Abstract
Background:
Studies have shown the risk factors for COVID-19 severity in children, including comorbidities, but information on the infection course in children with life-limiting conditions is sparse.
Aim:
To describe the effect of COVID-19 on pediatric patients receiving palliative care due to life-limiting conditions.
Design:
We conducted retrospective cohort study. The WHO Clinical Progression Scale was used to measure COVID-19 severity.
Setting/participants:
Seven of the 24 invited pediatric palliative care centers participated in this study. We analyzed the medical records of children under palliative care with confirmed COVID-19 (January 2020–April 2022)
Results:
Records of 60 patients with COVID-19 aged 0.24 to 21.6 years (mean (SD); 9.8 (6.6)) were collected. The largest group of patients with COVID-19 was children with congenital malformations and chromosomal abnormalities (42%); the most common manifestation was fever (85%). Bacterial coinfection was confirmed in 17 (28%) children. Fifteen (25%) children required hospitalization, including four admitted to the Intensive Care Unit. Mild COVID-19 was identified in 44 (73%) children, moderate in 11 (18%), severe in 3 (5%), and death in 2 (3%). Six of the 20 eligible children were vaccinated against SARS-CoV-2, followed by 16 mothers and fathers.
Conclusion:
In the study population initial presentation of COVID-19 was predominantly a mild; however, the small sample size precluded definitive conclusions. For children under palliative care, we should identify if they have an advance care plan for COVID-19, such as desires for intensive care support. Further studies are needed to define the short and long-term effects of COVID-19 in children with life-limiting conditions.
Preliminary evidence suggests that children with comorbidities have a higher risk of infection and a more severe course of COVID-19; however, there are no summarized data on the course of COVID-19 in children with life-limiting conditions.
Despite severe and untreatable disease, the course of COVID-19 was mild or asymptomatic in 75% of the study population. Every fourth patient with COVID-19 required hospitalization including intensive care.
Fatal outcome was observed in 3% patients with COVID-19.
Low level of vaccination in the study population was observed.
In the population of patients with life-limiting conditions under palliative care, we should identify whether they have an advance care plan for COVID-19, such as a desire for intensive care support.
Further studies are needed to define the short- and long-term effects of COVID-19 in children with life-limiting conditions.
There is a need to educate families about the benefits of vaccination.
Introduction
Children and adolescents under 18 years constituted 17% of COVID-19 cases with mortality less than 0.1%.,1,2 Many studies have shown a higher risk of severe COVID-19 in children with comorbidities but detailed information on the course of COVID-19 in children with life-limiting conditions is sparse.3–11 To date, there have been no reports on the course of COVID-19 in children qualified for palliative care due to LLC.
This study aimed to describe the effects of COVID-19 in children and adolescents with life-limiting conditions.
Methods
Design
We conducted retrospective cohort study.
Setting
Pediatric palliative care in Poland is provided in two models: a home-based care called “home hospice” and a stationary care called stationary hospice. Stationary hospices function like a hospital ward and are dedicated to children who cannot be treated at home, particularly for children who are orphaned or deprived of a legal guardian. Children are qualified for palliative care in accordance with the current standards. 12 We invited 24 pediatric palliative care centers to participate in the study.
Population and sampling
Seven centers participated in this study. Among all patients under palliative care (January 2020–April 2022), those with confirmed SARS-CoV-2 infection were included in the study.
Data collection
The electronic questionnaire was sent (Supplemental File), and we conducted a retrospective review of the medical records of children diagnosed with COVID-19. The WHO Clinical Progression Scale was used to measure COVID-19 severity. 13
Statistical analysis
Data were analyzed using STATISTICA 13·5 (StatSoft). For the analysis of dichotomous variables, such as the drugs used or symptoms of infection, the Chi2 test was used. The Mann-Whitney test was used to analyze continuous variables in the two groups’ differentiation, and Kruskal-Wallis statistics were used to compare many groups. All calculated data were considered significant at a 0.05 level.
Ethical issues
This retrospective study used data obtained for clinical purposes. We consulted the Bioethics Committee who determined that our study did not require ethical approval.
Results
Records of 60 patients with confirmed COVID-19, aged 0.24–21.6 years (mean (SD), 9.8(6.6)) were collected (Table 1).
Clinical characteristics of patients with confirmed SARS-CoV-2 infection.
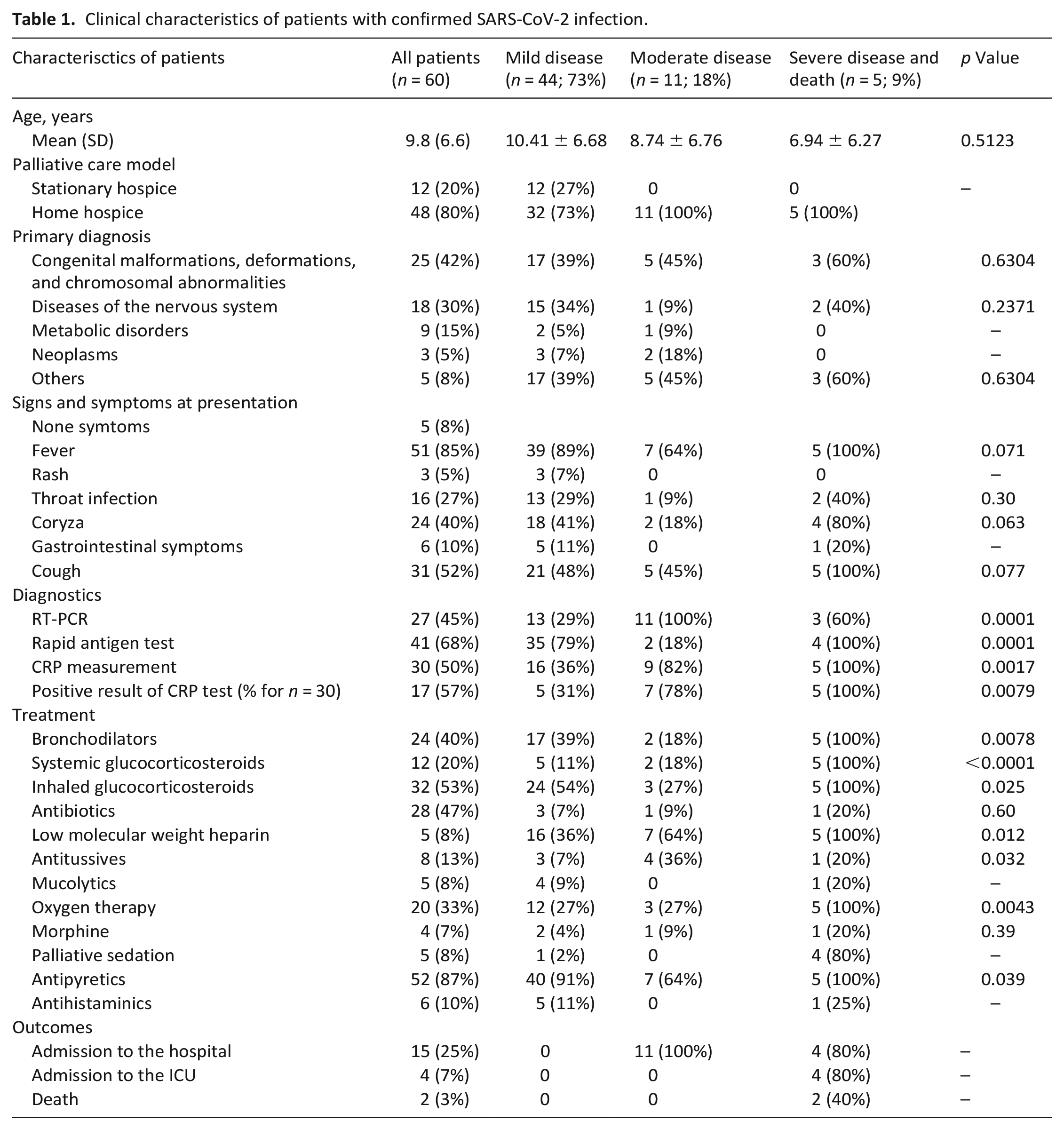
The most common manifestation was fever (Table 1).
Antipyretics were administered to 55 children, inhaled steroids to 32, inhaled bronchodilators to 24, and oxygen therapy to 20. A C-reactive protein (CRP) test was performed in 30 children; a positive result was obtained in 17 patients, and antibiotic therapy was administered in all patients. Additionally, antibiotic therapy was administered without CRP measurement in 12 patients treated at home. Compared to children without antibiotic therapy, they more frequently presented with cough (p = 0.0028), had higher punctuation on the WHO Clinical Progression Scale (p = 0.0083), and more frequently required treatment with bronchodilators (p = 0.0004), systemic glucocorticosteroids (p = 0.046), low-molecular-weight heparin, and oxygen (p = 0.0016).
Fifteen (25%) children required hospitalization, including four children admitted to the Intensive Care Unit (ICU). We observed a more frequent administration of antitussives, systemic glucocorticosteroids, and antibiotics in children treated at the hospital than in those treated at home (Table 2). All children admitted to the ICU required sedation, and two required morphine. Among children treated at home, one was sedated and required additional morphine administration. Among the ICU patients, one child died, one still required mechanical ventilation, and two children were discharged. The patient characteristics according to COVID-19 severity are shown in Table 1.
Clinical characteristics of patients treated at home and hospital.
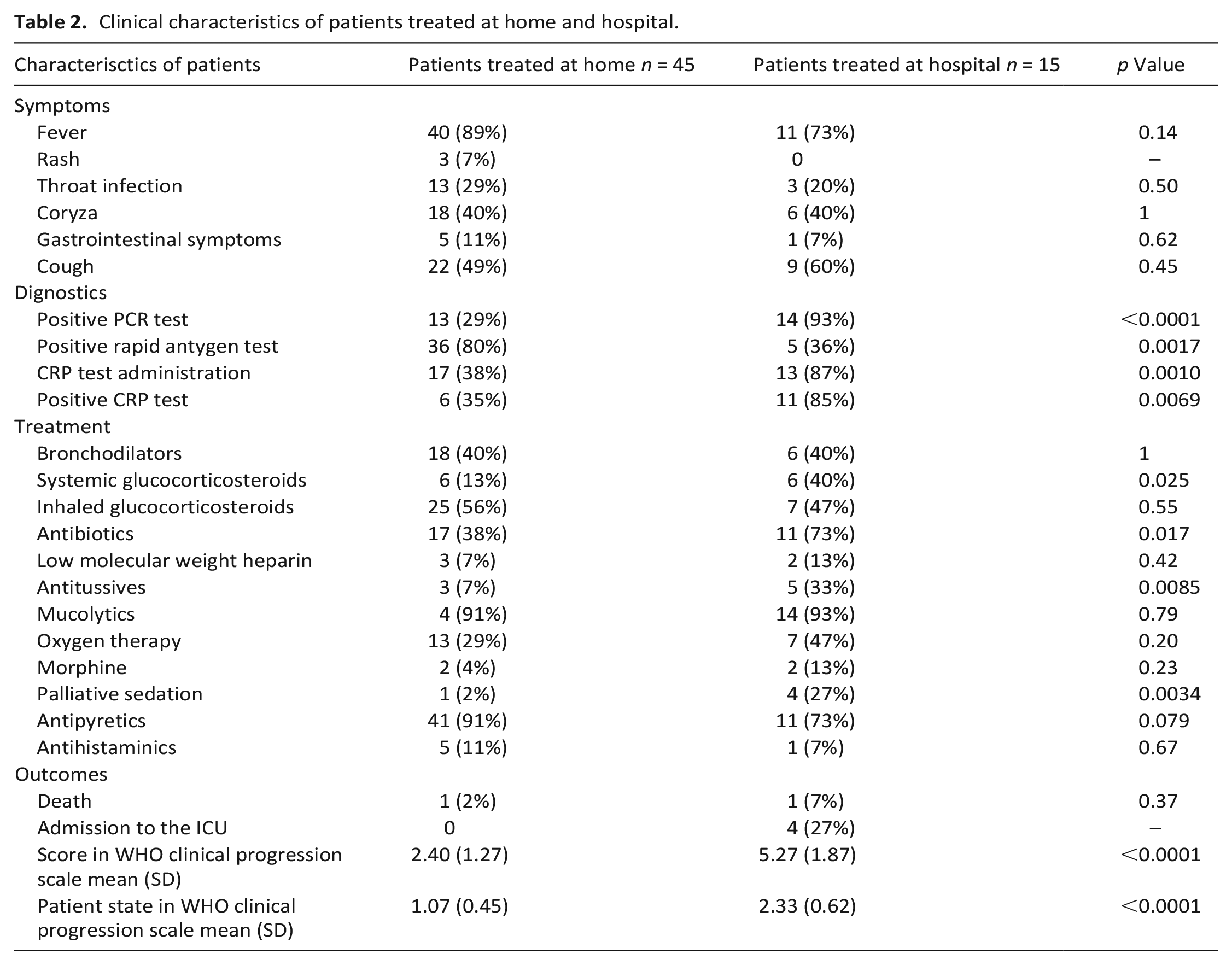
Two deaths were recorded during the fourth pandemic wave. First patient was transferred from home to the ICU, second died at home where the parents started ineffective resuscitation.
At the time of the COVID-19 diagnosis, 20 children were eligible for vaccination against SARS-CoV-2. Of the 48 home hospice families, 16 mothers (33%) and 16 fathers (33%) and only 2 siblings were vaccinated before child was diagnosed with COVID-19.
Discussion
Main findings of the study
We showed that despite severe and untreatable diseases, the course of COVID-19 was predominantly mild or asymptomatic however we observed a high rate (25%) of hospitalization and we describe fatal outcome in 3%. Only one third of eligible children and parents were vaccinated.
What this study adds?
To the best of our knowledge, this is the first multicenter study on COVID-19 in children and adolescents with life-limiting conditions under palliative care. There have been reports of a higher risk of infection and more severe course of COVID-19 in children with comorbidities.3–11 While most studies consider the impact of preexisting medical conditions on the course of SARS-CoV-2 infection, to the best of our knowledge, there are no summarized data on the course of COVID-19 in children with life-limiting conditions. We found only one study directly relevant to children with life-limiting conditions; the authors analyzed the sera of 39 patients and found a 26% seroprevalence for SARS-CoV-2, but all seropositive patients were previously unrecognized despite severe comorbidities. 14
Some studies present a severe course of COVID-19, including death in children with underlying conditions.5–7,15,16 In our study population, children with congenital malformations and chromosomal abnormalities most often suffered from COVID-19 and two patients from this group had a fatal outcome. Four of the 60 patients required admission to the ICU with one fatal outcome; another patient died at home after attempting resuscitation. The indications for intensive therapy in children diagnosed with incurable diseases should be considered. Patient, who died after respiratory therapy was in good condition before the infection and not in the terminal phase of the disease. The patient was under palliative care in accordance with the principles defined by WHO: “palliative care for children begins when illness is diagnosed and continues regardless of whether or not a child receives treatment directed at the disease”. 12 Thus, intensive treatment did not have the characteristics of futile therapy. In the second case, resuscitation was initiated by the parents. It is necessary to pay attention to the need for prior discussions with caregivers about the type and goals of treatment. 17 However, our long-term experience shows that not all parents are able to accept an unfavorable prognosis and not attempt resuscitation. Due to the limited resources of personnel and infrastructure, a difficult and probably controversial question should be raised regarding the validity of using intensive care in patients with an incurable disease. This aspect of care has also been recognized by the WHO, which recommends identifying, in all patients with COVID-19, whether they have an advance care plan for COVID-19 (such as desires for intensive care support) and respecting their priorities and preferences to tailor the care plan and provide the best care irrespective of treatment choice. 18
Low levels of vaccination were observed in the study population. Regarding information about COVID-19 vaccines, parents showed more trust in family doctors than in scientists and the government. 19 The most important reasons for the decline in vaccination were doubts about the safety and effectiveness of vaccines, fear of side effects, and the low self-perceived threat of COVID-19 for children. In the authors’ opinion, pediatric palliative care teams should persuade children’s families to vaccinate children under hospice care as the only way to significantly reduce the risk of hospitalization.
Strengths and limitations of the study
This study was limited by its retrospective design and small sample size, which precluded definitive conclusions. Nevertheless, from another point of view, it should be noted that 415 children were under palliative care in 2020, and 453 in 2021. In Poland 1963 children were treated in hospices in 2020 and 2125 in 2021; thus, the study population comprised 21% of all patients under palliative care. We did not collect data such as ethnicity—population with Polish national identifications constitutes 97.7% of the total population of Poland—and analysis of additional demographic factors associated with poorer outcomes was not possible.
Conclusions
We showed that most children in the study population presented with mild and moderate courses of COVID-19. The different courses of COVID-19 in children with LLC may suggest the presence of factors other than the coexistence of severe conditions that determine the more severe course of SARS-CoV-2 infection. However, further studies examining the effects of specific comorbidities are required to determine the effects of LLC on COVID-19 severity. In the population of patients with life-limiting conditions under palliative care, we should identify whether they have an advance care plan for COVID-19, such as a desire for intensive care support. Defining the high-risk group for severe COVID-19 could help in decision making regarding hospitalization priority in children with LLC. In the face of reports that children with preexisting conditions are at a greater risk of severe COVID-19 and its associated mortality, vaccination should be promoted with greater emphasis. Owing to the lack of literature data, further studies are needed to define the short- and long-term impact of COVID-19 infection in children with life-limiting conditions.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231206928 – Supplemental material for COVID-19 in pediatric palliative care patients: Multicenter, retrospective cohort study
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231206928 for COVID-19 in pediatric palliative care patients: Multicenter, retrospective cohort study by Aleksandra Korzeniewska-Eksterowicz, Olga Brzezinska, Urszula Dryja, Dominka Matczak, Andriy Sopilnyak, Eugenia Szuszkiewicz, Łukasz Przysło, Krzystof Szmyd, Katarzyna Jabłońska, Piotr Krych, Agnieszka Wojtków-Zielińska, Edyta Wąsińska and Maciej Niedźwiecki in Palliative Medicine
Footnotes
Author contributions
Dr. Aleksandra Korzeniewska-Eksterowicz and Olga Brzezinska conceptualized the study, contributed to the methodology, and performed the formal analysis. Dr. Aleksandra Korzeniewska-Eksterowicz, Dr. Urszula Dryja, Dr. Dominka Matczak, Dr. Andriy Sopilnyak, Dr. Eugenia Szuszkiewicz, Dr. Łukasz Przysło, Dr. Krzystof Szmyd, Dr. Katarzyna Jabłońska, Dr. Piotr Krych, Agnieszka Wojtków-Zielińska, Edyta Wąsińska, and Dr. Maciej Niedźwiecki contributed to the acquisition of data for the work. Dr Aleksandra Korzeniewska-Eksterowicz prepared the original draft of the manuscript. Dr. Aleksandra Korzeniewska-Eksterowicz and Dr. Maciej Niedźwiecki contributed to reviewing and editing the manuscript. All authors had full access to all the data in the study, participated in data interpretation, revised the manuscript, and approved the final version of the manuscript for publication. All authors contributed important intellectual content during manuscript drafting or revision, and accepted accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work were appropriately investigated and resolved. Dr. Aleksandra Korzeniewska-Eksterowicz is responsible for the honest, accurate, and transparent reporting of the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
This study was conducted retrospectively using data obtained for clinical purposes; according to the Bioethics Committee of the Medical University of Lodz guidelines, ethical approval was not required.
Data sharing
The data and analytical materials related to this study were maintained and managed according to organizational guidelines and ethical regulations. This information will not be made publicly available in the interest of patient confidentiality and anonymity. Requests for further information were directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
