outcome measure was very specific, only measuring sexual function, and was therefore not recommended for use.
For most of the measures identified it is unclear how young adults living with life-limiting conditions interpret the items, whether the items are perceived as relevant from their own experiences and whether the results are useful for clinical decision making. 71 Future studies should take existing patient reported outcome measures, covering all domains of palliative care (physical, psychological, social and spiritual) and ask young adults (aged 18–25 years) and professionals (clinicians and researchers) about the relevance, comprehensiveness and comprehensibility of items in line with COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidance. 30 Where young adults are unable to self-report, we recommend proxy-reporting by families or healthcare professionals where possible.
Most measures identified were two-dimensional either assessing physical and psychological outcomes, or psychological and social outcomes. Even within these dimensions, many measures only assessed one specific aspect, that is, physical fatigue. Only three patient reported outcome measures were holistic,45,52,59 assessing each domain of palliative care as defined by the World Health Organisation (physical, psychological, social and spiritual/existential) 29 and notably these measures were all developed and validated in cancer populations. Future studies may identify existing holistic measures that have been developed for adults or children and adapt them for use in young adult populations.
Strengths and limitations of the study
A strength of this review is that it adheres to the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) methodology for systematic reviews of patient reported outcome measures. Our search strategy was broad and was not limited by language or date restrictions to capture all relevant papers. Our search identified papers that developed, adapted or validated patient reported outcome measures for ages that spanned children, adolescents and young adults living with life-limiting conditions. In these studies, the age range was often 0–25 years or 14–30 years. This encompassed the age range for inclusion in our study (18–25 years), however, the papers did not specify the percentage of young adults aged 18–25 years in their sample, or the mean or median age fell outside our inclusion criteria (18–25 years). They were therefore excluded from this review. There may be high quality studies that have adequately assessed the measurement properties of holistic patient reported outcome measures, but because of our strict inclusion criteria they were not included in this review, but they may still be relevant to the young adult population.
Conclusion
We identified a number of one- and two-dimensional patient reported outcome measures for young adults living with life-limiting conditions. Most were validated in young adults living with cancer, with non-cancerous populations underrepresented, especially those with complex neurodisability. Future studies should examine existing patient reported outcome measures that cover all domains of palliative care (physical, psychological, social and spiritual) and assess their suitability for use in young adults. To standardise and facilitate the comprehensive reporting of validation studies, COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines should be utilised. This will support the selection of patient reported outcome measures for use in clinical practice and research studies.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163251340175 – Supplemental material for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review
Supplemental material, sj-docx-1-pmj-10.1177_02692163251340175 for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review by Rachel L Chambers, Mevhibe B Hocaoglu, Irene J Higginson, Katherine E Sleeman and Lorna K Fraser in Palliative Medicine
Supplemental Material
sj-docx-2-pmj-10.1177_02692163251340175 – Supplemental material for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review
Supplemental material, sj-docx-2-pmj-10.1177_02692163251340175 for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review by Rachel L Chambers, Mevhibe B Hocaoglu, Irene J Higginson, Katherine E Sleeman and Lorna K Fraser in Palliative Medicine
Supplemental Material
sj-docx-4-pmj-10.1177_02692163251340175 – Supplemental material for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review
Supplemental material, sj-docx-4-pmj-10.1177_02692163251340175 for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review by Rachel L Chambers, Mevhibe B Hocaoglu, Irene J Higginson, Katherine E Sleeman and Lorna K Fraser in Palliative Medicine
Supplemental Material
sj-xlsx-3-pmj-10.1177_02692163251340175 – Supplemental material for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review
Supplemental material, sj-xlsx-3-pmj-10.1177_02692163251340175 for Evaluating the measurement properties of patient-reported outcome measures for young adults with life-limiting conditions: A systematic review by Rachel L Chambers, Mevhibe B Hocaoglu, Irene J Higginson, Katherine E Sleeman and Lorna K Fraser in Palliative Medicine
Footnotes
Data availability
This review is registered on the PROSPERO database (CRD42023443273) and the full search strategy is provided in the Supplemental Material Files. Inclusion/exclusion criteria and quality appraisal results are available in the article. Additional data can be made available upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Rachel Chambers, Pre-doctoral Fellowship, NIHR302750 is funded by the National Institute for Health Research (NIHR) for this research project. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR, NHS or the UK Department of Health and Social Care. MBH is supported by the NIHR ARC SL. The views expressed in this article are those of the authors and not necessarily those of the NIHR, or the Department of Health and Social Care. KES is the Laing Galazka Chair in palliative care at King’s College London, funded through an endowment from Cicely Saunders International and the Kirby Laing Foundation. Lorna Fraser is funded by a National Institute for Health Research (NIHR) Career Development Fellowship (award CDF-2018-11-ST2-002). This publication presents independent research funded by the National Institute for Health Research (NIHR). The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care. IJH is a National Institute for Health Research (NIHR) Emeritus Senior Investigator and is supported by the NIHR Applied Research Collaboration (ARC) South London (SL) at King’s College Hospital National Health Service Foundation Trust. IJH leads the Palliative and End of Life Care theme of the NIHR ARC SL and co-leads the national theme in this. The views expressed in this article are those of the authors and not necessarily those of the NIHR, or the Department of Health and Social Care.
Ethical approval and consent to participate statements
Ethical approval was not required for this review as there was no direct patient contact or access to individual participant data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.