Abstract
Background:
Palliative care has the potential to address significant unmet needs in people with Parkinson’s disease and related disorders, but models that rely on in-person specialty palliative care teams have limited scalability.
Aim:
To describe patient and care partner experiences with a novel, community-based palliative care intervention for Parkinson’s disease.
Design:
Qualitative study embedded in a randomized clinical trial to document participant experiences with a novel palliative care intervention (community neurologist training and remote team-based specialist palliative care). Transcripts were coded and thematically analyzed through a combination of team-based inductive and deductive coding.
Setting/Participants:
Twenty-eight patients and 33 care partners purposively sampled from participants in a randomized clinical trial of a palliative care intervention for Parkinson’s disease and related disorders conducted at nine sites.
Results:
Benefits of the intervention included management of a wider range of non-motor symptoms, facilitation of conversations about the future, greater engagement with the health care team, and increased referrals to resources. Participants identified areas of improvement, including uptake of palliative care training by community neurologists, additional prognostic counseling, and clarity and timeliness of communication with the multidisciplinary team.
Conclusions:
Clinicians caring for people with Parkinson’s disease and related disorders should screen for non-motor symptoms, provide regular prognostic counseling, and refer to specialty palliative care services earlier in the course of illness. Future interventions should be designed to promote uptake of palliative care training by community neurologists and further optimize referral to and coordination with in-person or remote specialty palliative teams.
Parkinson’s disease and related disorders cause high symptom burden, decreased quality of life, and increased care partner strain. Palliative care has the potential to address significant unmet needs in Parkinson’s disease and related disorders, but models that rely on in-person, specialist palliative care teams have limited scalability.
Benefits of a novel, community-based palliative care intervention for Parkinson’s disease and related disorders included management of a greater range of needs, facilitation of conversations about the future, greater engagement with the health care team, and increased referrals to resources. Areas for improvement included uptake of palliative care training by community neurologists, additional anticipatory guidance about the expected disease course, and clarity and timeliness of communication with the multidisciplinary team.
All clinicians caring for people with Parkinson’s disease and related disorders should screen for symptoms beyond motor changes (such as pain, anxiety, and depression), provide prognostic counseling earlier and more regularly, and refer to specialty palliative careservices earlier in the course of illness. Future interventions should be designed to promote uptake of palliative care training by community neurologists, place an even larger emphasis on prognostic counseling, and optimize referral to specialty palliative care teams with improved clarity and timeliness of communication between multiple team members.
Introduction
Parkinson’s disease is the second most common neurodegenerative illness and growing in prevalence.1,2 People with Parkinson’s disease and related disorders and their care partners report multiple unmet needs under current models of care, including underrecognized symptoms, decreased quality of life, existential distress, and high care partner strain.3 –9 There is growing interest in the application of palliative care to improve care for people with Parkinson’s disease and related disorders, with two clinical trials of palliative care interventions showing improved patient and care partner outcomes.10,11 However, models that rely on in-person, specialist palliative care teams do not address the needs of people with Parkinson’s disease who lack access to these teams and have limited scalability due to a constrained number of palliative care specialists globally. 12
Given limited access to palliative care, training neurologists to provide primary palliative care supplemented by virtual access to specialty palliative care is a promising approach with the potential to increase access to palliative care. 13 We conducted a randomized clinical trial evaluating a novel intervention consisting of palliative care training for community neurologists along with telemedicine access to team-based, specialist-level palliative care support for patients and families, which showed improvement in patient quality of life and completion of advanced care directives. 14 Here, we present here the results of a qualitative program evaluation embedded in the randomized clinical trial, which was designed to supplement the quantitative results by obtaining narrative descriptions of the experiences of patients and care partners under usual care and the novel intervention. Our research aims were to investigate: (1) unmet needs under standard neurology care, (2) how the intervention addressed the needs of patients and care partners, and (3) the remaining needs under the intervention condition.
Methods
Study design
We conducted a descriptive qualitative study 15 embedded in a pragmatic, multisite, randomized controlled trial of community-based outpatient palliative care between June 2018 and October 2020. Study reporting adheres to the Consolidated Criteria for Reporting Qualitative Studies (COREQ). 16 Main results of the randomized controlled trial will be reported elsewhere.
Clinician participants
Nine community neurology practices in the Rocky Mountain region were enrolled through the University of Colorado Anschutz Medical Campus and six practices in California were enrolled through the University of California, San Francisco. Inclusion criteria for clinicians included: providers of care for patients with Parkinson’s disease and related disorders, willingness to refer at least 21 patients over 2.5 years, and willingness to undergo an 8-h palliative care training. Thirty-four clinicians (19 from the Rocky Mountain region and 15 from California) participated in the study.
Patient and care partner participants
Patient inclusion criteria included fluency in English, age over 18 years, meeting UK Brain Bank criteria for probable PD or standard criteria for progressive supranuclear palsy, multiple system atrophy, vascular parkinsonism, Lewy Body dementia, or corticobasal degeneration, and high potential palliative care needs as assessed by the Brief Needs Assessment Tool. Exclusion criteria included a plan to change neurologists in 6 months, presence of additional chronic medical illnesses contributing to palliative needs, already receiving palliative or hospice services, or a lack of identified palliative care needs. Care partners could participate if present and willing to join the study, but were not required.
Study design
The intervention included primary palliative care training for community neurologists (through the Education in Palliative and End-of-Life Care program modified for Parkinson’s disease and related disorders 17 ), access to palliative care coaching (email, phone, and telephone access for enrolled clinicians to a palliative care specialist and palliative care trained neurologist to discuss challenging cases), and ability to refer patients to a multidisciplinary telemedicine palliative care team (including a nurse, pharmacist, social worker, and chaplain). The study used a stepped-wedge design 18 (Supplemental Figure 1), so all participating sites eventually received the intervention. For the first 12 months, all participants were assigned to Arm 1 (usual care). Every 6 months during years 2 through 4, four to five independent participating community neurologists were randomized to begin the intervention and entered Arm 2 (usual care with palliative care).
Qualitative substudy
At the time of entry into the trial, we asked all participating patients and care partners if they would participate in semi-structured interviews. Sampling for interviews aimed to increase heterogeneity across study sites, age, sex, disease severity, symptom burden, living location, and cognitive status. Participants were purposively sampled from each phase of the trial to capture experiences under both usual care and the intervention. Participants were sampled separately and not interviewed twice. Recruitment for interviews ended when theoretical sufficiency was reached among participants from each trial arm (usual care and intervention).
Standard protocols, registrations, and participant consents
The study protocol was approved by the institutional review boards of all sites and posted on ClinicalTrials.gov (NCT03076671). Written or verbal consent was obtained from all care partners and patients (or legally authorized representatives if the patients lacked capacity).
Data collection
We used an interview guide to elicit perspectives from participants on their experience with their care teams with and without the intervention (Supplemental Figure 2). 19 The guide was iteratively adapted to allow for investigation of emergent themes with concurrent analysis described below.
Interviews lasting up to 60 min were conducted by teleconference or phone individually with participants or with the dyad if requested. Three study authors (MD, RA, and JJ), who were trained and experienced in qualitative research methodology, served as interviewers . Regular meetings were held to ensure consistent use of the interview guides. Interviews were digitally recorded and transcribed verbatim. Data were managed using Atlas.ti version 9.1.3.
Data analysis
We conducted a qualitative program evaluation with a postpositivist orientation. We conducted a combined inductive and deductive thematic analysis.20,21 To conduct our thematic analysis, we used a combination of a deductive approach (using predefined codes based on the current neuropalliative literature and our prior experience) and an inductive approach (using the 6-step inductive approach described by Braun and Clark 20 ). We identified patterns in the data and applied conceptual descriptions that went beyond pure labeling and served an interpretive function. However, we did not extend our process to a full interpretive explanation as we did not transform the data to produce grounded theories or other fully integrated explanations for our findings. We identified a combination of semantic themes (e.g. participants naming social work as a helpful member of the multidisciplinary team) and latent themes (e.g. participants expressing a need for more in-depth counseling about the future). 22 Supplemental Figure 1 displays our coding tree. To evaluate intercoder reliability and consistency, analysts independently double or triple coded the first five transcripts. Throughout the process, the analysts discussed and categorized emergent themes, engaged in diagramming, wrote memos to reflexively summarize data, and evaluated for disconfirming cases.21,23,24 We triangulated our findings from patients and care partners and interviewer field notes with additional field notes from conversations with study coordinators. The iterative analytic process continued until new themes or insights were no longer being identified and new data could be managed within existing categories, 25 indicating we had reached theoretical sufficiency. 26 Interval debriefing about the thematic analysis was completed throughout with the larger study team. We also used field notes from study coordinators who interacted closely with participants and received frequent informal feedback. The field notes represent observations of coordinators from their work with all participants captured in study visit files and reflections on their interactions. We also interviewed each study coordinator to further clarify these observations. These data were used to triangulate, support, and extend observations elicited from patients and care partners.
Results
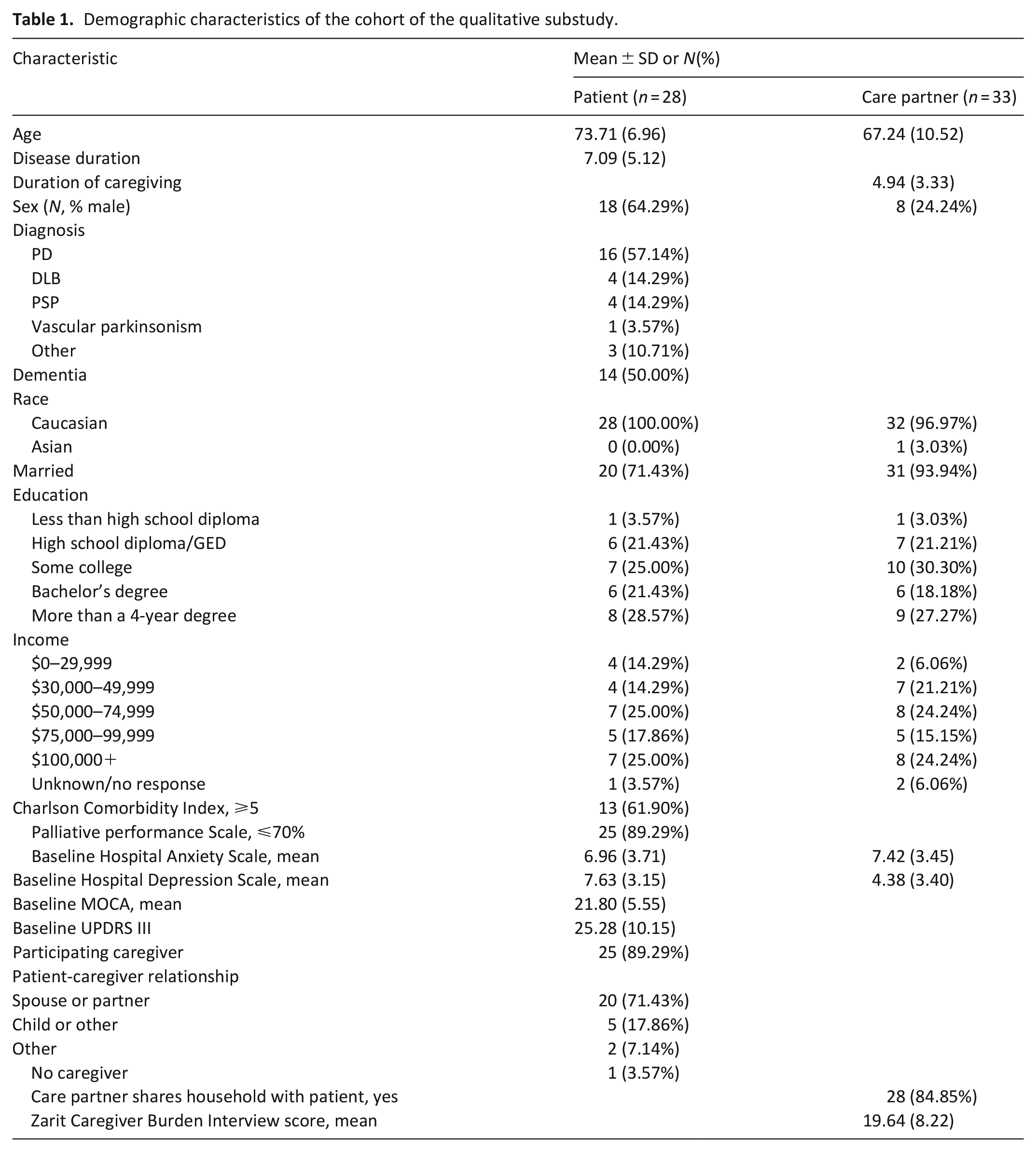
Clinical and demographic features of study participants
Of enrolled participants, 87.8% (354/403) patients and 90.4% (309/342) of care partners consented to be contacted about an interview. Participant characteristics are summarized in Table 1. Under usual care, we conducted 16 interviews with patients, 22 interviews with care partners, and 4 interviews with dyads. Under the intervention condition (mean time under intervention 127.2 days (SD = 120.5) for patients and 104.5 (SD = 95.9) days for care partners), we interviewed seven patients individually, nine care partners, and six dyads. We present themes regarding unmet needs under usual care, benefits of the intervention, and opportunities for improvement (Figure 1).
Demographic characteristics of the cohort of the qualitative substudy.
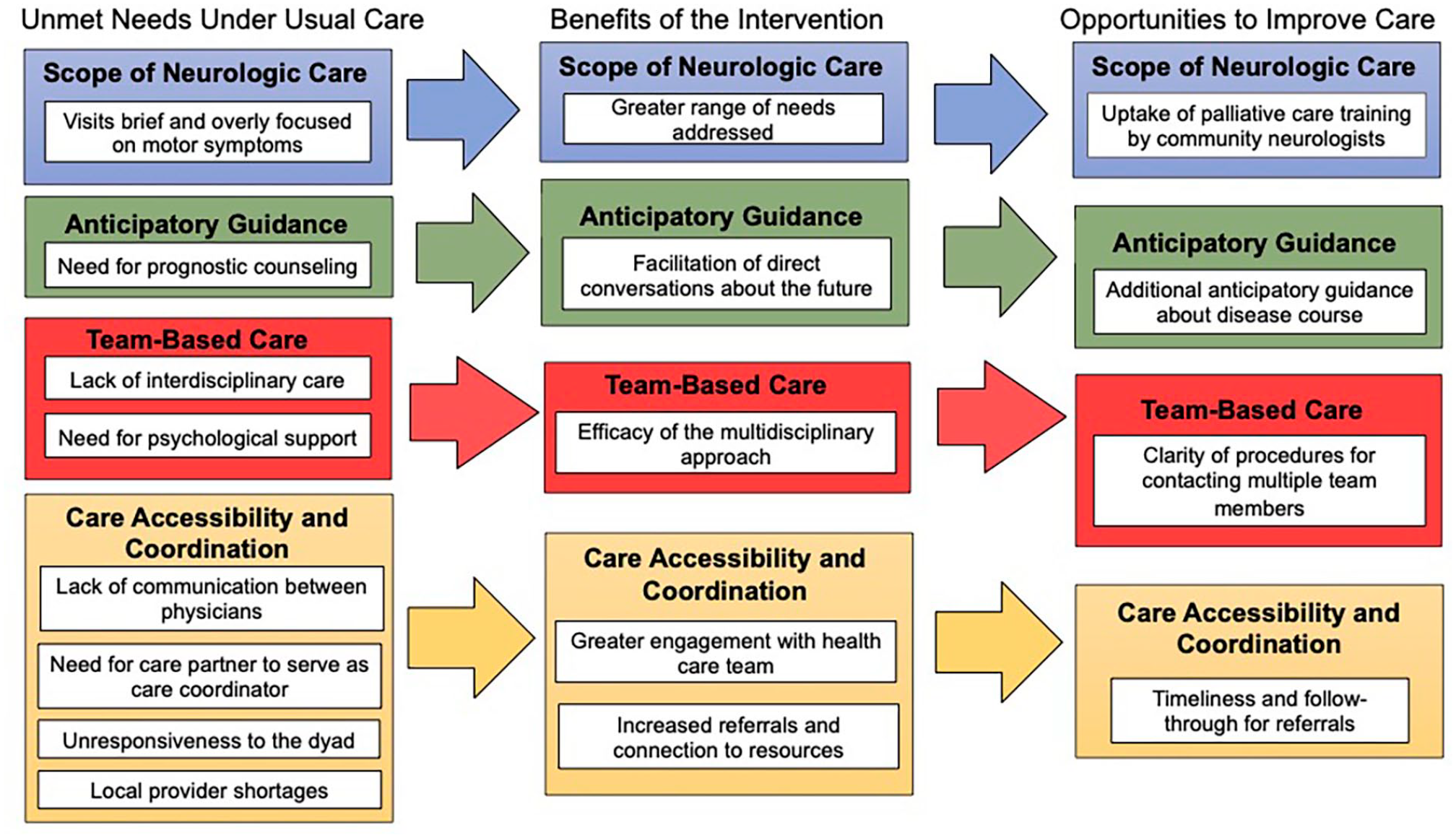
Emergent themes from patients and caregivers surrounding unmet needs under usual care, benefits of the palliative care intervention, and opportunities for improvement.
Unmet needs under usual care
Under usual care, we identified four main categories in participant descriptions of unmet needs. These are summarized in Table 2 and described in more nuance below.
Perspectives of people with Parkinson’s disease and related disorders and care partners about unmet needs under usual care.

Participants expressed appreciation for their neurologists and concern that their care is overly focused on motor symptoms. Primary neurologists were described as efficient and sometimes lacking in understanding about the full spectrum of needs created by Parkinson’s disease and related disorders. One care partner said: “We did not feel that the doctors here were. . .understanding how many difficulties he was having or challenges that he was having and we just wanted more help.” (Caregiver 20828). One patient described that the primary role of their neurologist is to “check the medicines to see how they’re working and to change them if we need to. And she kind of has me walk up and down the hall to see how much I have disintegrated from the last time. . .that’s pretty much it” (Patient 20307).
Multiple participants expressed a desire for anticipatory guidance about the disease course. One care partner said:
“[The patient] is constantly trying to get on the computer and research the disease and find out more information and so on and so forth, which puts her into these tailspins of repetitiveness and cyclical things in her brain. . .Any kind of educated opinion on what she’s going through would make her feel much, much more calm about the situation” (Care partner 10437)
Both patients and care partners wanted to learn from the experience of their clinicians about what to expect, even if their predictions were imperfect.
Most participants described their care team as consisting of themselves and physicians, with few mentions of other providers. One patient said “It kind of sounds funny to me, care team, because it’s really just the neurologist. . .I think it would be really swell if there was a counselor and a social worker there, and I’d be all over that but there isn’t” (Patient 20302). A common unmet need was the desire for counseling and mental health support to address mood symptoms and help to process the large changes in their lives.
Multiple participants were frustrated by the inaccessibility of their primary neurologist. One care partner said “there’s never an immediate answer to any kind of questions. . .you know appointments are always made, you know, sometimes months in advance. . .that just leaves us kind of spinning in the wind trying to deal with [the patient] and calm her down to whatever it is she’s freaking out about” (Care partner 10437). Participants in rural areas described local provider shortages, long wait times, neurologist turnover, and lack of relevant supportive services in their area.
Care partners in particular were frustrated about the lack of communication between different physicians. One care partner said “I’m managing all the different doctors. . .I always thought like the primary care doctor or the neurologist, main neurologist, would interact with all the different doctors and I don’t find that happening. . .there’s not one person looking out for him other than me. There’s not a medical person doing that and that’s frustrating” (Care partner 20828).
Benefits of the palliative care intervention
There were four main categories of perceived benefits of the palliative care intervention described by participants and affirmed by study coordinators. These are summarized in Table 3 and described in more nuance below.
Patient and care partner perspectives on benefits of the palliative care intervention.

Multiple patients and care partners expressed appreciation for spiritual care from the chaplain in reconnecting with things that are important in their life, regaining their sense of self, and processing disturbing thoughts about their mortality. A care partner said “the chaplain was really helpful in talking to [the patient] about coping with his problems and trying to help him with regaining his sense of self because there’s so many things [the patient] used to be able to do that he can’t do anymore” (Care partner 10429). One study coordinator mentioned that the chaplain was the person “people had the most alarm about seeing” initially but was “who most people wanted to see again and found the greatest benefit” (Coordinator 1). Study coordinators also specifically mentioned that the benefits of meeting with the pharmacist and social worker were initially underestimated by the patient.
Care partners appreciated more direct conversations about the predicted illness course. One described that “[the patient] had never really been told how it progresses and we got to hear that a little bit on Friday and to hear where he was in that progression, which was really great. . .[the study neurologist] was gentle, but she was direct and that was really helpful” (Care partner 21009).
Care partners appreciated extra time with providers and assistance with advance care planning. One explained that their primary neurologist “didn’t have enough time for all the questions I had so. . .he said I could do it at our next appointment with the [study team]. So, it’s just giving us extra support, so it’s wonderful.” (Care partner 20716) Many participants appreciated more points of contact with the health care team due to increased number of appointments and regular needs assessments.
Though these resources were not explicitly part of the intervention, participants described a helpful increase in outside referrals to physical therapy, exercises classes, and online educational resources. Within the intervention, study coordinators described that they often served “in a patient navigator role” to help participants connect with the appropriate members of the multidisciplinary team, saying that especially with overburdened care partners “sometimes they don’t even know what the team can do for them” (Coordinator 1).
Areas for improvement in the palliative care intervention
Areas for improvement identified by participants are summarized in Table 4 and described in more nuance below.
Patient and care partner perspectives on areas of improvement of the palliative care intervention.

One care partner expressed disappointment in the lack of change in their primary neurologist, saying “[the doctor] could have been under a rock for the couple years we were involved [in the study] because there’s nothing that. . .made any noticeable change” (Care partner 20311). Another patient stated, “I was told once that my [primary neurologist] completed the training. I think the care is good” (Patient 10207). There may have been variable uptake of the palliative care intervention between neurologists.
Most participants expressed appreciation for increased time spent on prognostication, but many desired even more information on this topic. One care partner expressed that they attended an appointment during the intervention to “find out what the future would hold. . .and I don’t think we got the answers we were requiring” (Care partner 10109).
The addition of the study team may have led to confusion about how to initiate contact with various team members or not knowing who to call. As all recommendations from the palliative care team had to be communicated through the primary neurologist’s office, several participants described perceived delays (of over one week) in receiving the referrals to outside services that had been discussed during their study appointment.
Discussion
Main findings
We present here the results of a qualitative study embedded in a randomized controlled trial to better understand patients and care partner experiences with a novel, multicomponent, community-based palliative care intervention. Perceived benefits of the palliative care intervention included the effectiveness of the multidisciplinary approach in addressing a wider range of needs from Parkinson’s disease and related disorders, facilitation of direct conversations about the future, greater engagement with the health care team, and increased referrals to community-based resources. These benefits were primarily related to interactions with members of the specialty palliative care team via telemedicine and telephone. Remaining areas of improvement included greater uptake of the intervention by community neurologists, additional anticipatory guidance about the future, and clarification of processes surrounding communication with the team. Our study provides novel evidence for the potential benefits, as well as limitations, of primary provider palliative care education and telehealth to support people with neurodegenerative illness receiving care outside of academic centers. This sheds additional light on the quantitative findings, which found that the intervention improved patient QOL but not care partner burden. 14 Our study was conducted in the US but has implications for care delivery internationally, particularly in resource-limited settings such as those targeted by Project ECHO to increase virtual care access.27 –29
What this study adds?
The emergent themes under standard of care corroborate existing literature showing many unmet palliative care needs in those with neurodegenerative illness in current chronic care models,30 –33 indicating that this was a prime population to undergo this intervention. Our findings show how interdisciplinary, specialty palliative care delivered by telehealth can address a wider array of symptoms, facilitate connection to community resources, increase engagement with the health care team, and improve understanding about what to expect in the future. The benefits from meeting with the specialty palliative care team via telemedicine highlight an opportunity to better serve geographic populations with lower access to care. Several participants initially underestimated the benefit of seeing certain providers, in particular the chaplain or pharmacist, suggesting that routine referrals to many of these services could be helpful. Spiritual care has been previously cited by people with Parkinson’s disease as an important source of coping and resilience, but is rarely incorporated into neurology appointments. 32 For participants in this study, chaplaincy helped with coping with loss of ability and reconnection with things that are important to them. Study coordinators noted they often served as de facto care navigators when participants were overwhelmed, highlighting a key unmet need in neurodegenerative disease 34 that is often addressed by overburdened care partners or not at all.
Our findings also indicate that this novel palliative care intervention can be further optimized. Firstly, the addition of another team, in absence of a care navigator, may have exacerbated care coordination issues. There were delays in receiving promised referrals, confusion about who to call for which issue, and a perceived lack of communication between providers. Care coordination issues have previously been described in home-based and other palliative care models; 35 efforts are underway to improve electronic and other information sharing. 36 In this intervention, the impact of the specialty-based palliative care team was more salient to patients and care partners than changes in their primary neurologist. Eight hours of training may be insufficient to significantly change practice but sufficient to convince neurologists of the potential benefits of referring to and collaborating closely with specialty palliative care teams. In another study, neurologists with low referral rates to palliative care were more likely to state that they already provided for the palliative needs of their patients, despite patients in the same study articulating many unmet needs. 37 Earlier incorporation of training may promote openness to palliative care approaches and is a gap in many neurology programs. 38 However, many of the perceived benefits that stemmed from the full multidisciplinary team would not be conferred solely by increased palliative care training for neurologists.
Despite participant report of improved counseling about the future, many participants expressed a remaining desire for more. Uncertainty about the future remains a core issue in Parkinson’s disease and related disorders even with increased palliative care support, which could be due to the need for more education around this recommendation, time barriers to implementation, or a need for intensive and ongoing prognostic counseling at many different points along the disease course.
Strength and limitations of the study
Strengths of our study include the inclusion of dyads, richness of the interview data, and triangulation of these findings by study coordinators. Our study was embedded in a randomized clinical trial, so our findings may not represent the full range of experiences of people with Parkinson’s disease and care partners. However, our recruitment from community practice provides perspectives less often captured in academic research. Our findings may not be generalizable to those with high medical comorbidity as they were excluded from this study and their palliative needs may be different.
In summary, our study highlights the relative strengths and weaknesses of a novel, multicomponent, community-based palliative care intervention that can inform clinical practice and future implementation research in this high need population, particularly for people receiving care outside of academic centers. All clinicians caring for people with Parkinson’s disease and related disorders should screen for bothersome non-motor symptoms, provide regular prognostic counseling, and refer to specialty palliative care services earlier in the course of illness. Future interventions should be designed to promote uptake of palliative care training by community neurologists, further optimize prognostic counseling, and streamline referral to specialty palliative care teams with improved care coordination.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231219923 – Supplemental material for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231219923 for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners by Meredith A Bock, Zachary A Macchi, Krista L Harrison, Maya Katz, Megan Dini, Jacqueline Jones, Roman Ayele, Jean S Kutner, Steven Z Pantilat, Christine Martin, Stefan Sillau and Benzi Kluger in Palliative Medicine
Supplemental Material
sj-pdf-3-pmj-10.1177_02692163231219923 – Supplemental material for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners
Supplemental material, sj-pdf-3-pmj-10.1177_02692163231219923 for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners by Meredith A Bock, Zachary A Macchi, Krista L Harrison, Maya Katz, Megan Dini, Jacqueline Jones, Roman Ayele, Jean S Kutner, Steven Z Pantilat, Christine Martin, Stefan Sillau and Benzi Kluger in Palliative Medicine
Supplemental Material
sj-pptx-2-pmj-10.1177_02692163231219923 – Supplemental material for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners
Supplemental material, sj-pptx-2-pmj-10.1177_02692163231219923 for Does a novel community-based outpatient palliative care intervention for Parkinson’s disease and related disorders improve care? Qualitative results from patients and care partners by Meredith A Bock, Zachary A Macchi, Krista L Harrison, Maya Katz, Megan Dini, Jacqueline Jones, Roman Ayele, Jean S Kutner, Steven Z Pantilat, Christine Martin, Stefan Sillau and Benzi Kluger in Palliative Medicine
Footnotes
Author contributions
Bock MA: Design, Execution, Analysis, Writing, and Editing of final version of manuscript. Macchi ZA: Execution, Analysis, and Editing of final version of manuscript. Harrison KL: Analysis, Writing, and Editing of final version of manuscript. Katz M: Design, Execution, and Editing of final version of manuscript. Dini M: Execution, Analysis, Editing of final version of manuscript. Jones J: Design, Analysis, Editing of final version of manuscript. Ayele R: Execution, Analysis, and Editing of final version of manuscript. Kutner JS: Design and Editing of final version of manuscript. Pantilat SZ: Design, Execution, and Editing of final version of manuscript. Martin C: Execution, Analysis, and Editing of final version of manuscript. Sillau S: Analysis and Editing of final version of manuscript. Kluger B: Design, Execution, Analysis, and Editing of final version of manuscript.
Consent and data sharing
Anonymized data not published within this article will be made available by reasonable request from qualified investigators.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work came from NINR R01NR016037.
Ethical approval
We have included details about IRB approval and consent in the Methods section.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
