Abstract
Background:
People with dementia requiring palliative care have multiple needs, which are amplified in long-term care settings. The European Association for Palliative Care White Paper offers recommendations for optimal palliative care in dementia integral for this population, providing useful guidance to inform interventions addressing their specific needs.
Aim:
The aim of this study is to describe the components of palliative care interventions for people with dementia in long-term care focusing on shared decision-making and examine their alignment to the European Association for Palliative Care domains of care.
Design:
Systematic review with narrative synthesis (PROSPERO ID: CRD42018095649).
Data sources:
Four databases (MEDLINE, CINAHL, PsycINFO and CENTRAL) were searched (earliest records – July 2019) for peer-reviewed articles and protocols in English, reporting on palliative care interventions for people with dementia in long-term care, addressing European Association for Palliative Care Domains 2 (person-centred) or 3 (setting care goals) and ⩾1 other domain.
Results:
Fifty-one papers were included, reporting on 32 studies. For each domain (1–10), there were interventions found aiming to address its goal, although no single intervention addressed all domains. Domain 7 (symptom management; n = 19), 6 (avoiding overly aggressive treatment; n = 18) and 10 (education; n = 17) were the most commonly addressed; Domain 5 (prognostication; n = 7) and 4 (continuity of care; n = 2) were the least addressed.
Conclusion:
Almost all domains were addressed across all interventions currently offered for this population to various degrees, but not within a singular intervention. Future research optimally needs to be theory driven when developing dementia-specific interventions at the end of life, with the European Association for Palliative Care domains serving as a foundation to inform the best care for this population.
Dementia is a progressive life-limiting illness, and a palliative approach to care is considered the best practice in its advanced stages.
The European Association for Palliative Care White Paper offers recommendations for optimal palliative care for people with dementia.
A review of current palliative care interventions for people with dementia is timely to better understand their design and examine how intervention components align with the European Association for Palliative Care framework domains.
This study indicates that a range of interventions is being developed for people with dementia receiving palliative care in long-term care.
This is the first review to use the European Association for Palliative Care White Paper to investigate the content being addressed in interventions for people receiving palliative care for their dementia in long-term care settings.
No single intervention addresses all 10 considered European Association for Palliative Care domains of interest, but all 10 domains are addressed to some extent across various interventions, and to various degrees.
The findings indicate that current research is more focussed on developing interventions that provide education and upskilling of health care teams, support optimal treatment of symptoms and comfort care, and those that seek to prevent aggressive treatment at the end of life. Fewer interventions focus on prognostication and continuity of care.
This article highlights a shift from prognosis-driven palliative care models to need-based care models for people with dementia in long-term care settings.
The European Association for Palliative Care framework offers a useful way to structure and consider complex, multicomponent interventions for people with dementia and palliative care needs, and to place such interventions in context, thus improving the generalisability and applicability of their findings.
Future research could highlight the inter-relatedness of domains, and how intervening at one domain may also improve outcomes in others.
Introduction
Dementia is a progressive life-limiting illness, and a palliative approach to care is considered best practice in its advanced stages 1 to help improve people’s quality of life, address their physical, psychosocial and spiritual issues, and support their carers. 2 Cognitive decline, the pattern of physical symptoms, inter-current problems such as infections, and high prevalence of comorbidities require a unique approach for the provision and delivery of optimal palliative care. As dementia progresses, the majority of this population in developed countries will be living in long-term care facilities at the end of life, a setting where palliative care is often suboptimal and/or deterioration precipitates acute hospitalisation (where receiving appropriate care is also limited).3,4
The multidimensional aspect of palliative care necessitates complex, multicomponent interventions to best address the needs of people with life-limiting illnesses. This also applies when considering the needs of specific populations, including those with dementia. Specifically, this requires a more in-depth understanding of how the elements contributing to tailored interventions can be optimally chosen, evaluated, implemented and described, to inform future research and also maximise knowledge translation into clinical practice. 1 A recent Cochrane review highlighted a paucity of high-quality data on the efficacy of palliative care interventions for people with dementia. 10 The review’s focus on efficacy limited the type of its included studies, which meant that studies focussing on how interventions have been designed to target dementia palliative care needs were not explored.
In 2014, the European Association for Palliative Care (EAPC) published a White Paper aiming to define optimal palliative care for people with dementia based on existing evidence and expert consensus. 6 The proposed framework outlined 11 domains, including 57 recommendations (‘subdomains’), with a view to guide clinical practice and policy change. In addition to its practice and policy agenda, the EAPC domains can provide a framework for the development of research in this context. A recent paper by Nakanishi et al. 5 has applied the EAPC domains to evaluate national dementia strategies and their palliative care content. Our review set out to identify the range of long-term care facility interventions addressing the palliative care needs of people with dementia and investigate their alignment with the domains of care as identified in the EAPC White Paper. In particular, we focused on interventions that fostered inclusiveness of the person with dementia and their families in decision-making and care planning, taking patient-centredness to be an essential pillar for care for people with dementia, who may have less capacity to provide information about their needs and to directly contribute to decision-making to their care. 7 In order to gain a more comprehensive description of current interventions available for this population, we sought to include a range of studies, including protocols, non-randomised studies and ongoing trials as well as randomised controlled studies. The review aimed to:
Describe the components of palliative care interventions that have been, or are being, developed for people with dementia in long-term care facilities, with a particular focus on those which included a focus on shared decision-making.
Examine the alignment of components with the EAPC framework domains. 6
Methods
A systematic review with a narrative approach to synthesis was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 8 guidelines.
Protocol and registration
The systematic review protocol was registered on the international prospective register of systematic reviews (PROSPERO) (ID: CRD42018095649).
Eligibility criteria
We included peer-reviewed primary studies, in English, focusing on palliative care interventions that address the needs of people with dementia living in long-term care facilities. Long-term care facilities were defined as settings where care is provided for older people 24 h a day, 7 days a week for an undefined period of time (e.g. care homes, nursing homes and residential aged care facilities). 9
Study design included qualitative, quantitative and mixed methods. The relevant population were people with any type of dementia in long-term care facilities who required and/or were receiving palliative care (as explicitly reported in the paper). Relevant interventions were those that supported palliative care for people with dementia, as defined by the EAPC White Paper domains as optimal care for this population. 6
To identify multicomponent interventions focusing on shared decision-making and implemented at a service level, studies were included if they described interventions addressing Domains 2 or 3 and at least one other domain. 6 Domain 2 (person-centred care and shared decision-making) and Domain 3 (setting care goals) were selected to capture interventions that included at least one element that acknowledged the review’s focus on the centrality of person with dementia in decision-making and care. In addition, the intervention(s) could address any other domain(s). Domain 11 was not considered as it relates to policy issues, and as such, is aligned with system level rather than service level interventions.
Information sources
Searches on the databases MEDLINE, CINAHL, PsycINFO and CENTRAL were conducted from earliest dates until 25 September 2017. The searches were updated for all databases from 1 September 2017 (or from 25 September 2019 where possible) to 11 July 2019. Collectively, dates searched were from 1946 – current for MEDLINE, 1980 – current for CINAHL, 1806 – current for PsycINFO and in Issue 10 October 2017 for CENTRAL.
Lateral searching was conducted and included: (1) Google Scholar with keywords adapted from Box 1; (2) checking reference lists of two recent systematic reviews,10,11 and their included studies; and (3) searching of clinical trials registries for Australia/New Zealand, 12 United States of America, 13 United Kingdom 14 and the European Union 15 to identify in-progress studies. Reference lists of relevant systematic reviews retrieved during the selection process were also searched. The systematic reviews themselves were not included.
Search strategy.

Search terms were informed by a recent Cochrane review, 10 modified to include non-randomised studies, Medical Subject Headings (MeSH) terms and keywords concerning palliative care and dementia, and search terms to capture studies describing interventions, strategies and their implementation. The MEDLINE search strategy is outlined in Box 1, with the modifications highlighted.
Study selection and data extraction
Search results were imported into EndNote X8 and selection criteria were applied independently by three reviewers (S.K., T.L. and M.V.G.). Two reviewers screened titles and abstracts (S.K. and M.V.G.) against inclusion criteria. Full text of potentially relevant studies was assessed for relevance by two reviewers (T.L. and M.V.G.). Any disagreements were resolved through discussion with a third reviewer (S.K.).
Data were extracted by three reviewers (S.K., T.L. and M.V.G.) using a Microsoft Office (2010) electronic proforma for EAPC Domains 1-10. 6 Data were extracted under the following headings: author, year, location, setting, study design, participants, outcomes, results (including efficacy, if evaluated) and conclusions. Intervention data were extracted using the Template for Intervention Description and Replication (TIDieR) reporting checklist. 16 Interventions were categorised based on the Cochrane Effective Practice and Organisation of Care (EPOC) Taxonomy for health interventions. 17
Data were only mapped to an EAPC domain if the intervention explicitly mentioned an aspect of palliative care aligned with the phrasing in that domain. A study had to meet at least one subdomain within the domain of interest, in order for that domain to be addressed. The content could apply to more than one subdomain within the domain of interest.
Synthesis of results
Synthesis of results followed a narrative approach, 18 structured around the aim, content and delivery of the intervention described in the included studies in relation to the EAPC domains. Results on efficacy were descriptively summarised, if available, to provide context for the content of those interventions.
Risk of bias in individual studies
The main focus of the review was to describe the components of interventions rather than determine their efficacy; thus, papers were not formally assessed for risk of bias.
Results
Study selection
Of 2221 references identified by database searches and an additional eight citations identified through lateral searching, 51 papers reporting on 32 studies met our inclusion criteria (Figure 1). The PRISMA flow chart details the original and updated search results for the review (Figure 1).
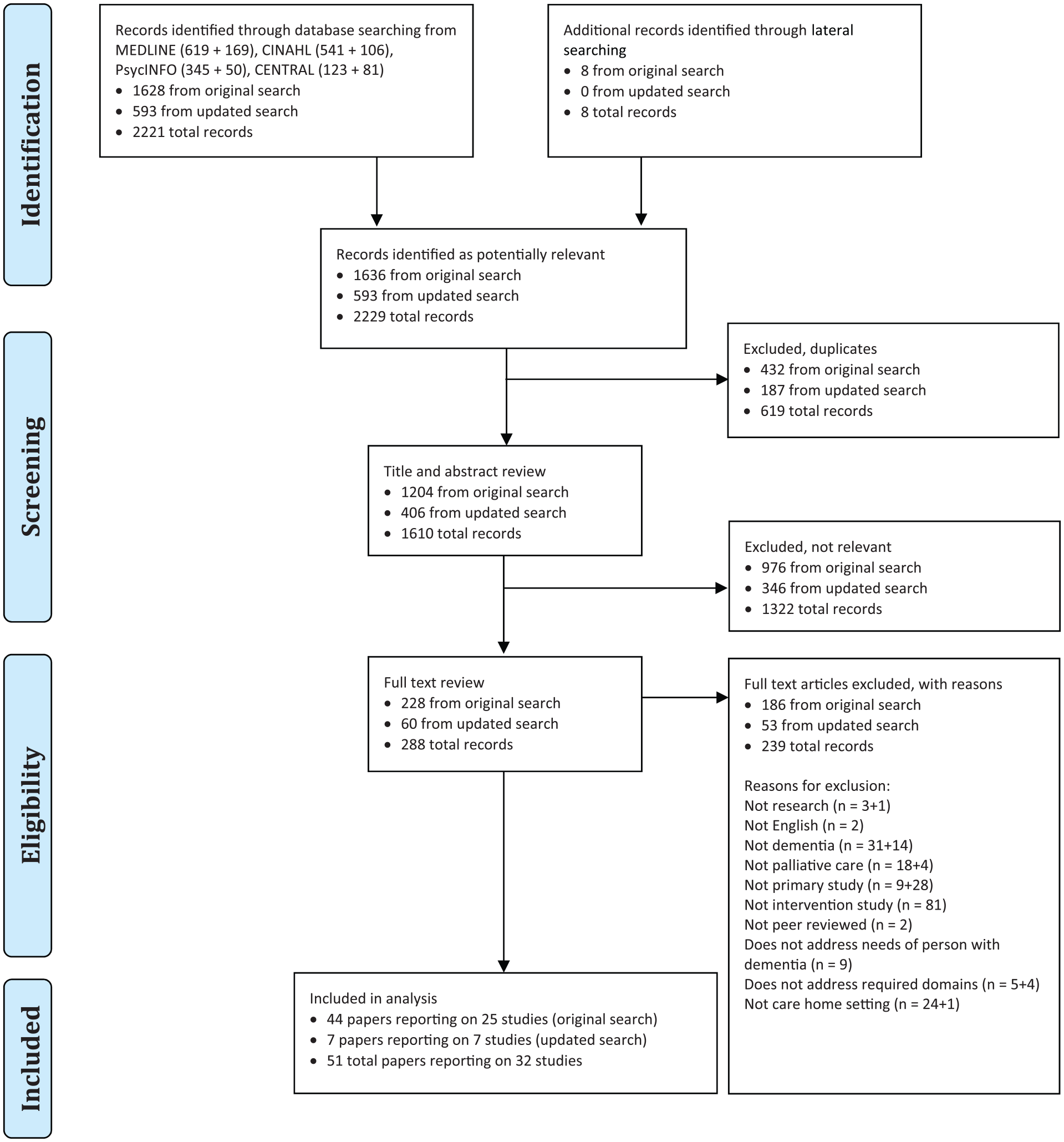
PRISMA flow diagram of included and excluded studies.
Study characteristics
Table 1 outlines the characteristics of the included studies.
Included studies were quantitative (n = 22),19–27,30,33,43,44,51,53,58,59,61,65–67 mixed methods (n = 10),28,30,31,34,38,45,49,54,56,60 qualitative (n = 2)42,52 and a description of an advance care plan process. 64 Studies included randomised control trials (n = 11)21–24,26,28,33,34,43,60,61 and non-randomised studies (n = 16).27,31,38,42,44,45,49,51,52,54,56,58,64–67 Studies originated from the United Kingdom (n = 7),22,33,38,42,44,45,54 United States (n = 5),19,23,25,34,61 Australia (n = 4),28,49,52,64 the Netherlands (n = 6),21,24,26,43,56,60 Canada (n = 3),27,56,65 Italy (n = 2),20,56 and one each from Belgium, 31 and Japan. 56 Eighteen interventions were specifically developed for people with advanced dementia.19,23,24,27,28,34,38,42,49,51,53,56,58,61,64–67 Number of partici-pants in the studies ranged from 16 to 688, with a median of 136 participants. Type of participants included residents, family members of residents, long-term care facility staff (clinical and non-clinical) and health professionals external to the long-term care facility.
Interventions specifically targeted long-term care facility staff members (n = 9),25,44,51,52,54,58,59,66,67 family members (n = 5)23,34,53,56,61 or a combination of both (n = 8).21,23,24,26,38,59,60 Three interventions involved long-term care facility staff, family members and external health professionals.33,49,60 Two interventions involved long-term care facility staff and external health professionals (general practitioners and district nurses),30,45 three involved long-term care facility-based physicians24,43,45 and one was delivered by an external agent. 38
Duration of follow-up to the intervention ranged from 7 days to 36 months (median 6 months) post-intervention implementation. Six studies did not report on the duration of follow-up.
Summary of included studies.
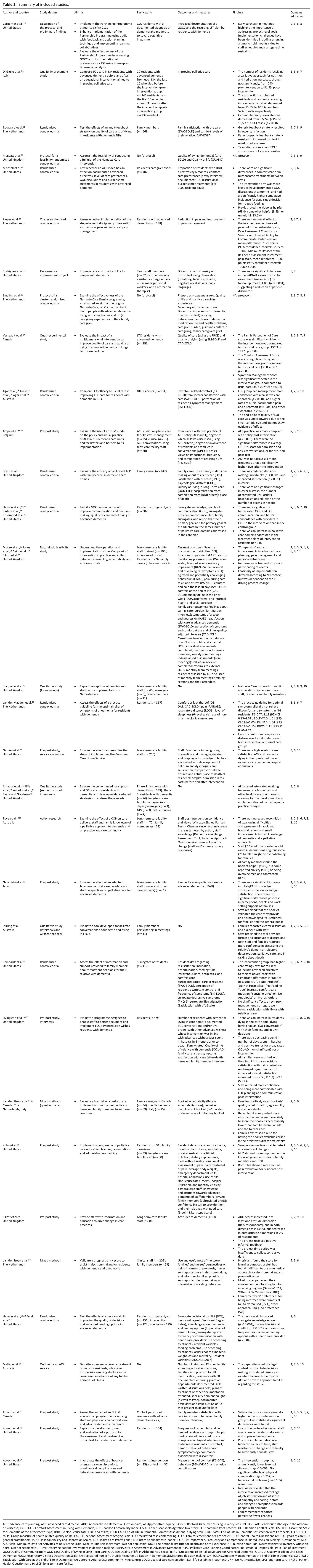
ACP: advance care planning; ACD: advanced care directive; ADQ: Approaches to Dementia Questionnaire; AI: Appreciative Inquiry; BANS-S: Bedford Alzheimer Nursing Severity Scale; BEHAVE-AD: Behaviour pathology in the Alzheimer’s disease; CAD-EOLD: Comfort Assessment in Dying with Dementia; CCI: Charlson Comorbidity Index; CMAI: Cohen-Mansfield Agitation Inventory; COP: community of practice; DCS: Decision Conflict Scale; DS-DAT: Discomfort Scale for Dementia of the Alzheimer’s Type; DNR: Do Not Resuscitate; EOL: end of life; EOLD-CAD: End-of-Life in Dementia-Comfort Assessment in Dying scale; EOLD-SWC: End-of-Life in Dementia-Satisfaction with Care scale; EQ-5D-5L: EuroQol Group measure of health related quality of life; FAST: Functional Assessment Staging Scale; FCC: facilitated case conferencing; FPCS: Family Perceptions of Care Scale; GHQ: General Health Questionnaire; GOC: goals of care; GP: general practitioner; HADS: Hospital Anxiety and Depression Scale; HCP: Health Care Professional; ICL: interdisciplinary care leader; IFC-SDM: Importance, Frequency and Competence in Shared Decision-Making Questionnaire; MDS ADL Scale: Minimum Data Set Activities of Daily Living Scale; MDT: multidisciplinary team; NA: not applicable; NICE: The National Institute for Health and Care Excellence; NH: nursing home; NPI: Neuropsychiatric Inventory Questionnaire; NR: not reported; OPTION: Observing patient involvement in decision-making; PAINAD: Pain Assessment in Advanced Dementia; PCPC: Palliative Care Planning Coordinator; PR: Person(s) Responsible; PoT: Plan of Treatment; QOC: Quality of Communication; QOD-LTC: Quality of Dying in Long-Term Care; QOL-AD: Quality of life in Alzheimer’s Disease; qPAD: Questionnaire on Palliative Care for Advanced Dementia; QUALID: Quality of Life in Late-Stage Dementia; RDOS: Respiratory Distress Observation Scale; RN: registered nurse; RUD-LITE: Resource Utilisation in Dementia; SDM: shared decision-making; SM-EOLD: Symptom Management at the End of Life in Dementia; SWC-EOLD: Satisfaction with Care at the End of Life in Dementia; VA: Veterans Affairs; CLC: community living centre; GOCC: goals of care conversation; LST: life-sustaining treatment; DNH: Do Not Hospitalise; LTC: long-term care; PHQ-9: Patient Health Questionnaire-9; LTCF: long-term care facility.
Synthesis of results
Interventions
Table 2 outlines the interventions described in the included studies.
Interventions involved educational material, including booklets (for staff and/or family),27,33,49,56,58,65 clinical practice guidelines (for physicians and/or long-term care facility staff),24,43,60,66 a communication tool (for staff), 52 and video decision aids (for family).34,61 Other interventions included educational outreach (for staff members),19,20,26,33,34,38,44,49,53,54,58,59,65,67 structured family meetings (for long-term care facility staff and family members),27,34,53,61 expansion of roles (e.g. training nurses to be case managers 67 or coordinators),28,29 or modification to long-term care facility environments.22,24,26,42,67
Some interventions focused on a particular need related to palliative care in dementia, such as facilitating communication with family members,28,31,52,53 advance care planning (ACP),28,29,31,33,44,54,64 symptom management24,30,49,51,56,58,65 and addressing psychosocial or spiritual needs.22,26,42 Other interventions were aimed at improving the overall knowledge regarding dementia and the role of palliative care in both long-term care facility staff20,34,49,58,59,67 and family.27,49,56,58,65
TIDieR table for included interventions.
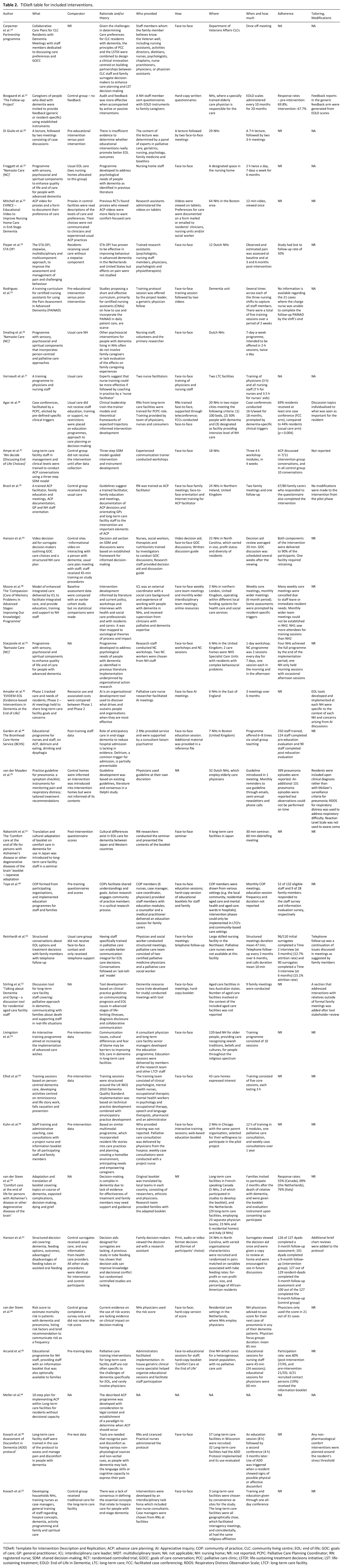
TIDieR: Template for Intervention Description and Replication; ACP: advance care planning; AI: Appreciative Inquiry; COP: community of practice; CLC: community living centre; EOL: end of life; GOC: goals of care; GP: general practitioner; ICL: interdisciplinary care leader; MDT: multidisciplinary team; NA: not applicable; NH: nursing home; NR: not reported; PCPC: Palliative Care Planning Coordinator; RN: registered nurse; SDM: shared decision-making; RCT: randomised controlled trial; GOCC: goals of care conversation; PCC: palliative care clinic; LSTDI: life-sustaining treatment decisions initiative; LST: life-sustaining treatment; EOLD: End-of-Life in Dementia; LTC: long-term care; FCC: facilitated case conferencing; RDOS: Respiratory Distress Observation Scale; LTCF: long-term care facility.
EAPC domains
Table 3 maps the interventions in relation to each of the EAPC domains they addressed. On average, the interventions addressed five domains each. Table 4 describes the components of interventions as they relate to the EAPC domains.
Intervention mapping to EAPC domains.
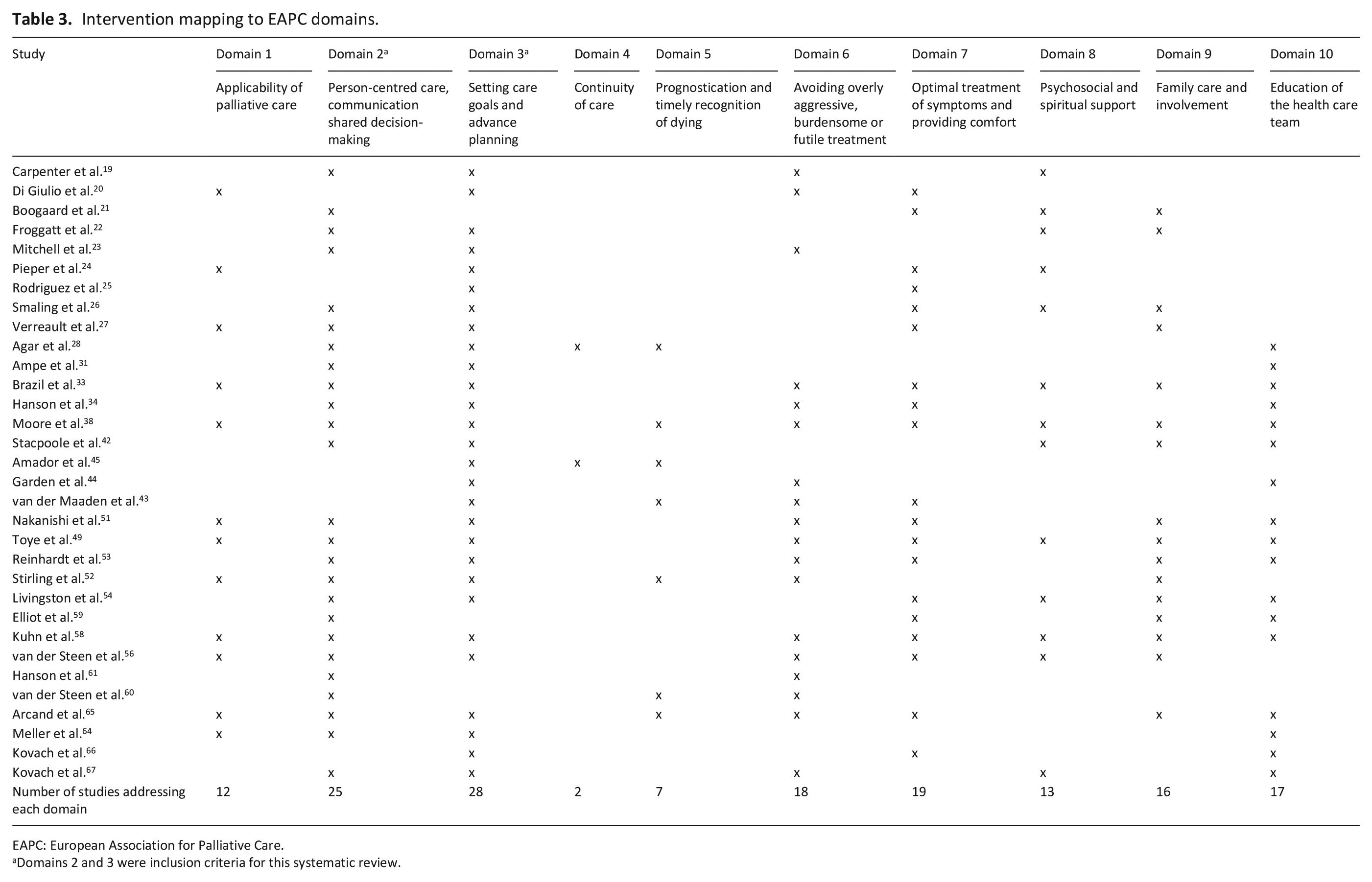
EAPC: European Association for Palliative Care.
Domains 2 and 3 were inclusion criteria for this systematic review.
Mapping of intervention components to EAPC domains.

EAPC: European Association for Palliative Care; ACP: advance care planning; AI: Appreciative Inquiry; COP: community of practice; EOL: end of life; EOLD: End of life in Dementia; GOC: goals of care; GP: general practitioner; ICL: interdisciplinary care leader; long-term care facility: long-term care facility; MDT: multidisciplinary team; NA: not applicable; NH: nursing home; NR: not reported; PCPC: Palliative Care Planning Coordinator; RN: registered nurse; SDM: shared decision-making; EOL: end of life; EOLD-SWC: End-of-Life in Dementia-Satisfaction with Care scale; EOLD-CAD: End-of-Life in Dementia-Comfort Assessment in Dying scale; ACD: advanced care directive.
All 10 domains were addressed to varying extent: Domain 1 (n = 12),20,24,27,33,38,49,51,52,56,58,64,65 Domain 2 (n = 25),19,21–23,26–28,31,33,34,38,42,49,51–54,56,58–61,64,65,67 Domain 3 (n = 28),19,20,22–28,31,33,34,38,42–45,49,51–54,56,58,64–67 Domain 4 (n = 3),28,45 Domain 5 (n = 7),28,38,43,45,52,60,65 Domain 6 (n = 18),19,20,33,34,38,43,44,49,51–53,56,58,60,61,65,67 Domain 7 (n = 19),20,21,24–27,33,34,38,43,51,53,54,56,58,59,65,66 Domain 8 (n = 13),19,21,22,24,26,33,38,42,49,54,56,58,67 Domain 9 (n = 16)21,22,26,27,33,38,42,49,51–54,56,58,59,65 and Domain 10 (n = 17).28,31,33,34,38,42,44,49,51,53,54,58,59,64–67
Studies implemented educational material addressing Domain 1 (n = 9),24,33,38,49,51,52,56,58,65 Domain 2 (n = 4),23,34,52,60 Domain 3 (n = 13),23–25,33,34,51,52,56,58,61,65–67 Domain 6 (n = 10),23,33,34,49,51,52,56,58,61,65 Domain 7 (n = 8),24,25,33,49,51,56,58,65 Domain 8 (n = 3),33,56,58 Domain 9 (n = 7)24,33,49,52,56,58,65 and Domain 10 (n = 4).34,49,58,65
Clinical practice guidelines addressed Domain 2, 60 Domain 3,43,66 Domain 5,43,60 Domain 6,43,60 Domain 743,66 and Domain 10. 66 Thirteen studies used educational outreach visits or educational meetings for long-term care facility staff and/or family members.19,27,33,34,38,44,49,53,54,58,59,65,67 These activities addressed Domain 1,27,49 Domain 2,19,27,31 Domain 3,19,27,31,33,44,54 Domain 5, 65 Domain 6,19,44,58,65,67 Domain 7,26,27,58,59,65,67 Domain 819,54,67 and Domain 9.27,49,54,58,59,65 By virtue of being education and training for staff regarding palliative care in dementia, these interventions also addressed Domain 10.28,31,33,34,38,42,44,49,51,53,54,58,59,64–67 Nine studies proposed interventions that facilitate better resident case management addressing Domain 1, 64 Domain 2,28,31,33,53,54 Domain 3,23,31,33,44,52–54 Domain 4,28,34,64 Domain 7,33,34,54 Domain 9 53 and Domain 10.28,31,44,64
Five interventions were considered to be resident/family mediated, addressing Domain 2,22,26 Domain 3, 23 Domain 5, 28 Domain 8 42 and Domain 9. 42 Two interventions involved the expansion or the creation of a role addressing Domain 2, 67 Domain 4 28 and Domain 10. 28 Four environment modification interventions addressed Domain 8.22,26,42,67
One audit and feedback intervention addressed Domains 2, 7, 8 and 9. 21 One intervention involved inter-professional communication between health professionals addressing Domains 3–5. 45 Other interventions involved telemedicine addressing Domain 9, 53 and monitoring health care delivery addressing Domain 7. 59 One intervention was informed by a national consensus process to form a model for end-of-life (EOL) care. 38
Domain coverage and theoretical underpinning
None of the studies addressed all ten considered EAPC domains as part of a single intervention. However, all ten EAPC domains were addressed to some extent across the included studies. The degree to which each study addressed each domain varied. The ‘Compassion Intervention’ had the most comprehensive coverage, supporting nine out of ten EAPC domains.38–41
Apart from Domains 2 and 3, the most commonly addressed domains were Domain 10 (n = 17),28,31,33,34,38,42,44,49,51,53,54,58,59,64–67 Domain 6 (n = 15)33,34,38,43,44,49,51–53,56,58,60,61,65,67 and Domain 7 (n = 14).21,33,34,38,43,49,51,53,54,56,58,59,61,65,66 In contrast, the least commonly addressed domains were Domain 4 (n = 2)28,45 and Domain 5 (n = 7).28,38,43,45,52,60,65
Seven of the included studies reported on the theoretical foundations of their interventions.28,31,34,38,42,45,49 Any available findings on intervention efficacy are summarised in Table 1. Included interventions were found to improve: staff competencies in various aspects of palliative care in dementia, 44 relatives’ satisfaction with the care, 27 symptom management 27 or comfort of the resident with dementia21,27,54 or observed pain24,25 or discomfort levels 67 in residents, to reduce decision-making uncertainty 33 or conflict, 61 to increase in the number of Do Not Resuscitate (DNR) decisions, 54 decrease in the number of resuscitations, 20 or to result in management more consistent with a palliative care approach. 28
There were non-significant trends towards higher effects on symptom management, surrogate well-being, life satisfaction or satisfaction with relatives’ care, 53 improved staff attitudes to dementia, 59 more routine pain evaluation, 58 increased satisfaction with pain control, emotional support, treatment of patient with respect, communication and provided information, 65 palliative approach to nutrition and hydration and proportion of tube-fed residents, 20 and in reduced behavioural problems. 67 Some interventions found no significant changes to ACP practice, 31 preferences for comfort care 23 or for withholding burdensome treatments,23,33,53 relatives’ satisfaction with resident pain control, 54 estimated pain, 24 number of physical iatrogenic problems experienced by residents, 66 or in overall quality of EOL care. 28
Discussion
Main findings
This is the first review to utilise the EAPC White Paper domains on optimal palliative care in dementia to explore the relevance and range of interventions in long-term care facilities. Although none of the interventions addressed all ten considered EAPC domains as part of a single intervention, these ten domains were addressed to some extent across various interventions, and to various degrees. Different domains were addressed by different types of interventions; with some highly focused, and others trying to address multiple aspects of palliative dementia care. Pragmatically, it is expected that no intervention can address all domains because it is difficult to achieve that level of change simultaneously, because of limits posed by finite resources or service model structure and processes, and also, importantly, because different interventions may prioritise different aspects of palliative care needs of people with dementia, and subsequently, relevant EAPC domains and recommendations. Equally, the development of multicomponent interventions is often incremental where one element or a simpler combination is developed and tested initially, and built on in subsequent studies. Our review has highlighted that when reporting on such studies, it is important for researchers to explicitly state why they have chosen specific domains, define the ‘active ingredient(s)’ within the complex intervention, and how those choices define the scope of the intervention, as well as its outcomes. Future work also needs to inform our understanding of ‘dose’ and ‘coverage’, that is how many components are needed (and at what breadth and depth) to best achieve the EAPC domain goals. The health economic aspect should also be considered when designing and implementing such interventions.
The review indicates that current research is more focussed on developing interventions that provide education and upskilling of health care teams, interventions that support optimal treatment of symptoms and comfort care, and interventions that seek to prevent aggressive treatment at the end of life. At the same time, less research seems to relate to continuity of care and prognostication. The small number of interventions relating to prognostication is consistent with the EAPC White Paper where prognostication was scored as the least important domain of interest. Given the uncertainly and difficulty in predicting survival, and the emphasis on identifying the (unmet) palliative care needs of people with dementia as best practice, it is encouraging that care interventions are focusing on addressing the (unmet) needs for this population rather than on models driven purely by prognosis.
Continuity of care was also less commonly explicitly addressed as a domain of interest. With our focus on long-term care facilities, the major transition from home for the person with dementia may have already occurred prior to this time, is complex to study longitudinally and care pathways may not always be linear. Acknowledging that avoidance of inappropriate hospitalisation is desirable (Domain 6), sometimes transition into the hospital environment will need to occur as it might be in the resident’s best interest and such occurrences should not be defined purely as ‘failures of the system’. Consideration of what defines continuity of care within the long-term care facility itself is needed and is likely multi-faceted (including continuity of information, continuity of staff and management). 68
The EAPC domains were proposed to inform clinical practice and policy by defining the relevant aspects of optimal palliative care for people with dementia and to articulate a research agenda. They were not developed to inform quality assessment of conducted research. However, this review supports the EAPC domains as useful guidance on issues critical for this population that new interventions should aim to address (and appropriately measure). This review does not argue for a minimum or maximum number of domains that need to be captured within individual interventions, but highlights the need for greater clarity around the intervention elements and their goals to encourage transparency and help replication and normalisation of the interventions. 69 In most cases, palliative care interventions for people with dementia will be complex, requiring multiple components. The EAPC framework offers a useful way to structure and consider interventions for people with dementia and palliative care needs, and to place such interventions in context. Similarly, this review did not set out to provide an optimal number of intervention components nor draw any correlations about types, numbers or combinations of intervention components and outcomes, but rather to conceptualise what current complex interventions are on offer for this population. This review however offers a starting point for informing future work in this field. The updated search identified seven studies for the period of 2018–2019, signalling that this is a research area of growing interest.
This review set out to describe the components of interventions within the framework posed by the EAPC White Paper. It was not in the remit of this review to address if greater domain coverage correlates with better outcomes for people with dementia (and their caregivers). Instead, the review set out to explore how thinking about these domains a priori would help inform study design, improve reporting, reduce the risk of duplication and enhance capacity to build systematically from prior intervention studies, as well as adapt or implement interventions in a variety of contexts. 69 Such approach will help increase generalisability and applicability of study findings in the clinical setting. Future research could highlight the inter-relatedness of domains, and how intervening at one domain may also improve outcomes in others.
This review explored whether proposed interventions were informed by theory. Strengthening the theoretical underpinnings in future research should extend beyond that of development and design of interventions. This may potentially involve using theory to inform the selection of outcomes, the interpretation and analysis of results, as well as to tailor the intervention as exemplified in studies such as FINCH, OPTIMAL and Namaste.70–72 Our review did not specifically explore the role of context. This can be a significant element for interventions, as exemplified by the ‘Compassion’ intervention where the impact of context was highlighted as critical to the intervention implementation. 38 Developing interventions with strong theoretical foundations would help bring clarity around how interventions are thought to achieve particular outcomes and the role of contextual factors. This, in turn, will help strengthen the evidence base for this population. 1 Theoretical frameworks of behaviour change at the levels of the long-term care facility, 69 clinician and family would be of particular interest.
Strengths and limitations
This review offers an initial, descriptive synthesis of interventions for people with dementia and palliative care needs using the domains of care proposed in the EAPC White Paper. The review was underpinned by the philosophy that at least one domain of such interventions should focus on shared decision-making or person-centredness, acknowledging the person with dementia as a partner in care. This may have led to exclusion of studies with a focus purely on a singular domain (e.g. symptom management).
The authors on this review were also investigators on some of the included studies. To minimise bias, decisions of whether or not such studies should be included and how content was to be mapped to EAPC domains was conducted by team members not involved in those studies.
This review used a modified search strategy from a recent Cochrane review 10 to ensure that the review findings build on existing evidence. The modification did not specifically include keywords on shared decision-making or person-centredness, as this could have potentially excluded studies of interest.
Data extraction and mapping of domains relied on the level of reporting in the included studies. To overcome this, content about each intervention was extracted from the primary study findings and then augmented by data reported in supplementary reports on those studies. Study authors were not contacted to provide further details with regard to the content of their interventions. Therefore, it is possible that some interventions may have addressed domains in addition to the ones reported in this review, but these details may have been missed because the information was not reported in the publications, or sufficient detail was not given about the intervention components. This may have affected how data were extracted against the EAPC domains.
Mapping of content to a singular domain for each intervention was difficult due to domain overlap. To maximise coverage within each domain, content was mapped to all relevant subdomains.
All of the included studies were set in long-term care facilities and interventions were set up to answer questions relevant to this setting. Interventions conducted in other clinical settings or in the community may be different, and therefore, may cover different domains.
This review focused on describing interventions rather than evidence for efficacy. Efficacy has been explored by previous reviews;10,11 this review sought to include emerging interventions (e.g. in protocols) as well-completed evaluations to ensure an up-to-date overview of the state of the science. Where available, included interventions showed mixed findings for efficacy, including the randomised controlled trials (RCTs) retrieved in this review. Included RCTs showed evidence for efficacy only on measures of process and very little evidence on efficacy on outcomes, highlighting the complex nature of this population and the included interventions, with a number of many interacting contextual factors. As many interventions in this field will require behaviour, attitudinal or system change, the mechanism to achieve this is also critical in intervention development.
What this study adds
The EAPC recommendations were derived from consensus and thus reflect participants’ (‘experts’) knowledge that is grounded in clinical experience and acknowledge the interaction and overlap between both the intent and integrated approach required to achieving the domain outcomes. Such frameworks will need to be responsive as the evidence evolves with further clarity about subdomains evolving as outcomes of different interventions are known. This can be augmented by a theory-driven approach which might articulate the inter-relationship and hierarchy of these subdomains in a more complex way. It may also highlight where there is large ‘return on investment’, if a singular component can tackle several important elements of care in an integrated manner, or if the degree of effort required to implement is lower and is thus associated with higher incremental gains in outcomes.
Conclusion
The design of complex interventions for people with dementia in long-term care facilities is itself an emerging field, and a primary focus on evaluative methods may stymie innovation through allowing the ‘tail to wag the dog’. Palliative care for long-term care facility residents with dementia represents a perfect storm of a complex intervention for a complex population in a complex setting. As such, improvements in care and optimal knowledge translation should be driven by a broader range of qualitative and quantitative methods including, but not exclusively driven by, randomised controlled trials, unified by a shared language of the elements of the intervention, with the EAPC domains informing the complex needs of people with dementia and their family members.
Footnotes
Acknowledgements
The authors acknowledge the generous assistance of Ms Ingrid Amgarth-Duff with data extraction and the preparation of this article for publication.
Authorship
M.R.A., C.G., T.L. and F.B. were responsible for study concept and design and for funding acquisition. S.K., M.V.G., C.G., F.B., T.L. and M.R.A. were responsible for the acquisition of data and for the drafting of the manuscript. All the authors contributed to analysis and interpretation of the data and the critical revision of the manuscript for important intellectual content. All the authors approved this version of the manuscript to be published.
Data management and sharing
All relevant data are within the manuscript. Any other data are available upon request from the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This review was conducted with funding from the University of Technology Sydney and the University of Hertfordshire Dean’s Health Futures Development Grant. C.G. is an NIHR Senior Investigator.
