Abstract
Background:
Trial participant recruitment is an interactional process between health care professionals, patients and carers. Little is known about how clinicians carry out this role in palliative care trials and the reasons why they do or do not recruit participants.
Aims:
To explore how clinicians recruit to palliative care trials, why they choose to implement particular recruitment strategies, and the factors that influence their choices.
Design:
A qualitative multiple case study of three UK palliative care trials. Data collection included interviews and study documentation. Analysis involved developing and refining theoretical propositions, guided by the ‘6Ps’ of the ‘Social Marketing Mix Framework’ as an a priori framework (identifying participants, product, price, place, promotion and working with partners). Framework Analysis guided within and then cross-case analysis.
Settings/participants:
Study investigators and research staff (n = 3, 9, 7) from trial coordinating centres and recruitment sites (hospice and hospital).
Results:
Cross-case analysis suggests the ‘Social Marketing Mix Framework’ is useful for understanding recruitment processes but wider contextual issues need to be incorporated. These include the ‘emotional labour’ of diagnosing dying and communicating palliative and end-of-life care to potential participants and how the recruitment process is influenced by the power relationships and hierarchies that exist among professional groups. These factors can lead to and support paternalistic practices.
Conclusions:
Those planning trials need to ensure that trial recruiters, depending on their experience and trial characteristics, have access to training and support to address the ‘emotional labour’ of recruitment. The type of training required requires further research.
Keywords
More high quality trials in palliative care are needed to improve the evidence base that underpins clinical practice but trials can struggle to achieve their recruitment targets.
Trial recruitment challenges, including clinician gatekeeping, can be amplified in the palliative care context.
Clinicians have a key role in the recruitment process, but the reasons why they do or do not recruit to palliative care trials is poorly understood.
The ‘Social Marketing Mix Framework’ is useful for understanding recruitment processes but wider contextual issues need to be incorporated.
The contextual issues include the ‘emotional labour’ of diagnosing dying, communicating palliative and end-of-life care to potential participants, and the power relationships and hierarchies that exist among professional groups.
These factors can lead to and support paternalistic recruitment practices.
Those planning trials need to ensure that trial recruiters have access to training and support to address the ‘emotional labour’ of recruitment.
Training will be dependent on trial characteristics and clinician experience but may need to include how to discuss a palliative care trial.
The type of training and support required to address this recruitment barrier requires further research.
Background
Recruiting sufficient numbers of participants, especially to randomised controlled trials, is a prominent challenge in palliative care research.1,2 Trials can be sub-optimal with fewer than 50% of randomised controlled trials achieving their recruitment targets 3 with reports of palliative care trials only reaching their targets in 37% of cases. 4 Struggling to achieve statistical power means the trial has the potential to be slow and expensive5 –7 and more importantly participants will have received an intervention with uncertain benefit and on study completion researchers may still be unable to determine whether the intervention does more good than harm.3,8
Why trials struggle to achieve their recruitment targets is complex with some issues not unique to palliative care research such as patient9,10 and clinician11,12 concerns about randomisation, blinding and placebos. Contextual issues, such as underfunding, 13 limited infrastructure,14,15 high attrition rates 16 and patient related factors such as a high prevalence of cognitive impairment 17 contribute to these recruitment challenges in palliative care. Patients are seen as vulnerable especially at the end of life which can lead to clinician gatekeeping. 18 Gatekeeping is not unique to palliative care research 19 but is a particularly challenging issue in this population.20,21
Clinicians play a key role in the recruitment process, but why they do or do not recruit to palliative care trials is poorly understood. Recruitment is not a single event and is often a lengthy and complex process typically involving three steps; identifying, approaching and consenting. 22 It occurs in real time, in real clinical settings and it can be a difficult activity as it disrupts the usual clinician/patient relationship. 23
What strategies may facilitate trial recruitment3,24 or to research studies in general 22 is hampered by a lack of high-quality evidence. A review exploring recruitment barriers and facilitators in palliative care trials using the ‘6 Ps’ of the ‘Social Marketing Mix Framework’ as a theoretical framework was conducted. 25 The ‘6 Ps’ are; identifying participants, product, price, place, promotion and working with partners (see Table 1 for definitions). 26 The review found that the evidence underpinning the barriers and facilitators to palliative care trial recruitment was largely anecdotal and more methodological research was needed. It suggested the ‘6 Ps’ may help researchers better understand recruitment processes but further exploration was required. 25
The ‘6 Ps’ of the ‘Social Marketing Mix Framework’. 26

This study aims to address this gap in knowledge by exploring how clinicians recruit patients and carers to palliative care trials, why they choose to implement particular recruitment strategies, and the factors that influence the decisions they make.
Methods
Research question
How do health care professionals recruit patients and their family carers to palliative care randomised controlled trials and why do they use certain strategies during the recruitment process?
Design
A retrospective, descriptive qualitative multiple case study was conducted following Yin’s 27 approach with a critical realist lens 28 being applied throughout the study (further details below).
Case definition
In this study cases were defined as; UK palliative care randomised controlled trials aimed at adult patients with incurable cancer and/or advanced, progressive non-malignant disease and their family carers; and either the primary endpoint was symptom control and/or quality of life; or it tested an intervention that was clearly a palliative care intervention and the study primary endpoint was survival (further details in Table 2).
Case and participant inclusion criteria.

Case selection
Cases were consecutively screened and identified by LD in consultation with CW and NP from the trial databases listed in Table 2. There were 18 eligible trials available at the time of screening (from 11/2016 to 10/2017). Cases were chosen that had a variety of study designs and were recruiting in different settings to reflect theoretical replication. Yin argues that study findings are more robust if cases corroborate each other despite having contrasting characteristics. 27 Practical issues also influenced case selection such as the number of eligible trials available in the UK at the time of sampling and a small number of trials could not be included because research team members were involved in the trial.
Case recruitment
The Chief Investigators of the three selected cases were approached by email and asked if they agreed for their trial to be a case.
Participant definition
Professionals with different recruitment roles, from both the study coordinating centre and clinical recruitment centres within each case, were eligible (see Table 2). A study coordinating centre was defined as overseeing the conduct of a trial 29 while a recruitment centre was a clinical setting where recruitment activity took place. 30
Participant recruitment
The Chief Investigator of the selected cases circulated study information to eligible participants. Participants were then asked to suggest other individuals with relevant experience. Recruitment ceased within the case when the pool of eligible participants who were willing to participate was exhausted. Participants provided verbal consent.
Theoretical propositions
Initial theoretical propositions (predicted theory about what may be learned from examining the case) were developed to guide data collection and analysis, influenced by the ‘Social Marketing Mix Framework’, 26 literature review findings 25 and wider trial recruitment literature. Yin recommends their use to explore the deeper reasons for what can be observed, reflecting a critical realist stance, and also so study findings can be generalised beyond the ‘cases’ through ‘analytical generalisation’. 27 Yin provides little practical guidance on how theoretical propositions work alongside an a priori theoretical framework, as in this study. This influenced the decision to develop only a small number of theoretical propositions relating to only three of the ‘Ps’ in the ‘Social Marketing Mix framework’. Product, working with partners and place were specifically chosen because of literature review findings and issues raised within the general trial literature. For example, being able to access dedicated research staff was the strategy most discussed in the literature review. 25
They were;
Study design influences how recruiting staff undertake the process of recruitment and the strategies they use. (Product)
The involvement of specific research staff in the recruitment process impacts on how well the trial meets its recruitment target. (Working with Partners)
How recruiting staff undertake the recruitment of patients or carers is influenced by their professional role. (Working with Partners)
Where recruitment activity takes place may influence the recruitment process. (Place)
Data collection
Data were collected from each case consecutively. A telephone interview was conducted with each participant to capture multiple perspectives of the recruitment process and trial documentation was collated to understand how these processes were formally communicated.27,31 Online trial documents available in the public domain were identified and participants were asked to suggest documents. The interview topic guide was iteratively developed throughout the study and covered; recruitment procedures, exploration of phraseology used to discuss the trial with participants, how well the trial had recruited, factors that helped or hindered recruitment, recruitment strategies and lessons learnt (see Supplemental Material). LD, a nurse researcher with specialist palliative care clinical and research experience, conducted the interviews for her PhD. Participants were not known to LD. Interviews were audio recorded, anonymised, transcribed and field notes were made.
Data analysis
Data analysis was an iterative process with framework thematic analysis 32 being used to compare and contrast data within and then across cases to identify patterns. This approach involves five interconnected stages: familiarisation, identifying a thematic framework, indexing, charting, and mapping and interpretation. 32 . The ‘6 Ps’ were used as the a priori analytical framework. Raw data was reviewed, labelled, sorted and synthesised; descriptive accounts were developed by identifying key dimensions/elements, refining categories and developing classifications and finally explanations were developed to account for the data. Analysis was carried out by LD with critical input from CW and NP.
Research ethics and approvals
Approval was obtained from Lancaster University Faculty of Health and Medicine Research Ethics Committee (Reference number: FHMREC15042, 22nd February 2016). The study is reported in line with the DESCARTE checklist. 33 Reflexivity was considered throughout the study, 34 including regular supervisor meetings, to try and limit subjectivity and bias as recommended by Yin. 27 Participation was voluntary and anonymisation was assured.
Findings
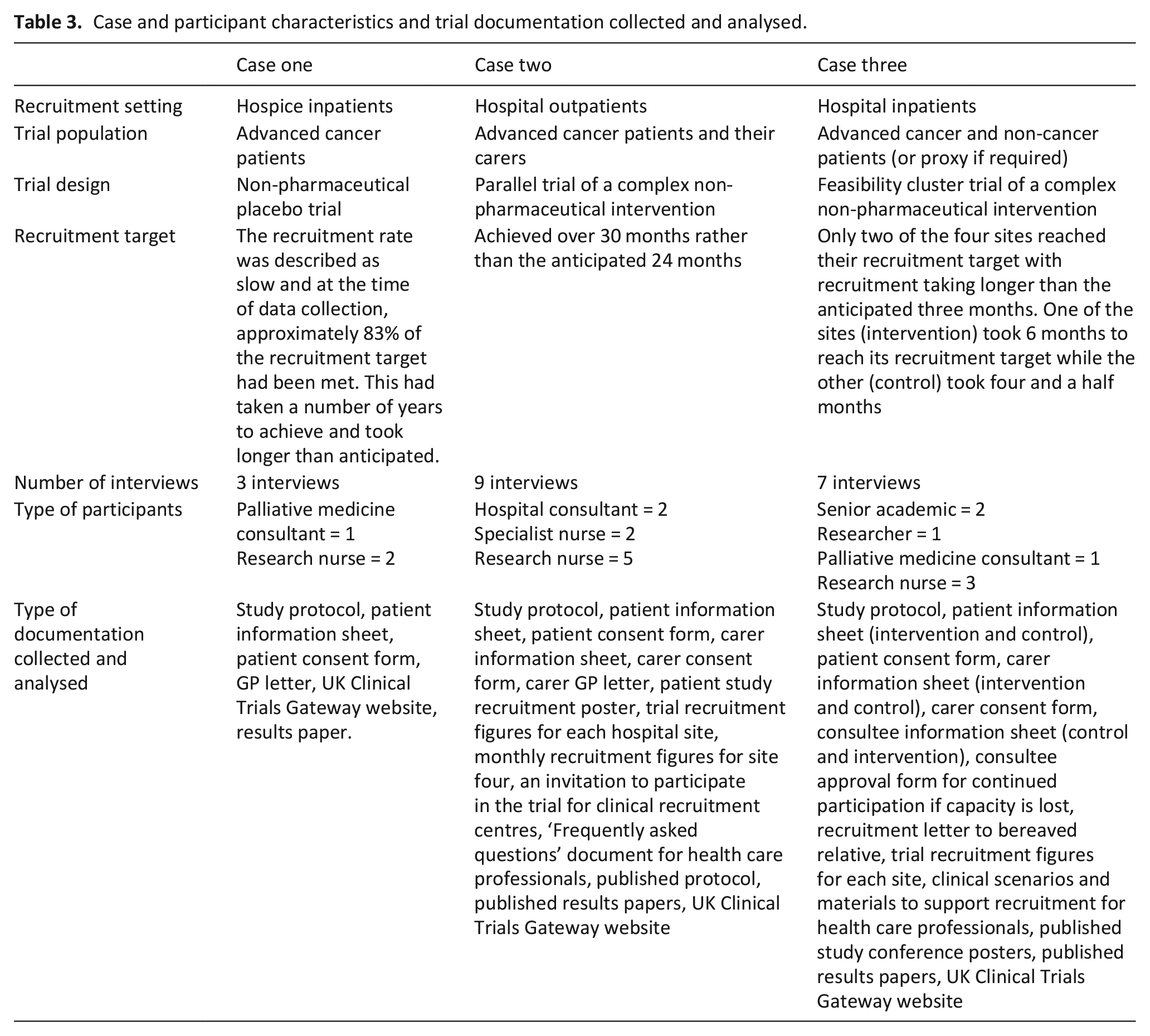
Data collection occurred between March 2017 and June 2018. Three cases were included and 19 participants (n = 3, 9, 7) took part with the mean interview length being 39 min (range 25–60 min). Study findings are presented as a cross case analysis with data from each of the cases dispersed throughout (see Table 3 for case and participant characteristics).
Case and participant characteristics and trial documentation collected and analysed.
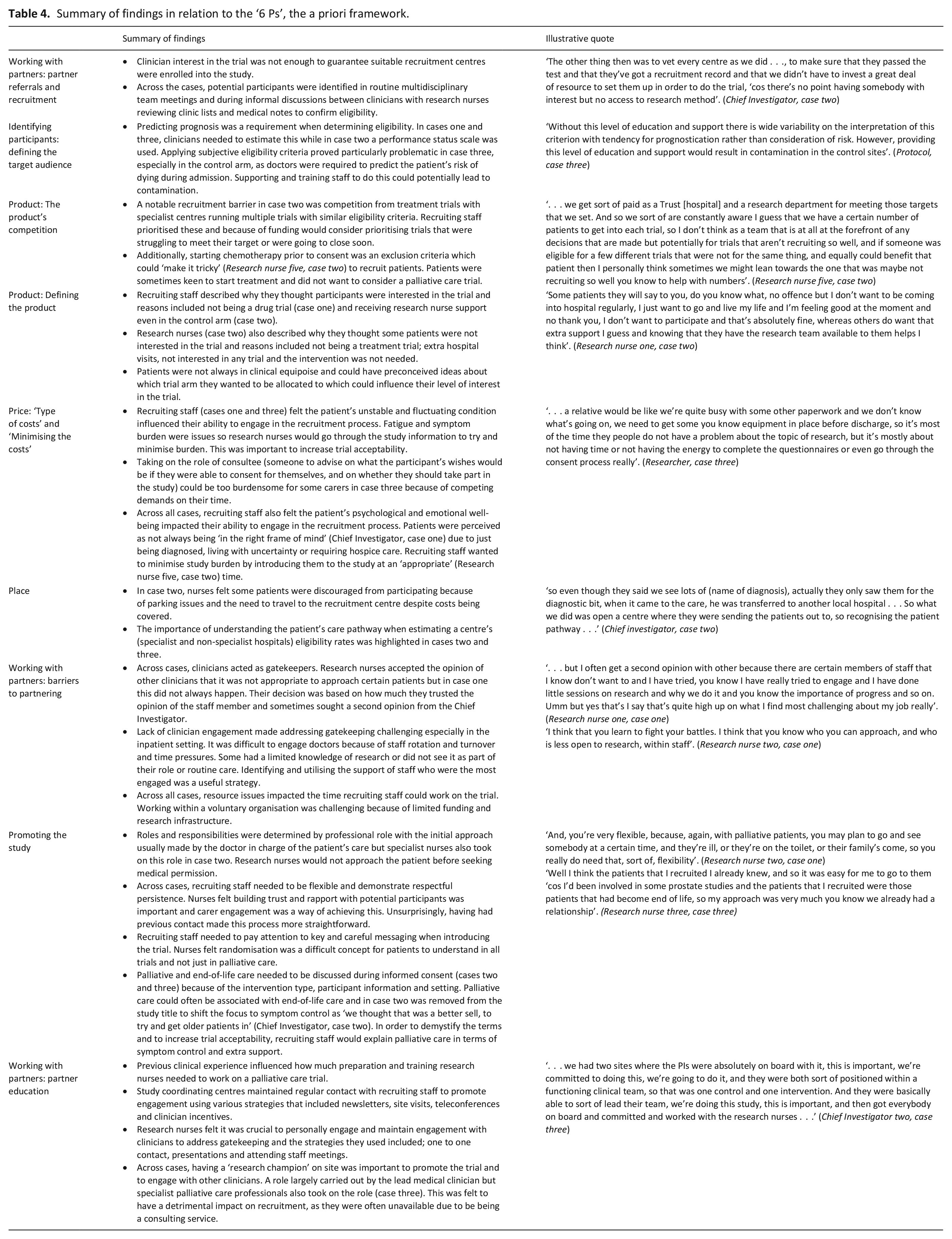
First, the findings are presented in relation to the ‘6 Ps’, the a priori framework, reordered to reflect the findings. These are summarised in Table 4. Second, three overarching classifications are presented that were derived interpretively from the data; ‘emotional labour’, ‘paternalism’ and ‘professional hierarchies and power relationships between clinicians’. How these map onto the ‘Social Marketing Mix Framework’ is presented in Figure 1.
Summary of findings in relation to the ‘6 Ps’, the a priori framework.
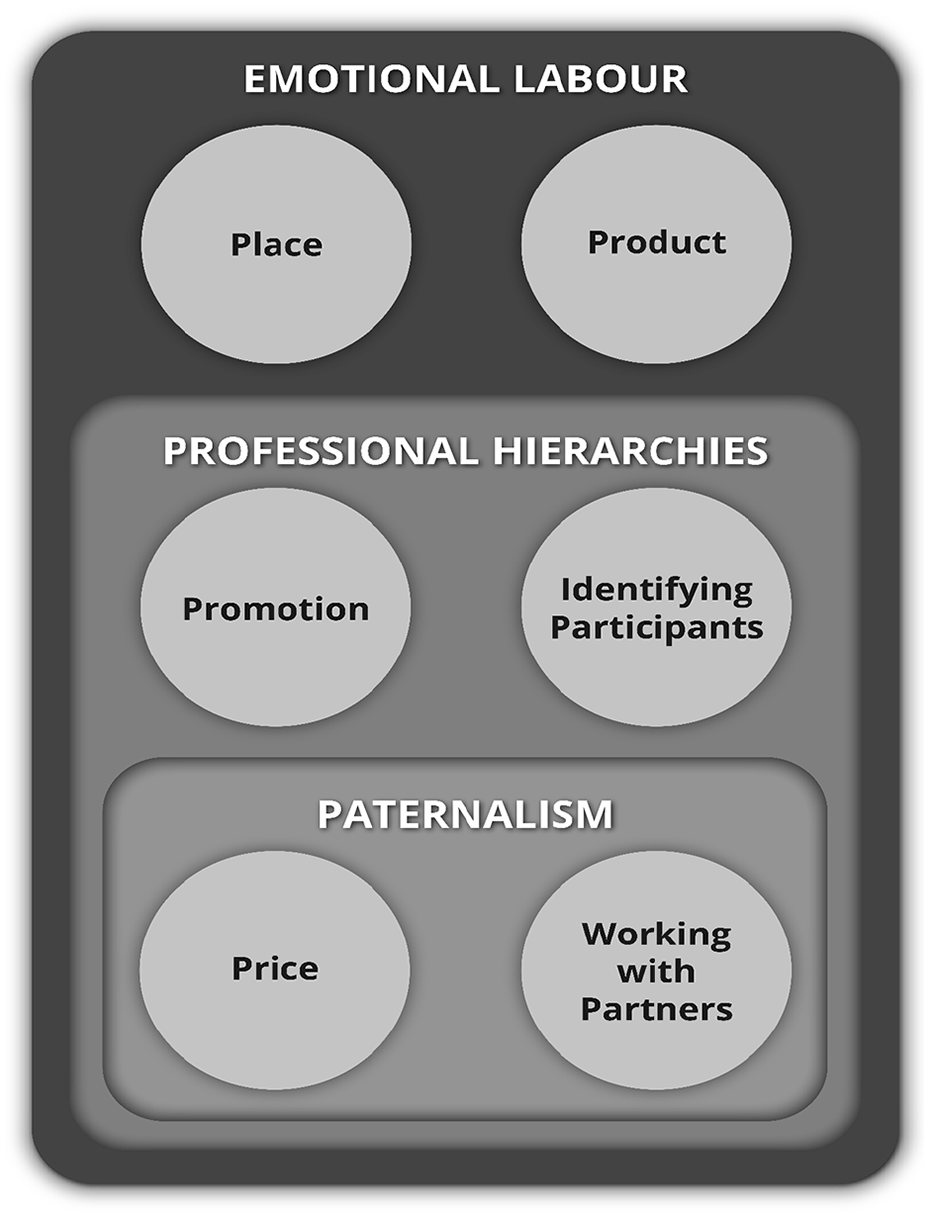
New palliative care trial recruitment framework proposed.
The ‘emotional labour’ of recruiting patients and carers to palliative care randomised controlled trials
‘Emotional labour’ involves the management and regulation of feelings in the workplace and is broadly defined as ‘the induction or suppression of feeling in order to sustain an outward appearance that produces in others a sense of being cared for in a convivial safe place’ (p. 11). 35 Across cases, recruiting staff had to manage the ‘emotional labour’ of approaching participants at a difficult time in their illness trajectory and they had to respond rapidly due to the risk of deterioration or start of treatment.
Costs for research nurses
The ‘Social Marketing Mix Framework’ does not acknowledge the ‘Price’ or ‘emotional labour’ of recruiting to a palliative care trial for research nurses, as ‘Price’ applies to patients and carers. Balancing the need to respond rapidly while taking account of the patient’s unstable condition was an issue in cases one and three which could be exacerbated by wider resource issues. Recruitment could be time consuming because of the need for multiple visits and the support patients required to go through study information: . . . you know they’ve said, you know I can’t actually, I’ve not felt up to reading it or you know I can’t read the information, you know so I have sort of read it to them and then gone through it with them so things just take a bit longer and you need oodles of sort of patience and also not giving up really. . . (Research nurse one, case one)
In case three, complex consent processes proved particularly challenging for some research nurses as they were required to seek consultee assent or if the patient had capacity, follow advance consent procedures. They could lack confidence and skill when assessing capacity and could be unclear about who could act as consultee. Lack of experience and training contributed to this recruitment barrier with the Chief Investigator reflecting on how they could have prepared the nurses better.
Multiple carer visits were required with some wanting to discuss it with other family members before making a decision. Allowing carers the time they needed while balancing the pressure to recruit within a short window of opportunity could lead the research nurses to face a difficult dilemma and experience increased emotional burden: . . . but I can’t say to somebody I know you want to discuss it with your brother but your mum’s only eligible right now and she could change tomorrow, do you know what I mean, I can’t say that to them. I just have to go that’s fine, they can discuss it with whoever they want, you know they can take as long as they want . . . (Research nurse one, case three)
Choosing the best time to approach patients
Across all cases, recruiting staff made judgements about when to introduce the trial based on their concerns about the patient’s physical condition, psychological and emotional wellbeing and practical considerations such as visiting times. They were concerned that patients were ‘overwhelmed’ (Research nurse one, case two) with information and so to increase trial acceptance nurses allowed patients time to process or digest what was happening to them before introducing the study, an approach some expressed they also used in non-palliative care trials. This approach also appeared beneficial for recruiting staff particularly the research nurses: People’s understanding of hospices, tends to be that it is a place you go to to die. So, when people are referred to the in-patient unit, and they’re admitted, there is that, sort of, automatic barrier comes up, so, oh my God, is this it? So, it’s, from my perspective, really helpful not to see somebody within the first 48 hours. Because, they need to gain a confidence in what we are doing, as an organisation, that the place is, you know, a good place to be, and they need to, sort of, feel safe and secure. (Research nurse two, case one)
When introducing the study, research nurses would vary the amount of information given depending on the patient’s response, level of understanding and their own level of comfort. Recruiting staff felt patients responded to being approached about a palliative care trial differently. Some were very accepting and viewed it positively while others were less open. Some were not ready to acknowledge or talk about their illness and a nurse described how a minority became distressed: . . . and then I had a couple of patients who actually got really angry in that leave my room, I don’t want to talk to you, I’m going to get better, how dare you start talking about such, you know, that I’m not going to get better, that’s negative thoughts, I don’t need that, go away, you know sort of real mixed kind of responses, sort of total denial so the patient had been told . . . (Research nurse one, case three)
Introducing the study appeared less demanding for those nurses who had had previous clinical contact with the patients as they could strike up an instant rapport and they knew what the patient understood about their condition.
Explaining palliative care
Recruiting staff (cases two and three) could find explaining palliative care challenging as it was difficult to discuss without acknowledging its association with end-of-life care. Research nurses had to broach issues that they would not routinely have to discuss which could cause worry and anxiety: . . . I actually think I made a mountain out of a molehill ‘cos actually I think the patients were quite fine about it, I think it was a lot of it was our worry about how they would feel, because we’d kind of never it was quite new to us . . . (Research nurse three, case two)
In case two, research nurses felt uncomfortable discussing a bereavement questionnaire as part of the consent process because they were apprehensive about how participants may react and how it may lead to difficult questions which they felt they did not have the skills to answer. The importance of highlighting the questionnaire to limit the potential for distress was discussed: . . . I had that discussion she burst into tears, so it wasn’t ideal, and she said to me afterwards she was like I’m really pleased that you highlighted that from the information because I wouldn’t have wanted to have been at home and burst into tears having read this, but she said that’s really upsetting and she sort of said I can see why you want to do it and it makes sense . . . (Research nurse five, case two)
Preparation for a sensitive conversation
Across all cases, those research nurses who had experience of talking to palliative care patients and carers appeared to find having sensitive conversations less emotionally demanding. Discomfort and inexperience manifested itself in staff declining to work on case three with centres having to use a core team of nurses who were comfortable working on the study: And the nature of the patients, some of our team didn’t like approaching them because they weren’t used to that type of patient, that was a problem as well. (Research nurse two, case three)
Despite previous experience, there were still concerns around how to broach conversations around death with participants in cases two and three. Research nurses prepared for sensitive discussions by discussing how best to approach these conversations within their own team and by seeking advice from the palliative care team. Working within a voluntary organisation as a research nurse, where resources are limited, could be challenging: . . . working on your own can be quite difficult and I’ve always been part of a research team so you know in my last job, . . . you’ve always got those colleagues that are working in exactly the same way as you that you can run things by or umm you know I’m very, I’m quite isolated really in my job and at times it can really upsetting and stressful and challenging and I don’t always feel that I really have anyone to share that with. (Research nurse one, case one)
The influence of ‘paternalism’ on palliative care trial recruitment
Paternalism refers to; ‘the intentional overriding of one person’s preferences or actions by another person, where the person who overrides justifies this action by appeal to the goal of benefitting or of preventing or mitigating harm to the person whose preferences or actions are overridden’ (p. 215). 36 There was evidence that clinician and carer paternalism could override patient autonomy in the trial recruitment process.
When screening, clinicians applied their own unofficial eligibility criteria and in case two, believing they were acting in the patient’s best interest, information was withheld from patients about all the trials they were eligible for. Treatment trials were seen as having the potential to ‘actually benefit (the patient) clinically’ (Research nurse three, case two) so were prioritised.
Carer gatekeeping was a particular issue in case three where patients were at risk of dying. Nurses sometimes needed to introduce themselves to the carer before or at the same time as speaking to the patient. A nurse described how she was fearful of approaching patients, as she was worried about the carer’s reaction, including concerns they may make a complaint. Families could became annoyed and it could be a dilemma balancing the right to approach a patient with managing the carer’s distress: I literally couldn’t even tell them who I was and what I was doing or finish the sentence before they were like now’s not the time, how dare you? Well it’s just we do have to ask at such a difficult time because of the timing of the research, I appreciate it’s difficult but the timing is necessary. But it’s not appropriate, they just weren’t listening. (Research nurse one, case three)
The influence of ‘professional hierarchies and power relationships between clinicians’ on recruitment practices
Professional hierarchies and power relationships influenced the recruitment of clinical recruitment centres, trial participants and the research champion within the research site. Chief Investigators who were also senior doctors used their medical contacts to identify research sites while the Chief Investigator from an academic background needed to negotiate access by building up relationships with clinicians which could take a long time: . . . So it was really just a question of convincing them and just negotiating with them and building levels of rapport which I think were really really critical. It’s not just a question of parachuting into a site, there’s so much ground work that needs to take place beforehand. (Chief Investigator one, case three)
Any care team member could identify participants but confirmation of trial eligibility was the responsibility of the lead medical clinician as they held overall ownership of the patient’s care. Medical confirmation that the patient was a palliative or end-of-life care patient was required. Research nurses used multi-disciplinary team meetings to identify eligible patients and to seek medical confirmation of eligibility. These meetings could be used as a forum for doctors to decide who the nurses could and could not approach: So if they were somebody that was likely to be fit enough for a chemotherapy trial, then we would get them to see the medical oncologist and the nurse specialist would not talk to them about (name of trial) . . . if we felt that they were not really suitable for either then we would then get the nurse specialist to discuss with them . . . (Doctor, case two)
Confirming eligibility was more problematic in case three as some doctors appeared reticent and fearful of making the decision that the patient may die under their care and how it could be difficult for recruiting staff to ‘get past people’s inherent optimism’ (Doctor, case three). In order to confirm eligibility a difficult conversation with the patient needed to occur and research nurses wanted to make sure this had happened before approaching but conversations could be poorly documented. They needed to see written confirmation that it was safe for them to approach the patient because of the sensitive nature of the information they needed to present. Similar concerns were raised by a palliative medicine doctor: . . .we couldn’t go to people who didn’t know, weren’t aware obviously that would come as a shock, perhaps they had been told but then forgotten, or that we were concerned that if they read that they you know they might not have realised . . . (Doctor, case three)
Research nurses also had the power to influence whether or not a potential participant was recruited as they felt they were ‘the patient’s advocate’ (Research nurse three, case two) and their role was to act in the patient’s best interest.
The requirement for the lead medical clinician to confirm eligibility influenced who was the most appropriate professional to act as ‘research champion’. In practice, who took on the role of Principal Investigator was often a practical decision, based on their enthusiasm. In case two, this role was carried out successfully by a specialist nurse reflecting their position within the multi-disciplinary team: . . . the MDT team makes a decision, the news is broken to the patient, and then ordinarily there’s like a small break out room where the patients can you know deal with . . . their emotions, and then the specialist nurses had a choice basically between either whether the patients were receptive at that stage to learn about the trial . . . (Chief Investigator, case two)
Research nurses valued the time specialist nurses spent talking to patients about their illness as it freed them up to focus on the more practical aspects of the recruitment process.
Figure 1 illustrates the new palliative care trial recruitment framework proposed that reflects the study findings. An adapted ‘Social Marketing Mix Framework’ that incorporates the wider overarching contextual issues of ‘emotional labour’, ‘paternalism’ and ‘professional hierarchies and power relationships between clinicians’.
Discussion
Main findings
The cross-case analysis suggests the ‘6 Ps’ are relevant in the context of palliative care trial recruitment but the concepts of ‘emotional labour’, ‘paternalism’ and ‘professional hierarchies and power relationships between clinicians’ are required as an additional theoretical lens. This is to better understand how health care professionals recruit patients and their family carers to palliative care trials and why they use certain strategies during the recruitment process. Clinicians experienced ‘emotional labour’ when they were ‘promoting’ and recruiting to a palliative care trial which lead to paternalistic recruitment practices. The ‘professional hierarchies and power relationships’ that existed between clinicians influenced how ‘emotional labour’ was experienced by medical and nursing staff and also facilitated and supported paternalistic recruitment practices. The final theoretical propositions that reflect the emerging findings and the wider literature are outlined in Table 5.
Final theoretical propositions.

What this study adds
The ‘emotional labour’ of trial recruitment is not new11,37 and unsurprisingly palliative care contextual factors contributed to this issue in this study. The ‘emotional labour’ of caring for the dying especially in the hospital setting has been recognised.35,38 Palliative care is carried out in an emotion laden context with clinicians often having to deal with appropriate but powerful patient and carer emotions. 39 Patients and carers often associate palliative care with death and dying40,41 and clinicians can find initiating end of life conversations challenging and can feel ill prepared.42,43 These issues are problematic in the recruitment context as an ‘active’ rather than a ‘passive’ communication stance is required. 44
Tailoring information according to levels of understanding and readiness is viewed as paramount in end-of-life communication45,46 but this can be difficult for those going through standardised participant information. Fluctuating levels of patient and carer awareness can also make it challenging for recruiting staff to ‘promote’ a palliative care trial. Respecting ‘awareness’ preferences is important 47 but must not be used by clinicians as a reason to avoid research discussions.
Doctors are viewed as largely being responsible for discussing prognosis and making treatment decisions48,49 but they can struggle to recognise when a patient is approaching the end of their life. 50 The ‘emotional labour’ of predicting a patient’s prognosis has been identified as a reason for doctors avoiding end-of-life care discussions 43 so according to Glaser and Strauss 51 perpetuating ‘closed awareness’. Closed awareness is when only clinicians and families are aware that the patient’s illness will lead to their death. 51 This has implications for the recruitment process as the lack of prognostic certainty can lead to a reluctance to confirm eligibility and ‘promote’ the trial to participants. There can also be tensions between consulting specialist palliative care services and those that focus on a more acute model of care. 47
Ethics committees can be concerned about involving patients and family carers in palliative and end of life care research. They can be fearful of overburdening vulnerable patients 21 and may insist on a clinician acting as a gatekeeper in the belief this will protect patients and reduce the risk of distress. This study has shown that health care professionals may have complex reasons for not approaching palliative care patients about research. This has important implications for the recruitment process as ethics committees may be inadvertently creating a barrier to participant enrolment by requiring clinicians to introduce a research study to the patient.
This study has highlighted the importance of incorporating ‘partner education’ into trial planning for recruiting staff to manage their emotional labour, including how to discuss a palliative care trial, echoing one of the recommendations from the updated MORECare project. 52 This is in addition to the generic trial training recommended in the general literature to improve recruitment rates.37,53 Training should reflect trial characteristics and clinician experience and may need to include how to; explain palliative and end-of-life care 54 ; assess the participants understanding of their condition 55 ; manage the psychological needs of patients and carers56,57; and assess capacity and enact proxy and advance consent procedures.52,58 Staff should also have the opportunity to reflect on their practice. 59 Integrating research into routine practice 60 will mean clinicians already have a rapport with participants.
Strengths and weaknesses of the study
To the author’s knowledge, this is the first qualitative multiple case study to produce new insights into the palliative care trial recruitment process. By using theoretical propositions and multiple diverse cases the findings can be used to understand trial recruitment processes beyond the three cases 27 and are relevant to palliative care research generally. An additional case would have been selected if resources had allowed as findings suggested pharmaceutical symptom control trials may raise particular issues for staff. The views of participants who agreed to take part may not be representative of others involved in the recruitment process. Patient and carer perspectives are not captured and they may be different to those expressed by clinicians. 61 This study took place in the UK and internationally communication preferences, 62 research ethics and governance requirements, 63 resources and services can differ which may affect the recruitment process.
Conclusion
To conclude, an adapted ‘Social Marketing Mix Framework’ incorporating the concepts of ‘emotional labour’, ‘paternalism’ and ‘professional hierarchies and power relationships between clinicians’ is a useful framework for planning and monitoring recruitment activity in a palliative care trial. Those planning trials need to ensure that clinicians, depending on their experience and trial characteristics, have access to training and support to address the ‘emotional labour’ of recruiting to a palliative care trial. The type of training and support required to address this recruitment barrier requires further research.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163231197917 – Supplemental material for Health care professional recruitment of patients and family carers to palliative care randomised controlled trials: A qualitative multiple case study
Supplemental material, sj-pdf-1-pmj-10.1177_02692163231197917 for Health care professional recruitment of patients and family carers to palliative care randomised controlled trials: A qualitative multiple case study by Lesley Dunleavy, Nancy Preston and Catherine Walshe in Palliative Medicine
Footnotes
Acknowledgements
LD would like to thank the Chief Investigators who agreed to support this study and the participants who agreed to take part.
Authorship
LD conceived the study, obtained ethics and organisational approval, collected and analysed the data, and prepared initial drafts of the manuscript. CW and NP advised on the study design, provided critical input into the data analysis process and iterations of the manuscript. All authors approved the final version of the submitted manuscript.
Data management and sharing
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethics and consent
Ethical approval was obtained from Lancaster University Faculty of Health and Medicine Research Ethics Committee (Reference number: FHMREC15042, 22nd February 2016). Verbal consent was provided by participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
