Abstract
Background:
Delirium is a complex condition, stressful for all involved. Although highly prevalent in palliative care settings, it remains underdiagnosed and associated with poor outcomes. Guideline-adherent delirium care may improve its detection, assessment and management.
Aim:
To inform a future definitive study that tests whether an implementation strategy designed to improve guideline-adherent delirium care in palliative care settings improves patient outcomes (reduced proportion of in-patient days with delirium).
Design:
With Patient Involvement members, we conducted a feasibility study to assess the acceptability of and engagement with the implementation strategy by hospice staff (intervention), and whether clinical record data collection of process (e.g. guideline-adherent delirium care) and clinical outcomes (evidence of delirium using a validated chart-based instrument;) pre- and 12-weeks post-implementation of the intervention would be possible.
Setting/participants:
In-patient admissions in three English hospices.
Results:
Between June 2021 and December 2022, clinical record data were extracted from 300 consecutive admissions. Despite data collection during COVID-19, target clinical record data collection (n = 300) was achieved. Approximately two-thirds of patients had a delirium episode during in-patient stay at both timepoints. A 6% absolute reduction in proportion of delirium days in those with a delirium episode was observed. Post-implementation improvements in guideline-adherent metrics include: clinical delirium diagnosis 15%–28%; delirium risk assessment 0%–16%; screening on admission 7%–35%.
Conclusions:
Collection of data on delirium outcomes and guideline-adherence from clinical records is feasible. The signal of patient benefit supports formal evaluation in a large-scale study.
Delirium is highly prevalent and associated with poor outcomes in palliative care.
Evidence-based delirium guidelines are not fully implemented in practice, and their implementation can be challenging.
Collecting data on delirium outcomes and guideline-adherence from hospice clinical records is feasible.
Demonstrated feasibility of a multi-professional (clinical, domestic and management) implementation strategy.
Methodological extension of implementation research beyond process to measure impact of implementation strategy on patient outcomes
There is disparity between need (high delirium-incidence) and documented action (low guideline-adherence).
Integration of a delirium assessment tool into patient documentation, identification of a delirium champion and management support are important for implementation of delirium guidelines.
A randomised multi-site effectiveness study is required to formally evaluate impact of the implementation strategy on patient outcomes.
Introduction
People nearing the end of life have a high risk of delirium, a syndrome of impaired awareness, attention and cognition. Delirium is distressing for patients and families, increases health professionals’ anxiety and stress, leads to poor clinical outcomes and higher care costs. Effective delirium care is driven by prevention, timely detection and non-pharmacological management, with pharmacological interventions if appropriate.1,2
One-third of people have delirium on admission to adult palliative care settings, while two-thirds develop delirium during their in-patient stay. 3 Although delirium assessment, management and prevention guidelines4,5 and standards 6 are evidence-based, implementation in practice is poor. The existence of guidelines alone does not appear to consistently translate into motivation or capacity of clinicians to deliver guideline-adherent delirium care. For example, continued routine use of antipsychotics to treat delirium, 7 despite evidence-based guidelines to the contrary.8,9 Slow practice change is also difficult if there are low levels of practical and emotional support. 10
In-patient palliative care settings have challenges for implementing delirium guidelines, including the complex interactions between patients, family members, clinical staff, volunteers and managers at some of the most sensitive times in the lives of patients and their families.10–12 Validated tools such as the 4AT 13 can be helpful to support delirium management and prevention, but even short tools have low rates of use in palliative care units. 14 Knowledge sharing within teams, incorporation of screening documentation into workflow and a culture of compassionate and collaborative working is important.15,16
It is therefore as vital to find strategies to implement guideline-adherent care as it is to identify the optimal way to assess, manage and prevent delirium through clinical interventions. The
In this paper, we report the findings of the feasibility study to test the feasibility of conductingbibr a definitive study to evaluate the impact of the implementation strategy (intervention) on process outcomes (delirium guideline adherence) and patient-relevant clinical outcomes (reduction in the proportion of delirium days during an admission). 19
Methods
Design
This study tests the feasibility of a future trial testing the effectiveness of an implementation strategy on process and clinical outcomes with regard to: approach to consent; staff engagement with the implementation strategy (intervention fidelity) and delivery of guideline-adherent delirium care (process outcomes); delirium retrospective diagnosis from in-patient clinical records (clinical outcome); costs of staff involvement in the implementation strategy; sample size estimation.
Public Involvement members were included in all aspects, with meetings held at six timepoints during the study and one member (co-author MO) joining the study team at monthly project management meetings.
We present a summary of data collection methods in relation to patients, staff and costs. Detailed methods are presented in the published protocol. 17 The design of the proposed future definitive trial is summarised in Supplemental 3.
Setting
Three adult independent charity-funded hospices in northern England (United Kingdom) located in an affluent rural/urban area, a socio-economically deprived urban city and a socio-economically deprived coastal town. All provided a range of in-patient and community care by a multi-disciplinary palliative care team. Hospices were of similar size (average bed capacity 20, reduced to ~5 during Covid restrictions).
Patients
Baseline in-patient data were collected from paper or electronic clinical records of 50 consecutive patients admitted to participating hospices who completed their in-patient stay immediately prior to the introduction of the CLECC-Pal implementation strategy. Post-implementation in-patient data were collected from 50 consecutive patients admitted to participating hospices from week 4 post-CLECC-Pal commencement. Approval was given by the Confidential Advisory Group of the Health Research Authority to collect clinical record data without consent providing an opt-out was available. Posters about the study were placed in visible public places, and information placed in every patient’s admission welcome pack, including that they could contact a member of clinical staff if they wished to opt-out of providing data.
Data collection
The non-clinical researcher (GJ) was given initial training by a clinical researcher (CJ) regarding how to use an expanded version of a validated chart-based instrument for detecting potential delirium from clinical records (clinical outcome), 20 and 10% of hospice one records were extracted by both researchers to look for agreement and need for additional training. The remaining in-patient data (paper-based or electronic, depending on the hospice), anonymised at the point of extraction, were collected by the non-clinical researcher (GJ) and where assumptions were ambiguous, consultation was held with the clinical researcher for clarification. Any discrepancies were resolved through consensus. The instrument enabled assessment of whether case-note recorded symptoms of delirium (which did not have to be identified as ‘delirium’ symptoms by the recording clinician) could be linked to actions taken (consistent or not with guidelines) during the person’s admission. We extracted demographic data, converting postcodes to Index of Multiple Deprivation (IMD) scores to preserve anonymity 21 (Table 1). Additional items about the presence or not of actions to support delirium assessment, management and prevention (for example, use of delirium screening tools, risk assessments and individualised delirium management care plans (Supplemental 1)) were included. Based on pilot work in a hospice of comparable size, a sample size of 50 patient records per hospice was sufficient to answer feasibility questions about data quality and for capture of care planning. The time required to extract data was recorded.
Patient demographics.
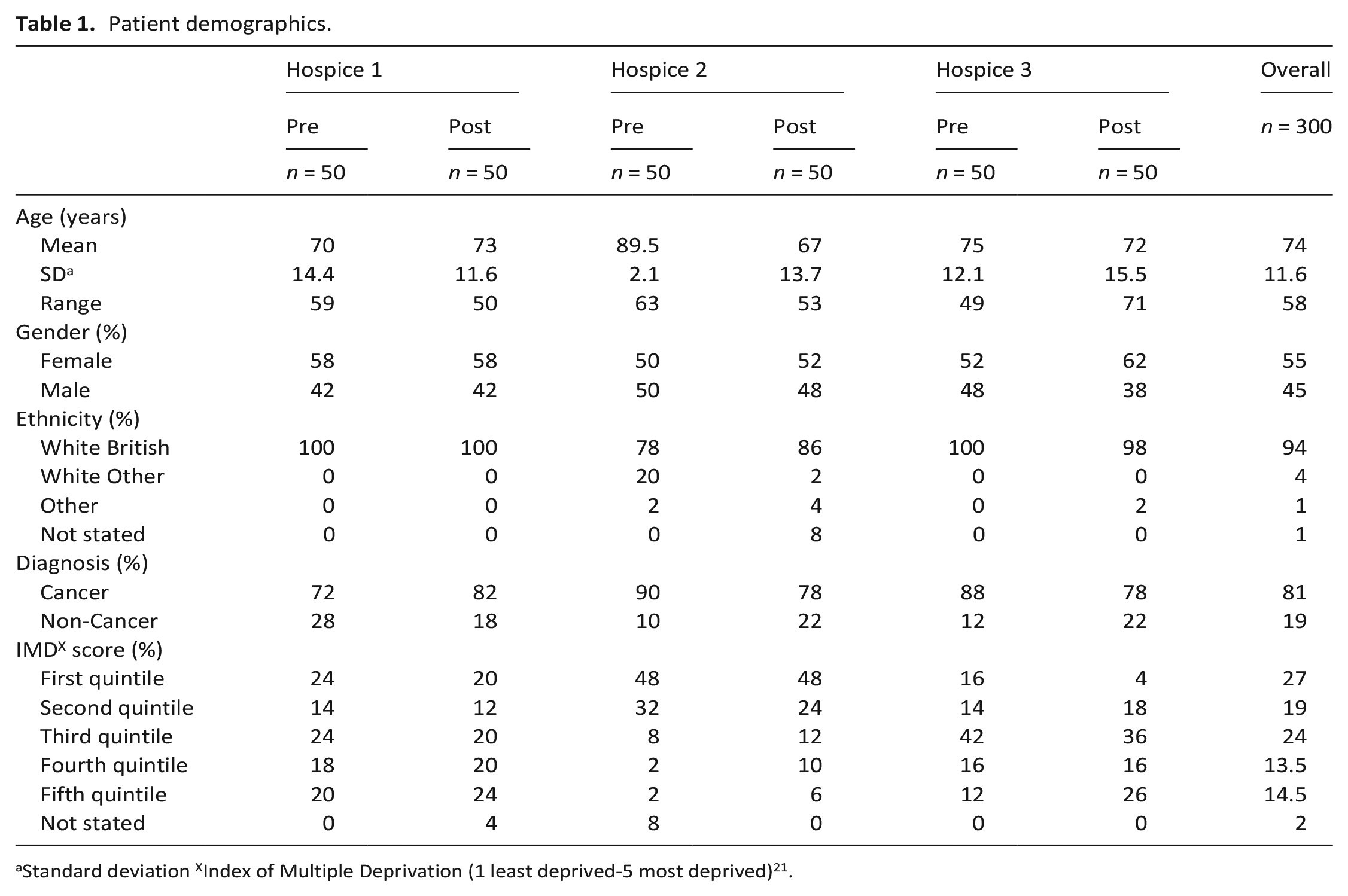
Standard deviation XIndex of Multiple Deprivation (1 least deprived-5 most deprived) 21 .
Implementation strategy (study intervention)
Co-designed development (CLECC-Pal) of the original CLECC strategy was used for 12-weeks at each hospice. CLECC-Pal aimed to create a secure learning environment that fosters new ways of working. Components are defined in the protocol, 17 but in summary included a study day (covering guideline-adherent delirium care training and CLECC components), mid-shift cluster discussion (ward-based delirium-focussed review), peer observation of practice (one-on-one clinical observation with feedback), reflective discussion (group reflection and learning facilitated by a clinical lead) and action learning sets (group action plan development to address identified problems). Clinical leads identified and invited staff members involved in the care of delirious patients to the study day aiming to achieve representation from a diverse range of roles. At the end of the study day an optional anonymous paper feedback form was distributed.
Staff
Data were collected about fidelity of CLECC-Pal use and levels of staff engagement through a weekly ‘rapid report’ by the clinical lead in each hospice throughout the implementation period. Data to be collected included the number of staff involved in each CLECC-Pal component, reasons for staff non-engagement/drop-out and modifications made in the use of CLECC-Pal.
Costs
Number of hours spent by members of staff in CLECC-Pal activities were collected to assess the feasibility of collecting data about the costs of using CLECC-Pal.
Analysis
Data are presented using descriptive statistics. Mean (SD) is reported for continuous data and raw count (number, percentage) for nominal data. Calculation of the variation around baseline proportion of delirium days/admission informs the sample size and number of hospices needed for the subsequent national study.
International relevance
An expert advisory group of senior (clinical) academics (Australia, Canada) met online five times during the study to provide advice and link the study to international developments in delirium management and prevention trials.
Ethical and regulatory approval
Health Research Authority Research Ethics Committee Wales REC7 (Ref. 21/WA/0180, 28/05/2021) and Health Research Authority Confidentiality Advisory Group (Ref. 21/CAG/0071, 14/06/2021; see above).
The study is reported in accordance with the Consolidated Standards of Reporting Trials (CONSORT) extension statement 22 and is registered (ISRCTN55416525).
Results
Clinical record data extraction
Patient data
During June 2021 to December 2022, patient data extraction tool was 100% completed for 50 in-patient episodes before/after implementation in each of three hospices (300 in-patient episodes; 55% female; mean age 74 years, range 24–96 years). A true consecutive sample was achieved; no patients or their representatives elected to opt-out. Most patients were older, white British, cancer patients. Socio-economic status varied; hospice two had more patients residing in the first IMD quintile indicating least deprived (Table 1). Clinical and non-clinical researcher agreement was unanimous (100%) for the 10% calibration data extraction. Out of 300 records 70 (23%) required discussion with the clinical researcher (CJ).
Delirium prevalence at different timepoints
Pre-implementation, overall delirium prevalence on admission for in-patients was 35%, post-implementation was 28%. Clinical records indicated delirium in around two-thirds of patients during their in-patient stay. Of those that experienced delirium during their in-patient stay, the proportion of in-patient days with delirium, that is, individual patient number of delirium days divided by their total number of in-patient stay (days), was on average 69% pre- and 63% post-intervention (6% absolute reduction; Table 2).
Before and after measures of delirium outcomes and management.
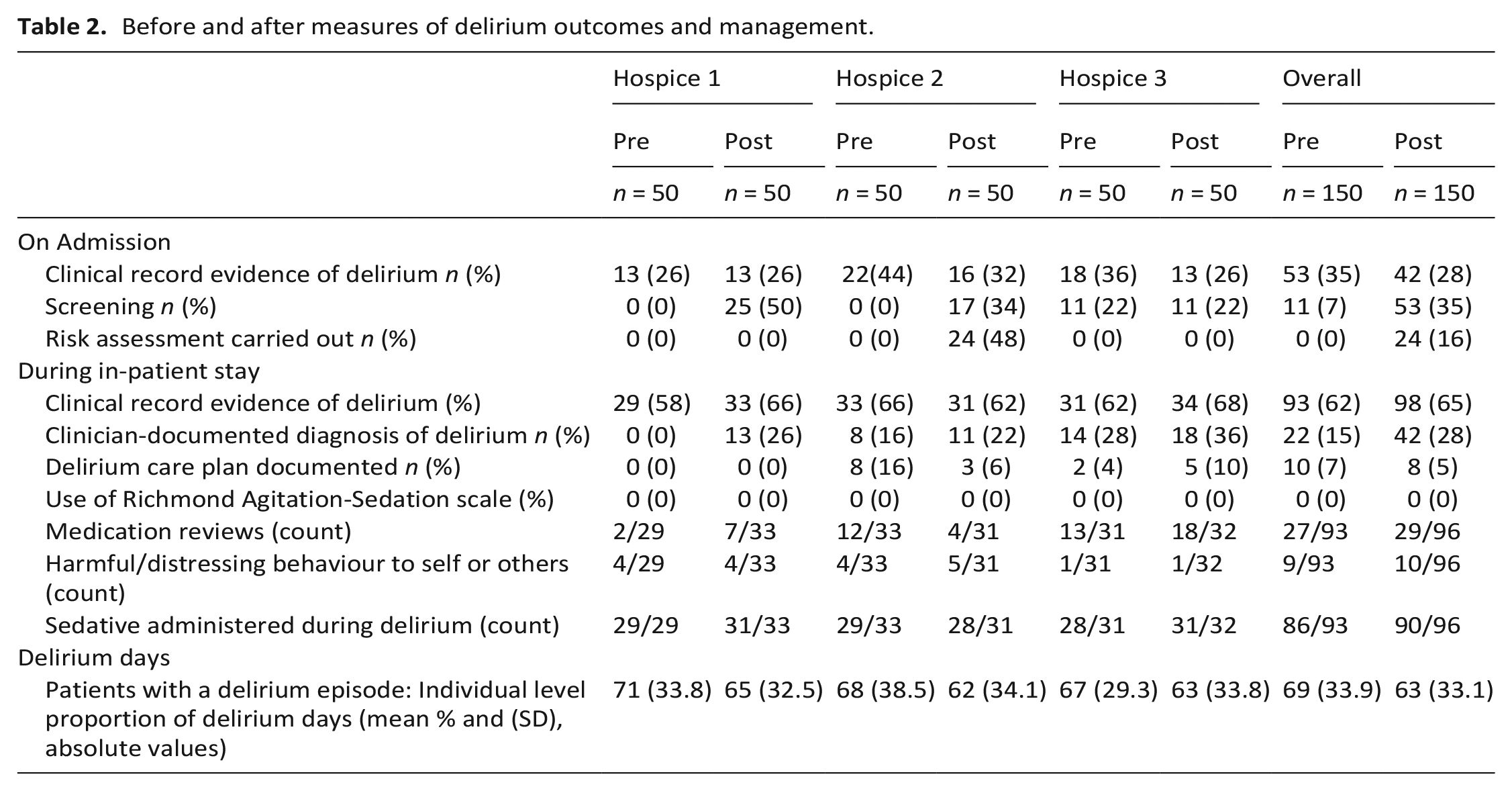
Delivery of guideline-adherent delirium care
There was evidence of improved guideline-adherence following CLECC-Pal (Table 2) Changes were observed for clinician-documented diagnosis of delirium (all hospices), delirium screening (two hospices) and risk assessment (one hospice). Overall, administration of sedatives was common (>90%) despite minimal evidence of harmful patient-behaviour (<16%).
Time to complete data collection
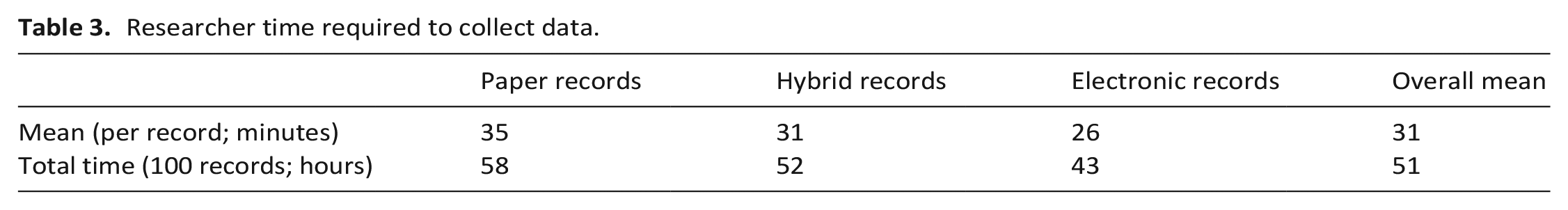
One hospice had fully paper records, one had electronic doctor records but nurse paper records, and the third fully electronic. Electronic records were accessible remotely. Time taken for data extraction (Supplemental 2) improved with researcher familiarity with clinical records. Overall, on average 31 minutes (range 3–150) per record.
Total time to data extract 100 records allows estimation of resource needed for a future definitive study (Table 3). Of note, estimate does not include researcher travel, or time spent at sites not directly data extracting.
Researcher time required to collect data.
CLECC-Pal participation
Staff (participation in CLECC-Pal components)
Study day
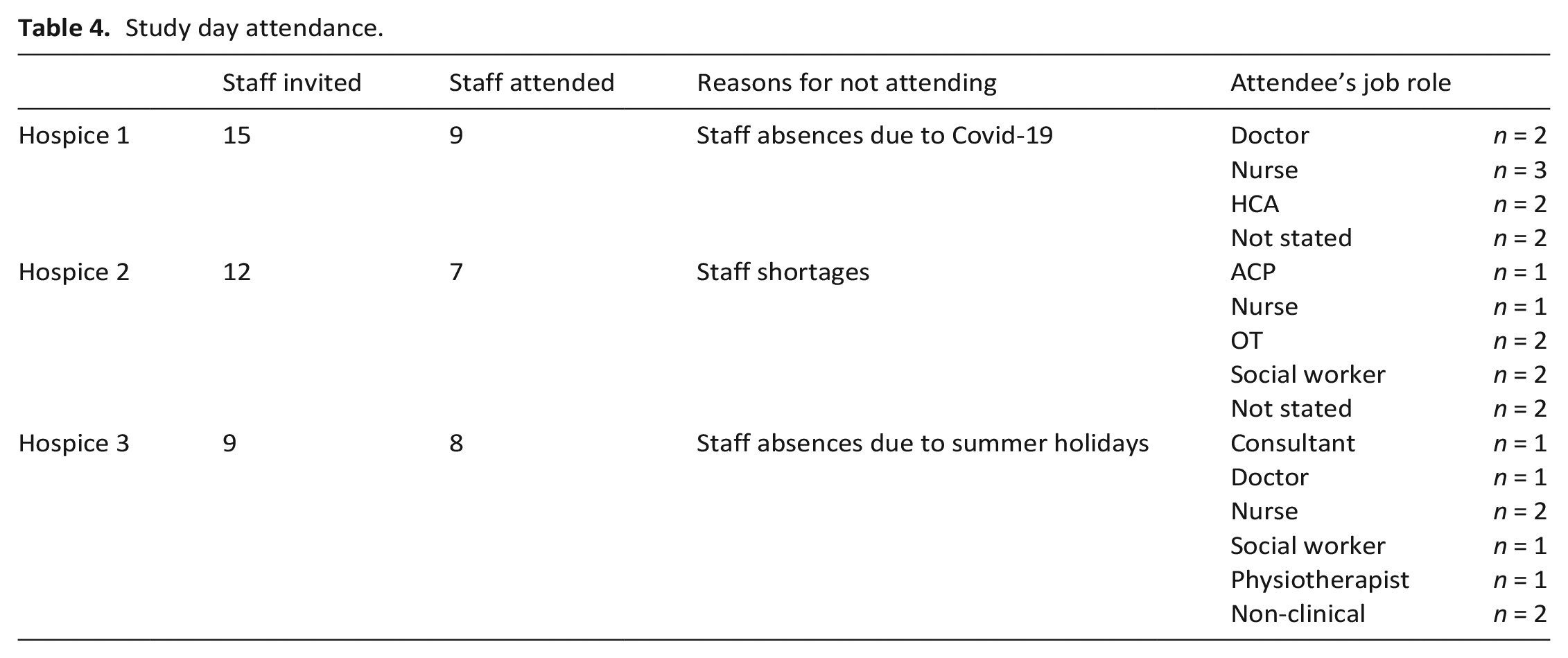
The intention was to deliver in-person study days. However, hybrid in-person/online delivery was necessary at one hospice due to COVID in the research team (GJ). The hospice clinical lead distributed study materials and facilitated online access. Co-chief investigator (MP) attended in person to co-facilitate the afternoon session. Clinical leads at each hospice invited staff members representing diverse roles. Over half of those invited attended (Table 4).
Study day attendance.
Study day format changes based on feedback
In response to feedback from the first study day, subsequent study days’ agenda was altered so that delirium education was the morning session and the afternoon session focussed on the CLECC-Pal implementation strategy and the individual components.
At the end of the study day an optional paper feedback form was distributed. Across the three hospices we received 23/24 completed feedback forms (one attendee left early). The form consisted of several open text questions relating to study day content and a free text box for any additional comments. Forms were not formally analysed, instead they were used as a temperature check for acceptability of study day content and format. Feedback about the day was positive, with most attendees finding presentations useful. From free text comments and feedback from clinical leads, meeting in-person, spaces for discussion, delirium presentations and CLECC-Pal activities/planning were particularly valued. Following co-design workshop feedback, we made study day presentations available online prior to the day (5/23 respondents accessed in advance).
Suggestions to optimise study day format for the future included: splitting the day into two separate sessions to achieve maximum attendance; further clarification and case examples of CLECC-Pal in practice; forming an initial plan and recruiting ‘champions’ at the study day.
Fidelity to other CLECC-Pal components
Rather than a formal mid-shift cluster discussion set up specifically for delirium care, all hospices adapted an existing meeting to include delirium discussion, such as handovers and/or safety huddle meetings. Reflective discussions had been used previously in all hospices prior to the pandemic, although not to discuss delirium. All recognised the value and planned to reinstate this component.
Action learning sets (ALS) and peer observation of practice (POP) were the least used components. Hospices found the purpose and value of ALS confusing. Opinions were divided on the value of POP with partial use reported in one hospice only. This hospice chose to use POP to help implement delirium screening tool use on admission. Although at that time POP was solely used by the doctors, they planned to roll it out to nurses. The other hospices initially viewed POP as a negative practice, and, although perceptions changed at the study day seeing the potential value, it was not used. Hospice one created a comprehensive sustainability plan during the implementation period, incorporating it into their existing audit plan. The other hospices did not devise sustainability plans due to focussing on other aspects of CLECC-Pal (Table 5).
Hospice use of CLECC-Pal components.

✓used no change used modified X Did not use *Partial.
used modified X Did not use *Partial.
CLECC-Pal participation costs
Although we received weekly feedback from 2/3 hospices (fortnightly at hospice three), details were insufficient to calculate costs. In consultation with the hospice leads, we estimated time and costs (mean of each band) for staff to engage in each CLECC-Pal component (Table 6).
CLECC-Pal time and cost estimates.
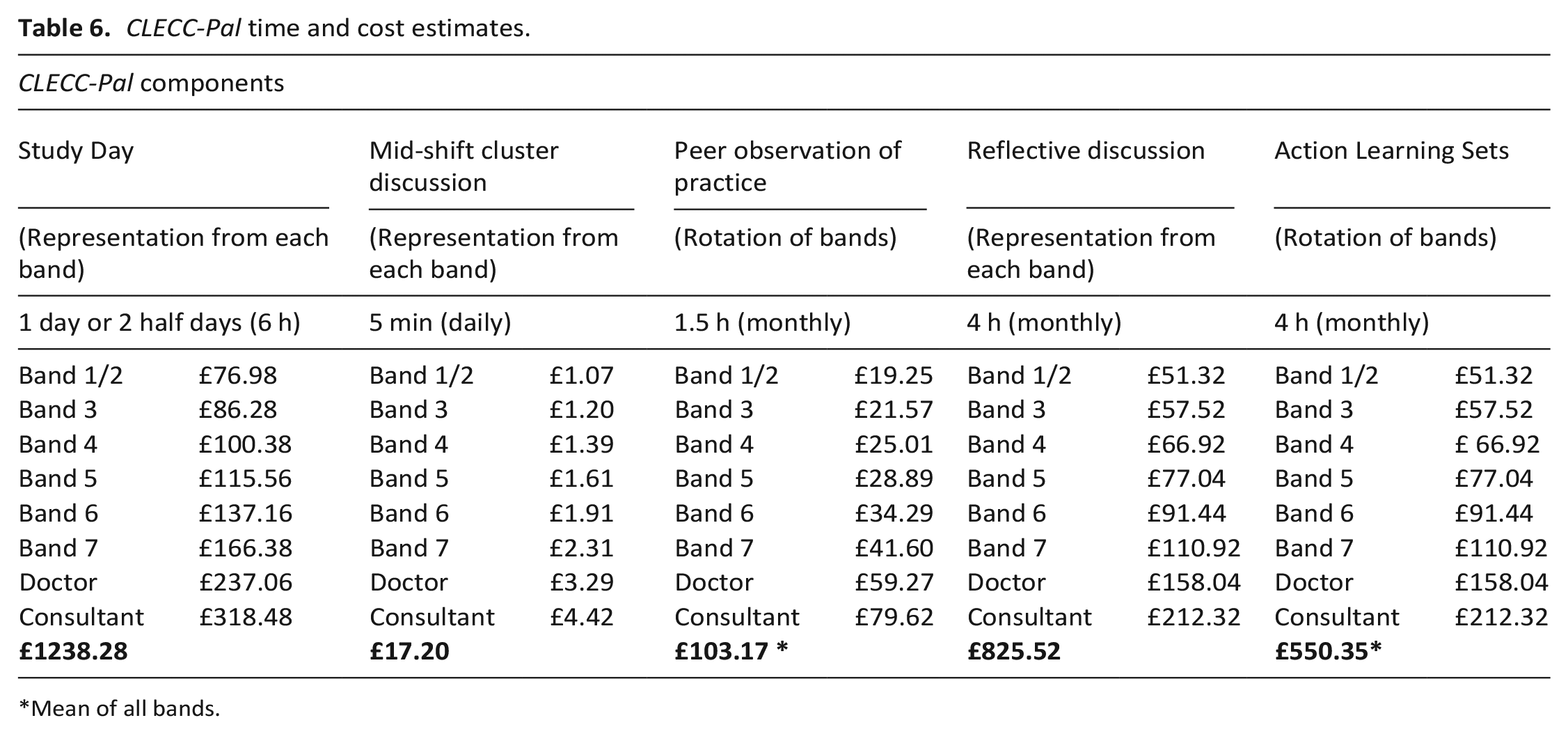
Mean of all bands.
Informing future definitive study design
Sample size
Based on the feasibility study data, we propose a cluster RCT as the optimal study design for a definitive study to test effectiveness of the CLECC-Pal implementation strategy in reducing proportion of delirium days (primary outcome). This definitive study will require a cluster of 30 patients with a delirium episode from each of 20 hospices (size of cluster = 30; number of hospice clusters for randomisation = 20) to provide 92.3% power (0.05 significance level; intraclass correlation coefficient of 0.03 in view of the clinical rather than process outcome) 23 to detect a 12% reduction of delirium days per episode of delirium (effect size 0.4). Based on delirium episodes in this feasibility study, a minimum of 50 consecutive patient records would be required (in each site, at each timepoint) to identify each sample of 30 delirium episodes.
Key components of CLECC-Pal
Feedback from hospices indicated a study day delivered flexibly (starting with delirium education), mid-shift discussions, reflective discussions, delirium champions and sustainability plans were the most implementable components. Peer observation of practice may be useful if staff concerns are addressed prior to introduction.
Key requirements for CLECC-Pal implementation
Based on clinical lead feedback and critical reflection by the study team, hospices starting implementation should consider:
How the 4AT tool will be integrated into clinical records.
Who has sufficient capacity, authority and motivation to lead each of the CLECC-Pal components.
How to ensure hospice managers are committed to supporting ways, such as CLECC-Pal, to optimise guideline-adherent delirium care.
Based on clinical lead feedback, critical reflection by the study team and the co-design workshops, maintenance of hospice staff engagement in a novel intervention such as CLECC-Pal requires:
Regular and personable contact between researchers and hospice leads.
Easily accessible study update materials, such as a bi-monthly newsletter. Other strategies suggested by hospice staff to increase study visibility include: computer screensavers or bite-size educational materials (e.g. ‘delirium point of the day’).
This study was conducted in the context of prior co-design workshops with participating hospices to develop CLECC -Pal which fostered engagement. In a definitive study, this will not be the case, and alternative ways to get initial ‘buy-in’ should be considered.
Discussion
A definitive study evaluating the effectiveness of an implementation strategy to facilitate delirium guideline-adherent care to reduce the proportion of delirium days per hospice admission is feasible. The opt-out approach to consent was acceptable (no opt-outs) and allowed a truly consecutive sample including those with reduced mental capacity – that is – those most likely to benefit from optimal delirium care, but most likely to be under-represented in study populations. Despite only modest improvements in documented guideline-adherent delirium care, a signal of patient benefit was seen supporting the need for a definitive trial to confirm or refute these findings.
The acceptability and benefits of opt-out instead of seeking patient consent to access patient records and collect delirium related data has been reported previously.24,25 Standard informed consent to participate, even with consultee or proxy provision for people with reduced mental capacity, runs the risk of an unrepresentative sample, 26 particularly of those living in deprived areas, with reduced mental capacity and/or from minoritised ethnic communities.
Using the clinical record data extraction tool, we were able to gain a useful clinical outcome (proportion of delirium days) from all patient records efficiently. Like others, we found frequency of keywords were higher in nursing notes than doctor’s notes. 27 Like others, 28 delirium awareness was low in our study; the word ‘delirium’ appeared rarely in baseline patient records. Gold standard for delirium diagnosis is clinical interview, but this would be a significant additional task for participating hospice clinicians and unlikely to happen consistently and would attract additional costs. Whilst other studies have used screening tools administered by participating clinicians, we did not use this approach due to concerns about reporting bias and missing data. 29 Funding sufficient trained researcher time to conduct patient assessments to diagnose delirium would be prohibitively expensive in a large, multisite trial, burdensome for patients and difficult to justify a waiver of opt-in consent. Research time needed for data extraction was documented, which will directly inform the funding request for a definitive trial.
The flexible CLECC-Pal strategy facilitated the ethos of individual ownership, expanding beyond simple transfer of knowledge to enable CLECC-Pal implementation into the existing hospice structure. The general consensus was this approach appeared helpful across participating hospices. Importance of recruiting delirium champions is acknowledged; 30 we suggest a delirium team including more than one nurse, allowing for staff changes, and absence. Senior management support was required to facilitate staff engagement with CLECC-Pal. This, and the recognition of the importance of delirium management, is consistent with systematic review findings regarding successful implementation of a delirium intervention. 31 Practical considerations to incorporate into a future study timeline included allowing for varying internal governance procedures to allow use of tools and delirium care plans. The differences in adherence and impact in the participating hospices reflect contextual differences between the hospices in terms of their ‘implementation readiness’ and staffing capacity. These contextual differences should be considered in the design and conduct of a definitive trial.
Despite an acknowledged need for a realistic timeframe to fully implement CLECC-Pal, it is worth noting that even with a 12-week implementation period in this feasibility study (which was not powered to detect benefit), we observed an absolute 6% reduction in delirium days post-implementation of CLECC-Pal for those patients that experienced delirium during their in-patient stay. This reduction in delirium days supports further investigation in a full-scale study.
Study strengths and limitations
This study has a number of strengths. Permission to use opt-out consent allowed recruitment of a truly representative sample. Contribution from members of the public with experience of delirium and hospice settings as service users strengthened our study design and rationale regarding public good that was important for approval of the opt-out approach to data collection. Our delirium data collection methods minimised burden to patients and staff and allowed efficient collection of a clinical outcome measure with sufficient robustness to indicate potential change. The chart-based tool we used was developed in the United States against the Confusion Assessment Method (CAM) 32 but has been validated (sensitivity 89%, specificity 75%) for a diagnosis of delirium. 20 Chart-based tools for delirium ascertainment have been used successfully to conduct large, multi-site hospital studies.33,34 As with our study, this approach also enabled identification of both clinically diagnosed and un-diagnosed delirium from clinical records (57% diagnosed clinically; 42% undiagnosed). Although chart-based methods may underestimate the true presence of delirium compared with a clinical assessment of delirium, 35 the agreement of clinical assessment and chart-based diagnoses was high (80%). Our dependence on clinical record documentation of guideline-adherent delirium care could be seen as a limitation. The documented evidence may lead to an underestimate of the good care delivered in practice and may explain in part why we only saw modest improvements in guideline-adherent delirium care. However, this underlines the importance of taking implementation research beyond process outcomes (delirium care) to clinical outcomes (reduction in delirium days); despite only a modest improvement in process, there was a signal of improvement with our clinical outcome.
Purposive inclusion of three diverse hospices allowed demonstration of the applicability of a flexible CLECC-Pal strategy in varying hospice settings and cultures. This supports the ability to scale-up to a multi-site hospice trial.
Study limitations included difficulty in collecting accurate cost data. We were able to collect some, but this issue requires further consideration for a future trial. We did not examine potential cost savings from reduction in delirium days; important to address due to other indications that optimal delirium care reduces healthcare costs. 28 Although, as a feasibility study, our study was not intended to assess effectiveness, we do not know if the signal of effect was because of CLECC-Pal implementation or the delirium research activity itself, or a chance finding. 36 Finally, there was minimal ethnic diversity in the hospice in-patient population despite significant ethnic diversity in the hospice catchment areas of two of the hospices, reflecting a more systemic issue of access to hospice in-patient care by people from minoritised ethnic communities. 37
Conclusion
A definitive multi-site trial of effectiveness of CLECC-Pal implementation strategy on the clinical outcome of reduced delirium days is feasible. Using a chart-based instrument to retrospectively diagnose delirium is an efficient data collection method, and an opt-out approach to consent access to clinical records appears acceptable to patients and effective in gaining a truly consecutive and representative sample. The signal of clinical benefit supports the need for a definitive trial.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163241236325 – Supplemental material for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care
Supplemental material, sj-pdf-1-pmj-10.1177_02692163241236325 for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care by Gillian P Jackson, Catriona E Jackson, Jason W Boland, Imogen Featherstone, Chao Huang, Margaret Ogden, Kathryn Sartain, Najma Siddiqi, Maureen Twiddy, Mark Pearson and Miriam J Johnson in Palliative Medicine
Supplemental Material
sj-pdf-2-pmj-10.1177_02692163241236325 – Supplemental material for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care
Supplemental material, sj-pdf-2-pmj-10.1177_02692163241236325 for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care by Gillian P Jackson, Catriona E Jackson, Jason W Boland, Imogen Featherstone, Chao Huang, Margaret Ogden, Kathryn Sartain, Najma Siddiqi, Maureen Twiddy, Mark Pearson and Miriam J Johnson in Palliative Medicine
Supplemental Material
sj-pdf-3-pmj-10.1177_02692163241236325 – Supplemental material for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care
Supplemental material, sj-pdf-3-pmj-10.1177_02692163241236325 for Improving the Detection, Assessment, Management and Prevention of Delirium in Hospices (the DAMPen-D study): Feasibility study of a flexible and scalable implementation strategy to deliver guideline-adherent delirium care by Gillian P Jackson, Catriona E Jackson, Jason W Boland, Imogen Featherstone, Chao Huang, Margaret Ogden, Kathryn Sartain, Najma Siddiqi, Maureen Twiddy, Mark Pearson and Miriam J Johnson in Palliative Medicine
Footnotes
Author contributions
All authors made significant contributions to the paper. MP and MJ led study conceptualisation and design, with contributions from CJ, JB and NS. GJ, MP and MJ wrote the first and subsequent drafts of this manuscript, with comments from the remaining authors. Data extraction and analysis was conducted by GJ with input from CJ, MP and MJ. GJ, MP, MJ and CH contributed to data interpretation. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Yorkshire Cancer Research (Award reference number HEND405DEL).
Permissions
Creating Learning Environments for Compassionate Care (CLECC) © 2020 University of Southampton. Used under non-exclusive licence.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
