Abstract
Background:
Patients with and without cancer are frequently hospitalized, and have specialist palliative care needs. In-hospital mortality can serve as a quality indicator of acute care. Trends in acute care outcomes have not previously been evaluated in patients with confirmed specialist palliative care needs or between diagnostic groups.
Aim:
To compare trends in discharge location between hospitalized patients with and without cancer who received specialist palliative care.
Design:
Retrospective cohort study. Association between diagnosis (cancer, non-cancer) and in-hospital mortality was assessed using multivariable logistic regression, controlling for demographic, clinical, and admission-specific information.
Setting/participants:
Patients who received specialist palliative care at an academic tertiary hospital in Toronto, Canada from 2013 to 2019.
Results:
The cohort comprised 6846 patients, 5024 with and 1822 without cancer. A higher proportion of patients without cancer had a Palliative Performance Scale score <30%, anticipated prognosis of <1 month, and were referred for end-of-life care (all p < 0.001). The adjusted odds of dying in hospital was 1.24-times higher among patients without cancer (95% CI: 1.05–1.46; p = 0.011). Though the proportion of patients without cancer who died in hospital decreased by 8.4% from 2013 to 2019, this proportion (41.2%) remained substantially higher compared to patients with cancer (14.0%) in 2019.
Conclusions:
Hospitalized patients without cancer were referred to specialist palliative care at a lower functional status, a poorer anticipated prognosis, and more likely for end-of-life care; and were more likely to die in hospital. Future studies are required to determine whether a proportion of hospital deaths in patients without cancer represent goal-discordant end-of-life care.
Keywords
Patients with and without cancer are frequently hospitalized and have specialist palliative care needs, which confirms the importance of evaluating acute care outcomes within and between these diagnostic groups.
In-hospital mortality can serve as a quality indicator of acute care.
Trends in acute care outcomes have not previously been evaluated in patients with confirmed specialist palliative care needs or between diagnostic groups.
Compared to patients with cancer, hospitalized patients without cancer were referred to specialist palliative care at a lower functional status, a poorer anticipated prognosis, and more likely for end-of-life care.
Relative to patients with cancer, patients without cancer had 1.24-times higher odds of in-hospital mortality controlling for demographic, clinical, and admission-specific information.
From 2013 to 2019, the proportion of patients without cancer dying in hospital remained substantially higher than that of patients with cancer.
Future studies are required to determine whether a proportion of hospital deaths in patients without cancer represent goal-discordant end-of-life care.
Hospital- and systems-level changes are required to increase early access to specialist palliative care services in the ambulatory and community settings among patients without cancer to mitigate possible health inequities observed at the end of life.
Introduction
Hospitalization is common across multiple patient populations,1 –6 particularly at the advanced stages of disease. For example, a population-level Danish study demonstrated that a substantial proportion of the last 6 months and 30 days of life were spent in hospital among patients with cancer, heart failure, and chronic obstructive pulmonary disease. 7 There is an increasing recognition of the specialist palliative care needs of patients without cancer, which substantiate the role of specialist palliative care teams in their care.8–11 Evaluating the acute care outcomes of patients with cancer versus without cancer who require specialist palliative care could shed light on differences in reasons for accessing acute care and in the role of palliative care teams during hospitalization.
In-hospital mortality has been used internationally as a quality indicator of acute care.12–20 Though in-hospital mortality has limitations as an indicator of care quality, it lends itself to being systematically collected over time, and can serve as an entry point to evaluate care quality, 21 in combination with other quantitative and qualitative metrics.22,23 Multiple studies have evidenced that most patients with and without cancer do not prefer to die in hospital.24–27 This has been substantiated by a systematic review of 210 studies from 33 countries that demonstrated moderate evidence that the majority of individuals preferred to die at home 28 and a meta-analysis of three studies that demonstrated that a slight majority of patients without cancer preferred to die at home. 29 To our knowledge, no study has demonstrated a predominant preference for death in the acute care setting.
There have been studies of trends in location of death30 –32 and in-hospital mortality specifically; 7 however, they were not performed in cohorts of patients with confirmed specialist palliative care needs nor compared between diagnostic groups. Therefore, we sought to evaluate trends in discharge location, specifically in-hospital mortality, in a cohort of patients with versus without cancer who received specialist palliative care from 2013 to 2019.
Methods
Design and setting
This was a retrospective cohort study performed in an academic tertiary hospital in Toronto, Ontario, Sunnybrook Health Sciences Centre. Toronto is Canada’s most populous city (6.2 million individuals in 2021 33 ) and houses a highly culturally diverse population (46.1% of the population identified as immigrants in 2016 34 ). The hospital includes a comprehensive specialist palliative care service. Most relevant to this study is the inpatient specialist palliative care team, 35 which is interprofessional, comprising physicians and advanced practice nurses. It provides consultation to all inpatient wards, including oncology, general internal medicine, critical care, emergency medicine, and surgery. The team works collaboratively with palliative care specialists in the ambulatory setting and the palliative care unit (PCU) and liaises with home-based palliative care providers from other healthcare institutions.
Population
The cohort comprised patients who were admitted to the hospital from January 1st, 2013 to December 31st, 2019 and who received specialist palliative care consultation during admission. All patients who received consultation were included in the cohort. There were no exclusion criteria.
Data collection
Data was collected from the palliative care database, which was established in 2012. The database includes detailed demographic, clinical, and admission-specific information from only a patient’s first hospitalization in which specialist palliative care was consulted. When a patient receives consultation during a subsequent hospitalization, this is noted in the database though detailed information is not collected. Therefore, if a patient was admitted and received consultation more than once during the study period, only their first hospitalization with consultation was included. Demographic information includes age and sex. Clinical information includes primary diagnosis, which is the diagnosis thought to be most responsible for the referral; Palliative Performance Scale (PPS) score; anticipated prognosis estimated by the specialist palliative care provider; and resuscitation status at the time of referral. Admission-specific information includes reason for referral; timing of admission, referral, and consultation; and discharge location. Discharge location is recorded in one of six general categories: in-hospital death, discharged home without support, discharged home with support, discharged to a PCU/hospice, other (rehabilitation, other hospital or healthcare institution), and signed off. The team signs off on a patient if they are considered to no longer require specialist palliative care during the hospitalization (e.g., a patient whose symptoms are controlled).
Data analysis
Descriptive statistics were used to characterize the cohort in terms of demographic, clinical, and admission-specific information, and to evaluate trends in discharge location over time. Differences between patients with and without cancer were assessed using the Wilcoxon two-sample test for continuous variables since none were normally distributed, and Chi-Square or Fisher’s Exact tests for categorical variables.
The association between diagnosis (cancer, non-cancer) and in-hospital mortality was assessed using multivariable logistic regression, controlling for age, sex, PPS, and year of consultation. As a sensitivity analysis, the logistic regression was performed on only patients with an established discharge location, excluding those on whom the team signed off. In the regression analyses, missing data were handled using complete case analysis considering that less than 0.1% of patients had a missing value of any variable in the model. The risk of overspecification was judged to be low considering the higher number of events in the cohort compared to the number of variables in the model. Statistical analysis was performed using SAS Studio (Cary, NC, USA).
Ethics
Research ethics approval was granted by the Research Ethics Board of Sunnybrook Health Sciences Centre (project identification number 5670).
Results
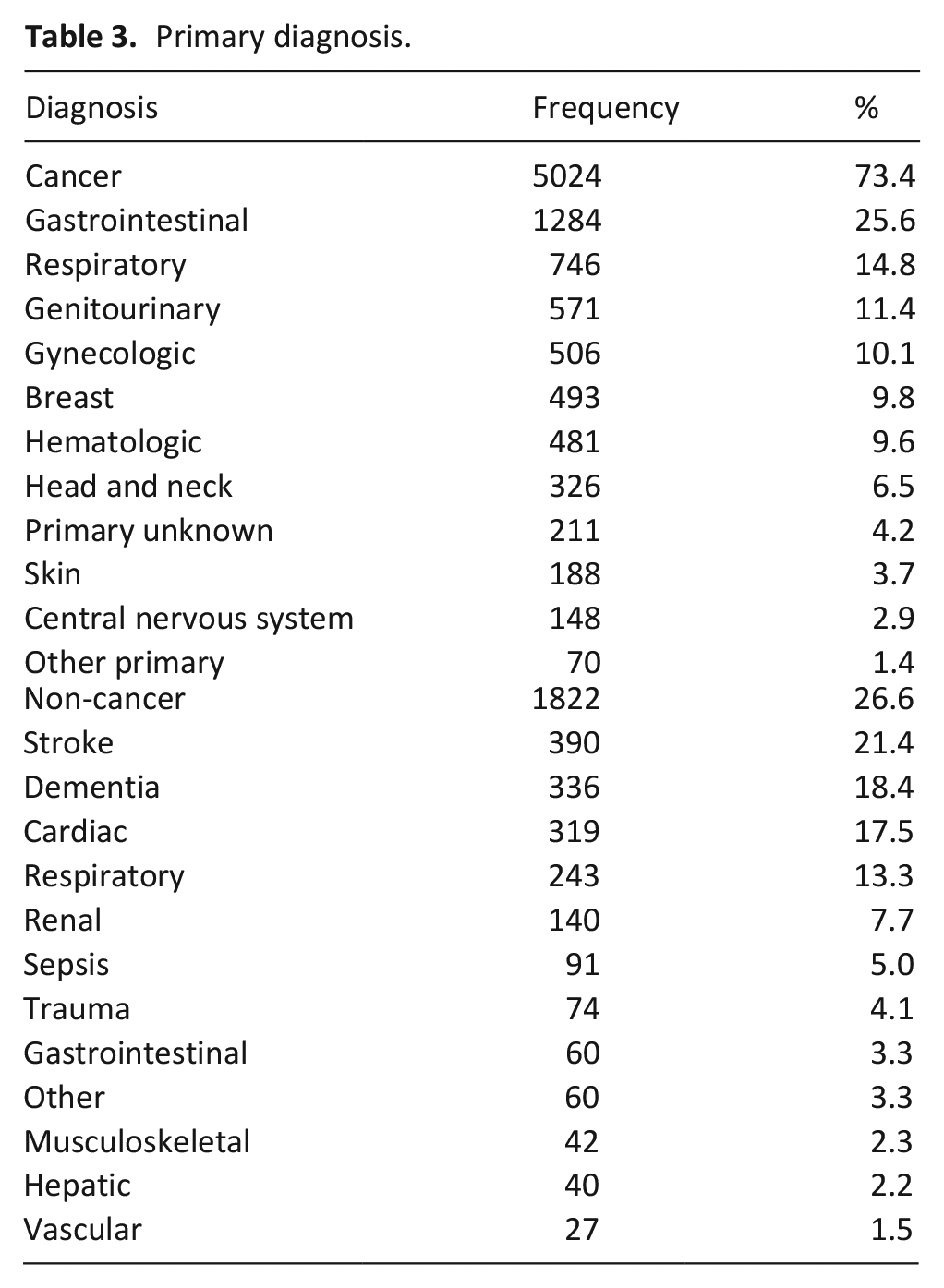
The cohort comprised 6846 unique patients, 5024 with a primary cancer diagnosis and 1822 with a primary non-cancer diagnosis (Tables 1 and 2). The median age was 80 (interquartile range (IQR) 23) years, with patients without cancer having an older age than patients with cancer (92 (IQR 14) and 75 (IQR 20) years, respectively; p < 0.001). A slight majority of the cohort was female (3578/6845, 52.3%) with no difference between groups (p = 0.31). Whereas 78.1% (1420/1818) of patients without cancer had a PPS <30%, only 26.7% (1342/5023) of patients with cancer did so (p < 0.001). Similarly, whereas most patients without cancer had an anticipated prognosis of <1 month (1151/1820, 63.2%), only 19.2% (962/5021) of patients with cancer did so (p < 0.001). A lower proportion of patients with cancer had an established do not resuscitate (DNR) order (2117/5020, 42.2%) compared to patients without cancer (1630/1819, 89.6%; p < 0.001). Primary diagnoses are listed in Table 3. Among patients with a primary cancer diagnosis, the most common primaries were gastrointestinal (25.6%), respiratory (14.8%), genitourinary (11.4%), gynecologic (10.1%), and breast (9.8%). Among patients with a primary non-malignant diagnosis, the most common diagnoses were stroke (21.4%), dementia (18.4%), cardiac (17.5%), respiratory (13.3%), and renal (7.7%).
Demographic and clinical information by diagnosis (cancer, non-cancer).
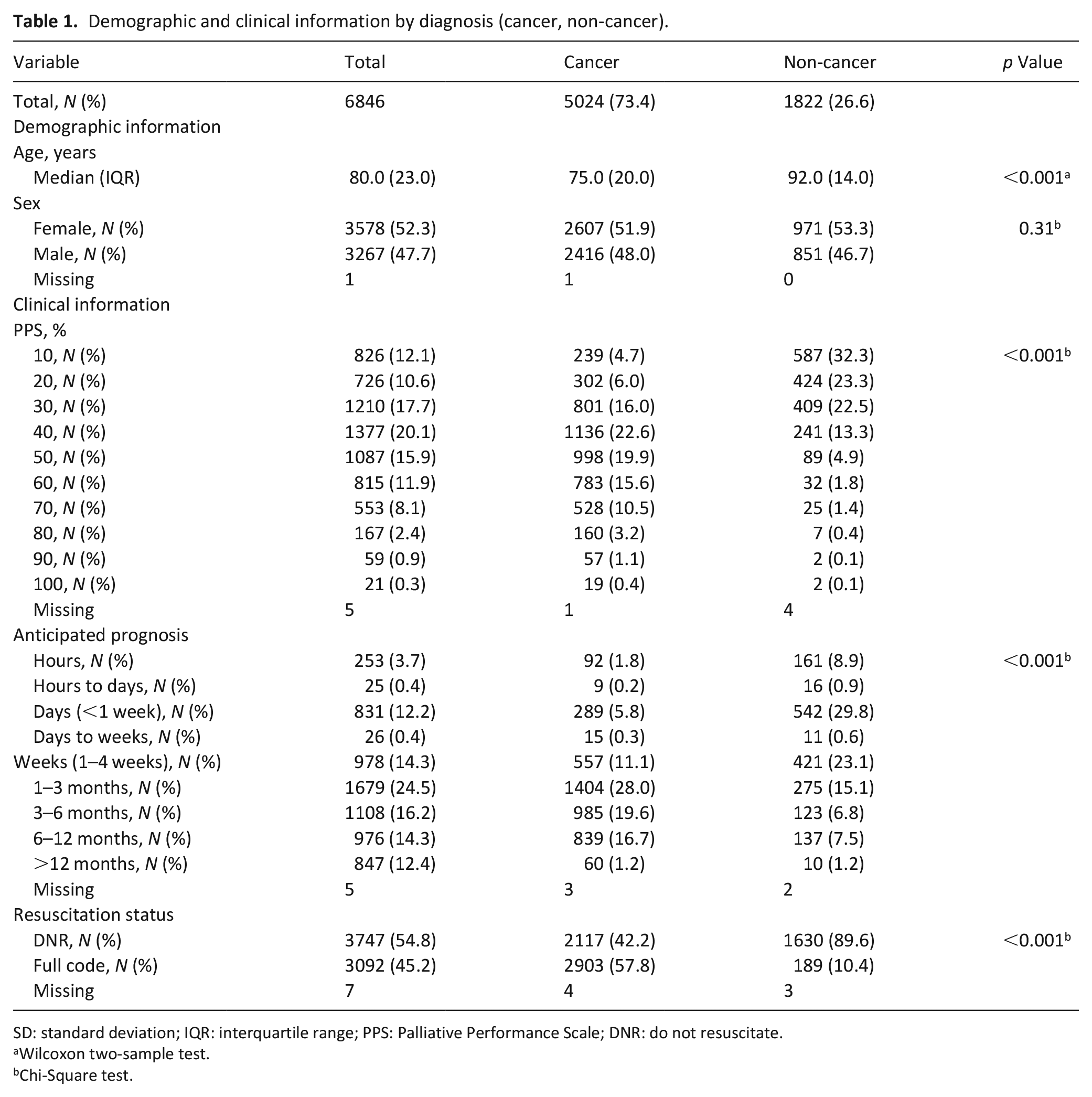
SD: standard deviation; IQR: interquartile range; PPS: Palliative Performance Scale; DNR: do not resuscitate.
Wilcoxon two-sample test.
Chi-Square test.
Admission information by diagnosis (cancer, non-cancer).
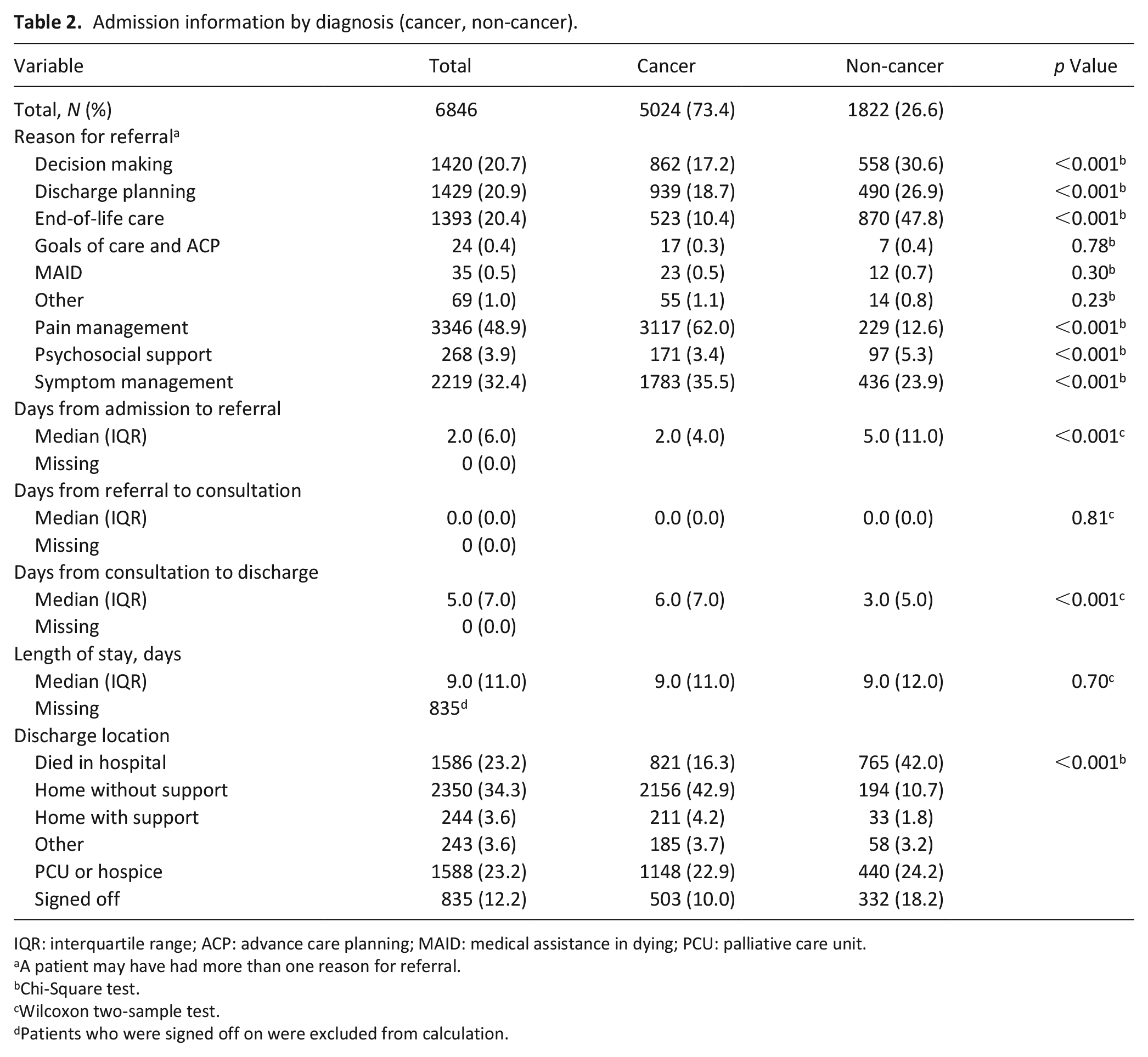
IQR: interquartile range; ACP: advance care planning; MAID: medical assistance in dying; PCU: palliative care unit.
A patient may have had more than one reason for referral.
Chi-Square test.
Wilcoxon two-sample test.
Patients who were signed off on were excluded from calculation.
Primary diagnosis.
A higher proportion of patients without cancer were referred for decision making (difference = 13.4%; p < 0.001), discharge planning (difference = 8.2%; p < 0.001), end-of-life care (difference = 37.4%; p < 0.001), and psychosocial support (difference = 1.9%; p < 0.001); whereas, a higher proportion of patients with cancer were referred for pain (difference = 49.4%; p < 0.001) and symptom (difference = 11.6%; p < 0.001) management. Similar proportions were referred for goals of care (difference = 0.1%; p = 0.78), medical assistance in dying (difference = 0.2%; p = 0.30), and other reasons (difference = 0.3%; p = 0.23).
The median number of days from admission to referral was higher among patients without cancer (5 (IQR 11)) than patients with cancer (2 (IQR 4); p < 0.001). The median number of days from consultation to discharge was higher among patients with cancer (6 (IQR 7)) than patients without cancer (3 (IQR 5); p < 0.001). There was no difference in time from referral to consultation (p = 0.81) or in length of stay (p = 0.70).
Association between diagnosis and in-hospital mortality
Univariate analysis demonstrated that a significantly higher proportion of patients without cancer (765/1822, 42.0%) died in hospital compared to patients with cancer (821/5024, 16.3%; p < 0.001) (Table 4). Compared to those who did not die in hospital, those who died in hospital were older (p < 0.001) and a higher proportion were male (p = 0.0059), had a low PPS (p < 0.001), and were hospitalized in earlier years (p = 0.0048).
Univariate and multivariate association between in-hospital mortality and demographic, clinical, and admission information.
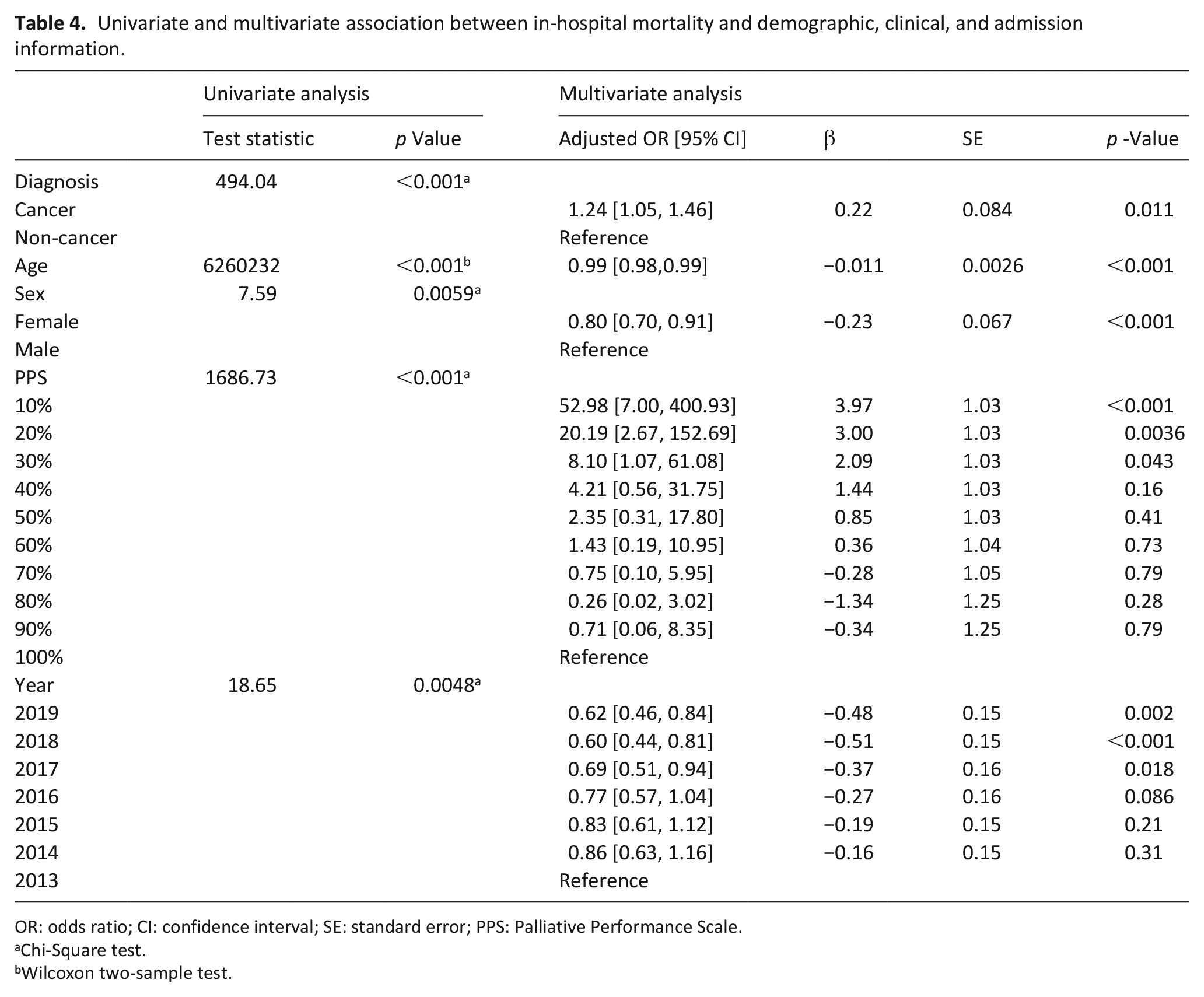
OR: odds ratio; CI: confidence interval; SE: standard error; PPS: Palliative Performance Scale.
Chi-Square test.
Wilcoxon two-sample test.
In multivariable analysis, all pre-specified demographic, clinical, and admission-specific variables were significantly associated with in-hospital mortality. Notably, diagnosis remained significantly associated with in-hospital mortality, controlling for age, sex, PPS, and year of consultation (p < 0.001). Specifically, the odds of dying in hospital was 1.24-times higher among patients without cancer than among patients with cancer (95% CI: 1.05, 1.46; p = 0.011). When excluding patients on whom the team signed off, the association between diagnosis and in-hospital mortality remained significant (adjusted OR 1.56 (95% CI: 1.31, 1.86; p < 0.001; Supplemental Table 1).
Trends in discharge location over time
In the total cohort (Figure 1(a)), the proportion of patients who died in hospital decreased over time from 29.5% in 2013 to 20.9% in 2019, while the proportion of those who were discharged to PCU/hospice increased (2013: 20.0%, 2019: 24.3%). Similarly, the proportion of patients who were discharged to another facility, either rehabilitation or another hospital or healthcare institution, increased (2013: 0.7%, 2019: 5.8%). The proportion of patients who were discharged home or on whom the team signed off was stable over time.
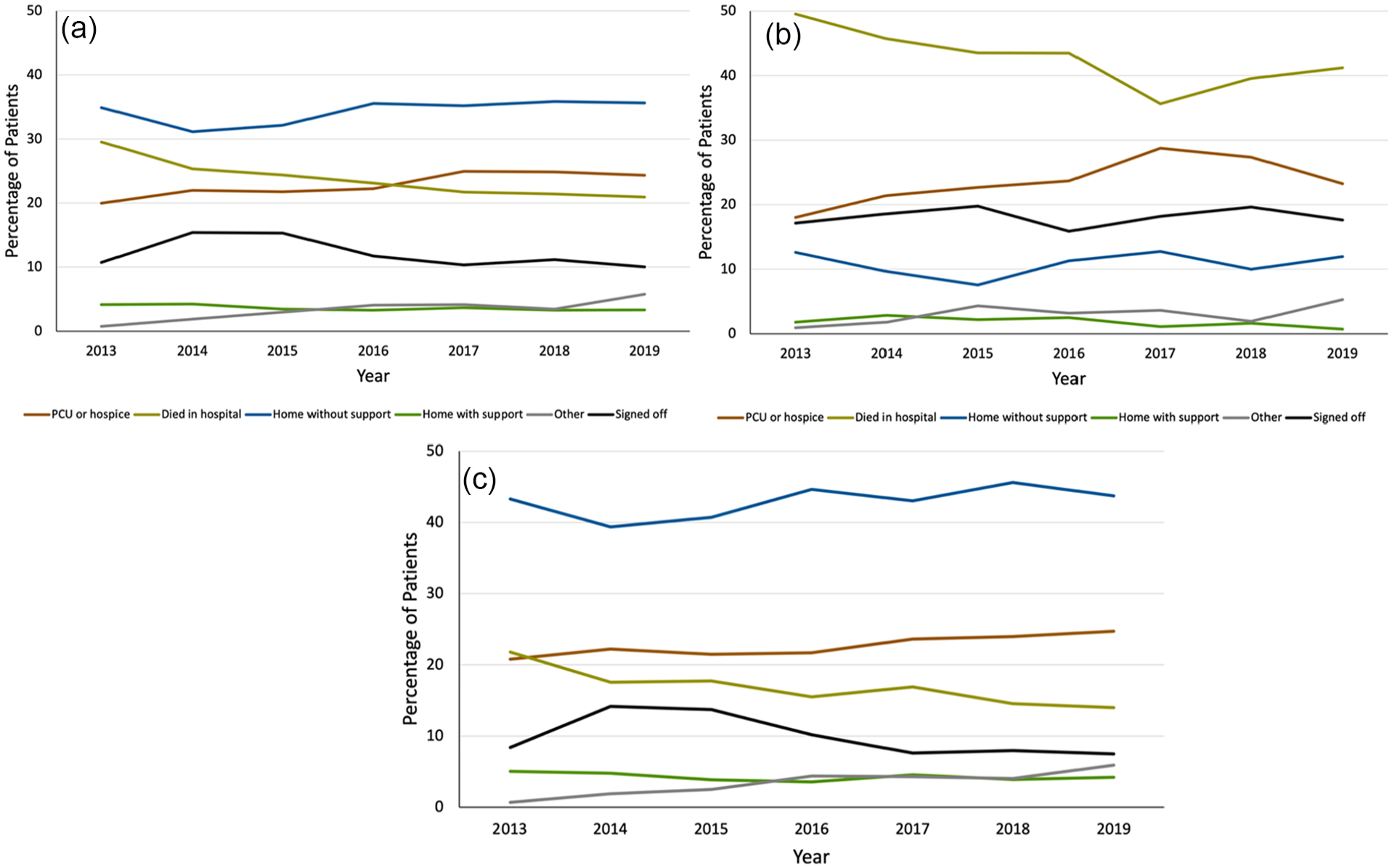
(a) Trend in discharge location from 2013 to 2019 among the total cohort, (b) trend in discharge location from 2013 to 2019 among patients with non-cancer diagnosis, and (c) trend in discharge location from 2013 to 2019 among patients with cancer diagnosis.
Similar trends were observed among diagnostic groups (Figure 1(b) and (c)). Though the proportion of patients without cancer who died in hospital decreased by 8.4% from 2013 to 2019, this proportion (41.2%) remained substantially higher compared to patients with cancer (14.0%) in 2019 (Figure 2). The proportion of patients without cancer who were discharged to PCU/hospice became comparable to that of patients with cancer over time, with about a quarter of patients being discharged thereto in later years. The proportion of patients who were discharged home did not change over time for either group, and remained consistently higher among patients with cancer than among patients without cancer.
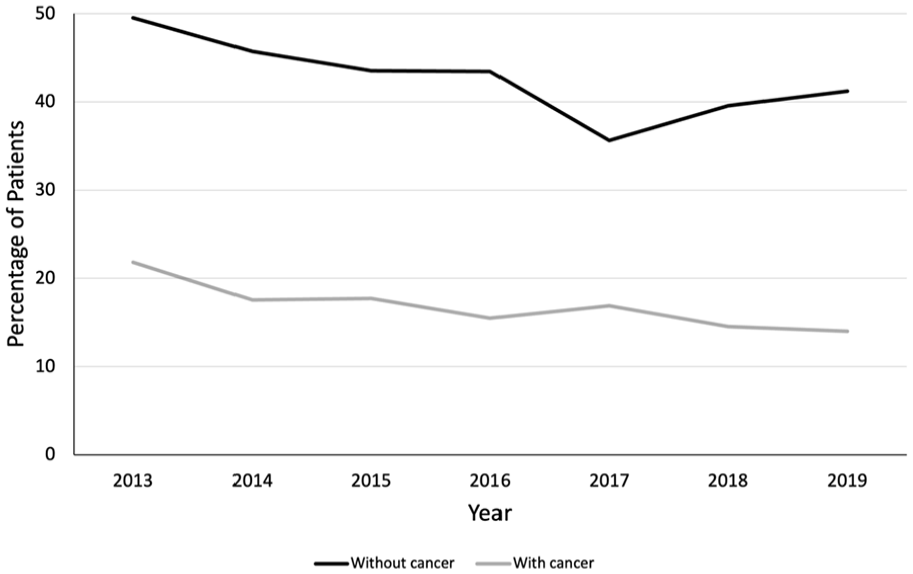
Trend in in-hospital mortality in patients with non-cancer and cancer diagnoses.
Discussion
Main findings
This six-year retrospective cohort study suggests discrepant acute care outcomes between patients with and without cancer who received specialist palliative care consultation. Specifically, at the time of consultation, patients with a primary non-cancer diagnosis were older, had a lower functional status, and had a poorer anticipated prognosis than patients with a primary cancer diagnosis. Accordingly, a higher proportion already had a DNR order and were referred for end-of-life care. Adjusting for demographic, clinical, and admission-specific information, patients without cancer had 24% higher odds of in-hospital mortality than patients with cancer. From 2013 to 2019, the proportion of patients without cancer who died in hospital remained substantially higher than the proportion of patients with cancer who did so.
The high frequency with which patients with and without cancer are hospitalized,1–7 and the lower frequency and later timing of palliative care access among patients without cancer36–41 have been previously established. This substantiates the importance of evaluating acute care outcomes within and between diagnostic groups. Our study achieves this imperative, demonstrating trends in in-hospital mortality among patients with and without cancer who have specialist palliative care needs. Case mix precludes direct comparisons between our findings and those of other hospitals. However, our study is in keeping with previous studies that have demonstrated differences in location of death between diagnostic groups. A population-level Dutch study demonstrated that the majority of groups without cancer had a higher proportion of hospital deaths (ranging from 22.2% in renovascular disease to 51.4% in liver disease) compared to patients with cancer (17.1%). 30 Similarly, a population-level American study demonstrated that a higher proportion of patients with frailty (64.5%), end-stage renal disease (60.8%), and cardiopulmonary disease (58.5%) died in hospital compared to patients with cancer (35.6%). Notably, family-reported quality of end-of-life care was higher when the location of death was not the acute care setting. 32 These studies focused on “palliative care-relevant” diagnoses or “serious illnesses” though patients did not have confirmed specialist palliative care needs. In addition, they did not investigate acute care outcomes specifically, but rather location of death.30,32 Finally, our finding that in-hospital mortality decreased over time is in keeping with a trend of decreasing hospital deaths in Ontario, from 58.2% in 2010/11 to 52.5% in 2017/18 in the general population. 42
The observed difference in acute care deaths between diagnostic groups invokes the thought that this represents a higher incidence of goal-discordant care in patients without cancer. In fact, a meta-analysis demonstrated that patients without cancer had a higher risk of dying in a non-preferred location compared to those with cancer. 43 Despite this knowledge, we cannot comment on the degree of goal-concordance in our study since we do not routinely collect information about preferred location of death in our database. Indeed, a recent review highlights the challenge of operationalizing goal concordance in palliative care research. 44 We also acknowledge that in-hospital mortality may be unavoidable secondary to the complicated nature of diseases that cannot be effectively supported in the community, and/or the lack or deficiency of community and hospice services, especially in low-resource settings. Particularly, the trajectories of non-cancer diseases, characterized by intermittent exacerbations requiring acute care,45,46 make hospitalizations, including a terminal one, challenging to avoid. In addition, about a quarter of the patients without cancer in our cohort had a stroke or trauma, diagnoses that require hospital-based care. If these events ultimately resulted in hospital deaths, then it would be inappropriate to conclude that this necessarily represented poor end-of-life care. However, this should not preclude the evaluation and management of potential inequities in palliative care access along diagnostic lines that could result in discrepant in-hospital mortality. Indeed, studies of home-based palliative care services in patients without cancer have been associated with decreased acute care utilization and in-hospital mortality.47,48 This provides evidence that enhancing palliative care access outside of the hospital setting may decrease acute care utilization and hospital deaths among patients without cancer.
Collectively, the results suggest that patients with non-cancer diagnoses are accessing specialist palliative care services later in the course of disease. There are variable reasons for later access to specialist palliative care services among patients with non-cancer diagnoses. First, there may be a deficiency of services for these patients in the ambulatory setting. At our center, there is a comprehensive specialist palliative service for outpatients with cancer that comprises a multidisciplinary team of physicians, nurses, and a social worker who collaborate closely with oncologists. We hypothesize that the discussions about goals of care that took place in this setting may partially drive the observed lower odds of in-hospital mortality among patients with cancer. Indeed, the benefits of early specialist palliative care in the outpatient setting are well established. 49 Equivalent services for patients without cancer are more sparse, 38 as demonstrated by a survey of cardiologists 50 and respirologists 51 across Canada that suggested poor availability of palliative care clinics. Fortunately, these services are increasing in number, including at our center, wherein there is now collaboration in clinics for patients with heart failure, end-stage kidney disease, and amyotrophic lateral sclerosis.
From 2013 to 2019, a lower proportion of patients without cancer died in hospital and a higher proportion were discharged to PCU/hospice. This is a striking finding considering that patients without cancer have had decreased access to PCU 52 and experience barriers to access. 53 These trends are potentially explained by interventions at a hospital- and systems-level. Specifically, since inception, the number of beds in the PCU at our center has doubled. These increases were designed to accommodate patients with anticipated prognosis >3 months who have complex specialist palliative care needs. In addition, over time, there has been a liberalization in the eligibility criteria for PCU that may have granted admission to patients with complex non-cancer disease. For example, the PCU accepts patients with end-stage kidney disease receiving hemodialysis, and those with heart failure who require intravenous diuresis. In addition, the increase in discharge to PCU/hospice may have been driven by a provincial campaign that promoted advance care planning (i.e., Speak Up 54 ). Finally, among patients in both diagnostic groups, there was a higher proportion who were discharged to rehabilitation or another healthcare institution during the study period. This may be a consequence of the launch of reactivation centers in 2017. These healthcare facilities were designed to safely transition patients from the acute care to the home setting. Taken together, this suggests that a combination of interventions at a hospital- and systems-level may be necessary to effect change in acute care outcomes among patients without cancer.
Strengths and limitations
This study is the first to compare trends in in-hospital mortality between diagnostic groups of patients who have confirmed specialist palliative care needs. It did so using routinely collected data from nearly 7,000 patients and over the span of 6 years.
Our study has limitations. First, despite the large sample size of the study, it was done at a single site. Therefore, our results cannot be readily generalized to other acute care institutions, particularly those in lower resource settings. Second, patients were categorized by their primary diagnosis (i.e., the diagnosis thought to be most responsible for the referral). Therefore, it is possible that a patient with a primary non-cancer diagnosis had a concomitant cancer. This potential bias would have attenuated the observed differences, such that our findings may be an underestimation of the discrepancies in acute care outcomes between diagnostic groups. Third, our logistic regression model estimating in-hospital mortality does not include variables that could have informed the observed association between diagnosis and in-hospital mortality. For example, it is known that race 55 and immigration status 56 are associated with the intensity of care received at the end of life; however, these variables are not collected in our database. Finally, patients’ preferences for acute care utilization and end-of-life care were unknown; therefore, in-hospital mortality may have constituted goal-concordant care rather than have been a sign of a poor health outcome.
Conclusions
This six-year retrospective cohort study demonstrated that among patients receiving inpatient specialist palliative care consultation, those with a primary non-cancer diagnosis were referred at an older age, a lower functional status, and a poorer anticipated prognosis than those with a primary cancer diagnosis. These patients were disproportionately referred for end-of-life care and had 24% higher odds of in-hospital mortality. While changes at the hospital- and systems-level may have decreased the proportion of patients without cancer dying in hospital, in-hospital mortality remained substantially higher than that among patients with cancer. Further studies are required to explain why these differences were observed, including the possibility that a proportion of hospital deaths represent goal-discordant end-of-life care.
Supplemental Material
sj-docx-1-pmj-10.1177_02692163231183009 – Supplemental material for Differences in trends in discharge location in a cohort of hospitalized patients with cancer and non-cancer diagnoses receiving specialist palliative care: A retrospective cohort study
Supplemental material, sj-docx-1-pmj-10.1177_02692163231183009 for Differences in trends in discharge location in a cohort of hospitalized patients with cancer and non-cancer diagnoses receiving specialist palliative care: A retrospective cohort study by Michael Bonares, Kalli Stillos, Lise Huynh and Debbie Selby in Palliative Medicine
Footnotes
Acknowledgements
We acknowledge Fanchea Lau for aggregating the data from the palliative care database. We acknowledge the members of the specialist palliative care team at Sunnybrook Health Sciences Centre, past and present, who have contributed to the database.
Authorship
MB contributed to study design, data acquisition, data analysis, and data interpretation; and drafted the article. KS contributed to study design, data acquisition, and data interpretation; and revised the article. LH contributed to study design, data acquisition, and data interpretation; and revised the article. DS contributed to data interpretation and revised the article. All authors approved the version to be published and participated sufficiently in the work to take public responsibility for the content.
Data sharing
Requests for data sharing should be directed to MB (
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics
Research ethics approval was granted by the Research Ethics Board of Sunnybrook Health Sciences Centre (project identification number 5670).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
