Abstract
Background:
Persons with advanced cancer may participate in Phase 1 clinical trials – first-in-human trials that are conducted with the main objectives of safety and dosing. The motivations for participation are not well understood and may include hope for cure.
Aim:
To explore the perspectives of persons with advanced cancer in order to understand the motivations for participating in Phase 1 clinical trials, experiences while being on trial and views on palliative care provision.
Design:
Qualitative study with a constructivist stance, using thematic analysis based upon the grounded theory approach.
Setting/participants:
20 persons with advanced cancer who were participating in a Phase 1 clinical trial.
Results:
Many participants described how Phase 1 clinical trial participation was their last hope, as they were cognisant of their advanced disease. Information-seeking needs differed – some needed comprehensive information while others relied on the doctor’s recommendation. Participants experienced varied negative and positive physical and psycho-emotional concerns, and needed to draw on multiple sources of support such as family, friends and healthcare professionals. Some could list potential benefits of palliative care but felt they did not require it yet. The overarching theme was hope and positive thinking as a way of coping.
Conclusions:
The concepts of hope as a way of coping and the supportive presence of healthcare professionals could be weaved into a future model of palliative care to improve the illness journey for patients considering Phase 1 clinical trial participation and other persons with advanced cancer.
Persons with advanced cancer may participate in Phase 1 clinical trials with unrealistic hope regarding trial outcomes.
Persons who participate in Phase 1 clinical trials are cognisant of the advanced nature of their disease, but nevertheless hold on to hope as a way of coping.
The supportive presence of doctors and clinical research coordinators are a source of encouragement for patients.
The concepts of hope as a way of coping and the supportive presence of healthcare professionals could be weaved into the development of palliative care to improve the illness journey and possibly subsequent health outcomes of Phase 1 clinical trial participants and other persons with advanced cancer.
Introduction
Persons with advanced cancer who have exhausted conventional treatments may participate in clinical trials, which are increasing in number due to new therapeutic targets for metastatic disease. 1 Indeed, later phase clinical trials are perceived by oncology healthcare professionals and patients to offer the best treatment available despite limited evidence to suggest that trial participation prolongs survival.2–4 Patients who are not able to access or who progress through these later phase clinical trials may opt to participate in Phase 1 clinical trials. 5 As part of new drug development in a first-in-human setting, the objectives of Phase 1 clinical trials are primarily safety and dosing, with minimal or no therapeutic intent, especially in the early stages when the dose has not been determined. 6 Patients who are deciding whether or not to participate in Phase 1 clinical trials should have a good understanding of the research trial aims, but it is uncertain if they are fully aware about the purposes, risks and benefits of trial participation.7,8
Even though trial drugs have no proven clinical benefit, many persons with advanced cancer may still choose to participate in Phase 1 clinical trials, motivated mainly by hopes of personal therapeutic benefit, altruism in contributing to scientific knowledge, financial provisions, access to healthcare and having a sense of control.2,4,7,9–13 However, many are motivated by unrealistic hope regarding trial outcomes, fuelled by misconceptions about trial aims and outcomes.7,8,14,15 Even where there is adequate information provided and a realistic understanding of prognosis, patients may still hold on to apparently incongruous hopes for extraordinary outcomes.12,16–18
Exploration of patient’s values and goals while navigating the decision-making process of whether or not to participate in a Phase 1 clinical trial may be supported by healthcare professionals in palliative care.19,20 Palliative care may also offer wider benefits to these patients who have advanced cancer and limited prognosis in view of failure to respond to conventional lines of treatment.7,21 Indeed, these patients with limited survival prognosis may have unmet quality of life needs and distress that could be addressed by a palliative care approach.5,21–23 Therefore, the early provision of palliative care could potentially improve outcomes of reduced symptom burden and better psychological wellbeing for patients with advanced cancer who are contemplating participation in Phase 1 clinical trials.24–26 However, the views of persons with advanced cancer on receiving palliative care whilst pursuing Phase 1 clinical trials is largely unexplored.27,28
The overall aim of this study was to explore the perspectives of patients with advanced cancer in order to understand their motivations for participating in Phase 1 clinical trials, experiences while being on trial and views on palliative care provision. Findings from this study could be used to inform the development of palliative care interventions to support these patients in order to improve their outcomes.
Methods
Study design
This was a qualitative study, presented here in accordance with the consolidated criteria for reporting qualitative research (COREQ) guidelines. 29 Semi-structured one-on-one interviews were conducted in English with patients who had stage 4 solid tumours and were registered in the phase 1 trial clinic to explore (i) their motivations and experiences in phase 1 clinical trial participation; (ii) their main concerns and care needs; (iii) their views on palliative care.
Study setting
The study was conducted in National Cancer Centre Singapore, the largest provider of public cancer care in Singapore. Approximately 300 patients are reviewed annually for consideration of phase 1 trial participation, where about 90 patients are enrolled. At any one time, the Experimental Cancer Therapeutics Unit has 30–40 phase I trials open.
Participants
Eligible patients were invited to participate in the study. Inclusion criteria were: (a) aged over 21 years; (b) diagnosed with stage four solid tumour; (c) able to communicate in English and/or Chinese; (d) no cognitive impairment that would prevent them from completing the face-to-face interviews; (e) able to give written informed consent; (f) ongoing participation in a Phase 1 clinical trial. Recruitment of patients was conducted using a purposive sampling approach in order to ensure representation of age, gender and primary cancer type.
Data collection
Eligible patients were first approached face-to-face by their treating physician, who informed them of the study purpose, invited them to participate and sought their permission to be referred to the study team. Written informed consent was obtained. Face-to-face interviews were then conducted by a female research coordinator (HRT), who had training in qualitative interviewing, in a quiet room or a suitable location within the hospital premises chosen by the patients. Family caregivers were allowed to sit in for the interview if this was requested for by the patient. The interviewer (HRT) had no clinical healthcare experience; she did not have any prior relationship with the participants and was not involved in clinical care of the patients. A semi-structured interview guide was used to enable patients to respond in their own words and set their own emphases while providing a frame of orientation. 30 (See Table 1 for sample interview questions). Interviews with participants lasted 30–60 min; they were audio-recorded and later transcribed by HRT.
Sample interview questions.

Data analysis
The transcripts were analysed using thematic analysis with a constructivist stance and based upon the grounded theory approach.31,32 In open coding, two of the investigators (JT and GMY) independently read the transcripts and identified themes using a constant comparative approach and developed codes from these themes. 33 JT is a female research coordinator with 3 years of experience in conducting palliative care research; GMY is female physician with training in specialist palliative medicine. Using Microsoft Excel to facilitate data management, they grouped the codes into categories, and axial coding was used to identify properties of each category and the interrelationship of core categories. Data analysis was an iterative process in which codes were discussed and the categories and sub-categories were adapted throughout the process. Regular discussions were held among members of the study team (JT, GMY and WYO) to resolve differences and generate consensus. Data saturation was reached when no new themes were identified.
This study was reviewed and approved by the SingHealth Centralised Institutional Review Board (CIRB reference number: 2018/2080).
Results
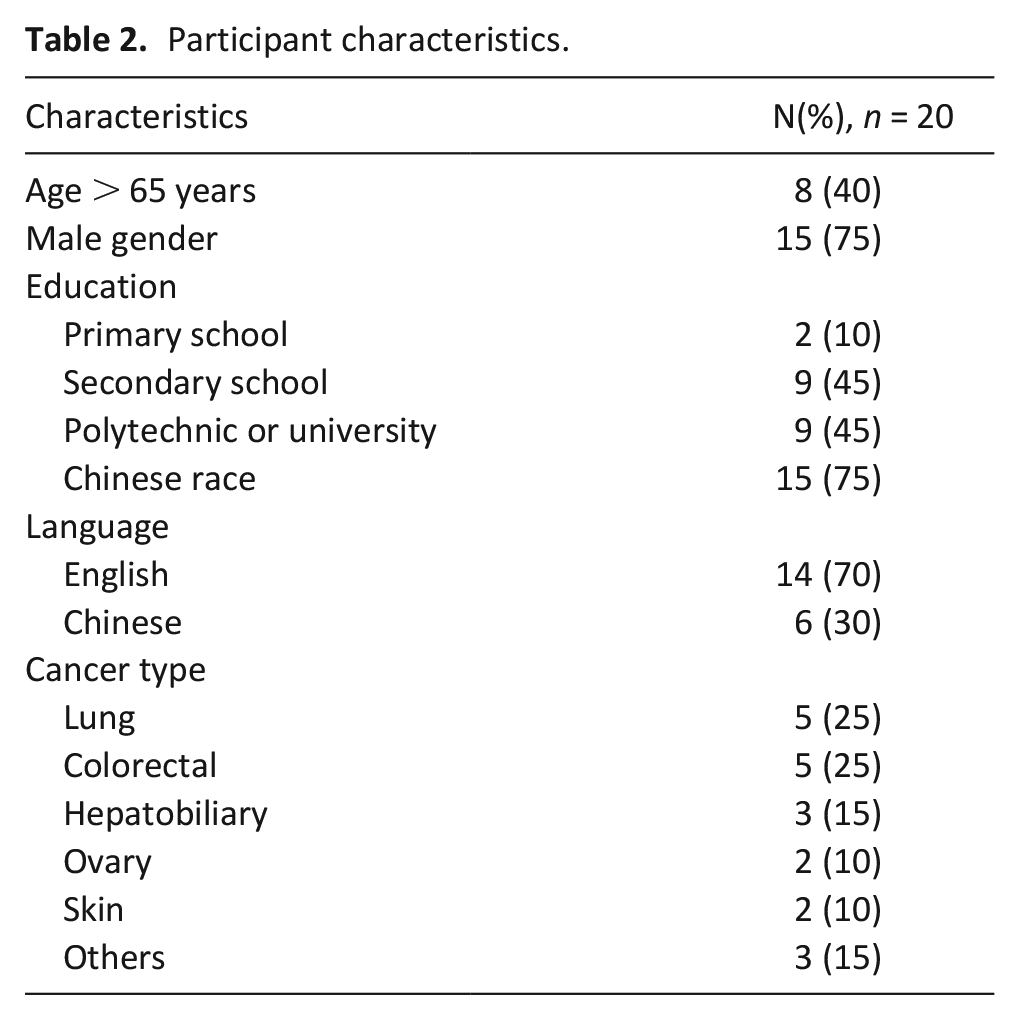
Thirty-four eligible patients were approached and 14 declined to participate for reasons of being uninterested (n = 10) or busy (n = 4). Twenty patients who were recruited from May 2018 to October 2019 shared their perspectives on participating in a Phase 1 clinical trial. The demographic and clinical characteristics of study participants are shown in Table 2. The main themes were: hope as a motivation for Phase 1 clinical trial participation; wide range of information needs to support decision-making; physical and psycho-emotional concerns; needing to draw on multiple sources of support; views on palliative care. The overarching theme was hope and positive thinking as a way of coping.
Participant characteristics.
Hope as a motivation for Phase 1 clinical trial participation
Many patients described how they had exhausted other treatment options and had no choice but to participate in a Phase 1 clinical trial because it was the last and only option and they had nothing to lose. P38 said ‘at least there is a drug to treat. I want to be treated even if it is one percent’. As P56 expressed, ‘there is no more medicine, so if you want to live on, you just have to try’. Several participants hoped that they could achieve some tumour control and some even hoped for cure. P13 said ‘my main thing is to conquer the cancer’ and P46 articulated ‘hope can cure my sickness’.
Other than hope for cure, some were motivated by hope that their participation would benefit future patients and contribute to medical progress – ‘if you don’t have patients coming forward for the trial, then you do not have samples to do the clinical study’ (P64). Participants’ willingness to go ahead with the trial drug was also fuelled by perceived milder side effects compared to conventional chemotherapy and flexibility of withdrawing anytime. P02 shared, ‘Well one of the reasons why I wanted to get on this trial is: There is a Facebook group for this cancer, and everyone on this trial drug said the side effects were minimal’. P41 said, ‘I am allowed withdraw anytime. So it is ok if I feel like I have any profound side effects that I cannot tolerate. That’s why I decided to enrol’. Furthermore, trial participation was free of charge: ‘the two options were this trial and chemotherapy. . . [the doctor] asked me to come for the trial first, because it’s free and it’s open for me. For the chemotherapy, I have to pay expensive money you know. It’s expensive’ (P06).
Information needs to support decision-making
In terms of their main concerns and needs, some participants described needing adequate information before they decided whether or not to participate in the Phase 1 clinical trial. Sources of information included doctors who were able to answer all their questions and address any worries that they had, trial coordinators, brochures and the internet. P34 said, ‘[the doctor] gave me a very detailed account. . . and the information tallied with the information on the internet, so it boosted my confidence to actually go for the trial’.
Although information was helpful in supporting decision-making, many relied on the recommendation of their doctor when ultimately making the decision to participate in a Phase 1 clinical trial. One of the participants (P42) felt that there was too much information: ‘They will introduce to you, introduce, introduce, introduce. I cannot bear to continue listening to him. Why should I listen?’ P42 described the crux of the decision as ‘The doctor asks me to inject this medication. If I don’t inject this medication, I wait for death. If I try and it is good, I can live longer. It is just that simple. Telling me so much, say this, say that, say this, say that, say until I have no interest to listen’.
Physical and psycho-emotional concerns
Participants experienced a range of negative and positive physical concerns which may be due to the underlying cancer or cancer treatment. For example, some described weakness, tiredness, vomiting, diarrhoea, constipation and insomnia; others described better appetite, pain relief, weight gain and feeling stronger. P38 felt that the trial drug was ‘a lot better than chemotherapy’ and the positive effects meant that P34 was even able to go for a ‘one-week holiday with my family’.
Participants also described experiencing psycho-emotional issues such as low mood or anxiety. P41 said, ‘Maybe at times when you are alone. . . or when you see other cancer patients, you think depressing thoughts. But I do not indulge myself, you know, I do not soak myself too much in depression. Really you have to do some self-control, you know, because emotions will kill you’. P38 reflected, ‘Don’t go and think too much. Because I have seen many patients who would think. . . in the end, they worry about this, worry about that’. In a similar vein, P04 decided, ‘I say, every day you feel happy! You have food to eat, you eat. Don’t overthink. If you have money for a holiday, you go for a holiday, nothing wrong. . . don’t restrict yourself’.
While negative symptoms like tiredness meant that P42 was not able to work anymore because ‘even going to the coffee shop is very tiring’, positive physical effects enabled P03 to ‘do all my activities as normal’. ‘You feel better, you can do your normal daily activities, you don’t feel lethargic, you don’t feel let down’ (P06). Several participants also described how objective improvement in terms of reduction in tumour size maintained their hope for cancer control, as articulated by P34 who said ‘So now we are seeing improvement, I am more confident that I will recover fully’. The exact measurements were recalled by P15: ‘In October my tumour size was 7.5cm, but after I got six months of the infusion, it became 3.9cm’.
Needing to draw on multiple sources of support
Family, friends and work colleagues were sources of support for participants. This support could be in the form of companionship. For example, P04 described how interactions with ‘my grandson – everyday talk to me, sing song to me, then play piano’ led to the feeling of being happy. The support could also be expressed through words of encouragement, prayers, social visits, gestures of care and attention and practical provisions at work such as light duty or a flexible working schedule.
Participants also felt supported by healthcare professionals, particularly research coordinators who helped to navigate the complicated healthcare system. Phase 1 clinical trial participation took up a lot of time and involved many visits for numerous investigations and assessments. However, this burden was lightened by the research coordinators who arranged the logistics of multiple appointments and investigations, and managed side effects with advice from doctors. P03 described, ‘the coordinator contacts me, for information, for dates and so on. They are the link, the bridge, and they are very useful, very helpful’. P34 also shared ‘if I have any questions, I ask [the research coordinator], then she would ask the doctor and come back to us’,
Other than logistical assistance, participants also felt supported when healthcare professionals showed empathetic honesty, kindness and built good relationships with the patients and families. P38 articulated, ‘Actually the people in the hospital are very important. The doctors, the nurses, the coordinator – if they care for you, that will be the best. Previously when I was staying in the hospital, the nurse will come and say, “Are you well already? Are you better?” A greeting like this is honestly very good to a patient’. P13 expressed, ‘These two go together – doctor advice and doctor encouragement. These two combined can provide a medical miracle!’
Views on palliative care
When asked for their views on whether palliative care could offer support for patients like them, some participants were unsure about what palliative care was, while others could list some potential benefits of palliative care but felt that they did not require it. Possible benefits suggested by participants included pain relief, psychological support and caregiver support. However, palliative care was perceived as something that is only appropriate for the ‘end of life’ or ‘terminal’ phase, and that it was not applicable to them yet. For example, P15 initially shared their understanding about what is palliative care – ‘Palliative care is for people who have terminal disease. You go to palliative care to relieve the suffering and not to cure the disease’ and went on to describe how palliative care may be helpful in pain management or provision of psychological support. However, when asked if it would be useful, P15 said, ‘for myself, I don’t think I will need. Because I know all the things about my disease. . . I have positive thinking’.
Overarching theme of hope and positive thinking as a way of coping
The overarching theme that emerged was that of hope – hope that motivated initial participation in Phase 1 clinical trials, hope that sustained them through the challenges, hope that was maintained by multiple sources of support from family, friends, work colleagues and healthcare professionals. Several participants described how a mindset of positive thinking and staying optimistic was central to what helped them to cope in their cancer journey. This positivity was juxtaposed with a cognisance that their cancer was very advanced – ‘although the percentage [of cure] won’t be that high, you have to accept it. . . continue with life. . . don’t give up’ (P19). P03 recognised that the trial drug was ‘experimental’, but shared their outlook that ‘positive thinking is very important. We cannot let whatever illness or disease take over and defeat us’. P44 said ‘I don’t have any expectations. . . the only thing I have is I can’t go in there feeling negative’.
Discussion
Hope as a way of coping
This study explored the perspectives of patients with advanced cancer participating in a Phase 1 clinical trial. We found that hope was the main motivation for deciding to participate in Phase 1 clinical trials – hope for cure or at least tumour control.11,34 The primary objective of phase 1 clinical trials is dose-finding and toxicity, and while it is possible that patients had inadequate information to make an informed decision regarding Phase 1 clinical trial participation, the emerging experience with biomarker-driven studies of novel anti-cancer agents demonstrating early successes even in Phase 1 trials may have shaped this perspective.8–10,35,36 Provisions for identification and selection of patients through these biomarkers means that Phase 1 clinical trials may be offered to patients with therapeutic intent, and it may not be unreasonable to harbour hopes of clinical benefit.6,9,10
For participants in our study, their hope for cure was juxtaposed with a cognisance that their disease was very advanced – should patients be allowed to hope for a hopelessly optimistic outcome? 37 Hope is a multifaceted concept with three main forms being described in the literature. First, unrealistic or irrational hope is where the expectation of cure may preclude mental preparation for poorer and more likely outcomes, and has been blamed for inappropriate treatment decisions and overly aggressive care at the end of life. 38 Second, realistic or rational hope is where a person may understand that the prognosis but nonetheless still refuses to ‘give up’.11,12 This kind of hope may be a coping strategy that makes an otherwise bleak situation bearable, and may even contribute to greater productivity and wellbeing.39–41 Third, a trusting kind of hope goes beyond expectation of a particular outcome and is therefore a more enduring kind of hope. This is defined by Herth as ‘an inner power directed towards enrichment of being’, and can be bolstered by trusting relationships with God, family or the healthcare team.42–44 For patients with advanced cancer who nonetheless hold on to hope, perhaps hope can be iteratively reframed from an unrealistic hope to a more enduring kind of hope that extends beyond the expectation of a specific outcome.37,43
Relevance of palliative care in Phase 1 clinical trials
The question of how hope could be fostered in the context of advanced cancer is an important subject in the field of palliative care.37,45–47 In cases where patients start out having unrealistic hopes, the role of the clinician is then to help nurture a greater range of hopes that would include realistic hopes. 37 However, doctors and nurses themselves may have unrealistic hopes of survival benefit from clinical trial participation, making it difficult for healthcare personnel on the ground to shift from supporting unrealistic hopes to nurturing realistic hopes. 2 In future palliative models, healthcare staff could be trained to widen their provision of hope by being present and demonstrating caring, conveying non-abandonment that is comforting and strengthening for the patient.43,44,48 Through interactions between patients, families and the healthcare team, hope is collectively cultivated and expressed, and subsequently drawn on as a source of solidarity and support. 47
Even though palliative care was recognised as a source of support by participants in our study, they also felt that palliative care was not relevant to them. In recent years, palliative care has been increasingly recognised as being beneficial to persons who have advanced cancer, even if they were concurrently receiving chemotherapy or other cancer-directed therapy.49–52 However, there may still be a reluctance to accept palliative care among Phase 1 trial participants due to perceived conflict between the goals of pursing aggressive treatment to prolong life in a Phase 1 clinical trial versus prioritising quality of when receiving palliative care. 25 Nonetheless, the pursuit of Phase 1 clinical trials and palliative care are not as incongruent as they appear to be.5,27,28 Indeed, palliative care interventions for patients on Phase 1 studies were reported to result in reduction of distress and improvement in some aspects of quality of life for patients and their caregivers.24,53 For reasons of feasibility and scalability, future palliative care models may include generalist palliative care provided by the oncologists or even clinical trial coordinators, given their central role in Phase 1 clinical trials.54,55
Strengths and limitations
Our study used qualitative methodology to explore the perspectives of persons with advanced cancer to gain a deeper understanding of their motivations, experiences and views on palliative care. There are however a few limitations. First, we recruited a small number of patients from a single site, therefore their views may not be representative of patients from other geographic regions. Furthermore, majority of participants were male, although five females were included. Second, only one interview was conducted for each patient, therefore changes in perspectives over time were not captured. Third, the views of family caregivers who may have different needs and challenges were not elicited, as this study focused on the experiences of patients themselves are salient to understanding the illness journey.
Conclusions
In conclusion, while participants of Phase 1 clinical trials were cognisant of the advanced stage of their cancer, many described how hope helped them cope in an otherwise hopeless situation. Good relationships with doctors and clinical research coordinators was also vital in supporting patients and families through the challenges of living with advanced cancer. Even though this study explored the perspectives of Phase 1 clinical trial participants, the findings could be extrapolated to patients with advanced cancer who have progressed through initial lines of cancer treatment. The concepts of hope as a way of coping and the supportive presence of healthcare professionals could weaved into future iterations of palliative care models to improve the illness journey and possibly subsequent health outcomes for patients with advanced cancer.
Footnotes
Acknowledgements
The authors wish to thank the patients who participated in the study, and Ms Han Rou Teo (HRT) for conducting the interviews.
Author contributions
GMY obtained funding for this research study. JT, WYO and GMY contributed to the data analysis and interpretation and drafted the initial manuscript. All authors – GMY, WYO, JT, JD, SH, DT, PN – contributed to the study conceptualisation and design, critically reviewed and revised the manuscript, approved the final manuscript for submission, and participated sufficiently to take responsibility for the content of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Singapore Ministry of Health’s (MOH) National Medical Research Council (NMRC) under the NMRC grant (grant number: NMRC/SEEDFUNDING/021/2017) and by NCCS Cancer Fund. GMY received salary support from Lien Centre for Palliative Care, Duke-NUS Medical School.
Ethics and consent
This study was reviewed and approved by the SingHealth Centralised Institutional Review Board (CIRB reference number: 2018/2080).
Data sharing
The data of this study are stored at the National Cancer Centre of Singapore. Data are available upon reasonable request to the corresponding author.
