Abstract
Background:
Patients with chronic obstructive pulmonary disease (COPD) face limited treatment options and inadequate access to palliative care.
Aim:
To provide a pragmatic overview of clinical guidelines and produce evidence-based recommendations for severe COPD. Interventions for which there is inconsistent evidence to support their use and areas requiring further research were identified.
Design:
Practice review of guidelines supported by scoping review methodology to examine the evidence reporting the use of guideline-recommended interventions.
Data sources:
An electronic search was undertaken in MEDLINE, EMBASE, PsycINFO, CINAHL and The Cochrane Database of Systematic Reviews, complemented by web searching for guidelines and publications providing primary evidence (July 2021). Guidelines published within the last 5 years and evidence in the last 10 years were included.
Results:
Severe COPD should be managed using a multidisciplinary approach with a holistic assessment. For stable patients, long-acting beta-agonist/long-acting muscarinic antagonist and pulmonary rehabilitation are recommended. Low dose opioids, self-management, handheld fan and nutritional support may provide small benefits, whereas routine corticosteroids should be avoided. For COPD exacerbations, systematic corticosteroids, non-invasive ventilation and exacerbation action plans are recommended. Short-acting inhaled beta-agonists and antibiotics may be considered but pulmonary rehabilitation should be avoided during hospitalisation. Long term oxygen therapy is only recommended for patients with chronic severe hypoxaemia. Short-acting anticholinergic inhalers, nebulised opioids, oral theophylline or telehealth are not recommended.
Conclusions:
Recommended interventions by guidelines are not always supported by high-quality evidence. Further research is required on efficacy and safety of inhaled corticosteroids, antidepressants, benzodiazepines, mucolytics, relaxation and breathing exercises.
Patients with severe COPD face frequent exacerbations, hospitalisation and shortened survival but limited access to palliative care.
Neither standardised guidelines nor synthesis is available on recommendations for severe COPD.
Many clinicians are unsure how to best treat or support severe COPD.
Not all guidelines are developed based on robust evidence.
There is a lack of high-quality studies focussing on patients with severe COPD.
Evidence supports the multidisciplinary approach with combination inhaled therapies.
Practical recommendations are made for the management of stable COPD and COPD exacerbations respectively balancing the efficacy and harms.
Severe COPD should be managed by a multidisciplinary approach with a holistic assessment.
Stable patients will benefit from combination inhaled therapies such as long-acting beta-agonist/long-acting muscarinic antagonist are preferred over monotherapies, low dose opioids and non-pharmacological interventions such as self-management, pulmonary rehabilitation, a handheld fan can provide small benefits, but routine corticosteroids should be avoided.
For COPD exacerbations, systematic corticosteroids, non-invasive ventilation and exacerbation action plans are recommended, short-acting inhaled beta-agonists and antibiotics may provide benefits but pulmonary rehabilitation should be avoided during hospitalisation.
Long term oxygen therapy is only recommended for patients with chronic severe hypoxaemia.
Short-acting anticholinergic inhalers, nebulised opioids, oral theophylline or telehealth are not recommended.
Further research is required to investigate the efficacy and safety of inhaled corticosteroids, antidepressants, benzodiazepines, mucolytics, relaxation and breathing exercises in patients with severe COPD.
Introduction
Chronic obstructive pulmonary disease (COPD) is a leading cause of death worldwide. 1 Poorly managed severe COPD (forced expiratory volume in 1 s (FEV1) <50% predicted 2 ) may lead to frequent exacerbations, hospitalisation and shortened survival. 3 Patients experience a similar symptom burden to those with lung cancer with unmet palliative care needs, 4 yet have limited access and lower uptake of palliative care often due to unpredictable prognosis. 5
Palliative care has predominately focussed on malignant disease, neither standardised guidelines nor synthesis is available for end of life of non-malignant diseases or the management of patients with severe COPD concerning early palliative care measures. In addition, not all guidelines are evidence-based. Although a growing body of research supports early palliative care approaches for better health outcomes, 6 many health systems are not set up to provide trained palliative care clinicians to patients and many clinicians experience difficulties in when, how and what to initiate palliative care, 7 leading to inadequate provision of palliative care for COPD patients. Palliative care is shown only frequently to be provided within the last few weeks of life in patients with severe COPD instead of their last year of life, and it is more likely for patients to receive it if a lung cancer co-diagnosis exists. 5 We aim to (1) provide a critical yet pragmatic overview of clinical guidelines, (2) produce evidence-based recommendations for practice and (3) identify gaps that require further research.
Methods
This practice review is to provide an overview of current guidelines with a supporting evidence based summary of recommendations drawn from the available evidence. National and international guidelines that described the management of COPD in adults were searched and examined and interventions recommended by guidelines were identified. A scoping review was then undertaken to provide evidence base supporting the recommendation of these interventions and identify areas where the evidence-base is lacking, and further research is required. A series of searches in MEDLINE, EMBASE, PsycINFO, CINAHL and The Cochrane Database of Systematic Reviews were made using a range of keywords and subject headings (July 2021, Supplemental Material 1). References lists of guidelines and supporting evidence were scrutinised. Guideline and supporting study eligibility are shown in Table 1. It is worth noting that asthma and COPD are different disorders although they may coexist, in which asthma guidelines for pharmacotherapy plus pharmacological and non-pharmacological approaches for COPD should be used. 2
Eligibility of guideline and supporting study.

Identified citations were exported to EndNote for deduplication and selected after independent screening of titles and abstracts by two researchers (YF, EJC) any discrepancies resolved by discussion with a third author. Results are presented according to Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews was followed 8 (Supplemental Material 2).
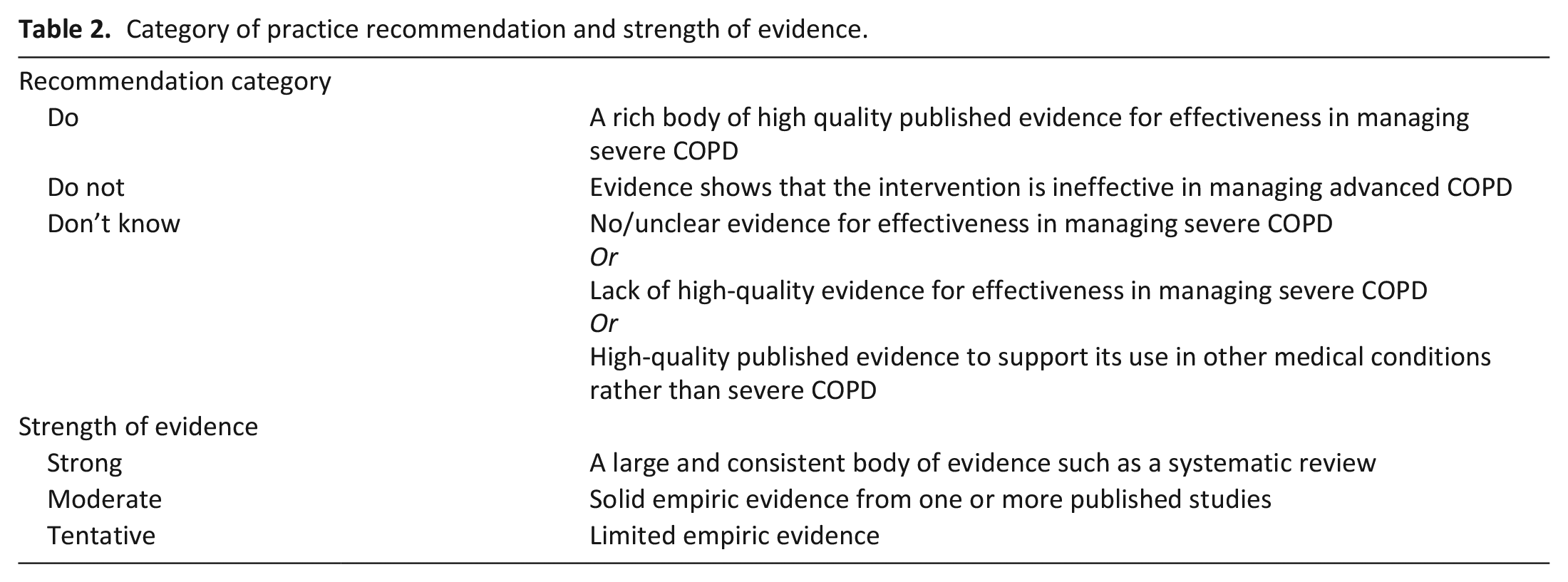
Each intervention was allocated to a practice recommendation of ‘Do’, ‘Don’t’ or ‘Don’t know’ based on consistency and quality of the supporting empirical evidence. The strength of recommendation was rated as ‘strong’, ‘moderate’ and ‘tentative’ balancing benefits and harms according to criteria listed in Table 2.
Category of practice recommendation and strength of evidence.
Results
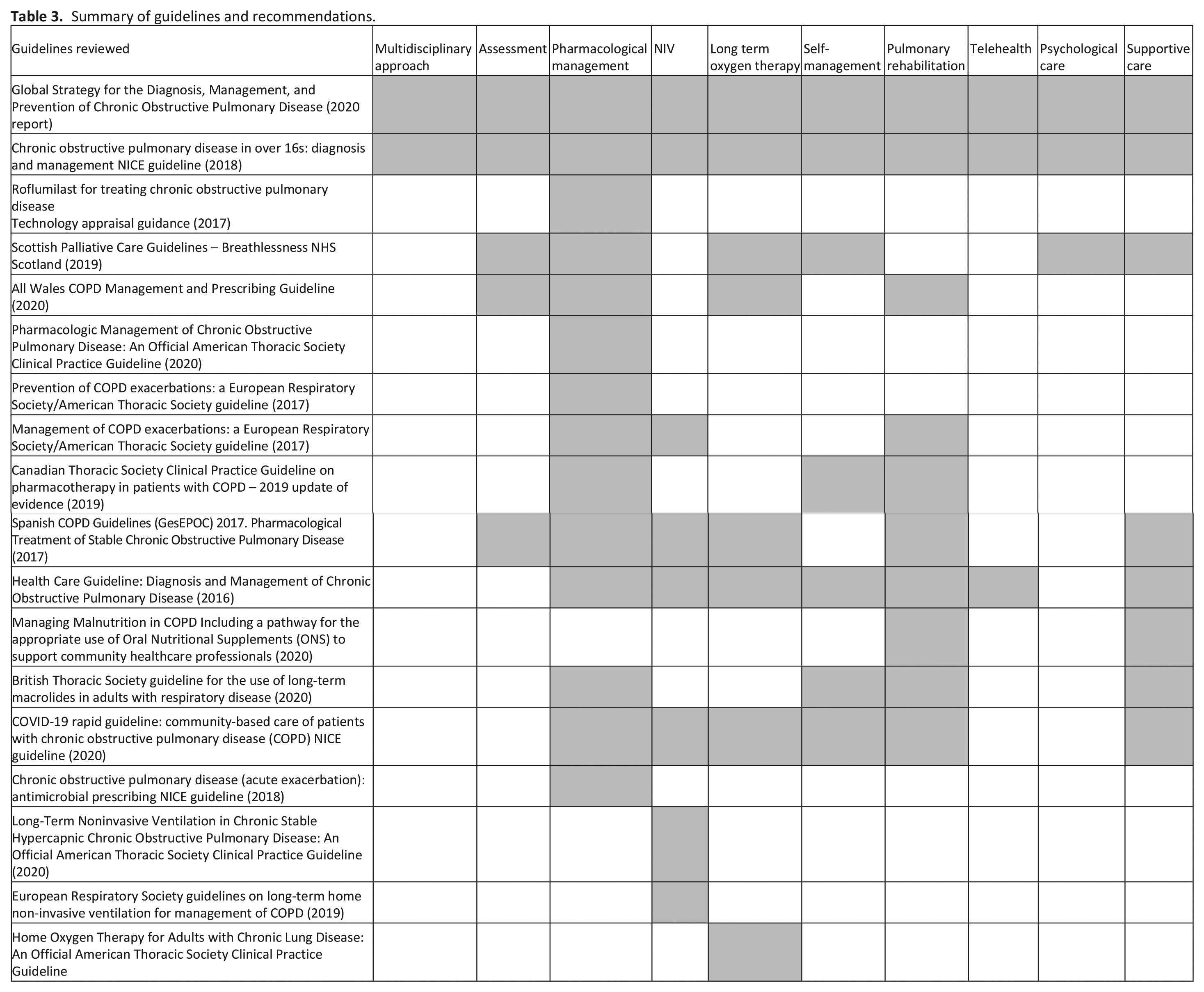
Twenty four guidelines were identified but 18 were included as six guidelines used other guidelines as the prime evidence base. A summary of guidelines with recommendations is presented in Table 3. The strength of each recommendation is presented in Table 4. Evidence used to support practical recommendations is presented in Supplemental Material 3.
Summary of guidelines and recommendations.
Practice recommendations and strength of evidence.
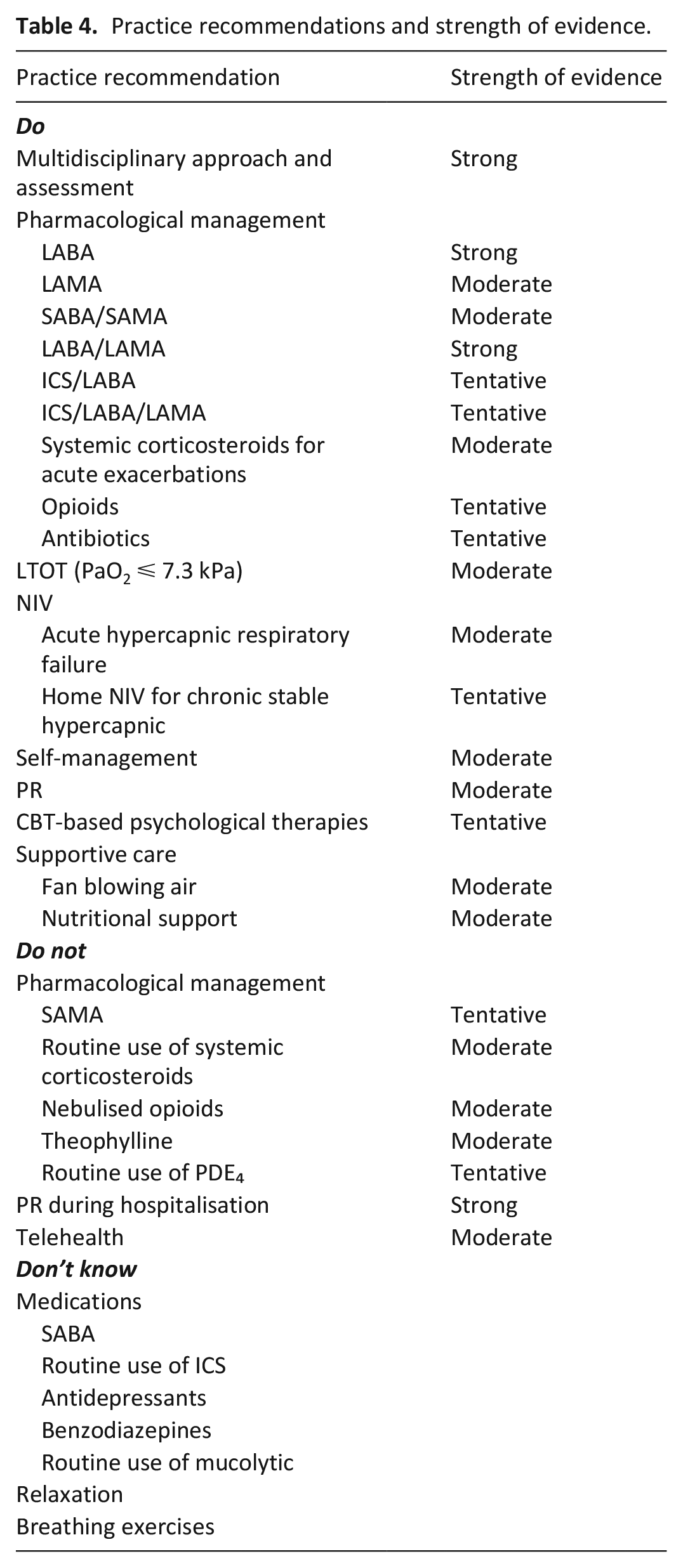
Do
Multidisciplinary management and assessment
A multidisciplinary approach is strongly recommended. Multidisciplinary and multi-treatment improve breathlessness, dysponea, fatigue, disease impact and activity and reduce hospital admissions and hospitalisation days. Mixed evidence on emotional and symptom mastery are observed.9,10 A randomised controlled trial (RCT) finds early integration of palliative care increases survival rate for patients. 11
Holistic assessment is recommended and severity of airflow limitation should be indicated. A comprehensive assessment of symptoms measuring breathlessness by MRC scale 12 and symptom burden by COPD Assessment Test (CAT), 13 exercise capacity by the 6-minute walking distance,14,15 oxygen saturation, 16 history of hospital admissions and exacerbations17,18 should be undertaken. Comorbidities 19 and Alpha-1 antitrypsin deficiency 20 should be assessed. Blood eosinophil counts 21 should also be used in combination with clinical assessment of exacerbation risk to predict the treatment effect of inhaled corticosteroids containing regimes.
Pharmacological management
Beta-agonist
Cochrane reviews provide moderate to high-quality evidence that long-acting beta-agonist (LABA) is associated with improvement in lung function, exacerbations and quality of life (QoL), but no impact on mortality.22,23
Anticholinergics
Long-acting muscarinic antagonist (LAMA) are recommended to reduce exacerbation.2,24 Tiotropium reduces risk of exacerbations (and related hospitalisations) and clinical deterioration, with an increase in clinical improvement and QoL.25–27 An associated risk of increased mortality is observed in patients with soft mist inhaler compared with placebo 25 but no difference in deaths with ipratropium bromide. 26
Combination inhaled therapy
Guidelines2,28–32 recommend the use of a combination of medications over monotherapies with different mechanisms that may increase overall efficacy and lower adverse effects.
Short acting beta agonists (SABA)/short-acting anticholinergic (SAMA)
Recent evidence is limited on the use of SABA/SAMA but suggested a potential effect on exacerbation, patients also showed a preference for using Respimat inhaler. 34
LABA/LAMA
Long term efficacy and safety of LABA/LAMA are demonstrated across multiple trials for bronchodilation and dyspnoea33–36 in moderate and severe patients, leading to a strong Do. Cochrane reviews further suggest a small improvement in lung function and QoL with LABA/LAMA but inconsistent effects on exacerbations which may be caused by heterogeneity.37,38 Compared with nebulised SABA/SAMA, effect on FEV1 is not superior over 6h with a slower reaching of lower peak FEV1 in dry powder with LABA/LAMA. 39
ICS/LABA
Cochrane reviews suggest that ICS/LABA reduces exacerbations and potentially improves mortality, lung function, symptoms and QoL, compared with ICS, LABA or usual care.40–42 However an increased risk of pneumonia is noted in patients on combined inhalers. 41
ICS/LABA/LAMA
Triple therapy decreases the risk of exacerbations and improves QoL in patients with and without a history of exacerbations in the past year, however it increases the risk of pneumonia compared with LABA/LAMA.43–45 A Cochrane review provides good quality evidence ICS/LABA/tiotropium has a small effect on lung function and QoL compared with tiotropium alone. No conclusion could be drawn on impact on exacerbations due to heterogeneity between trials. 46
Systemic corticosteroids in acute exacerbations
There is high-quality evidence to recommend systemic corticosteroids for acute exacerbations, which improve symptoms and lung function, reduce treatment failure and relapse and duration of hospitalisation. 47 Short courses of oral corticosteroids are in favour leading to a decrease in pneumonia admissions and all-cause mortality. 48 Risk of adverse effects such as hyperglycaemia is increased.
Opioids
A tentative recommendation is made for low dose (10–30 mg daily) opioids. 49 Reviews suggest a reduction in breathlessness with systemic opioids in stable severe COPD without effect on exercise capacity. 50 Opioid-related adverse events including nausea, vomiting and constipation are common but often self-limiting on withdrawal. 49 Trials suggest improvement in patient’s breathlessness with low dose opioids.51,52 Future studies to investigate longer-term benefits and risks are needed.
Antibiotics
Antibiotics are tentatively recommended concerning antibiotic resistance and other adverse events.2,24,28,32,53–57 A Cochrane review finds small effects in outpatients on treatment failure, no effect in inpatients with severe exacerbations, but a large effect among patients in the intensive care unit patients. Routine use of macrolides shows a clinically meaningful reduction in exacerbations.58–60 Serious adverse events are reported including hearing impairment, long QTc and tinnitus.59,60
Long term oxygen therapy (LTOT)
Oxygen for more than 15 h per day is recommended for patients with chronic severe hypoxaemia (a PaO2 ⩽ 7.3 kPa). 61 Cochrane reviews and trials suggest survival benefits in patients with severe hypoxaemia 62 but improve breathlessness only during exercise in mildly hypoxaemic and non-hypoxaemic patients. 63 No effect is observed on QoL or hospitalisation, although serious adverse events are rare.63–65 For patients with advanced life-limiting diseases with PaO2 > 7.3 kPa, palliative oxygen does not improve breathlessness relief compared with air65,66 although studies are undertaken with a majority of cancer patients. Regular assessment and support should be undertaken to ensure compliance, along with monitoring for hypercapnia and optimal oxygen saturation.
Non-invasive ventilation (NIV)
NIV is recommended for patients with acute hypercapnic respiratory failure in patients hospitalised for acute exacerbations. 67 A Cochrane review 68 with moderate evidence suggests NIV decreases risk of mortality, needing endotracheal intubation and complications and reduces length of hospital stay. It is also associated with improved pH and partial pressure of oxygen. For stable patients, a tentative recommendation is made due to weak evidence of efficacy of long term home NIV, showing a small effect on QoL, possible fewer hospitalisations but no survival benefit.69,70 Mild but manageable adverse effects are common. 71
Self-management
Self-management is often delivered in an interactive process covering multi interventions including an action plan for acute exacerbation. Cochrane reviews72,73 with low to moderate quality evidence show self-management reduces respiratory-related hospital admissions and improves QoL and dyspnoea. The use of COPD exacerbation action plans with a single brief educational component, in conjunction with ongoing support, is found to reduce in-hospital healthcare utilisation and increases treatment of COPD exacerbations with corticosteroids and antibiotics. 74 Heterogeneity in interventions components, duration and outcome measures requires further studies to inform the most optimised content.
Pulmonary rehabilitation
Pulmonary rehabilitation has a strong focus on physical exercise and has benefits on dyspnoea, fatigue, emotional function and symptom mastery, and QoL in studies of very low to moderate quality. 75 In patients with a recent hospitalisation for an acute exacerbation, moderate to large effects are observed on QoL and exercise capacity. Pulmonary rehabilitation also reduces readmissions but does not improve mortality, but results are heterogeneous. 76 However trials suggest mixed findings of the effect on long-term clinical outcomes.77,78 Further evidence is needed to determine the effective components and format.
Cognitive Behavioural Therapy (CBT) based psychological therapies
A Cochrane review with low-quality evidence demonstrates a small effect on depression. 79 Other studies suggest a small impact on anxiety,80–82 but there is one review that reports a non-significant effect of using CBT on patients’ emotional status. 83 High-quality studies are needed to confirm effectiveness of psychological therapies and to identify comparative effectiveness.
Supportive care
Handheld fan
Small trials suggest use of a handheld fan directed to the face may improve patients’ breathlessness.84–87 Patients show favour in routine use considering low costs and an absence of risks.
Nutritional support
Moderate-quality evidence supports an association between supplementation and weight gain, respiratory muscle strength, walking and QoL with larger effects if malnourished.88,89 A review using individual patient data suggests vitamin D reduces moderate and severe exacerbations in patients with baseline 25-hydroxyvitamin D levels <25 nmol/L. 90 Common adverse effects may include bloating, leading to low supplementation adherence.
Do not
SAMA
A Cochrane review with moderate to high quality of evidence suggests ipratropium bromide monotherapy is less effective in improving lung function, hospital admissions, exacerbations and QoL, compared to tiotropium. 26
Routine use of systemic corticosteroids
A Cochrane review finds that longer courses (10–14 days) of systemic corticosteroids (both oral and intravenous) do not reduce the risk of treatment failure, relapse or time to re-exacerbation compared with shorter courses in severe or very severe patients. No difference in the likelihood of adverse events is found, including hyperglycaemia, hypertension, gastrointestinal tract bleeding and symptoms of congestive heart failure or ischaemic heart disease. 91 A moderate Do not recommendation is made due to lack of efficacy and potential toxicity.
Nebulised opioids
A Cochrane review with low-quality of evidence finds no evidence that nebulised opioids are more effective than placebo although a wide range of doses is delivered (1–50 mg). 92 Another review demonstrates a lack of benefit on exercise capacity and breathlessness. Common adverse effects include lightheadedness, dizziness and mild nausea. 50 A moderate Do not recommendation is made due to lack of clinical benefit in studies of poor quality.
Theophylline
Recent research is limited and not in favour of theophylline concerning the likelihood of serious adverse events and no additional benefit in reducing exacerbations.93,94 A possible increased risk of overall exacerbations is reported. 93
Routine use of phosphodiesterase-4 (PDE₄)
Routine use of PDE₄ should be avoided given the small benefit but a range of adverse events. A Cochrane review 95 suggests PDE₄ is associated with a small improvement in FEV1, QoL and exacerbation frequency in moderate to very severe patients. No effects are observed on symptoms or exercise tolerance. However patients experience increased depressive symptoms and insomnia, weight loss and diarrhoea, nausea and vomiting.
Pulmonary rehabilitation during hospitalisation
When Pulmonary rehabilitation is initiated early during acute admission for exacerbation, no consistent improvement is observed on mortality, hospital readmission, QoL or exercise capacity. 55 Trials demonstrate no difference in readmission or recovery of physical function but a slightly higher mortality rate. 96 Higher rates of adverse events relating to fall, blood pressure, heart rate although not severe are reported.55,97
Telehealth
Telehealth is remote delivery of healthcare or monitoring disease, 98 which has no impact on mortality but a possible improvement on QoL and emergency department and hospital attendance, with no excessive costs incurred. 99 However, a recent review suggests a lack of effect compared with usual care. 98 A large RCT finds no benefit on psychological outcomes or QoL. 100
Don’t know
Short acting beta agonists (SABA)
There is some evidence for a beneficial effect of albuterol with side effects reported related to heart rate in non COPD contexts.101–103 The potential benefit for severe COPD needs careful consideration and high quality evidence is urgently needed.
Routine use of ICS
Inconsistent recommendation is made for use of ICS2,56 with conflicting evidence. A Cochrane review with good quality evidence suggests routine use of ICS reduces mean rate of exacerbations with no benefits on FEV1 and mortality. Although there is a slower decline in QoL, likelihood of oropharyngeal candidiasis, hoarseness and pneumonia increases. 104 A possible higher mortality rate is reported in a trial in patients with fluticasone propionate alone compared with placebo. 105 Research on long term adverse effects is needed.
Antidepressants
Short term nortriptyline has some effect on depressive symptoms but not on dyspnoea, FEV1, hospital utilisation, exercise tolerance or QoL. Evidence supporting use of selective serotonin reuptake inhibitors is lacking with reported nausea and dizziness. 106
Benzodiazepines
Benzodiazepines do not improve breathlessness in patients with advanced cancer and COPD regardless of type of benzodiazepine, dose, delivery route and frequency or duration with more adverse events of drowsiness compared with placebo but less compared to morphine. 107 A longitudinal cohort study observes an association between higher benzodiazepines doses and increased mortality. 108
Routine use of mucolytics
Mucolytics may reduce likelihood of an acute exacerbation. A possible benefit on days of disability and hospitalisations is observed with no association with increased adverse events. 109
Relaxation
Although relaxation is recommended as a way to strengthen coping and functional ability however no high-quality evidence is available to support its routine practice. 110 A meta-analysis with a high degree of heterogeneity in intervention components concludes only small effects on FEV1, anxiety and QoL. 111 An explorative RCT suggests benefit of a brief relaxation exercise. 112 RCT’s evidence is needed to determine comparative effectiveness for this low risk and inexpensive intervention.
Breathing exercises
No consistent effects of breathing exercises (with or without supervision) can be drawn on patients’ dyspnoea or QoL, 113 from a Cochrane review with low-quality evidence. Another review suggests no improvement in dyspnoea but a beneficial effect on respiratory rate. 114 Further evidence is needed due to large variability among studies examined in current reviews. 115
Limitations
A scoping review rather than a systematic review was undertaken. No quality appraisal was performed on supporting evidence. Not all evidence focussed solely on patients with severe COPD. Concerns were given to the current validity of guidelines, all guidelines published within the last 5 years and more than half published within the last 3 years, which is within the timeframe that they may be considered current. 116
Summary
A multidisciplinary approach with holistic assessment should be implemented. Treatment with LABA or LAMA is effective, but combination inhaled therapies provide greater benefits without additional adverse effects. However an ICS as part of combination therapy needs to be used with caution considering risk of pneumonia.
For stable patients, LABA/LAMA and pulmonary rehabilitation are recommended, low dose opioids, self-management, handheld fan and nutritional support may be beneficial. However routine corticosteroids should be avoided.
For COPD exacerbations, systematic corticosteroids, non-invasive ventilation and exacerbation action plans are recommended, short-acting inhaled beta-agonists and antibiotics may be beneficial but pulmonary rehabilitation should be avoided during hospitalisation.
Long term oxygen therapy is only recommended for patients with chronic severe hypoxaemia.
Short-acting anticholinergic inhalers, nebulised opioids, oral theophylline or telehealth are not recommended.
Recommendations by guidelines are not always consistent with robust evidence. There is a lack of high-quality studies focussing on patients with severe COPD. More research is needed to investigate efficacy and safety of ICS, antidepressants, benzodiazepines, mucolytics and supportive care targetting severe COPD.
Supplemental Material
sj-pdf-1-pmj-10.1177_02692163221079697 – Supplemental material for Evidence-based management approaches for patients with severe chronic obstructive pulmonary disease (COPD): A practice review
Supplemental material, sj-pdf-1-pmj-10.1177_02692163221079697 for Evidence-based management approaches for patients with severe chronic obstructive pulmonary disease (COPD): A practice review by Yu Fu, Emma J Chapman, Alison C Boland and Michael I Bennett in Palliative Medicine
Footnotes
Author contributions
YF developed the review aims and undertook literature search, data synthesis and preparation of the manuscript. EJC checked guidelines identified and contributed to data synthesis. ACB and MIB checked data synthesis and contributed to clinical interpretation. All authors revised and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project received funding from the European Union’s Horizon 2020 research and innovation programme (Grant 825750).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
